Lingonberry Improves Hepatic Lipid Metabolism by Targeting Notch1 Signaling
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animal Model
2.2. Cell Culture
2.3. Triglyceride and Total Cholesterol Assays
2.4. Western Immunoblotting
2.5. Real-Time qPCR
2.6. Histological Staining
2.7. Statistical Analysis
3. Results
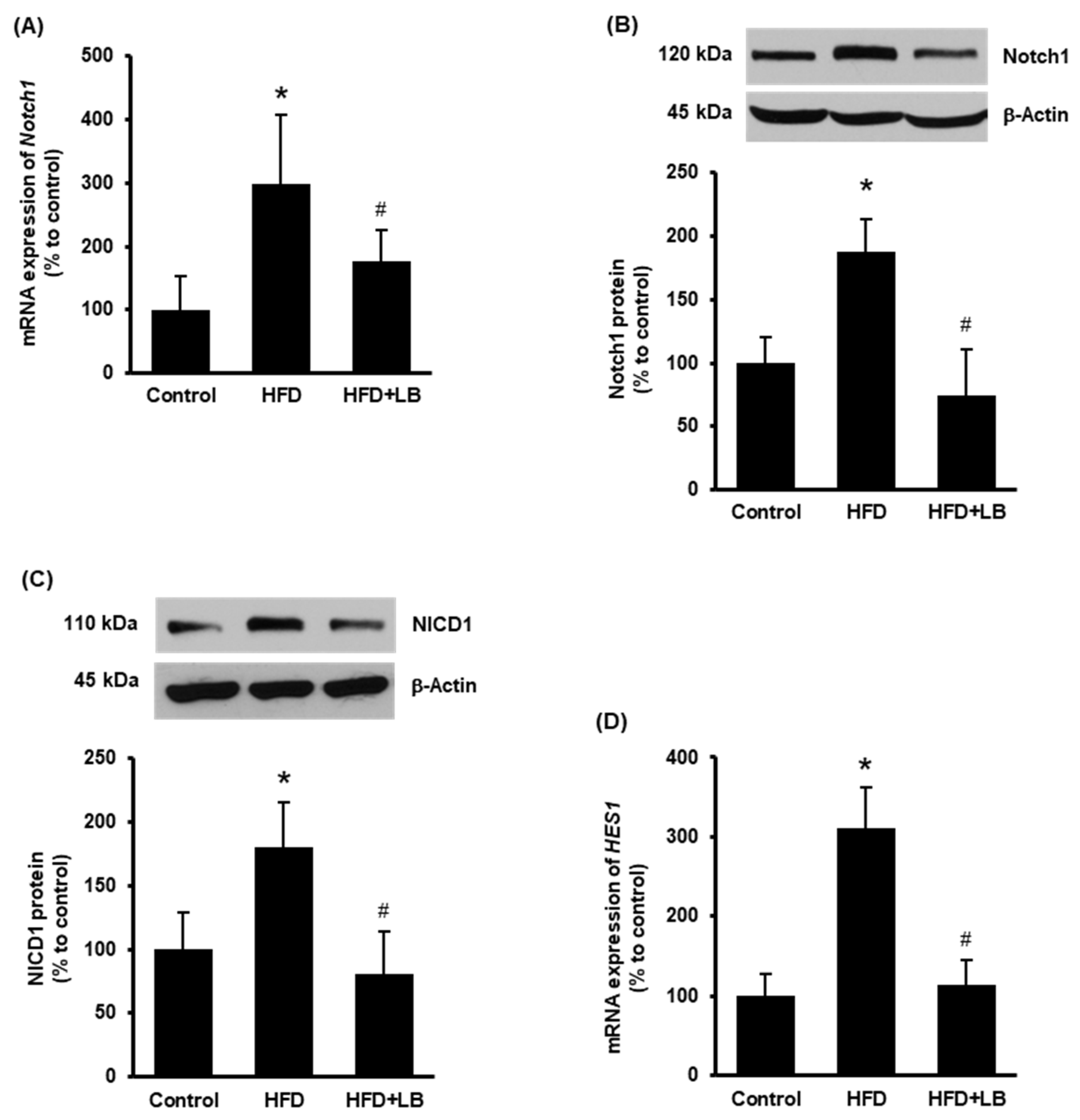
3.1. Lingonberry Supplementation Inhibits Hepatic Notch1 Signaling
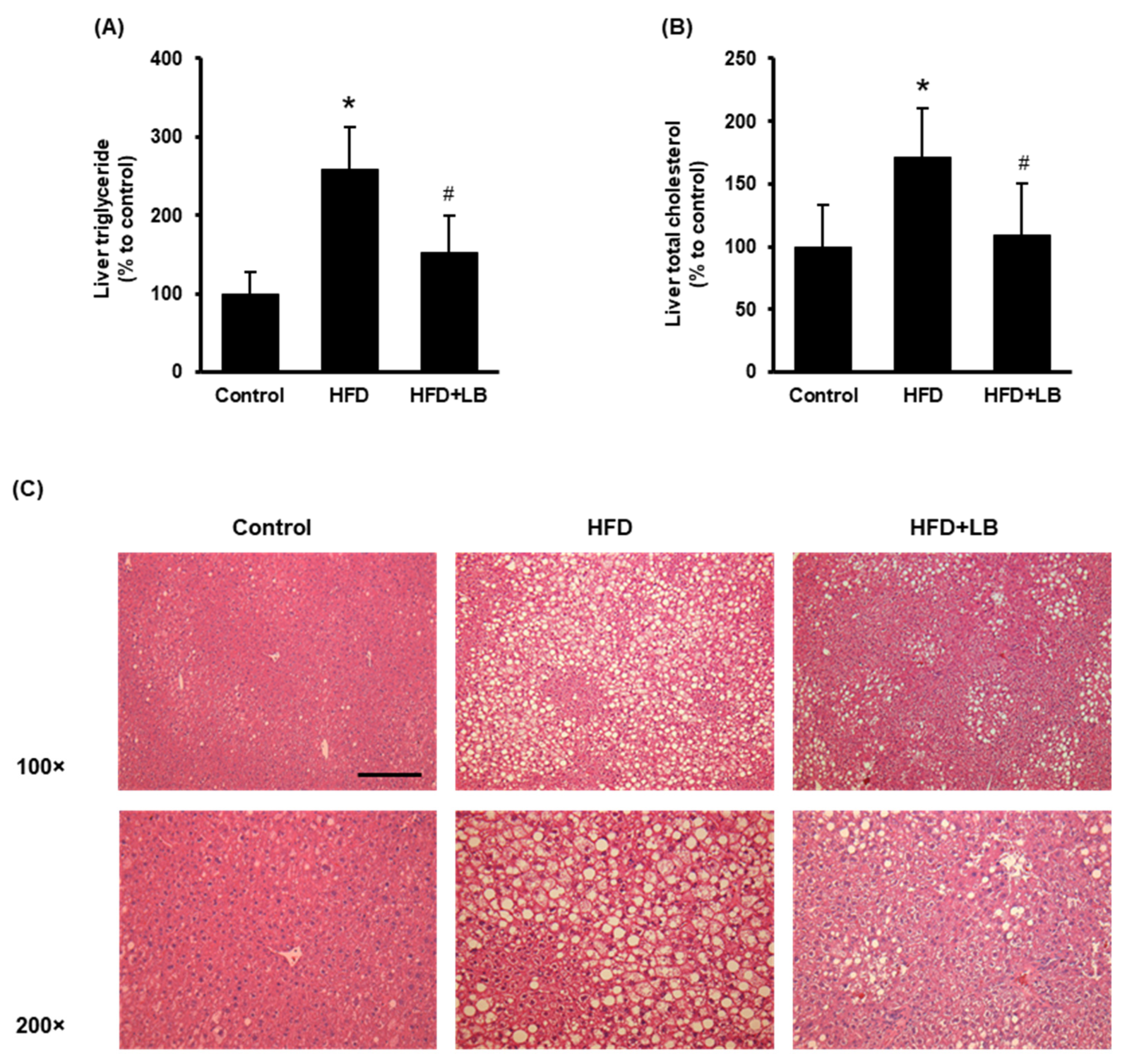
3.2. Lingonberry Supplementation Attenuates Liver Lipid Accumulation
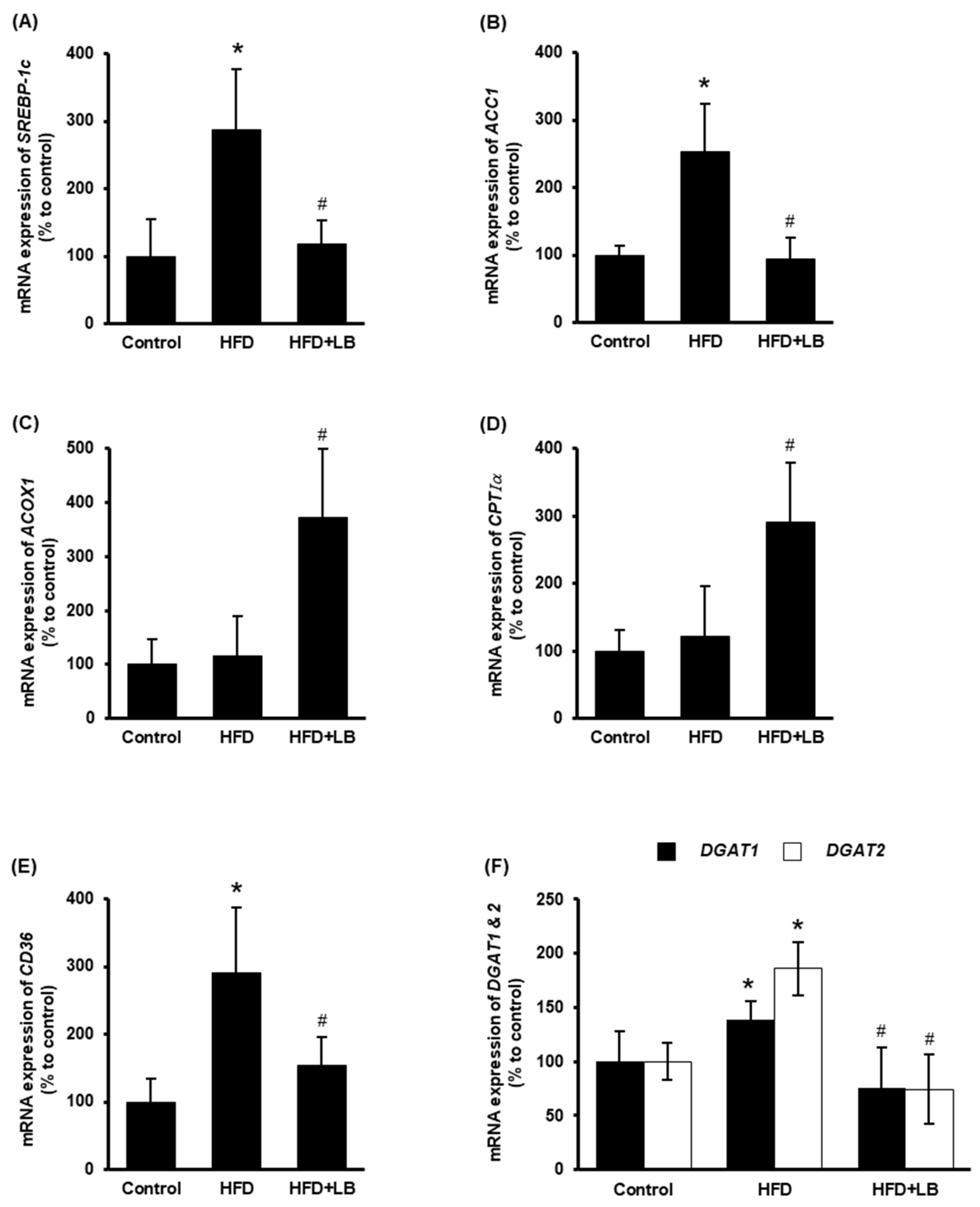
3.3. Lingonberry Supplementation Improves Liver Lipid Metabolism
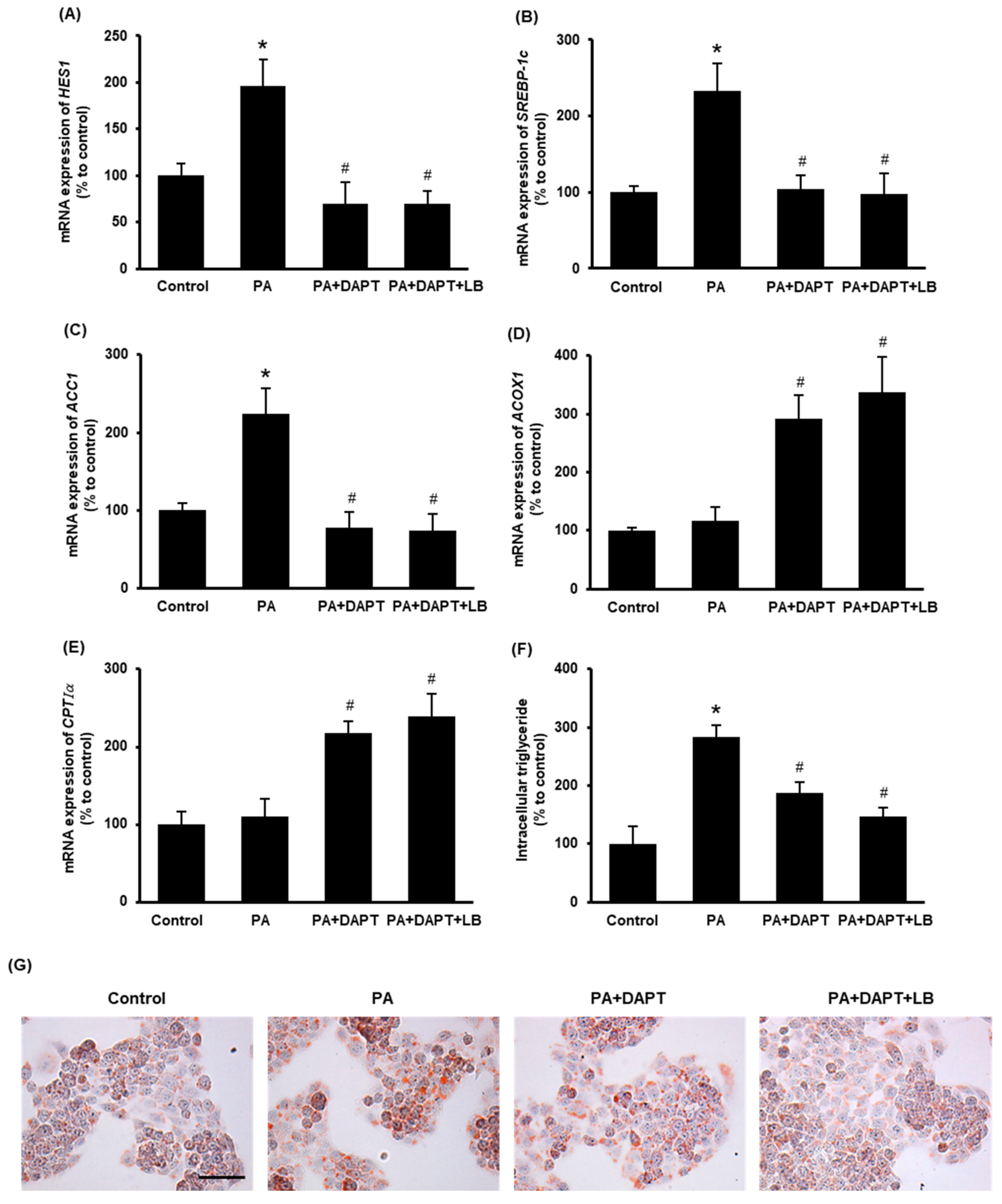
3.4. Lingonberry Extract and C3Glu Reduce Intracellular Lipid Accumulation and Suppress Notch1 and SREBP-1c Signaling
3.5. Inhibition of Notch Signaling Improves Cellular Lipid Metabolism
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Younossi, Z.M. Non-alcoholic fatty liver disease—A global public health perspective. J. Hepatol. 2019, 70, 531–544. [Google Scholar] [CrossRef] [Green Version]
- Loomba, R.; Sanyal, A.J. The global NAFLD epidemic. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 686–690. [Google Scholar] [CrossRef] [PubMed]
- Chalasani, N.; Younossi, Z.; LaVine, J.E.; Diehl, A.M.; Brunt, E.M.; Cusi, K.; Charlton, M.; Sanyal, A.J. The diagnosis and management of non-alcoholic fatty liver disease: Practice guideline by the American Association for the Study of Liver Diseases, American College of Gastroenterology, and the American Gastroenterological Association. Hepatology 2012, 55, 2005–2023. [Google Scholar] [CrossRef]
- Reddy, J.K.; Rao, M.S. Lipid metabolism and liver inflammation. II. Fatty liver disease and fatty acid oxidation. Am. J. Physiol. Liver Physiol. 2006, 290, G852–G858. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lambert, J.E.; Ramos-Roman, M.A.; Browning, J.D.; Parks, E.J. Increased de novo lipogenesis is a distinct characteristic of individuals with nonalcoholic fatty liver disease. Gastroenterology 2014, 146, 726–735. [Google Scholar] [CrossRef]
- Shimano, H.; Horton, J.D.; Shimomura, I.; Hammer, R.E.; Brown, M.S.; Goldstein, J.L. Isoform 1c of sterol regulatory element binding protein is less active than isoform 1a in livers of transgenic mice and in cultured cells. J. Clin. Investig. 1997, 99, 846–854. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Esler, W.P.; Bence, K.K. Metabolic targets in nonalcoholic fatty liver disease. Cell. Mol. Gastroenterol. Hepatol. 2019, 8, 247–267. [Google Scholar] [CrossRef] [Green Version]
- Bray, S. Notch signalling in context. Nat. Rev. Mol. Cell Biol. 2016, 17, 722–735. [Google Scholar] [CrossRef]
- Zong, Y.; Panikkar, A.; Xu, J.; Antoniou, A.; Raynaud, P.; Lemaigre, F.; Stanger, B.Z. Notch signaling controls liver development by regulating biliary differentiation. Development 2009, 136, 1727–1739. [Google Scholar] [CrossRef] [Green Version]
- Geisler, F.; Strazzabosco, M. Emerging roles of Notch signaling in liver disease. Hepatology 2015, 61, 382–392. [Google Scholar] [CrossRef] [Green Version]
- Romeo, S. Notch and nonalcoholic fatty liver and fibrosis. N. Engl. J. Med. 2019, 380, 681–683. [Google Scholar] [CrossRef] [PubMed]
- Kopan, R.; Ilagan, M.X.G. The canonical Notch signaling pathway: Unfolding the activation mechanism. Cell 2009, 137, 216–233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valenti, L.; Mendoza, R.M.; Rametta, R.; Maggioni, M.; Kitajewski, C.; Shawber, C.J.; Pajvani, U.B. Hepatic Notch signaling correlates with insulin resistance and nonalcoholic ffatty liver disease. Diabetes 2013, 62, 4052–4062. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pajvani, U.B.; Shawber, C.J.; Samuel, V.T.; Birkenfeld, A.L.; Shulman, G.I.; Kitajewski, J.; Accili, D. Inhibition of Notch signaling ameliorates insulin resistance in a FoxO1-dependent manner. Nat. Med. 2011, 17, 961–967. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, C.; Kim, K.; Wang, X.; Bartolome, A.; Salomao, M.; Dongiovanni, P.; Meroni, M.; Graham, M.J.; Yates, K.P.; Diehl, A.M.; et al. Hepatocyte Notch activation induces liver fibrosis in nonalcoholic steatohepatitis. Sci. Transl. Med. 2018, 10, eaat0344. [Google Scholar] [CrossRef]
- Chen, Y.; Zheng, S.; Qi, D.; Zheng, S.; Guo, J.; Zhang, S.; Weng, Z. Inhibition of Notch signaling by a γ-secretase inhibitor attenuates hepatic fibrosis in rats. PLoS ONE 2012, 7, e46512. [Google Scholar] [CrossRef]
- Debnath, S.C. Genetic diversity and erosion in berries. In Genetic Diversity and Erosion in Plants; Ahuja, M.R., Jain, S.M., Eds.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 75–129. [Google Scholar]
- Alhamimi, S.; Heyman-Lindén, L.; Plaza, M.; Turner, C.; Berger, K.; Spégel, P. Alterations in the plasma metabolite profile associated with improved hepatic function and glycemia in mice fed lingonberry supplemented high-fat diets. Mol. Nutr. Food Res. 2017, 61, 1600442. [Google Scholar] [CrossRef]
- Heyman, L.; Axling, U.; Blanco, N.; Sterner, O.; Holm, C.; Berger, K. Evaluation of beneficial metabolic effects of berries in high-fat fed C57BL/6J mice. J. Nutr. Metab. 2014, 2014, 403041. [Google Scholar] [CrossRef]
- Ryyti, R.; Hämäläinen, M.; Peltola, R.; Moilanen, E. Beneficial effects of lingonberry (Vaccinium vitis-idaea L.) supplementation on metabolic and inflammatory adverse effects induced by high-fat diet in a mouse model of obesity. PLoS ONE 2020, 15, e0232605. [Google Scholar] [CrossRef]
- Isaak, C.K.; Petkau, J.C.; O, K.; Debnath, S.C.; Siow, Y.L. Manitoba lingonberry (Vaccinium vitis-idaea) bioactivities in ischemia-reperfusion injury. J. Agric. Food Chem. 2015, 63, 5660–5669. [Google Scholar] [CrossRef]
- Isaak, C.K.; Petkau, J.C.; Blewett, H.; O, K.; Siow, Y.L. Lingonberry anthocyanins protect cardiac cells from oxidative-stress-induced apoptosis. Can. J. Physiol. Pharmacol. 2017, 95, 904–910. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Madduma Hewage, S.; Prashar, S.; Debnath, S.C.; O, K.; Siow, Y. Inhibition of inflammatory cytokine expression prevents high-fat diet-induced kidney injury: Role of lingonberry supplementation. Front. Med. 2020, 7, 80. [Google Scholar] [CrossRef]
- Madduma Hewage, S.; Prashar, S.; O, K.; Siow, Y. Lingonberry improves non-alcoholic fatty liver disease by reducing hepatic lipid accumulation, oxidative Stress and inflammatory response. Antioxidants 2021, 10, 565. [Google Scholar] [CrossRef] [PubMed]
- Kowalska, K.; Dembczyński, R.; Gołąbek, A.; Olkowicz, M.; Olejnik, A. ROS modulating effects of lingonberry (Vaccinium vitis-idaea L.) polyphenols on obese adipocyte hypertrophy and vascular endothelial dysfunction. Nutrients 2021, 13, 885. [Google Scholar] [CrossRef]
- Zhou, X.-L.; Wu, X.; Zhu, R.-R.; Xu, H.; Li, Y.-Y.; Xu, Q.-R.; Liu, S.; Lai, S.-Q.; Xu, X.; Wan, L.; et al. Notch1–Nrf2 signaling crosstalk provides myocardial protection by reducing ROS formation. Biochem. Cell Biol. 2020, 98, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef]
- Sarna, L.K.; Wu, N.; Wang, P.; Hwang, S.-Y.; Siow, Y.L.; O, K. Folic acid supplementation attenuates high fat diet induced hepatic oxidative stress via regulation of NADPH oxidase. Can. J. Physiol. Pharmacol. 2012, 90, 155–165. [Google Scholar] [CrossRef]
- Sid, V.; Shang, Y.; Siow, Y.L.; Madduma Hewage, S.; House, J.D.; O, K. Folic acid supplementation attenuates chronic hepatic inflammation in high-fat diet fed mice. Lipids 2018, 53, 709–716. [Google Scholar] [CrossRef]
- Chomzynski, P.; Mackey, K. Modification of the TRI Reagent [TM] Procedure for isolation of RNA from polysaccharide-and proteoglycan-rich sources. Biotechniques 1995, 19, 942–945. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Woo, C.W.H.; Siow, Y.L.; Pierce, G.N.; Choy, P.C.; Minuk, G.Y.; Mymin, D.; O, K. Hyperhomocysteinemia induces hepatic cholesterol biosynthesis and lipid accumulation via activation of transcription factors. Am. J. Physiol. Metab. 2005, 288, E1002–E1010. [Google Scholar] [CrossRef] [PubMed]
- Ferre, P.; Foufelle, F. Hepatic steatosis: A role for de novo lipogenesis and the transcription factor SREBP-1c. Diabetes Obes. Metab. 2010, 12, 83–92. [Google Scholar] [CrossRef]
- Cook, G.A.; Park, E.A. Expression and regulation of carnitine palmitoyltransferase-lα and -1β genes. Am. J. Med. Sci. 1999, 318, 43–48. [Google Scholar] [CrossRef]
- Aoyama, T.; Souri, M.; Kamijo, T.; Ushikubo, S.; Hashimoto, T. Peroxisomal acyl-coenzyme A oxidase is a rate-limiting enzyme in a very long-chain fatty acid β-oxidation system. Biochem. Biophys. Res. Commun. 1994, 201, 1541–1547. [Google Scholar] [CrossRef] [PubMed]
- Wilson, C.G.; Tran, J.L.; Erion, D.M.; Vera, N.B.; Febbraio, M.; Weiss, E.J. Hepatocyte-specific disruption of CD36 attenuates fatty liver and improves insulin sensitivity in HFD-fed mice. Endocrinology 2016, 157, 570–585. [Google Scholar] [CrossRef] [Green Version]
- Li, C.; Li, L.; Lian, J.; Watts, R.; Nelson, R.; Goodwin, B.; Lehner, R. Roles of acyl-CoA:diacylglycerol acyltransferases 1 and 2 in triacylglycerol synthesis and secretion in primary hepatocytes. Arter. Thromb. Vasc. Biol. 2015, 35, 1080–1091. [Google Scholar] [CrossRef] [Green Version]
- Pajvani, U.B.; Qiang, L.; Kangsamaksin, T.; Kitajewski, J.; Ginsberg, H.N.; Accili, D. Inhibition of Notch uncouples Akt activation from hepatic lipid accumulation by decreasing mTorc1 stability. Nat. Med. 2013, 19, 1054–1060. [Google Scholar] [CrossRef] [Green Version]
- Song, N.-J.; Hyun, B.S.; Yang, S.; Wu, C.; Seo, C.-R.; Gwon, A.-R.; Baik, S.-H.; Choi, Y.; Choi, B.Y.; Bahn, G.; et al. Notch1 deficiency decreases hepatic lipid accumulation by induction of fatty acid oxidation. Sci. Rep. 2016, 6, 19377. [Google Scholar] [CrossRef] [Green Version]
- Li, D.; Wang, P.; Luo, Y.; Zhao, M.; Chen, F. Health benefits of anthocyanins and molecular mechanisms: Update from recent decade. Crit. Rev. Food Sci. Nutr. 2017, 57, 1729–1741. [Google Scholar] [CrossRef]
- Osipo, C.; Zlobin, A.; Olsauskas-Kuprys, R. Gamma secretase inhibitors of Notch signaling. OncoTargets Ther. 2013, 6, 943–955. [Google Scholar] [CrossRef] [Green Version]
- Kratz, E.M.; Sołkiewicz, K.; Kubis-Kubiak, A.; Piwowar, A. Sirtuins as important factors in pathological states and the role of their molecular activity modulators. Int. J. Mol. Sci. 2021, 22, 630. [Google Scholar] [CrossRef] [PubMed]
- Tarantino, G.; Finelli, C.; Scopacasa, F.; Pasanisi, F.; Contaldo, F.; Capone, D.; Savastano, S. Circulating levels of sirtuin 4, a potential marker of oxidative metabolism, related to coronary artery disease in obese patients suffering from NAFLD, with normal or slightly increased liver enzymes. Oxidative Med. Cell. Longev. 2014, 2014, 920676. [Google Scholar] [CrossRef] [Green Version]
- Huynh, F.; Hu, X.; Lin, Z.; Johnson, J.D.; Hirschey, M.D. Loss of sirtuin 4 leads to elevated glucose- and leucine-stimulated insulin levels and accelerated age-induced insulin resistance in multiple murine genetic backgrounds. J. Inherit. Metab. Dis. 2018, 41, 59–72. [Google Scholar] [CrossRef] [PubMed]
- Purushotham, A.; Schug, T.T.; Xu, Q.; Surapureddi, S.; Guo, X.; Li, X. Hepatocyte-specific deletion of SIRT1 alters fatty acid metabolism and results in hepatic steatosis and inflammation. Cell Metab. 2009, 9, 327–338. [Google Scholar] [CrossRef] [Green Version]
- Kim, K.-H. Regulation of mammalian acetyl-coenzyme A. Annu. Rev. Nutr. 1997, 17, 77–99. [Google Scholar] [CrossRef] [PubMed]
- Stiede, K.; Miao, W.; Blanchette, H.S.; Beysen, C.; Harriman, G.; Harwood, H.J.; Kelley, H.; Kapeller, R.; Schmalbach, T.; Westlin, W.F. Acetyl-coenzyme A carboxylase inhibition reduces de novo lipogenesis in overweight male subjects: A randomized, double-blind, crossover study. Hepatology 2017, 66, 324–334. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tessari, P.; Coracina, A.; Cosma, A.; Tiengo, A. Hepatic lipid metabolism and non-alcoholic fatty liver disease. Nutr. Metab. Cardiovasc. Dis. 2009, 19, 291–302. [Google Scholar] [CrossRef] [PubMed]
- Alves-Bezerra, M.; Cohen, D.E. Triglyceride metabolism in the liver. Compr. Physiol. 2017, 8, 1–22. [Google Scholar] [CrossRef]
- Ryyti, R.; Pemmari, A.; Peltola, R.; Hämäläinen, M.; Moilanen, E. Effects of lingonberry (Vaccinium vitis-idaea L.) supplementation on hepatic gene expression in high-fat diet fed mice. Nutrients 2021, 13, 3693. [Google Scholar] [CrossRef]
- Nguyen, P.; Leray, V.; Diez, M.; Serisier, S.; Le Bloc’H, J.; Siliart, B.; Dumon, H. Liver lipid metabolism. J. Anim. Physiol. Anim. Nutr. 2008, 92, 272–283. [Google Scholar] [CrossRef]
- Foster, D.W. Malonyl-CoA: The regulator of fatty acid synthesis and oxidation. J. Clin. Investig. 2012, 122, 1958–1959. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Primer | Forward Sequence (5′–3′) | Reverse Sequence (5′–3′) | Accession Number |
|---|---|---|---|
| Human | |||
| Notch1 | CAATGTGGATGCCGCAGTTGTG | CAGCACCTTGGCGGTCTCGTA | NM_017617.5 |
| HES1 | TCAACACGACACCGGATAAA | CCGCGAGCTATCTTTCTTCA | NM_005524.4 |
| ACOX1 | GGCGCATACATGAAGGAGACCT | AGGTGAAAGCCTTCAGTCCAGC | NM_001185039.2 |
| CPTIα | CGATGTTACGACAGGTGGTTTGACA | AGTGCCCATCCTCCGCATAG | NM_001876.4 |
| ACC1 | TTCACTCCACCTTGTCAGCGGA | GTCAGAGAAGCAGCCCATCACT | NM_198838.2 |
| SREBP-1c | ACACAGCAACCAGAAACTCAAG | AGTGTGTCCTCCACCTCAGTCT | NM_001005291.3 |
| Mouse | |||
| Notch1 | CCAGCAGATGATCTTCCCGTAC | TAGACAATGGAGCCACGGATGT | NM_008714.3 |
| HES1 | CCCCAGCCAGTGTCAACAC | TGTGCTCAGAGGCCGTCTT | D16464.1 |
| DGAT1 | TTCCGCCTCTGGGCATT | AGAATCGGCCCACAATCCA | XM_006520405.4 |
| DGAT2 | AGTGGCAATGCTATCATCATCGT | AAGGAATAAGTGGGAACCAGATCA | NM_026384.3 |
| CD36 | TGTGCTAGACATTGGCAAATG | CTTCTCCTAAAGATAGGTGTG | XM_030254088.1 |
| ACOX1 | GCCTTTGTTGTCCCTATCCGT | CTTCAGGTAGCCATTATCCATCTCT | NM_001271898.1 |
| CPTIα | CATGATTGCAAAGATCAATCGG | CTTGACATGCGGCCAGTG | NM_013495.2 |
| ACC1 | CGGACCTTTGAAGATTTTGTGAGG | GCTTTATTCTGCTGGGTGAACTCTC | XM_030245463.1 |
| SREBP-1c | GGAGCCATGGATTGCACATT | GGCCCGGGAAGTCACTGT | NM_001358314.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Madduma Hewage, S.; Au-Yeung, K.K.W.; Prashar, S.; Wijerathne, C.U.B.; O, K.; Siow, Y.L. Lingonberry Improves Hepatic Lipid Metabolism by Targeting Notch1 Signaling. Antioxidants 2022, 11, 472. https://doi.org/10.3390/antiox11030472
Madduma Hewage S, Au-Yeung KKW, Prashar S, Wijerathne CUB, O K, Siow YL. Lingonberry Improves Hepatic Lipid Metabolism by Targeting Notch1 Signaling. Antioxidants. 2022; 11(3):472. https://doi.org/10.3390/antiox11030472
Chicago/Turabian StyleMadduma Hewage, Susara, Kathy K. W. Au-Yeung, Suvira Prashar, Charith U. B. Wijerathne, Karmin O, and Yaw L. Siow. 2022. "Lingonberry Improves Hepatic Lipid Metabolism by Targeting Notch1 Signaling" Antioxidants 11, no. 3: 472. https://doi.org/10.3390/antiox11030472
APA StyleMadduma Hewage, S., Au-Yeung, K. K. W., Prashar, S., Wijerathne, C. U. B., O, K., & Siow, Y. L. (2022). Lingonberry Improves Hepatic Lipid Metabolism by Targeting Notch1 Signaling. Antioxidants, 11(3), 472. https://doi.org/10.3390/antiox11030472







