Comparative Study of Antioxidant Power, Polyphenols, Flavonoids and Betacyanins of the Peel and Pulp of Three Tunisian Opuntia Forms
Abstract
:1. Introduction
2. Experimental
2.1. Plant Material
2.2. Solvents and Standard Phenolics
2.3. Sample Preparation
2.4. DPPH Radical Scavenging Activity Assay
2.5. Determination of the Total Phenolic Content
2.6. Determination of Total Flavonoid Content
2.7. RP-HPLC and ESI-MS Analysis
2.8. Statistical Analysis
3. Results and Discussion
3.1. DPPH Radical Scavenging Activity
| DPPH Antioxidant Scavenging Capacity IC50% (mg·mL−1) a | |||
|---|---|---|---|
| Tissue Fruit | Peel | Pulp | |
| Species-Forms | |||
| O. ficus indica “spiny” | 0.54 b ± 0.04 | 0.51 c ± 0.01 | |
| O. ficus indica “thornless” | 0.57 b ± 0.02 | 0.56 c ± 0.01 | |
| O. stricta | 0.40 b ± 0.03 | 0.43 c ± 0.01 | |
| Trolox | 0.33 ± 0.01 | ||
3.2. Total Phenolic Contents
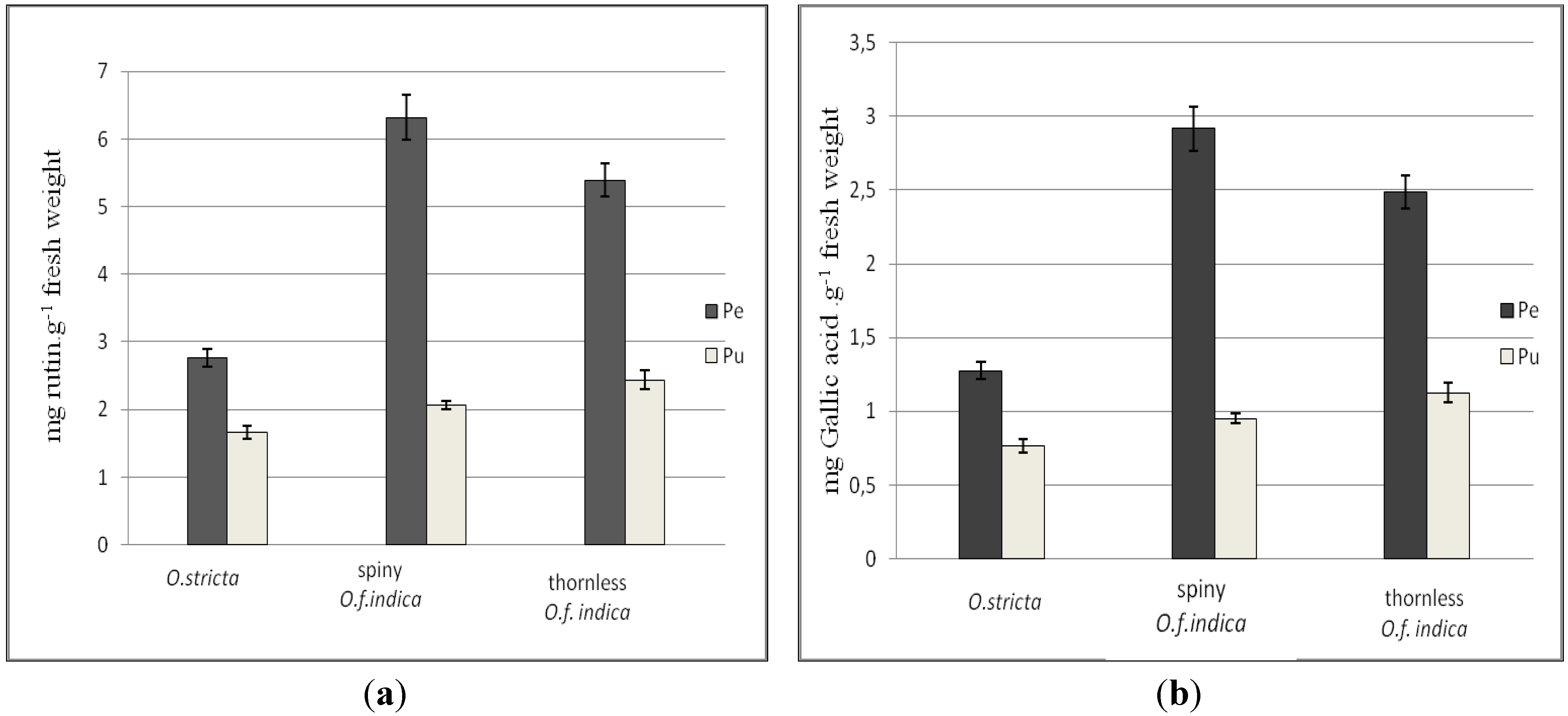
3.3. Total Flavonoid Content
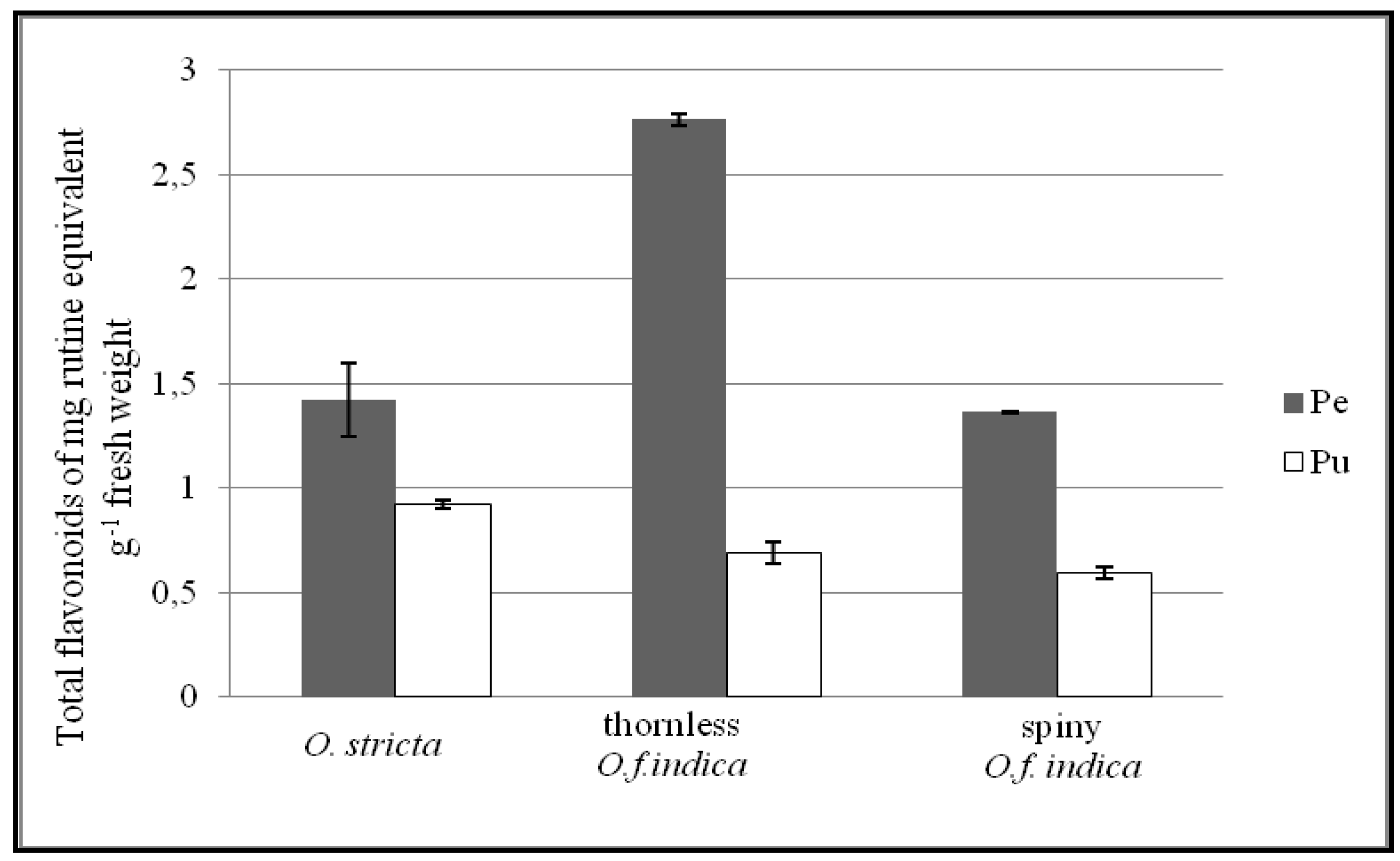
3.4. RP-HPLC and ESI-MS Analysis
| Tunisian Opuntia | Tissues | Major Peaks | Bioactive Compounds | RT (min) | MS (m/z) | MS2 (m/z) | λmax (mn) | Amount (μg·g−1 Fresh Weight) M ± SD |
|---|---|---|---|---|---|---|---|---|
| Spiny O. ficus indica | Peel | A31 | unidentified flavonol | 18.45 | 741 | 300 | 352 | 3.13 ± 0.04 |
| A32 | Isorhamnetin diglycoside | 20.68 | 769 | 314–605 | 355 | 14.60 ± 0.009 | ||
| A33 | Isorhamnetin diglycoside | 21.05 | 755 | 315–605 | 354 | 45.59 ± 0.01 | ||
| A34 | unidentified flavonol | 22.43 | 609 | 315–314 | 263–353 | 17.48 ± 0.07 | ||
| A35 | Isorhamnetin-3- O-rutinoside | 23.75 | 623 | 315–623 | 352 | 2 ± 0.03 | ||
| A36 | Isorhamnetin-3- O-rutinoside | 24.03 | 623 | 315–623 | 352 | 1.98 ± 0.002 | ||
| A37 | Isorhamnetin-3- O-glucoside | 24.93 | 477 | 314–477 | 352 | 1.08 ± 0.01 | ||
| A38 | phenolic acid | 28.68 | 431 | 193–236 | 328 | 1.81 ± 0.02 | ||
| A51 | Indicaxanthin | 7.22 | 309 | 265 | 485 | n.q | ||
| Pulp | A61 | Indicaxanthin | 6.98 | 309 | 239 | 485 | n.q | |
| Thornless O. ficus indica | Peel | B31 | phenolic acid | 12.55 | 239 | 239 | 328 | 2.44 ± 0.06 |
| B32 | phenolic acid | 14 | 355 | 193–355 | 326 | 4.69 ± 0.02 | ||
| B33 | phenolic acid | 15.53 | 489 | 235–193 | 325 | 8.64 ± 0.03 | ||
| B34 | Isorhamnetin diglycoside | 20.58 | 769 | 315–605 | 355 | 13.07 ± 0.05 | ||
| B35 | Isorhamnetin diglycoside | 20.95 | 755 | 315–605 | 354 | 35.09 ± 0.01 | ||
| B36 | Isorhamnetin diglycoside | 21.2 | 755 | 315–605 | 353 | 10.13 ± 0.02 | ||
| B37 | quercetine-3- O-rutinoside | 22.35 | 609 | 301 | 350 | 5.60 ± 0.05 | ||
| B38 | Isorhamnetin-3- O-rutinoside | 23.97 | 623 | 315 | 354 | 18.74 ± 0.08 | ||
| B39 | phenolic acid | 28.65 | 431 | 193–237 | 326 | 5.43 ± 0.04 | ||
| B51 | Betanin (betanidin-5- O-β-glucoside) | 9.52 | 551 | 475–312 | 530 | n.q | ||
| Pulp | B41 | phenolic acid | 20.6 | 613 | 562–477 | 270–332 | 0.34 ± 0.001 | |
| B42 | phenolic acid | 20.97 | 565 | 339 | 325 | 0.75 ± 0.003 | ||
| B43 | Isorhamnetin3- O-rutinoside | 23.97 | 623 | 315 | 355 | 0.39 ± 0.005 | ||
| B61 | Betanin | 7.43 | 551 | 389 | 537 | n.q | ||
| B62 | Isobetanin (isobetanidin-5- O-β-glucoside) | 8.43 | 551 | 389 | 537 | n.q | ||
| B63 | betanidin | 9.63 | 389 | 389 | 541 | n.q | ||
| O. stricta | Peel | C31 | phenolic acid | 12.57 | 395 | 349 | 325 | 4.15 ± 0.002 |
| C32 | unidentified compound | 16.2 | 977 | 815 | 346 | n.q | ||
| C33 | unidentified compound | 18.68 | 639 | 477 | 346 | n.q | ||
| C34 | unidentified compound | 19.27 | 611 | 431 | 345 | n.q | ||
| C35 | Isorhamnetin-3- O-rutinoside | 23.68 | 623 | 315 | 352 | 4.95 ± 0.01 | ||
| C36 | Isorhamnetin-3- O-rutinoside | 23.97 | 623 | 315 | 355 | 14.27 ± 0.02 | ||
| C51 | Betanin | 7.72 | 551 | 389 | 533 | n.q | ||
| C52 | Isobetanin | 8.58 | 551 | 389 | 533 | n.q | ||
| C53 | betanidin | 9.78 | 389 | 343–150 | 539 | n.q | ||
| Pulp | C41 | phenolic acid | 12.62 | 395 | 395–349 | 326 | 0.45 ± 0.003 | |
| C42 | phenolic acid | 16.08 | 977 | 815 | 330 | 0.41 ± 0.01 | ||
| C43 | phenolic acid | 18.58 | 639 | 477 | 330 | 0.43 ± 0.02 | ||
| C44 | phenolic acid | 19.17 | 611 | 431 | 334 | 0.43 ± 0.009 | ||
| C45 | Isorhamnetin-3- O-rutinoside | 23.68 | 623 | 315 | 350 | 0.85 ± 0.01 | ||
| C46 | Phenolic acid | 24.87 | 477 | 314 | 331 | 2 ± 0.02 | ||
| C61 | Betanin | 7.53 | 551 | 389 | 538 | n.q | ||
| C62 | Isobetanin | 8.4 | 551 | 389 | 533 | n.q | ||
| C63 | betanidin | 9.87 | 389 | 343 | 525 | n.q |
- (a)
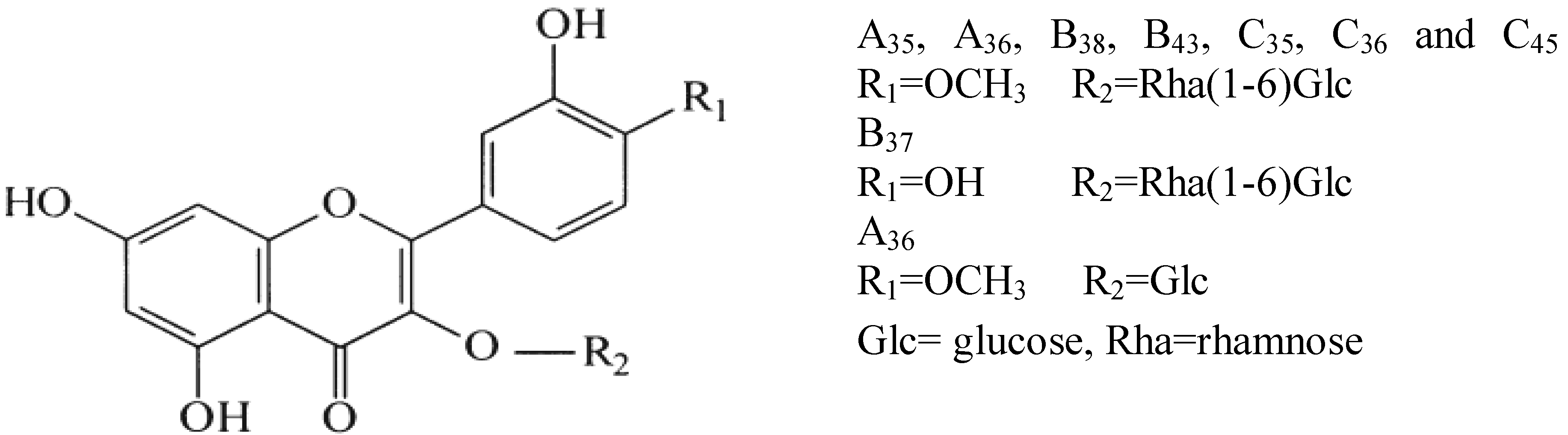
- Flavonol glycosyl: The flavonol glycosil class was indicated by absorbance at 255–263 nm and 350–355 nm in accordance with the work of Wollenweber [48]. He analyzed the flavonoids in the fronds of Cheilanthes farinosa (Polypodiaceae) and detected such peaks at the mentioned wavelength. Results show that the peel of the thornless O. ficus indica has the highest amount of phenolic compounds. The most dominant flavonol glycosil are isorhamnetin derivatives. They are found in the peels but not in the pulp (very small amount of isorhamnetin-3-O-rutinoside in the thornless forms and O. stricta). The content of these compounds are as follows: thornless O. ficus indica: 77.03 μg·g−1 spiny O. ficus indica: 65.25 μg·g−1; and O. stricta 19.22 μg·g−1. The peel also contained a compound not found in the spiny O. ficus indica and the O. stricta. This compound was identified as quercetin 3-O-rutinoside (5.60 ± 0.05 μg·g−1 FW). Compared to Tunisian fruits that contained more isorhamnetin, less quercetin and no kaempferol, Kuti [14] showed that the Texas fruit has more quercetin, less isorhamnetin, and some kaempferol. Fernández-López et al. [49] reported for Opuntia fruits significant amounts of flavonoids, being quercetin in the predominant type followed by isorhamnetin, luteolin and kaempferol. These results indicate that the phenols are region-specific. Our MS results showed a signal at m/z 769 (A32 and B34). The corresponding MS2 spectrum exhibited a main product ion at m/z 605 and 314. These results could be correlated to the presence of isorhamnetin [28]. The signal at m/z 605 corresponds to the loss of a fragment with −146 amu (atomic mass unit) in accordance with the mass of a deoxyhexose moiety flowed by one water molecule ([M − H − 146 − 18]−). The fragment at 314 corresponds to the molar mass of an ionic isorhamnetin. The MS spectrum of peaks A33, B35 and B36 produced a pseudomolecular ion [M − H]− at m/z 755, releasing a major MS2 fragment at m/z 605 = ([M − H −132 − 18]−) and 315, −132 amu is the molar mass of a pentose which loses a water molecule, thus allowing us to hypothesize that those signals correspond to an isorhamnetin diglycoside containing one pentose molecule. All these MS results prove the presence of isorhamnetin derivatives.
- (b)
- Phenolic acids: The highest content of total phenolic acids was found in thornless O. ficus indica peel (21.2 μg·g−1 FW) followed by O. stricta peel (4.15 μg·g−1 FW) and pulp (3.72 μg·g−1 FW). No phenolic acid was detected in the spiny O. ficus indica pulp.
- (c)
- Betalain: The A61 showed a pseudomolecular ion [M + H]+ at m/z 309. The corresponding MS2 spectrum exhibited a main product ion at m/z 239. The A61 spectrum (MS and MS2) was indicative of indicaxanthin. The MS spectrum of peaks B51, B61, C51, and C61 produced a pseudomolecular ion [M + H]+ at m/z 551 and the MS2 spectrum showed produced ions at m/z 389. The peaks were identified as betanin. The chromatographic profile of betalain peel showed the richness of the spiny O. ficus indica in indicaxanthin (A51 at 470 nm) and the O. stricta in betacyanin (C51, C52 and C53 at 540 nm). Table 2 revealed a wealth in betacyanin in O. stricta and thornless O. ficus indica pulp. Our results are in agreement with the results of Castellar et al. [31], who found that the level of betanin and isobetanin were around five times higher in Murcia O. stricta (southeast of Spain) than in the red-purple fruits of O. ficus indica.
4. Conclusions
Acknowledgments
References
- Gurbachan, S.; Felker, P. Cactus: New world foods. Indian Hortic. 1998, 43, 29–31. [Google Scholar]
- Han, H.; Felker, P. Field validation of water-use efficiency of a CAM plant Opuntia ellisiana in south Texas. J. Arid Environ. 1997, 36, 133–148. [Google Scholar] [CrossRef]
- Espinosa, J.; Borrocal, R.; Jara, M.; Zorrilla, C.; Medina, J. Some properties and preliminary tries of preservation of fruits and juice of Opuntia (O. ficus indica). Fruits 1973, 28, 285–289. [Google Scholar]
- Ennouri, M.; Bourret, E.; Baklouti, S.; Damak, K.; Rebaï, A.; Attia, H. Rheology of microfiltrated Opuntia juices. Sci. Aliment. 2006, 26, 337–348. [Google Scholar] [CrossRef]
- Bustos, E.O. Alcoholic beverage from Chilean Opuntia ficus indica. Am. J. Enol. Vitic. 1981, 32, 228–229. [Google Scholar]
- Sawaya, W.N.; Khatchadorian, H.A.; Almuhammad, H.M. Chemical characterization of Opuntia pulp, Opuntia ficus indica, and the manufacturing of Opuntia jam. J. Food Technol. 1983, 18, 183–193. [Google Scholar]
- Saenz, C.; Sepulveda, E. Cactus-pear juices. J. Prof. Assoc. Cactus. Dev. 2001, 4, 3–10. [Google Scholar]
- Castellar, M.R.; Obón, J.M.; Fernández-López, J.A. Color properties and stability of betacyanins from Opuntia fruits. J. Agric. Food Chem. 2003, 51, 2772–2776. [Google Scholar] [CrossRef]
- Diaz, F.; Santos, E.M.; Filardo, S.; villagómez, R.; Scheinvar, L. Colorant extraction from red prickley peat (Opuntia lasiacantha) for food application. J. Agric. Food Chem. 2006, 5, 1330–1337. [Google Scholar]
- Strack, D.; Vogt, T.; Schliemann, W. Recent advances in betalain research. Phytochemistry 2003, 62, 247–269. [Google Scholar] [CrossRef]
- Mizrahi, Y.; Nerd, A.; Nobel, P.S. Cacti as crops. Hortic. Rev. 1997, 18, 291–320. [Google Scholar]
- Stintzing, F.C.; Schieber, A.; Carle, R. Evaluation of colour properties and chemical quality parameters of cactus juices. Eur. Food Res. Technol. 2003, 216, 303–311. [Google Scholar]
- Stintzing, F.C.; Carle, R. Cactus fruits—More than colour. Fruit Process. 2006, 16, 166–171. [Google Scholar]
- Kuti, J.O. Antioxidant compounds from four Opuntia cactus pear fruit varieties. Food Chem. 2004, 85, 527–533. [Google Scholar] [CrossRef]
- Zakharova, N.S.; Petrova, T.A. Relationship between the structure and antioxidant activity of various betalains. Prikl. Biokhim. Mikrobiol. 1998, 34, 199–202. [Google Scholar] [PubMed]
- Gentile, C.; Tesoriere, L.; Allegra, M.; Livrea, M.A.; D’Alessio, P. Antioxidant betalains from cactus pear (Opuntia ficus indica) inhibit endothelial ICAM-1 expression. Ann. N. Y. Acad. Sci. 2004, 1028, 481–486. [Google Scholar] [CrossRef] [Green Version]
- Andres-Lacueva, C.; Zamora-Ros, R. Wanted: Specific nutritional biomarkers for food consumption for the study of its protective role in health. Br. J. Nutr. 2010, 103, 307–308. [Google Scholar] [CrossRef]
- Wallace, R.S. Biochemical taxonomy and the Cactaceae. Cactus Succul. J. 1986, 58, 35–38. [Google Scholar]
- Kuti, J.O. Antioxidant activity of Opuntia cactus pear. Hort Sci. 2000, 35, 433. [Google Scholar]
- Jacob, J.K.; Hakimuddin, F.; Paliyath, G.; Fisher, H. Antioxidant and antiproliferative activity of polyphenols in novel high-polyphenol grape lines. Food Res. Intern. 2008, 41, 419–428. [Google Scholar] [CrossRef]
- Lampila, P.; Lieshout, M.; Germmen, B.; Lähteenmäki, L. Consumer attitudes towards enhanced flavonoid content in fruit. Food Res. Intern. 2009, 42, 122–129. [Google Scholar] [CrossRef]
- Trejo-Gonzalez, A.; Gabriel-Ortiz, G.; Puebla-Perez, A.M. A purified extract from prickly pear cactus (Opuntia fuliginosa) controls experimentally induced diabetes in rats. J. Ethnopharmacol. 1996, 55, 27–33. [Google Scholar] [CrossRef]
- Galati, E.M.; Mondello, M.R.; Guifferida, D.; Dugo, G.; Miceli, N.; Pergolizzi, S.; Taviano, M.F. Chemical characterization and biological effects of Sicilian Opuntia ficus indica (L.) Mill. Fruit Juice: Antioxidant and antiulcerogenic activity. J. Agric. Food Chem. 2003, 51, 4903–4908. [Google Scholar] [CrossRef]
- Butera, D.; Tesoriere, L.; Di Gaudio, F.; Bongiorno, M.; Allegra, M.; Pintaudi, A.M.; Kohen, R.; Livrea, M.A. Antioxidant activities of Sicilian prickly pear (Opuntia ficus indica) fruit extracts and reducing properties of its betalains: Betanin and indicaxanthin. J. Agric. Food Chem. 2002, 50, 6895–6901. [Google Scholar] [CrossRef]
- Kuti, J.O. Growth and compositional changes during the development of prickly pear fruit. J. Hortic. Sci. 1992, 67, 861–868. [Google Scholar]
- Shahidi, F.; Janitha, P.K.; Wanasundara, P.D. Phenolic antioxidanta, critical previous term reviews next term. Food Sci. Nutr. 1992, 32, 67–103. [Google Scholar]
- Aires, V.; Adote, S.; Hichami, A.; Moutairou, K.; Boustani, E.S.E.; Khan, N.A. Modulation of intracellular calcium concentrations and T cell activation by prickly pear polyphenols. Mol. Cell. Biochem. 2004, 260, 103–110. [Google Scholar] [CrossRef]
- Di Cesare, L.F.; Nani, R. Analysis of volatile constituents of prickly pear juice (Opuntia ficus indica var. Fructa sanguineo). Fruit Process. 1992, 2, 6–8. [Google Scholar]
- El Kossori, R.L.; Villaume, C.; El Boustani, E.; Sauvaire, Y.; Mejean, L. Composition of pulp, skin and seeds of prickly pears fruit (Opuntia ficus indica sp.). Plant Foods Hum. Nutr. 1998, 52, 263–270. [Google Scholar] [CrossRef]
- Russel, C.E.; Felker, P. The prickley pear (Opuntia spp., Cactaceae): A source of human and animal food in semi-arid regions. Econ. Bot. 1987, 41, 433–445. [Google Scholar] [CrossRef]
- Castellar, M.R.; Solano, F.; Obón, J.M. Betacyanin and other production during growth of Opuntia stricta (Haw.) fruits. Plant Foods Hum. Nutr. 2012, 64, 337–343. [Google Scholar] [CrossRef]
- De Leo, M.; Bruzual de Abreu, M.; Pawlowska, A.M.; Cioni, P.L.; Barca, A. Profiling the chemical content of Opuntia ficus-indica flowers by HPLC-PDA-ESI-MS and GC/EIMS analyses. Phytochem. Lett. 2010, 3, 48–52. [Google Scholar] [CrossRef]
- Yeddes, N.; Chérif, J.K.; Guyot, S.; Baron, A.; Trabelsi-Ayadi, M. Phenolic profile of Tunisian Opuntia ficus indica thornless form flowers via chromatographic and spectral analysis by RP-HPLC-UV-PDA and ESI-MS. Int. J. Food Prop. 2012. submitted for publication. [Google Scholar]
- Guyot, S.; Marnet, N.; Sanoner, P.; Drilleau, J.F. Variability of the polyphenolic composition of cider apple (Malus domestica) fruits and juices. J. Agric. Food Chem. 2003, 51, 6240–6247. [Google Scholar] [CrossRef]
- Yen, G.C.; Duth, P.D. Scavenging effect of methanolic extracts of peanut hulls on free radical and active-oxygen species. J. Agric. Food Chem. 1994, 42, 629–632. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic phosphotungustic acid reagents. Am. J. Enol. Viticul. 1965, 16, 144–158. [Google Scholar]
- Bahorun, T.; Grinier, B.; Trotin, F.; Brunet, G.; Pin, T.; Luncky, M.; Vasseur, J.; Cazin, M.; Cazin, C.; Pinkas, M. Oxygen species scavenging activity of phenolic extracts from hawthorn fresh plant organs and pharmaceutical preparations. Arzneim. Forsch. 1996, 46, 1086–1089. [Google Scholar]
- SPSS Software, version 11.5; SPSS Inc.: Chicago, IL, USA.
- Moussa-Ayoub, T.E.; El-Samahy, S.K.; Rohn, S.; Kroh, L.W. Flavonols, betacyanins content and antioxidant activity of Cactus Opuntia macrorhiza fruits. Food Res. Int. 2011, 44, 2169–2174. [Google Scholar] [CrossRef]
- Maatoui, B.S.; Hmyene, A.; Hilali, S. Activités anti-radiculaires d’extraits de jus de fruits du figuier de Barbarie (Opuntia ficus indica). Leban. Sci. J. 2006, 7, 3–8. [Google Scholar]
- Ammar, I.; Ennouri, M.; Khemakhem, B.; Yangui, T.; Attia, H. Variation of chemical composition and biological activities of two species of Opuntia flowers at four stages of flowering. Ind. Crop. Prod. 2012, 37, 34–40. [Google Scholar] [CrossRef]
- Díaz-Medina, E.M.; Rodríguez-Rodríguez, E.M.; Díaz-Romero, C. Chemical characterization of Opuntia dillenii and Opuntia ficus-indica fruits. Food Chem. 2007, 103, 38–45. [Google Scholar] [CrossRef]
- Ndhlala, A.R.; Kasiyamhuru, A.; Mupure, C.; Chitindingu, K.; Benhura, M.A.; Muchuweti, M. Phenolic composition of Flacourtia indica, Opuntia megacantha and Sclerocarya birrea. Food Chem. 2007, 103, 82–87. [Google Scholar] [CrossRef]
- Chiva-Blanch, G.; Visioli, F. Polyphenols and health: Moving beyond antioxidants. J. Berry Res. 2012, 2, 63–71. [Google Scholar]
- Alvarez-Suarez, J.M.; Giampieri, F.; González-Paramás, A.M.; Damiani, E.; Astolfi, P.; Marinez-Sanchez, G.; Bompadre, S.; Quiles, J.L.; Santos-Buelga, C.; Battino, M. Phenolics from monofloral honeys protect human erythrocyte membranes against oxidative damage. Food Chem. Toxicol. 2012, 50, 1508–1516. [Google Scholar] [CrossRef]
- Niki, E. Antioxidant capacity: Which capacity and how to asses it? J. Berry Res. 2011, 1, 169–176. [Google Scholar]
- Cai, Y.Z.; Sun, M.; Corke, H. Antioxidant activity of betalains from plants of Amaranthaceae. J. Agric. Food Chem. 2003, 51, 2288–2294. [Google Scholar] [CrossRef]
- Wollenweber, E. Exudates flavonoids of Mexican ferns as chemotaxonomic markers. Rev. Latinoamer. Quim. 1984, 15, 3–11. [Google Scholar]
- Fernández-López, J.A.; Almela, L.; Obón, J.M.; Castellar, R. Determination of antioxidant constituents in cactus pear fruits. Plant Foods Hum. Nutr. 2010, 65, 253–259. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yeddes, N.; Chérif, J.K.; Guyot, S.; Sotin, H.; Ayadi, M.T. Comparative Study of Antioxidant Power, Polyphenols, Flavonoids and Betacyanins of the Peel and Pulp of Three Tunisian Opuntia Forms. Antioxidants 2013, 2, 37-51. https://doi.org/10.3390/antiox2020037
Yeddes N, Chérif JK, Guyot S, Sotin H, Ayadi MT. Comparative Study of Antioxidant Power, Polyphenols, Flavonoids and Betacyanins of the Peel and Pulp of Three Tunisian Opuntia Forms. Antioxidants. 2013; 2(2):37-51. https://doi.org/10.3390/antiox2020037
Chicago/Turabian StyleYeddes, Nizar, Jamila K. Chérif, Sylvain Guyot, Hélène Sotin, and Malika T. Ayadi. 2013. "Comparative Study of Antioxidant Power, Polyphenols, Flavonoids and Betacyanins of the Peel and Pulp of Three Tunisian Opuntia Forms" Antioxidants 2, no. 2: 37-51. https://doi.org/10.3390/antiox2020037




