An Experimental Study on Antioxidant Enzyme Gene Expression in Trematomus newnesi (Boulenger, 1902) Experimentally Exposed to Perfluoro-Octanoic Acid
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Procedures
2.2. Sampling Activity
2.3. Primer Design; Total RNA Extraction; and sod1, sod2, gpx1, and gpx4 cDNA Synthesis
2.4. qRT-PCR Analysis
2.5. Statistical Analysis
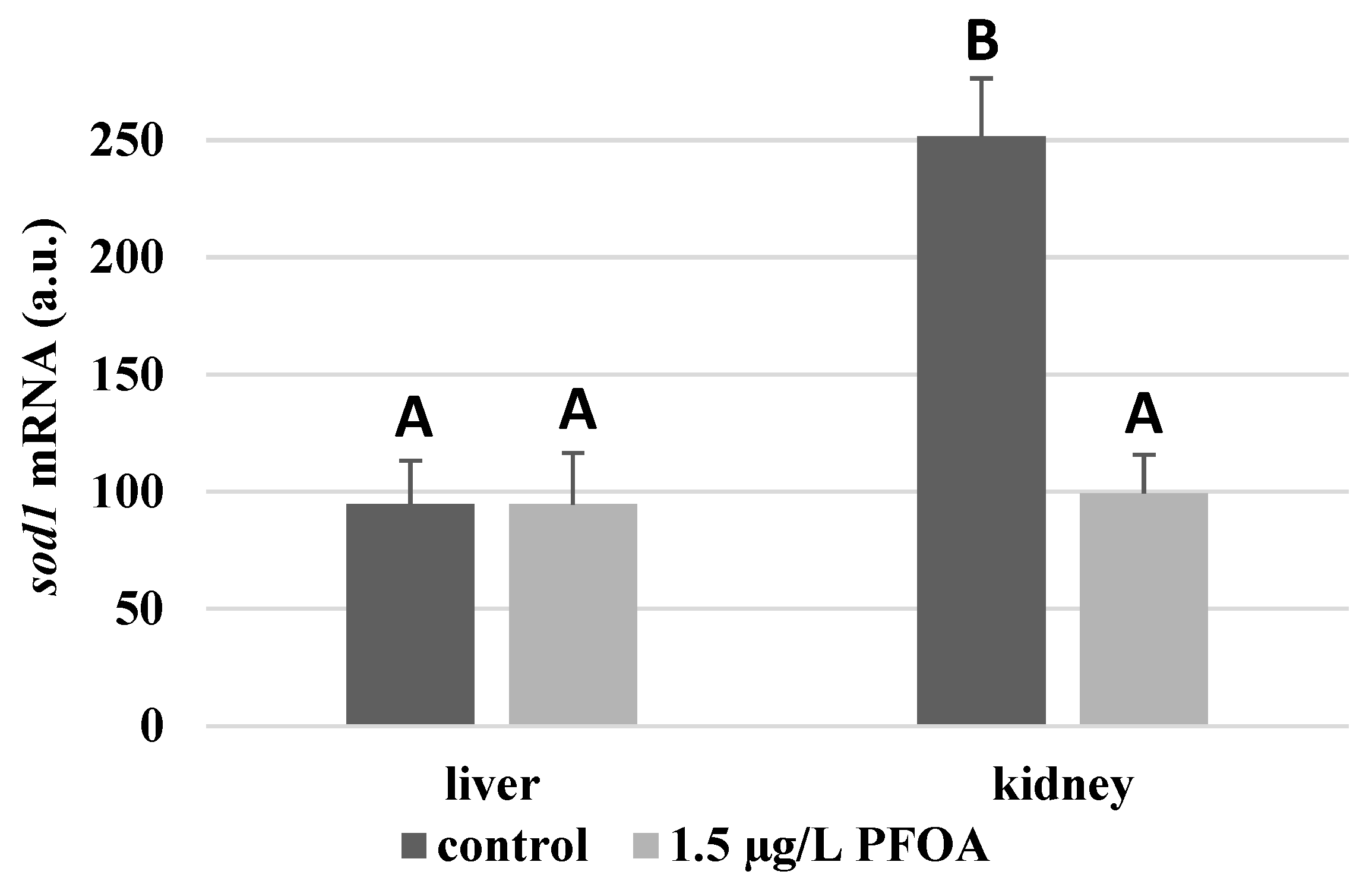
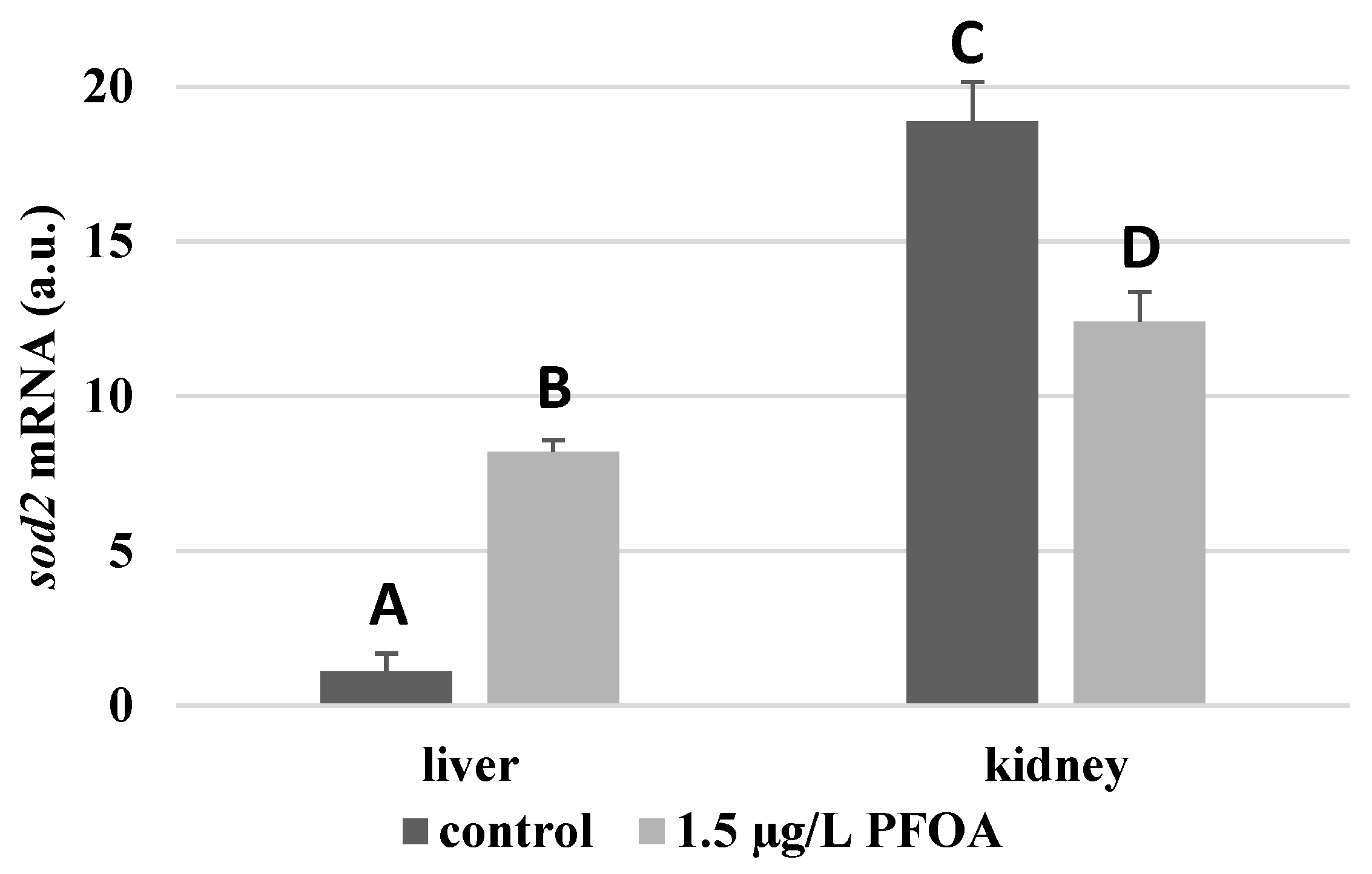
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Corsolini, S.; Ademollo, N. POPs in Antarctic ecosystems: Is climate change affecting their temporal trends? Environ. Sci. Process. Impacts 2022, 24, 1631–1642. [Google Scholar] [CrossRef]
- Marrone, A.; La Russa, D.; Brunelli, E.; Santovito, G.; La Russa, M.F.; Barca, D.; Pellegrino, D. Antarctic Fish as a Global Pollution Sensor: Metals Biomonitoring in a Twelve-Year Period. Front. Mol. Biosci. 2021, 8, 794946. [Google Scholar] [CrossRef] [PubMed]
- Bonato, M.; Corrà, F.; Bellio, M.; Guidolin, L.; Tallandini, L.; Irato, P.; Santovito, G. PFAS Environmental Pollution and Antioxidant Responses: An Overview of the Impact on Human Field. Int. J. Environ. Res. Public Health 2020, 17, 8020. [Google Scholar] [CrossRef] [PubMed]
- Stockholm Convention. Available online: http://chm.pops.int/TheConvention/ThePOPs/AllPOPs/tabid/2509/Default.aspx (accessed on 4 November 2022).
- Casal, P.; González-Gaya, B.; Zhang, Y.; Reardon, A.J.F.; Martin, J.W.; Jiménez, B.; Dachs, J. Accumulation of Perfluoroalkylated Substances in Oceanic Plankton. Environ. Sci. Technol. 2017, 51, 2766–2775. [Google Scholar] [CrossRef] [PubMed]
- Kurwadkar, S.; Dane, J.; Kanel, S.R.; Nadagouda, M.N.; Cawdrey, R.W.; Ambade, B.; Struckhoff, G.C.; Wilkin, R. Per- and polyfluoroalkyl substances in water and wastewater: A critical review of their global occurrence and distribution. Sci. Total Environ. 2022, 809, 151003. [Google Scholar] [CrossRef]
- Gao, K.; Miao, X.; Fu, J.; Chen, Y.; Li, H.; Pan, W.; Fu, J.; Zhang, Q.; Zhang, A.; Jiang, G. Occurrence and Trophic Transfer of Per- and Polyfluoroalkyl Substances in an Antarctic Ecosystem. Environ. Pollut. 2020, 257, 113383. [Google Scholar] [CrossRef]
- O’Brien, K.M.; Oldham, C.A.; Sarrimanolis, J.; Fish, A.; Castellini, L.; Vance, J.; Lekanof, H.; Crockett, E.L. Warm Acclimation Alters Antioxidant Defences but Not Metabolic Capacities in the Antarctic Fish, Notothenia coriiceps. Conserv. Physiol. 2022, 10, coac054. [Google Scholar] [CrossRef]
- Daane, J.M.; Detrich, H.W., 3rd. Adaptations and Diversity of Antarctic Fishes: A Genomic Perspective. Annu. Rev. Anim. Biosci. 2022, 10, 39–62. [Google Scholar] [CrossRef]
- Vacchi, M. I pesci e la pesca. PNRA, Museo Nazionale dell’Antartide Felice Ippolito. Available online: https://mna.it/sites/default/files/in_antartide/pesci_pesca.pdf (accessed on 6 June 2022).
- Di Prisco, G. I pesci—Evoluzione e Adattamento alle Basse Temperature. PNRA, Museo Nazionale dell’Antartide Felice Ippolito. Available online: https://mna.it/sites/default/files/in_antartide/pesci_evoluzione_adatt.pdf (accessed on 6 June 2022).
- Bakiu, R.; Boldrin, F.; Pacchini, S.; Schumann, S.; Piva, E.; Tolomeo, A.M.; Ferro, D.; Grapputo, A.; Santovito, G.; Irato, P. Molecular Evolution of Metallothioneins of Antarctic Fish: A Physiological Adaptation to Peculiar Seawater Chemical Characteristics. J. Mar. Sci. Eng. 2022, 10, 1592. [Google Scholar] [CrossRef]
- Zorov, D.B.; Juhaszova, M.; Sollott, S.J. Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS release. Physiol Rev. 2014, 94, 909–950. [Google Scholar] [CrossRef]
- Irato, P.; Santovito, G. Enzymatic and non-enzymatic molecules with antioxidant function. Antioxidants 2022, 10, 579. [Google Scholar] [CrossRef]
- Di Giulio, R.D. Indices of Oxidative Stress as Biomarkers for Environmental Contamination. In Aquatic Toxicology and Risk Assessment: Fourteenth Volume; Mayes, M.A., Barron, M.G., Eds.; ASTM International: West Conshohocken, PA, USA, 1991; pp. 15–31. [Google Scholar]
- Benedetti, M.; Giuliani, M.E.; Regoli, F. Oxidative metabolism of chemical pollutants in marine organisms: Molecular and biochemical biomarkers in environmental toxicology. Ann. N. Y. Acad. Sci. 2015, 1340, 8–19. [Google Scholar] [CrossRef]
- Hu, X.-Z.; Hu, D.-C. Effects of Perfluorooctanoate and Perfluorooctane Sulfonate Exposure on Hepatoma Hep G2 Cells. Arch. Toxicol. 2009, 83, 851–861. [Google Scholar] [CrossRef]
- Piva, E.; Schumann, S.; Dotteschini, S.; Brocca, G.; Radaelli, G.; Marion, A.; Irato, P.; Bertotto, D.; Santovito, G. Antioxidant Responses Induced by PFAS Exposure in Freshwater Fish in the Veneto Region. Antioxidants 2022, 11, 1115. [Google Scholar] [CrossRef]
- Wilhelm Filho, D. Reactive Oxygen Species, Antioxidants and Fish Mitochondria. Front. Biosci. 2007, 12, 1229–1237. [Google Scholar] [CrossRef]
- Gon, O.; Heemstra, P.C. Fishes of the Southern Ocean; J.L.B. Smith Institute of Ichthyology: Grahamstown, South Africa, 1990. [Google Scholar]
- Miller, R. History and Atlas of the Fishes of the Antarctic Ocean; Foresta Institute for Ocean and Mountain Studies: Carson City, NV, USA, 1993. [Google Scholar]
- Sidell, B.D. Life at body temperatures below 0 degrees C: The physiology and biochemistry of Antarctic fishes. Gravit. Space Biol. Bull. 2000, 13, 25–34. [Google Scholar]
- Peck, L.S. Prospects for surviving climate change in Antarctic aquatic species. Front. Zool. 2005, 2, 9. [Google Scholar] [CrossRef]
- Ricci, F.; Lauro, F.M.; Grzymski, J.J.; Read, R.; Bakiu, R.; Santovito, G.; Luporini, P.; Vallesi, A. The antioxidant defense system of the marine polar ciliate Euplotes nobilii: Characterization of the msrB gene family. Biology 2017, 6, 4. [Google Scholar] [CrossRef]
- Chatzidimitriou, E.; Bisaccia, P.; Corrà, F.; Bonato, M.; Irato, P.; Manuto, L.; Toppo, S.; Bakiu, R.; Santovito, G. Copper/zinc superoxide dismutase from the crocodile icefish Chionodraco hamatus: Antioxidant defense at constant sub-zero temperature. Antioxidants 2020, 9, 325. [Google Scholar] [CrossRef]
- Choi, E.M.; Suh, K.S.; Rhee, S.Y.; Oh, S.; Woo, J.-T.; Kim, S.W.; Kim, Y.S.; Pak, Y.K.; Chon, S. Perfluorooctanoic Acid Induces Mitochondrial Dysfunction in MC3T3-E1 Osteoblast Cells. J. Environ. Sci. Health A 2017, 52, 281–289. [Google Scholar] [CrossRef]
- Suh, K.S.; Choi, E.M.; Kim, Y.J.; Hong, S.M.; Park, S.Y.; Rhee, S.Y.; Oh, S.; Kim, S.W.; Pak, Y.K.; Choe, W.; et al. Perfluorooctanoic Acid Induces Oxidative Damage and Mitochondrial Dysfunction in Pancreatic β-Cells. Mol. Med. Rep. 2017, 15, 3871–3878. [Google Scholar] [CrossRef] [PubMed]
- Ku, T.; Zhou, M.; Hou, Y.; Xie, Y.; Li, G.; Sang, N. Tebuconazole Induces Liver Injury Coupled with ROS-Mediated Hepatic Metabolism Disorder. Ecotoxicol. Environ. Saf. 2021, 220, 112309. [Google Scholar] [CrossRef] [PubMed]
- Ploch, S.A.; Lee, Y.-P.; MacLean, E.; Di Giulio, R.T. Oxidative Stress in Liver of Brown Bullhead and Channel Catfish Following Exposure to Tert-Butyl Hydroperoxide. Aquat. Toxicol. 1999, 46, 231–240. [Google Scholar] [CrossRef]
- Motta, C.M.; Simoniello, P.; Di Lorenzo, M.; Migliaccio, V.; Panzuto, R.; Califano, E.; Santovito, G. Endocrine disrupting effects of copper and cadmium in the oocytes of the Antarctic Emerald rockcod Trematomus bernacchii. Chemosphere 2021, 268, 129282. [Google Scholar] [CrossRef] [PubMed]
- Bakiu, R.; Pacchini, S.; Piva, E.; Schumann, S.; Tolomeo, A.M.; Ferro, D.; Irato, P.; Santovito, G. Metallothionein Expression as a Physiological Response against Metal Toxicity in the Striped Rockcod Trematomus hansoni. Int. J. Mol. Sci. 2022, 23, 12799. [Google Scholar] [CrossRef]
- Du, J.; Cai, J.; Wang, S.; You, H. Oxidative Stress and Apotosis to Zebrafish (Danio rerio) Embryos Exposed to Perfluorooctane Sulfonate (PFOS) and ZnO Nanoparticles. Int. J. Occup. Med. Environ. Health 2017, 30, 213–229. [Google Scholar] [CrossRef]
- Lushchak, V.I.; Bagnyukova, T.V. Effects of Different Environmental Oxygen Levels on Free Radical Processes in Fish. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2006, 144, 283–289. [Google Scholar] [CrossRef]
- Santovito, G.; Piccinni, E.; Boldrin, F.; Irato, P. Comparative study on metal homeostasis and detoxification in two Antarctic teleosts. Comp. Biochem. Physiol. C 2012, 155, 580–586. [Google Scholar] [CrossRef]
- Sattin, G.; Bakiu, R.; Tolomeo, A.M.; Carraro, A.; Coppola, D.; Ferro, D.; Patarnello, T.; Santovito, G. Characterization and Expression of a New Cytoplasmic Glutathione Peroxidase 1 Gene in the Antarctic Fish Trematomus bernacchii. Hydrobiologia 2015, 761, 363–372. [Google Scholar] [CrossRef]
- Sattin, G.; Santovito, G.; Cassini, A. Physiological Antioxidant Responses against High Environmental Oxygen Concentration: Glutathione Peroxidase from the Antarctic Teleost Trematomus eulepidotus. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2008, 151, S27. [Google Scholar] [CrossRef]
- Fields, L.G.; DeVries, A.L. Variation in blood serum antifreeze activity of Antarctic Trematomus fishes across habitat temperature and depth. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2015, 185, 43–50. [Google Scholar] [CrossRef]
- Tolomeo, A.M.; Carraro, A.; Bakiu, R.; Toppo, S.; Garofalo, F.; Pellegrino, D.; Gerdol, M.; Ferro, D.; Place, S.P.; Santovito, G. Molecular characterization of novel mitochondrial peroxiredoxins from the Antarctic emerald rockcod and their gene expression in response to environmental warming. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2019, 255, 108580. [Google Scholar] [CrossRef]
- Lavut, A.; Raveh, D. Sequestration of Highly Expressed MRNAs in Cytoplasmic Granules, P-Bodies, and Stress Granules Enhances Cell Viability. PLoS Genet. 2012, 8, e1002527. [Google Scholar] [CrossRef]
- Olszewska, M.; Bujarski, J.J.; Kurpisz, M. P-Bodies and Their Functions during MRNA Cell Cycle: Mini-Review. Cell Biochem. Funct. 2012, 30, 177–182. [Google Scholar] [CrossRef]
- Drago, L.; Peronato, A.; Franchi, N.; Ballarin, L.; Bakiu, R.; Santovito, G. Stress granules in Ciona robusta: First evidences of TIA-1-related nucleolysin and tristetraprolin gene expression under metal exposure. Comp. Biochem. Physiol. C 2021, 243, 108977. [Google Scholar] [CrossRef]
- Nicorelli, E.; Gerdol, M.; Buonocore, F.; Pallavicini, A.; Scapigliati, G.; Guidolin, L.; Irato, P.; Corrà, F.; Santovito, G. First Evidence of T Cell Restricted Intracellular Antigen (TIA) Protein Gene Expression in Antarctic Fish. XIX Incontr. Sci. Della Soc. Ital. Di Immunobiol. Comp. E Dello Svilupp. Genova 2018, 15, 127. [Google Scholar]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pacchini, S.; Piva, E.; Schumann, S.; Irato, P.; Pellegrino, D.; Santovito, G. An Experimental Study on Antioxidant Enzyme Gene Expression in Trematomus newnesi (Boulenger, 1902) Experimentally Exposed to Perfluoro-Octanoic Acid. Antioxidants 2023, 12, 352. https://doi.org/10.3390/antiox12020352
Pacchini S, Piva E, Schumann S, Irato P, Pellegrino D, Santovito G. An Experimental Study on Antioxidant Enzyme Gene Expression in Trematomus newnesi (Boulenger, 1902) Experimentally Exposed to Perfluoro-Octanoic Acid. Antioxidants. 2023; 12(2):352. https://doi.org/10.3390/antiox12020352
Chicago/Turabian StylePacchini, Sara, Elisabetta Piva, Sophia Schumann, Paola Irato, Daniela Pellegrino, and Gianfranco Santovito. 2023. "An Experimental Study on Antioxidant Enzyme Gene Expression in Trematomus newnesi (Boulenger, 1902) Experimentally Exposed to Perfluoro-Octanoic Acid" Antioxidants 12, no. 2: 352. https://doi.org/10.3390/antiox12020352
APA StylePacchini, S., Piva, E., Schumann, S., Irato, P., Pellegrino, D., & Santovito, G. (2023). An Experimental Study on Antioxidant Enzyme Gene Expression in Trematomus newnesi (Boulenger, 1902) Experimentally Exposed to Perfluoro-Octanoic Acid. Antioxidants, 12(2), 352. https://doi.org/10.3390/antiox12020352








