Improvement in the Biosynthesis of Antioxidant-Active Metabolites in In Vitro Cultures of Isatis tinctoria (Brassicaceae) by Biotechnological Methods/Elicitation and Precursor Feeding
Abstract
1. Introduction
2. Materials and Methods
2.1. Establishment of In Vitro Cultures
2.2. Optimization of Growth Conditions of Different Types of Cultures
2.2.1. Stationary Culture
2.2.2. Agitated Culture
2.3. Elicitation of Cultures
2.3.1. Elicitors Preparation
2.3.2. Elicitation Procedure
2.4. Precursors Feeding of Cultures
2.5. Extracts Preparation
2.6. Phytochemical Analysis
2.6.1. Determination of Total Phenolic Content
2.6.2. HPLC Analysis
2.7. Antioxidant Activity
2.7.1. Free Radical Scavenging Activity
2.7.2. Reducing Power
2.7.3. Ferrous Ion (Fe2+) Chelating Activity
2.8. Statistical Analysis
3. Results
3.1. Establishment of In Vitro Cultures
3.2. Optimization of Growth Conditions of Different Types of Cultures
3.2.1. Agar Culture
3.2.2. Agitated Culture
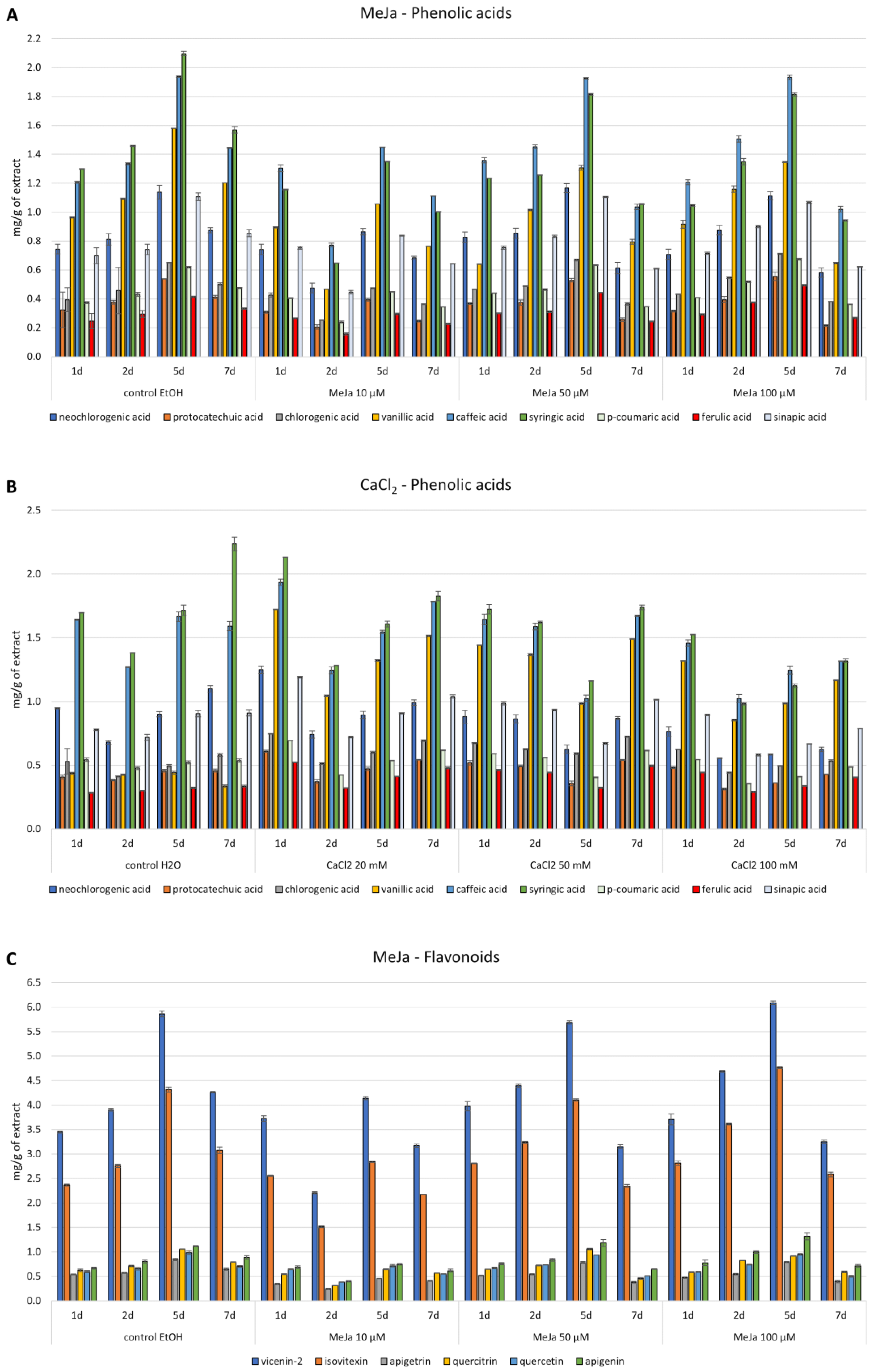
3.3. Elicitation of Cultures
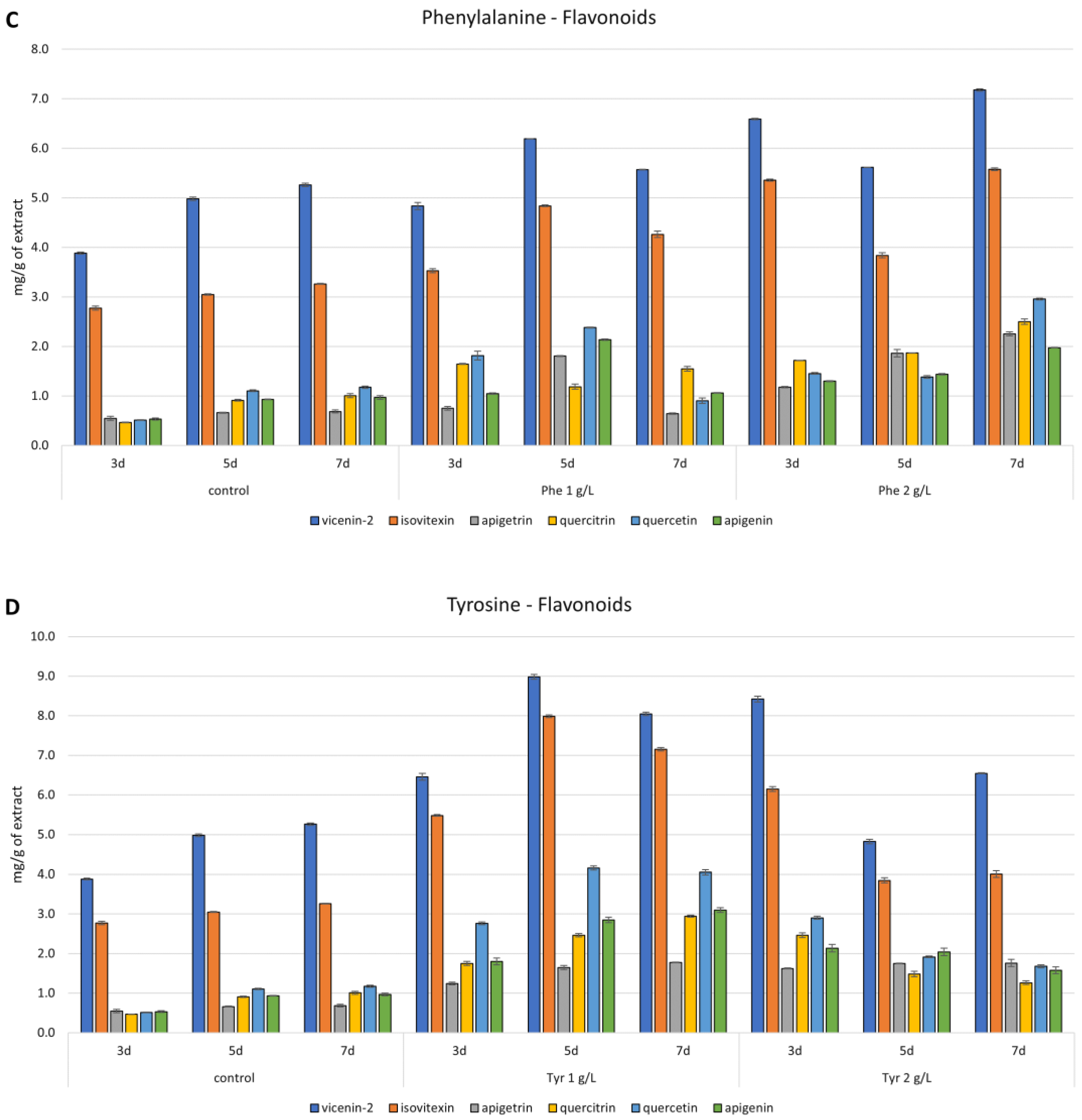
3.4. Precursors Feeding of Cultures
3.5. Phytochemical Investigations
3.5.1. Determinations of Total Phenolic Content
3.5.2. HPLC Analysis
3.6. Antioxidant Activity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ASE | Ascorbic acid equivalent |
| BAP | 6-Benzylaminopurine |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| DW | Dry Weight |
| GAE | Gallic Acid Equivalents |
| IBA | Indole-3-butyric acid |
| KIN | Kinetin |
| Tyr | L-Tyrosine |
| Phe | L-Phenylalanine |
| MeJa | Methyl Jasmonate |
| MS | Murashige and Skoog |
| NAA | 1-naphthaleneacetic acid |
| TPC | Total Phenolic Content |
| TE | Trolox equivalents |
| YE | Yeast |
References
- Speranza, J.; Miceli, N.; Taviano, M.F.; Ragusa, S.; Kwiecień, I.; Szopa, A.; Ekiert, H. Isatis tinctoria L. (Woad): A review of its botany, ethnobotanical uses, phytochemistry, biological activities, and biotechnological studies. Plants 2020, 9, 298. [Google Scholar] [CrossRef]
- Gai, Q.Y.; Jiao, J.; Wang, X.; Zang, Y.P.; Niu, L.L.; Fu, Y.J. Elicitation of Isatis tinctoria L. hairy root cultures by salicylic acid and methyl jasmonate for the enhanced production of pharmacologically active alkaloids and flavonoids. Plant Cell Tissue Organ Cult. 2019, 137, 77–86. [Google Scholar] [CrossRef]
- Zhang, D.; Ruan, D.; Li, J.; Chen, Z.; Zhu, W.; Guo, F.; Chen, K.; Li, Y.; Wang, R. Four undescribed sulfur-containing indole alkaloids with nitric oxide inhibitory activities from Isatis tinctoria L. roots. Phytochemistry 2020, 174, 112337. [Google Scholar] [CrossRef]
- European Directorate for the Quality of Medicines. Isatis root (Isatidis radix). In European Pharmacopoeia, 7th ed.; Suppl. 7.3 (2566); European Directorate for the Quality of Medicines: Strasburg, Germany, 2011; pp. 3866–3867. [Google Scholar]
- Kim, E.J.; Woo, J.; Shin, S.; Choi, H.; Kim, Y.; Kim, J.; Kang, C. A focused natural compound screen reveals senolytic and senostatic effects of Isatis tinctoria. Anim. Cells Syst. 2022, 26, 310–317. [Google Scholar] [CrossRef]
- Mohn, T.; Plitzko, I.; Hamburger, M. A comprehensive metabolite profiling of Isatis tinctoria leaf extracts. Phytochemistry 2009, 70, 924–934. [Google Scholar] [CrossRef]
- Xu, J.; Lan, Z.; Wang, S.; Guo, Y.; Ning, J.; Song, Y.; Liu, M.; Tian, C. The pharmaceutical applications of total flavonoids extract from Isatis tinctoria L. leaves. Pharmacol. Res. Mod. Chin. Med. 2022, 3, 100122. [Google Scholar] [CrossRef]
- Miceli, N.; Filocamo, A.; Ragusa, S.; Cacciola, F.; Dugo, P.; Mondello, L.; Celano, M.; Maggisano, V.; Taviano, M.F. Chemical characterization and biological activities of phenolic-rich fraction from cauline leaves of Isatis tinctoria L. (Brassicaceae) growing in Sicily, Italy. Chem. Biodivers. 2017, 14, e1700073. [Google Scholar] [CrossRef]
- Nicosia, N.; Kwiecień, I.; Mazurek, J.; Mika, K.; Bednarski, M.; Miceli, N.; Ragusa, S.; Ekiert, H.; Maes, M.; Kotańska, M. Hydroalcoholic Leaf Extract of Isatis tinctoria L. via Antioxidative and Anti-Inflammatory Effects Reduces Stress-Induced Behavioral and Cellular Disorders in Mice. Oxid Med. Cell Longev. 2022, 2022, 3567879. [Google Scholar] [CrossRef]
- Chen, G.; Wang, S.; Huang, X.; Hong, J.; Du, L.; Zhang, L.; Ye, L. Environmental factors affecting growth and development of Banlangen (Radix Isatidis) in China. Afr. J. Plant Sci. 2015, 9, 421–426. [Google Scholar]
- Isah, T.; Umar, S.; Mujib, A.; Sharma, M.P.; Rajasekharan, P.E.; Zafar, N.; Frukh, A. Secondary metabolism of pharmaceuticals in the plant in vitro cultures: Strategies, approaches, and limitations to achieving higher yield. Plant Cell Tissue Organ Cult. (PCTOC) 2018, 132, 239–265. [Google Scholar] [CrossRef]
- Gai, Q.Y.; Jiao, J.; Luo, M.; Wang, W.; Ma, W.; Zu, Y.G.; Fu, Y.J. Establishment of high-productive Isatis tinctoria L. hairy root cultures: A promising approach for efficient production of bioactive alkaloids. Biochem. Eng. J. 2015, 95, 37–47. [Google Scholar] [CrossRef]
- Gai, Q.Y.; Jiao, J.; Luo, M.; Wei, Z.F.; Zu, Y.G.; Ma, W.; Fu, Y.J. Establishment of hairy root cultures by Agrobacterium rhizogenes mediated transformation of Isatis tinctoria L. for the efficient production of flavonoids and evaluation of antioxidant activities. PLoS ONE 2015, 10, e0119022. [Google Scholar] [CrossRef]
- Jiao, J.; Gai, Q.Y.; Wang, X.; Qin, Q.P.; Wang, Z.Y.; Liu, J.; Fu, Y.J. Chitosan elicitation of Isatis tinctoria L. hairy root cultures for enhancing flavonoid productivity and gene expression and related antioxidant activity. Ind. Crops Prod. 2018, 124, 28–35. [Google Scholar] [CrossRef]
- Jiao, J.; Gai, Q.-Y.; Yao, L.-P.; Niu, L.-L.; Zang, Y.-P.; Fu, Y.-J. Ultraviolet radiation for flavonoid augmentation in Isatis tinctoria L. hairy root cultures mediated by oxidative stress and biosynthetic gene expression. Ind. Crops Prod. 2018, 118, 347–354. [Google Scholar] [CrossRef]
- Jiao, J.; Gai, Q.-Y.; Wang, W.; Zang, Y.-P.; Niu, L.-L.; Fu, Y.-J.; Wang, X. Remarkable enhancement of flavonoid production in a co-cultivation system of Isatis tinctoria L. hairy root cultures and immobilized Aspergillus niger. Ind. Crops Prod. 2018, 112, 252–261. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Leal, F.; Cipriano, J.; Carnide, V.; Pinto-Carnide, O. In vitro culture establishment of woad (Isatis tinctoria L.). In III International Symposium on Acclimatization and Establishment of Micropropagated Plants; ISHS: Leuven, Belgium, 2007; pp. 121–124. [Google Scholar]
- Ge, X.; Wu, J. Tanshinone production and isoprenoid pathways in Salvia miltiorrhiza hairy roots induced by Ag+ and yeast elicitor. Plant Sci. 2005, 168, 487–491. [Google Scholar] [CrossRef]
- DuBois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.T.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Buitelaar, R.M.; Casário, M.T.; Tramper, J. Elicitation of thiophene production by hairy roots of Tagetes patula. Enzyme Microb. Technol. 1992, 14, 2–7. [Google Scholar] [CrossRef]
- Isah, T.; Masood, S.; Umar, S. Biomass and camptothecin production in the calcium chloride elicited and liquid medium overlayed Nothapodytes nimmoniana (J. Graham) Mabberly callus cultures. Vegetos 2022, 35, 104–114. [Google Scholar] [CrossRef]
- Li, B.; Wang, B.; Li, H.; Peng, L.; Ru, M.; Liang, Z.; Yan, X.; Zhu, Y. Establishment of Salvia castanea Diels f. tomentosa Stib. hairy root cultures and the promotion of tanshinone accumulation and gene expression with Ag+, methyl jasmonate, and yeast extract elicitation. Protoplasma 2016, 253, 87–100. [Google Scholar] [CrossRef]
- Mendoza, D.; Cuaspud, O.; Arias, J.P.; Ruiz, O.; Arias, M. Effect of salicylic acid and methyl jasmonate in the production of phenolic compounds in plant cell suspension cultures of Thevetia peruviana. Biotechnol. Rep. 2018, 19, e00273. [Google Scholar] [CrossRef]
- Das, D.; Bandyopadhyay, M. Novel approaches towards over-production of andrographolide in in vitro seedling cultures of Andrographis paniculata. S. Afr. J. Bot. 2020, 128, 77–86. [Google Scholar] [CrossRef]
- Zhao, J.L.; Zhou, L.G.; Wu, J.Y. Effects of biotic and abiotic elicitors on cell growth and tanshinone accumulation in Salvia miltiorrhiza cell cultures. Appl Microbiol Biotechnol. 2010, 87, 137–144. [Google Scholar] [CrossRef]
- Herrmann, K.M. The shikimate pathway: Early steps in the biosynthesis of aromatic compounds. Plant Cell 1995, 7, 907–919. [Google Scholar] [CrossRef]
- Kwiecień, I.; Miceli, N.; D’Arrigo, M.; Marino, A.; Ekiert, H. Antioxidant potential and enhancement of bioactive metabolite production in in vitro cultures of Scutellaria lateriflora L. by biotechnological methods. Molecules 2022, 27, 1140. [Google Scholar] [CrossRef]
- Dewanjee, S.; Gangopadhyay, M.; Das, U.; Sahu, R.; Khanra, R. Enhanced rosmarinic acid biosynthesis in Solenostemon scutellarioides culture: A precursor-feeding strategy. Nat. Prod. Res. 2014, 28, 1691–1698. [Google Scholar] [CrossRef]
- Inyai, C.; Yusakul, G.; Komaikul, J.; Kitisripanya, T.; Likhitwitayawuid, K.; Sritularak, B.; Putalun, W. Improvement of stilbene production by mulberry Morus alba root culture via precursor feeding and co-elicitation. Bioprocess Biosyst. Eng. 2021, 44, 653–660. [Google Scholar] [CrossRef]
- Taviano, M.F.; Filocamo, A.; Ragusa, S.; Cacciola, F.; Dugo, P.; Mondello, L.; Paterniti Mastrazzo, G.; De Rose, R.F.; Celano, M.; Lombardo, G.E.; et al. Phenolic profile, antioxidant and cytotoxic properties of polar extracts from leaves and flowers of Isatis tinctoria L. (Brassicaceae) growing in Sicily. Plant Biosyst. Int. J. Deal. All Asp. Plant Biol. 2018, 152, 795–803. [Google Scholar] [CrossRef]
- Gao, X.; Ohlander, M.; Jeppsson, N.; Björk, L.; Trajkovski, V. Changes in antioxidant effects and their relationship to phytonutrients in fruits of sea buckthorn (Hippophae rhamnoides L.) during maturation. J. Agric. Food Chem. 2000, 48, 1485–1490. [Google Scholar] [CrossRef]
- Ellnain-Wojtaszek, M.; Zgórka, G. High-performance liquid chromatography and thin-layer chromatography of phenolic acids from Ginkgo biloba L. leaves collected within vegetative period. J. Liq. Chromatogr. Relat Technol. 1999, 22, 1457–1471. [Google Scholar] [CrossRef]
- Ohnishi, M.; Morishita, H.; Iwahashi, H.; Toda, S.; Shirataki, Y.; Kimura, M.; Kido, R. Inhibitory effects of chlorogenic acids on linoleic acid peroxidation and haemolysis. Phytochemistry 1994, 36, 579–583. [Google Scholar] [CrossRef]
- Oyaizu, M. Studies on products of browning reaction antioxidative activities of products of browning reaction prepared from glucosamine. Jpn. J. Nutr. Diet. 1986, 44, 307–315. [Google Scholar] [CrossRef]
- Decker, E.A.; Welch, B. Role of ferritin as a lipid oxidation catalyst in muscle food. J. Agric. Food Chem. 1990, 38, 674–677. [Google Scholar] [CrossRef]
- Nguyen, T.K.; Jamali, A.; Grand, E.; Morreel, K.; Marcelo, P.; Gontier, E.; Dauwe, R. Phenylpropanoid profiling reveals a class of hydroxycinnamoyl glucaric acid conjugates in Isatis tinctoria leaves. Phytochemistry 2017, 144, 127–140. [Google Scholar] [CrossRef]
- Platzer, M.; Kiese, S.; Herfellner, T.; Schweiggert-Weisz, U.; Eisner, P. How Does the Phenol Structure Influence the Results of the Folin-Ciocalteu Assay? Antioxidants 2021, 10, 811. [Google Scholar] [CrossRef]
- Guan, Y.; Hu, W.; Jiang, A.; Xu, Y.; Sa, R.; Feng, K.; Zhao, M.; Yu, J.; Ji, Y.; Hou, M.; et al. Effect of Methyl Jasmonate on Phenolic Accumulation in Wounded Broccoli. Molecules 2019, 24, 3537. [Google Scholar] [CrossRef]
- Poulev, A.; O’Neal, J.M.; Logendra, S.; Pouleva, R.B.; Timeva, V.; Garvey, A.S.; Gleba, D.; Jenkins, I.S.; Halpern, B.T.; Kneer, R.; et al. Elicitation, a new window into plant chemo diversity and phytochemical drug discovery. J. Med. Chem. 2003, 46, 2542–2547. [Google Scholar] [CrossRef]
- Angelova, Z.; Georgiev, S.; Roos, W. Elicitation of plants. Biotechnol. Biotechnol. Equip. 2006, 20, 72–83. [Google Scholar] [CrossRef]
- Namdeo, A.G. Plant cell elicitation for production of secondary metabolites: A review. Pharmacog. Rev. 2007, 1, 69–79. [Google Scholar]
- Naik, P.M.; Al-Khayri, J.M. Abiotic and biotic elicitors–role in secondary metabolites production through in vitro culture of medicinal plants. In Abiotic and Biotic Stress in Plants-Recent Advances and Future Perspectives; Shanker, A.K., Shanker, C., Eds.; InTech: Rijeka, Croatia, 2016; pp. 247–277. [Google Scholar]
- Thakur, M.; Bhattacharya, S.; Khosla, P.K.; Puri, S. Improving production of plant secondary metabolites through biotic and abiotic elicitation. J. App. Res. Med. Aromat. Plants 2019, 12, 1–12. [Google Scholar] [CrossRef]
- Kumari, N.; Varghese, B.A.; Khan, M.A.; Jangra, S.; Kumar, A. Biotic elicitation: A tool for producing bioactive compounds. Plant Arch. 2020, 20, 3385–3391. [Google Scholar]
- Kikowska, M.; Thiem, B.; Jafernik, K.; Klimek-Szczykutowicz, M.; Studzińska-Sroka, E.; Ekiert, H.; Szopa, A. Effect of Elicitation with (+)-Usnic Acid on Accumulation of Phenolic Acids and Flavonoids in Agitated Microshoots of Eryngium alpinum L. Molecules 2021, 26, 5532. [Google Scholar] [CrossRef]
- Arya, S.S.; Rookes, J.E.; Cahill, D.M.; Lenka, S.K. Chitosan nanoparticles and their combination with methyl jasmonate for the elicitation of phenolics and flavonoids in plant cell suspension cultures. Int. J. Biol. Macromol. 2022, 214, 632–641. [Google Scholar] [CrossRef]
- Kawka, B.; Kwiecień, I.; Ekiert, H. Production of specific flavonoids and verbascoside in shoot cultures of Scutellaria baicalensis. In Plant Cell and Tissue Differentiation and Secondary Metabolites; Reference Series in Phytochemistry; Ramawat, K., Ekiert, H., Goyal, S., Eds.; Springer Nature: Geneva, Switzerland, 2021; pp. 249–272. [Google Scholar]
- Rodriguez Basso, A.; Marrassini, C.; Anesini, C.; Gorzalczany, S. Acute Hypotensive, Diuretic and Antioxidant Activities Induced by Urtica circularis. Eur. J. Med. Plants 2016, 14, 1–9. [Google Scholar] [CrossRef]
- Cai, Y.-Z.; Sun, M.; Xing, J.; Luo, Q.; Corke, H. Structure-Radical Scavenging Activity Relationships of Phenolic Compounds from Traditional Chinese Medicinal Plants. Life Sci. 2006, 78, 2872–2888. [Google Scholar] [CrossRef]
- Velozo, L.S.M.; Ferreira, M.J.P.; Santos, M.I.S.; Moreira, D.L.; Guimaraes, E.F.; Emerenciano, V.P.; Kaplan, M.A.C. C-Glycosyl Flavones from Peperomia blanda. Fitoterapia 2009, 80, 119–122. [Google Scholar] [CrossRef]
- Platzer, M.; Kiese, S.; Herfellner, T.; Schweiggert-Weisz, U.; Miesbauer, O.; Eisner, P. Common Trends and Differences in Antioxidant Activity Analysis of Phenolic Substances Using Single Electron Transfer Based Assays. Molecules 2021, 26, 1244. [Google Scholar] [CrossRef]
- Sroka, Z.; Cisowski, W. Hydrogen peroxide scavenging, antioxidant and antiradical activity of some phenolic acids. Food Chem. Toxicol. 2003, 41, 753–758. [Google Scholar] [CrossRef]





| GRs BAP + NAA (mg/L) | Fresh Weight (g) After 2 Weeks | Dried Weight (g) After 2 Weeks | Time Fold |
|---|---|---|---|
| 2.0/1.0 | 3.371 ± 0.154 a | 0.258 ± 0.032 a | 3.189 ± 0.487 a |
| 1.0/0.5 | 3.467 ± 0.386 a | 1.011 ± 0.022 a | 3.295 ± 0.369 a |
| 1.0/1.0 | 3.797 ± 0.750 a | 0.290 ± 0.045 a | 3.579 ± 0.705 a |
| 0/1.0 | 1.959 ± 0.363 b | 0.173 ± 0.027 b | 1.840 ± 0.345 b |
| 1.0/0 | 2.785 ± 0.794 a,b | 0.187 ± 0.069 a,b | 2.622 ± 0.752 a,b |
| 0.5/0 | 3.538 ± 0.827 a | 0.264 ± 0.036 a | 3.321 ± 0.775 a |
| PGRs BAP + NAA (mg/L) | Fresh Weight (g) After 2 Weeks | Dried Weight (g) After 2 Weeks | Time Fold |
|---|---|---|---|
| 2.0/1.0 | 5.927 ± 2.014 a | 0.447 ± 0.134 a | 11.663 ± 4.046 a |
| 1.0/0.5 | 4.424 ± 3.353 a,b | 0.425 ± 0.212 a | 8.654 ± 6.586 a,b |
| 1.0/1.0 | 5.038 ± 2.272 a | 0.401 ± 0.146 a | 9.973 ± 4.524 a |
| 0/1.0 | 0.996 ± 0.589 b | 0.092 ± 0.051 b | 1.964 ± 1.154 b |
| 1.0/0 | 5.589 ± 1.511 a | 0.467 ± 0.110 a | 11.027 ± 2.996 a |
| 0.5/0 | 3.092 ± 1.646 a,b | 0.223 ± 0.118 a,b | 6.117 ± 3.252 a,b |
| Elicitor | Dry Biomass Weight (g) | ||||
|---|---|---|---|---|---|
| 24 h | 48 h | 120 h | 168 h | ||
| MeJa | 10 µM | 1.324 ± 0.097 | 0.586 ± 0.381 | 1.216 ± 0.447 | 0.678 ± 0.371 |
| 50 µM | 1.230 ± 0.025 | 1.076 ± 0.741 | 0.967 ± 0.754 | 0.335 ± 0.195 | |
| 100 µM | 1.329 ± 0.239 | 0.961 ± 0.847 | 0.922 ± 0.600 | 0.463 ± 0.649 | |
| Ctr EtOH 50% | 0.830 ± 0.404 | 0.784 ± 0.345 | 1.064 ± 0.634 | 0.379 ± 0.058 | |
| CaCl2 | 20 mM | 1.215 ± 0.552 | 0.251 ± 0.096 | 0.258 ± 0.116 | 0.305 ± 0.067 |
| 50 mM | 1.553 ± 0.060 | 0.273 ± 0.038 | 0.317 ± 0.093 | 0.556 ± 0.120 | |
| 100 mM | 1.038 ± 0.832 | 0.346 ± 0.157 | 0.383 ± 0.252 | 0.537 ± 0.184 | |
| Ctr H2O | 0.746 ± 0.650 | 0.490 ± 0.354 | 0.836 ± 0.636 | 0.835 ± 0.616 | |
| AgNO3 | 0.5 mM | 1.112 ± 0.166 | 0.684 ± 0.242 | 0.430 ± 0.057 | |
| 1 mM | 1.347 ± 0.144 | 1.306 ± 0.126 | 0.965 ± 0.276 | ||
| 2 mM | 0.660 ± 0.351 | 0.709 ± 0.092 | 0.618 ± 0.185 | ||
| Ctr H2O | 0.746 ± 0.650 | 0.490 ± 0.354 | 0.836 ± 0.636 | ||
| YE extract | 50 mg/L | 0.609 ± 0.150 | 0.422 ± 0.171 | 0.515 ± 0.069 | |
| 200 mg/L | 0.378 ± 0.028 | 0.532 ± 0.345 | 0.707 ± 0.201 | ||
| 300 mg/L | 0.410 ± 0.142 | 0.534 ± 0.267 | 0.567 ± 0.051 | ||
| Ctr H2O | 0.490 ± 0.354 | 0.836 ± 0.636 | 0.835 ± 0.616 | ||
| Precursor (g/L) | Dry Biomass Weight (g) | |||
|---|---|---|---|---|
| 72 h | 120 h | 168 h | ||
| Phe | 1 | 1.210 ± 0.637 | 1.358 ± 0.246 | 0.852 ± 0.334 |
| 2 | 1.290 ± 0.684 | 1.096 ± 0.203 | 1.136 ± 0.225 | |
| Ctr H2O | 0.426 ± 0.083 | 0.836 ± 0.636 | 0.835 ± 0.616 | |
| Tyr | 1 | 1.485 ± 0.225 | 1.288 ± 0.183 | 0.643 ± 0.260 |
| 2 | 1.082 ± 0.269 | 0.779 ± 0.319 | 0.319 ± 0.098 | |
| Ctr H2O | 0.426 ± 0.083 | 0.836 ± 0.636 | 0.835 ± 0.616 | |
| MS Medium Variant BAP/NAA (mg/L) | Total Phenolic Content (TPC) mg GAE/g Extract (DW) |
|---|---|
| 2.0/1.0 | 13.833 ± 0.506 a,b |
| 1.0/0.5 | 13.306 ± 0.973 a,b |
| 1.0/1.0 | 14.451 ± 1.043 a |
| 1.0/0 | 12.411 ± 0.566 b |
| 0.5/0 | 12.818 ± 0.161 b |
| 0/1.0 | 9.604 ± 0.555 c |
| Elicitor | Total Phenolic Content (TPC) mg GAE/g Extract DW | ||||
|---|---|---|---|---|---|
| 24 h | 48 h | 120 h | 168 h | ||
| MeJa | 10 µM | 18.91 ± 0.21 aA | 11.92 ± 0.20 aB | 24.96 ± 0.34 aC | 16.27 ± 0.07 aA |
| 50 µM | 22.58 ± 1.15 abA | 24.82 ± 0.69 bA | 33.36 ± 1.65 bB | 16.14 ± 0.27 aC | |
| 100 µM | 22.99 ± 0.46 abA | 26.91 ± 2.09 bAB | 28.42 ± 2.95 aB | 17.45 ± 0.28 aC | |
| CtrEtOH50% | 16.80 ± 1.54 acA | 19.24 ± 1.01 cA | 36.54 ± 0.22 bB | 23.19 ± 0.55 bC | |
| CaCl2 | 20 mM | 30.43 ± 1.06 aA | 20.52 ± 0.27 aB | 25.51 ± 0.02 aC | 29.51 ± 1.66 aD |
| 50 mM | 29.87 ± 1.07 aA | 26.51 ± 0.54 bB | 19.15 ± 1.32 bC | 31.57 ± 1.47 aA | |
| 100 mM | 25.73 ± 0.26 bA | 16.67 ± 1.55 cB | 19.80 ± 0.65 bC | 24.13 ± 0.79 bA | |
| Ctr H2O | 24.74 ± 0.84 bA | 20.23 ± 0.86 aB | 29.56 ± 1.44 cC | 32.93 ± 0.42 aC | |
| AgNO3 | 0.5 mM | 17.85 ± 0.72 aA | 17.04 ± 0.62 aA | 19.79 ± 0.07 aA | |
| 1 mM | 18.67 ± 0.86 aA | 14.88 ± 0.14 bB | 17.75 ± 0.51 bA | ||
| 2 mM | 15.68 ± 0.26 bA | 13.52 ± 0.74 bB | 16.30 ± 0.42 bA | ||
| Ctr H2O | 24.74 ± 0.84 cA | 20.23 ± 0.86 dB | 29.56 ± 1.44 cC | ||
| YE extract | 50 mg/L | 18.85 ± 0.97 aA | 17.77 ± 0.03 aA | 20.70 ± 0.02 aA | |
| 200 mg/L | 18.93 ± 3.12 aA | 17.50 ± 0.19 aA | 15.57 ± 0.23 bA | ||
| 300 mg/L | 18.58 ± 0.10 aA | 16.03 ± 0.18 bA | 20.91 ± 0.46 aA | ||
| Ctr H2O | 20.23 ± 0.86 aA | 29.56 ± 1.44 cB | 32.93 ± 0.42 cC | ||
| Precursor (g/L) | Total Phenolic Content (TPC) mg GAE/g Extract DW | |||
|---|---|---|---|---|
| 72 h | 120 h | 168 h | ||
| Phe | 1 | 27.28 ± 1.20 aA | 36.00 ± 0.11 aB | 25.73 ± 1.14 aA |
| 2 | 36.36 ± 2.76 bA | 32.585 ± 1.00 aA | 41.43 ± 6.45 bB | |
| Ctr H2O | 17.75 ± 0.48 cA | 29.556 ± 1.44 aB | 32.93 ± 0.42 abB | |
| Tyr | 1 | 44.48 ± 1.15 aA | 58.65 ± 0.91 aB | 60.36 ± 4.97 aB |
| 2 | 49.37 ± 0.93 aA | 36.52 ± 6.99 bB | 34.97 ± 0.45 bB | |
| Ctr H2O | 17.75 ± 0.48 bA | 29.56 ± 1.44 cB | 32.93 ± 0.42 bB | |
| MS Medium Variant BAP/NAA (mg/L) | DPPH Test mg TE/g Extract | Chelating Activity IC50 (mg/mL) |
|---|---|---|
| 2.0/1.0 | 7.38 ± 0.34 a,c | 0.80 ± 0.05 a,e |
| 1.0/0.5 | 7.98 ± 0.45 a | 0.83 ± 0.07 a |
| 1.0/1.0 | 9.65 ± 0.26 b | 0.64 ± 0.01 b |
| 1.0/0 | 9.81 ± 0.69 b | 1.01 ± 0.01 c |
| 0.5/0 | 6.53 ± 0.33 c | 1.40 ± 0.06 d |
| 0/1.0 | 6.39 ± 0.42 c | 0.75 ± 0.01 e |
| Elicitor | DPPH Test mg TE/g Extract | ||||
|---|---|---|---|---|---|
| 24 h | 48 h | 120 h | 168 h | ||
| MeJa | 10 µM | 14.90 ± 0.84 aA | 8.37 ± 0.70 aB | 18.90 ± 0.51 aC | 16.39 ± 0.64 aD |
| 50 µM | 15.48 ± 1.17 bA | 21.12 ± 0.78 bB | 24.20 ± 0.66 bC | 13.46 ± 0.09 bD | |
| 100 µM | 17.11 ± 0.54 bA | 18.57 ± 0.37 cA | 24.79 ± 0.38 bB | 6.50 ± 0.03 cC | |
| Ctr EtOH 50% | 15.43 ± 0.51 bA | 14.28 ± 0.28 dA- | 25.27 ± 0.43 bB | 12.15 ± 0.59 bdC | |
| CaCl2 | 20 mM | 21.91 ± 0.65 aA | 13.16 ± 0.34 aB | 16.81 ± 0.46 aC | 20.99 ± 0.91 aA |
| 50 mM | 25.14 ± 0.35 bA | 15.89 ± 0.49 bB | 11.35 ± 0.41 bC | 23.86 ± 0.84 bA | |
| 100 mM | 19.22 ± 0.31 cA | 16.03 ± 0.51 bB | 12.38 ± 0.25 cC | 18.71 ± 1.07 cA | |
| Ctr H2O | 18.44 ± 0.01 cA | 18.01 ± 0.13 cA | 24.49 ± 0.088 dB | 25.24 ± 0.63 bC | |
| AgNO3 | 0.5 mM | 11.08 ± 0.97 aA | 9.72 ± 0.78 aB | 6.62 ± 0.03 aC | |
| 1 mM | 9.20 ± 0.15 bA | 11.88 ± 0.19 bB | 10.92 ± 0.28 bB | ||
| 2 mM | 7.12 ± 0.70 cA | 5.19 ± 0.17 cB | 4.25 ± 0.58 cB | ||
| Ctr H2O | 18.44 ± 0.01 dA | 18.01 ± 0.13 dA | 24.49 ± 0.09 dB | ||
| YE extract | 50 mg/L | 17.65 ± 0.46 aA | 19.73 ± 0.26 aB | 19.70 ± 0.44 aB | |
| 200 mg/L | 12.28 ± 0.01 bA | 22.03 ± 0.79 bB | 15.54 ± 0.11 bC | ||
| 300 mg/L | 15.71 ± 0.31 cA | 19.01 ± 0.23 bB | 18.25 ± 0.01 cB | ||
| Ctr H2O | 18.01 ± 0.13 aA | 24.49 ± 0.09 cB | 25.24 ± 0.63 dC | ||
| Precursor (g/L) | DPPH Test mg TE/g Extract | |||
|---|---|---|---|---|
| 72 h | 120 h | 168 h | ||
| Phe | 1 | 12.70 ± 0.02 aA | 19.36 ± 0.94 aB | 12.4 ± 0.45 aA |
| 2 | 19.51 ± 0.55 bA | 21.85 ± 0.15 bB | 17.44 ± 0.03 bC | |
| Ctr H2O | 7.23 ± 0.40 cA | 24.49 ± 0.09 cB | 25.24 ± 0.63 cB | |
| Tyr | 1 | 13.98 ± 0.90 aA | 16.27 ± 0.35 aB | 17.49 ± 0.04 aB |
| 2 | 17.63 ± 0.08 bA | 18.82 ± 0.23 bA | 6.70 ± 0.95 bB | |
| Ctr H2O | 7.23 ± 0.40 cA | 24.49 ± 0.09 cB | 25.24 ± 0.63 cB | |
| Elicitor | Chelating Activity IC50 (mg/mL) | ||||
|---|---|---|---|---|---|
| 24 h | 48 h | 120 h | 168 h | ||
| MeJa | 10 µM | 0.51 ± 0.01 aA | 1.57 ± 0.04 aB | 0.64 ± 0.02 aC | >2 aD |
| 50 µM | 0.94 ± 0.01 bA | 1.32 ± 0.01 bB | 1.22 ± 0.02 bC | >2 aD | |
| 100 µM | 1.17 ± 0.01 cA | 1.11 ± 0.02 cA | 0.99 ± 0.02 cB | Na | |
| Ctr EtOH 50% | 0.89 ± 0.01 bA | 0.91 ± 0.01 dA | 0.76 ± 0.01 dB | 1.18 ± 0.02 bC | |
| CaCl2 | 20 mM | Na | Na | Na | Na |
| 50 mM | Na | Na | Na | Na | |
| 100 mM | Na | Na | Na | Na | |
| Ctr H2O | >2 A | >2 A | 0.97 ± 0.01 B | 1.74 ± 0.04 C | |
| AgNO3 | 0.5 mM | >2 a | >2 a | >2 a | |
| 1 mM | 1.53 ± 0.02 bA | 0.76 ± 0.05 bB | 1.35 ± 0.02 bC | ||
| 2 mM | >2 aA | 1.14 ± 0.03 cB | >2 aA | ||
| Ctr H2O | >2 cA | >2 aA | 0.97 ± 0.01 cB | ||
| YE extract | 50 mg/L | >2 a | >2 a | >2 a | |
| 200 mg/L | >2 a | >2 a | >2 a | ||
| 300 mg/L | Na | Na | Na | ||
| Ctr H2O | >2 aA | 0.97 ± 0.01 bB | 1.74 ± 0.04 bC | ||
| Precursor (g/L) | Chelating Activity IC50 (mg/mL) | |||
|---|---|---|---|---|
| 72 h | 120 h | 168 h | ||
| Phe | 1 | 1.48 ± 0.05 aA | 1.33 ± 0.03 aB | 1.63 ± 0.05 aC |
| 2 | 1.06 ± 0.01 bA | 1.05 ± 0.03 bA | 0.85 ± 0.01 bB | |
| Ctr H2O | >2 cA | 0.97 ± 0.01 bB | 1.74 ± 0.04 cC | |
| Tyr | 1 | 0.54 ± 0.03 aA | 0.69 ± 0.02 aB | 1.42 ± 0.02 aC |
| 2 | 0.27 ± 0.01 bA | 0.57 ± 0.03 bB | 1.86 ±0.07 bC | |
| Ctr H2O | >2 cA | 0.97 ± 0.01 cB | 1.74 ± 0.04 cC | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miceli, N.; Kwiecień, I.; Nicosia, N.; Speranza, J.; Ragusa, S.; Cavò, E.; Davì, F.; Taviano, M.F.; Ekiert, H. Improvement in the Biosynthesis of Antioxidant-Active Metabolites in In Vitro Cultures of Isatis tinctoria (Brassicaceae) by Biotechnological Methods/Elicitation and Precursor Feeding. Antioxidants 2023, 12, 1111. https://doi.org/10.3390/antiox12051111
Miceli N, Kwiecień I, Nicosia N, Speranza J, Ragusa S, Cavò E, Davì F, Taviano MF, Ekiert H. Improvement in the Biosynthesis of Antioxidant-Active Metabolites in In Vitro Cultures of Isatis tinctoria (Brassicaceae) by Biotechnological Methods/Elicitation and Precursor Feeding. Antioxidants. 2023; 12(5):1111. https://doi.org/10.3390/antiox12051111
Chicago/Turabian StyleMiceli, Natalizia, Inga Kwiecień, Noemi Nicosia, Jasmine Speranza, Salvatore Ragusa, Emilia Cavò, Federica Davì, Maria Fernanda Taviano, and Halina Ekiert. 2023. "Improvement in the Biosynthesis of Antioxidant-Active Metabolites in In Vitro Cultures of Isatis tinctoria (Brassicaceae) by Biotechnological Methods/Elicitation and Precursor Feeding" Antioxidants 12, no. 5: 1111. https://doi.org/10.3390/antiox12051111
APA StyleMiceli, N., Kwiecień, I., Nicosia, N., Speranza, J., Ragusa, S., Cavò, E., Davì, F., Taviano, M. F., & Ekiert, H. (2023). Improvement in the Biosynthesis of Antioxidant-Active Metabolites in In Vitro Cultures of Isatis tinctoria (Brassicaceae) by Biotechnological Methods/Elicitation and Precursor Feeding. Antioxidants, 12(5), 1111. https://doi.org/10.3390/antiox12051111








