Plant- and Animal-Based Antioxidants’ Structure, Efficacy, Mechanisms, and Applications: A Review
Abstract
:1. Introduction
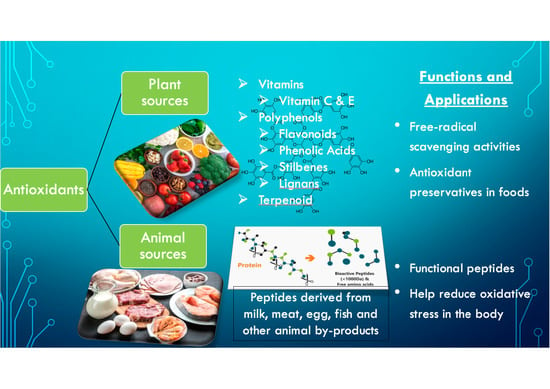
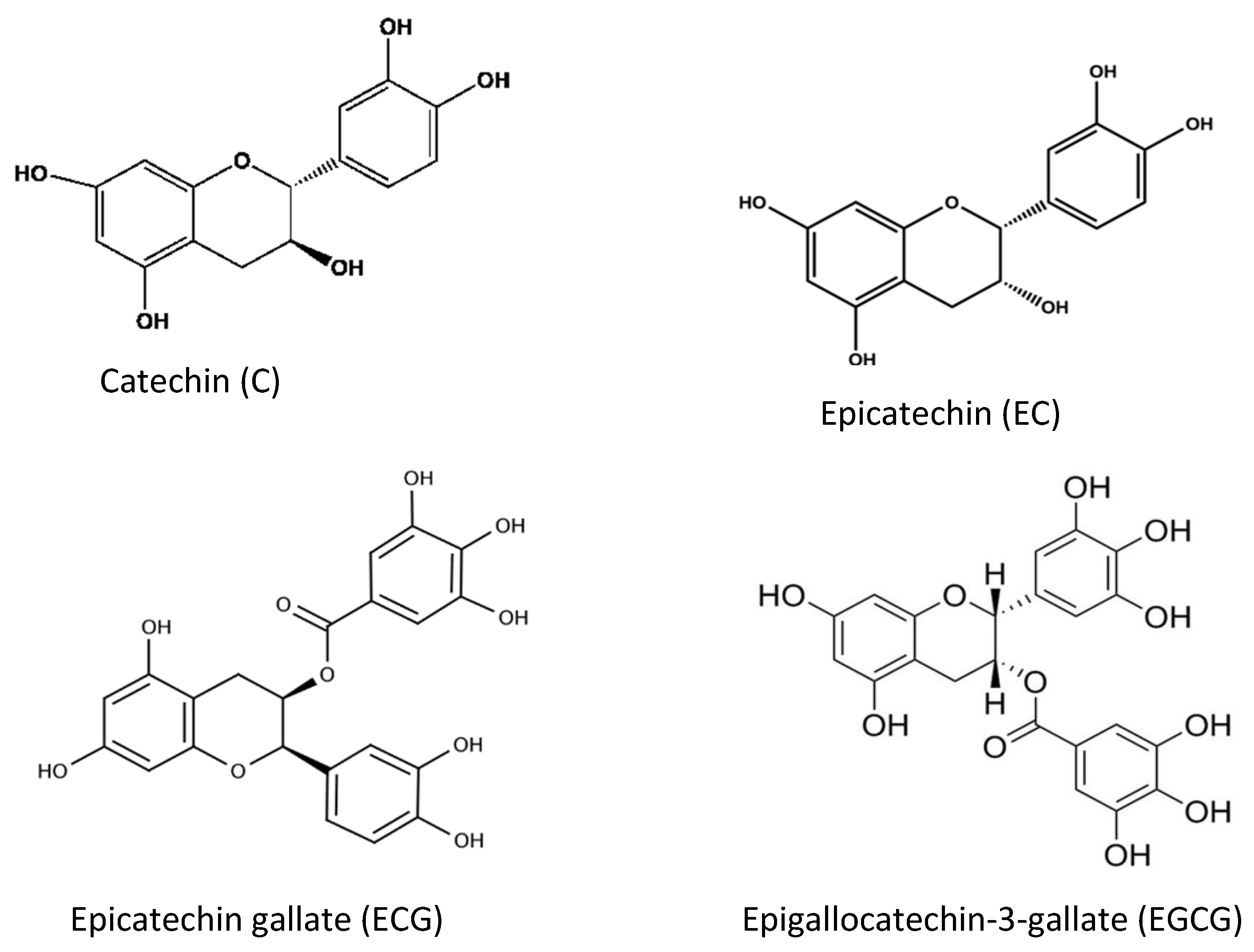
2. Antioxidants from Plant Sources
3. Antioxidants from Animal Sources
Antioxidant Animal Proteins and Peptides
4. Differences between the Antioxidants from Plant and Animal Sources
5. Applications of Antioxidants
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Hazra, B.; Biswas, S.; Mandal, N. Antioxidant and free radical scavenging activity of Spondias pinnata. BMC Complement. Altern. Med. 2008, 8, 63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mirzaei, M.; Mirdamadi, S.; Ehsani, M.R.; Aminlari, M. Antioxidant, ACE-inhibitory and antibacterial activities of Kluyeromyces marzianus protein hydrolysates and their peptide fractions. Funct. Foods Health Dis. 2016, 6, 425–439. [Google Scholar] [CrossRef]
- Kulczyński, B.; Sidor, A.; Gramza-Michałowska, A. Characteristics of selected antioxidative and bioactive compounds in meat and animal origin products. Antioxidants 2019, 8, 335. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zeb, A. Concept, mechanism, and applications of phenolic antioxidants in foods. J. Food Biochem. 2020, 44, e13394. [Google Scholar] [CrossRef]
- Abeyrathne, E.D.N.S.; Nam, K.C.; Ahn, D.U. Analytical methods for lipid oxidation and antioxidant capacity in food systems—A review. Antioxidants 2021, 10, 1587. [Google Scholar] [CrossRef]
- Abourashed, E.A. Bioavailability of plant-derived antioxidants. Antioxidants 2013, 2, 309–325. [Google Scholar] [CrossRef] [Green Version]
- Orian, M.; Escriche, I. Antioxidants: Characterization, natural sources, extraction, and analysis. Food Res. Int. 2015, 74, 10–36. [Google Scholar] [CrossRef]
- Hernández-Ruiz, K.L.; Ruiz-Cruz, S.; Cira-Chávez, L.A.; Gassos-Ortega, L.E.; Ornelas-Paz, J.J.; del-Toro-Sánchez, C.L.; Márquez-Ríos, E.; López-Mata, M.A.; Rodríguez-Félix, F. Evaluation of antioxidant capacity, protective effect on human erythrocytes and phenolic compounds identification in two varieties of plum fruit (Spondias spp.) by UPLC-MS. Molecules 2018, 23, 3200. [Google Scholar] [CrossRef] [Green Version]
- Munekata, P.E.S.; Gullón, B.; Pateiro, M.; Tomasevic, I.; Domínguez, R.; Lorenzo, J.S. Natural antioxidants from seeds and their applications in meat products. Antioxidants 2020, 9, 815. [Google Scholar] [CrossRef]
- Mojzer, E.B.; Hrnčič, M.K.; Škerget, M.; Knez, Ž.; Bren, U. Polyphenols: Extraction methods, Antioxidative action, bioavailability, and anticarcinogenic effects. Molecules 2016, 21, 901. [Google Scholar] [CrossRef]
- Alshkh, N.; de Camargo, A.D.; Shahidi, F. Phenolics of selected lentil cultivars: Antioxidant activities and inhibition of low-density lipoprotein and DNA damage. J. Funct. Foods 2015, 18, 1022–1038. [Google Scholar] [CrossRef]
- Falowo, A.B.; Fayemi, P.O.; Muchenje, V. Natural antioxidants against lipid-protein oxidative deterioration in meat and meat products. A review. Food Res. Int. 2014, 64, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Dziedzinski, M.; Kobus-Cisowska, J.; Szymanowska, D.; Stuper-Szablewska, K.; Baranowska, M. Identification of polyphenols from coniferous shoots as natural antioxidants and antimicrobial compounds. Molecules 2020, 25, 3527. [Google Scholar] [CrossRef] [PubMed]
- Sant’Ana, A.S. Introduction to the special issue: Salmonella in foods: Evolution, strategies, and challenges. Food Res. Int. 2012, 45, 451–454. [Google Scholar] [CrossRef]
- Jideani, A.I.O.; Silungwe, H.; Takalani, T.; Omolola, A.O.; Udeh, H.O.; Anyashi, T.A. Antioxidant-rich natural fruit and vegetable products and human health. Int. J. Food Prop. 2021, 24, 41–67. [Google Scholar] [CrossRef]
- Chen, X.; Touyz, R.M.; Park, J.B.; Schiffrin, E.L. Antioxidant effects of vitamin C and E are associated with altered activation of vascular NADPH oxidase and superoxide dismutase in stroke-prone SHR. Hypertension 2001, 38, 606–611. [Google Scholar] [CrossRef] [Green Version]
- Traber, M.G.; Stevens, J.F. Vitamin C and E: Beneficial effects from a mechanistic perspective. Free Radic. Biol. Med. 2011, 51, 1000–1013. [Google Scholar] [CrossRef] [Green Version]
- Leopoldina, M.; Marino, T.; Russo, N.; Toscano, M. Antioxidant properties of phenolic compounds: H-atom versus electron transfer mechanism. J. Phys. Chem. A 2004, 108, 4916–4922. [Google Scholar] [CrossRef]
- Klein, E.; Lukeš, V. DFT/B3LYP study of the substituent effect on the reaction enthalpies of the individual steps of single-electron transfer-proton transfer and sequential proton loss electron transfer mechanisms of phenols antioxidant action. J. Phys. Chem. A 2006, 110, 12312–12320. [Google Scholar] [CrossRef]
- Abdel-Shafy, H.; Mansour, M.S.M. Polyphenols: Properties, occurrence, Content in food, and potential effects. In Environmental Science and Engineering; Volume 6: Toxicology; Chandra, R., Gurjar, B.R., Govil, J.N., Eds.; Studium Press LLC: Houston, TX, USA, 2017; pp. 232–261. [Google Scholar]
- Single, R.K.; Dubey, A.K.; Garg, A.; Sharma, R.K.; Fiorino, M. Natural polyphenols: Chemical Classification, definition of classes, subcategories, and structure. J. AOAC Int. 2019, 102, 1397–1400. [Google Scholar] [CrossRef]
- Dimitrios, B. Sources of natural phenolic antioxidants. Trends Food Sci. Technol. 2006, 17, 505–512. [Google Scholar] [CrossRef]
- Dirimanov, S.; Högger, P. Screening of inhibitory effects of polyphenols on Akt-phosphorylation in endothelial cells and determination of structure-activity features. Biomolecules 2019, 9, 219. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amarowicz, R.; Pegg, R.B. Natural antioxidants of plant origin. In Advances in Food and Nutrition Research; Ferreira, I.C.F.R., Barros, L., Eds.; Academic Press: Cambridge, UK, 2019; pp. 1–81. [Google Scholar]
- Kumar, N.; Goel, N. Phenolic acids: Natural versatile molecules with promising therapeutic applications. Biotechnol. Rep. 2019, 24, e00370. [Google Scholar] [CrossRef]
- Shan, B.; Cai, Y.Z.; Brooks, J.D.; Corke, H. Antibacterial and antioxidant effects of five spice and herb extracts as natural preservatives of raw pork. J. Sci. Food Agric. 2009, 89, 1879–1885. [Google Scholar] [CrossRef]
- Markus, M.A.; Morris, B.J. Resveratrol in prevention and treatment of common clinical conditions of aging. Clin. Interv. Aging 2008, 3, 331–339. [Google Scholar] [PubMed]
- Pandey, K.B.; Rizvi, S.I. Resveratrol may protect plasma proteins from oxidation under conditions of oxidative stress in vitro. J. Braz. Chem. Soc. 2010, 21, 909–913. [Google Scholar] [CrossRef] [Green Version]
- Nagapan, T.S.; Ghazali, A.R.; Basri, D.F.; Lim, W.N. Photoprotective effect of stilbenes and its derivatives against ultraviolet radiation-induced skin disorders. Biomed. Pharmacol. J. 2018, 11, 1199–1208. [Google Scholar] [CrossRef]
- Liu, Z.; Sarrinen, N.M.; Thompson, L.U. Sesamin is one of the major precursors of mammalian lignans in sesame seed (Sesamum indicum), as observed in vitro and in rats. J. Nutr. 2006, 136, 906–912. [Google Scholar] [CrossRef] [Green Version]
- Wanasundara, P.K.J.P.D.; Shahidi, F. Process-induced change in edible oils. In Process-Induced Chemical Changes in Food; Shahidi, F., Ho, C.T., Chuyen, N.V., Eds.; Plenum Publishers: New York, NY, USA, 1998; pp. 135–160. [Google Scholar]
- Kumar, M.C.; Singh, S.A. Bioactive lignans from sesame (Sesamum indicum L.): Evaluation of their antioxidant and antibacterial effects for food applications. J. Food Sci. Technol. 2015, 52, 2934–2941. [Google Scholar] [CrossRef] [Green Version]
- Rosalina, R.; Weerapreeyakul, N. An Insight into sesamolin: Physicochemical properties, pharmacological activities, and future research prospects. Molecules 2021, 26, 5849. [Google Scholar] [CrossRef]
- Takahashi, M.; Nishizaki, Y.; Sugimoto, N.; Takeuchi, H.; Nakagawa, K.; Akiyama, H.; Sato, K.; Inoue, K. Determination and purification of sesamin and sesamolin in sesame seed oil unsaponified matter using reversed-phase liquid chromatography coupled with photodiode array and tandem mass spectrometry and high-speed counter-current chromatography. J. Sep. Sci. 2016, 39, 3898–3905. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, I.I.; Quax, W.J. A Glimpse into the biosynthesis of terpenoids. In NRLS Conference Proceedings, International Conference on Natural Resources and Life Science (2016). KnE Life Sciences; Knoledge E Publisher: Dubai, United Arab Emirates, 2017; pp. 81–98. [Google Scholar]
- Hatano, T.; Kusuda, M.; Inada, K.; Ogawa, T.O.; Shiota, S.; Tsuchiya, T.; Yoshida, T. Effects of tannins and related polyphenols on methicillin-resistant Staphylococcus aureus. Phytochemistry 2005, 66, 2047–2055. [Google Scholar] [CrossRef] [PubMed]
- Amarowicz, R.; Estrella, I.; Hernández, T.; Troszynska, A. Antioxidant activity of extract of adzuki bean and its fractions. J. Food Lipids 2008, 15, 119–136. [Google Scholar] [CrossRef]
- Azman, N.A.M.; Gallego, M.G.; Segovia, F.; Abdullah, S.; Md Shaarani, S.; Pablos, M.P.A. Study of the properties of bearberry leaf extract as a natural antioxidant in model foods. Antioxidants 2016, 5, 11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jabri, M.-A.; Rtibi, K.; Ben-Said, A.; Aouadhi, C.; Hosni, K.; Sally, M.; Sebai, H. Antidiarrhoeal, antimicrobial and antioxidant effects of myrtle berries (Myrtus communis L.) seeds extract. J. Pharm. Pharmacol. 2016, 68, 264–274. [Google Scholar] [CrossRef]
- Amarowicz, R.; Karamać, M.; Dueňas, M.; Pegg, R.B. Antioxidant activity and phenolic composition of a red bean (Phaseolus Vulgaris) extract and its fractions. Nat. Prod. Commun. 2017, 12, 541–544. [Google Scholar]
- Amarowicz, R.; Shahidi, F. Antioxidant activity of Faba bean extract and fractions thereof. J. Food Bioact. 2018, 1, 112–118. [Google Scholar] [CrossRef] [Green Version]
- Patel, M.; Naik, S.N. Gamma-oryzanol from rice bran oil—A review. J. Sci. Ind. Res. 2004, 63, 569–578. [Google Scholar]
- Lee, S.H.; Kim, H.W.; Lee, M.K.; Lee, M.K.; Kim, Y.J.; Asamenew, G.; Cha, Y.S.; Kim, J.B. Phenolic profiling and quantitative determination of common sage (Salvia plebeian R. Br.) by UPLC-DAD-QTOF/MS. Eur. Food Res. Technol. 2018, 244, 1637–1646. [Google Scholar] [CrossRef] [Green Version]
- Oniga, I.; Puşcaş, C.; Silaghi-Dumitrescu, R.; Olah, N.-K.; Sevastre, B.; Marica, R.; Marcus, I.; Sevastre-Berghian, A.C.; Benedec, D.; Pop, C.E.; et al. Origanum vulgare ssp. vulgare: Chemical composition and biological studies. Molecules 2018, 23, 2077. [Google Scholar] [CrossRef] [Green Version]
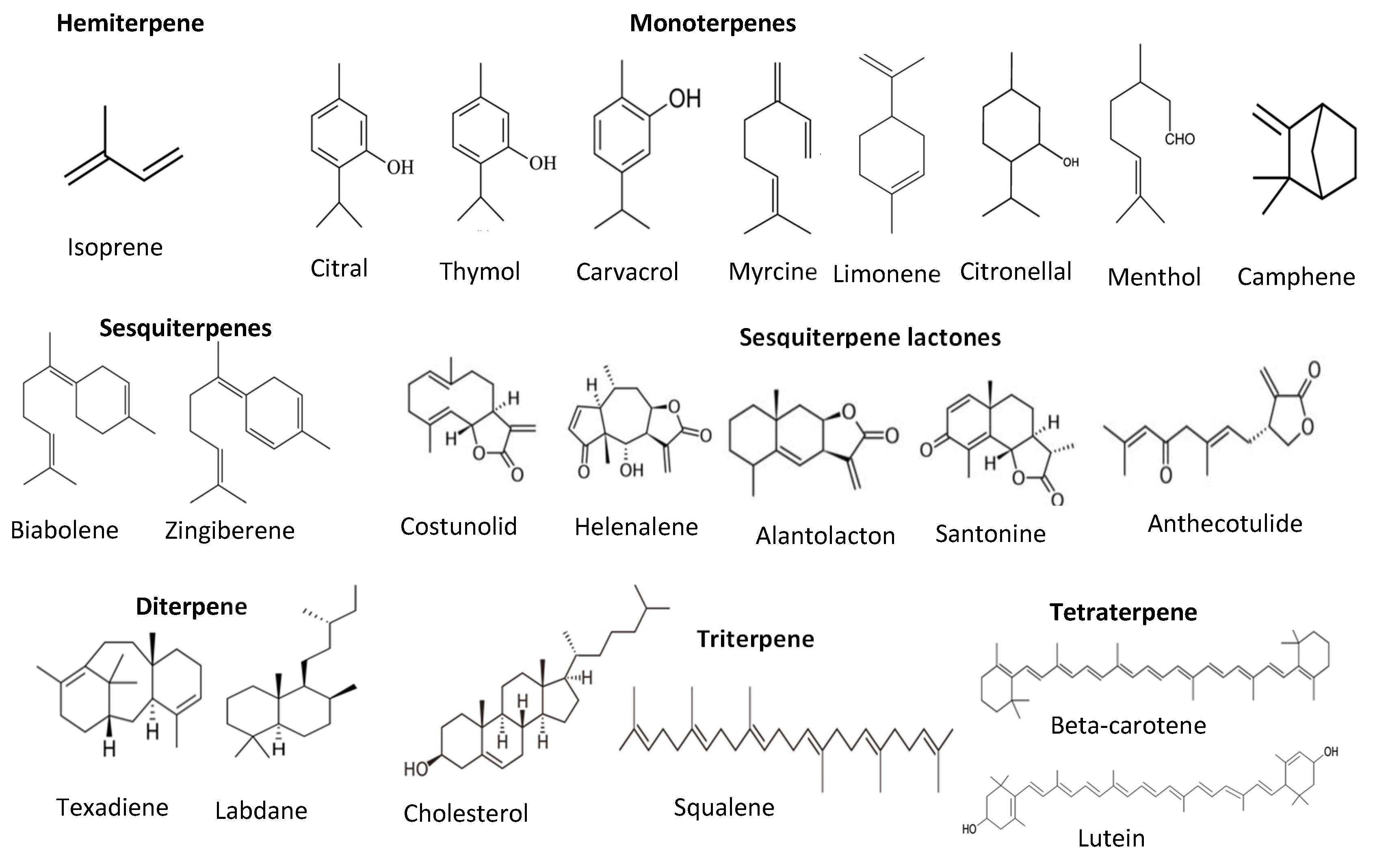
- Jaeger, R.; Cuny, E. Terpenoids with special pharmacological significance: A review. Nat. Prod. Commun. 2016, 11, 1373–1390. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prishtina, E.; Plyusnin, S.; Babak, T.; Lashmanova, E.; Maganova, F.; Koval, L.; Platonova, E.; Shaposhnikov, M.; Moskalev, A. Terpenoids as potential geroprotectors. Antioxidants 2020, 9, 529. [Google Scholar]
- Terpins, P.; Čeh, B.; Ulrich, N.P.; Abramovič, H. Studies of the correlation between antioxidant properties and the total phenolic content of different oil cake extracts. Ind. Crops Prod. 2012, 39, 210–217. [Google Scholar] [CrossRef]
- Szydłowska-Czerniak, A. Rapeseed and its products-sources of bioactive compounds: A review of their characteristics and analysis. Crit. Rev. Food Sci. Nutr. 2013, 53, 307–330. [Google Scholar] [CrossRef] [PubMed]
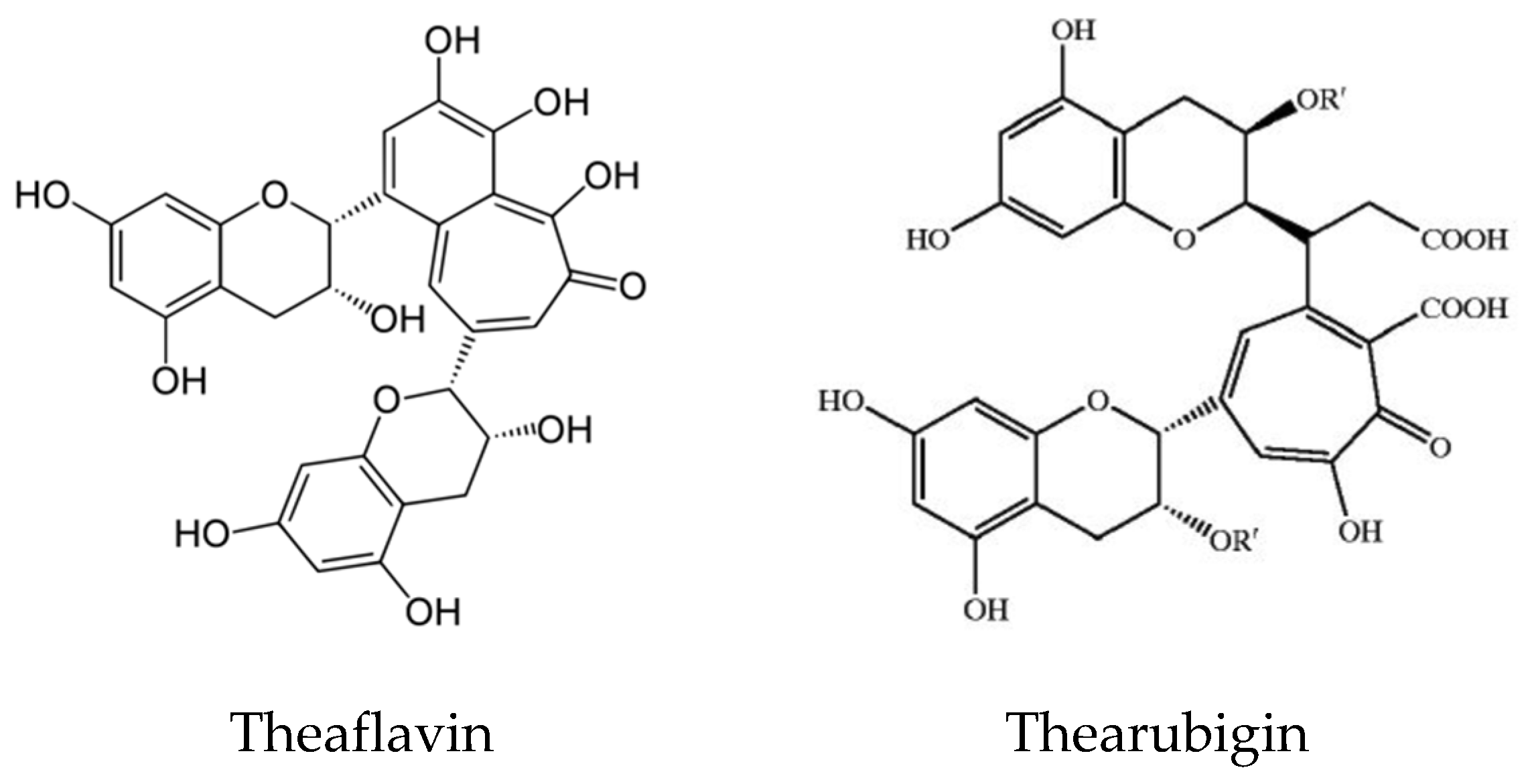
- Lee, L.S.; Kim, Y.C.; Park, J.D.; Kim, Y.B.; Kim, S.H. Changes in major polyphenolic compounds of tea (Camellia sinensis) leaves during the production of black tea. Food Sci. Biotechnol. 2016, 25, 1523–1527. [Google Scholar] [CrossRef] [PubMed]
- Koch, W.; Kukula-Koch, W.; Komsta, Ł.; Maezec, Z.; Szwarc, W.; Głowniak, K. Green tea quality evaluation based on its catechins and metals composition in combination with chemometric analysis. Molecules 2018, 23, 1689. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takemoto, M.; Takemoto, H. Synthesis of theaflavins and their functions. Molecules 2018, 23, 918. [Google Scholar] [CrossRef] [Green Version]
- Moreira, A.S.P.; Nunes, F.M.; Domingues, M.R.; Coimbra, M.A. Coffee melanoidins: Structures, mechanisms of formation and potential health impacts. Food Funct. 2012, 3, 903–915. [Google Scholar] [CrossRef]
- Farah, A.; Donangelo, C.M. Phenolic compounds in coffee. Braz. J. Plant Biol. 2007, 18, 23–36. [Google Scholar] [CrossRef]
- Fanali, C.; Tripodo, G.; Russo, M.; Della Posta, S.; Pasqualetti, V.; de Gara, L. Effect of solvent on the extraction on the phenolic compounds and antioxidant capacity of the hazelnut kernel. Electrophoresis 2018, 39, 1683–1691. [Google Scholar] [CrossRef]
- Rusu, M.E.; Gheldiu, A.M.; Mocan, A.; Moldovan, C.; Popa, D.S.; Tomita, I.; Vlase, L. Process optimization for improved phenolic compounds recovery from walnut (Juglans regia L.) septum: Phytochemical profile and biological activities. Molecules 2018, 23, 2814. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vrhovsek, U.; Rigo, A.; Tonon, D.; Mattivi, F. Quantitation of polyphenols in different apple varieties. J. Agric. Food Chem. 2004, 52, 6532–6538. [Google Scholar] [CrossRef] [PubMed]
- Skrovankova, S.; Sumczynski, D.; Macek, J.; Jurikova, T.; Sochor, J. Bioactive compounds and antioxidant activity in different types of berries. Int. J. Mol. Sci. 2015, 16, 24673–24706. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khoshnoudi-Na, S.; Niakosari, M.; Tahiri, Z. Subcritical water technology for extraction phytochemical compound. J. Med. Plants 2017, 9, 94–107. [Google Scholar]
- Marchante, L.; Izquierdo-Caǹas, P.M.; Gómez-Alonso, S.; Alaǹón, S.; García-Romero, M.E.; Pérez-Coello, M.S.; Díaz-Maroto, M. Oenological potential of extracts from winery and cooperage by-products in combination with colloidal silver as natural substitutes to sulphur dioxide. Food Chem. 2019, 276, 485–493. [Google Scholar] [CrossRef]
- Nick, M.; Shahidi, F. Phenolics in cereals, fruits, and vegetables: Occurrence, extraction, and analysis. J. Pharm. Biomed. Anal. 2006, 41, 1523–1542. [Google Scholar]
- Liang, J.; Zago, E.; Nandasiri, R.; Khattab, R.; Eskin, N.A.M.M.; Eck, P.; Holländer, U.T. Effect of solvent preheating temperature and time on the ultrasonic extraction of phenolic compounds from cold-pressed hempseed cake. J. Am. Oil Chem. Soc. 2018, 95, 1319–1327. [Google Scholar] [CrossRef]
- Acosta-Estrada, B.A.; Gutierrez-Uribe, J.A.; Serna-Saldívar, S.O. Bound phenolics in foods. A review. Food Chem. 2014, 152, 46–55. [Google Scholar] [CrossRef]
- Mullen, W.; Nemzer, B.; Ou, B.; Stalmach, A.; Hunter, J.; Clifford, M.; Combet, E. The antioxidant and chlorogenic acid profiles of whole coffee fruits are influenced by extraction procedures. J. Agric. Food Chem. 2011, 59, 3754–3762. [Google Scholar] [CrossRef]
- Lee, S.G.; Terrence, M.; Vance, T.M.; Nam, T.G.; Kim, D.O.; Koo, S.I.; Chun, O.K. Contribution of anthocyanin composition of total antioxidant capacity of berries. Plant Food Hum. Nutr. 2005, 25, 523–1527. [Google Scholar] [CrossRef]
- Kozłowska, M.; Gruczynska, E. Comparison of the oxidative stability of soybean and sunflower oils enriched with herbal plant extracts. Chem. Pap. 2018, 72, 2607–2615. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dos Reis, L.C.R.; Facco, E.M.P.; Flôres, S.H.; Rios, A.D.O. Stability of functional compounds and antioxidant activity of fresh and pasteurized orange passion fruit (Passiflora caerulea) during cold storage. Food Res. Int. 2018, 106, 481–486. [Google Scholar] [CrossRef] [PubMed]
- İnan, Ö.; Özczn, M.M.; Aljuhaimi, F. Effect of location and Citrus species on total phenolic, antioxidant and radical scavenging activities of some Citrus seed and oil. J. Food Process. Preserv. 2018, 42, 1215–1219. [Google Scholar] [CrossRef]
- Do Couto, C.A.; de Souza, E.R.B.; Morgado, C.M.A.; Ogata, T.; Cunha Júnior, L.C. Citrus Sinensis cultivars: Alternatives for diversification of Brazilian orchards. Rev. Bras. Food Sci. 2018, 50, 905–991. [Google Scholar] [CrossRef] [Green Version]
- Konieczyski, P.; Viapiana, A.; Mark Wesolowski, M. Comparison of infusions from black and green teas (Camellia sinensis L. Kuntze) and erva-mate (Ilex paraguariensis A. St.-Hil) based on the content of essential elements, secondary metabolites, and antioxidant activity. Food Anal. Methods 2017, 10, 3063–3070. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.-F.; Oey, I.; Bremer, P.; Carne, A.; Silcock, P. Bioactive peptides derived from egg proteins: A review. Crit. Rev. Food Sci. Nutr. 2018, 58, 2508–2530. [Google Scholar] [CrossRef]
- Nowicka, A.; Kucharska, A.Z.; Sokół-Łętowska, A.; Fecka, I. Comparison of polyphenol content and antioxidant capacity of strawberry fruit from 90 cultivars of Fragaria × ananassa Duch. Food Chem. 2019, 270, 32–46. [Google Scholar] [CrossRef]
- Lourenço, S.C.; Moldão-Martins, M.; Alves, V.D. Antioxidants of natural plant origins: From sources to food industry applications. Molecules 2019, 24, 4132. [Google Scholar] [CrossRef] [Green Version]
- Wijeratne, S.S.K.; Amarowicz, R.; Shahidi, F. Antioxidant activity of almonds and their by-products in food model. J. Am. Oil Chem. Soc. 2006, 83, 223–230. [Google Scholar] [CrossRef]
- Rojas, M.C.; Brewer, M.S. Effect of natural antioxidants on oxidative stability of cooked, refrigerated beef and pork. J. Food Sci. 2007, 72, S282–S288. [Google Scholar] [CrossRef]
- Oswell, N.J.; Thippareddi, H.; Pegg, R.B. Practical use of natural antioxidants in meat products in the US. A Review. Meat Sci. 2017, 145, 469–479. [Google Scholar] [CrossRef] [PubMed]
- Gramza-Michałowska, A.; Korczak, J.; Reguła, J. Use of plant extracts in summer and winter season butter oxidative stability improvement. Asia Pac. J. Clin. Nutr. 2007, 16 (Suppl. 1), 85–88. [Google Scholar] [PubMed]
- Maqsood, S.; Benjakul, S.; Abushelaibi, A.; Alam, A. Phenolic compounds and plant phenolic extracts as natural antioxidants in prevention of lipid oxidation in seafood: A detailed review. Compr. Rev. Food Sci. Food Saf. 2014, 13, 1125–1140. [Google Scholar] [CrossRef]
- Your, M.; Howell, N. Antioxidant and ACE inhibitory bioactive peptides from purified egg yolk proteins. Int. J. Mol. Sci. 2015, 16, 29161–29178. [Google Scholar]
- Abeyrathne, E.D.N.S.; Huang, X.; Ahn, D.U. Antioxidant, angiotensin-converting enzyme inhibitory activity, and other functional properties of egg white proteins and their derived peptides. Poult. Sci. 2018, 97, 1462–1468. [Google Scholar] [CrossRef]
- Rathnapala, E.C.N.; Ahn, D.U.; Abeyrathne, E.D.N.S. Functional properties of ovotransferrin from chicken egg white and its derived peptides- A Review. Food Sci. Biotechnol. 2021, 30, 619–630. [Google Scholar] [CrossRef]
- Huang, X.; Ahn, D.U. Lipid oxidation and its implications to meat quality and human health. Food Sci. Biotechnol. 2019, 28, 1275–1285. [Google Scholar] [CrossRef]
- Sarmadi, B.; Ismail, A. Antioxidative peptides from food proteins: A review. Peptides 2010, 31, 1949–1956. [Google Scholar] [CrossRef]
- Benedé, S.; Molina, E. Chicken egg proteins and derived peptides with antioxidant properties. Foods 2020, 9, 735. [Google Scholar] [CrossRef]
- Shahidi, F.; Zhong, Y. Bioactive peptides. J. AOAC Int. 2008, 91, 914–931. [Google Scholar] [CrossRef] [Green Version]
- Bhat, Z.F.; Kumat, S.; Bhat, H.F. Bioactive peptides of animal origin: A review. J. Food Sci. Technol. 2015, 52, 5377–5392. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maestri, E.; Pavlicevic, M.; Montorsi, M.; Marmiroli, N. Meta-Analysis for correlating structure of bioactive peptides in foods of animal origin with regard to effect and stability. Compr. Rev. Food Sci. Food Saf. 2018, 18, 3–30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiang, L.J.; Hu, Y.Y.; Hu, H.Y.; Ge, Q.F.; Zhou, G.H.; Zhang, W.G. Purification and identification of antioxidative peptides from dry-cured Xuanwei ham. Food Chem. 2016, 194, 951–958. [Google Scholar] [CrossRef] [PubMed]
- Toldrá, F.; Gallego, M.; Reig, M.; Aristoy, M.C.; Mora, L. Recent progress in enzymatic release of peptides in foods of animal origin and assessment of bioactivity. J. Agric. Food Chem. 2020, 68, 12842–12855. [Google Scholar] [CrossRef] [PubMed]
- Akbarian, M.; Khani, A.; Eghbalpour, S.; Uversky, V.N. Bioactive peptides: Synthesis, source, application and proposed mechanisms of action. Int. J. Mol. Sci. 2022, 23, 1445. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.W.; Nam, M.S. Bioactive peptides in milk and dairy products: A review. Korean J. Food Sci. Anim. Resour. 2015, 35, 831–840. [Google Scholar] [CrossRef] [Green Version]
- Albenzio, M.; Santillo, A.; Caroprese, M.; della Malva, A.; Marino, R. Bioactive peptides in animal food proteins. Foods 2017, 6, 35. [Google Scholar] [CrossRef] [Green Version]
- Corrȇa, A.P.E.; Dariot, D.J.; Coelho, J.; Meirs, S.M.M.; Lopes, F.C.; Segalin, J. Antioxidant, antihypertensive and antimicrobial properties of ovine milk caseinate hydrolyzed with microbial proteinase. J. Sci. Food Agric. 2011, 91, 2247–2254. [Google Scholar]
- Wali, A.; Yanhua, G.; Ishimov, U.; Yili, A.; Aisa, H.A.; Salikhov, S. Therapeutics, Isolation, and identification of three novel antioxidant peptides from the Bactrian camel milk hydrolysates. Int. J. Pept. Res. Ther. 2020, 26, 641–650. [Google Scholar] [CrossRef]
- Shanmugam, V.P.; Kapila, S.; Sonfack, T.K.; Kapila, R. Antioxidative peptides derived from enzymatic digestion of buffalo casein. Int. Dairy J. 2015, 42, 1–5. [Google Scholar] [CrossRef]
- Chakrabarti, S.; Jahandideh, F.; Wu, J. Food-derived bioactive peptides on inflammation and oxidative stress. BioMed Res. Int. 2014, 2014, 608979. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tonolo, F.; Folda, A.; Cesaro, L.; Scalcon, V.; Marin, O.; Ferro, S.; Bindoli, A.; Rigobello, M.P. Milk-derived bioactive peptides exhibit antioxidant activity through the Keap1-Nrf2 signaling pathway. J. Funct. Foods 2020, 64, 103696. [Google Scholar] [CrossRef]
- Huma, N.; Rafiq, S.; Sameen, A.; Pasha, I.; Khan, M.I. Antioxidant potential of buffalo and cow milk cheddar cheeses to tackle human colon adenocarcinoma (Caco-2) cells. Asian-Australas. J. Anim. Sci. 2018, 31, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Aloğlu, H.Ş.; Öner, Z. Determination of the antioxidant activity of bioactive peptides fractions obtained from yogurt. J. Dairy Sci. 2011, 94, 5305–5314. [Google Scholar] [CrossRef] [Green Version]
- Fu, Y.; Jette, F.Y.; Therkildsen, M. Bioactive peptides from meat muscle and by-products: Endogenous generation through postmortem aging. Meat Sci. 2017, 23, 134–142. [Google Scholar] [CrossRef]
- Susanto, E.; Fadlilah, A.; Amin, M.F. Synthesis, extraction, and identification of meat bioactive peptides: A review. IOP Conf. Series. Earth Environ. Sci. 2021, 888, 012058. [Google Scholar]
- Xing, L.; Liu, R.; Cao, S.; Zhang, W.; Zhu, G.H. Meat protein-based bioactive peptides and their potential functional activity: A review. Int. J. Food Sci. Technol. 2019, 54, 1956–1966. [Google Scholar] [CrossRef] [Green Version]
- Di Bernadini, R.; Mullen, A.M.; Bolton, D.; Kerry, J.; O’Neill, E.; Hayes, M. Assessment of angiotensin-I-converting enzyme (ACE-1) inhibitory and antioxidant activities of hydrolysates of bovine brisket sarcoplasmic proteins produced by papain and characterization of associated bioactive peptides fractions. Meat Sci. 2012, 90, 226–235. [Google Scholar] [CrossRef]
- Saiga, A.; Tanabe, S.; Nishimura, T. Antioxidant activity of peptides obtained from porcine myofibrilla proteins by protease treatment. J. Agric. Food Chem. 2003, 51, 3661–3667. [Google Scholar] [CrossRef]
- Ryan, J.T.; Ross, R.P.; Bolton, D.; Fitzgerald, G.F.; Stanton, C. Bioactive peptides from muscle sources: Meat and Fish. Nutrients 2011, 3, 765–791. [Google Scholar] [CrossRef] [Green Version]
- Wu, H.C.; Pan, B.S.; Chang, C.L.; Shiau, C.Y. Low-molecular-weight peptides as related to antioxidant properties of chicken essence. J. Food Drug Anal. 2005, 13, 176–183. [Google Scholar] [CrossRef]
- Stadnik, J.; Kęska, P. Meat and fermented meat products as a source of bioactive peptides. Acta Sci. Pol. Technol. Aliment. 2015, 14, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Broncano, J.M.; Otte, J.; Petrón, M.J.; Parra, V.; Timón, M.L. Isolation and identification of low molecular weight antioxidant compounds from fermented “chorizo” sausage. Meat Sci. 2012, 90, 494–501. [Google Scholar] [CrossRef] [PubMed]
- Escudero, E.; Mora, L.; Fraser, P.D.; Aristory, M.C. Identification of novel antioxidant peptides generated in Spanish dry-cured ham. Food Chem. 2013, 138, 1282–1288. [Google Scholar] [CrossRef]
- Liu, R.; Wang, M.; Duan, J.A.; Guo, G.M.; Tang, Y.P. Purification and identification of three novel antioxidant peptides from Cornu bubali (water buffalo horn). Peptides 2010, 31, 786–793. [Google Scholar] [CrossRef]
- Nadalian, M.; Kamaruzaman, N.; Yusop, M.S.M.; Babji, A.S.; Yusop, S.M. Isolation, purification, and characterization of antioxidative bioactive Elastin peptides from poultry skin. Food Sci. Anim. Resour. 2019, 39, 966–979. [Google Scholar] [CrossRef] [Green Version]
- Ao, J.; Li, B. Amino acid composition and antioxidant activities of hydrolysates and peptides fractions from porcine collagen. Food Sci. Technol. Int. 2012, 18, 425–434. [Google Scholar] [CrossRef]
- Gómez-Sampedro, L.J.; Zapata-Montoya, J.E. Obtaining of antioxidant peptide from bovine plasma hydrolysates and effect of the degree of hydrolysis on antioxidant capacity. Rev. Mex. De Ing. Química 2016, 15, 101–109. [Google Scholar]
- Bah, C.S.F.; Bekhit, E.A.; Crne, A.; McConnell, M.A. Slaughterhouse blood: An emerging source of bioactive compounds. Compr. Rev. Food Sci. Food Saf. 2013, 12, 314–331. [Google Scholar] [CrossRef]
- Yu, H.C.; Hsu, J.L.; Chang, C.I.; Tan, F.J. Antioxidant properties of porcine liver proteins hydrolyzed using Monascus purpureus. Food Sci. Biotechnol. 2017, 26, 1217–1225. [Google Scholar] [CrossRef]
- Liu, Q.; Tang, G.Y.; Zhao, C.N.; Feng, X.L.; Xu, X.Y.; Cao, S.Y.; Meng, X.; Li, S.; Gan, R.Y.; Li, H.B. Comparison of antioxidant activities of different grape varieties. Molecules 2018, 23, 2432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shen, S.; Chahal, B.; Majumder, K.; You, S.J.; Wu, J. Identification of novel antioxidative peptides derived from a thermolytic hydrolysate of ovotransferrin by LC-MS/MS. J. Agric. Food Chem. 2010, 58, 7664–7672. [Google Scholar] [CrossRef] [PubMed]
- Ayala, A.; Muñoz, M.F.; Argüelles, S. Lipid peroxidation: Production, metabolism and signaling mechanisms of malondialdehyde and 4-Hydroxy-2-Nonenal. Oxidative Med. Cell. Longev. 2014, 2014, 360438. [Google Scholar] [CrossRef] [PubMed]
- He, L.; He, T.; Farrar, S.; Ji, L.; Liu, T.; Ma, X. Antioxidants maintain cellular redox homeostasis by elimination of reactive oxygen species. Cell. Physiol. Biochem. 2017, 44, 532–553. [Google Scholar] [CrossRef]
- Jiang, H.; Tong, T.; Sun, J.; Xu, Y.; Zhao, Z.; Liao, D. Purification and characterization of antioxidative peptides from round scad (Decapterus maruadsi) muscle protein hydrolysate. Food Chem. 2014, 154, 158–163. [Google Scholar] [CrossRef]
- Lorenzo, J.M.; Munekata, P.E.S.; Gómez, B.; Barba, F.J.; Mora, L.; Pérez-Santaescolástica, C.; Toldrá, F. Bioactive peptides as natural antioxidants in food products—A review. Trends Food Sci. Technol. 2018, 79, 136–147. [Google Scholar] [CrossRef]
- Barreca, D. Mechanisms of Plant antioxidants actions. Plants 2021, 10, 35. [Google Scholar] [CrossRef]
- Zhu, C.Z.; Zhang, W.G.; Kanf, Z.L.; Zhou, G.H.; Xu, S.L. Stability of antioxidant peptides extracted from Jinhua ham. Meat Sci. 2014, 96, 783–789. [Google Scholar] [CrossRef]
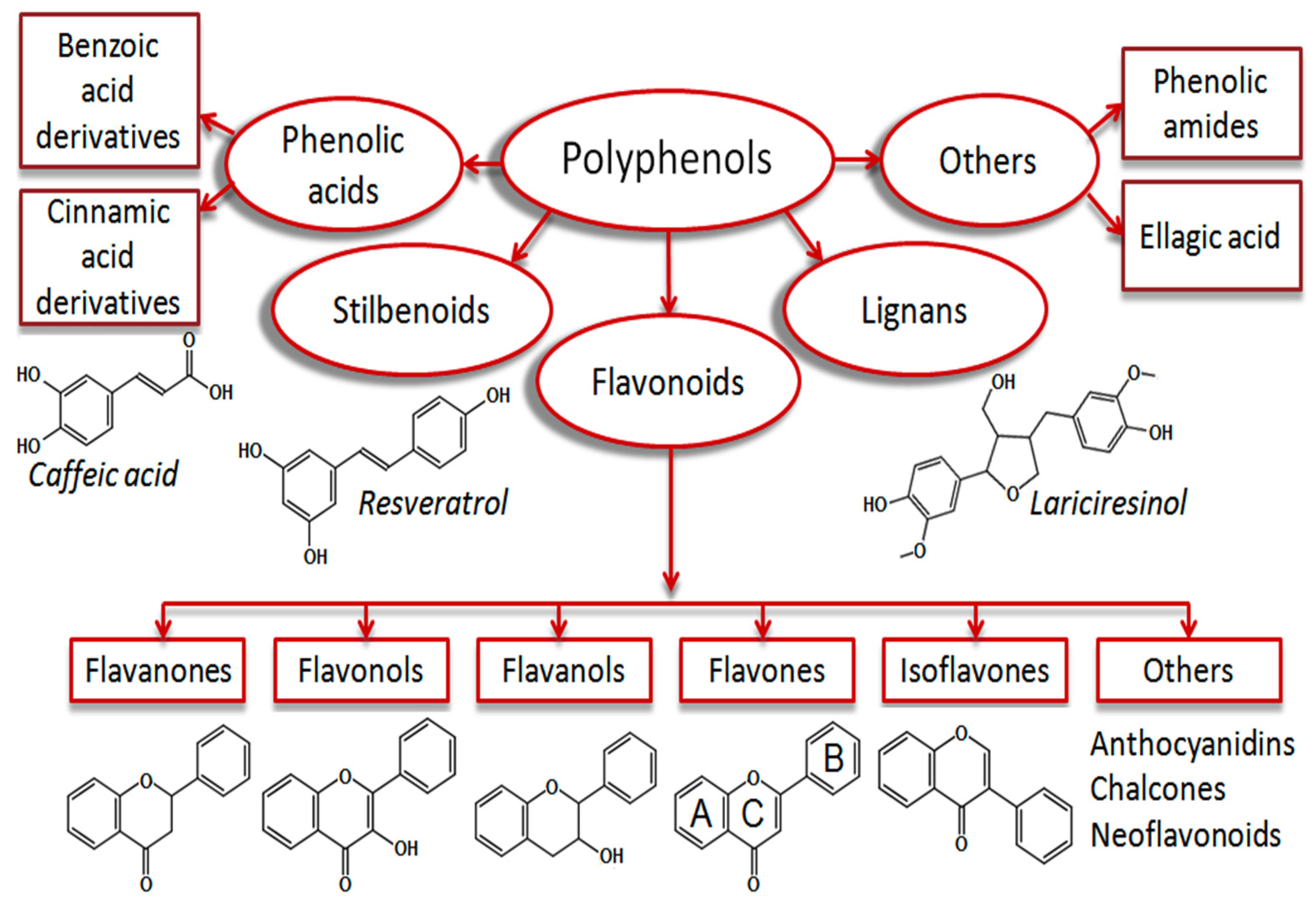
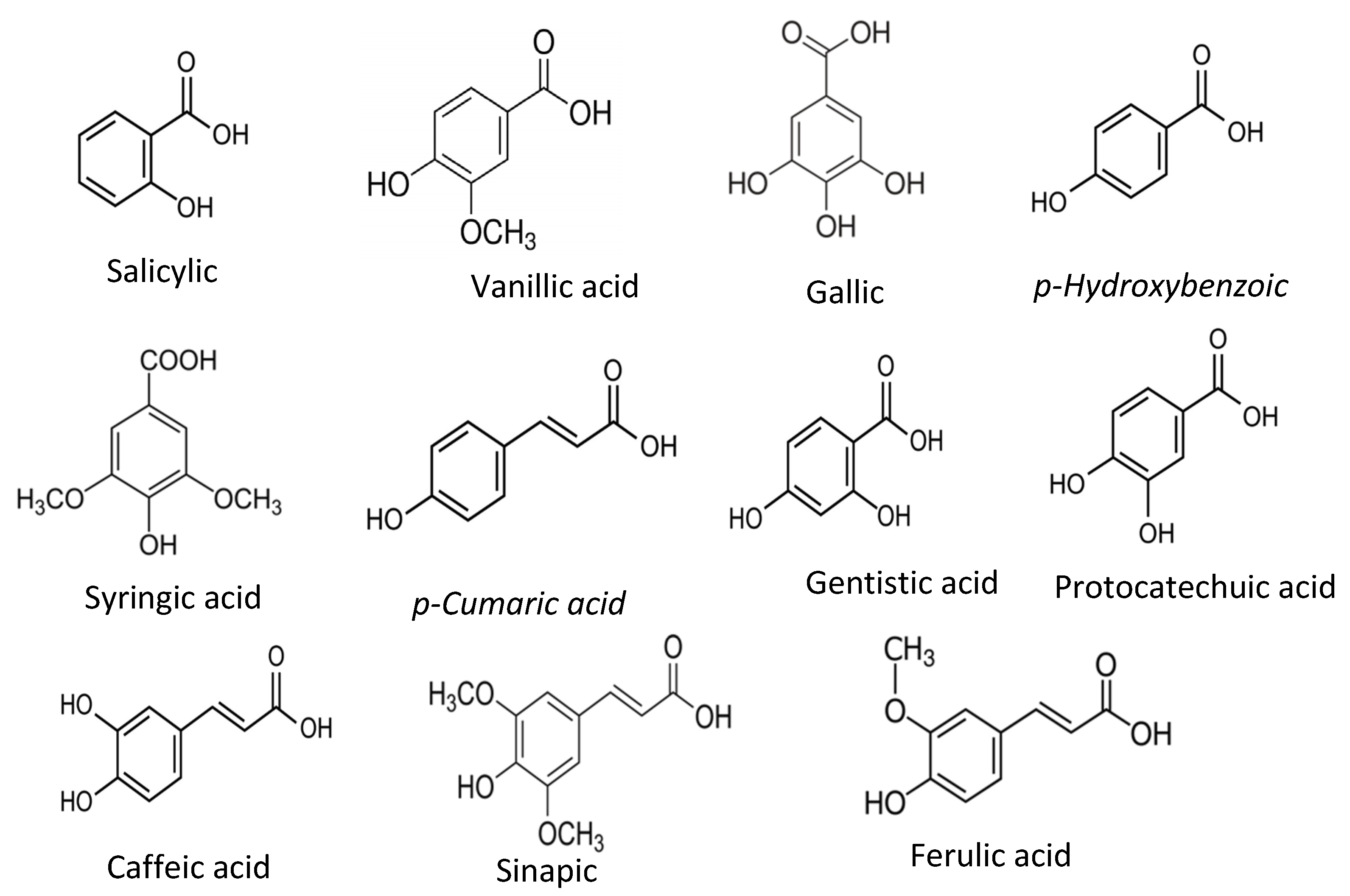
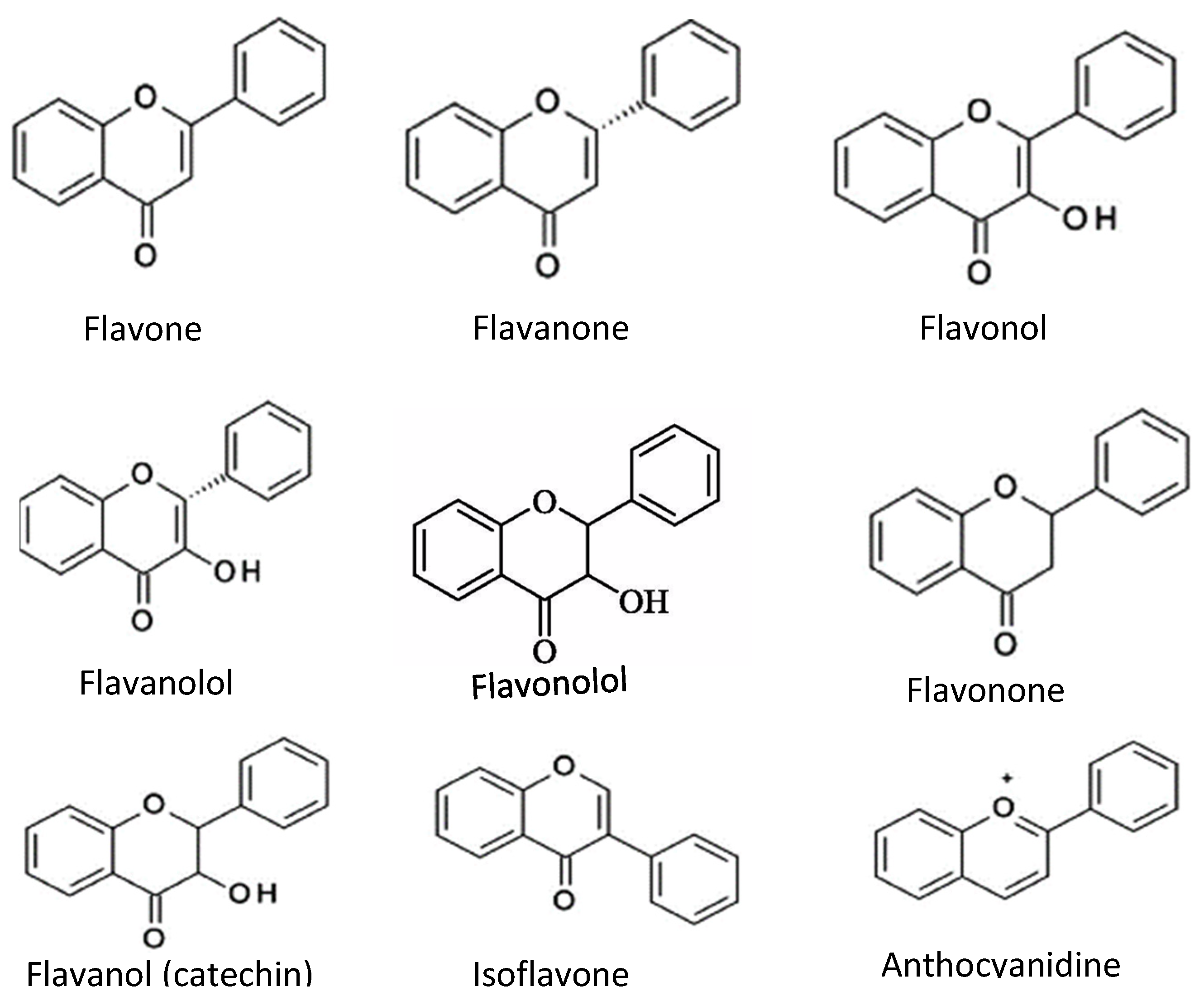

| Antioxidant Type | Subgroups | Examples | Applications | References |
|---|---|---|---|---|
| Phenolic compounds | Phenolic acids | Salicylic acid, Gentisic acid, p-Hydoxybenzoic acid, Protocatechuic acid, Vanillic acid, Syringic acid, Gallic acid, p-coumaric acid, Ferulic acid, Caffeic acid, Sinapic acid | As naturally present, primary antioxidant. | [21,25,35] |
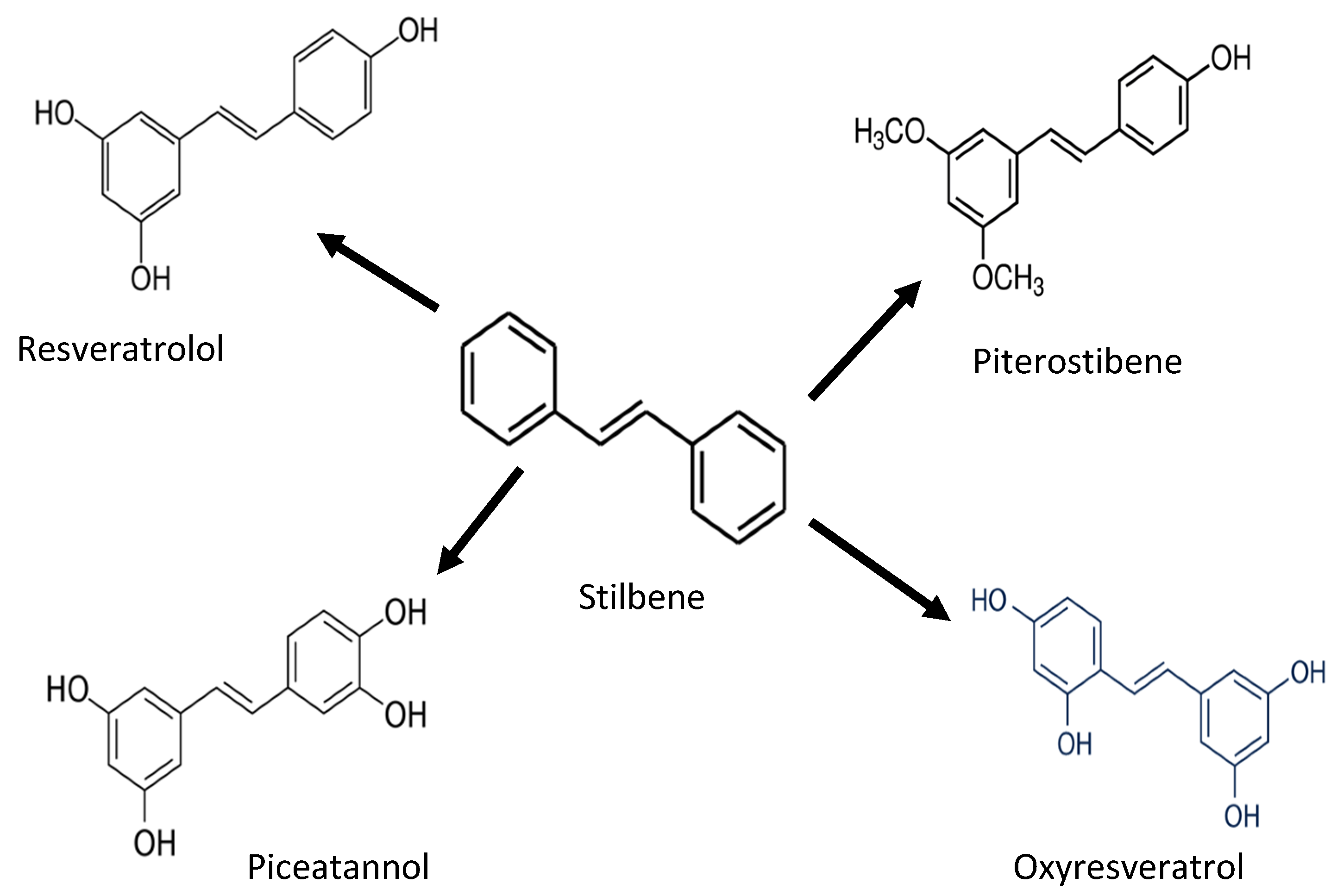
| Stilbenes | Piceid, Resveratrol, Piceatannol, Pterostilbene | Antioxidant activity against proteins and lipids | [27,28] | |
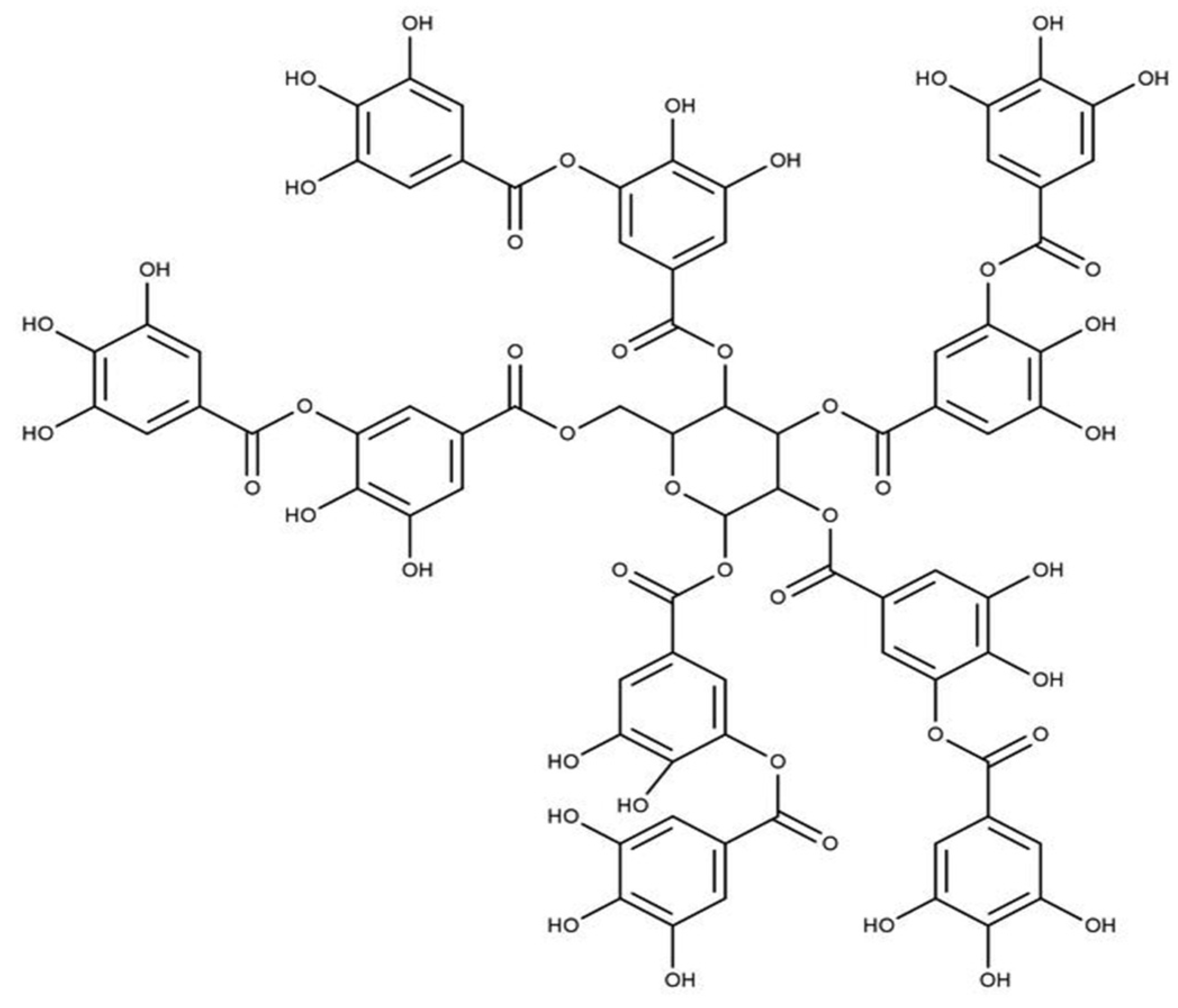
| Tannins | Biopolymers based on flavan-3-ols and Gallic and Ellagic acid | Strong antioxidant activities than flavonoids and phenolic acids | [36,37,38,39,40,41] | |
| Flavonoids | Flavone, Flavanol, Flavanone, Flavanonol, Flavonone, Flanononol, Flavanol (catechin), Isoflavone, Anthocyanidin | Act as antioxidant compounds if fruits, berries | [24,26,42,43,44] | |
| Lignans | Secoisolariciresinol, Matairesinol, Pinoresinol, Lariciresinol, sesamin, sesamolin | Strong antioxidant activities | [30,32] |
| Origin | Example Bioactive Proteins/Peptides | References | |
|---|---|---|---|
| Milk, milk-based products, and milk by-products | VLPVPQK, RLDGQGRPRVWLGR, KVLPVPEK, TPDNIDIWLGGIAEPQVKR, AVPYPQR, NPYVPR, ARHPHPHLSFM, VAYSDDGENWTEYRDQGAVEGK, YFYPEL | [85,92,93,95,97,98] | |
| Meat and meat products | Naturally present peptides | Carnosine (β-alanyl-L-histidine), Anserine (N-β-alanyl-1-methyl-L-histidine), Glutathione (GSH, Glu-Cys-Gly) | [106,117,118] |
| Synthetic Peptides | DAQEKLE, AKHPSDFGADAQ, SLTA, VT, AKHPSDFGADAQA, KAPVA, PTPVT, RPR, GLSDGEWQ, GFHI, DFHING, FHG, DSGVT, IEAEGE, EELDNALN, VPSIDDQEELM, DAQEKLE, DSGVT, IEAEGE, EELDNALN, VPSIDDQEELM, DAQEKLE, ATA, SLTA, VT, SAGNPN. GLAGA, DLEE, FGG, DM, ALTA | [3,85,87,88,91,99,100,101,104,105,106,107,108] | |
| Slaughterhouse by-products | QGAR, LQGM, LQGMH, HC, QYDQGV, YEDCTDCHN, AADNANELFPPN, GAHTHPRLPFKPR, GMPGFDVR, ADASVLPK, GAHQPSG, QQPVRDOQ | [84,86,88,101,109,110,111,113,114] | |
| Chicken eggs | Proteins | Ovotransferrin, Phosvitin | [79,80] |
| Peptides | White–WNIP, GWNI, IRW, LKP, VYLPR, YLGAK, GGLEPINFN, ESKPV, DVYSF, DSTRTQ Yolk–LMSYMWSTSM, KLSDW, RASDPLLSV, QSLVSVPGMS, LELHKLRSSHWFSRR, YINQMPQKSRE, RNDDLNYIQ, YINQMPQKSREA, VTGRFAGHPAAQ, LAPSLPGKPKPD, KGLWE, AGTTCLFTPLALPYDYSH, YIEAVNKVSPRAGQF | [78,79,80,91,115,116] | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abeyrathne, E.D.N.S.; Nam, K.; Huang, X.; Ahn, D.U. Plant- and Animal-Based Antioxidants’ Structure, Efficacy, Mechanisms, and Applications: A Review. Antioxidants 2022, 11, 1025. https://doi.org/10.3390/antiox11051025
Abeyrathne EDNS, Nam K, Huang X, Ahn DU. Plant- and Animal-Based Antioxidants’ Structure, Efficacy, Mechanisms, and Applications: A Review. Antioxidants. 2022; 11(5):1025. https://doi.org/10.3390/antiox11051025
Chicago/Turabian StyleAbeyrathne, Edirisinghe Dewage Nalaka Sandun, Kichang Nam, Xi Huang, and Dong Uk Ahn. 2022. "Plant- and Animal-Based Antioxidants’ Structure, Efficacy, Mechanisms, and Applications: A Review" Antioxidants 11, no. 5: 1025. https://doi.org/10.3390/antiox11051025
APA StyleAbeyrathne, E. D. N. S., Nam, K., Huang, X., & Ahn, D. U. (2022). Plant- and Animal-Based Antioxidants’ Structure, Efficacy, Mechanisms, and Applications: A Review. Antioxidants, 11(5), 1025. https://doi.org/10.3390/antiox11051025