Abstract
The rapid spread of antibiotic resistance and lack of effective drugs for treating infections caused by multi-drug resistant bacteria in animal and human medicine have forced us to find new antibacterial strategies. Natural products have served as powerful therapeutics against bacterial infection and are still an important source for the discovery of novel antibacterial drugs. Curcumin, an important constituent of turmeric, is considered safe for oral consumption to treat bacterial infections. Many studies showed that curcumin exhibited antibacterial activities against Gram-negative and Gram-positive bacteria. The antibacterial action of curcumin involves the disruption of the bacterial membrane, inhibition of the production of bacterial virulence factors and biofilm formation, and the induction of oxidative stress. These characteristics also contribute to explain how curcumin acts a broad-spectrum antibacterial adjuvant, which was evidenced by the markedly additive or synergistical effects with various types of conventional antibiotics or non-antibiotic compounds. In this review, we summarize the antibacterial properties, underlying molecular mechanism of curcumin, and discuss its combination use, nano-formulations, safety, and current challenges towards development as an antibacterial agent. We hope that this review provides valuable insight, stimulates broader discussions, and spurs further developments around this promising natural product.
1. Introduction
There is an urgent unmet medical need for new antibiotics for infections caused by multidrug-resistant (MDR) Gram-negative ‘superbugs’ Pseudomonas aeruginosa, Acinetobacter baumannii, and Klebsiella pneumoniae and Gram-positive methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant S. aureus (VRSA), and mobilized colistin resistance gene (MCR)-producing Enterobacteriaceae bacteria, which are resistant to almost all available antibacterial drugs [1]. The coronavirus disease 2019 (COVID-19) pandemic especially led to the increased clinical use of all antibiotics, which further promoted the development of bacterial resistance, highlighting the unmet medical need for new antibiotics [2].
Since the golden age of antibiotic discovery in the mid-20th century, natural products have served as the major foundation for the development of the majority of antibiotic drugs in clinical use to this very day [3]. Natural product antibiotics act by directly inhibiting the growth or killing the bacteria, acting as potentiators that augment or transform other agents or as immunomodulators to host cells or block pathogen virulence [1]. For example, a recent study from our group showed that two natural products, α-mangostin and isobavachalcone, could rapidly kill several types of MDR bacteria, including MRSA, VRSA, and MCR-producing Enterobacteriaceae bacteria in vitro [4].
Curcumin ((1E,6E)-1,7-bis-(4-hydroxy -3-methoxyphenyl)-hepta-1,6-diene -3,5-dione) is one of major active ingredients of turmeric extract, which is acquired from Curcuma longa, a type of herb belonging to the ginger family and widely growing in southern and south-western tropical Asia regions [5]. Curcumin is usually used as a coloring agent in foods or cooking. In China, curcumin has been approved as a food additive to improve animal’s productive performance [6]. Curcumin has been shown to possess direct broad spectrum antibacterial activities against Gram-negative and Gram-positive bacteria [6,7,8,9,10]. Curcumin also acts as an immunomodulator whereby it ameliorates bacterial infections by blocking the pathogen’s virulence factors and augmenting host-mediated immunity [11]. As a potential broad-spectrum antibacterial adjuvant to permeabilize the bacterial membrane, curcumin has a marked synergistic or additive anti-bacterial activity in combination with some traditional antibacterial drugs (e.g., polymyxin B, colistin, ciprofloxacin, and tetracycline) and natural active substances (e.g., berberine, and epigallocatechin gallate) [9,12]. Animal studies showed that topical curcumin was an effective treatment for localized trauma-caused skin infections [13]. Importantly, human trials showed that oral administration of curcumin was safe and effective for skin diseases, including psoriasis, infection, acne, skin inflammation, and skin cancer [14]. Bacterial infections also contribute to tumor formation, and curcumin combinations with some drugs have both anti-cancer and antibacterial activities that would provide a novel thyroid cancer treatment strategy [15]. In the present review, we survey the literature on the antibacterial properties and current underlying molecular mechanism of curcumin per se, curcumin-based combinations, and its nano-formulations, clinical trials, and major challenges, aiming to provide a prospective into the further clinical application of this promising antimicrobial candidate or broad-spectrum antibacterial adjuvant.
2. Chemical Structure and Biological Activity of Curcumin
The chemistry and structure of curcumin was first characterized in 1910 by Lampe and Milobedeska. Three years later, in 1913, they reported the synthesis of curcumin and confirmed its structure [16]. In 1953, Srinivasan reported the separation and quantification of components of curcumin using chromatography [17]. Curcumin is a mixture largely composed of three hydrophobic curcuminoids, namely, demethoxycurcumin (DMC), bisdemethoxycurcumin (BDMC), and curcumin, in the proportion of 17: 3: 77 (Figure 1) [18]. From a structural viewpoint, curcumin, DMC, and BDMC all contain two aromatic feruloyl rings with orthomethoxy phenolic OH groups (Figure 2). The highly polar aromatic rings are symmetrically connected via a seven-carbon aliphatic chain and two α, -unsaturated carbonyl groups (e.g., β-diketonemoiety) [19]. This seven-carbon aliphatic chain structure is responsible for the hydrophobic nature of curcumin, which makes it practically insoluble in water; however, solubility can be achieved in ethanol, dimethyl sulfoxide (DMSO), methanol, and acetone [19]. Curcumin displays a maximum ultraviolet (UV)-absorption (λmax) peak at 430 nm, which is due to the two feruloyl aromatic ring structure [19]. Curcumin has two molecular configurations, bis-keto and enolate. Its bis-keto form predominates under acidic, neutral, and solid phase conditions, whereas its enolic form is predominantly found under alkaline conditions [20].
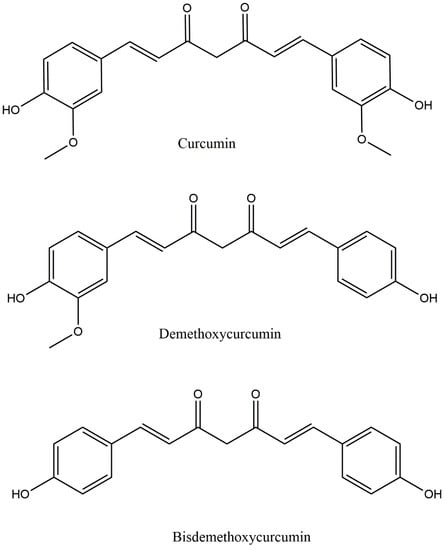
Figure 1.
Chemical structures of curcuminoids.
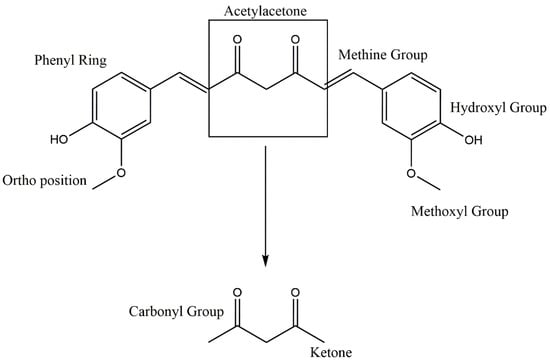
Figure 2.
Functional groups in the molecular structure of curcumin.
Documented biological activities of curcumin include antimicrobial, antioxidant, anti-inflammatory, neuroprotective, anticancer, and immuno-modulatory activities [20]. Due to its various biological activities, curcumin has been used extensively in traditional medicine for the treatment of various illnesses including autoimmune, neurological, diabetic, cardiovascular, and infectious disease [5,21]. In the proceeding discussions, we elaborate on the antibacterial activities of curcumin, its mechanism of action, and barriers associated with its clinical application as an antibiotic therapy.
3. Antibacterial Activity of Curcumin
In 1949, Schraufstatter and colleagues were the first to report the antibacterial properties of curcumin [22]. In the past seventy years, there have been several studies of the broad-spectrum inhibitory effect that curcumin exhibits against various Gram-negative and Gram-positive bacteria, including A. baumannii, E. faecalis, K. pneumoniae, P. aeruginosa, Bacillus subtilis (B. subtilis), Staphylococcus epidermidis, Bacillus cereus (B. cereus), Listeria innocua, Streptococcus pyogenes, S. aureus, Helicobacter pylori (H. pylori), Escherichia coli (E. coli), Salmonella enterica serotype Typhimurium, and Streptococcus mutans (Details shown in Table 1) [6,8,10,23,24]. Importantly, curcumin also exhibits marked antibacterial activities against MDR-isolates, such as polymyxin-resistant K. pneumoniae and MRSA [9,10,24]. A recent study by Batista de Andrade Neto et al., reported that minimum inhibitory concentration (MIC) values for curcumin against clinical isolates of MRSA were in the range of 125–500 μg/mL [25]. Another study by Yasbolaghi Sharahi et al., reported that MICs of curcumin against MDR-A. baumannii, P. aeruginosa and K. pneumoniae were in the range of 128–512 μg/mL [8]. Notably, there were significant differences in the MICs of curcumin against certain stains reported by different research groups [26]. This may be due to the difference in solubility of curcumin in the different vehicles (e.g., water, DMSO, and ethanol) used by each research group [26]. In addition, these differences may be related to the MIC test methodology, impact of the vehicle against the bacterial outer membrane, and purity of the curcumin used in the study [27].

Table 1.
Documented antibacterial activities of curcumin.
4. Mechanisms of the Antibacterial Action of Curcumin
4.1. Cell Membrane Disruption
Curcumin and its two analogs, DMC and BDMC, have been shown to possess antibacterial activity against a wide range of bacteria [23]. Studies have shown that curcumin can damage the permeability and integrity of bacterial cell membranes in both Gram-positive and -negative bacteria, finally leading to bacterial cell death [47]. Curcumin’s lipophilic structure allows it to directly insert into liposome bilayers, which in turn enhances the bilayer permeability [47]. Solid-state nuclear magnetic resonance (NMR) spectroscopy studies revealed that curcumin can insert deep into the membrane in a trans-bilayer orientation, resulting in disordering 1,2-dipalmitoyl-sn-glycero-3-phosphocholine (DPPC) membranes and influencing exocytotic and membrane fusion processes [48]. Tyagi et al., demonstrated that curcumin at a concentration of 100 μM can induce permeabilization of both S. aureus and E. coli cell walls [49]. This membrane permeabilization property could account for the direct bacterial killing effect of curcumin against Gram-positive and -negative bacteria [49]. Indeed, the increase in membrane permeabilization of bacteria caused by curcumin could increase the uptake of other drugs [50]. This is a critical mechanism to explain the synergistic effect of curcumin combination therapy with other antibiotic drugs or natural products, as discussed in detail below.
4.2. Inhibition of Bacterial Quorum Sensing System and Biofilm Formation
Quorum sensing (QS) system is a cell–cell communication system that is ubiquitously used in microbial communities to monitor their population density and adapt to external environment [51]. To date, there are three main QS systems, (1) the acylhomoserine lactone (AHL) QS system in Gram-negative bacteria; (2) the autoinducing peptide (AIP) QS system in Gram-positive bacteria, and (3) the autoinducer-2 (AI-2) QS system, which is in both Gram-negative and Gram-positive bacteria. It is well-known that QS systems play critical role in the formation and maturation of bacterial biofilms, which are associated with about 80% microbial infections [52]. Bacteria growing in biofilms are largely protected from antibiotics or host immune cells, leading to the failure of antimicrobial therapy [52]. QS systems are the master controllers for the entire process of biofilm formation, including bacterial adhesion, biofilm development, and maturation. Therefore, the discovery of new inhibitory compounds targeting bacterial QS systems is an important strategy to control bacterial biofilm formation and resistance.
Several studies reported that curcumin inhibits bacterial QS systems/biofilm formation and prevents bacterial adhesion to host receptors in various species, including S. aureus, E. faecalis, E. coli, Streptococcus mutans, Listeria monocytogenes, H. pylori, P. aeruginosa, Serratia marcescens, Aeromonas hydrophila and A. baumannii [36,38,50,53,54,55]. We have summarized the QS system’s curcumin targets in various bacteria in Table 2. In addition, Figure 3 provides an overview of the inhibitory mechanisms of curcumin against biofilm formation, inhibition of bacterial swimming/clustering behaviors, and inhibition of virulence [35,36,38,50,53,54,55]. Interestingly, available data suggest that the autoxidation of curcumin could also contribute to the inhibition of biofilm formation [56]. For example, curcumin was shown promote the production of lactate dehydrogenase (LDH) in P. aeruginosa, S. aureus, and E. faecalis, wherein the curcumin/LDH complex exhibited antibacterial and anti-biofilm activities [56]. Clearly, the anti-biofilm properties of curcumin increase its potential as a tractable anti-infective agent.

Table 2.
Targets or action model of curcumin in the inhibition of biofilm in various bacteria.
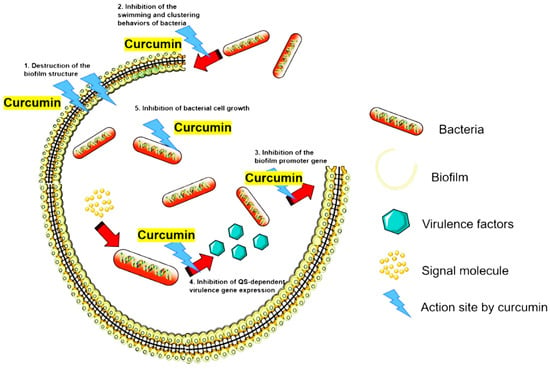
Figure 3.
Inhibitory effect of curcumin against the bacterial quorum sensing (QS) system. The main mechanisms of curcumin in QS inhibition involve (1), destruction of the biofilm structure; (2) inhibition of bacterial swimming and clustering behavior; (3) inhibition of the expression of biofilm promotor genes; (4) inhibition of the gene expression of QS-dependent virulence; (5) inhibition of bacterial cell growth [35,36,38,50,53,54,55].
4.3. Inhibition of Cell Division
Inhibition of bacterial cell division is an important mechanism of curcumin’s antibacterial activity [23,64]. Filament temperature-sensitive protein Z (FtsZ) is shown to be essential for bacterial cell division [64,65]. It consists of an N-terminal polymerization domain connected to a highly conserved C-terminal peptide (CCTP) of ~eight amino acids by an intrinsically disordered linker region of variable length (50 amino acids in E. coli). FtsZ associates in a GTP-dependent manner to form polymers [64]. This process is coupled to the conversion between closed and open conformations of FtsZ and plays a critical role in the formation of the Z ring of FtsZ. The polymerized FtsZ filaments attach to the cytoplasmic membrane through membrane anchors ZipA and FtsA, mediated by the CCTP of FtsZ (Figure 4). Rai et al., showed that curcumin blocks the formation of the cytokinetic Z ring through direct interaction with FtsZ in B. subtilis and E.coli [64]. In addition, curcumin also increased the GTPase activity of FtsZ, which in turn aborted the polymerization process [64]. Molecular docking of curcumin to the E. coli FtsZ structure suggests binding occurs within the GTPase catalytic pocket, with the curcumin molecule making key contacts with Gly20, Gly21, Gly109, Thr132, and Asn165 and residues at the sites of Gly21, Gly22, Gly72, Thr133, and Asn166 in B. subtilis FtsZ (Figure 4) [66]. More recently, Morão et al., showed that a molecular simplified version of curcumin where its β-diketone moiety had been substituted with a monocarbonyl group could disrupt the divisional septum of B. subtilis without exerting a direct inhibition of FtsZ. These findings suggest that the simplified curcumin exerts its antibacterial action largely through membrane permeabilization, with disruption of the membrane potential necessary for FtsZ intra-cellular localization [23].
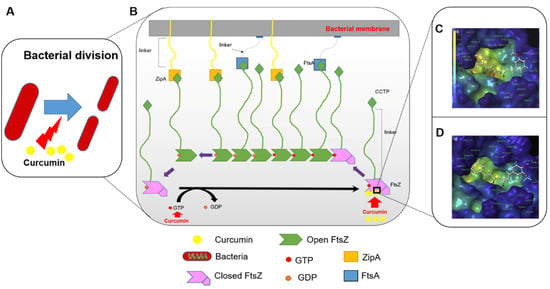
Figure 4.
Curcumin inhibits the bacterial division by blocking FtsZ assembly. (A), working model of curcumin for the inhibition of bacterial division. (B), curcumin can activate the activity of GTP and interact with FtsZ, blocking the FtsZ assembly [65]. (C,D), the interaction site of FtsZ with curcumin in E. coli and B. subtilis strains, respectively [66].
4.4. Induction of Oxidative Stress and Programmed Cell Death
Traditionally, programmed cell death (PCD) is an important biological and pathological process in the life-cycle of eukaryotic multicellular organisms [67]. Similarly, monocellular organism such as bacteria can activate signaling pathways, leading to cell death within a colony. In bacteria, many factors, including stress response, developmental phase, genetic transformation, and biofilm formation contribute to the induction of bacterial programmed apoptotic-like death processes [67]. The physiological and biochemical hallmarks of apoptotic-like death in terminally stressed E. coli involve the production of reactive oxygen species (ROS), chromosomal condensation, extracellular exposure of phosphatidylserine, DNA fragmentation, membrane potential (ΔΨ) dissipation, and loss of structural integrity, all markers of eukaryotic apoptosis [68].
ROS-mediated cell death results from the damaging effects of the superoxide anions (O2•−), hydrogen peroxide (H2O2), and hydroxyl radicals (OH•) on bacterial cellular components (DNA, membrane lipids, and proteins) [69]. Curcumin at MIC concentrations induces the production of ROS in bacterial cells, resulting in an apoptosis-like response in E. coli, including the accumulation of ROS, membrane depolarization, and increase of Ca2+ influx [50]. At the genetic level, curcumin induced the upregulation of RecA protein expression, which mediates apoptotic-like death processes in bacteria [50]. In line with this finding, E. coli RecA knock-outs displayed curcumin resistance, consolidating the conclusion that curcumin-induced cell death in E. coli is dependent on apoptotic pathways [50]. In addition, curcumin has been shown to downregulate the expression of genes that mediate the SOS response in bacteria, which rescues the cell from DNA damage and is involved in biofilm formation and division [68]. LexA is a DNA-binding transcriptional repressor that regulates genes involved in the SOS response [70]. Recent studies indicated that curcumin inhibited the SOS responses caused by UV-induced DNA damage in Salmonella typhimurium and E. coli by suppressing the expression of LexA. The inhibitory effects of curcumin on biofilm formation and cell division mentioned above are likely associated with its inhibitory effects on the bacterial SOS response. Curcumin has also been shown to directly interact with bacterial DNA to produce a bacteriostatic effect [50]. We have provided an overview of curcumin-induced bacterial cell death in Figure 5.
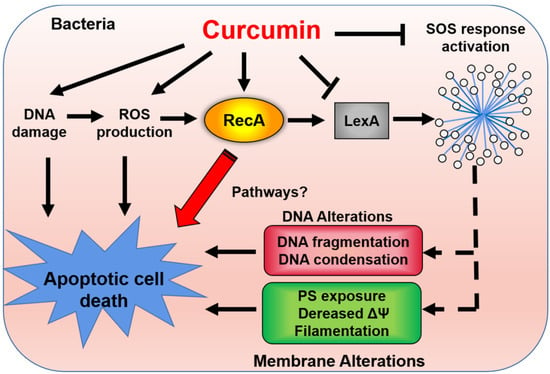
Figure 5.
Curcumin induces oxidative stress, DNA damage, and apoptotic-like death in bacterial cells. Stress caused by drugs or other factors in bacterial cells could induce the production of ROS or DNA damage, following by activating the expression of RecA, DNA fragmentation, decreased membrane potential (ΔΨ), and formation of filamentation, finally leading to cell death. In response to this stress, the SOS response network is usually activated and plays a protective role in bacterial survival [68].
4.5. Phototoxicity
Curcumin absorbs blue light in the range of 455–460 nm and can be employed as an effective photosensitizer to promote the success of photodynamic processing [19]. This photosensitizing property has been exploited to induce phototoxicity in Gram-positive and -negative bacterial cells under blue light irradiation [50,71]. It is noteworthy to mention that Gram-positive bacteria are known to be more sensitive and are easily killed by photosensitizers compared to Gram-negative bacteria [50]. This difference may be related to the more robust outer membrane structure of Gram-negative bacteria compared to the more porous cytoplasmic membrane structure of Gram-positive cells in which allowed photosensitizers more easily penetrate into cells [72]. Recently, it was found that ethylene diamine tetraacetic acid (EDTA), which permeabilizes the cell membrane, could significantly enhance the antibacterial effect of blue light-activated curcumin in S. aureus and S. mutans cells [73].
In the past 10 years, researchers have developed a working understanding of the molecular mechanisms of curcumin-induced phototoxicity, although the precise molecular mechanism is still unclear [19]. It has been demonstrated that the antibacterial effect of blue light-activated curcumin involves an autoxidation to generate ROS, which in turn damage lipids, protein, and DNA, finally leading to bacterial cell death [50]. Jiang et al., showed that blue light-activated curcumin could significantly increase the levels of intracellular ROS and membrane damage in S. aureus [74]. A recent study showed that curcumin-mediated phototoxicity involves the direct induction of DNA damage and protein degradation, eradication of biofilms and inhibition of virulence genes (e.g., inlA, hlyA, and plcA) in Listeria monocytogenes [75]. Chen et al., showed that the process of curcumin-mediated phototoxicity is temperature dependent [76]. Very recently, it was reported that curcumin could be employed as a coating on the surface of the endotracheal tube, (which was considered the primary cause of ventilator-associated pneumonia), capable of a robust photodynamic inactivation under blue light activation (at 450 nm) against E. coli, S. aureus, and P. aeruginosa [77]. This photodynamic activity of curcumin provided a novel application in avoiding ventilator-associated pneumonia in patients.
4.6. Curcumin Perturbs Bacterial Cell Metabolism
Many antibiotics, such as β-lactams, aminoglycosides, and quinolones, have been widely used in clinical practice, and the primary mechanisms of action have been well-established [1]. However, more recent metabolomics studies from high-throughput technologies have indicated that, in addition to these distinct mechanisms, subsequent metabolic changes that occur downstream of the interaction of the antibiotics with their primary targets also play an important role in their antibacterial-killing mechanism [78]. It has been reported that L-serine supplementation could sensitize E. coli to gentamicin by promoting the production of NADH and ROS production, which also mediated the bacterial killing of curcumin [79]. Adeyemi et al., reported that curcumin treatment of S. aureus impacts the levels of kynurenine, nitric oxide, and total thiol levels, indicating that perturbations in the aforementioned metabiotic pathways contribute to the antibacterial killing mechanism of curcumin [80]. The activation of the kynurenine pathway likely produces a decrease in the cellular L-tryptophan pool available to support bacterial growth, thereby starving bacterial cells of an essential nutrient [80].
4.7. Curcumin Regulates Intracellular Bacterial Proliferation
Curcumin is a powerful immune-regulator, with a proven ability to modulate host defenses against intracellular bacterial infections [81]. Marathe et al., showed that pre-treatment of macrophages with curcumin attenuated Listeria monocytogenes and Shigella flexneri intracellular infection, albeit the pre-treatment had the opposite effect on infection by Salmonella enterica serovar Typhimurium, S. aureus, and Yersinia enterocolitica, which were aggravated by curcumin [81]. This differential effect may be attributed to the membrane-stabilizing effect of curcumin wherein S. enterica serovar Typhimurium, S. aureus, and Y. enterocolitica have acquired machinery that inhibits the fusion of the pathogen-containing vacuole with lysosomes [82]. By contrast, Listeria monocytogenes and S. flexneri in the host cells can escape into the cytosol and prevent lysosomal degradation [83]. Recent studies also indicated that curcumin can protect human macrophages against Mycobacterium tuberculosis infection by inducing apoptosis, autophagy, and the activation of nuclear factor-kappa B (NF-κB) [84]. To date, the key targets of curcumin in the host that governs the growth and proliferation of intracellular pathogens are still unclear, and the precise molecular mechanisms require further investigation.
5. Synergistic Antibacterial Effects of Curcumin with Antibacterial or Non-Antibacterial Agents
Synergistic antibacterial effects between antibiotics are strictly defined microbiological phenomena, requiring two bioactive agents to exhibit a greater effect in bacterial killing than the added effects of each constituent [85].
Several studies have shown that curcumin exhibits synergistic antibacterial effects when combined with traditional antibacterial agents (e.g., polymyxins, meropenem, oxacillin, tetracycline, ciprofloxacin, ampicillin, norfloxacin), natural products (e.g., epigallocatechin gallate, berberine) or metals (e.g., Cu2+, Zn2+, and Fe3+) [86,87,88,89]. In the proceeding discussion, we summarized these potential combinations and discussed their various mechanisms of action.
5.1. Synergistic Effect between Curcumin and Antibacterial Agents
5.1.1. Curcumin and Polypeptide Antibacterial Drugs
In the clinic, vancomycin and polymyxins (including polymyxin B and E, also called colistin) are commonly employed as antibacterial drugs against MDR Gram-negative and Gram-positive bacteria, respectively [90]. The emergence of polymyxin- and vancomycin-resistant bacteria has posed a huge challenge and medical burden.
The well-accepted primary mechanism of action of polymyxins is through spatially displacing the cations (e.g., Ca2+ and Mg2+) in the Gram-negative outer membrane and binding to the lipid A component of the lipopolysaccharide (LPS), subsequently disrupting the stability of both the outer and inner membranes, ultimately leading to bacterial cell lysis [91]. Recent studies also indicated that polymyxins can also induce the production of excessive ROS (i.e., OH•) in bacterial cells, leading to oxidative stress-dependent cell death [92]. Polymyxin B in combination with curcumin showed a marked synergetic effect against polymyxin-susceptible and -resistant Gram-positive (e.g., Enterococcus, S. aureus, and Streptococcus) and Gram-negative (e.g., A. baumannii, E. coli, P. aeruginosa, and S. maltophilia) bacterial isolates associated isolated from traumatic wound infections [32]. This synergistic effect may be due to curcumin’s ability to permeabilize the outer membrane, which facilitates the entry of the secondary agent to enter the bacterial cells and cause cell death [24]. In addition, this synergistic effect could be attributed to the inhibitory effect of curcumin on the activities of efflux pumps [9,24]. Curcumin and polymyxin combination treatment for bacterial infections may have another advantage, i.e., significant improvement in the therapeutic index of polymyxins by additionally inhibiting polymyxin-induced cytotoxicity, neurotoxicity, and nephrotoxicity, which is beyond antibacterial activity [93]. This combination may have a powerful application in clinical practice and warrants clinical trials.
Vancomycin is a glycopeptide antibiotic that inhibits a specific step in the synthesis of the peptidoglycan layer in Gram-positive bacteria. It has been reported that curcumin combined with vancomycin showed a synergistic effect against MDR clinical K. pneumoniae isolates [94]. This potential mechanism may be dependent on the synergistic effect of cell membrane permeability [94]. Moreover, curcumin could also attenuate vancomycin-induced nephrotoxicity by inhibiting oxidative stress and the inflammation response in a rat model [94].
5.1.2. Curcumin and β- Lactam Antibacterial Drugs
β-lactam antibiotics are the most widely used antibacterial agents worldwide. β-lactamases confer significant antibiotic resistance to their bacterial hosts by hydrolyzing the amide bond of the four-membered β-lactam ring of β-lactam antibiotics, which include four classes of drugs, i.e., penams (penicillins), cephems (cephalosporins), monobactams, and carbapenems [95]. It has reported that a curcumin and meropenem combination displayed markedly synergistic or additive effects against antibiotic-susceptible and -resistant Gram-positive (E. faecalis) and carbapenem-associated MDR A. baumannii, P. aeruginosa, and K. pneumoniae isolates via the observation of MICs [86]. A report by Yadav et al., showed that a water-soluble curcumin derivative could reverse meropenem resistance by targeting the activity of carbapenemases and the AcrAB-TolC multidrug efflux pump system [96]. Mun et al. showed that a curcumin combination with oxacillin and ampicillin exhibited a marked synergistic effect against S. aureus ATCC (American Type Culture Collection) 25,923 (methicillin-sensitive strain) [97]. Similarly, in another study, BDMC in combination with oxacillin showed a marked synergistic effect against S. aureus ATCC 33,591 (methicillin-resistant strain) and clinical MRSA isolates [98]. The potential mechanism may be dependent on the expression of the mecA gene that encodes penicillin-binding protein 2a (PBP2a), which governs the resistance of MRSA isolates to β-lactam antibiotics [98]. Sasidharan et al. found that curcumin in combination with third-generation cephalosporins (e.g., cefaclor, cefodizime, and cefotaxime) showed marked synergistic effect against S. aureus, B. subtilis, and E. coli, which are also associated with infectious diarrhea [87]. There was no increased toxic effects between these combinations [87]. These results indicated curcumin and cephalosporin combination are promising therapeutic options for infectious diarrhea disease.
5.1.3. Curcumin and Aminoglycoside Antibacterial Drugs
Aminoglycosides are potent, broad-spectrum antibiotics that act through inhibition of protein synthesis by irreversibly binding to 30S ribosomal subunits [99]. A report by Teow et al., stated that curcumin in combination with two aminoglycoside antibiotics (e.g., amikacin and gentamicin) showed a powerful synergistic effect against S. aureus strains, and these synergistic effects were stronger than that of curcumin in combination with ciprofloxacin [100]. Notably, this difference in synergistic effect may be related to the difference in the primary targets between quinolone and aminoglycosides against bacteria [101]. The potential action mechanism is related to the inhibition of biofilm formation, which was evident by the significant inhibition of their combination of the swarming motilities and the mRNA expression of several key QS regulatory genes (e.g., lasI, lasR, rhlI, and rhlR) [100]. In addition, it has been reported that curcumin can also attenuate gentamicin-induced nephrotoxicity and neurotoxicity by inhibiting oxidative stress and cell apoptosis in a rat model [102]. Therefore, the combination between curcumin and aminoglycosides can not only improve the antibacterial effectiveness but can also decrease the toxic effects of gentamicin.
5.1.4. Curcumin and Macrolide Antibacterial Drugs
Azithromycin is a macrolide antibiotic, which can exhibit a good antibacterial effect by inhibiting bacterial protein synthesis, quorum-sensing, and the formation of biofilms. In clinical practice, azithromycin has been used in treating respiratory, urogenital, dermal, and other bacterial infections [103]. Bahari et al., found that curcumin in combination with azithromycin showed a synergistic effect against P. aeruginosa PAO1, and the value of FICI was 0.25 [100]. The potential action mechanism may be similar to the above-mentioned combination of curcumin and gentamicin [100]. Erythromycin is in a class of medications called macrolide antibiotics. The action mechanism involves the blockade of bacterial growth. In a rat model, oral administration of curcumin (50 mg/kg) and erythromycin (20 mg/kg) significantly inhibited the growth of MRSA isolates in bone tissue compared to either administered alone [11]. The curcumin and erythromycin combination also significantly alleviated bone infection and the inflammatory response [11].
5.1.5. Curcumin and Quinolone Antibacterial Drugs
There was a marked synergistic effect in curcumin combination with two quinolone antibiotics (e.g., ciprofloxacin and norfloxacin) against the S. aureus ATCC 33,591 strain and clinical MRSA isolates [97]. On the contrary, curcumin treatment reduced the antimicrobial activity of ciprofloxacin against Salmonella typhimurium and Salmonella typhi [97]. This may be related to the antioxidant property of curcumin and its inhibition of the expression of interferon γ (IFNγ) in vitro and in a mouse model [97].
5.2. Curcumin and Natural Products
5.2.1. Curcumin and Berberine
Berberine is a benzylisoquinoline alkaloid compound and has antimicrobial properties against both Gram-negative and Gram-positive bacteria [104]. Berberine has been widely used in traditional Chinese and native American medicines. FtsZ protein is an important target of berberine in inhibiting bacterial division [105]. Interesting, co-encapsulation of berberine and curcumin in liposomes decreased their MICs against MRSA by 87% and 96%, respectively, as compared to their free forms, with an FICI of 0.13, indicating a synergistic effect [88]. However, the synergistic effect in their combination in native form was not detected. In addition, co-treatment of berberine and curcumin in liposomes also significantly improved intracellular infection and the inflammation response in macrophages following MRSA infection. Mechanically, the synergistic effect between curcumin and berberine is partly dependent on the inhibition of biofilm formation and improvement of their solubilities [88]. Additionally, similar to curcumin, berberine is also an FtsZ inhibitor and inhibits bacterial cell division [104]. Therefore, this synergistic effect between curcumin and berberine may also be partly dependent on the inhibition of FtsZ assembly.
5.2.2. Curcumin and Epigallocatechin Gallate
Epigallocatechin-3-gallate (EGCG) is a polyphenol found in green tea, which, similar to curcumin, has been linked with health benefits and has significant antimicrobial activity against some MDR pathogens, including MDR S. maltophilia, A. baumannii, and S. aureus [106]. In vitro, it has been found that curcumin in combination with EGCG exhibited a marked synergistic effect against MDR A. baumannii [107]. A possible explanation for the synergy between curcumin and EGCG could be disruption of the outer membrane and facilitation of curcumin to enter bacterial cells [108]. In another study, it was suggested that inhibition of acylhomoserine lactone-mediated biofilm formation may contribute to this synergistic effect, and investigations of precise mechanisms are still required [109].
5.3. Curcumin and Metals
Many metals have been used as antimicrobial agents due to the antiquity and potential molecular mechanism involved in oxidative stress, protein dysfunction or membrane damage in bacterial cells [110]. A copper (II) sulfate pentahydrate–curcumin complex (Cu-CUR), iron (III) nitrate nonahydrate–curcumin complex (Fe-CUR), and zinc (II) chloride–curcumin complex (Zn-CUR) all significantly inhibited cell growth in P. aeruginosa PAO1 compared to curcumin treatment alone [111,112]. Furthermore, the authors found that the Cu–CUR complex significantly inhibited the formation of the biofilm and the production of QS-related virulence factors of P. aeruginosa PAO1 [89]. Consistently, the synergistic activity of curcumin and silver/copper nanoparticles (NPs) was detected against the cell growth and biofilm formation of S. aureus and P. aeruginosa compared to curcumin, AgNPs or CuNPs alone [113]. These marked synergistic effects may be related to the improvement of curcumin or intracellular uptake of curcumin [114].
6. Safety of Curcumin
Curcumin has been proven to be safe and tolerable across various animal studies as well as clinical trials [115,116,117]. Orally administered curcumin at the dose of 50, 250, 480, and 1300 mg/kg body weight for 13 weeks did not exhibit acute toxicity in rats [118]. However, some abnormal effects including increased liver weight, stained fur, discolored faces, and hyperplasia of mucosal epithelium in the cecum and colon were observed in animals from the highest dosage group (2600 mg/kg body weight). Orally administered curcumin at 100, 200 or 400 mg/kg/day has been shown to effectively inhibit acute liver damage, nephrotoxicity, and nerve damage caused by colistin, aflatoxin B1, carbon tetrachloride, and cadmium [21,119,120,121,122] in rat or mouse models. In an infection model, oral administration of curcumin at 25 or 50 mg/kg body weight for two weeks could significantly ameliorate the H. pylori infection-induced inflammation response in gastric tissues of mice [123]. A phase I human trial showed that oral administration of curcumin in some cancer patients at a dose of 8 g/day for three months did not show any adverse effects, albeit some adverse effects were detected when the patients were administered a higher dose of 12 g/day [124]. The results of a 4-month phase I clinical trial in cancer patients showed that oral curcumin at a dose of 3.6 g/day significantly inhibited levels of serum prostaglandin E 2 (PGE2) production, a biomarker of the inflammatory response. Notably, no adverse effects were reported in the curcumin treatment cohort [125]. Consistently, a triple blinded clinical trial showed that a combination of 500 mg curcumin (equal to 8.33 mg/kg body weight) and 40 mg famotidine daily for one month significantly decreased the rate of H. pylori infection in patients [126]. Collectively, these studies indicated that the therapeutic dose of curcumin is far lower than the dosages at which toxicity is observed, thus giving curcumin a good therapeutic index.
7. Nano-Formulations of Curcumin
Curcumin has low water solubility (about 11 ng/mL), which results in its poor bioavailability under oral consumption [127]. Additionally, curcumin degrades rapidly, resulting in low concentrations in the blood or organs of the body, making it difficult to reach the effective concentration to treat the bacterial infection in the liver, lungs, or other organs [128]. To overcome this insufficiency of bioavailability, scientists have developed various nan-formulations of curcumin, such as lipid-based nanocarriers (e.g., liposomes, solid lipid nanoparticles, nanostructured lipid carriers, and nano-emulsion), biopolymers (e.g., nanocomposite, polymeric nanoparticles, hydrogel, and polymeric micelles), technique-based nanoparticles (e.g., spray-dried nano-formulation of curcumin, and nanofibers), and other miscellaneous types of nanocurcumin (curcumin nanocrystals, quantum dots, and graphene oxide) [18,129,130,131,132]. In addition, nanomaterial-based combinations of curcumin with other anti-bacterial agents that are effective against bacteria were also developed. Most of them are used in cancer therapy [133]. Here, we summarized the main types of nanocurcumin that are applied due to their antibacterial effect, as shown in Table 3. Their special characteristics and antibacterial activities have been well-described and addressed (see Sharifi et al.’s review paper) [132]. It is notable that there was no clinical trial to test the effectiveness of these various nano-formulations of curcumin, although they exhibited a better antibacterial effect in vitro and animal experiments by improving the solubility and biocompatibility. Therefore, more clinical trials are still required.
In addition, beyond the development of nano-formulations, other types of new formulations (e.g., inclusion technology, solid dispersion technology, microspheres, and microcapsules) were also developed to improve the solubility and bioavailability of curcumin. For example, Yaday et al. found that various cyclodextrin (CD) complexes of curcumin could enhance the solubility of curcumin > 100-fold compared with curcumin per se in water [134]. However, similar to the nano-formulations, the development of these new formulations of curcumin remains at the laboratory research stage, and there are no necessary clinical studies.

Table 3.
Nano-formulations of curcumin and their antibacterial effects.
Table 3.
Nano-formulations of curcumin and their antibacterial effects.
| Type (or Name Present in Published Literatures) | Preparation and Characterizations | Improvement in Antibacterial Activity (Accessed by MICs or Biofilm Formation) | Reference |
|---|---|---|---|
| Curcumin nanoparticles (curc-np) | Curcumin was encapsulated into a silane-hydrogel nanoparticle vehicle. Average hydrodynamic diameter at the range of 222 ± 14 nm. | In vitro, curc-np significantly inhibited the growth of MRSA and P. aeruginosa isolates compared to native curcumin. In a mouse model: significantly reduced bacterial burden in MRSA-infected burn wounds compared to native curcumin administration. | [29] |
| Nanoparticles of curcumin (nanocurcumin) | A wet-milling technique was used to make the particle size of curcumin 2–40 nm, and nanocurcumin was freely dispersible in water. | The MICs of nanocurcumin in water were 100 μg/mL, 75 μg/mL, 250 μg/mL, 200 μg/mL, 350 μg/mL against S. aureus, B. subtilis, E. coli, P. aeruginosa, A. niger, much higher than native curcumin in DMSO (the corresponding MICs were 150, 100, 300, 250 and 400 μg/mL). | [28,130] |
| Microcapsule curcumin | Microcapsule curcumin could be prepared with gelatin and porous starch as a wall system by a spray-drying method. The size was not reported. | The MICs were 250, 250, 62.5, 125, 125, 15,7, 31.3 and 31.3 μg/mL against E.coli, Yersinia enterocolitica, S. aureus, B. subtilis, B. cereus, A. niger, P. notatum, and S. cerevisiae. There was no comparation with native curcumin. | [135] |
| Sodium carboxylmethyl cellulose silver nanocomposite films-curcumin (SCMC-SNCF-CM) | SCMC-SNCF were developed from sodium carboxylmethyl cellulose (SCMC), N, N1-methylenebisacrylamide (MBA), and silver nitrate solution. Curcumin loading into SCMC–SNCF was achieved by a diffusion mechanism. The size was not reported. | SCMC-SNCF-CM composite showed 86% inhibition growth against E. coli. There was no comparation with native curcumin. | [136] |
| Curcumin Quantum Dots (CurQDs) | A newer two-step, bottom-up wet milling approach was used to prepare curcumin quantum dots (CurQDs), and acetone was used as a primary solvent. Average size was about 2.5 nm | The MIC of CurQDs significantly decreased to the range of 1.96–15.65 μg/mL from 175–300 μg/mL for native curcumin against all tested bacteria, including S. aureus, MRSA, E. faecalis, K. Pneumoniae, and P. aeruginosa | [137] |
| Poly-(lactic-co-glycolic acid) Curcumin nanocapsules(PLGA-CUR-NCs) | Curcumin (CUR) nanocapsules (NCs) were prepared by the solvent displacement method with some modifications. The detailed information has been described in a published paper. The solubility in water increased to 591–928 μg/mL, and its solubility could be regulated by changes in the oil and water ratio. The sizes were in the range of 100–1000 nm, dependent on the ratio of glucose. | The MICs of PLGA-CUR-NCs against E. coli, Salmonella, and P. aeruginosa decreased from 300 μg/mL to 100 μg/mL, and against S. aureus, B. sonorensis, and B. licheniformis decreased from 100 μg/mL to 75 μg/mL. | [138] |
| Nano-sized particles of curcumin | Colloids of curcumin nanoparticles with an average diameter of 20–40 nm were prepared in accordance with the method (a wet-milling technique). | Nano-curcumin could enhance the inhibition of biofilm formation in P. aeruginosa. | [139] |
| Cur/PVA/collagen composite films (CPCF) | A composite film (CPCF) containing curcumin nanoparticles, collagen, and polyvinyl alcohol (PVA). The diameter and polydispersity of the Cur/poly(ε-caprolactone)-poly (ethylene glycol)-poly(ε-caprolactone) PCEC nanoparticles were 43.63 ± 13.22 nm and 0.334 ± 0.403 nm, respectively. | There was no marked change in the MICs. The cytotoxicity of CPCF significantly decreased in human skin fibroblasts compared to native curcumin. | [140] |
| Curcumin-chitosan-zinc oxide (CCZ) | Curcumin and chitosan were layered on a hexagonal ZnO, and the particles were sized to about 48 ± 2 nm. | Increased antibacterial activity of the CCZ against MRSA and E. coli compared to native curcumin or ZnO. | [141] |
| Pectin/curcumin/sulfur nanoparticles films | pH-responsive pectin-based functional films were prepared by incorporating curcumin and sulfur nanoparticles (SNP). Curcumin and SNP were uniformly dispersed in the pectin to form a composite film. | The composite film exhibited enhanced inhibitory effect against E. coli and L. monocytogenes, with enhanced strong antioxidant activity. | [131] |
8. Conclusions and Perspectives
In the past decades, the potential molecular mechanisms of curcumin’s antibacterial activities have been extensively studied, involving the disruption of the bacterial membrane, the inhibition of the production of bacterial virulence factors and bacterial biofilm formation, induction of oxidative stress leading to programmed cell death, bacterial metabolic disturbance, and phototoxicity. These characteristics also contribute to explain how curcumin acts a broad-spectrum antibacterial adjuvant, which was evidenced by the markedly additively or synergistically effect with various conventional antibiotics or non-antibiotic compounds, such as antibacterial agents, natural products, and metals. Animal experiments and human clinical trials reveal that curcumin has high safety. However, unlike curcumin as a chemotherapy drug in cancer therapy, curcumin as a potential antibacterial therapy still has many challenges: (1) the critical targets of curcumin alone or combination in bacteria and precise molecular mechanisms are poorly understood; (2) the poor solubility, low bioavailability, and rapid degradation in humans or animals when curcumin was consumed orally; (3) no effective clinical trials. In order to overcome the poor solubility of curcumin, scientists have developed various curcumin nano-formulations and they indeed exhibited better solubility and antibacterial activity compared to native curcumin. However, there is a lack of evidence-based randomized investigation especially exploring the therapeutic roles of the nanocarrier-based delivery systems in enhancing anti-bacterial actions; therefore, much needs to be explored.
Author Contributions
Conceptualization, C.D.; software, J.L., Y.W. and H.L.; data curation, C.D.; writing—original draft preparation, C.D. and Z.S.; writing—review and editing, C.D. and T.V.; project administration, C.D.; funding acquisition, J.S. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by Guangdong Major Project of Basic and Applied Basic Research, No. 2020B0301030007. This study was also supported by the National Natural Science Foundation of China (Award number 32102724).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Theuretzbacher, U.; Outterson, K.; Engel, A.; Karlen, A. The global preclinical antibacterial pipeline. Nat. Rev. Microbiol. 2020, 18, 275–285. Available online: https://www.ncbi.nlm.nih.gov/pubmed/31745331 (accessed on 3 February 2022). [CrossRef] [PubMed] [Green Version]
- Lai, C.C.; Chen, S.Y.; Ko, W.C.; Hsueh, R.P. Increased antimicrobial resistance during the COVID-19 pandemic. Int. J. Antimicrob. Agents 2021, 57, 106324. [Google Scholar] [CrossRef] [PubMed]
- Rossiter, S.E.; Fletcher, M.H.; Wuest, M.W. Natural products as platforms to overcome antibiotic resistance. Chem. Rev. 2017, 117, 12415–12474. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Liu, Y.; Li, T.; Liu, X.; Hao, Z.; Ding, S.; Panichayupakaranant, P.; Zhu, K.; Shen, J. Plant natural flavonoids against multidrug resistant pathogens. Adv. Sci. 2021, 8, 2100749. Available online: https://www.ncbi.nlm.nih.gov/pubmed/34041861 (accessed on 3 February 2022). [CrossRef]
- Kocaadam, B.; Sanlier, N. Curcumin, an active component of turmeric (curcuma longa), and its effects on health. Crit. Rev. Food Sci. Nutr. 2017, 57, 2889–2895. Available online: https://www.ncbi.nlm.nih.gov/pubmed/26528921 (accessed on 3 February 2022). [CrossRef] [PubMed]
- Liu, M.; Lu, Y.; Gao, P.; Xie, X.; Li, D.; Yu, D.; Yu, M. Effect of curcumin on laying performance, egg quality, endocrine hormones, and immune activity in heat-stressed hens. Poult. Sci. 2020, 99, 2196–2202. [Google Scholar] [CrossRef]
- Praditya, D.; Kirchhoff, L.; Bruning, J.; Rachmawati, H.; Steinmann, J.; Steinmann, E. Anti-infective properties of the golden spice curcumin. Front. Microbiol. 2019, 10, 912. Available online: https://www.ncbi.nlm.nih.gov/pubmed/31130924 (accessed on 3 February 2022). [CrossRef] [Green Version]
- Yasbolaghi Sharahi, J.; Aliakbar Ahovan, Z.; Taghizadeh Maleki, D.; Riahi Rad, Z.; Riahi Rad, Z.; Goudarzi, M.; Shariati, A.; Bostanghadiri, N.; Abbasi, E.; Hashemi, A. In vitro antibacterial activity of curcumin-meropenem combination against extensively drug-resistant (xdr) bacteria isolated from burn wound infections. Avicenna J. Phytomed. 2020, 10, 3–10. [Google Scholar]
- Sundaramoorthy, N.S.; Sivasubramanian, A.; Nagarajan, S. Simultaneous inhibition of marr by salicylate and efflux pumps by curcumin sensitizes colistin resistant clinical isolates of enterobacteriaceae. Microb. Pathog. 2020, 148, 104445. [Google Scholar] [CrossRef]
- Taghavifar, S.; Afroughi, F.; Saadati Keyvan, M. Curcumin nanoparticles improved diabetic wounds infected with methicillin-resistant staphylococcus aureus sensitized with hamlet. Int. J. Low. Extrem. Wounds 2020. [Google Scholar] [CrossRef]
- Zhou, Z.; Pan, C.; Lu, Y.; Gao, Y.; Liu, W.; Yin, P.; Yu, X. Combination of erythromycin and curcumin alleviates staphylococcus aureus induced osteomyelitis in rats. Front. Cell. Infect. Microbiol. 2017, 7, 379. [Google Scholar] [CrossRef]
- Itzia Azucena, R.C.; José Roberto, C.L.; Martin, Z.R.; Rafael, C.Z.; Leonardo, H.H.; Gabriela, T.P.; Araceli, R.C. Drug susceptibility testing and synergistic antibacterial activity of curcumin with antibiotics against enterotoxigenic escherichia coli. Antibiotics 2019, 8, 43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marulasiddeshwara, R.; Jyothi, M.S.; Soontarapa, K.; Keri, R.S.; Velmurugan, R. Nonwoven fabric supported, chitosan membrane anchored with curcumin/TiO2 complex: Scaffolds for mrsa infected wound skin reconstruction. Int. J. Biol. Macromol. 2020, 144, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Panahi, Y.; Fazlolahzadeh, O.; Atkin, S.L.; Majeed, M.; Butler, A.E.; Johnston, T.P.; Sahebkar, A. Evidence of curcumin and curcumin analogue effects in skin diseases: A narrative review. J. Cell Physiol. 2019, 234, 1165–1178. [Google Scholar] [CrossRef] [PubMed]
- Altundağ, E.M.; Toprak, K.; Şanlıtürk, G.; Güran, M.; Özbilenler, C.; Kerküklü, N.R.; Yılmaz, A.M.; Yalçın, S.A. Synergistic combination of histone deacetylase inhibitor suberoylanilide hydroxamic acid and natural flavonoid curcumin exhibits anticancer and antibacterial activity. Anticancer Agents Med. Chem. 2021, 21, 1301–1308. [Google Scholar] [CrossRef]
- Lampe, V.; Milobedzka, J. Studien über curcumin. Berichte der deutschen chemischen Gesellschaft 1913, 46, 2235–2240. Available online: https://chemistry-europe.onlinelibrary.wiley.com/doi/abs/10.1002/cber.191304602149 (accessed on 3 February 2022). [CrossRef]
- Srinivasan, K.R. A chromatographic study of the curcuminoids in Curcuma longa, L. J. Pharm. Pharmacol. 1953, 5, 448–457. [Google Scholar] [CrossRef]
- Ma, Z.; Wang, N.; He, H.; Tang, X. Pharmaceutical strategies of improving oral systemic bioavailability of curcumin for clinical application. J. Control. Release 2019, 316, 359–380. Available online: https://www.ncbi.nlm.nih.gov/pubmed/31682912 (accessed on 3 February 2022). [CrossRef]
- Seidi Damyeh, M.; Mereddy, R.; Netzel, M.E.; Sultanbawa, Y. An insight into curcumin-based photosensitization as a promising and green food preservation technology. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1727–1759. [Google Scholar] [CrossRef]
- Prasad, S.; Gupta, S.C.; Tyagi, A.K.; Aggarwal, B.B. Curcumin, a component of golden spice: From bedside to bench and back. Biotechnol. Adv. 2014, 32, 1053–1064. [Google Scholar] [CrossRef]
- Dai, C.; Xiao, X.; Zhang, Y.; Xiang, B.; Hoyer, D.; Shen, J.; Velkov, T.; Tang, S. Curcumin attenuates colistin-induced peripheral neurotoxicity in mice. ACS Infect. Dis. 2020, 6, 715–724. [Google Scholar] [CrossRef] [PubMed]
- Schraufstatter, E.; Bernt, H. Antibacterial action of curcumin and related compounds. Nature 1949, 164, 456. [Google Scholar] [CrossRef] [PubMed]
- Morão, L.G.; Polaquini, C.R.; Kopacz, M.; Torrezan, G.S.; Ayusso, G.M.; Dilarri, G.; Cavalca, L.B.; Zielińska, A.; Scheffers, D.J.; Regasini, L.O.; et al. A simplified curcumin targets the membrane of bacillus subtilis. Microbiologyopen 2019, 8, e00683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaur, A.; Sharma, P.; Capalash, N. Curcumin alleviates persistence of acinetobacter baumannii against colistin. Sci. Rep. 2018, 8, 11029. [Google Scholar] [CrossRef] [PubMed]
- Batista de Andrade Neto, J.; Pessoa de Farias Cabral, V.; Brito Nogueira, L.F.; Rocha da Silva, C.; Gurgel do Amaral Valente Sá, L.; Ramos da Silva, A.; Barbosa da Silva, W.M.; Silva, J.; Marinho, E.S.; Cavalcanti, B.C.; et al. Anti-mrsa activity of curcumin in planktonic cells and biofilms and determination of possible action mechanisms. Microb. Pathog. 2021, 155, 104892. [Google Scholar] [CrossRef] [PubMed]
- Yadav, S.; Singh, A.K.; Agrahari, A.K.; Sharma, K.; Singh, A.S.; Gupta, M.K.; Tiwari, V.K.; Prakash, P. Making of water soluble curcumin to potentiate conventional antimicrobials by inducing apoptosis-like phenomena among drug-resistant bacteria. Sci. Rep. 2020, 10, 14204. Available online: https://www.ncbi.nlm.nih.gov/pubmed/32848171 (accessed on 3 February 2022). [CrossRef]
- Teow, S.Y.; Liew, K.; Ali, S.A.; Khoo, A.S.; Peh, C.S. Antibacterial action of curcumin against staphylococcus aureus: A brief review. J. Trop. Med. 2016, 2016, 2853045. [Google Scholar] [CrossRef] [Green Version]
- Bhawana; Basniwal, R.K.; Buttar, H.S.; Jain, V.K.; Jain, N. Curcumin nanoparticles: Preparation, characterization, and antimicrobial study. J. Agric. Food Chem. 2011, 59, 2056–2061. Available online: https://www.ncbi.nlm.nih.gov/pubmed/21322563 (accessed on 3 February 2022). [CrossRef]
- Krausz, A.E.; Adler, B.L.; Cabral, V.; Navati, M.; Doerner, J.; Charafeddine, R.A.; Chandra, D.; Liang, H.; Gunther, L.; Clendaniel, A.; et al. Curcumin-encapsulated nanoparticles as innovative antimicrobial and wound healing agent. Nanomedicine 2015, 11, 195–206. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Yan, M.; Ma, R.; Ma, S. Synthesis and antibacterial activity of novel 4-bromo-1h-indazole derivatives as ftsz inhibitors. Arch. Pharm. 2015, 348, 266–274. [Google Scholar] [CrossRef]
- Hegge, A.B.; Bruzell, E.; Kristensen, S.; Tønnesen, H.H. Photoinactivation of staphylococcus epidermidis biofilms and suspensions by the hydrophobic photosensitizer curcumin—Effect of selected nanocarrier: Studies on curcumin and curcuminoides xlvii. Eur. J. Pharm. Sci. 2012, 47, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Betts, J.W.; Sharili, A.S.; La Ragione, R.M.; Wareham, W.D. In vitro antibacterial activity of curcumin-polymyxin b combinations against multidrug-resistant bacteria associated with traumatic wound infections. J. Nat. Prod. 2016, 79, 1702–1706. [Google Scholar] [CrossRef] [PubMed]
- Gunes, H.; Gulen, D.; Mutlu, R.; Gumus, A.; Tas, T.; Topkaya, E.A. Antibacterial effects of curcumin: An in vitro minimum inhibitory concentration study. Toxicol. Ind. Health 2016, 32, 246–250. [Google Scholar] [CrossRef]
- Wang, X.; Ip, M.; Leung, A.W.; Yang, Z.; Wang, P.; Zhang, B.; Ip, S.; Xu, C. Sonodynamic action of curcumin on foodborne bacteria bacillus cereus and escherichia coli. Ultrasonics 2015, 62, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Dogra, N.; Choudhary, R.; Kohli, P.; Haddock, J.D.; Makwana, S.; Horev, B.; Vinokur, Y.; Droby, S.; Rodov, V. Polydiacetylene nanovesicles as carriers of natural phenylpropanoids for creating antimicrobial food-contact surfaces. J. Agric. Food Chem. 2015, 63, 2557–2565. [Google Scholar] [CrossRef] [PubMed]
- Bonifácio, D.; Martins, C.; David, B.; Lemos, C.; Neves, M.; Almeida, A.; Pinto, D.; Faustino, M.A.F.; Cunha, Â. Photodynamic inactivation of listeria innocua biofilms with food-grade photosensitizers: A curcumin-rich extract of curcuma longa vs commercial curcumin. J. Appl. Microbiol. 2018, 125, 282–294. [Google Scholar] [CrossRef]
- Sarkar, A.; De, R.; Mukhopadhyay, K.A. Curcumin as a potential therapeutic candidate for helicobacter pylori associated diseases. World J. Gastroenterol. 2016, 22, 2736–2748. [Google Scholar] [CrossRef] [PubMed]
- Darmani, H.; Smadi, E.A.M.; Bataineh, S.B.M. Blue light emitting diodes enhance the antivirulence effects of curcumin against helicobacter pylori. J. Med. Microbiol. 2020, 69, 617–624. [Google Scholar] [CrossRef]
- De, R.; Kundu, P.; Swarnakar, S.; Ramamurthy, T.; Chowdhury, A.; Nair, G.B.; Mukhopadhyay, K.A. Antimicrobial activity of curcumin against helicobacter pylori isolates from india and during infections in mice. Antimicrob. Agents Chemother. 2009, 53, 1592–1597. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Yin, L.; Ramage, G.; Li, B.; Tao, Y.; Zhi, Q.; Lin, H.; Zhou, Y. Assessing the impact of curcumin on dual-species biofilms formed by streptococcus mutans and candida albicans. Microbiologyopen 2019, 8, e937. [Google Scholar] [CrossRef] [Green Version]
- Marathe, S.A.; Balakrishnan, A.; Negi, V.D.; Sakorey, D.; Chandra, N.; Chakravortty, D. Curcumin reduces the motility of salmonella enterica serovar typhimurium by binding to the flagella, thereby leading to flagellar fragility and shedding. J. Bacteriol. 2016, 198, 1798–1811. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dahl, T.A.; McGowan, W.M.; Shand, M.A.; Srinivasan, S.V. Photokilling of bacteria by the natural dye curcumin. Arch. Microbiol. 1989, 151, 183–185. [Google Scholar] [CrossRef] [PubMed]
- Raorane, C.J.; Lee, J.H.; Kim, Y.G.; Rajasekharan, S.K.; García-Contreras, R.; Lee, J. Antibiofilm and antivirulence efficacies of flavonoids and curcumin against acinetobacter baumannii. Front. Microbiol. 2019, 10, 990. [Google Scholar] [CrossRef] [PubMed]
- Marini, E.; Di Giulio, M.; Magi, G.; Di Lodovico, S.; Cimarelli, M.E.; Brenciani, A.; Nostro, A.; Cellini, L.; Facinelli, B. Curcumin, an antibiotic resistance breaker against a multiresistant clinical isolate of mycobacterium abscessus. Phytother. Res. 2018, 32, 488–495. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Izui, S.; Sekine, S.; Maeda, K.; Kuboniwa, M.; Takada, A.; Amano, A.; Nagata, H. Antibacterial activity of curcumin against periodontopathic bacteria. J. Periodontol. 2016, 87, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Mody, D.; Athamneh, A.I.M.; Seleem, N.M. Curcumin: A natural derivative with antibacterial activity against clostridium difficile. J. Glob. Antimicrob. Resist. 2020, 21, 154–161. Available online: https://www.ncbi.nlm.nih.gov/pubmed/31622683 (accessed on 3 February 2022). [CrossRef] [PubMed]
- Varshney, G.K.; Saini, R.K.; Gupta, P.K.; Das, K. Effect of curcumin on the diffusion kinetics of a hemicyanine dye, lds-698, across a lipid bilayer probed by second harmonic spectroscopy. Langmuir 2013, 29, 2912–2918. [Google Scholar] [CrossRef]
- Barry, J.; Fritz, M.; Brender, J.R.; Smith, P.E.; Lee, D.K.; Ramamoorthy, A. Determining the effects of lipophilic drugs on membrane structure by solid-state nmr spectroscopy: The case of the antioxidant curcumin. J. Am. Chem. Soc. 2009, 131, 4490–4498. [Google Scholar] [CrossRef] [Green Version]
- Tyagi, P.; Singh, M.; Kumari, H.; Kumari, A.; Mukhopadhyay, K. Bactericidal activity of curcumin i is associated with damaging of bacterial membrane. PLoS ONE 2015, 10, e0121313. [Google Scholar] [CrossRef] [Green Version]
- Zheng, D.; Huang, C.; Huang, H.; Zhao, Y.; Khan, M.R.U.; Zhao, H.; Huang, L. Antibacterial mechanism of curcumin: A review. Chem. Biodivers. 2020, 17, e2000171. Available online: https://www.ncbi.nlm.nih.gov/pubmed/32533635 (accessed on 3 February 2022). [CrossRef]
- Deryabin, D.; Galadzhieva, A.; Kosyan, D.; Duskaev, G. Plant-derived inhibitors of ahl-mediated quorum sensing in bacteria: Modes of action. Int. J. Mol. Sci. 2019, 20, 5588. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brackman, G.; Coenye, T. Quorum sensing inhibitors as anti-biofilm agents. Curr. Pharm. Des. 2015, 21, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Ding, T.; Li, T.; Li, J. Impact of curcumin liposomes with anti-quorum sensing properties against foodborne pathogens aeromonas hydrophila and serratia grimesii. Microb. Pathog. 2018, 122, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Rocha, M.P.; Santos, M.S.; Rodrigues, P.L.F.; Araújo, T.S.D.; de Oliveira, J.M.; Rosa, L.P.; Bagnato, V.S.; da Silva, F.C. Photodynamic therapry with curcumin in the reduction of enterococcus faecalis biofilm in bone cavity: Rmicrobiological and spectral fluorescense analysis. Photodiagn. Photodyn. Ther. 2021, 33, 102084. [Google Scholar] [CrossRef] [PubMed]
- Santos, C.A.; Lima, E.M.F.; Franco, B.; Pinto, M.U. Exploring phenolic compounds as quorum sensing inhibitors in foodborne bacteria. Front. Microbiol. 2021, 12, 735931. [Google Scholar] [CrossRef] [PubMed]
- Gayani, B.; Dilhari, A.; Wijesinghe, G.K.; Kumarage, S.; Abayaweera, G.; Samarakoon, S.R.; Perera, I.C.; Kottegoda, N.; Weerasekera, M.M. Effect of natural curcuminoids-intercalated layered double hydroxide nanohybrid against staphylococcus aureus, pseudomonas aeruginosa, and enterococcus faecalis: A bactericidal, antibiofilm, and mechanistic study. Microbiologyopen 2019, 8, e00723. [Google Scholar] [CrossRef]
- Niu, X.; Gao, Y.; Yu, Y.; Yang, Y.; Wang, G.; Sun, L.; Wang, H. Molecular modelling reveals the inhibition mechanism and structure-activity relationship of curcumin and its analogues to staphylococcal aureus sortase A. J. Biomol. Struct. Dyn. 2019, 37, 1220–1230. [Google Scholar] [CrossRef]
- Packiavathy, I.A.; Priya, S.; Pandian, S.K.; Ravi, V.A. Inhibition of biofilm development of uropathogens by curcumin–An anti-quorum sensing agent from curcuma longa. Food Chem. 2014, 148, 453–460. [Google Scholar] [CrossRef]
- Hu, P.; Huang, P.; Chen, W.M. Curcumin reduces streptococcus mutans biofilm formation by inhibiting sortase a activity. Arch. Oral Biol. 2013, 58, 1343–1348. [Google Scholar] [CrossRef]
- Hu, P.; Huang, P.; Chen, M.W. Curcumin inhibits the sortase a activity of the streptococcus mutans ua159. Appl. Biochem. Biotechnol. 2013, 171, 396–402. [Google Scholar] [CrossRef]
- Li, B.; Li, X.; Lin, H.; Zhou, Y. Curcumin as a promising antibacterial agent: Effects on metabolism and biofilm formation in s. Mutans. Biomed. Res. Int. 2018, 2018, 4508709. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Magesh, H.; Kumar, A.; Alam, A.; Sekar, U.; Priyam; Sumantran, V.N.; Vaidyanathan, R. Identification of natural compounds which inhibit biofilm formation in clinical isolates of klebsiella pneumoniae. Indian J. Exp. Biol. 2013, 51, 764–772. [Google Scholar] [PubMed]
- Zhu, H.; He, C.C.; Chu, H.Q. Inhibition of quorum sensing in chromobacterium violaceum by pigments extracted from auricularia auricular. Lett. Appl. Microbiol. 2011, 52, 269–274. Available online: https://www.ncbi.nlm.nih.gov/pubmed/21204879 (accessed on 3 February 2022). [CrossRef] [PubMed]
- Rai, D.; Singh, J.K.; Roy, N.; Panda, D. Curcumin inhibits ftsz assembly: An attractive mechanism for its antibacterial activity. Biochem. J. 2008, 410, 147–155. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Du, S.; Lutkenhaus, J. At the heart of bacterial cytokinesis: The z ring. Trends Microbiol. 2019, 27, 781–791. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.; Modi, N.H.; Panda, D.; Roy, N. Probing the binding site of curcumin in escherichia coli and bacillus subtilis ftsz – A structural insight to unveil antibacterial activity of curcumin. Eur. J. Med. Chem. 2010, 45, 4209–4214. [Google Scholar] [CrossRef]
- Allocati, N.; Masulli, M.; Di Ilio, C.; De Laurenzi, V. Die for the community: An overview of programmed cell death in bacteria. Cell Death Dis. 2015, 6, e1609. [Google Scholar] [CrossRef] [Green Version]
- Dwyer, D.J.; Camacho, D.M.; Kohanski, M.A.; Callura, J.M.; Collins, J.J. Antibiotic-induced bacterial cell death exhibits physiological and biochemical hallmarks of apoptosis. Mol. Cell 2012, 46, 561–572. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ezraty, B.; Gennaris, A.; Barras, F.; Collet, F.J. Oxidative stress, protein damage and repair in bacteria. Nat. Rev. Microbiol. 2017, 15, 385–396. [Google Scholar] [CrossRef]
- Butala, M.; Zgur-Bertok, D.; Busby, J.S. The bacterial lexa transcriptional repressor. Cell Mol. Life Sci. 2009, 66, 82–93. [Google Scholar] [CrossRef]
- Ghasemi, M.; Khorsandi, K.; Kianmehr, Z. Photodynamic inactivation with curcumin and silver nanoparticles hinders pseudomonas aeruginosa planktonic and biofilm formation: Evaluation of glutathione peroxidase activity and ros production. World J. Microbiol. Biotechnol. 2021, 37, 149. [Google Scholar] [CrossRef] [PubMed]
- Ghate, V.S.; Zhou, W.; Yuk, G.H. Perspectives and trends in the application of photodynamic inactivation for microbiological food safety. Compr. Rev. Food Sci. Food Saf. 2019, 18, 402–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nima, G.; Soto-Montero, J.; Alves, L.A.; Mattos-Graner, R.O.; Giannini, M. Photodynamic inactivation of streptococcus mutans by curcumin in combination with edta. Dent. Mater. 2021, 37, e1–e14. [Google Scholar] [CrossRef]
- Jiang, Y.; Leung, A.W.; Hua, H.; Rao, X.; Xu, C. Photodynamic action of led-activated curcumin against Staphylococcus aureus involving intracellular ros increase and membrane damage. Int. J. Photoenergy 2014, 2014, 637601. [Google Scholar] [CrossRef] [Green Version]
- Huang, J.; Chen, B.; Li, H.; Zeng, Q.-H.; Wang, J.J.; Liu, H.; Pan, Y.; Zhao, Y. Enhanced antibacterial and antibiofilm functions of the curcumin-mediated photodynamic inactivation against listeria monocytogenes. Food Control 2020, 108, 106886. Available online: https://www.sciencedirect.com/science/article/pii/S095671351930475X (accessed on 3 February 2022). [CrossRef]
- Chen, B.; Huang, J.; Liu, Y.; Liu, H.; Zhao, Y.; Wang, J.J. Effects of the curcumin-mediated photodynamic inactivation on the quality of cooked oysters with vibrio parahaemolyticus during storage at different temperature. Int. J. Food Microbiol. 2021, 345, 109152. Available online: https://www.sciencedirect.com/science/article/pii/S0168160521001112 (accessed on 3 February 2022). [CrossRef]
- Zangirolami, A.C.; Dias, L.D.; Blanco, K.C.; Vinagreiro, C.S.; Inada, N.M.; Arnaut, L.G.; Pereira, M.M.; Bagnato, S.V. Avoiding ventilator-associated pneumonia: Curcumin-functionalized endotracheal tube and photodynamic action. Proc. Natl. Acad. Sci. USA 2020, 117, 22967–22973. [Google Scholar] [CrossRef]
- Belenky, P.; Ye, J.D.; Porter, C.B.; Cohen, N.R.; Lobritz, M.A.; Ferrante, T.; Jain, S.; Korry, B.J.; Schwarz, E.G.; Walker, G.C.; et al. Bactericidal antibiotics induce toxic metabolic perturbations that lead to cellular damage. Cell Rep. 2015, 13, 968–980. [Google Scholar] [CrossRef] [Green Version]
- Duan, X.; Huang, X.; Wang, X.; Yan, S.; Guo, S.; Abdalla, A.E.; Huang, C.; Xie, J. L-serine potentiates fluoroquinolone activity against escherichia coli by enhancing endogenous reactive oxygen species production. J. Antimicrob. Chemother. 2016, 71, 2192–2199. [Google Scholar] [CrossRef] [Green Version]
- Adeyemi, O.S.; Obeme-Imom, J.I.; Akpor, B.O.; Rotimi, D.; Batiha, G.E.; Owolabi, A. Altered redox status, DNA damage and modulation of l-tryptophan metabolism contribute to antimicrobial action of curcumin. Heliyon 2020, 6, e03495. [Google Scholar] [CrossRef]
- Marathe, S.A.; Sen, M.; Dasgupta, I.; Chakravortty, D. Differential modulation of intracellular survival of cytosolic and vacuolar pathogens by curcumin. Antimicrob. Agents Chemother. 2012, 56, 5555–5567. Available online: https://www.ncbi.nlm.nih.gov/pubmed/22890770 (accessed on 3 February 2022). [CrossRef] [Green Version]
- Case, E.D.; Smith, J.A.; Ficht, T.A.; Samuel, J.E.; de Figueiredo, P. Space: A final frontier for vacuolar pathogens. Traffic 2016, 17, 461–474. Available online: https://www.ncbi.nlm.nih.gov/pubmed/26842840 (accessed on 3 February 2022). [CrossRef] [PubMed] [Green Version]
- Ogawa, M.; Sasakawa, C. Intracellular survival of shigella. Cell Microbiol. 2006, 8, 177–184. Available online: https://www.ncbi.nlm.nih.gov/pubmed/16441429 (accessed on 3 February 2022). [CrossRef] [PubMed]
- Bai, X.; Oberley-Deegan, R.E.; Bai, A.; Ovrutsky, A.R.; Kinney, W.H.; Weaver, M.; Zhang, G.; Honda, J.R.; Chan, D.E. Curcumin enhances human macrophage control of mycobacterium tuberculosis infection. Respirology 2016, 21, 951–957. Available online: https://www.ncbi.nlm.nih.gov/pubmed/27012592 (accessed on 3 February 2022). [CrossRef]
- Bush, K. Synergistic antibiotic combinations. In Antibacterials; Fisher, J.F., Mobashery, S., Miller, J.M., Eds.; Springer International Publishing: Berlin/Heidelberg, Germany, 2018; pp. 69–88. [Google Scholar]
- Gülen, D.; Şafak, B.; Erdal, B.; Günaydın, B. Curcumin-meropenem synergy in carbapenem resistant klebsiella pneumoniae curcumin-meropenem synergy. Iran. J. Microbiol. 2021, 13, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Sasidharan, N.K.; Sreekala, S.R.; Jacob, J.; Nambisan, B. In vitro synergistic effect of curcumin in combination with third generation cephalosporins against bacteria associated with infectious diarrhea. Biomed. Res. Int. 2014, 2014, 561456. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhatia, E.; Sharma, S.; Jadhav, K.; Banerjee, R. Combinatorial liposomes of berberine and curcumin inhibit biofilm formation and intracellular methicillin resistant staphylococcus aureus infections and associated inflammation. J. Mater. Chem. B 2021, 9, 864–875. [Google Scholar] [CrossRef]
- Gholami, M.; Zeighami, H.; Bikas, R.; Heidari, A.; Rafiee, F.; Haghi, F. Inhibitory activity of metal-curcumin complexes on quorum sensing related virulence factors of pseudomonas aeruginosa pao1. AMB Express 2020, 10, 111. [Google Scholar] [CrossRef]
- Jawetz, E. Polymyxins, colistin, bacitracin, ristocetin and vancomycin. Pediatr. Clin. N. Am. 1968, 15, 85–94. [Google Scholar] [CrossRef]
- Dixon, R.A.; Chopra, I. Polymyxin b and polymyxin b nonapeptide alter cytoplasmic membrane permeability in escherichia coli. J. Antimicrob. Chemother. 1986, 18, 557–563. Available online: https://www.ncbi.nlm.nih.gov/pubmed/3027012 (accessed on 3 February 2022). [CrossRef]
- Sampson, T.R.; Liu, X.; Schroeder, M.R.; Kraft, C.S.; Burd, E.M.; Weiss, S.D. Rapid killing of acinetobacter baumannii by polymyxins is mediated by a hydroxyl radical death pathway. Antimicrob. Agents Chemother. 2012, 56, 5642–5649. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dai, C.; Wang, Y.; Sharma, G.; Shen, J.; Velkov, T.; Xiao, X. Polymyxins-curcumin combination antimicrobial therapy: Safety implications and efficacy for infection treatment. Antioxidants 2020, 9, 506. [Google Scholar] [CrossRef] [PubMed]
- Ahmida, M.H. Protective role of curcumin in nephrotoxic oxidative damage induced by vancomycin in rats. Exp. Toxicol. Pathol. 2012, 64, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Blumenthal, K.G.; Peter, J.G.; Trubiano, J.A.; Phillips, J.E. Antibiotic allergy. Lancet 2019, 393, 183–198. [Google Scholar] [CrossRef]
- Yadav, S.; Singh, A.K.; Agrahari, A.K.; Pandey, A.K.; Gupta, M.K.; Chakravortty, D.; Tiwari, V.K.; Prakash, P. Galactose-clicked curcumin-mediated reversal of meropenem resistance among klebsiella pneumoniae by targeting its carbapenemases and the acrab-tolc efflux system. Antibiotics 2021, 10, 388. [Google Scholar] [CrossRef] [PubMed]
- Mun, S.H.; Joung, D.K.; Kim, Y.S.; Kang, O.H.; Kim, S.B.; Seo, Y.S.; Kim, Y.C.; Lee, D.S.; Shin, D.W.; Kweon, K.T.; et al. Synergistic antibacterial effect of curcumin against methicillin-resistant staphylococcus aureus. Phytomedicine 2013, 20, 714–718. [Google Scholar] [CrossRef]
- Wang, S.; Kim, M.C.; Kang, O.H.; Kwon, Y.D. The mechanism of bisdemethoxycurcumin enhances conventional antibiotics against methicillin-resistant staphylococcus aureus. Int. J. Mol. Sci. 2020, 21, 7945. [Google Scholar] [CrossRef]
- Stokes, J.M.; Lopatkin, A.J.; Lobritz, M.A.; Collins, J.J. Bacterial metabolism and antibiotic efficacy. Cell Metab. 2019, 30, 251–259. [Google Scholar] [CrossRef]
- Bahari, S.; Zeighami, H.; Mirshahabi, H.; Roudashti, S.; Haghi, F. Inhibition of pseudomonas aeruginosa quorum sensing by subinhibitory concentrations of curcumin with gentamicin and azithromycin. J. Glob. Antimicrob. Resist. 2017, 10, 21–28. [Google Scholar] [CrossRef]
- Kohanski, M.A.; Dwyer, D.J.; Collins, J.J. How antibiotics kill bacteria: From targets to networks. Nat. Rev. Microbiol. 2010, 8, 423–435. [Google Scholar] [CrossRef] [Green Version]
- Abd-Elhakim, Y.M.; Abdel-Motal, S.M.; Malhat, S.M.; Mostafa, H.I.; Moselhy, A.A.A.; Beheiry, R.R.; Said, N.E. Curcumin mitigates neurotoxic and neurobehavioral changes of gentamicin and sodium salicylate in rats by adjusting oxidative stress and apoptosis. Life Sci. 2021, 265, 118824. [Google Scholar] [CrossRef]
- Parnham, M.J.; Erakovic Haber, V.; Giamarellos-Bourboulis, E.J.; Perletti, G.; Verleden, G.M.; Vos, R. Azithromycin: Mechanisms of action and their relevance for clinical applications. Pharmacol. Ther. 2014, 143, 225–245. [Google Scholar] [CrossRef] [PubMed]
- Sun, N.; Chan, F.Y.; Lu, Y.J.; Neves, M.A.; Lui, H.K.; Wang, Y.; Chow, K.Y.; Chan, K.F.; Yan, S.C.; Leung, Y.C.; et al. Rational design of berberine-based ftsz inhibitors with broad-spectrum antibacterial activity. PLoS ONE 2014, 9, e97514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Domadia, P.N.; Bhunia, A.; Sivaraman, J.; Swarup, S.; Dasgupta, D. Berberine targets assembly of escherichia coli cell division protein ftsz. Biochemistry 2008, 47, 3225–3234. [Google Scholar] [CrossRef] [PubMed]
- Gordon, N.C.; Wareham, W.D. Antimicrobial activity of the green tea polyphenol (-)-epigallocatechin-3-gallate (egcg) against clinical isolates of stenotrophomonas maltophilia. Int. J. Antimicrob. Agents 2010, 36, 129–131. [Google Scholar] [CrossRef]
- Betts, J.W.; Wareham, W.D. In vitro activity of curcumin in combination with epigallocatechin gallate (egcg) versus multidrug-resistant acinetobacter baumannii. BMC Microbiol. 2014, 14, 172. [Google Scholar] [CrossRef] [Green Version]
- Hatano, T.; Tsugawa, M.; Kusuda, M.; Taniguchi, S.; Yoshida, T.; Shiota, S.; Tsuchiya, T. Enhancement of antibacterial effects of epigallocatechin gallate, using ascorbic acid. Phytochemistry 2008, 69, 3111–3116. [Google Scholar] [CrossRef]
- Lade, H.; Paul, D.; Kweon, H.J. Combined effects of curcumin and (-)-epigallocatechin gallate on inhibition of n-acylhomoserine lactone-mediated biofilm formation in wastewater bacteria from membrane bioreactor. J. Microbiol. Biotechnol. 2015, 25, 1908–1919. [Google Scholar] [CrossRef]
- Lemire, J.A.; Harrison, J.J.; Turner, J.R. Antimicrobial activity of metals: Mechanisms, molecular targets and applications. Nat. Rev. Microbiol. 2013, 11, 371–384. [Google Scholar] [CrossRef]
- Lyu, Y.; Yu, M.; Liu, Q.; Zhang, Q.; Liu, Z.; Tian, Y.; Li, D.; Changdao, M. Synthesis of silver nanoparticles using oxidized amylose and combination with curcumin for enhanced antibacterial activity. Carbohydr. Polym. 2020, 230, 115573. [Google Scholar] [CrossRef]
- Song, Z.; Wu, Y.; Wang, H.; Han, H. Synergistic antibacterial effects of curcumin modified silver nanoparticles through ros-mediated pathways. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 99, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Targhi, A.A.; Moammeri, A.; Jamshidifar, E.; Abbaspour, K.; Sadeghi, S.; Lamakani, L.; Akbarzadeh, I. Synergistic effect of curcumin-cu and curcumin-ag nanoparticle loaded niosome: Enhanced antibacterial and anti-biofilm activities. Bioorg. Chem. 2021, 115, 105116. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Saha, T.; Behera, S.; Gupta, S.; Das, S.; Mukhopadhyay, K. Enhanced efficacy of a Cu2+ complex of curcumin against gram-positive and gram-negative bacteria: Attributes of complex formation. J. Inorg. Biochem. 2021, 222, 111494. [Google Scholar] [CrossRef]
- Salehi, B.; Stojanović-Radić, Z.; Matejić, J.; Sharifi-Rad, M.; Anil Kumar, N.V.; Martins, N.; Sharifi-Rad, J. The therapeutic potential of curcumin: A review of clinical trials. Eur. J. Med. Chem. 2019, 163, 527–545. [Google Scholar] [CrossRef] [PubMed]
- Nelson, K.M.; Dahlin, J.L.; Bisson, J.; Graham, J.; Pauli, G.F.; Walters, A.M. The essential medicinal chemistry of curcumin. J. Med. Chem. 2017, 60, 1620–1637. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, J.; Rayess, Y.E.; Rizk, A.A.; Sadaka, C.; Zgheib, R.; Zam, W.; Sestito, S.; Rapposelli, S.; Neffe-Skocińska, K.; Zielińska, D.; et al. Turmeric and its major compound curcumin on health: Bioactive effects and safety profiles for food, pharmaceutical, biotechnological and medicinal applications. Front. Pharmacol. 2020, 11, 01021. [Google Scholar] [CrossRef]
- Evaluation of Certain Food Additives and Contaminants; WHO Technical Report Series; WHO: Geneva, Switzerland, 2013; pp. 1–75.
- Momeni, H.R.; Eskandari, N. Curcumin protects the testis against cadmium-induced histopathological damages and oxidative stress in mice. Hum. Exp. Toxicol. 2020, 39, 653–661. [Google Scholar] [CrossRef]
- Kim, K.S.; Lim, H.J.; Lim, J.S.; Son, J.Y.; Lee, J.; Lee, B.M.; Chang, S.C.; Kim, S.H. Curcumin ameliorates cadmium-induced nephrotoxicity in sprague-dawley rats. Food Chem. Toxicol. 2018, 114, 34–40. [Google Scholar] [CrossRef]
- Li, S.; Liu, R.; Wei, G.; Guo, G.; Yu, H.; Zhang, Y.; Ishfaq, M.; Fazilani, S.A.; Zhang, X. Curcumin protects against aflatoxin b1-induced liver injury in broilers via the modulation of long non-coding rna expression. Ecotoxicol. Environ. Saf. 2021, 208, 111725. [Google Scholar] [CrossRef]
- Cheng, P.; Ishfaq, M.; Yu, H.; Yang, Y.; Li, S.; Li, X.; Fazlani, S.A.; Guo, W.; Zhang, X. Curcumin ameliorates duodenal toxicity of afb1 in chicken through inducing p-glycoprotein and downregulating cytochrome p450 enzymes. Poult. Sci. 2020, 99, 7035–7045. [Google Scholar] [CrossRef] [PubMed]
- Kundu, P.; De, R.; Pal, I.; Mukhopadhyay, A.K.; Saha, D.R.; Swarnakar, S. Curcumin alleviates matrix metalloproteinase-3 and -9 activities during eradication of helicobacter pylori infection in cultured cells and mice. PLoS ONE 2011, 6, e16306. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, A.L.; Hsu, C.H.; Lin, J.K.; Hsu, M.M.; Ho, Y.F.; Shen, T.S.; Ko, J.Y.; Lin, J.T.; Lin, B.R.; Ming-Shiang, W.; et al. Phase i clinical trial of curcumin, a chemopreventive agent, in patients with high-risk or pre-malignant lesions. Anticancer Res. 2001, 21, 2895–2900. [Google Scholar] [PubMed]
- Sharma, R.A.; Euden, S.A.; Platton, S.L.; Cooke, D.N.; Shafayat, A.; Hewitt, H.R.; Marczylo, T.H.; Morgan, B.; Hemingway, D.; Plummer, S.M.; et al. Phase i clinical trial of oral curcumin: Biomarkers of systemic activity and compliance. Clin. Cancer Res. 2004, 10, 6847–6854. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Panahi, Y.; Karbasi, A.; Valizadegan, G.; Ostadzadeh, N.; Soflaei, S.S.; Jamialahmadi, T.; Majeed, M.; Sahebkar, A. Effect of curcumin on severity of functional dyspepsia: A triple blinded clinical trial. Adv. Exp. Med. Biol. 2021, 1308, 119–126. [Google Scholar] [CrossRef]
- Taghavi Kevij, H.; Salami, M.; Mohammadian, M.; Khodadadi, M. Fabrication and investigation of physicochemical, food simulant release, and antioxidant properties of whey protein isolate-based films activated by loading with curcumin through the ph-driven method. Food Hydrocoll. 2020, 108, 106026. Available online: https://www.sciencedirect.com/science/article/pii/S0268005X20306044 (accessed on 3 February 2022). [CrossRef]
- Soleimani, V.; Sahebkar, A.; Hosseinzadeh, H. Turmeric (curcuma longa) and its major constituent (curcumin) as nontoxic and safe substances: Review. Phytother. Res. 2018, 32, 985–995. [Google Scholar] [CrossRef]
- Kotha, R.R.; Luthria, L.D. Curcumin: Biological, pharmaceutical, nutraceutical, and analytical aspects. Molecules 2019, 24, 2930. [Google Scholar] [CrossRef] [Green Version]
- Adahoun, M.A.; Al-Akhras, M.H.; Jaafar, M.S.; Bououdina, M. Enhanced anti-cancer and antimicrobial activities of curcumin nanoparticles. Artif. Cells Nanomed. Biotechnol. 2017, 45, 98–107. Available online: https://www.ncbi.nlm.nih.gov/pubmed/26747522 (accessed on 3 February 2022). [CrossRef]
- Ezati, P.; Rhim, W.J. Ph-responsive pectin-based multifunctional films incorporated with curcumin and sulfur nanoparticles. Carbohydr. Polym. 2020, 230, 115638. [Google Scholar] [CrossRef]
- Sharifi, S.; Fathi, N.; Memar, M.Y.; Hosseiniyan Khatibi, S.M.; Khalilov, R.; Negahdari, R.; Zununi Vahed, S.; Maleki Dizaj, S. Anti-microbial activity of curcumin nanoformulations: New trends and future perspectives. Phytother. Res. 2020, 34, 1926–1946. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Lu, Y.; Lee, R.J.; Xiang, G. Nano encapsulated curcumin: And its potential for biomedical applications. Int. J. Nanomed. 2020, 15, 3099–3120. [Google Scholar] [CrossRef] [PubMed]
- Yadav, V.R.; Suresh, S.; Devi, K.; Yadav, S. Effect of cyclodextrin complexation of curcumin on its solubility and antiangiogenic and anti-inflammatory activity in rat colitis model. AAPS PharmSciTech 2009, 10, 752–762. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Lu, Z.; Wu, H.; Lv, F. Study on the antibiotic activity of microcapsule curcumin against foodborne pathogens. Int. J. Food Microbiol. 2009, 136, 71–74. Available online: https://www.ncbi.nlm.nih.gov/pubmed/19775769 (accessed on 3 February 2022). [CrossRef]
- Varaprasad, K.; Vimala, K.; Ravindra, S.; Narayana Reddy, N.; Venkata Subba Reddy, G.; Mohana Raju, K. Fabrication of silver nanocomposite films impregnated with curcumin for superior antibacterial applications. J. Mater. Sci. Mater. Med. 2011, 22, 1863–1872. Available online: https://www.ncbi.nlm.nih.gov/pubmed/21681658 (accessed on 3 February 2022). [CrossRef]
- Singh, A.K.; Prakash, P.; Singh, R.; Nandy, N.; Firdaus, Z.; Bansal, M.; Singh, R.K.; Srivastava, A.; Roy, J.K.; Mishra, B.; et al. Curcumin quantum dots mediated degradation of bacterial biofilms. Front. Microbiol. 2017, 8, 1517. Available online: https://www.ncbi.nlm.nih.gov/pubmed/28848526 (accessed on 3 February 2022). [CrossRef] [Green Version]
- Gao, M.Q.; Long, X.; Du, J.; Teng, M.T.; Zhang, W.C.; Wang, Y.T.; Wang, X.Q.; Wang, Z.Y.; Zhang, P.; Li, J. Enhanced curcumin solubility and antibacterial activity by encapsulation in plga oily core nanocapsules. Food Funct. 2020, 11, 448–455. [Google Scholar] [CrossRef]
- Shariati, A.; Asadian, E.; Fallah, F.; Azimi, T.; Hashemi, A.; Sharahi, J.Y.; Moghadam, T.M. Evaluation of nano-curcumin effects on expression levels of virulence genes and biofilm production of multidrug-resistant pseudomonas aeruginosa isolated from burn wound infection in tehran, iran. Infect. Drug Resist. 2019, 12, 2223–2235. [Google Scholar] [CrossRef] [Green Version]
- Leng, Q.Q.; Li, Y.; Pang, X.L.; Wang, B.Q.; Wu, Z.X.; Lu, Y.; Xiong, K.; Zhao, L.; Zhou, P.; Fu, Z.S. Curcumin nanoparticles incorporated in pva/collagen composite films promote wound healing. Drug Deliv. 2020, 27, 1676–1685. [Google Scholar] [CrossRef]
- Karthikeyan, C.; Varaprasad, K.; Akbari-Fakhrabadi, A.; Hameed, A.S.H.; Sadiku, R. Biomolecule chitosan, curcumin and zno-based antibacterial nanomaterial, via a one-pot process. Carbohydr. Polym. 2020, 249, 116825. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).