Abstract
Plant bioactive compounds have gained global significance in terms of both medicinal and economic ramifications due to being easily accessible and are believed to be effective with fewer side effects. Growing relevant clinical and scientific evidence has become an important criterion for accepting traditional health claims of medicinal plants and also supports the traditional uses of Corchorus as folk medicine. C. capsularis and C. olitorius have broad applications ranging from textile to biocomposite, and young leaves and shoots are used as healthy vegetables and have long been used as traditional remedies for fever, ascites, algesia, liver disorders, piles, and tumors in many cultures. This review systematically summarized and emphasized the nutritional attributes, mostly available bioactive compounds, and biological and potential pharmaceutical properties of C. capsularis and C. olitorius, disclosed to users and non-users. Results suggest that various phytochemicals such as cardiac glycosides, phenols, flavonoids, sterols, lipids, and fatty acids were found or analytically identified in different plant parts (leaf, stem, seed, and root), and many of them are responsible for pharmacological properties and their antitumor, anticancer, antioxidant, antinociceptive, anti-inflammatory, analgesic, antipyretic, antiviral, antibacterial, anticonvulsant, antidiabetic and antiobesity, and cardiovascular properties help to prevent and cure many chronic diseases. In addition to their use in traditional food and medicine, their leaves have also been developed for skin care products, and some other possible uses are described. From this review, it is clear that the isolated compounds of both species have great potential to prevent and treat various diseases and be used as functional foods. In conclusion, this comprehensive review establishes a significant reference base for future research into various medical and functional food applications.
1. Introduction
Plants have provided nourishment and other health benefits to humans and animals for as long as they have existed. According to reports, around 80% of the world’s population relies on natural products and plant-based medicines as their primary source of medication [1], and about 25% of all medications are derived from 500 various herbal plant species [2]. People are becoming more aware of the side effects of synthetic drugs, as well as high cost, unavailability, inaccessibility, and unexpected loss of efficacy, so there is a growing interest in these traditional medicines [3]. The rising costs of health care and mainstream medicine’s failure to treat certain diseases have also resulted in the increased use of traditional medicine in disease prevention. So, the scientific community is searching to find an alternative source such as a plant to develop a more stable and efficient drug to treat various diseases.
Corchorous is a genus of annual herbs belonging to the Tiliaceae family that contains about 50–60 species, but only two species, C. olitorius and C. capsularis, are well-known for commercially bast fiber production and are distributed throughout the world’s tropics, subtropics, and warm-temperate regions [4,5]. The major jute-growing countries are India, Bangladesh, China, Thailand, and Nepal (Figure 1). In jute-producing countries, short and branched stems with mucilaginous leaves of these species are used as vegetables in foods to promote community [6,7]. In addition, the traditional uses of corchorus as folk and ayurvedic medicine to treat a variety of human diseases has been made evident, as more people recognize natural products are also effective, safe, non-narcotic, and affordable, without any kind of side effects [8].
Figure 1.
Jute-producing countries in the world.
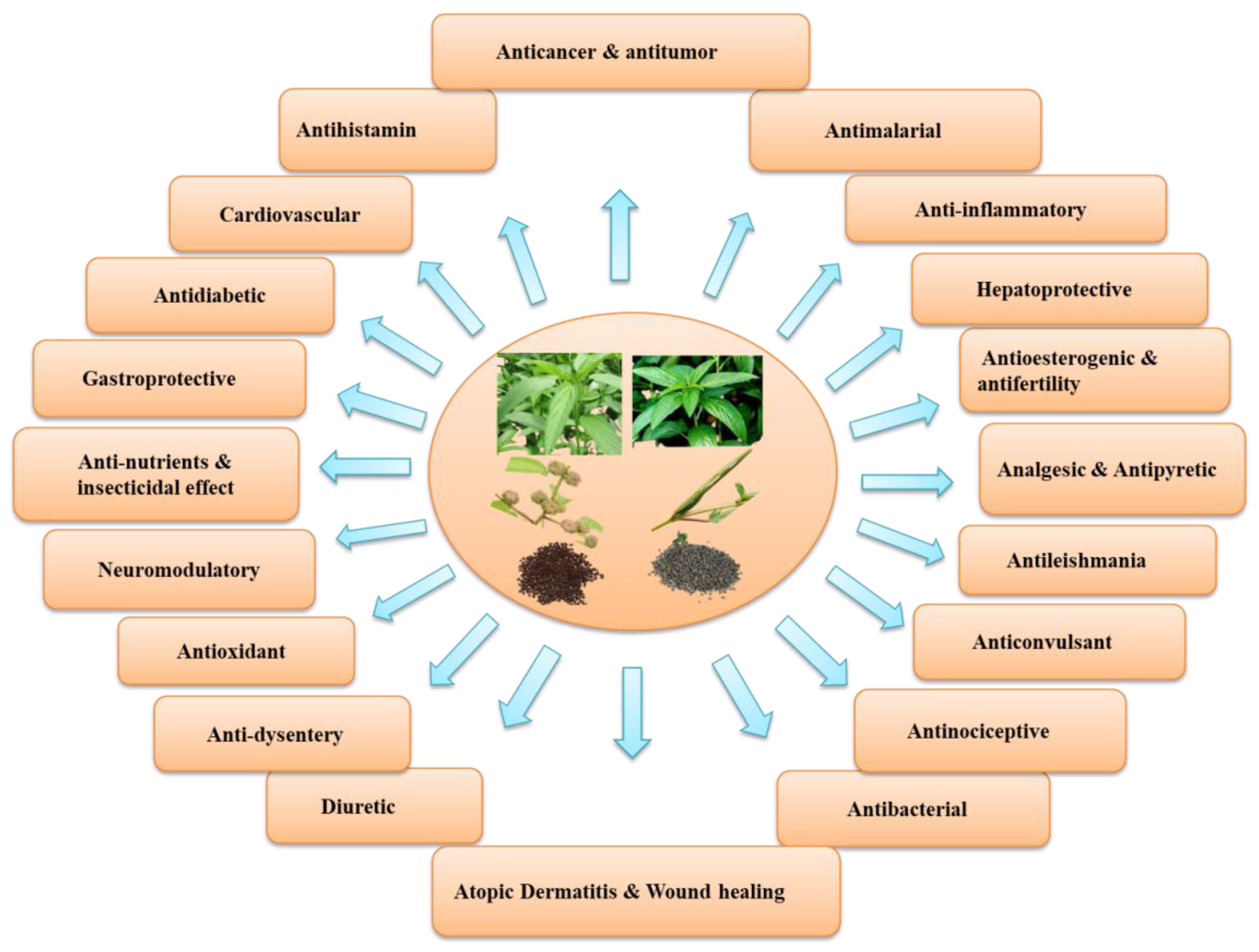
It has been reported that the leaves of edible species of Corchorus are an excellent source of protein, vitamins, and certain hormone precursors and are rich in minerals [9,10]. In addition to the micro and macronutrients, it contains a wide range of bioactive compounds such as glycosides, phenolics, flavonoids, tannins, saponins, sterols, triterpenoids, ionones, fatty acids, and carbohydrates, all of which have been reported from different the plant parts and seeds of two commonly cultivated species. These substances have powerful antipyretic, diuretic, analgesic, antioxidant, antimicrobial, and antitumor properties [11]. In addition, the leaves of C. capsularis and C. olitorius were reported to exhibit protective effects, mainly antitumor and anticancer promoting activity as well as antioxidant, anti-inflammatory, antitumor, antifungal, gastroprotective, antinociceptive, analgesic, and antibacterial activities [12,13,14] (Figure 2). Moreover, both species inhibit α-glucosidase and α-amylase activities [15] and are linked to a reduction in oxidative stress and an increase in β-oxidation in the liver [16].
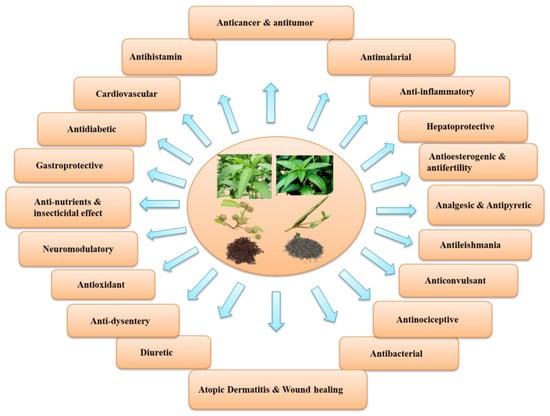
Figure 2.
An overview of various biological, pharmacological, and medicinal attributes of C. capsularis and C. olitorius.
Despite therapeutic and prophylactic uses, little is known about Corchorus leaf-derived active principles, their biological roles, and toxicological limits. Several previously published reports have provided fragmentary summaries of morphological, phytochemical, and ethnomedicinal properties of C. capsularis and C. olitorius in general. Such information is crucial for achieving the potential commercialization of leaves as well as different parts of both species. However, a systematic review of the literature that is organized, comprehensive, and targeted has not been conducted to assess the complete attributes of the nutritional, bioactive compounds and pharmaceutical properties of C. capsularis and C. olitorius. Therefore, this article seeks to fill this gap by presenting a comprehensive and up-to-date overview of the nutritional and phytochemical profiles, and biological as well as pharmacological features, of both species. For this, an unbiased literature search was conducted using several well-known scientific search engines with comprehensive coverage for a literature search, including Google, Google Scholar, ResearchGate, PubMed, Web of Science, and Scopus using different combinations of the following: C. capsularis, C. olitorius, nutrient, phytochemistry, ethnomedicine, health effects, biological activities, pharmacological activities, in vitro, and in vivo. The authors did not attempt to contact the investigators or identify any unpublished data. A detailed bibliographic search was carried out, and information was examined, critically analyzed, and collated systematically to ensure the reproducibility of the literature review process. This review aims to take advantage of the key findings concerning phytoconstituents, and emphasizes their therapeutic potential based on pharmacognosy, traditional medicine, and experimental knowledge. Another aim is to identify the information gaps and provide a valuable guide for future scientific research and the pharmaceutical industry as well as contributions to the field.
2. Use in Local and Traditional Food and Medicine
Corchorus spp., especially the leaves of C. olitorius and C. capsularis, are used as leafy vegetables in many Asian, African, and European countries [4,17,18,19] (Table 1). It is simple to prepare and may be incorporated in a variety of dishes to enable people to take advantage of its vitamins and minerals. Due to their mucilaginous texture, the leaves have a viscous consistency and are widely consumed as soup in Middle Eastern countries. The leaves are used as a condiment in Bengal, where it is considered a tonic, and frequently incorporated into rice for the daily diet. Dried leaves are used in herbal tea, while seeds are used as a flavoring agents [20].

Table 1.
Application of different parts as food and its ethnobotanical uses [8,18,28].
The dried leaves are a favorite of the Boros of northeast India, who make narji, a mucilaginous preparation made with fatty pork and lye. Most often, it is lightly sauteed and eaten with rice or rice gruel in Sambalpur and the western part of Odisha in summer. The leaves of C. olitorius are commonly consumed together with bamboo shoots in the Philippines, whereas in Thailand they are eaten with plain rice congee and tastes like spinach and samphire [8,21]. In Vietnamese cuisine, it is known as rau đay and is made into a soup with shrimp. The Luhya people of Western Kenya eat jute leaves with starchy foods such as ugali, a staple food for most Kenyan communities. Nigerian cuisine is usually used as a condiment for other starchy foods such as amala, cassava, yam or millet as well as used in ethno-medicine to facilitate labor and smooth child delivery, especially in the Yoruba people [22]. In addition, some tribes of Nigeria used leaf extract to treat a menstrual disorder that is associated with excessive uterine contractions during menstruation [23] and to arrest threatened miscarriage and induce tocolysis [24]. Moreover, its leaf decoctions are used to treat iron deficiency and folic acid deficiency and in the treatment of anemia. In many West African cooking traditions, the leaves are used as an ingredient to make a traditional slimy soup or sauce. It has been recognized and popular as an Egyptian national dish in Egypt. In Turkey and Cyprus, it is typically cooked into a kind of chicken stew and flavored with lemon and olive oil [25]. Mallow leaf stews with rice are a known cuisine of the Middle East [8,26]. Recently, molokhia leaves were also used to develop sushi wraps as a promising viable substitute for Nori [27,28]. The young leaves of C. capsularis are the most edible part, mainly consumed in a salad or cooked as a pot herb, as they have been in many parts of the world for a long time. In Japan, leaves of both species were regarded as healthy food, and young dried leaves are considered an alternative to coffee or tea and a thickener in soups [29,30].
Consumption of the C. olitorius and C. capsularis leaves are reported to be a diuretic, demulcent, deobstruent, purgative, refrigerant, carminative, bitter tonic, lactagogue, and blood purifier; the leaf twigs are used for cardiac problems and the stems for treating cardiovascular disorder [31]. The seeds are used as a laxative and the roots for treating toothache, and a decoction of the roots is used as a tonic to increase strength. In Tanzania, leaf infusions are used as a tonic, appetite stimulant, and to treat constipation [32,33]. Both of these plants’ leaves are used in ethnomedicine to treat aches and pains, swellings, cystitis, dysuria, gastroenteritis, dysentery, malaria, enteritis, fever, gonorrhea, diabetes, dyspepsia, liver disorders, pectoral pains, and tumors [29,30,33,34,35,36]. C. olitorius is one of the plants used in folk medicine to treat measles [37]. Furthermore, it is also a component in facial moisturizers, lotions, hair tonics, and hand creams [38].
3. Nutritional Composition of Jute
Edible species of Corchorus, especially C. olitorius and C. capsularis, are used as a vegetable in jute growing as excellent sources of proteins, dietary fiber and carotene, and vitamins (A, C, E), and they are also rich in mineral nutrients such as K, Ca, P, and Fe [9,10] (Table 2). In addition to this, C. olitorius content has a high amount of iron, folate, amino acids, and essential minerals [39], which are essential for preventing anemia [9]. It has been reported that C. olitorius leaves contain a significant amount of 17 macro and micronutrients, including carbohydrates, protein, fat, fiber, calcium, phosphorous, potassium, ascorbic acid, beta-carotene, riboflavin, thiamine, niacin, etc [8]. Regarding the micronutrients, the leaves of both species had a high concentration of Fe and Se compared with other leafy vegetables, whereas Zn and Se content varied from 0.91–7.35 mg/100 DW and 85.51–168.85 μg/100 g DW, respectively [40]. This variation may be the genetic variations among the cultivars and environmental growing conditions. A number of studies reported that C. olitorious and C. capsularis were found to be crude protein (14%, 3.86%), potassium (44.73 mg, 40.43 mg), and beta-carotene (8.13 mg, 6.13 mg) per 100 g, respectively [8,41]. It was stated that beta-carotene is a group of phytochemicals that are liable for different colors of the fruits and vegetables, playing a vital role in the prevention of mortal disease and that C. olitorius’ content is higher than other Corchorus spp. as well as other leafy vegetables [42]. It was found that 100 g of C. capsularis leaves contained 80.4–84.1 g H2O, 43–58 calories, 4.5–5.6 g protein, 7.6–12.4 g carbohydrate, 0.3 g fat, 1.7–2.0 g fiber, 2.4 g ash, 266–366 mg Ca, 97–122 mg P, 7.2–7.7 mg Fe, 12 mg Na, 444 mg K, 6.41–7.85 mg beta-carotene equivalent, 53–80 mg ascorbic acid, 0.26–0.53 mg riboflavin, 0.13–0.15 mg thiamine, and 1.1–1.2 mg niacin and folic acid content was considerably higher compared with other folacin-rich vegetables [43]. It was reported that jute leaves as saluyot (boiled/100 g) contain 43–58 of food energy (cal), 4.5–5.6 g of protein, 1.7–2.0 g of fiber, 2.4 g of ash, 7.6–12.4 g of total carbohydrates, 97–122 mg of phosphorous, 11.6 mg of iron, 12 mg of sodium, 444 mg of potassium, 6390 of Vit-A (I.U), 15 mg of thiamine, 28 mg of riboflabin, 1.5 mg of niacin, and 95 mg of ascorbic acid [8]. In addition, it was reported that both the raw and cooked fruit of C. olitorius composed appreciable amounts of protein, fat, fibre, ash, carbohydrate, and energy [44]. It stated that the boiled leaf saluyot of C. olitorius contains higher ascorbate than fresh leaves. A group of researchers identifies the leaves contained ascorbic acid (258 mg/100 g) and α-tocopherol (14 mg/100 g) [45]. The findings suggest that ascorbic acid presence in leaves might greatly contribute to the antioxidant activity and, regularly consumed, may provide enough to meet the recommended daily allowance of 60 mg for adults [46].

Table 2.
Nutritional comparison of C. capsularis and C. olitorius mg per 100 g [8,40,46,47].
4. Bioactive Compounds in Jute
4.1. Polyhenols
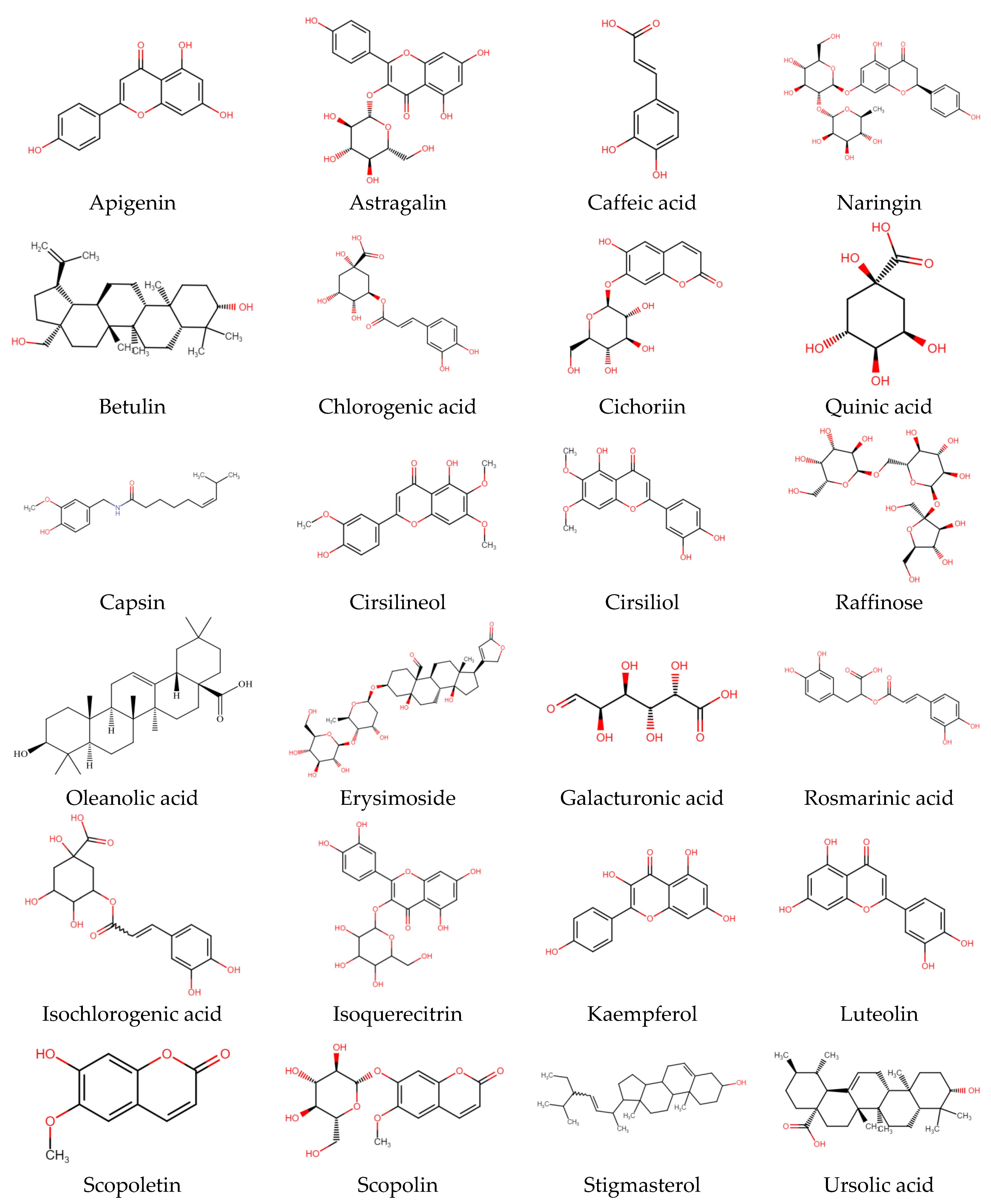
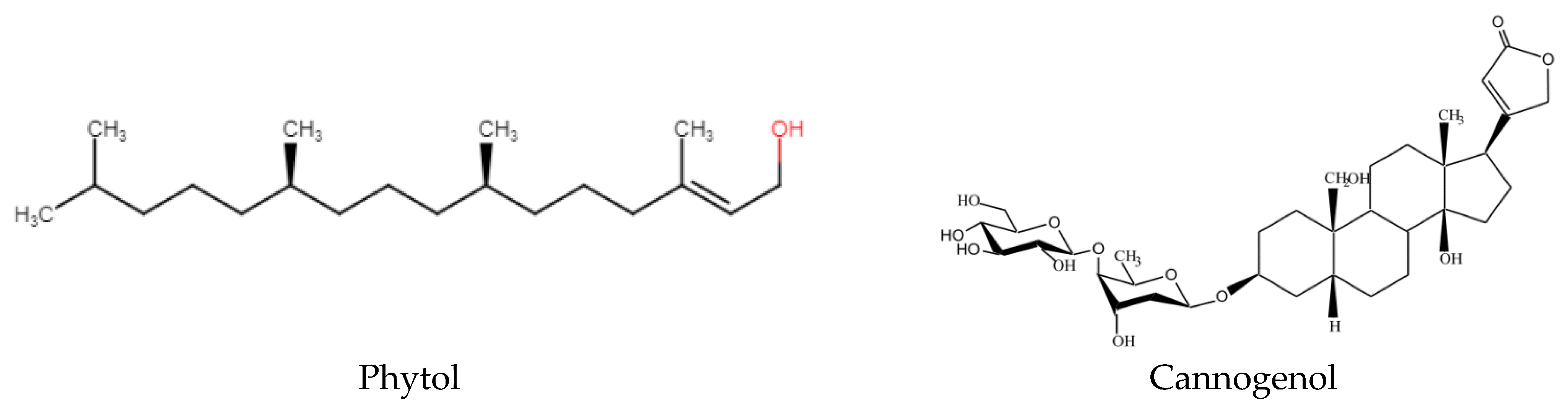
Phenolic compounds are a large class of plant secondary metabolites that are important for the quality of plant-based foods, and along with other dietary reducing agents, are regarded as antioxidants. They protect the tissues of the body from oxidative stress-related pathologies such as as cancers, inflammation, and coronary heart disease. Phenolic acids are cyanidin and cyanidin glucosides from the bark and leaf of C. capsularis whereas astragalin, tolifolin, isoquercitin, and jugulanin come from C. olitorius leaves, as reported by researchers [47,48]. From the leaves of C. olitorius, a number phenolic compounds such as chlorogenic acid (383.9 mg/100 g FW), 3,5-dicaffeoylquinic acid (102.1 mg/100 g), quinic acid, gallic acid, protocatechuic acid, 4-O-caffeoylquinic acid, caffeic acid, 1,3-di-O-caffeoylquinic acid, feruloyl-quinic acids, and 4,5-di-O-caffeoylquinic acid were identified [47,49]. According to the literature, chlorogenic acid may be the most predominant antioxidant [45,47,50], whereas caffeoylquinic acids and dicaffeoylquinic acids have been reported to inhibit human low-density lipoprotein oxidation in vitro; there are limited studies investigating the effects in vivo [51,52]. Total phenols (9.20 mg GAE/g DW) were reported in C. olitorius from ethanolic acid, and recently new phenolic compounds such as trans-ferulic acid, p-coumaric acid, and rosmarinic acid were detected from the C. olitorius [41,53]. In a separate study, it was recorded that the most abundant polyphenolic compound was caffeic acid, trans-ferulic acid, rutin hydrate, ellagic acid, and quercetin hydrate in the leaves of C. capsularis and C. olitorius [54]. Most common bioactive compounds available in C. capsularis and C. olitorius and their chemical structure are illustrated in Figure 3, and isolated compounds from different plant parts with extraction technology are presented in Table 3.
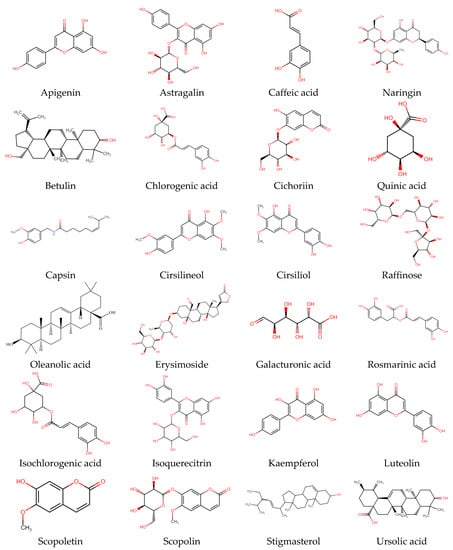
Figure 3.
Chemical structures of the most common bioactive compound available in C. capsularis and C. olitorius.

Table 3.
Chemicals isolated from different parts of C. capsularis and C. olitorius.
4.2. Flavanoids
Flavonoids are biologically important compounds with high antioxidant properties that protect cells from free-radical damage caused by reactive oxygen species (ROS) and have antioxidant effects connected to several diseases, including cancer, atherosclerosis, and Alzheimer’s disease (AD) [55,56]. The leaves of C. olitorius contain flavanones (naringenin, naringin), flavones (cirsilineol and cirsiliol), flavones glycosides (apigenin, apegenin-7-O-glucoside), tolifolin (kaempferol-3-O-β-D-galactopyranoside), astragalin (kaempferol-3-O-β-D-glucopyranoside), and jugulanin (kaempferol- 3-O-β-L-arabinopyranoside) [47,53]. A number of researchers have reported that flavonoid glycosides, flavonol glucosides astragalin, quercetrin, isoquercitin, quercetin 3-galactoside, quercetin 3 glucoside, quercetin 3-(6 malonylglucoside) and quercetin 3-(6 malonylgalactoside), quercetin 3-O-galactoside (hyperoside), and catechin were identified from the leaves of C. olitorius [49,50,53,56,57]. The content of quercetin glycosides in C. olitorius leaves was 233 mg/100 g of FW, almost twice that of onion, another significant source of quercetin glycosides [52]. Authors have isolated scopoletin and fusidic acid from C. capsularis [58], and ethanolic extract of C. olitorius leaves produced the highest flavonoids content (4.10 mg QE/g) [53], which was lower than mucilaginous polysaccharides (PSc) extracts (6 mg QE/g) conducted by Yakoub et al. (2020) [41].
4.3. Cardiac Glycosides and Their Aglycones
Cardiac glycosides are a class of organic compounds that enhance the output force and rate of contraction of the heart by inhibiting the Na+/K+ ATPases in cardiac myocytes and are employed in heart failure and arrhythmias treatment. Several glycosidic compounds such as capsularin, chorchoritin, and corchularin have been isolated from different Corchorus spp. as well as a variety of algycones, including corchsugenin, corchortoxin, and corchorgenin [48]. The chemical identification of these aglycones as strophanthidin 1, the well-known aglycone of the cardiac glycoside strophanthin, was a significant advancement. From C. capsularis and C. olitorius, two digitalis glycosides have been derived, such as corchoroside A2 (mp 188–190⁰, [α]D20 = +11° (MeOH)) and corchoroside B1520 (mp 222–24°, [α]D20 = +68°), respectively [48]. Researchers also identified and confirmed the presence of a monoglucoside of corchoroside A in C. olitorius seeds, as well as a diglucoside and a triglucoside of corchoroside A [29,48]. After enzymatic hydrolysis, the seed extract of C. capsularis and C. olitorius gave a fair yield of corchoroside A. In addition to erysimoside in the seeds of C. olitorius, there is the presence of cardenolide glycoside, trioside of strophantidin, which has a similar structure to the polar glycoside of C. capsularis. A rare cardiac glycoside such as corchoroside-A and cannogenol is found in the roots extract of C. capsularis [58]. Among the numerous components of Vietnamese C. olitorius leaves, researchers first detected and identified four ionone glucosides called corchoionosides A, B, and C, (6S,9R)-reseoside, a monoterpene glucoside betulalbuside A, two flavonol glucosides isoquercitin and astragalin, and two coumarin glucosides, cichoriine and scopolin [36], whereas new coumarin 4,7-dihydroxycoumarin had been isolated from the chloroform extract of defatted seeds of C. olitorius [47,59]. C. capsularis leaves resulted in glycosides, capsulason, corchorol, capsularol, and a few sugar fragments, namely glucose, galactose, and arabinose-free sugars when treated with 4% KCl. Acid hydrolysis of capsularol produces glucose and an aglycone, capsularogenin [48]. A polar glycoside was obtained from the seeds of C. capsularis, and energetic hydrolysis resulted in glucose, while mild acid hydrolysis yielded aglycon strophanthidine, whereas controlled enzymic hydrolysis with β-glucosidase formulated olitoribose, glucose, and boivinose, implying that the sugar residue is gluco-olitoribose [48].
It was stated that cardenolide glycosides such as olitoriside, erysimoside, corchoroside A, and coroloside were isolated from the seeds of C. olitorius; helveticoside was also isolated from autofermented seeds, and deglucocoroloside was identified as an artifact through hydrolysis of the extract of seed [60]. Three new cardenolide glycosides were discovered by a group of researchers, and their structures were established based on chemical and spectroscopic evidence as cannogenol 3-O-β-D glucopyranosyl-(1→4)-O-β-D-boivinopyranoside, periplogenin 3-O-β-D-glucopyranosyl-(1→4)-O-β-D digitoxopyranoside, and digitoxigenin 3-O-β-D-glucopyranosyl-(1→6)-O-β-D-glucopyranosyl-(1→4)-O-β-D-digitoxopyranoside, as well as some new cardiotonic oligoglycosides [60]. It has been stated that dark grayish-green seeds contain a greater concentration of cardiac glucoside than dark greyish-green or yellowish-green seeds [60]. Digitoxigenin glycosides, coroloside, and glucoevatromonoside, as well as strophanthidin glycosides (erysimoside, olitoriside, biosides, trioside, corchoroside A, and helveticoside), were isolated from the methanolic extract of seeds, which were the major cardiac glycosides [60,61].
Polar glycosides A and B were produced by chloroform–butanol (1:3) fractions of C. capsularis seeds, while glycoside B and a new polar glycoside C were produced by chloroform–alcohol (2:1) extract. One study on C. olitorius seeds isolated and identified strophanthidin, strophanthidol, corchoroside A, helveticoside, and olitorin [48].
4.4. Triterpenoids
Triterpenoids are compounds with a carbon skeleton based on six isoprene and promising agents for the prevention of diabetic complications as well as hepatoprotective, cardiotonic, sedative, and tonic effects. Corosic acid (C30H44O6) was yielded when triterpenoid corosin isolated from C. capsularis root was refluxed with HCl [62]. A group of researchers, isolated urosolic acid, corosolic acid, betulinic acid, and oxo-corosin in fresh, undried roots of C. capsularis and C. olitorius [58,62]. Capsugenin (30-O-glucopyranoside), one more triterpine glucoside, was isolated from the mature leaves [63,64]. It also was reported that Egyptian origin C. olitorius leaves have been found to contain oleanolic acid [65]. A new dammarane triterpine glycoside, a capsin bitter-tasting substance of the leaves was identified as the 3-glucoside of 20,24-epoxy-3β,12β,25, 30-tetrahydroxydammarane (capsugenin). Later, another new triterpene glucoside, capsugenin 30-O-glucopyranoside, was confined from the leaves of C. capsularis [64,66,67]. The roots of both C. capsularis and C. olitorius were reported to contain three ursane triterpenes: corrosion, corosolic acid, and ursolic acid.
Both C. capsularis and C. olitorius roots have been reported to possess three ursane triterpenes: corrosion, corosolic acid, and ursolic acid [66]. Ramadevi (2013) stated that terpenoids such as betulinic acid and oleanolic acid were found in the roots of C. capsularis [58].
4.5. Organic Acid/Essential Oils
Chemical compositions of the essential oils of C. olitorius and C. capsularis leaves resulted in the identification of 11 and 21 compounds, with total concentrations of 24.7 and 62.9%, respectively, while 29 and 19 compounds were still unidentified for both species. It was also observed that edrane-5-one (17.7%), followed by methyl tiglate (2.73%) and trans-phytol (0.99%) of C. olitorius and γ-terpinene (12.1%), followed by carvacrol methyl ether (10.01%) of C. capsularis, represent each plant’s major compounds, respectively [65]. In other research, it was stated that 43 components were identified from C. olitorius leaves and flowers, the most abundant of which were benzaldehyde (56%), methyl 4-methoxysalicylate (6.55%), and carvacrol (4.75%) [68].
4.6. Fatty Acid
Fatty acids are ubiquitous to all living organisms, and plants synthesize a huge variety. Due to emulsification properties, it can serve as a thickening agent in cosmetic industries as well as food. It has been confirmed that the leaves of C. olitorius yield four higher fatty acids with a trienone function, corchorifatty acid A, B, C, and D, an undecanoic acid, a trihydroxy fatty acid, and corchorifatty acid E and F [36,69]. It was reported that there is the presence of glyceryl monopalmitate in the leaves of Egyptian C. olitorius [65], where β-sitosteryl fatty acid is isolated from the stem [45]. Yousef investigated the dry oil of the leaves and stem of C. olitorius by GC-MS and claimed both were high in hydrocarbons and fatty acids, but the leaves contained a higher concentration of hexadecanoic acid (28.52%) and 2,4-di tert-butyl phenol (15.01%) as a main component, whereas stem dry oil revealed ethyl palmitate (26.19%), 2,4-di tert-butyl phenol (14.35%), 1-eicosanol (8.27%), tetratetracontane (7.02%), 8-heptadecene (5.33%), and hexatriacontane (5.16%) in high quantities [70].
The author stated that fatty acid methyl esters of the leaves of C. olitorius are rich in ω3-octadecatriene fatty acid or α-linolenic acid (49%) [71], where seeds contained linoleic acid, linolenic acid, oleic acid, palmitic acid, stearic acid, and behenic acid. Linoleic acid, one of the two essential fatty acids for human health, is required for the immune system to function properly and help regulate blood pressure. A group of research and epidemiological studies have established the essentiality of ω-3 fatty acids for the normal development of premature infants’ retina and brain, as well as hypotriglyceridemic, anti-inflammatory, and antithrombotic properties [72,73]. The total fatty acid content (57.26–125.1 mg/100 g wet weight) in the leaf development stages of C. olitorius were found to be the highest 50 days after being sown [31]. In newer research, different parts of C. olitorius contained six fatty acids, namely palmitic acid, linoleic acid, heptadecanoic acid, oleic acid, steric acid, and arachidic acid. Among these, stearic acid represented the major compound in the case of the seeds (49.48%), followed by the roots (68.80%) and the leaves (58.67%), whereas palmitic acid (59.94%) was the major compound in the stems [65]. Previous research has demonstrated that seed fats of both species contain a number of fatty acids, where linoleic acid was more prominent, and C. capsularis seed fat is therefore, to some extent, similar to cotton seed oil, whereas in some respects, C. olitorius is more like sunflower-seed oil [74].
In another research, it was reported that the total fatty acids of C. capsularis were found to be 0.02 and 0.03% (DW) in the seeds and the vegetative parts, respectively, whereas palmitic acid was represented the most abundantly in the seeds (81.68%) and the vegetative part (54.10%) [75]. In addition, both the seeds and vegetative parts contained 87.70 and 79.62% saturated fatty acids, and 12.30 and 22.38% unsaturated fatty acids.
4.7. Lipid Composition
Lipids are the basic unit of membranes and our bodies’ energy source, as small hydrophobic molecules are excellent chemical messengers between organelles and cells. A group of researchers found a total lipid content ranging from (16.75–23.10 g/100 g) and lipid-like TAG (Triacylglycerol), DAG (Diacylglycerol), FFA (Free fatty acids), and PTS (Phytosterol) was determined from Thai jute cultivars’ seed oil using Iatroscan (TLC/FID), where four types of lipids significantly differed. This variation is caused by inherent differences between varieties and/or the environment in which the seed grew. A wide range of DAG concentrations (9.49%) was observed in jute seed oil, where the highest DAG was similar to that in cottonseed oil [72]. It was reported that lipids in diacylglycerol (DAG) form were found to significantly lower serum triacylglycerol levels [76,77,78]. To explain the mechanism, researchers propose that dietary DAG hypotriacylglycerolemia is caused by decreased intestinal re-esterification and chylomicron assembly or decreased chylomicron circulation secretion [78].
Triacylglycerol was the most abundant lipid in jute seed oil, accounting for 70% to 74% of the total, with phytosterol (12% to 28%) and diacylglycerol (0% to 9%) as minor components. Linoleic acid (18:2n-6), which accounts for 40–67% of total fatty acid, was the most abundant polyunsaturated fatty acid (PUFA), α-linolenic acid (18:3n-3). The findings indicated that jute seed oil could be a source of edible PUFAs [72].
4.8. Phytosterols and Hydrocarbons
Sterols are a subclass of steroids with the formula C17H28O that contain a hydroxyl group at the 3-position of A-ring. Hasan and Kadhim (2018) found total sterol content was 1.8, 1.6, 1.4, and 0.6% (DW) in the seeds, leaves, stems, and root petroleum ether extract of C. olitorius and 0.05 and 0.12 (DW) in the seeds and vegetative parts of C. capsularis, respectively [75], which was higher than earlier research [79]. In a recent study, β-sitosterol, a novel antileishmanial agent, was isolated and fully characterized based on spectroscopic analysis (FTIR, H NMR, C NMR, and GC–MS) from the chloroform extract of C. capsularis [80]. It has been reported that steroids such as β-sitosterol and β-sitosterol-D-glucoside have been isolated from the leaves and roots of both species, with β-sitosterol 3-O-β-D-glucoside from the Egyptian origin C. olitorius leaves [65] and the roots of C. capsularis [48,81].
Estimation of hydrocarbon contents showed that octadecane is the major compound in the roots (55.54%), followed by the stems (13.81%) and leaves (5.12%), and a large quantity of tetradecane was recorded in the seeds (33.58%), followed by the roots (22.79%). The investigated parts of C. capsularis found that the seeds are composed mainly of cholesterol (28.52%), whereas the vegetative part is composed of cholesterol (12.46%), campasterol (24.98%), stigmasterol (4.38%), and β-sitosterol (10.39%). Hydrocarbon contents of the examined parts of C. capsularis showed that octadecane is the major compound in the seeds (45.13%) and heptadecane was only present in the vegetative part [65].
Several researchers investigated unsaponifiable fractions by GLC and showed that the seeds and roots contain mainly cholesterol and the triterpenoid compound β-amyrin, where cholesterol, β-sitosterol, and β-amyrin were detected from the leaves. It was noticed that cholesterol represents the major compound in the leaves (19.28%), whereas β-amyrin represents the major compound in (23.18%) the stems. β-Sitosterol is present in stems (8.39%) and leaves (7.62%), in contrast, stigmasterol is only in the stems (9.37%) [65].
4.9. Polysaccharides and Other Sugars
Polysaccharide has been reported from the mucilage extract of dried leaf of C. olitorious; it is rich in uronic acid (65%) and contains glucose, rhamnose, galacturonic acid, and gluconic acid in a molar ratio of 1.0:0.2:0.2:0.9:1.7, as well as the acetyle group [20]. In the leaves of C. olitorius, monosaccharide analysis revealed that galactose was predominant, followed by rhamnose, glucose, arabinose, and mannose, with the least amount of glucuronic acid [41]. Compared with the latest results, galactose, arabinose, and mannose were not detected in earlier research. The seed of C. capsularis and C. olitorius comprised free sugars, glucose, sucrose, fructose, raffinose, arabinose, and galactose, while the root extract consisted of glucose and fructose arabinose and raffinose [62], while fructose and galactose were also identified in the leaves and bark. This polysaccharide promotes the proliferation of murine splenocytes. The leaves of C. capsularis and C. olitorius contain two active components: monogalactosyldiacylglycerol (1,2-di-O-α-linolenoyl-3-O-β-D-galactopyranosyl-sn-glycerol) and phytol (3,7,11,15-tetramethyl-2-hexadecen-1-ol) [82], and concentration increased with hot water treatment.
4.10. Volatile Compounds
It was stated that a total of 45 and 49 components were identified in natural fresh leaves and cupric chloride-treated leaves of C. olitorius by GC-MS analysis, respectively. In fresh leaves, cis-3-Hexen-1-ol, cis-4-hexen-1-ol, terpinolene, sabinene, and phytol were the major compounds, whereas the major compounds found in treated leaves were cis-4-hexen-1-ol, cis-3-hexen-1-ol, tetradecanal, and phytol [83]. In a another study, five compounds were identified as scopoletin, isopimpinellin, xanthotoxol, fraxinol, and peucedanol and showed a strong antimicrobial activity [84].
5. Biological Activity
5.1. Antitumor and Anticancer Promoting Activity
Recent epidemiological studies have found that consuming a lot of plant leaves and fruits may be linked to a lower risk of cancer, particularly in the gastrointestinal tract. Two active components named phytol (a side chain of chlorophylls) and mono-galactosyldiacylglycerol have been identified in the leaves of C. capsularis and C. olitorius [82]. Reportedly, phytol and mono-galactosyldiacylglycerol at concentrations 15 g/mL and 30 g/mL completely inhibited the induction of EBV early antigen, respectively. Earlier, it was stated that phytol’s cytotoxicity on cancer cells was caused by apoptosis induction, whereas mono-galactosyldiacylglycerol inhibits the mammalian DNA polymerases activities, including DNA polymerase β related to repair, and made a strong apoptosis in gastric cancer cells. In a study by Li et al. (2012), human hepatocellular carcinoma (HepG2) cells treated with C. olitorius ethanol extract (ECO) at 12.5 µg/mL concentration demonstrated a significant reduction in cell viability, and by increasing caspase-9 activity and cytochrome c leakage from mitochondria with reduced membrane potential indicated triggering apoptosis, while normal FL83B hepatocytes were unaffected. Thence, apoptosis in ECO-treated HepG2 cells through caspase-dependent mitochondrial pathway may be induced partially by phytol and mono-galactosyldiacylglycerol [14]. In the evaluation of nutraceutical properties, polyphenol-enriched extracts (PEEs) of C. olitorius showed cytotoxicity against the colorectal Caco-2 cell line by triggering oxidative stress without disruption of the healthy CCD841 line and inhibition of the activity of glutathione-independent antioxidant enzymes [49]. Therefore, C. olitorius is suggested as a foodstuff plant with the potential to develop chemopreventive agents for human cancers.
In research, the cytotoxic inhibitory potential of C. olitorius seed extract and the genotoxic potential of the leaf, as well seed extract, were displayed on human multiple myeloma-derived ARH-77 cells in a dose-dependent manner [91]. This finding is supported by Newman et al. (2003), who reported that medicinal plants can exert a significant anticarcinogenic effect [92]. Similarly, the potential protective effects was observed against the foodborne mycotoxins of AFB1 and/or FB1 in H4IIE-luc rat hepatoma cells, where C. olitorius aqueous extract treated cells were statistically more viable cells and had less DNA damage in H4IIE-Luc cells exposed to mycotoxins than not treated with plant extract. This might be due to the abundant presence of flavonoid compounds and the antioxidant capacity [93]. Therefore, the result suggests that the aqueous extract of C. olitorius possesses cancer chemo-preventative agents.
In a newer research, it was recorded that chlorogenic acid (CGA) and isoquercetin (IQ) derived from the aqueous extract of C. olitorius significantly inhibited proliferation in vitro of human melanoma (A-375), gastric cancer (AGS), and pancreatic cancer (SUIT-2) cells and suppressed the growth of A-375 and AGS tumor xenografts in a CAM model by inducing apoptosis and antiangiogenetic properties [94]. Soykut et al. (2018), found that aqueous extract of C. olitorius leaf effectively inhibits both primary (Colo-320) and metastatic (Colo-741) colon adenocarcinoma cells at 50 µg/mL concentration in the case of in vitro anticancer and apoptotic induction by increasing caspase-3, cytochromec, and FasL immunoreactivity significantly in Colo-741 cells, indicating activation of extrinsic and intrinsic apoptotic pathways and being more effective in metastatic colon adenocarcinoma cell lines [12]. These apoptopic induction properties are due to the presence of natural anticancers such as quercetin, isoquercetin, and chlorogenic acid in the extract. Quercetin induction of apoptosis by increasing intracellular ROS, as well as increasing Bax protein expression and inhibiting Bcl-2 in HCT-116 via the intrinsic pathway, has been reported. Chlorogenic acid had cytotoxic effects in HT-29 colon cancer cells by increased caspase-3, while decreasing Bcl-2 activity and suppressing heat shock protein 70. As a result, tumor growth was inhibited [95]. Thus, the results indicate that extracts may have anticancer properties and may be used as a guide for treating colon cancer with phytomedicines rather than chemotherapy. Researchers have established the cytotoxicity of two identified compounds named Methyl-1,4,5-tri-O-caffeoyl quinate and trans-3-(4-Hydroxy-3-methoxyphenyl) acrylic anhydride (a popular chemo-preventive curcumin in cancer therapy) against HeLa, HL460 lung cancer cell line, and PC3 prostate cancer cell line [13]. When tested at concentrations up to 1.6 mM, the compounds were found to have mild cytotoxic activity against HeLa cells at ≥800 mM. A study determined the cytotoxicity of the different extracts from C. capsularis leaves by Brine shrimp lethality bioassay; the result reveal that butanol extract showed the most potent 71.14% inhibition at a concentration of 1.25 mg/mL, followed by ethyl acetate and methanol with inhibitions of 28.57% and 14.28%, respectively [96]. Based on the above discussion, it can be concluded that C. olitorius may be a source of naturally occurring “lead” compounds with antitumor activity. A summary of the biological activities of C. capsularis and C. olitorius are presented in Table 4.
5.2. Gastroprotective Effect
A peptic ulcer is an ulcer of one of the areas of the gastrointestinal tract that occurs as a result of excess acid, a breakdown in mucosal defenses, and peptic activity in gastric juice [97]. The results obtained by researchers point out that an extract of C. olitorius leaf demonstrated potential antiulcer activity in rats by decreasing acid secretion, improving mucosal defense, and increasing in vivo antioxidant status [98], where it was found that mucus productions increased dose-dependently. The highest dose (400 mg/kg) of plant extract resulted in mucus production increase up to 162.90 mg and a highly significant reduction in ulcers, with a corresponding curing rate of 94.08% by increasing the antioxidant enzymes activities. These findings indicating C. olitorius may accelerate ulcer healing by boosting angiogenesis, cell proliferation, migration, and maturation of the granulation tissue. A similar finding was also stated by a group of researcher, where the extract’s ability to inhibit gastric acid secretion and lesser gastric ulcer areas was found against pylorus ligation-induced gastric lesions [99,100]. Results also show that oral extract administration reduces the ulcer indices dose-dependently substantially, with significant increases in the production of gastric mucosa in rats. The decreased gastric acidity indicates that the extract’s cytoprotective mechanism, similar to that of endogenous prostaglandins, may involve either direct inhibition of gastric secretion or simple neutralization of the acid secreted by parietal cells, as well as reinforcement of the in vivo antioxidant status by the increase in SOD and a decrease in MDA. In addition to histological analysis, immunohistochemistry confirmed the protection-mediated up-regulation of Hsp70 and the down-regulation of Bax proteins. In conclusion, C. olitorius reduces gastric motility by reducing the area of the gastric wall of ulcerated areas and decreasing the infiltration of edema and leukocyte into submucosal layers.
5.3. Antinociceptive, Anti-Inflammatory, and Antipyretic
Antinociceptive activity was determined using formalin tests, writhing, and hot plate, whereas anti-inflammatory activity was found using the carrageenan-induced paw edema and antipyretic activity in brewer’s yeast-induced pyrexia tests [29,30]. Different doses of air-dried leaves extract (20, 100, and 200 mg/kg) exhibited significant (p < 0.05) antinociceptive, anti-inflammatory, and antipyretic activities independent of dosage. A separate study by Zakaria et al. (2005), suggested that the antinociceptive properties of C. olitorius are enhanced by temperature and may be mediated peripherally, rather than centrally, at least in part, via the involvement of the opioid receptor system [101]. This suggests that the heating process may enhance the breakdown of the effective compound responsible for the antinociceptive activity of COAE. It also found that the highest antinociception was achieved by 75% concentration of COAE treatment, while the highest concentration (100%) was found to produce no antinociceptive activity. This is the possibility that the loss of antinociceptive activity of the COAE for the highest concentrations of bioactive compound present in the extract causes deactivation of the antinociceptive-inducing receptors within the peritoneal cavity as a result of the presence of a high concentration of the bioactive compound of the respective extracts [101].
Similarly, Oyewole et al. (2015) provided the information that C. olitorius root is regarded as antipyretic and anti-inflammatory, where pyrexia and inflammation were induced by injecting 10 mL/kg of 20% (w/v) suspension of brewer’s yeast and 0.1% carrageenan, respectively [102]. The results indicate that C. olitorius root extract significantly (p < 0.05) decreased the elevated temperature following brewer’s yeast injection, and that in inflammatory tests, compared with the control group, the test group had significantly (p < 0.05) smaller granuloma weights and paw sizes. Thus, the above finding confirmed that C. olitorius root is a strong source of phytomedicine that can effectively treat inflammation and pyrexia associated with certain diseases that support its folklore use. Yan et al. (2013) reported that phenolic crude extracts C. olitorius significantly reduced LPS-induced protein expression of nitric oxide synthase (iNOS) and cyclooxygenase 2 (COX-2) in LPS-stimulated J774A.1, and prostaglandin E2 (PGE2) synthesis was suppressed in dose-dependent manners [103].
Recent studies have reported that C. olitorius leaf extracts significantly reduced the amplitudes of uterine tissue contraction dose-dependently, with the highest dose (666.67 μg/mL) achieving a 100% inhibitory effect, and significant inhibition of oxytocin-induced contractions was also seen [22]. Significant smooth relaxing effects of the uterine and gastrointestinal muscles, possibly due to their molecular interactions with receptors, inhibited contractions of the smooth muscles for extracts rich in flavonoids [104]. According to Parvin et al. 2015, a methanolic extract of C. olitorius aerial parts had a significant impact on reducing the number of abdominal constrictions induced by acetic acid through intraperitoneal administration [105]. It was stated that 100 mg/kg MECO was better than 200 mg/kg aspirin in relieving pain. Due to relaxing effects on the uterine, C. olitorius is used in ethnomedicine to facilitate labor and promote the smooth delivery of babies, particularly among the Yoruba people [106].
5.4. Antioxidant Activity
Free radicals are generated in large quantities due to different reactions within our bodies. In normal metabolic processes, free radicals cause pathological changes as a result of their interaction with various biological compounds both inside and outside living cells [107]. Consuming dietary antioxidants has been shown to reduce the risk of developing oxidative stress-related diseases such as diabetes, cardiovascular disease, and cancer by scavenging free radicals and reducing oxidative stress [108]. It was reported that C. capsularis leaves contained higher levels of total antioxidant capacity than that of C. olitorius leaves [54]. In the methanolic leaf extracts of both of the species, there was a higher percentage of radical scavenging activity compared with superoxide anion and DPPH radical scavenging assays, indicating that they are rich in phytochemicals with potent natural antioxidative activities [96,109]. A study by Yan et al. (2013) stated that a fraction of the whole plant, stem, and leaves showed significantly higher DPPH radical scavenging effects that are related to higher content of phenolics [103]. In addition, linoleic acid autoxidation inhibition was greater in all fractions than in α-tocopherol. A new study undertaken by Biswas et al. (2020) reported the leaves of ethanolic extract of C. olitorius demonstrated better DPPH, ABTS, and FRAP activities than C. capsularis, and a strong positive correlation exists between TFC and FRAP, followed by TPC and FRAP and TFC and ABTS [40]. Therefore, these correlations suggest that the presence of phenolic compounds such as flavonoids and polyphenols may be accountable for antioxidant activities and react with free radicals. Another study revealed that the methanolic extract of immature leaf extracts demonstrated the highest DPPH radical scavenging activities from any other growth stages that are nearly close to the value of α-tocopherol [110].
Polyphenols are frequently linked to the antioxidant capacity of the corchorous species; there are few studies on the role of other chemical components. Oboh et al. (2009) reported that polar aqueous extract exhibited significantly higher DPPH radical scavenging activity, Fe2+ chelating ability, and trolox equivalent antioxidant capacity than nonpolar hexane extracts [111]. The high polar polyphenols (flavonoids and nonflavonoid polyphenols) and vitamin C are responsible for the antioxidant activity of the hydrophilic extract, while the presence of carotenoids may have been responsible for the antioxidant activity of hexane extract [112]. The evaluation of the reducing power with the DPPH method revealed that the hydroethanolic extract of C. olitorius was the most active (IC50 = 45.58 µg/mL), which was in agreement with the results obtained by the FRAP method [112]. Among phytochemicals, phenolic compounds are the main contributors to antioxidant activity in plant extracts due to their high total content, interaction, redox property of an individual or combination of their diverse chemical structures, and synergistic effectiveness as hydrogen donors, reducing agents, and free radical scavengers [113,114].yos.
It was observed in the in vitro study that ethanolic extract (EE) C. olitorius exhibited overall better antioxidant activities (91.65% against DPPH radicals, 86.19% against β-carotene, and 70.24% against lipid peroxidation at 500 μg/mL) than the EAE and AE, dose-dependently [53]. Similar antioxidant activities were the half-maximal inhibitory concentration (IC50) value for DPPH, and ABTS radical scavenging activity was 37.65 µg/mL and 33.19 µg/mL, respectively, with the methanolic extract of C. olitorius [115]. Yakoub et al. (2020) reported that 1.5 mg/mL mucilaginous polysaccharides (PSc) extract of C. olitorius revealed 90%, 78%, and 69% against DPPH radical scavenging, lipid peroxidation, and β-carotene, respectively [41]. The same concentration of PSc also demonstrated an effective response against hydroxyl radicals and DNA breakage with a FRAP assay. This is consistent with previous research that reported better chelating efficiency on ferrous ions, this may due to the presence of condensed tannins at a high level [103]. Azuma (1999) reported that 5-caffeoylquinic acid showed a predominant phenolic antioxidant with stronger scavenger properties than ascorbic acid [47].
5.5. Cardiovascular Activity
Apart from its antioxidant capacity, the jute leaf has received a lot of attention in possible cardiovascular-protective activities. In the literature, it was reported that Corchortoxin (strophanthidin) is a cardiac, isolated from C. capsularis seeds, which exhibits cardiac activity comparable to digitalis genius [88]. In 2000 patients with cardiac decompensation, the clinical effects of Soviet-produced strophanthoids (cardiac glycosides with strophanthin-like effect speed, coronary-vessel effect, diastolic and diuretic effect degrees, etc.) have been studied. The most active drugs used in cardiac problems are olitoriside and corchoriside, isolated from C. olitorius [48]. Corchoroside A and B isolated from C. olitorius seeds provide digitalis-like action. The esterification of OH group in 3′ and 4′ position of Corchoroside A and in the position of 3′, 4′, and 19 of Corchorosol A by acetic acid resulted in the creation of new acetylated cardenolide. Corchorosol derivatives from C. capsularis are more effective than corchorosides in treating cardiovascular diseases.
In a study, the authors stated that C. olitorius seeds displayed strong inhibitory action against Na and K-ATPase, which correspond to digitoxin ouabain for the presence of cardenolides and oligoglycosides (corchorusosides A–E) [89]. These act on the contractile force of the cardiac muscle because of their ability to disrupt the heart function, most of them are highly toxic. Jute seed extract has shown greater activity than corchortoxin. The result of a study by Langendroff, where rabbit hearts were tested with the seed extract at different concentrations, shows that left ventricular pressure and coronary blood flow increase at low concentrations, where high doses were significantly decreased with a sharp increase in heart rates with ventricular fibrillation. Khatib et al. (1998) reported that the presences of active ingredients in the seed extract of C. olitorius are associated with direct effects on the cardiac myocardium. They also stated that C. olitorius seed oil exhibits estrogenic activity and is capable of treating cardiovascular diseases because of a high amount of active cardiac principles, particularly olitoriside, which demonstrated equivalent strophanthin effects in chronic cardiac patients [70].
According to one study by Airaodion et al. (2019), C. olitorius leaf extract has significant health benefits in terms of hyperglucosemia and hyperlipidaemia by significantly lowering fasting blood sugar, total cholesterol, LDL-cholesterol, and triglyceride, while significantly increasing HDL-cholesterol and HDL/LDL cholesterol ratio when compared with the control group [116]. This indicates that leaf extract is exceedingly helpful in controlling obesity and hypertension by inhibiting multiple targets and drug development from natural plant products.
It has been reported that Methyl-1,4,5-tri-O-caffeoyl quinate top is a ligand that is the major compound in the methanolic extract of C. olitorius and has a stronger bond and can block the receptor ACE2. Thus, it could make it difficult for coronavirus to enter cells, which could at least significantly slow the epidemic until the virus disappears and allow us to propose a methanolic extract of C. olitorius, and suggested as a natural and reliable treatment during the first stage of COVID-19 [117].
5.6. Antidiabetic and Bioadsorbent Properties
Plants that are rich in diverse polyphenolic compounds with the ability to interact with proteins and suppress carbohydrate-hydrolysing enzymes are important for their possible antidiabetic effect [118]. Besides antioxidant potential, there are considerable ethnomedicinal reports regarding the treatment of diabetes owning to the potential antidiabetic action of Corchorus spp. In a study, Oboh et al. (2012) evaluated the antidiabetic and antihypertensive inhibitory effects of soluble-free and bound polyphenol-rich C. olitorius leaf extracts on pancreatic α-amylase, α-glucosidase, and angiotensin I converting enzyme [119]. The result shows that extracts inhibited α-amylase, α-glucosidase (12.5–50.0 µg/mL), and ACE (10.0–50.0 µg/mL) in a dose-dependent manner and inhibitory activities disclosure by IC50, and free polyphenol-rich extracts had significantly (p < 0.05) high α-amylase, α-glucosidase, and ACE at 17.5 µg/mL, 11.4 µg/mL, and 15.7 µg/mL, respectively, in term of IC50. The inhibitory action of the extracts of C. olitorius consists of earlier studies where findings claimed that plant phytochemicals are mild inhibitors of α-amylase and powerful inhibitors of α-glucosidase [120,121], suggesting polyphenol-rich extracts of C. olitorius may help address a major draw-back associated with synthetic α-glucosidase inhibitors. Authors reported that all Corchorus species had a significant (p < 0.05) inhibitory effect on α-amylase and α-glucosidase activities at low concentrations (0–8 μg/mL) along with exhibiting antioxidant properties [15]. These findings can explain the biochemical justification behind the use in the management of T2DM in folklore medicine.
It has been reported that the ethanolic extract of the C. olitorius leaf significantly (p < 0.05) elevated levels of serum and liver bilirubin (direct and total), transaminases (AST and ALT), alkaline phosphatase, urea, creatinine, total cholesterol, triglyceride, and LDL-C, as well as reduced levels of total protein, globulin, albumin, and HDL-C in the alloxan-induced untreated diabetic rats normalized upon treatment with the ethanolic extract of C. olitorius leaf. Thus, the ethanolic extract of C. olitorius leaf exhibited antihyperglycemic and anti-dyslipidemic effects, and hence it could be considered safe for use as an antidiabetic recipe [122]. Another group of researchers pointed out that ethanolic seed extract of C. olitorius is a valuable antidiabetic agent due to its ability to lower blood glucose levels [32]. A significant difference (p ˂ 0.001) was observed in blood sugar levels between the control and treatment group from the 30th minute and 2nd hour in the glucose-loaded hyperglycaemic test (OGTT), where regular rats were administrated with glucose 30 min after orally administrated ethanolic extracts of C. olitorius. These findings show that C. olitorius leaves play a crucial role in health. A separate study reported that C. olitorius could aggregate blood plates and significantly reduce blood glucose levels in mice [105,123]. Authors Ozioma et al. (2014) investigated anti-hyperglycemic properties and antioxidant enzymes with the ethanolic extract of C. olitorius leaves orally administrated to streptozotocin-induced diabetic rats and obtained promising results [123]. The result indicate a high safety index (no toxicity or fatality) after oral administration of 5000 mg/kg body weight of the ethanol extract, and the extract reduced blood glucose levels dose-dependently and reduced superoxide dismutase, catalase activities, and liver function enzymes assayed ALT, AST, and ALP when compared with nontreated groups. A similar trend was noted in the methanol extract of aerial parts, which significantly decreased blood glucose levels, and the highest dose of extract had better antihyperglycemic activity than with the drug glibenclamide loaded in mice [105]. The IC50 for α-amylase and α-glucosidase was found to be 27.95 µg/mL and 41.64 µg/mL, respectively, with the methanolic extract of C. olitorius exhibiting good antidiabetic potential [115].
A group of researchers investigated the antiobesity of C. olitorius leaf polyphenolic compounds in LDLR (low-density lipoprotein receptor), demonstrating that the effect was associated with a decrease in oxidative stress and an increase in β-oxidation in the liver [16]. The gene expression of the PPARα and CPT1A regulatory molecule in fatty acid β-oxidation significantly up-regulated by 1.56 and 1.61 fold, respectively, in the treated group, while gp91phox, a key subunit of NADPH oxidase and involved in oxidative stress, was significantly down-regulated (−1.61-fold) compared with the control group. The finding suggests that leaf extract possesses an anti-hyperglycemic effect, which is linked to managing diabetes mellitus and metabolic disorders in animals and helpful in preventing obesity caused by diet. In another study, researchers found that the C. olitorius leaf extract significantly reduced weight gain and visceral white adipose tissue in obese rats [124]. The levels of serum glucose, triglycerides, total cholesterol, lipoprotein-low cholesterol (LLC), free fatty acids, IL-1β, inflammatory cytokines (TNF-α and IL-6), insulin, HOMA-IR index, and leptin were significantly reduced in obese rat groups, along with a significant increase in high-density lipoprotein cholesterol (HDL-C) and the number of adipocytes. The findings coincide with other reports that the treatment of diabetes and obesity-accompanying insulin resistance and metabolic disorders is the most responsible component of phenolic compounds as well as phytol, including terpenes and phenolic compounds [125]. Therefore, the authors suggest leaf extract is effective as orlistat in preventing and ameliorating obesity, steatosis, fatty liver, hyperlipidaemia, and insulin resistance in rats fed a high-fat diet (HFD) by suppressing pancreatic lipase activity, TNF-α, IL-1β, and leptin resistance along with the up-regulation of adiponectin.
A group of researchers studied the effect of C. olitorius in structural changes of testicular tissue in streptozotocin-induced diabetic rats [126]. They found that blood glucose levels significantly decreased in the treatment groups compared with the diabetic group; seminiferous tubule degenerations were prevented, and apoptotic cell numbers were reduced. Results indicate that C. olitorius decreases the complications of diabetes mellitus-induced rat testis, and precise information of the experiment indicates possible therapeutic effects in diabetes treatment. A group of researchers demonstrated, significantly ameliorating inflammation-mediated obesity in high-fat diet mice that is associated with the inactivation of inflammatory reactions in gut serum, as well as 3T3-L1 cells, and an inhibition of metabolic endotoxemia and colonic inflammation as well as a rebalance of gut microflora, including an increase in Lactobacillus and decrease in Desulfovibrio [127]. It has been reported that alcoholic and chloroform extracts of C. olitorius (100 mg/kg) protected against diabetic complications such as hyperlipidemia and determinant effects on the liver tissue. These pharmacological effects are due to the presence of some bioactive phytochemical constituents such as theophylline, trans-2, 3-dimethoxycinnamic acid, 7-hydroxy-4-methyl coumarin, apigenin 7-glucoside, and glycitein [128].
5.7. Hepatobiliary, Renal, and Haematological Activity
This key metabolic organ detoxifies toxic metabolites, synthesizes proteins, and produces essential compounds required by the body for proper functioning [129]. In diabetes mellitus, impaired liver function is associated with decreased hepatic delta-aminolevulinic acid dehydratase (δ-ALAD) activity. In a recent study, it was reported that water-soluble extracts of C. olitorius leaves ameliorated the alcohol-induced increase in hepatic inflammation and lipogenic protein levels by improving the gut–liver axis [50]. Saliu et al. (2019) reported that supplementation of jute leaf (C. olitorius) to streptozotocin-induced diabetes rats at 100 mg/g for 30 days significantly (p < 0.05) restored the decreased hepatic δ-ALAD activity and increased hepatic catalase and SOD activity accompanying the decrease in serum AST and AST activities [130]. Thus, the finding suggested that C. olitorius leaf extract has hepatoprotective properties and could protect the liver from diabetes-induced oxidative damage as well as improve the activities of some critical enzyme systems required for normal hepatic function, such as delta-aminolevulinic acid dehydratase. Laskar et al. (1986) reported that after feeding a protein-enriched diet made from C. olitorius seeds bodyweight, including the weight of the liver, increased in test animals and also significantly increased AST, ALT, and total lipid of the liver, whereas the AST and ALT of serum decreased. It was found that a cholesterol-free diet containing powdered green leaves of C. olitorius in rats helps to reduced hepatic cholesterol and increased fecal bile acid via inhibition of enterohepatic circulation condition and neutral sterol excretion [131]. Earlier studies discovered that rats with hypercholesterolemia had lower levels of serum and hepatic cholesterol and increased fecal steroid excretion [132], where rats fed a diet JML (fridze dried leaf extract) containing a water-soluble viscous polysaccharides containing dietary fibers are the principal effective component.
A study conducted by Mazumder et al. (2003) observed the impacts of different doses of C. olitorius on liver, kidney, and hematological parameters [133]. Results point out high doses significantly increased WBC counts, whereas SGOT, SGPT, NPN, and plasma cholesterol levels increased to their highest levels when given in medium and high doses. A group of authors demonstrated that the aqueous extract of C. olitorius, after administration for 30 days on haematological and plasma biochemical parameters in male albino rats, was found to have no significant effect of PCV, RBC, MCV, MCHC, platelet, neutrephil, lymphocyte, oenophile, and monocyte values at various doses in their respective tests, but the TWBC count was substantially decreased compared with the controls [134]. So, it may be recommended for people with blood disorders to consume. A histopathological study supported that AECO markedly mitigated chemically induced liver toxicity and preserved the histo-architecture of hepatic tissue [135]. Do et al. (2021) reported that the water-soluble extract of C. olitorius leaves ameliorated the alcohol-induced increase in hepatic inflammation and lipogenic protein levels by significantly down-regulating the expression of inflammatory cytokines, improving the gut microbial composition that helps to improve gut health [50].
5.8. Anticonvulsant Activity
Methanolic extract of C. olitorius seed (MECO) provided significant protection against chemo-convulsive agent-induced convulsions in mice by altering catecholamine and brain amino acid levels [133]. Results suggest significantly increased catecholamine levels in extract-treated mice, and GABA (γ-amino butyric acid) increased in mice brains after six-week treatment. Along with GABA, increased glutamate resulting from conversion of α-ketoglutarate to the glutamic transmission of glutamine levels may be associated with MECO’s anticonvulsant properties. So, it has been suggested that extract processed significant roles in CNS depressant and anticonvulsive properties against chemoconvulsions in mice, probably due to increased catecholamines and 5-HT in mice. Das and his colleagues figured out how to handle C. olitorius leaves’ aqueous extract cardiotoxicity mediated against sodium arsenite in experimental rats [136], suggesting leaf extract has a significant defense effect against myocardial injury caused by arsenic.
5.9. Neuromodulatory Activity
Dietary polyphenols are gaining scientific attention for their protective effects against degenerative diseases and cognitive decline due to its antioxidants properties [137]. A group of researchers revealed that hydroalcohol extract of C. olitorius leaves had inhibitory effects on neuroinflammation in a mouse model of lipopolysaccharide (LPS)-induced neuroinflammation [138]. They characterized 25 metabolites, and one of the major constituents (5.7%) was 1,5-dicaffeoylquinic acid, which significantly protected microglia from H2O2-induced cytotoxicity. The LPS group showed stronger GFAP immunoreactivity when compared with the control group and reduced GFAP expression in Co extract in the pretreated group compared with the LPS group. Immunohistochemistry showed decreased expression of the astrocytic marker glial fibrillary acidic protein, which is linked to inflammatory activity, and the inflammatory marker cyclooxygenase-2, all of which support the findings of previous research. These activities indicate several polyphenols such as mono- and dicaffeoylquinic acid that protect MC65 cells from Aβ-induced cell death and lead to controlling oxidative stress, in addition to other effects such as anti-Aβ fibrillation, and delay the aging process as well as neurogenerative disorder development [139,140]. Findings of the study correlated with significant improvement of cognitive function as well as abatement of GFAP and COX-2 expression and reduction in LPS-induced neurodegeneration in Co-treated mice. This may be due to the abundance of flavonoids in C. olitorius leaves and demonstrated neuroprotective properties by increasing neuronal viability, decreasing dopaminergic neuron loss in the brain, acting as an antiamyloidogenic agent, and ameliorating cognitive impairment [141]. Therefore, phenolic compounds have achieved attention in the medicinal field due to lowering risk by altering the progression of multifactorial diseases such as neurodegenerative and cardiovascular diseases and cancer.
5.10. Antioesterogenic and Antifertility Activity
It has been found that the methanolic extract of C. olitorius seed arrested the normal estrus cycle in female mice, and tremendously delayed sexual maturation was proven by the age at the vaginal opening and first oestrus appearance [142]. On administration of the methanolic extract of C. olitorius, the decreased weight of ovaries and uterus was observed in the adult. In treated mice, the content of cholesterol and ascorbic acid in the ovaries significantly increased, and two key enzymes (∆5-3-β hydrxysteroid dehydrogenase (HSD) and glucose-6-phosphate dehydrogenase (G-6-PD) decreased significantly, along with the decreased weight of the ovary, uterus, and pituitary. On the basis of available data, it is hypothesized that depression and reduction in ovarian steroidogenesis are the causes of delayed maturation in mice treated by the extract of C. olitorius seed. The improved substrate and decreased enzyme concentration is an indication that steroidogenesis is inhibited, and it may be induced by the presence of flavonoids in C. olitorius leaf extracts [143].
On the other hand, the fertility test by MECO treated with medium and high dose in male mice showed 100% negative results, and different dose levels resulted in a simultaneous decrease in testicular Δ5-3β-hydroxyl steroid dehydrogenase and glucose-6-phosphate dehydrogenase activities, both of which are involved in testicular steroidogenesis [144]. In the testis of MECO-treated mice, the activities of ascorbic acid oxidase, lactate dehydrogenase, and malic dehydrogenase were decreased, whereas carbonic anhydrase activity was significantly increased. All of these findings suggest that the methanol extract of C. olitorius seed inhibited gonadal steroidogenesis and thus produced antifertility activity in sexually matured male mice. All of these findings suggest that MECO inhibited gonadal steroidogenesis, resulting in an antifertility effect in sexually mature male mice.
5.11. Dermatitis and Wound Healing Activities
Wound healing is the process by which the skin and other soft tissues are repaired following injury. Authors Yokoyama et al. (2014) investigated the skin hydration on atopic dermatitis in mice using C. olitorius leaf extract excluding high MW (COEW), and the results show a possible therapeutic benefit for atopic dermatitis due to decreased plasma immunoglobulin E (IgE) level as well as mast cell degranulation [145]. In this study, mice treated with the COEW cream experienced significantly increased skin hydration and reduced transepidermal water loss (TEWL) compared with control mice, but IgE concentrations were not affected, whereas NC/Nga mice showed a decreased AD score. It has been stated that C. olitorius leaves comprise high fatty acid counts, specially α-linolenic and linoleic acid, and high quantities of antioxidant molecules [31]; these compounds have the ability to reduce inflammation by suppressing activation of ROS and lead to the aggravation of AD [146,147]. According to Ohtani et al. (1995), polysaccharides are known to facilitate skin hydration, and C. olitorius leaves contain a significant amount of mucilaginous polysaccharides, providing adequate skin hydration and reducing skin dryness [20].
Similar studies have shown that the optical densities of the plasma of rats treated with different doses have decreased over time, suggesting C. olitorius has a platelet-aggregative influence [123]. A separate study stated that both plant powder and aqueous extracts of C. olitorius exhibited a high level of antioxidant activity and significant wound healing activity in an excision model [148], where on the 18th day the percentage of wound contraction was determined to be 100% for powdered plant ointment, 5% for powdered plant ointment, and 100 mg/mL for aqueous extract, respectively. The microbial load on the wound surface was estimated similarly to the wound healing capability, and research findings indicated traditional uses of this plant for wound treatment. Induced wound healing and the regeneration of injured tissue characteristics, alongside avoiding side effects and resistance of synthetic medicines, mean C. olitorius could be an alternative source of wound healing medicine to develop.
5.12. Antimicrobial Activity
Phytochemicals are considered to be attractive sources of natural antimicrobial compounds for combating the future public health challenge of antibiotic resistance. In order to test the efficacy of C. olitorius leaf extracts as antimicrobials, two different assays were used: agar diffusion and tube dilution, and both aqueous and methanolic extracts towards several bacteria such as Escherichia coli, Salmonella typhi, Klebsiella pneumoniae, and Staphylococcus aureus have been well-reported, with methanolic extracts exhibiting greater inhibition and activity indices than aqueous extracts. Susceptibility increased with concentration, with E. coli exhibiting the greatest susceptibility. This can be explained by the existence of phytochemical elements and is prophylactically significant [149], and the choice of extraction solvents influences antimicrobial and antibacterial activities. Following that, the effect of three differently derived extractions (EE, AEE, and AE) of C. olitorius against five Gram-negative and three Gram-positive bacteria by Yakub et al. (2018) [53] suggests that both EE and AEE have the potential to fight all tested bacteria, as well as demonstrate a notable increase in the zone of inhibition with an increasing concentration of extract. EE had the highest zone of inhibition, followed by AEE and AE against all strains, where maximal inhibitions were noticed for S. aureus (34.5 mm), K. pneumonia (23.5 mm), and M. luteus (22 mm) at 50 mg/mL. It was also found that mucilaginous polysaccharides (PSc) extract had a broad spectrum of antibacterial activities against all (Gram+ and Gram−) bacteria tested, whereas soluble fraction (SF) extract was effective against Salmonella enterica and Klebsiella pneumoniae at a concentration of 25 mg/mL [41]. In another study, Rume et al. (2016) reported that the N-hexane fraction of a methanolic extract of C. capsularis leaves exhibited antibacterial and antifungal activity with a zone of inhibition of 0.9–1.5 mm, followed by the hexane extract.
The authors described the in vitro interaction of C. olitorius leaf extract with five antibiotics on methicillin-resistant and methicillin-sensitive Staphyloccus aureus [150]. The most remarkable activity was against all bacterial strains tested, specially S. aureus and E. coli, which were unable to grow in the presence of components of stem dry oil, while components of leaves and stem oil had antimicrobial properties with MIC values ranging from 0.40–0.8 and 1.6- > 3.2 mg/mL, respectively [70]. The study concluded that petroleum ether extracted leaves offered an excellent activity against E. coli, S. aureus, and Y. enterocolitica, 20 mm, 19 mm, and 19 mm, respectively.
Conditions of extraction and the type of solvent influence the jute-leaf antimicrobial activity, indicating the active compounds’ biochemical nature. Abir and other coauthors fractionated lipophilic leaf extracts of C. olitorius and C. capsularis were tested against S. aureus and E. coli along with a control organism, E. coli DH5± [151]. The results reveal that fractions of C. olitorius leaves were more effective against S. aureus (19 ± 2.80 mm inhibition zone), whereas fractions of C. capsularis leaves have been more effective against E. coli (15 ± 2.3 mm inhibition zone). Comparing the above findings, the antibacterial effects of C. olitorius leaf extract was shown to be higher than those of C. capsularis leaves. Additional analysis should be carried out on isolated fractions to introduce lead components of new-generation antibacterial drugs, which account for promising findings on antibacterial efficacy. In addition to antimicrobial activity, antiviral properties of C. olitorius extracts have been investigated. The aqueous layer of C. olitorius leaves against the measles virus was reported by a group of researchers [75].
5.13. Antimalarial Activity
According to Sathiamoorthy et al. (2007, 1999), an aqueous extract of C. olitorius exhibited strong growth of inhibition (>96%) against malaria parasite plasmodium falciparum [152]. In a recent study, the extract of C. capsularis’ ability to control two common malarial vectors, Anopheles stephensi and a dengue vector A. aegypti, was reported [153], where C. capsularis ethyl acetate extract has been more prominent than acetone and methanol extracts in larvicidal activity. In terms of ovicidal activity, no hatchability was attained at 300–450 ppm in An. Stephensi, and 375–450 ppm in Ae. Aegypti, due to the action of phytochemicals present in the extract interfering with chorion of the eggs. A similar result was also recorded for the ethanolic extract of Andrographis paniculata against An. stephensi in ovicidal and gravid mortality [154]. Sathiyamoorthy also stated that aqueous leaf, stem, and seed extract of C. olitorius was found to have >96% growth inhibition against the malaria parasite Plasmodium falciparum [155]. The leaf extracts of C. olitorius were revealed to posses antimicrobial activity against different Gram-positive (Enterrococcus fecealis and Bacillus subtilis) and Gram-negative bacteria (Escherichia coli, Proteus vulgaris, Klebsiella spp., and Serratia marcescens) [156]. These finding suggest that C. capsularis and C. olitorius could be used to a greater extent to control and kill the mosquito menace. Since mosquitoes developed a resistance to all and altered the behavior of nontarget organisms, it is urgent to develop alternative sources of environmentally friendly drugs, preferably plants from nature.
5.14. Antinutrients and Insecticidal Effect
It has been reported that cyanide concentration in the fresh sample (128 mg/Kg) of C. olitorius was less than the maximum permissible level of 200 mg/kg of fresh weight vegetables [46]. This finding suggests that the cyanide concentrations in the fresh and pressed samples are insufficient to cause toxicity in humans. In other words, compared with processed samples, the total concentrations of nitrate, soluble oxalate, and oxalates in fresh leaves are higher than permissible levels. However, moderate cooking can effectively reduce plant toxins to acceptable levels without altering the plant’s nutritional potential.
In a separate study, Roy (2014) assessed the role of C. capsularis leaf phytochemicals in Diacrisia casignetum Kollar feeding, growth, and reproduction [157]. When compared with young and senescent leaf-fed insects, mature jute-leaf-fed insects appeared to have shorter larval and postlarval developmental durations but longer adult longevity (p < 0.05). The highest fecundity was observed on mature leaves, followed by young and senescent leaves. The result suggests that the growth and development of D. casignetum are associated with nutrient content in relation to the secondary metabolites of three types of jute leaves. It has been reported that consumption of higher quantities of secondary chemicals has been shown to significantly decrease adult longevity and fertility and delay larval growth [158]. It was concluded that mature leaves with high nutritional factors and low levels of total phenols and other secondary chemicals contributed to a shorter developmental period, increased growth rate, fecundity, and cumulative D. casignetum, followed by young and senescent leaves.
In a laboratory condition, the potency of C. capsularis petroleum ether extract of three stored product pests (Callosobruchus chinensis, Sitophilus oryzae L, and Tribolium castaneum Herbst) was evaluated in adult phase. It was pointed out that the highest efficacy was recorded against C. chinensis with LD50 (μg/cm) being 93.82 after 24 h and 5.63 after 48 h, followed by S. oryzae, while LD50 was 77.63 after 24 h and 23.32 after 48 h, with minimum performance against T. castaneum [159]. The results given support the findings of Mahfuz and Khanam (2007) [160], who stated that C. capsularis were extremely toxic to adults T. confusum.
5.15. Toxicology
Corchorus leaves have a long history of use in medicine and are widely assumed to be safe. However, the available toxicological data is limited, but it generally supports this assessment. In an As-contaminated rice-induced toxicities study, rats of groups II, III, and IV were fed standard laboratory pellets supplied with As-contaminated rice, 4% C. olitorius leaf powder and As-contaminated rice + 4% C. olitorius leaf powder, while Group I was nourished with laboratory pellets [21]. It was observed that Group II had significantly decreased Hb concentration, WBC and RBC count, and that platelet count decreased nonsignificantly compared with the control. Hematological abnormalities and other serum indices were significantly improved by C. olitorius leaves with arsenic-contaminated rice. The levels of TG, TC, and LDL-C were significantly higher in rats treated with As-contaminated rice (Group II) compared with controls. This change is most likely caused by increased proteolytic activity and/or decreased protein synthesis or destruction of hepatic protein-synthesizing subcellular structures. A possible explanation for the increased serum proteins could be the result of antioxidant activity of C. olitorius leaves, which comprise large amount of α-tocopherol equivalent Vit-E [8], which have an effect on hepatic insulin resistance and increase serum proteins by stimulating the incorporation of amino acids into protein [161]. A similar result was obtained by Adedosu et al. [162], where sodium arsenite elevated serum enzymes in rats, C. olitorius leaf extract restored them nearly to their control levels. A group of researchers also stated that aqueous C. olitorius leaf extract (AECO) significantly restores the biochemical and haematological parameters near to the normal status in Pb-acetate and CdCl2 intoxication-tested rats through antioxidant activity and/or by preventing bioaccumulation of Pb within the tissues of experimental rats [163,164]. Additionally to this, AECO could significantly reduce the Cd-induced increase in the Bad/Bcl-2 ratio and the over-expression of NF-jB, caspase 3, and caspase 9.
In another study, the authors demonstrated the effects of C. olitorius leaves (AECO) against sodium arsenite (NaAsO2)-induced cardiotoxicity in experimental rats [136]. The treatment of rats with AECO (50 and 100 mg/kg) for 15 days prior to NaAsO2 intoxication significantly protected cardiac tissue against arsenic-induced cardiac oxidative impairment, cardiac arsenic content, and DNA fragmentation. In addition, pretreatment significantly prevented the arsenic-induced alterations of lipid profiles as well as enhancement of MDA and protein carbonyl content, while GSH, GST, GPx, and GR significantly improved in cardiac tissue. Histological studies of the organs supported the protective role of jute leaves, probably due to the presence of the substantial quantity of antioxidants and its free radical scavenging properties of flavonoids and phenolic compounds. It is possible that supplementing with antioxidant-enriched C. olitorius leaves may be a potential therapy and directly connected to the amelioration of chronic Pb, As, and Cd intoxication. Thus, there is a tremendous opportunity to develop this vegetable as a dietary composition that contains nutritional value as well as free radical detoxification capability to help neutralize the negative effects of contaminants such as heavy metals, metalloids, and other pollutants.
Negm et al. (1980), concluded that seed extract was the most toxic preparation because it contains the highest percentage of cardiac steroids, followed by leaves, stems, and roots [79]. The leaf of C. capsularis contains HCN, which may be responsible for cytotoxicity. A lethal dose (LD50) of Corchoroside A in mice is 0.053–0.0768 mg/kg, and in the case of Corchoroside B, it is 0.059–0.1413 mg/kg [48].


Table 4.
A summary of the biological activities of C. capsularis and C. olitorius.
Table 4.
A summary of the biological activities of C. capsularis and C. olitorius.
| Biological Activities | Species | Plant Parts | Extraction Solvent/Fed Diet | Main Findings | References |
|---|---|---|---|---|---|
| Antioxidant activity | C. olitorius | Leaf stem | 96% alcohol | Both leaves and stem displayed DPPH radical scavenging (95.1% and 97.1%), respectively, at 400 µg/mL. | [70] |
| ,, | Leaf | Distilled water | Mucilaginous polysaccharides (PSc) showed more fantastic antioxidant activities than soluble fraction (SF) extract. Antioxidant activities were about 90% against DPPH•, 78% against lipid peroxidation, and 69% against β-carotene at 1.5 mg/mL. | [41] | |
| Cardiovascular activity | C. olitorius | Seed | Methanol | Compounds such as Corchoroside A, Corchoroside B, Strophanthidin trioside, Coroloside, and Olitoriside are widely used for heart failure treatment. | [87] |
| Antitumor and anticancer promoting activity | C. capsularis C. olitorius | Leaf | Methanol | Phytol and mono galactosyldiacylglycerol at concentrations of 15 g/mL and 30 g/mL completely inhibited the induction of Epsterin-Barr virus (EBV) early antigen in Raji cells, and viable cells decreased about 20% in the inducer-treated Raji cells. | [17] |
| C. olitorius | Leaf | Ethanol | ECO treatment had a dose-dependent effect on HepG2 cell proliferation and 12.5 µg/mL effectively triggered apoptosis by increasing caspase-9 activity mitochondria as well as caspase-mediated pathways. | [14] | |
| C. olitorius | Whole plant | Methanol | Cytotoxicity against HeLa, HL460 lung cancer cell line, and PC3 prostate cancer cell line, indicating antitumor potential and Galactolipid 1 antitumor promoting activity. | [13] | |
| C. olitorius | Leaf | Dichloromethane (DCM) and aqueous | Two extract-induced apoptotic in Colo-320 and Colo-741 cells lines at 50 µg/mL concentration, and extract-induced apoptosis was more effective in metastatic Colo-741, colon adenocarcinoma cell lines. | [12] | |
| C. olitorius | Stem | Dichloromethane | Extracts have growth-inhibiting effects on humans MCF-7 and MDA-MB-231 adenocarcinoma. Stigmasterol demonstrated cytostatic activity against Hep-2 and McCoy cells. | [45] | |
| C. olitorius | Leaf | 67% methanol and chloroform | Polyphenol-enriched extracts (PEEs) of C. olitorius leaves significantly reduced the viability of tumor Caco-2 cancer cells without any detrimental effects on the healthy CCD841 line. | [49] | |
| C. olitorius | Leaf | Aqueous | Significant antiproliferative effects on SUIT-2, A-375 and AGS cells at a concentration as low as 2.54 mg/mL. Moreover, extracts strongly inhibited angiogenesis and the growth of A-375 and AGS tumors. | [94] | |
| Antiulcer activity | C.olitorius | Leaf | Aqueous | Oral aqueous extract dose-dependently inhibited gastric ulcers. The extract (400 mg/kg) had the highest cure rate (94.08%) and (33.75%) for acetic acid and ethanol/aspirin-induced ulcers, respectively. | [97] |
| Antidiabetic and bioadsorbent properties | C. olitorius | Leaf | Methanol | The dose-dependent inhibition of α-amylase and α-glucosidase (12.5–50.0 µg/mL), as well as ACE (10.0–50.0 µg/mL), was observed. | [65] |
| C. olitorius | Leaf | Phytol and terpenes have hepatoprotective and antiadipogenic properties and may help manage insulin resistance and metabolic disorders associated with diabetes and obesity. | [84] | ||
| C. olitorius | Leaf | Methanol | The extract reduced blood glucose on a dose-dependent basis. | [105] | |
| C. olitorius | Leaf | Ethanol | Extract significantly reduced (p > 0.05) the activities of catalase, SOD, and liver function enzymes. Oral administration of extract had an anti-hyperglycemic effect in streptozotocin-induced diabetics, suggesting it could be used to treat diabetes mellitus. | [165] | |
| C. olitorius | Seed | Ethanol | Significantly reduced blood sugar levels in normoglycaemic, OGTT, and diabetic rats, as well as a suppressed postprandial increase in glucose-loaded rats, and a decreased blood glucose level in diabetic rats. | [32] | |
| Antiobesity effect | C. olitorius | Leaf | Ethanol | It reduces oxidative stress and increases β-oxidation in the liver, which helps prevent diet-induced obesity. | [16] |
| Stem Leaf | Dichloromethane | Oleanolic acid possesses glucose-lowering properties. | [45] | ||
| Antifertility activity | C. olitorius | Seed | Methanol | Seed extract inhibited male reproductive capacity in sexually mature mice, since it interferes with gonadal steroidogenesis. | [144] |
| Antinociceptive, anti-inflammatory, and antipyretic | C. capsularis | Leaf | Chloroform | Reduce the number of abdominal constrictions that was confirming its traditional use of inflammatory and pain-related diseases. It is also linked to curing chronic urinary bladder inflammation. | [29] |
| Anti-inflammatory and antipyretic | C. olitorius | Root | Distilled water | Extract significantly lowered the elevated temperature after the brewer’s injection. Compared with controls, approximately 50 mg/kg of active ingredient had the least granuloma weight. | [166] |
| C. olitorius | Aerial part | Aqueous ethanol | Isoquercetin-rich extracts reduced carrageenan-induced rat paw edema in inflammatory exudates and demonstrated activity against metastatic melanoma, leukemia, and osteosarcoma cell lines. | [57] | |
| C. olitorius | Leaf | chloroform: methanol (2:1, v/v) | α-linolenic acid and linoleic acid ability to anti-inflammatory properties and wound healing promotion. | [72] | |
| Proliferative activity | C. olitorius | Leaf | Chloroform and methanol | Polysaccharides rich in uronic acid showed proliferative activity toward the murine splenocyte. | [20] |
| Anticonvulsant activity | C. olitorius | Seed | Methanol | Seed extract significantly increased the level of catecholamines in mice brain after a 6-week treatment. | [165] |
| Gastroprotective activity | C. olitorius | Leaf | Ethanol | Extract administration significantly inhibited gastric wall mucus depletion, and a group treated with 400 mg/kg produced a significant amount of gastric mucus. | [100] |
| Hepatobiliary, renal and haematological activity | C. olitorius | Leaf | Leaf powder | Reduces hepatic cholesterol while increasing neutral fecal bile acid and neutral sterol excretion. | [131] |
| C. olitorius | Seed | Powder | A significant increase was seen in AST, ALT, as well as total lipid of liver, while serum AST and ALT dropped. | [48] | |
| C. olitorius Leaf | Leaf | Water-soluble extract | Reduced the levels of serum biomarkers for liver injury and reduced the overexpression of inflammatory cytokines and lipid metabolism-related proteins in the liver. | [50] | |
| C. olitorius | Leaf | Leaf powder | Increased δ-ALAD, hepatic catalase, and SOD activities were observed in association with a decrease in serum AST and AST activity. | [130] | |
| Wound healing | C. olitorius | Leaf | Methanol and aqueous | An excision wound model showed significant wound healing activity for both powder and aqueous extract. | [148] |
| Neuromodulatory activity | C. olitorius | Leaf | Ethanol | Extracts significantly correlated with improved cognitive function and reduced neurodegeneration induced by LPS. | [138] |
| Phytoalexin activity | C. olitorius | Fresh young leaf | 95% ethanol | Isolated stress metabolites and volatile compounds had good activity against the microorganisms. Antifungal substances are released or fungioxic compounds are synthesized in response to infection or injury. | [84] |
| Antileishmanial activity | C. capsularis | Leaf | Chloroform | Showed potent antileishmanial activity against L. donovani promastigotes with an IC50 value of 79 μg/mL and exhibited very specific apoptotic features by targeting LdTryR. | [80,167] |
| Antibacterial activity | C. capsularis C. olitorius | Leaf | Lipophilic extracts | Both of the species of Corchorus showed antibacterial activity against (Gram+ and Gram−) bacteria. Fractions of C. capsularis leaves extract were found to be more effective against E. coli. | [151] |
| C. olitorius | Leaf | Petroleum ether, methanol, and ethyl acetate + water | Antibacterial or antifungal activity was observed in all extracts, and petroleum ether was demonstrated with zone diameters of 14 to 20 mm. | [166] | |
| C. olitorius | Leaf | Ethanol | Simultaneous administration of antibiotics to patients who eat C. olitorius regularly needs to be reappraised because of possible synergism and antagonism. | [150] | |
| C. olitorius | Seed | Petroleum ether, chloroform, methanol | Isolated cardenolide glycosides at 150 g/mL were the most effective against the bacteria tested, with a 20–25 mm zone of inhibition. | [168] | |
| C. olitorius | Leaf | 95% ethanol | Volatile components are effective against Gram-positive and Gram-negative bacteria. | [84] | |
| Antimalarial activity | C. capsularis | Leaf | Acetone, ethyl acetate, ethanol | Different extracts demonstrate mosquitocidal activity against Anopheles stephensi, a common malaria vector, and Aedes aegypti, a dengue vector. | [153] |
| Antimicrobial activity Antibacterial activity | C. olitorius | Leaf stem | 96% alcohol | Leaf and stem oil components showed significant antibacterial activity with MIC values of 0.40–0.8 and 1.6- > 3.2 mg/mL, respectively. | [70] |
| C. capsularis | Leaf | Methanol | The highest activities against Gram-positive, Gram-negative bacteria and fungi, with a 0.9 to 1.5 mm zone of inhibition. | [96] | |
| C. olitorius | Leaf | Methanol and aqueous | Both extracts had antimicrobial activities, but methanolic extracts displayed more comprehensive inhibition and activity indices. | [149] | |
| Antifungal activity | C. olitorius | Leaf | Petroleum ether, methanol, and ethyl acetate + water | All extracts displayed varying antifungal activity, and ethyl acetate + water extract showed prominent activity against Geotrichum candidum. | [166] |
6. Conclusions and Future Perspectives
In terms of nutritional value, the leaves of C. olitorius and C. capsularis are valued for their high dietary fiber, protein, vitamins, iron, and folate. Based on previous scientific reports on phytoconstitute and biological activities, C. capsularis and C. olitorius have potential to be used as a substitute ingredient in pharmaceutical and nutraceutical industries. Two species have various major phytoconstituents, namely chlorogenic acid, 3,5-dicaffeoylquinic, quercetin, flavone, tannins, diacylglycerol, uronic acid, α-linolenic acid, and β-sitosterol. Therefore, this justifies the therapeutic activity against a wide array of diseases due to the presence of numerous bioactive constituents and their antioxidant, anticancer, antiulcer, antidiabetic, antinociceptive, and antibacterial properties that can prevent a number of diseases. Biological mechanisms have been discovered for a small number of bioactive compounds, but many studies have shown that isolation and characterization of these compounds results in limited information. Additionally, the majority of these studies used crude extracts and, in some cases, fractions of the leaves without properly characterizing or standardizing the phytochemical constituents, which analyses of which might yield inconsistent or inaccurate results. Additional investigation into the specific phytochemicals present in C. capsularis and C. olitorius extracts may lead to the development of effective therapeutics for the prevention and/or treatment of these disease states. More research should be conducted to evaluate its usefulness as a means of preventing oxidative stress, cancer, ulcer, diabetes, inflammation, neuroinflammation, and microbial infections. Even with the promising findings obtained from the in vivo and in vitro studies, it is still important to conduct clinical trials that are randomized, controlled, and sufficiently large to obtain reliable therapeutic data. On the other hand, the mechanism of action of the chemical components and molecular mechanisms in combating different diseases or disorders should be explored by application of different techniques, such as genomics, transcriptomics, proteomics, and metabolomics. Based on the available literature and previously published scientific findings, it is proposed that both species have significant potential for human nutrition, health, and beauty care, as well as for development as functional ingredients due to their nutritional properties and bioactive compound composition.
Author Contributions
Conceptualized the idea and methodology and performed the literature search, A.B. and S.D.; literature search, Z.M.B. and J.Z.; wrote the first draft of the manuscript, A.B. and S.D.; critically reviewed and revised the manuscript, L.L., Z.M.B., and D.A.; project administration and investigation, S.H. and Y.D.; funding acquisition and supervision, D.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the Chinese Agriculture Technology Research System (CARS-16-E04) and Agricultural Science and Technology Innovation Program (ASTIP-IBFC03).
Conflicts of Interest
The authors declare that there are no conflict of interest.
Abbreviations
| ABTS | 2,2’-azino-bis(3 ethylbenzothiazoline-6-sulphonic acid) |
| AFB1 | Aflatoxin B |
| δ-ALAD | Hepatic delta-aminolevulinic acid dehydratase |
| CPT1A | Carnitine palmitoyl transferase 1A |
| COX-2 | Cyclooxigenase-2 |
| DAG | Diacylglycerol |
| DCM | dichloromethane; |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| DW | Dry weight |
| EC50 | Half maximal effective concentration |
| EGFR | Epidermal growth factor receptor |
| FFA | Free fatty acids |
| FRAP | Ferric reducing ability of plasma |
| GABA | γ-amino butyric acid |
| G-6-PD | Glucose-6-phosphate dehydrogenase |
| GPx | Glutathione peroxidase |
| GSH | Glutathione |
| GST | Glutathione S-transferase |
| HE | Hydrophilic extract |
| HFD | High-fat diet |
| HPLC | High-performance liquid chromatography |
| HDL-C | High-density lipoprotein cholesterol |
| HepG2 | Human hepatocellular carcinoma |
| IC50 | Half maximal inhibitory concentration |
| IgE | Immunoglobulin E |
| ICAD | Inhibitor of caspase-activated DNase |
| LDLR | Low-density lipoprotein receptor-deficient |
| LE | Lipophilic extract |
| LLC | Lipoprotein-low cholesterol |
| LPS | Lipopolysaccharide |
| iNOS | Inducible nitric oxide synthase |
| LC-MS | Liquid chromatography-mass spectrometry |
| LPS | Lipopolysaccharide |
| MAPK | Mitogen-activated protein kinase |
| MECO | Methanolic extract of C. olitorius |
| NMR | Nuclear Magnetic Resonance |
| PARP | Poly ADP-ribose polymerase |
| PEEs | Polyphenol-enriched extracts |
| PGE2 | Prostaglandin E2 |
| PSc | Polysaccharides |
| PTS | Phytosterol |
| PPARα | Peroxisome proliferator activated receptor alpha |
| QE/g | Quercetin equivalent per gram |
| ROS | Reactive oxygen species |
| SGOT | Serum glutamic oxaloacetic transaminase |
| SGPT | Serum glutamic pyruvic transaminase |
| SOD | Superoxide dismutase |
| STAT | Signal transducer and activator of transcription |
| TAG | Triacylglycerol |
| TFC | Total flavonoids content |
| T2DM | Type 2 diabetes mellitus |
References
- Beyene, B. Review on Application and Management of Medicinal Plants for the Livelihood of the Local Community. J. Resour. Dev. Manag. 2016, 22, 33–39. [Google Scholar]
- Shityakov, S.; Bigdelian, E.; Hussein, A.A.; Hussain, M.B.; Tripathi, Y.C.; Khan, M.U.; Shariati, M.A. Phytochemical and pharmacological attributes of piperine: A bioactive ingredient of black pepper. Eur. J. Med. Chem. 2019, 176, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Akomas, S.; Ijioma, S.; Emelike, C. In-vivo and In-vitro spasmolytic Effect of Ficus sur Forssk Ethanol Leaf Extract on the Gastrointestinal Tract. Br. Biotechnol. J. 2014, 4, 1182–1190. [Google Scholar] [CrossRef]
- Loumerem, M.; Alercia, A. Descriptors for jute (Corchorus olitorius L.). Genet. Resour. Crop Evol. 2016, 63, 1103–1111. [Google Scholar] [CrossRef]
- Kumari, N.; Choudhary, S.B.; Sharma, H.K.; Singh, B.K.; Kumar, A.A. Health-promoting properties of Corchorus leaves: A review. J. Herb. Med. 2019, 15, 100240. [Google Scholar] [CrossRef]
- Choudhary, S.B.; Sharma, H.K.; Karmakar, P.G.; Anil Kumar, A.; Saha, A.R.; Hazra, P.; Mahapatra, B.S. Nutritional profile of cultivated and wild jute (Corchorus) species. Aust. J. Crop Sci. 2013, 7, 1973–1982. [Google Scholar]
- Choudhary, S.B.; Sharma, H.K.; Anil Kumar, A.; Maruthi, R.T.; Karmakar, P.G. The genus Corchorus L. (Malvaceae) in India: Species distribution and ethnobotany. Genet. Resour. Crop Evol. 2017, 64, 1675–1686. [Google Scholar] [CrossRef]
- Islam, M.M. Biochemistry, Medicinal and Food values of Jute (Corchorus capsularis L. and C. olitorius L.) leaf: A Review. Int. J. Enhanc. Res. Sci. Technol. Eng. 2013, 2, 35–44. [Google Scholar]
- Steyn, N.P.; Olivier, J.; Winter, P.; Burger, S.; Nesamvuni, C. A survey of wild, green, leafy vegetables and their potential in combating micronutrient deficiencies in rural populations. S. Afr. J. Sci. 2001, 97, 276–278. [Google Scholar]
- Dansi, A.; Adjatin, A.; Adoukonou-Sagbadja, H.; Faladé, V.; Yedomonhan, H.; Odou, D.; Dossou, B. Traditional leafy vegetables and their use in the Benin Republic. Genet. Resour. Crop Evol. 2008, 55, 1239–1256. [Google Scholar] [CrossRef]
- Zakaria, Z.A.; Somchit, M.N.; Zaiton, H.; Mat Jais, A.M.; Sulaiman, M.R.; Farah, W.O.; Nazaratulmawarina, R.; Fatimah, C.A. The in vitro antibacterial activity of Corchorus olitorius extracts. Int. J. Pharmacol. 2006, 2, 213–215. [Google Scholar] [CrossRef]
- Soykut, G.; Becer, E.; Calis, I.; Yucecan, S.; Vatansever, S. Apoptotic effects of Corchorus olitorius L. leaf extracts in colon adenocarcinoma cell lines. Prog. Nutr. 2018, 20, 689–698. [Google Scholar] [CrossRef]
- Taiwo, B.J.; Taiwo, G.O.; Olubiyi, O.O.; Fatokun, A.A. Polyphenolic compounds with anti-tumour potential from Corchorus olitorius (L.) Tiliaceae, a Nigerian leaf veget. Bioorg. Med. Chem. Lett. 2016, 26, 3404–3410. [Google Scholar] [CrossRef] [PubMed]
- Li, C.J.; Huang, S.Y.; Wu, M.Y.; Chen, Y.C.; Tsang, S.F.; Chyuan, J.H.; Hsu, H.Y. Induction of apoptosis by ethanolic extract of Corchorus olitorius leaf in human hepatocellular carcinoma (HepG2) cells via a mitochondria-dependent pathway. Molecules 2012, 17, 9348–9360. [Google Scholar] [CrossRef]
- Ademiluyi, A.O.; Oboh, G.; Aragbaiye, F.P.; Oyeleye, S.I.; Ogunsuyi, O.B. Antioxidant properties and in vitro α-amylase and α-glucosidase inhibitory properties of phenolics constituents from different varieties of Corchorus spp. J. Taibah Univ. Med. Sci. 2015, 10, 278–287. [Google Scholar] [CrossRef]
- Wang, L.; Yamasaki, M.; Katsube, T.; Sun, X.; Yamasaki, Y.; Shiwaku, K. Antiobesity effect of polyphenolic compounds from molokheiya (Corchorus olitorius L.) leaves in LDL receptor-deficient mice. Eur. J. Nutr. 2011, 50, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Furumoto, T.F.; Ang, R.W.; Kazaki, K.O.; Asan, A.F.M.F.H.; Li, M.I.A.; Ondo, A.K. Antitumor Promoters in Leaves of Jute (Corchorus capsularis and Corchorus olitorius). Food Sci. Technol. Res. 2002, 8, 239–243. [Google Scholar] [CrossRef]
- Oyedele, D.J.; Asonugho, C.; Awotoye, O.O. Heavy metals in soil and accumulation by edible vegetables after phosphate fertilizer application. Electron. J. Environ. Agric. Food Chem. 2006, 5, 1446–1453. [Google Scholar]
- Velempini, P.; Riddoch, I.; Batisani, N. Seed treatments for enhancing germination of wild okra (Corchorus olitorius). Exp. Agric. 2003, 39, 441–447. [Google Scholar] [CrossRef]
- Ohtani, K.; Okai, K.; Yamashita, U.; Yuasa, I.; Misaki, A. Characterization of an Acidic Polysaccharide Isolated from the Leaves of Corchorus olitorius (Moroheiya). Biosci. Biotechnol. Biochem. 1995, 59, 378–381. [Google Scholar] [CrossRef]
- Hosen, S.M.I.; Das, D.; Kobi, R.; Chowdhury, D.U.S.; Alam, M.J.; Rudra, B.; Bakar, M.A.; Islam, S.; Rahman, Z.; Al-Forkan, M. Study of arsenic accumulation in rice and evaluation of protective effects of Chorchorus olitorius leaves against arsenic contaminated rice induced toxicities in Wistar albino rats. BMC Pharmacol. Toxicol. 2016, 17, 46. [Google Scholar] [CrossRef] [PubMed]
- Orieke, D.; Ohaeri, O.C.; Ijeh, I.I.; Ijioma, S.N. Gastrointestinal and uterine smooth muscles relaxant and anti-inflammatory effects of Corchorus olitorius leaf extract in laboratory animal models. J. Ethnopharmacol. 2020, 247, 112224. [Google Scholar] [CrossRef] [PubMed]
- Nduche, M.U.; Omosun, G.; Okwulehie, I.C. Ethnobotanical Survey of Plants Used as Remedy for Fertility Conditions in Ebonyi State of Nigeria. Sch. Acad. J. Biosiences. 2015, 3, 214–221. [Google Scholar]
- Akaneme, F.I. Identification and preliminary phytochemical analysis of herbs that can arrest threatened miscarriage in Orba and Nsukka towns of Enugu State. African J. Biotechnol. 2008, 7, 006–011. [Google Scholar] [CrossRef]
- Sadat, A.; Chakraborty, K. Jute-A biological elixir with multifaceted applications: An overview. Int. J. Res. Pharm. Sci. 2015, 6, 323–332. [Google Scholar]
- Whitlock, B.A.; Karol, K.G.; Alverson, W.S. Chloroplast DNA sequences confirm the placement of the enigmatic Oceanopapaver within Corchorus (Grewioideae: Malvaceae S.L., formerly Tiliaceae). Int. J. Plant Sci. 2003, 164, 35–41. [Google Scholar] [CrossRef]
- Ahmed, F. Nutraceutical Potential of Molokhia (Corchorus olitorius L.): A Versatile Green Leafy Vegetable. Pharmacogn. Res. 2021, 10, 24–30. [Google Scholar] [CrossRef]
- Castro, M.P.T.; Obtinalla, C.O.; Lucente, M.E. Evaluation of Jute (Corchorus olitorius) Leaves as a Sushi Wrap. Adv. Nutr. Food Sci. 2018, 3, 1–5. [Google Scholar] [CrossRef]
- Zakaria, Z.A.; Sulaiman, M.R.; Gopalan, H.K.; Abdul Ghani, Z.D.F.; Raden Nur, R.N.S.; Mat Jais, A.M.; Abdullah, F.C. Antinociceptive and anti-inflammatory properties of Corchorus capsularis leaves chloroform extract in experimental animalmodels. Yakugaku Zasshi. 2007, 127, 359–365. [Google Scholar] [CrossRef]
- Zakaria, Z.A.; Kumar, G.H.; Nor, R.N.S.R.M.; Sulaiman, M.R.; Fatimah, C.A.; Jais, A.M.M.; Somchit, M.N.; Ismail, M.S. Antinociceptive, anti-inflammatory and antipyretic properties of an aqueous extract of Corchorus capsularis leaves in experimental animal models. Pharm. Biol. 2009, 47, 104–110. [Google Scholar] [CrossRef]
- Zeghichi, S.; Kallithraka, S.; Simopoulos, A.P. Nutritional composition of molokhia (Corchorus olitorius) and stamnagathi (Cichorium spinosum). World Rev. Nutr. Diet. 2003, 91, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Osaronowen, M.; Udo, E.; Oricha, S. Anti-diabetic Activity of Ethanolic Seed Extract of Corchorus olitorius. IJSBAR 2013, 12, 8–22. [Google Scholar]
- Adebo, H.O.; Ahoton, L.E.; Quenum, F.J.B.; Adoukonou-Sagbadja, H.; Bello, D.O.; Chrysostome, C.A.A.M. Ethnobotanical Knowledge of Jute (Corchorus olitorius L.) in Benin. European J. Med. Plants. 2018, 26, 1–11. [Google Scholar] [CrossRef]
- Adefegha, S.A.; Oboh, G. Inhibition of key enzymes linked to type 2 diabetes and sodium nitroprusside-induced lipid peroxidation in rat pancreas by water extractable phytochemicals from some tropical spices. Pharm. Biol. 2012, 50, 857–865. [Google Scholar] [CrossRef] [PubMed]
- Al-snafi, A.E. The Constituents and Pharmacology of Corchorus aestuans-A Review. Pharm. Chem. J. 2016, 3, 208–214. [Google Scholar]
- Yoshikawa, M.; Shimada, H.; Saka, M.; Yoshizumi, S. Medicinal Foodstuffs. V. 1 Moroheiya. (1): Absolute Stereostructures of Corchoionosides A, B, and C, Histamine Release Inhibitors from the Leaves of Vietnamese Corchorus olitorius L. (Tiliaceae). Chem. Pharm. Bull. 1997, 45, 464–469. [Google Scholar] [CrossRef] [PubMed]
- Sonibare, M.A.; Moody, J.O.; Adesanya, E.O.; Momordica, L.; Seem, P.B.; Ocimum, L. Use of medicinal plants for the treatment of measles in Nigeria. J. Ethnopharmacol. 2009, 122, 268–272. [Google Scholar] [CrossRef] [PubMed]
- Hadid, A.F.A.; El-shinawy, M.; El-beltagy, A.S.; Medany, M. Studies on the production of off-season Jew’s Mallow “Melokhia” in Egypt. Egypt. J. Hort. 1994, 21, 187–193. [Google Scholar]
- Idirs, S.; Yisa, J.; Ndamitso, M.M. Nutritional composition of Corchorus olitorius leaves. J. Anim. Prod. Adv. 2009, 5, 2. [Google Scholar] [CrossRef]
- Biswas, A.; Dey, S.; Li, D.; Liu, Y.; Zhang, J.; Huang, S.; Pan, G.; Deng, Y. Comparison of Phytochemical Profile, Mineral Content, and in Vitro Antioxidant Activities of Corchorus capsularis and Corchorus olitorius Leaf Extracts from Different Populations. J. Food Qual. 2020. [Google Scholar] [CrossRef]
- Ben Yakoub, A.R.; Abdehedi, O.; Jridi, M.; Elfalleh, W.; Bkhairia, I.; Nasri, M.; Ferchichi, A. Bioactive polysaccharides and their soluble fraction from Tossa jute (Corchorus olitorius L.) leaves. Food Biosci. 2020, 37, 100741. [Google Scholar] [CrossRef]
- Gupta, S.; Prakash, J. Studies on Indian green leafy vegetables for their antioxidant activity. Plant Foods Hum. Nutr. 2009, 64, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Tabassum, T. Extraction, Identification and Estimation of Caffeine and Catechin from Corchorus capsularis Leaves Extract. Ph.D. Thesis, East West University, Dhaka, Bangladesh, 2014. [Google Scholar]
- Samuel, F.O.; Ayoola, P.B.; Ejoh, S.I. Nutrient, antinutrient and sensory evaluation of Corchorus olitorius fruit. Ife J. Agric. 2020, 32, 13–20. [Google Scholar]
- Ragasa, C.Y.; Vivar, J.L.A.; Tan, M.C.S.; Shen, C.C. Chemical constituents of Corchorus olitorius L. Int. J. Pharmacognf. Phytochem. Res. 2016, 8, 2085–2089. [Google Scholar]
- Musa, A.; Ogbadoyi, E.O. Effect of Cooking and Sun Drying On Micronutrients, Antinutrients an Toxic Substances in Corchorus olitorius (Jute Mallow). J. Nutr. Food Sci. 2012, 2, 1000140. [Google Scholar] [CrossRef]
- Azuma, K.; Nakayama, M.; Koshioka, M.; Ippoushi, K.; Yamaguchi, Y.; Kohata, K.; Yamauchi, Y.; Ito, H.; Higashio, H. Phenolic antioxidants from the leaves of Corchorus olitorius L. J. Agric. Food Chem. 1999, 47, 3963–3966. [Google Scholar] [CrossRef]
- Khan, M.S.Y.; Bano, S.; Javed, K.; Asad Mueed, M. A comprehensive review on the chemistry and pharmacology of Corchorus species - A source of cardiac glycosides, triterpenoids, ionones, flavonoids, coumarins, steroids and some other compounds. J. Sci. Ind. Res. (India) 2006, 65, 283–298. [Google Scholar]
- Guzzetti, L.; Panzeri, D.; Ulaszewska, M.; Sacco, G.; Forcella, M.; Fusi, P.; Tommasi, N.; Fiorini, A.; Campone, L.; Labra, M. Assessment of Dietary Bioactive Phenolic Compounds and Agricultural Sustainability of an African Leafy Vegetable Corchorus olitorius L. Front. Nutr. 2021, 8, 667812. [Google Scholar] [CrossRef]
- Do, M.H.; Lee, H.H.L.; Kim, Y.; Lee, H.B.; Lee, E.; Park, J.H.; Park, H.Y. Corchorus olitorius L. ameliorates alcoholic liver disease by regulating gut-liver axis. J. Funct. Foods 2021, 85, 104648. [Google Scholar] [CrossRef]
- Meyer, A.S.; Donovan, J.L.; Pearson, D.A.; Waterhouse, A.L.; Frankel, E.N. Fruit Hydroxycinnamic Acids Inhibit Human Low-Density Lipoprotein Oxidation in Vitro. J. Agric. Food Chem. 1998, 46, 1783–1787. [Google Scholar] [CrossRef]
- Price, K.R.; Rhodes, M.J.C. Analysis of the major flavonol glycosides present in four varieties of onion (Allium cepa) and changes in composition resulting from autolysis. J. Sci. Food Agric. 1997, 74, 331–339. [Google Scholar] [CrossRef]
- Ben Yakoub, A.R.; Abdehedi, O.; Jridi, M.; Elfalleh, W.; Nasri, M.; Ferchichi, A. Flavonoids, phenols, antioxidant, and antimicrobial activities in various extracts from Tossa jute leave (Corchorus olitorus L.). Ind. Crops Prod. 2018, 118, 206–213. [Google Scholar] [CrossRef]
- Ali, M.M.; Ahmed, K.S.; Hossain, H.; Roy, B.; Rokeya, B.; Rahman, M.T.; Jahan, I.A.; Rahman, M.M. Total antioxidant capacity and profiling of polyphenolic compounds in jute leaves by hplc-dad. Food Res. 2021, 5, 343–348. [Google Scholar] [CrossRef]
- Chhikara, N.; Devi, H.R.; Jaglan, S.; Sharma, P.; Gupta, P.; Panghal, A. Bioactive compounds, food applications and health benefits of Parkia speciosa (stinky beans): A review. Agric. Food Secur. 2018, 7, 46. [Google Scholar] [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5. [Google Scholar] [CrossRef]
- Handoussa, H.; Hanafi, R.; Eddiasty, I.; El-Gendy, M.; El Khatib, A.; Linscheid, M.; Mahran, L.; Ayoub, N. Anti-inflammatory and cytotoxic activities of dietary phenolics isolated from Corchorus olitorius and Vitis vinifera. J. Funct. Foods. 2013, 5, 1204–1216. [Google Scholar] [CrossRef]
- Ramadevi, D. Phytochemical examination of Corchorus olitorius leaves. Int. J. Curr. Pharm. Rev. Res. 2013, 4, 42–46. [Google Scholar]
- Mukherjee, K.K.; Mitra, S.K.; Ganguly, S.N. A new coumarin from the seeds of jute (Corchorus olitorius L.). Nat. Prod. Sci. 1998, 4, 51–52. [Google Scholar]
- Nakamura, T.; Goda, Y.; Sakai, S.; Kondo, K.; Akiyama, H.; Toyoda, M. Cardenolide glycosides from seeds of Corchorus olitorius. Phytochemistry 1998, 49, 2097–2101. [Google Scholar] [CrossRef]
- Mahato, S.B.; Sahu, N.P.; Roy, S.K.; Pramanik, B.N. Cardiac Glycosides from Corchorus olitorius. J. Chem. Soc. 1998, 2065–2068. [Google Scholar] [CrossRef]
- Manzoor-i-Khuda, M.; Habermehl, G. Chemical Constituents of Corchorus capsularis and C.olitorius (Jute Plant), Part IV* Isolation of Corosolic Acid, Ursolic Acid and Oxo-corosin and Correlation of Corosin with Tormentic Acid. Z. Fur Nat. Sect. B J. Chem. Sci. 1979, 34, 1320–1325. [Google Scholar] [CrossRef]
- Chen, S.; Wu, B.; Fang, J.; Liu, Y.; Zhang, H.; Fang, L. Analysis of flavonoids from lotus (Nelumbo nucifera) leaves using high performance liquid chromatography/photodiode array detector tandem electrospray ionization mass spectrometry and an extraction method optimized by orthogonal design. J. Chromatogr. A. 2012, 1227, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Quader, M.A.; Ahmed, M.D.; Hasan, C.M.; Waterman, P.G. Capsugenin-30-O-β-glucopyranoside: A new glucoside from the leaves of Corchorous capsularis. J. Nat. Prod. 1987, 50, 479–481. [Google Scholar] [CrossRef]
- Hassan, A.; Mekhael, M.; Hanna, A.; Simon, A.; Tóth, G.; Duddeck, H. Phytochemical investigation of Corchorus olitorius and Corchorus capsularis (Family Tiliaceae) that grow in Egypt. Egypt. Pharm. J. 2019, 18, 123–134. [Google Scholar] [CrossRef]
- Hasan, C.M.; Islam, A.; Ahmed, M.; Ahmed, M.D.; Waterman, P.G. Capsugenin, a dammarane triterpene from Corchorus capsularis. Phytochemistry 1984, 23, 2583–2587. [Google Scholar] [CrossRef]
- Quader, M.A.; Gray, A.I.; Layud, C.; Massiot, G.; Hasan, C.M.; Ahmed, M.D. Capsugenin-25,30-O-β-diglucopyranoside: A new glycoside from the leaves of Corchorus capsularis. J. Nat. Prod. 1990, 53, 527–530. [Google Scholar] [CrossRef]
- Driss, D.; Kaoubaa, M.; Ben Mansour, R.; Kallel, F.; eddine Abdelmalek, B.; Ellouz Chaabouni, S. Antioxidant, Antimutagenic and Cytotoxic Properties of Essential Oil from Corchorus olitorius L. Flowers and Leaf. Free Radic. Antioxid. 2016, 6, 34–43. [Google Scholar] [CrossRef]
- Yoshikawa, M.; Murakami, T.; Shimada, H.; Yoshizumi, S. Medicinal Foodstuffs. XIV. On the Bioactive Constituents of Moroheiya. (2): New Fatty Acids, Corchorifatty Acids A, B, C, D, E, and F, from the Leaves of Corchorus olitorius L. (Tiliaceae): Structures and Inhibitory Effect on NO Production. Chem. Pharm. Bull. 1998, 46, 1008–1014. [Google Scholar] [CrossRef]
- Al-Yousef, H.M.; Amina, M.; Ahamad, S.R. Comparative study on the chemical composition of Corchorus olitorius leaf and stem dry oils. Biomed. Res. 2017, 28, 4581–4587. [Google Scholar]
- Mahmoud, A.S.; Thao, N.; Mario, A. Corchorus olitorius Linn: A Rich Source of Ω3-Fatty Acids. Pharm. Anal. Acta 2016, 7, 6–8. [Google Scholar] [CrossRef]
- Siriamornpun, S.; Li, D.; Yang, L.; Kanchanamayoon, W.; Suttajit, M. Thai jute seed oil: A potential polyunsaturated fatty acid source. Songklanakarin J. Sci. Technol. 2006, 28, 11–15. [Google Scholar]
- Simopoulos, A.P. Omega-3 fatty acids in health and disease and in growth and development. Am. J. Clin. Nutr. 1991, 54, 438–463. [Google Scholar] [CrossRef] [PubMed]
- Dale, A.P.; Meara, M.L. The component fatty acids and glycerides of coconut oils. J. Sci. Food Agric. 1952, 6, 237–240. [Google Scholar] [CrossRef]
- Hasan, H.T.; Kadhim, E.J. Phytochemical investigation of leaves and seeds of Corchorus olitorius l. Cultivated in iraq. Asian J. Pharm. Clin. Res. 2018, 11, 408–417. [Google Scholar] [CrossRef]
- Oikawa, T. NII-Electronic Library Service. Chem. Pharm. Bull. 2002, 2091. [Google Scholar]
- Yamamoto, K.; Asakawa, H.; Tokunaga, K.; Watanabe, H.; Matsuo, N.; Tokimitsu, I.; Yagi, N. Long-term ingestion of dietary diacylglycerol lowers serum triacylglycerol in type II diabetic patients with hypertriglyceridemia. J. Nutr. 2001, 131, 3204–3207. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, H.; Watanabe, H.; Onizawa, K.; Nagao, T.; Gotoh, N.; Yasukawa, T.; Tsushima, R.; Shimasaki, H.; Itakura, H. Double-blind controlled study on the effects of dietary diacylglycerol on postprandial serum and chylomicron triacylglycerol responses in healthy humans. J. Am. Coll. Nutr. 2000, 19, 789–796. [Google Scholar] [CrossRef]
- Negm, S.; El Shabrawy, O.; Arbid, M.; Radwan, A.S. Toxicological study of the different organs of Corchorus olitorius L. plant with special reference to their cardiac glycosides content. Z. Ern&Hrungswiss. 1980, 19, 28–32. [Google Scholar]
- Pramanik, P.K.; Chakraborti, S.; Bagchi, A.; Chakraborti, T. Bioassay-based Corchorus capsularis L. leaf-derived β-sitosterol exerts antileishmanial effects against Leishmania donovani by targeting trypanothione reductase. Sci. Rep. 2020, 10, 20440. [Google Scholar] [CrossRef]
- Ramadevi, D.; Lakshmi Sirisha, K.; Dhorababu, D.; Sampath Kumar, G.V. Phytochemical Examination of Corchorus capsularis Roots. Int. J. Pharmacogn. Phytochem. Res. 2014, 6, 137–140. [Google Scholar]
- Furumoto, T.; Wang, R.; Okazaki, K.; Hasan, A.; Ali, M.; Kondo, A.; Fukui, H. Antitumor Promoters in Leaves of Jute (Corchorus capsularis and Corchorus olitorius). Food Sci. Technol. Res. 2002, 8, 239–243. [Google Scholar] [CrossRef]
- Takahashi, K.; Tanaka, Y.; Hosoi, S.; Hidaka, T.; Usui, S. Identification and Determination of Unknown Chemicals in Crops by Dual-Column GC System Equipped with MS and ECD. J Food Hyg Soc Japan. 2002, 43, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Zeid, A.H.S.A. Stress metabolites from Corchorus olitorius L. leaves in response to certain stress agents. Food Chem. 2002, 76, 187–195. [Google Scholar] [CrossRef]
- Ogawa, M.; Hayasi, K.; Tomimori, S.; Konishi, N.; Nakayama, O. Contents of strophanthidin glycosides and digitoxigenin glycosides in”moroheiya” (Corchorus olitorius L.) and its products. J. Jpn. Soc. Food Sci. Technol. (Japan) 2002, 49, 282–287. [Google Scholar] [CrossRef]
- Mazumder, U.K.; Gupta, M.; Pal, D.; Bhattacharya, S. Chemical and toxicological evaluation of methanol extract of Cuscuta reflexa Roxb. stem and Corchorus olitorius Linn. seed on hematological parameters and hepatorenal functions in mice. Acta Pol. Pharm.-Drug Res. 2003, 60, 317–323. [Google Scholar]
- Matsufuji, H.; Sakai, S.; Chino, M.; Goda, Y.; Toyoda, M.; Takeda, M. Relationship between cardiac glycoside contents and color of Corchorus olitorius seeds. J. Health Sci. 2001, 47, 89–93. [Google Scholar] [CrossRef][Green Version]
- Al-Snafi, A.E. The contents and pharmacological importance of Corchorus capsularis-A review. IOSR J. 2016, 6, 58–63. [Google Scholar]
- Goda, Y.; Sakai, S.; Nakamura, T.; Akiyama, H.; Toyoda, M. Identification and Analyses of Main Cardiac Glycosides in Corchorus olitorius Seeds and Their Acute Oral Toxicity in mice. F. Hyg. Saf. Sci. 1377, 68–70. [Google Scholar] [CrossRef]
- Handoussa, H.; Hanafi, R.S.; El-Khatib, A.H.; Linscheid, M.W.; Mahran, L.G.; Ayoub, N.A. Computer-assisted HPLC method development using DryLab for determination of major phenolic components in Corchorus olitorius and Vitis vinifera by using HPLC-PDA-ESI-TOF- MSn. Res. Rev. J. Bot. Sci. 2017, 6, 9–16. [Google Scholar]
- Darcansoy İşeri, Ö.; Yurtcu, E.; Sahin, F.I.; Haberal, M. Corchorus olitorius (jute) extract induced cytotoxicity and genotoxicity on human multiple myeloma cells (ARH-77). Pharm. Biol. 2013, 51, 766–770. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M.; Snader, K.M. Natural products as sources of new drugs over the period 1981-2002. J. Nat. Prod. 2003, 66, 1022–1037. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.; Pieters, R.; Abdel-Aziem, S.; Van der Walt, A.; Bezuidenhout, C.; Giesy, J.; Abdel-Wahhab, M. Aqueous extract of Corchorus olitorius decreases cytotoxicity of aflatoxin B1 and fumonisin B1 in H4IIE-luc cells. Hepatoma Res. 2015, 1, 75. [Google Scholar] [CrossRef]
- Tosoc, J.P.S.; Nuñeza, O.M.; Sudha, T.; Darwish, N.H.E.; Mousa, S.A. Anticancer effects of the Corchorus olitorius aqueous extract and its bioactive compounds on human cancer cell lines. Molecules 2021, 26, 6033. [Google Scholar] [CrossRef] [PubMed]
- Choi, D.W.; Lim, M.S.; Lee, J.W.; Chun, W.; Lee, S.H.; Nam, Y.H.; Park, J.M.; Choi, D.H.; Kang, C.D.; Lee, S.J.; et al. The cytotoxicity of kahweol in HT-29 human colorectal cancer cells is mediated by apoptosis and suppression of heat shock protein 70 expression. Biomol. Ther. 2015, 23, 128. [Google Scholar] [CrossRef] [PubMed]
- Rume, J.M. Phytochemical, Antimicrobial and Biological Investigations of Methanolic Extract of Leavrs of Corchorus Capsularis. Master Thesis, East West University, Dhaka, Bangladesh, 2010. [Google Scholar]
- Malfertheiner, P.; Chan, F.K.; McColl, K.E. Peptic ulcer disease. Lancet 2009, 374, 1449–1461. [Google Scholar]
- Mezui, C.; Amang, A.P.; Nkenfou, C.; Sando, Z.; Betou, D.; Moulioum, H.T.P.V. Anti-Ulcer and Antioxidant Activities of the Leaf Aqueous Extract of Corchorus olitorius (Tiliaceae) in Rats. Int. J. Phytopharm. 2016, 7, 17–28. [Google Scholar]
- Devaraj, V.C.; Asad, M.; Prasad, S. Effect of leaves and fruits of Moringa oleifera on gastric and duodenal ulcers. Pharm. Biol. 2007, 45, 332–338. [Google Scholar] [CrossRef]
- Al-Batran, R.; Al-Bayaty, F.; Ameen Abdulla, M.; Jamil Al-Obaidi, M.M.; Hajrezaei, M.; Hassandarvish, P.; Fouad, M.; Golbabapour, S.; Talaee, S. Gastroprotective effects of Corchorus olitorius leaf extract against ethanol-induced gastric mucosal hemorrhagic lesions in rats. J. Gastroenterol. Hepatol. 2013, 28, 1321–1329. [Google Scholar] [CrossRef]
- Zakaria, Z.A.; Safarul, M.; Valsala, R.; Sulaiman, M.R.; Fatimah, C.A.; Somchit, M.N.; Mat Jais, A.M. The influences of temperature and naloxone on the antinociceptive activity of Corchorus olitorius L. in mice. Naunyn. Schmiedebergs. Arch. Pharmacol. 2005, 372, 55–62. [Google Scholar] [CrossRef]
- Oyewole, A.L.; Owoyele, B.V.; Alimi, M.L.; Sanni, S.A.; Oyeleke, S.A. Anti-inflammatory and antipyretic properties of Corchorus olitorius aqueous root extract in Wistar rats. J. Basic Clin. Physiol. Pharmacol. 2015, 26, 363–368. [Google Scholar] [CrossRef]
- Yan, Y.Y.; Wang, Y.W.; Chen, S.L.; Zhuang, S.R.; Wang, C.K. Anti-inflammatory effects of phenolic crude extracts from five fractions of Corchorus olitorius L. Food Chem. 2013, 138, 1008–1014. [Google Scholar] [CrossRef] [PubMed]
- Ijioma, N.; Madubuike, K.; Nwankudu, O.; Nwosu, C.; Emelike, C. Uterine Relaxation Potential of Ethanol Leaf Extract of Moringa oleifera Lam. VIA the Muscarinic Receptor Pathway. Br. J. Pharm. Res. 2014, 4, 2455–2462. [Google Scholar] [CrossRef]
- Parvin, S.; Marzan, M.; Rahman, S.; Das, A.K.; Haque, S.; Rahmatullah, M. Preliminary phytochemical screening, antihyperglycemic, analgesic and toxicity studies on methanolic extract of aerial parts of Corchorus olitorius L. J. Appl. Pharm. Sci. 2015, 5, 068–071. [Google Scholar] [CrossRef]
- Borokini, T.I.; Lawal, I.O. Traditional medicine practices among the Yoruba people of Nigeria: A historical perspective. J. Med. Plants Stud. 2014, 2, 20–33. [Google Scholar]
- Sharma, S.; Ali, A.; Ali, J.; Sahni, J.K.; Baboota, S. Rutin: Therapeutic potential and recent advances in drug delivery. Expert Opin. Investig. Drugs 2013, 22, 1063–1079. [Google Scholar] [CrossRef]
- Nisha, P.; Mini, S. In vitro antioxidant and antiglycation properties of methanol extract and its different solvent fractions of Musa paradisiaca L. (Cv.Nendran) inflorescence. Int. J. Food Prop. 2014, 17, 399–409. [Google Scholar] [CrossRef]
- Mibei, E.K.; Ojijo, N.K.O.; Karanja, S.M.; Kinyua, J.K. Phytochemical and antioxidant analysis of methanolic extracts of four african indegenous leafy vegetables. Ann. Food Sci. Technol. 2012, 13, 37–42. [Google Scholar]
- Sadat, A.; Hore, M.; Chakraborty, K.; Roy, S. Phytochemical Analysis and Antioxidant Activity of Methanolic Extract of Leaves of Corchorus olitorius. Int. J. Curr. Pharm. Res. 2017, 9, 59–63. [Google Scholar] [CrossRef]
- Oboh, G.; Raddatz, H.; Henle, T. Characterization of the antioxidant properties of hydrophilic and lipophilic extracts of Jute (Corchorus olitorius) leaf. Int. J. Food Sci. Nutr. 2009, 60, 124–134. [Google Scholar] [CrossRef]
- Kaboré, S.; Touré, A.; Kouamé, B.K.; Bamba, A.E.; Kabran, A.F.; Kadio, B.D.; Konan, D.J.; Attioua, K.B.; Coulibaly, W.K.; Kablan, A.L.; et al. Phytochemical screening and antioxidant activity of leaves of Amaranthus hybridus L., Corchorus olitorius L and Hibiscus sabdariffa L. grown in northen of Côte d’Ivoire. GSC Biol. Pharm. Sci. 2021, 16, 182–189. [Google Scholar] [CrossRef]
- Morsy, N.E.; Rayan, A.M.; Youssef, K.M. Physico Chemical Properties, Antioxidant Activity, Phytochemicals and Sensory Evaluation of Rice-Based Extrudates Containing Dried Corchorus olitorius L. Leaves. J. Food Process. Technol. 2015, 6, 1. [Google Scholar] [CrossRef]
- Hussien, N.M.; Labib, S.E.; Hefnawy, H.T.M. Phytochemical studies and antioxidant activity of leaves extracts of Corchorus olitorius L. (Molokhia). Zagazig J. Agric. Res. 2017, 44, 2231–2239. [Google Scholar] [CrossRef]
- Chigurupati, S.; Aladhadh, H.S.; Alhowail, A.; Selvarajan, K.K.; Bhatia, S. Phytochemical composition, antioxidant and antidiabetic potential of methanolic extract from Corchorus olitorius Linn. Grown in Saudi Arabia. Med. Plants 2020, 12, 71–76. [Google Scholar] [CrossRef]
- Airaodion, A.I.; Ogbuagu, E.O.; Ewa, O.; Ogbuagu, U.; Awosanya, O.O.; Adekale, O.A. Increase in Tannin Content of Some Selected Nigerian Vegetables during Blanching and Juicing. Asian J. Biochem. Genet. Mol. Biol. 2019, 2, 1–10. [Google Scholar] [CrossRef]
- Mesli, F.; Ghalem, M.; Daoud, I.; Ghalem, S. Potential inhibitors of angiotensin converting enzyme 2 receptor of COVID-19 by Corchorus olitorius Linn using docking, molecular dynamics, conceptual DFT investigation and pharmacophore mapping. J. Biomol. Struct. Dyn. 2021, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Suryanarayana, P.; Anil Kumar, P.; Saraswat, M.; Petrash, J.M.; Reddy, G.B. Inhibition of aldose reductase by tannoid principles of Emblica officinalis: Implications for the prevention of sugar cataract. Mol. Vis. 2004, 10, 148–154. [Google Scholar]
- Oboh, G.; Ademiluyi, A.O.; Akinyemi, A.J.; Henle, T.; Saliu, J.A.; Schwarzenbolz, U. Inhibitory effect of polyphenol-rich extracts of jute leaf (Corchorus olitorius) on key enzyme linked to type 2 diabetes (α-amylase and α-glucosidase) and hypertension (angiotensin I converting) in vitro. J. Funct. Foods 2012, 4, 450–458. [Google Scholar] [CrossRef]
- Oboh, G.; Akinyemi, A.J.; Ademiluyi, A.O.; Adefegha, S.A. Inhibitory effects of aqueous extracts of two varieties of ginger on some key enzymes linked to type-2 diabetes in vitro. Journal of Food and Nutrition Research. J. Food Nutr. Res. 2010, 49, 14–20. [Google Scholar]
- Kwon, Y.I.; Apostolidis, E.; Kim, Y.C.; Shetty, K. Health benefits of traditional corn, beans, and pumpkin: In vitro studies for hyperglycemia and hypertension management. J. Med. Food. 2007, 10, 266–275. [Google Scholar] [CrossRef]
- Olusanya, A.R.; Ifeoluwa, B.S. Antidiabetic and safety properties of ethanolic leaf extract of Corchorus olitorius in alloxan-induced diabetic rats. Diabetes Food Plan. 2018, 11, 57. [Google Scholar]
- Ozioma, O.K.; Solomon, O.A.; Ocholi, E.D.; Somadina, C.R. Platelet Aggregation Effect of Corchorus olitorus Extract as an Index of Managing Blood Clotting Disorders. IOSR J. Dent. Med. Sci. 2014, 13, 58–60. [Google Scholar] [CrossRef]
- Gomaa, A.A.; El-Sers, D.A.; Al-Zokeim, N.I.; Gomaa, M.A. Amelioration of experimental metabolic syndrome induced in rats by orlistat and Corchorus olitorius leaf extract; role of adipo/cytokines. J. Pharm. Pharmacol. 2019, 71, 281–291. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Hu, X.; Ai, W.; Zhang, F.; Yang, K.; Wang, L.; Zhu, X.; Gao, P.; Shu, G.; Jiang, Q.; et al. Phytol increases adipocyte number and glucose tolerance through activation of PI3K/Akt signaling pathway in mice fed high-fat and high-fructose diet. Biochem. Biophys. Res. Commun. 2017, 489, 432–438. [Google Scholar] [CrossRef] [PubMed]
- Mercan, N.; Toros, P.; Söyler, G.; Hanoglu, A.; Kükner, A. Effects of Corchorus olitorius and protacatechuic acid on diabetic rat testis tissue. Int. J. Morphol. 2020, 38, 1330–1335. [Google Scholar] [CrossRef]
- Lee, H.B.; Oh, M.J.; Do, M.H.; Kim, Y.S.; Park, H.Y. Molokhia leaf extract prevents gut inflammation and obesity. J. Ethnopharmacol. 2020, 257, 112866. [Google Scholar] [CrossRef]
- Abdallah, H.M.I.; Abdel Jaleel, G.A.; Mohammed, H.S.; Mahmoud, S.S.; Yassin, N.A.; Gamal El Din, A.A.; Shaffie, N.M.; Bassyouni, F.A. Phytochemical screening, gas chromatography-mass spectrometry analysis, and antidiabetic effects of Corchorus olitorius leaves in rats. Open Access Maced. J. Med. Sci. 2020, 8, 385–394. [Google Scholar] [CrossRef]
- Gupta, A.; Pandey, A.K. Plant Secondary Metabolites With Hepatoprotective Efficacy. In Nutraceuticals and Natural Product Pharmaceuticals; Academic Press: London, UK, 2019; ISBN 9780128164501. [Google Scholar]
- Saliu, J.A.; Ademiluyi, A.O.; Boligon, A.A.; Oboh, G.; Schetinger, M.R.C.; Rocha, J.B.T. Dietary supplementation of jute leaf (Corchorus olitorius) modulates hepatic delta-aminolevulinic acid dehydratase (δ-ALAD) activity and oxidative status in high-fat fed/low streptozotocin-induced diabetic rats. J. Food Biochem. 2019, 43, e12949. [Google Scholar] [CrossRef]
- Innami, S.; Tabata, K.; Shimizu, J.; Kusunoki, K.; Ishida, H.; Matsuguma, M.; Wada, M.; Sugiyama, N.; Kondo, M. Dried green leaf powders of Jew’s mellow (Corchorus), persimmon (Diosphyros kaki) and sweet potato (Ipomoea batatas poir) lower hepatic cholesterol concentration and increase fecal bile acid excretion in rats fed a cholesterol-free diet. Plant Foods Hum. Nutr. 1998, 52, 55–66. [Google Scholar] [CrossRef]
- Innami, S.; Nakamura, K.; Tabata, K.; Wada, M.; Takita, T. Water-soluble viscous substance of Jew’s mellow leaves lowers serum and liver cholesterol concentrations and increases fecal steroid excretion in rats fed a high cholesterol diet. J. Nutr. Sci. Vitaminol. 1995, 41, 465–475. [Google Scholar] [CrossRef]
- Gupta, M.; Mazumder, U.K.; Pal, D.; Bhattacharya, S.; Chakrabarty, S. Studies on brain biogenic amines in methanolic extract of Cuscuta reflexa Roxb. and Corchorus olitorius linn. seed treated mice. Acta Pol. Pharm. Drug Res. 2003, 60, 207–210. [Google Scholar]
- Oyedeji, K.O.; Bolarinwa, A.F.; Akinbode, A.A. Effect of Corchorus olitorius extract on reproductive functions in male albino rats. Int. J. Pharm. Pharm. Sci. 2013, 5, 427–431. [Google Scholar]
- Haridy, L.A.M.; Shakerali, S.; Alghamdi, R.K. Protective Role of Corchorus olitorius L. Leaves Extract against Experimentally-induced Hepatotoxicity. 2020. Available online: https://eijppr.com/IHPNOqf (accessed on 30 May 2022).
- Das, A.K.; Sahu, R.; Dua, T.K.; Bag, S.; Gangopadhyay, M.; Sinha, M.K.; Dewanjee, S. Arsenic-induced myocardial injury: Protective role of Corchorus olitorius leaves. Food Chem. Toxicol. 2010, 48, 1210–1217. [Google Scholar] [CrossRef] [PubMed]
- Colizzi, C. The protective effects of polyphenols on Alzheimer’s disease: A systematic review. Alzheimer’s Dement. Transl. Res. Clin. Interv. 2019, 5, 184–196. [Google Scholar] [CrossRef] [PubMed]
- Wagdy, R.; Abdelkader, R.M.; El-Khatib, A.H.; Linscheid, M.W.; Hamdi, N.; Handoussa, H. Neuromodulatory Activity of Dietary Phenolics Derived from Corchorus olitorius L. J. Food Sci. 2019, 84, 1012–1022. [Google Scholar] [CrossRef]
- Gay, N.H.; Phopin, K.; Suwanjang, W.; Songtawee, N.; Ruankham, W. Neuroprotective Effects of Phenolic and Carboxylic Acids on Oxidative Stress-Induced Toxicity in Human Neuroblastoma SH-SY5Y Cells. Neurochem. Res. 2018, 43, 619–639. [Google Scholar] [CrossRef]
- Gray, N.E.; Morré, J.; Kelley, J.; Maier, C.S.; Stevens, J.F.; Quinn, J.F.; Soumyanath, A. Caffeoylquinic acids in Centella asiatica protect against amyloid-β toxicity. J. Alzheimer’s Dis. 2014, 40, 359–373. [Google Scholar] [CrossRef]
- Putteeraj, M.; Lim, W.L.; Teoh, S.L.; Yahaya, M.F. Flavonoids and its Neuroprotective Effects on Brain Ischemia and Neurodegenerative Diseases. Curr. Drug Targets. 2018, 19, 1710–1720. [Google Scholar] [CrossRef]
- Gupta, M.; Mazumder, U.K.; Pal, D.K.; Bhattacharya, S. Onset of puberty and ovarian steroidogenesis following adminstration of methanolic extract of Cuscuta reflexa Roxb. stem and Corchorus olitorius Linn. seed in mice. J. Ethnopharmacol. 2003, 89, 55–59. [Google Scholar] [CrossRef]
- Yadav, S.B.; Tripathi, V.; Singh, R.K.; Pandey, H.P. Antioxidant activity of Cuscuta reflexa stems. Indian J. Pharm. Sci. 2000, 62, 477–478. [Google Scholar]
- Pal, D.; Gupta, M.; Mazumder, U. Effects of methanol extracts of Cuscuta reflexa Roxb. stem and Corchorus olitorius Linn. seed on male reproductive system of mice. Orient. Pharm. Exp. Med. 2009, 9, 49–57. [Google Scholar] [CrossRef]
- Yokoyama, S.; Hiramoto, K.; Fujikawa, T.; Kondo, H.; Konishi, N.; Sudo, S.; Iwashima, M.; Ooi, K. Skin Hydrating Effects of Corchorus olitorius; Extract in a Mouse Model of Atopic Dermatitis. J. Cosmet. Dermatological Sci. Appl. 2014, 4, 42341. [Google Scholar] [CrossRef]
- McCusker, M.M.; Grant-Kels, J.M. Healing fats of the skin: The structural and immunologic roles of the Ω-6 and Ω-3 fatty acids. Clin. Dermatol. 2010, 28, 440–451. [Google Scholar] [CrossRef] [PubMed]
- Yen, C.H.; Dai, Y.S.; Yang, Y.H.; Wang, L.C.; Lee, J.H.; Chiang, B.L. Linoleic acid metabolite levels and transepidermal water loss in children with atopic dermatitis. Ann. Allergy Asthma Immunol. 2008, 100, 66–73. [Google Scholar] [CrossRef]
- Vya, B.; Boye, A.; Quansah, N. Antioxidant and wound healing studies on the extracts of Corchorus olitorius leaf. Sci. Res. Rev. J. 2013, 1, 67–73. [Google Scholar]
- Adegoke, A.A.; Adebayo-Tayo, B.C. Phytochemical composition and antimicrobial effects of C. orchorous olitorius leaf extracts on four bacterial isolates. J. Med. Plants Res. 2009, 3, 155–159. [Google Scholar]
- Senu, A.J.; Olusola, E.M.; Afolashade, O.O.; Biliaminu; Ajoke, S. The in-vitro effect of Corchorus olitorius (Linn.) on the antibacterial activities of five antibiotics. Glob. Res. J. Microbiol. 2012, 2, 113–117. [Google Scholar]
- Abir, R.R.; Marjia, M.; Rakhi, N.N.; Saha, O.; Hossain, M.A.; Rahaman, M.M. In Vitro Comparative Analysis of Antibacterial Activity of Different Fractions of Corchorus capsularis and Corchorus olitorius Leaves Extracts. Bangladesh J. Microbiol. 2020, 36, 69–73. [Google Scholar] [CrossRef]
- Sathiamoorthy, B.; Gupta, P.; Kumar, M.; Chaturvedi, A.K.; Shukla, P.K.; Maurya, R. New antifungal flavonoid glycoside from Vitex negundo. Bioorganic Med. Chem. Lett. 2007, 17, 239–242. [Google Scholar] [CrossRef]
- Elangovan, A.; Dhanasekaran, S.; Anandan, A.; Krishnappa, K.; Gokulakrishnan, J.; Elumalai, K. Mosquitocidal Activities of Corchorus capsularis L (Malvaceae) against a Common Malarial Vector, Anopheles Stephensi (Liston) and a Dengue Vector Aedes Aegypti (L) (Diptera: Culicidae). Int. J. Recent Sci. Res. 2012, 3, 564–568. [Google Scholar]
- Chenniappan, K.; Kadarkarai, M. Oviposition deterrent, ovicidal and gravid mortality effects of ethanolic extract of Andrographis paniculata Nees against the malarial vector Anopheles stephensi Liston (Diptera: Culicidae). Entomol. Res. 2008, 38, 119–125. [Google Scholar] [CrossRef]
- Sathiyamoorthy, P.; Lugasi-Evgi, H.; Schlesinger, P.; Kedar, I.; Gopas, J.; Pollack, Y.; Golan-Goldhirsh, A. Screening for cytotoxic and antimalarial activities in desert plants of the Negev and Bedouin market plant products. Pharm. Biol. 1999, 37, 188–195. [Google Scholar] [CrossRef]
- Mohammed, R.M.O. Phytochemical Investigation of Antimicrobial and Antioxidant Activity Leaves Extracts of Corchorus olitorius. OALib 2016, 3, 1–5. [Google Scholar] [CrossRef]
- Roy, N. Role of Chorchorus capsularis phytochemicals on the feeding dynamics of Diacrisia casignetum Kollar (Lepidoptera: Arctiidae). J. Entomol. Zool. Stud. 2014, 2, 227–236. [Google Scholar]
- Schoonhoven, L.; Van Loon, J.J.A.; Dicke, M. Insect Plant Biology, 2nd ed.; Oxford University Press: Oxford, UK, 2005; Available online: https://www.scirp.org/(S(351jmbntvnsjt1aadkozje))/reference/referencespapers.aspx?referenceid=1939403 (accessed on 30 May 2022).
- Talukder, D.; Muslima Khanam, L.A. Toxicity of four plant based products against three stored product pests. J. Bio-Sci. 2009, 17, 149–153. [Google Scholar] [CrossRef]
- Mahfuz, I.; Khanam, L.A.M. Toxicity of Some Indigenous Plant Extracts Against Tribolium. J. Bio-Sci. 2007, 15, 133–138. [Google Scholar] [CrossRef]
- Manning, P.J.; Sutherland, W.H.; Walker, R.J.; Williams, S.M.; De Jong, S.A.; Ryalls, A.R.; Berry, E.A. Effect of High-Dose Vitamin E on Insulin. Emerg. Treat. Technol. 2004, 27, 2166–2171. [Google Scholar]
- Adedosu, O.T.; Akanni, O.E.; Afolabi, O.K.; Adedeji, A.L. Effects of Corchorus olitorius Extract on Certain Antioxidants and Biochemical Indices in Sodium Arsenite Exposed Rats. Am. J. Phytomed. Clin. Ther. 2015, 245–256. [Google Scholar]
- Dewanjee, S.; Gangopadhyay, M.; Sahu, R.; Karmakar, S. Cadmium induced pathophysiology: Prophylactic role of edible jute (Corchorus olitorius) leaves with special emphasis on oxidative stress and mitochondrial involvement. Food Chem. Toxicol. 2013, 60, 188–198. [Google Scholar] [CrossRef]
- Dewanjee, S.; Sahu, R.; Karmakar, S.; Gangopadhyay, M. Toxic effects of lead exposure in Wistar rats: Involvement of oxidative stress and the beneficial role of edible jute (Corchorus olitorius) leaves. Food Chem. Toxicol. 2013, 55, 78–91. [Google Scholar] [CrossRef]
- Omeje, K.O.; Ezike, T.C.; Omeje, H.C.; Ezema, B.O. Effect of ethanol extract of Corchorus olitorus leaf on glucose level and antioxidant enzymes of Streptozotocin-induced hyperglycemic rat. Biokemistri 2016, 28, 121–127. [Google Scholar] [CrossRef]
- Ilhan, S.; Savarouglu, F.; CColak, F. Antibacterial and Antifungal Activity of Corchorus olitorius L. (Molokhia) Extracts. Int. J. Nat. Eng. Sci. 2007, 1, 59–61. [Google Scholar]
- Pramanik, P.K.; Paik, D.; Pramanik, A.; Chakraborti, T. Parasitology International White jute (Corchorus capsularis L.) leaf extract has potent leishmanicidal activity against Leishmania donovani. Parasitol. Int. 2019, 71, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Pal, D.K.; Mandal, M.; Senthilkumar, G.P.; Padhiari, A. Antibacterial activity of Cuscuta reflexa stem and Corchorus olitorius seed. Fitoterapia 2006, 77, 589–591. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).