Redox Properties, Bioactivity and Health Effects of Indicaxanthin, a Bioavailable Phytochemical from Opuntia ficus indica, L.: A Critical Review of Accumulated Evidence and Perspectives
Abstract
1. Introduction
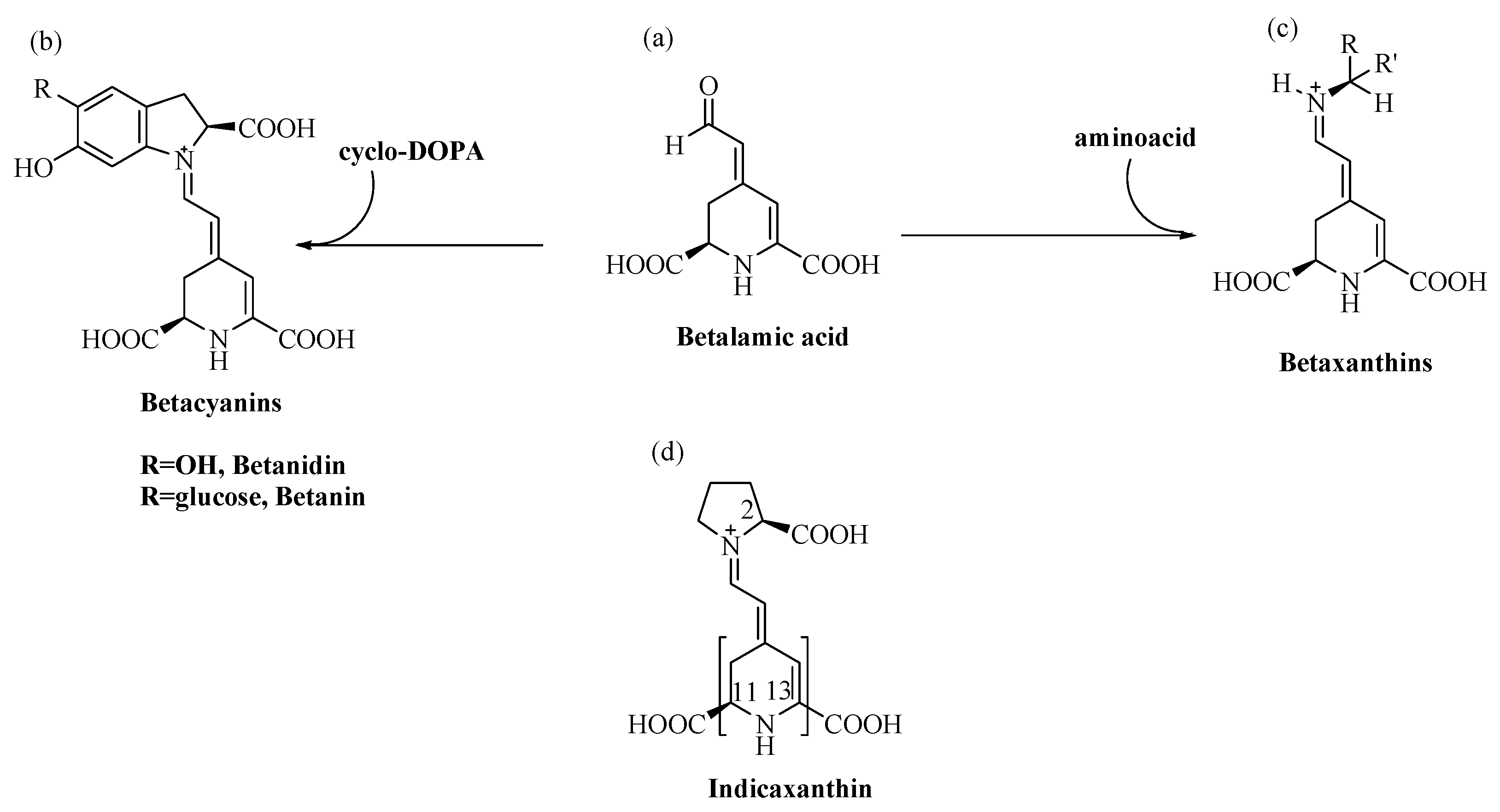
2. The Betalain Indicaxanthin
3. Redox Chemistry and Physicochemical Characteristics of Indicaxanthin
3.1. Redox Chemistry of Indicaxanthin in Solution
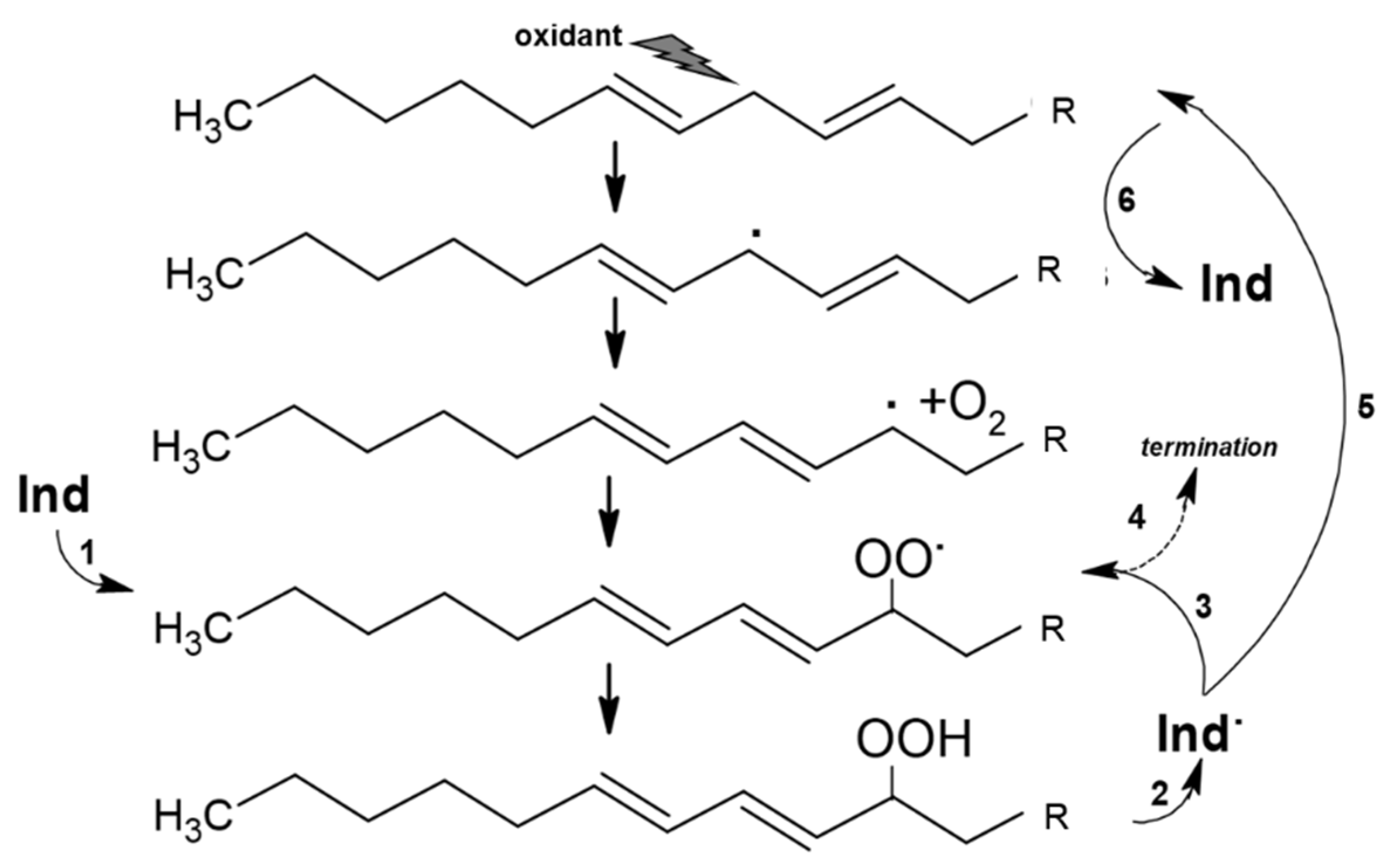
3.2. Physicochemical Properties and Interactions of Indicaxanthin with Lipid Systems
4. Binding of Indicaxanthin to Proteins: In Silico Molecular Modeling and Test Tube Experiments
5. Bioavailability of Indicaxanthin
6. Modes of Action of Indicaxanthin in Cells and Tissues
6.1. Indicaxanthin, Redox Balance and Oxidative Stress in Healthy Cells
6.1.1. Indicaxanthin between Antioxidant vs. Pro-Oxidant Activity
6.1.2. Indicaxanthin and Cell Redox Homeostasis: ROS- and RNS-Producing Enzymes and Redox Signaling
6.2. Indicaxanthin Activity in Organ Bath
6.3. Indicaxanthin and Oxidative Stress in Tumor Cells
7. Indicaxanthin In Vivo: Activity in a Model Organism and Pharmacological Effects in Animal Models
7.1. Caenorhabditis Elegans: A New Paradigm for Indicaxanthin?
7.2. Pharmacological Activity of Indicaxanthin in Murine Models
7.2.1. Indicaxanthin in a Pleurisy Model
7.2.2. Indicaxantin in a Melanoma Model
7.2.3. Indicaxanthin in a Metabolic Syndrome Model
7.2.4. Neuro-Physiological Activity of Indicaxanthin
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Das, K.; Roychoudhury, A. Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Front. Environ. Sci. 2014, 2, 53. [Google Scholar] [CrossRef]
- Trchounian, A.; Petrosyan, M.; Sahakyan, N. Plant cell redox homeostasis and reactive oxygen species. In Redox State as a Central Regulator of Plant-Cell Stress Responses; Springer International Publishing: Cham, Switzerland, 2016; pp. 25–50. [Google Scholar]
- Xie, X.; He, Z.; Chen, N.; Tang, Z.; Wang, Q.; Cai, Y. The roles of environmental factors in regulation of oxidative stress in plant. BioMed Res. Int. 2019, 2019, 9732325. [Google Scholar] [CrossRef] [PubMed]
- Forman, H.J. Redox signaling: An evolution from free radicals to aging. Free Radic. Biol. Med. 2016, 97, 398–407. [Google Scholar] [CrossRef]
- Anjum, N.A.; Khan, N.A.; Sofo, A.; Baier, M.; Kizek, R. Redox homeostasis managers in plants under environmental stresses. Front. Environ. Sci. 2016, 4, 35. [Google Scholar] [CrossRef]
- Ursini, F.; Maiorino, M.; Forman, H.J. Redox homeostasis: The Golden Mean of healthy living. Redox. Biol. 2016, 8, 205–215. [Google Scholar] [CrossRef]
- Forman, H.J.; Davies, K.J.; Ursini, F. How do nutritional antioxidants really work: Nucleophilic tone and para-hormesis versus free radical scavenging in vivo. Free Radic. Biol. Med. 2014, 66, 24–35. [Google Scholar] [CrossRef]
- Howitz, K.T.; Sinclair, D.A. Xenohormesis: Sensing the chemical cues of other species. Cell 2008, 133, 387–391. [Google Scholar] [CrossRef]
- Surh, Y.J. Xenohormesis mechanisms underlying chemopreventive effects of some dietary phytochemicals. Ann. N. Y. Acad. Sci. 2011, 1229, 1–6. [Google Scholar] [CrossRef]
- Davies, K.J.A. Adaptive homeostasis. Mol. Aspects Med. 2016, 49, 1–7. [Google Scholar] [CrossRef]
- Calabrese, V.; Cornelius, C.; Dinkova-Kostova, A.T.; Iavicoli, I.; Di Paola, R.; Koverech, A.; Cuzzocrea, S.; Rizzarelli, E.; Calabrese, E.J. Cellular stress responses, hormetic phytochemicals and vitagenes in aging and longevity. Biochim. Biophys. Acta 2012, 1822, 753–783. [Google Scholar]
- Carlos-Reyes, Á.; López-González, J.S.; Meneses-Flores, M.; Gallardo-Rincón, D.; Ruíz-García, E.; Marchat, L.A.; Astudillo-de la Vega, H.; Hernández de la Cruz, O.N.; López-Camarillo, C. Dietary Compounds as Epigenetic Modulating Agents in Cancer. Front. Genet. 2019, 10, 79. [Google Scholar] [CrossRef]
- Slimen, I.B.; Najar, T.; Abderrabba, M. Chemical and Antioxidant Properties of Betalains. J. Agric. Food Chem. 2017, 65, 675–689. [Google Scholar] [CrossRef]
- Inglese, P. Orchard planting and management. In Agro-Ecology, Cultivation and Uses of Cactus Pear; Barbera, G., Inglese, P., Pimienta-Barrios, E., Eds.; FAO Plant Production and Protection Paper; FAO: Rome, Italy, 1995; Volume 132, pp. 78–91. [Google Scholar]
- Livrea, M.A.; Tesoriere, L. Indicaxanthin Dietetics: Past, Present, and Future. In Pigments in Fruits and Vegetables; Chen, C., Ed.; Springer: New York, NY, USA, 2015; pp. 141–163. [Google Scholar]
- Gutzeit, H.O.; Ludwig-Muller, J. Plant Natural Product: Synthesis, Biological Functions and Practical Applications, 1st ed; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2014. [Google Scholar]
- Sachdev, S.; Ansari, S.A.; Ansari, M.I.; Fujita, M.; Hasanuzzaman, M. Abiotic Stress and Reactive Oxygen Species: Generation, Signaling, and Defense Mechanisms. Antioxidants 2021, 10, 277. [Google Scholar] [CrossRef]
- Khan, M.I.; Sri Harsha, P.S.C.; Giridhar, P.; Ravishankar, G.A. Pigment identification, nutritional composition, bioactivity, and in vitro cancer cell cytotoxicity of Rivina humilis L. berries, potential source of betalains. LWT—Food Sci. Technol. 2012, 47, 315–323. [Google Scholar] [CrossRef]
- Khan, M.I. Plant betalains: Safety, antioxidant activity, clinical efficacy and bioavailability. Compr. Rev. Food Sci. Food Saf. 2016, 15, 316–330. [Google Scholar] [CrossRef]
- Khan, M.I.; Kumar, A.; Giridhar, P. Betalains and expression of antioxidant enzymes during development and abiotic stress in Rivina humilis L. berries. Turk. J. Bot. 2016, 40, 28–36. [Google Scholar] [CrossRef]
- Hoang, X.L.T.; Nhi, D.N.H.; Thu, N.B.A.; Thao, N.P.; Tran, L.-S.P. Transcription factors and their roles in signal transduction in plants under abiotic stress. Curr. Genom. 2017, 18, 479–483. [Google Scholar] [CrossRef]
- Gliszczyńska-Świgło, A.; Szymusiak, H.; Malinowska, P. Betanin, the main pigment of red beet: Molecular origin of its exceptionally high free radical-scavenging activity. Food Addit. Contam. 2006, 11, 1079–1087. [Google Scholar] [CrossRef]
- Impellizzeri, G.; Piattelli, M. Biosynthesis of indicaxanthin in Opuntia ficus-indica fruits. Phytochemistry 1972, 11, 2499–2502. [Google Scholar] [CrossRef]
- Allegra, M.; Tutone, M.; Tesoriere, L.; Almerico, A.M.; Culletta, G.; Livrea, M.A.; Attanzio, A. Indicaxanthin, a multi-target natural compound from Opuntia ficus-indica fruit: From its poly-pharmacological effects to biochemical mechanisms and molecular modelling studies. Eur. J. Med. Chem. 2019, 179, 753–764. [Google Scholar] [CrossRef]
- Tesoriere, L.; Allegra, M.; Butera, D.; Gentile, C.; Livrea, M.A. Kinetics of the lipoperoxyl radical-scavenging activity of indicaxanthin in solution and unilamellar liposomes. Free Radic. Res. 2007, 41, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Allegra, M.; Furtmüller, P.G.; Jantschko, W.; Zederbauer, M.; Tesoriere, L.; Livrea, M.A.; Obinger, C. Mechanism of interaction of betanin and indicaxanthin with human myeloperoxidase and hypochlorous acid. Biochem. Biophys. Res. Commun. 2005, 332, 837–844. [Google Scholar] [CrossRef]
- Tesoriere, L.; Butera, D.; D’Arpa, D.; Di Gaudio, F.; Allegra, M.; Gentile, C.; Livrea, M.A. Increased resistance to oxidation of betalain-enriched human low-density lipoproteins. Free Radiac. Res. 2003, 37, 689–696. [Google Scholar] [CrossRef]
- Tesoriere, L.; Butera, D.; Allegra, M.; Fazzari, M.; Livrea, M.A. Distribution of betalain pigments in red blood cells after consumption of cactus pear fruits and increased resistance of the cells to ex vivo induced oxidative hemolysis in humans. J. Agric. Food Chem. 2005, 53, 1266–1270. [Google Scholar] [CrossRef]
- Tesoriere, L.; Allegra, M.; Butera, D.; Gentile, C.; Livrea, M.A. Cytoprotective effects of the antioxidant phytochemical indicaxanthin in β-thalassemia red blood cells. Free Radic. Res. 2006, 40, 753–761. [Google Scholar] [CrossRef]
- Gomez-Maqueo, A.; Garcia-Cayuela, T.; Fernandez-Lopez, R.; Welti-Chanes, J.; Cano, M.P. Inhibitory potential of prickly pears and their isolated bioactives against digestive enzymes linked to type 2 diabetes and inflammatory response. J. Sci. Food Agric. 2019, 99, 6380–6391. [Google Scholar] [CrossRef]
- Tesoriere, L.; Allegra, M.; Butera, D.; Livrea, M.A. Absorption, escretion and distribution in low density lipoproteins of dietary antioxidant betalains. Potential health effects of betalains in humans. Am. J. Clin Nutr. 2004, 80, 941–945. [Google Scholar] [CrossRef]
- Tesoriere, L.; Attanzio, A.; Allegra, M.; Gentile, C.; Livrea, M.A. Indicaxanthin inhibits NADPH oxidase (NOX)-1 activation and NF-κB-dependent release of inflammatory mediators and prevents the increase of epithelial permeability in IL-1β-exposed Caco-2 cells. Br. J. Nutr. 2014, 111, 415–423. [Google Scholar] [CrossRef]
- Tesoriere, L.; Attanzio, A.; Allegra, M.; Gentile, C.; Livrea, M.A. Phytochemical indicaxanthin suppresses 7-ketocholesterol-induced THP-1 cell apoptosis by preventing cytosolic Ca2+ increase and oxidative stress. Br. J. Nutr. 2013, 110, 230–240. [Google Scholar] [CrossRef]
- Baldassano, S.; Tesoriere, L.; Rotondo, A.; Serio, R.; Livrea, M.A.; Mulè, F. Inhibition of the mechanical activity of mouse ileum by cactus pear (Opuntia Ficus Indica, L, Mill.) fruit extract and its pigment indicaxanthin. J. Agric. Food Chem. 2010, 58, 7565–7571. [Google Scholar] [CrossRef]
- Campisi, A.; Raciti, G.; Sposito, G.; Grasso, R.; Chiacchio, M.A.; Spatuzza, M.; Attanzio, A.; Chiacchio, U.; Tesoriere, L.; Allegra, M.; et al. Amyloid-Beta Induces Different Expression Pattern of Tissue Transglutaminase and Its Isoforms on Olfactory Ensheathing Cells: Modulatory Effect of Indicaxanthin. Int. J. Mol. Sci. 2021, 22, 3388–3409. [Google Scholar] [CrossRef]
- Allegra, M.; Carletti, F.; Gambino, G.; Tutone, M.; Attanzio, A.; Tesoriere, L.; Ferraro, G.; Sardo, P.; Almerico, A.M.; Livrea, M.A. Indicaxanthin from Opuntia ficus-indica Crosses the Blood-Brain Barrier and Modulates Neuronal Bioelectric Activity in Rat Hippocampus at Dietary-Consistent Amounts. J. Agric. Food Chem. 2015, 63, 7353–7360. [Google Scholar] [CrossRef]
- Gambino, G.; Allegra, M.; Sardo, P.; Attanzio, A.; Tesoriere, L.; Livrea, M.A.; Ferraro, G.; Carletti, F. Brain Distribution and Modulation of Neuronal Excitability by Indicaxanthin from Opuntia Ficus Indica administered at Nutritionally-Relevant Amounts. Front. Aging. Neurosci. 2018, 10, 133. [Google Scholar] [CrossRef]
- Tesoriere, L.; Attanzio, A.; Allegra, M.; Livrea, M.A. Dietary indicaxanthin from cactus pear (Opuntia ficus-indica L. Mill) fruit prevents eryptosis induced by oxysterols in a hypercholesterolaemia relevant proportion and adhesion of human erythrocytes to endothelial cell layers. Br. J. Nutr. 2015, 114, 368–375. [Google Scholar] [CrossRef]
- Attanzio, A.; Frazzitta, A.; Busa’, R.; Tesoriere, L.; Livrea, M.A.; Allegra, M. Indicaxanthin from Opuntia ficus indica (L. Mill) Inhibits Oxidized LDL-Mediated Human Endothelial Cell Dysfunction through Inhibition of NF-κB Activation. Oxid. Med. Cell. Longev. 2019, 18, 3457846. [Google Scholar] [CrossRef]
- Gentile, C.; Tesoriere, L.; Allegra, M.; Livrea, M.A.; D’Alessio, P. Antioxidant betalains from cactus pear (Opuntia ficus indica) inhibit endothelial ICAM-1 expression. Ann. N. Y. Acad. Sci. 2004, 1028, 481–486. [Google Scholar] [CrossRef]
- Allegra, M.; D’Acquisto, F.; Tesoriere, L.; Attanzio, A.; Livrea, M.A. Pro-oxidant activity of indicaxanthin from Opuntia ficus indica modulates arachidonate metabolism and prostaglandin synthesis through lipid peroxide production in LPS-stimulated RAW 264.7 macrophages. Redox. Biol. 2014, 2, 892–900. [Google Scholar] [CrossRef][Green Version]
- Naselli, F.; Tesoriere, L.; Caradonna, F.; Bellavia, D.; Attanzio, A.; Gentile, C.; Livrea, M.A. Anti-proliferative and pro-apoptotic activity of whole extract and isolated indicaxanthin from Opuntia ficus-indica associated with re-activation of the onco-suppressor p16(INK4a) gene in human colorectal carcinoma (Caco-2) cells. Biochem. Biophys. Res. Commun. 2014, 450, 652–658. [Google Scholar] [CrossRef]
- Allegra, M.; D’Anneo, A.; Frazzitta, A.; Restivo, I.; Livrea, M.A.; Attanzio, A.; Tesoriere, L. The Phytochemical Indicaxanthin Synergistically Enhances Cisplatin-Induced Apoptosis in HeLa Cells via Oxidative Stress-Dependent p53/p21waf1 Axis. Biomolecules 2020, 10, 994. [Google Scholar] [CrossRef]
- Allegra, M.; De Cicco, P.; Ercolano, G.; Attanzio, A.; Busà, R.; Girino, G.; Tesoriere, L.; Livrea, M.A.; Ianaro, A. Indicaxanthin from Opuntia Ficus Indica (L. Mill) impairs melanoma cell proliferation, invasiveness, and tumor progression. Phytomedicine 2018, 15, 19–24. [Google Scholar] [CrossRef]
- Baldassano, S.; Rotondo, A.; Serio, R.; Livrea, M.A.; Tesoriere, L.; Mulè, F. Inhibitory effects of indicaxanthin on mouse ileal contractility: Analysis of the mechanism of action. Eur. J. Pharmacol. 2011, 658, 200–205. [Google Scholar] [CrossRef]
- Naselli, F.; Belshaw, N.J.; Gentile, C.; Tutone, M.; Tesoriere, L.; Livrea, M.A.; Caradonna, F. Phytochemical Indicaxanthin Inhibits Colon Cancer Cell Growth and Affects the DNA Methylation Status by Influencing Epigenetically Modifying Enzyme Expression and Activity. J. Nutr. Nutrgenom. 2015, 8, 114–127. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Rubio, M.A.; Hernández-García, S.; García-Carmona, F.; Gandía-Herrero, F. Extension of life-span using a RNAi model and in vivo antioxidant effect of Opuntia fruit extracts and pure betalains in Caenorhabditis elegans. Food Chem. 2019, 274, 840–847. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Rubio, M.A.; Hernandez-Garcia, S.; Escribano, J.; Jimenez-Atienzae, M.; Cabanes, J.; Garcia-Carmona, F.; Gandia-Herrero, F. Betalain health promoting effects after ingestion in Caenorhabditis elegans are mediated by DAF-16/FOXO and SKN-1/Nrf2 transcription factors. Food Chem. 2020, 330, 127228. [Google Scholar] [PubMed]
- Allegra, M.; Ianaro, A.; Tersigni, M.; Panza, E.; Tesoriere, L.; Livrea, M.A. Indicaxanthin from cactus pear fruit exerts anti-inflammatory effects in carrageenin-induced rat pleurisy. J. Nutr. 2014, 144, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Terzo, S.; Attanzio, A.; Calvi, P.; Mulè, F.; Tesoriere, L.; Allegra, M.; Amato, A. Indicaxanthin from Opuntia ficus-indica Fruit Ameliorates Glucose Dysmetabolism and Counteracts Insulin Resistance in High-Fat-Diet-Fed Mice. Antioxidants 2021, 11, 80. [Google Scholar] [CrossRef] [PubMed]
- Tesoriere, L.; Butera, D.; Pintaudi, A.M.; Allegra, M.; Livrea, M.A. Supplementation with cactus pear (Opuntia ficus-indica) fruit decreases oxidative stress in healthy humans: A comparative study with vitamin C. Am. J. Clin Nutr. 2004, 80, 391–395. [Google Scholar] [CrossRef]
- Kanner, J.; Harel, S.; Granit, R. Betalains—A new class of dietary antioxidants. J. Agric. Food Chem. 2001, 49, 5178–5185. [Google Scholar] [CrossRef]
- Tutone, M.; Lauria, A.; Almerico, A.F. Theoretical Determination of the PK a Values of Betalamic Acid Related to the Free Radical Scavenger Capacity: Comparison Between Empirical and Quantum Chemical Methods. Interdiscip. Sci. 2016, 8, 177–185. [Google Scholar] [CrossRef]
- Escribano, J.; Pedreno, M.A.; Garcia-Carmona, F.; Munoz, R. Characterization of the antiradical activity of betalains from Beta vulgaris, L. roots. Phytochem. Anal. 1998, 9, 124–127. [Google Scholar] [CrossRef]
- Gandia-Herrero, F.; Escribano, J.; Garcia-Carmona, F. The role of phenolic hydroxy groups in the free radical scavenging activity of betalains. J. Nat. Prod. 2009, 72, 1142–1146. [Google Scholar] [CrossRef]
- Butera, D.; Tesoriere, L.; Di Gaudio, F.; Bongiorno, A.; Allegra, M.; Pintaudi, A.M.; Livrea, M.A. Antioxidant activities of Sicilian prickly pear (Opuntia ficus indica) fruit extracts and reducing properties of its betalains: Betanin and indicaxanthin. J. Agric. Food Chem. 2002, 50, 6895–6901. [Google Scholar] [CrossRef]
- Spiegel, M.; Gamian, A.; Sroka, Z. Antiradical Activity of Beetroot (Beta vulgaris L.) Betalain. Molecules 2021, 26, 2439. [Google Scholar] [CrossRef]
- Buettner, G.R. The pecking order of free radicals and antioxidants: Lipid peroxidation, alpha-tocopherol, and ascorbate. Arch. Biochem. Biophys. 1993, 300, 535–543. [Google Scholar] [CrossRef]
- Wang, Y.; Fernando, G.S.N.; Sergeeva, N.N.; Vagkidis, N.; Chechik, V.; Do, T.; Marshall, L.J.; Boesch, C. Uptake and immunomodulatory properties of Betanin, Vulgaxanthin I and Indicaxanthin towards Caco-2 intestinal cells. Antioxidants 2022, 11, 1627–1646. [Google Scholar] [CrossRef]
- Tesoriere, L.; Gentile, C.; Angileri, F.; Attanzio, A.; Tutone, M.; Allegra, M.; Livrea, M.A. Trans-epithelial transport of the betalain pigments indicaxanthin and betanin across Caco-2 cell monolayers and influence of food matrix. Eur. J. Nutr. 2013, 52, 1077–1087. [Google Scholar] [CrossRef]
- Dressman, J.B.; Berardi, R.R.; Dermentzoglou, L.C.; Russel, T.L.; Schmaltz, S.P.; Barnett, J.L.; Jarvenpaa, K.M. Upper gastrointestinal (GI) pH in young, healthy men and women. Pharm. Res. 1990, 7, 756–761. [Google Scholar] [CrossRef]
- Lucas, M. Determination of acid surface pH in vivo in rat proximal jejunum. Gut 1983, 24, 734–739. [Google Scholar] [CrossRef]
- Turco-Liveri, M.L.; Sciascia, L.; Lombardo, R.; Tesoriere, L.; Passante, E.; Livrea, M.A. Spectrophotometric evidence for the solubilization site of betalain pigments in membrane biomimetic systems. J. Agric. Food Chem. 2007, 55, 2836–2840. [Google Scholar] [CrossRef]
- Turco-Liveri, M.L.; Sciascia, L.; Allegra, M.; Tesoriere, L.; Livrea, M.A. Partition of indicaxanthin in membrane biomimetic systems. A kinetic and modeling approach. J. Agric. Food Chem. 2009, 57, 10959–10963. [Google Scholar] [CrossRef]
- Barclay, L.R.C.; Baskin, K.A.; Kong, G.; Locke, S.J. Autoxidation of model membranes. The kinetics and mechanism of autoxidation of mixed phospholipid bilayers. Can. J. Chem. 1987, 65, 2541–2550. [Google Scholar] [CrossRef]
- Barclay, L.R.C. Syntex Award Lecture Model biomembranes: Quantitative studies of peroxidation, antioxidant action, partitioning, and oxidative stress. Can. J. Chem. 1992, 71, 97624269. [Google Scholar]
- Rietjens, I.M.; Boersma, M.G.; de Haan, L.; Spenkelink, B.; Awad, H.M.; Cnubben, N.H.; van Zanden, J.J.; van der Woude, H.; Alink, G.M.; Koeman, J.H. The pro-oxidant chemistry of the natural antioxidants vitamin C, vitamin E, carotenoids and flavonoids. Environ. Toxicol. Pharmacol. 2002, 11, 321–333. [Google Scholar] [CrossRef] [PubMed]
- Patel, R.P.; Boersma, B.J.; Crawford, J.H.; Hogg, N.; Kirk, M.; Kalyanaraman, B.; Parks, D.A.; Barnes, S.; Darley-Usmar, V. Antioxidant mechanisms of isoflavones in lipid systems: Paradoxical effects of peroxyl radical scavenging. Free Radic. Biol. Med. 2001, 31, 1570–1581. [Google Scholar] [CrossRef]
- Brigelius-Flohé, R. Vitamin E: The shrew waiting to be tamed. Free Radic. Biol. Med. 2009, 46, 543–554. [Google Scholar] [CrossRef]
- Tesoriere, L.; D’Arpa, D.; Re, R.; Livrea, M.A. Antioxidant reactions of all-trans retinol in phospholipid bilayers: Effect of oxygen partial pressure, radical fluxes, and retinol concentration. Arch. Biochem. Biophys. 1997, 343, 13–18. [Google Scholar] [CrossRef]
- Yeum, K.J.; Aldini, G.; Russell, R.M.; Krinsky, N.I. Antioxidant/Pro-oxidant Actions of Carotenoids. In Carotenoids; Birkhäuser: Basel, Switzerland, 2009. [Google Scholar]
- Graham, H.N. Green tea composition, consumption, and polyphenol chemistry. Prev. Med. 1992, 21, 334–350. [Google Scholar] [CrossRef]
- Esterbauer, H.; Gebicki, J.; Puhl, H.; Jurgens, G. The role of lipid peroxidation and antioxidants in oxidative modification of LDL. Free Radic. Biol. Med. 1992, 13, 341–390. [Google Scholar] [CrossRef]
- Casares, D.; Escriba, P.V.; Rossello, C.A. Membrane lipid composition: Effect on membrane and organelle structure, function and compartmentalization and therapeutic avenues. Int. J. Mol. Sci. 2019, 20, 2167. [Google Scholar] [CrossRef]
- Niki, E. Role of vitamin E as a lipid-soluble peroxyl radical scavenger: In vitro and in vivo evidence. Free Radic. Biol. Med. 2014, 66, 3–12. [Google Scholar] [CrossRef]
- Francis, S.H.; Blount, M.A.; Corbin, J.D. Mammalian cyclic nucleotide phosphodiesterases: Molecular mechanisms and physiological functions. Physiol. Rev. 2011, 91, 651–690. [Google Scholar] [CrossRef] [PubMed]
- Lugnier, C. Cyclic nucleotide phosphodiesterase (PDE) superfamily: A new target for the development of specific therapeutic agents. Pharmacol. Ther. 2006, 109, 366–398. [Google Scholar] [CrossRef] [PubMed]
- Tutone, M.; Virzì, A.; Almerico, A.M. Reverse screening on indicaxanthin from Opuntia ficus indica as natural chemoactive and chemopreventive agent. J. Theoretical. Biol. 2018, 455, 147–160. [Google Scholar] [CrossRef] [PubMed]
- Allegra, M.; Tutone, M.; Tesoriere, L.; Attanzio, A.; Culletta, G.; Almerico, A.M. Evaluation of the IKKβ Binding of Indicaxanthin by Induced-Fit Docking, Binding Pose Metadynamics, and Molecular Dynamics. Front. Pharmacol. 2021, 12, 701568. [Google Scholar] [CrossRef]
- Yu, H.; Lin, L.; Zhang, Z.; Zhang, H.; Hu, H. Targeting NF-κB pathway for the therapy of diseases: Mechanism and clinical study. Signal. Transduct. Target. Ther. 2020, 5, 209. [Google Scholar] [CrossRef]
- Cheplick, S.; Kwon, Y.I.; Bhowmik, P.; Shetty, K. Phenolic-linked variation in strawberry cultivars for potential dietary management of hyperglycemia and related complications of hypertension. Bioresour. Technol. 2010, 101, 404–413. [Google Scholar] [CrossRef]
- Puls, W.; Keup, U.; Krause, H.P.; Thomas, G.; Hoffmeister, F. Glucosidase inhibition. A new approach to the treatment of diabetes, obesity, and hyperlipoproteinaemia. Naturwissenschaften 1977, 64, 536–537. [Google Scholar] [CrossRef]
- Ndlovu, G.; Fouche, G.; Tselanyane, M.; Cordier, W.; Steenkamp, V. In vitro determination of the anti-aging potential of four southern African medicinal plants. BMC Complement Altern. Med. 2013, 13, 304–310. [Google Scholar] [CrossRef]
- Fronza, M.; Caetano, G.F.; Leite, M.N.; Bitencourt, C.S.; Paula-Silva, F.W.; Andrade, T.A.; Frade, M.A.; Merfort, I.; Faccioli, L.H. Hyaluronidase modulates inflammatory response and accelerates the cutaneous wound healing. PLoS ONE 2014, 9, 112297. [Google Scholar] [CrossRef]
- Lee, M.T.; Lin, W.C.; Yu, B.; Lee, T.T. Antioxidant capacity of phytochemicals and their potential effects on oxidative status in animals—A review. Asian. Australas. J. Anim Sci. 2017, 30, 299–308. [Google Scholar] [CrossRef]
- Manach, C.; Williamson, G.; Morand, C.; Scalbert, A.; Rémésy, C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am. J. Clin. Nutr. 2005, 81, 230S–242S. [Google Scholar] [CrossRef] [PubMed]
- Tesoriere, L.; Fazzari, M.; Angileri, F.; Gentile, C.; Livrea, M.A. In vitro digestion of betalainic foods. Stability and bioaccessibility of betaxanthins and betacyanins and antioxidative potential of food digesta. J. Agric. Food Chem. 2008, 56, 10487–10492. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Maqueo, A.; Antunes-Ricardo, M.; Welti-Chanes, J.; Piòar-Cano, M. Digestive stability and bioaccessibiity of antioxidants in prickly pear fruits from the Canary Islands: Healthy foods and ingredients. Antioxidants 2020, 9, 164–181. [Google Scholar] [CrossRef] [PubMed]
- Montiel-Sánchez, M.; García-Cayuela, T.; Gómez-Maqueo, A.; García, H.S.; Cano, M.P. In vitro gastrointestinal stability, bioaccessibility and potential biological activities of betalains and phenolic compounds in cactus berry fruits (Myrtillocactus geometrizans). Food Chem. 2021, 342, 128087–128098. [Google Scholar] [CrossRef] [PubMed]
- Trachootham, D.; Lu, W.; Ogasawara, M.A.; Nilsa, R.D.; Huang, P. Redox regulation of cell survival. Antioxid. Redox. Signal. 2008, 10, 1343–1374. [Google Scholar] [CrossRef] [PubMed]
- Lambeth, J.D. Nox enzymes, ROS, and chronic disease: An example of antagonistic pleiotropy. Free Radic. Biol. Med. 2007, 43, 332–347. [Google Scholar] [CrossRef]
- West, A.P.; Brodsky, I.E.; Rahner, C.; Woo, D.K.; Erdjument-Bromage, H.; Tempst, P.; Walsh, M.C.; Choi, Y.; Shadel, G.S.; Ghosh, S. TLR signalling augments macrophage bactericidal activity through mitochondrial ROS. Nature 2011, 472, 476–480. [Google Scholar] [CrossRef]
- Canton, M.; Sánchez-Rodríguez, R.; Spera, I.; Venegas, F.C.; Favia, M.; Viola, A.; Castegna, A. Reactive Oxygen Species in Macrophages: Sources and Targets. Front. Immunol. 2021, 12, 734229. [Google Scholar] [CrossRef]
- Harry, R.S.; Hiatt, L.A.; Kimmel, D.W.; Carney, C.K.; Halfpenny, K.C.; Cliffel, D.E.; Wright, D.W. Metabolic impact of 4-hydroxynonenal on macrophage-like RAW 264.7 function and activation. Chem. Res. Toxicol. 2012, 25, 1643–1651. [Google Scholar] [CrossRef][Green Version]
- Poli, G.; Leonarduzzi, G.; Biasi, F.; Chiarpotto, E. Oxidative stress and cell signaling. Curr. Med. Chem. 2004, 11, 1163–1182. [Google Scholar] [CrossRef]
- Forman, H.J.; Fukuto, J.M.; Miller, T.; Zhang, H.; Rinna, A.; Levy, S. The chemistry of cell signaling by reactive oxygen and nitrogen species and 4-hydroxynonenal. Arch. Biochem. Biophys. 2008, 477, 183–195. [Google Scholar] [CrossRef]
- Lopez Bernardo, E.; Anedda, A.; Sánchez-Pérez, P.; Acosta-Iborra, B.; Cadenas, S. 4-Hydroxynonenal induces Nrf2-mediated UCP3 upregulation in mouse cardiomyocytes. Free Radic. Biol. Med. 2015, 88, 427–438. [Google Scholar] [CrossRef]
- Kumagai, T.; Matsukawa, N.; Kaneko, Y.; Kusumi, Y.; Mitsumata, M.; Uchida, K. A lipid peroxidation-derived inflammatory mediator: Identification of 4-hydroxy-2-nonenal as a potential inducer of cyclooxygenase-2 in macrophages. J. Biol. Chem. 2004, 279, 48389–48396. [Google Scholar]
- Bochkov, V.N.; Leitinger, N. Anti-inflammatory properties of lipid oxidation products. J. Mol. Med. 2003, 81, 613–626. [Google Scholar]
- Esser-von Bieren, J. Immune-regulation and functions of eicosanoid lipid mediators. Biol. Chem. 2017, 398, 1177–1191. [Google Scholar] [PubMed]
- Dyall, S.C.; Balas, L.; Bazan, N.G.; Brenna, J.T.; Chiang, N.; da Costa Souza, F.; Dalli, J.; Durand, T.; Galano, J.M.; Lein, P.J.; et al. Polyunsaturated fatty acids and fatty acid-derived lipid mediators: Recent advances in the understanding of their biosynthesis, structures, and functions. Prog. Lipid Res. 2022, 86, 101165. [Google Scholar] [CrossRef]
- Sies, H. Hydrogen peroxide as a central redox signaling molecule in physiological oxidative stress: Oxidative eustress. Redox. Biol. 2017, 11, 613–619. [Google Scholar] [CrossRef]
- Kanner, J. Polyphenols by Generating H2O2, Affect Cell Redox Signaling, Inhibit PTPs and Activate Nrf2 Axis for Adaptation and Cell Surviving: In Vitro, In Vivo and Human Health. Antioxidants 2020, 9, 797. [Google Scholar] [CrossRef] [PubMed]
- Brandes, R.P.; Weissmann, N.; Schröder, K. Nox family NADPH oxidases: Molecular mechanisms of activation. Free Radic. Biol. Med. 2014, 76, 208–226. [Google Scholar] [CrossRef]
- Skonieczna, M.; Hejmo, T.; Poterala-Hejmo, A.; Cieslar-Pobuda, A.; Buldak, R.J. NADPH Oxidases: Insights into Selected Functions and Mechanisms of Action in Cancer and Stem Cells. Oxid. Med. Cell. Longev. 2017, 2017, 9420539. [Google Scholar] [CrossRef]
- Jaquet, V.; Scapozza, L.; Clark, R.A.; Krause, K.H.; Lambeth, J.D. Small-molecule NOX inhibitors: ROS-generating NADPH oxidases as therapeutic targets. Antioxid. Redox. Signal. 2009, 11, 2535. [Google Scholar] [CrossRef]
- Krause, K.H.; Lambeth, D.; Krönke, M. NOX enzymes as drug targets. Cell. Mol. Life Sci. 2012, 69, 2279–2282. [Google Scholar] [CrossRef]
- Szanto, I.; Rubbia-Brandt, L.; Kiss, P.; Steger, K.; Banfi, B.; Kovari, E.; Herrmann, F.; Hadengue, A.; Krause, K.H. Expression of NOX1, a superoxide-generating NADPH oxidase, in colon cancer and inflammatory bowel disease. J. Pathol. 2005, 207, 164–176. [Google Scholar] [CrossRef]
- Ligumsky, M.; Simon, P.L.; Karmeli, F.; Rachmilewitz, D. Role of interleukin-1 in inflammatory bowel-disease-enhanced production during active disease. Gut 1990, 31, 686–689. [Google Scholar] [CrossRef]
- Reinecker, H.C.; Steffen, M.; Witthoeft, T.; Pflueger, I.; Schreiber, S.; MacDermott, R.P.; Raedler, A. Enhanced secretion of tumor-necrosis-factor-alpha, IL-6, and IL-1 beta by isolated lamina propria mononuclear-cells from patients with ulcerative-colitis and Crohn’s-disease. Clin. Exp. Immunol. 1993, 94, 174–181. [Google Scholar] [CrossRef]
- Böcker, U.; Damião, A.; Holt, L.; Han, D.S.; Jobin, C.; Panja, A.; Mayer, L.; Sartor, R.B. Differential expression of interleukin 1 receptor antagonist isoforms in human intestinal epithelial cells. Gastroenterology 1998, 115, 1426–1438. [Google Scholar] [CrossRef]
- Brahmi, F.; Vejux, A.; Sghaier, R.; Zarrouk, A.; Nury, T.; Meddeb, W.; Rezig, L.; Namsi, A.; Sassi, K.; Yammine, A.; et al. Prevention of 7-ketocholesterol-induced side effects by natural compounds. Crit. Rev. Food Sci. Nutr. 2019, 59, 3179–3198. [Google Scholar] [CrossRef]
- Martyn, K.D.; Frederick, L.M.; von Loehneysen, K.; Dinauer, D.C.; Knaus, U.G. Functional analysis of Nox4 reveals unique characteristics compared to other NADPH oxidases. Cell Signal. 2006, 18, 69–82. [Google Scholar] [CrossRef]
- Leto, T.L.; Morand, S.; Hurt, D.; Ueyama, T. Targeting and regulation of reactive oxygen species generation by Nox family NADPH oxidases. Antioxid. Redox Signal. 2009, 11, 2607–2619. [Google Scholar] [CrossRef]
- Oakley, F.D.; Smith, R.L.; Engelhardt, J.F. Lipid rafts and caveolin-1 coordinate interleukin-1beta (IL-1beta)-dependent activation of NFkB by controlling endocytosis of Nox2 and IL-1beta receptor 1 from the plasma membrane. J. Biol. Chem. 2009, 284, 33255–33264. [Google Scholar] [CrossRef]
- Gustafson, S.J.; Dunlap, K.L.; McGill, C.M.; Kuhn, T.B. A nonpolar blueberry fraction blunts NADPH oxidase activation in neuronal cells exposed to tumor necrosis factor-α. Oxid. Med. Cell. Longev. 2012, 2012, 768101. [Google Scholar] [CrossRef]
- Berthier, A.; Lemaire-Ewing, S.; Prunet, C.; Monier, S.; Athias, A.; Bessède, G.; Pais de Barros, J.-P.; Laubriet, A.; Gambert, P.; Lizard, G.; et al. Involvement of a calcium-dependent dephosphorylation of BAD associated with the localization of Trpc-1 within lipid rafts in 7-ketocholesterol-induced THP-1 cell apoptosis. Cell Death Differ. 2004, 11, 805–897. [Google Scholar] [CrossRef]
- Massey, J.B.; Pownall, H.J. Structures of biologically active oxysterols determine their differential effects on phospholipid membranes. Biochemistry 2006, 45, 10747–10758. [Google Scholar] [CrossRef]
- Olkkonen, V.M.; Hynynen, R. Interactions of oxysterols with membranes and proteins. Mol. Asp. Med. 2009, 30, 123–133. [Google Scholar] [CrossRef]
- Massey, J.B.; Pownall, H.J. The polar nature of 7-ketocholesterol determines its location within membrane domains and the kinetics of membrane microsolubilization by apolipoprotein A-I. Biochemistry 2005, 44, 10423–10433. [Google Scholar] [CrossRef] [PubMed]
- Han, W.; Li, H.; Villar, V.A.; Pascua, A.M.; Dajani, M.I.; Wang, X.; Natarajan, A.; Quinn, M.T.; Felde, R.A.; Jose, P.A.; et al. Lipid rafts keep NADPH oxidase in the inactive state in human renal proximal tubule cells. Hypertension 2008, 51, 481–487. [Google Scholar] [CrossRef] [PubMed]
- Shao, D.; Segal, A.W.; Dekker, L.V. Lipid rafts determine efficiency of NADPH oxidase activation in neutrophils. FEBS Lett. 2003, 550, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Lang, F.; Lang, K.S.; Lang, P.A.; Huber, S.M.; Wieder, T. Mechanisms and significance of eryptosis. Antioxid. Redox Signal. 2006, 8, 1183–1192. [Google Scholar] [CrossRef] [PubMed]
- Tesoriere, L.; Attanzio, A.; Allegra, M.; Cilla, A.; Gentile, C.; Livrea, M.A. Oxysterol mixture in a hypercholesterolaemia-relevant proportion causes oxidative stress-dependedt eryptosis. Cell Physiol. Biochem. 2014, 34, 1075–1089. [Google Scholar] [CrossRef]
- Attanzio, A.; Frazzitta, A.; Cilla, A.; Livrea, M.A.; Tesoriere, L.; Allegra, M. 7-Keto-Cholesterol and Cholestan-3beta, 5alpha, 6beta-Triol Induce Eryptosis through Distinct Pathways Leading to NADPH Oxidase and Nitric Oxide Synthase Activation. Cell. Physiol. Biochem. 2019, 53, 933–947. [Google Scholar]
- Negre-Salvayre, A.; Augé, N.; Camaré, C.; Bacchetti, T.; Ferretti, G.; Salvayre, R. Dual signalling evoked by oxidized LDLs in vascular cells. Free Radic. Biol. Med. 2017, 106, 118–133. [Google Scholar] [CrossRef] [PubMed]
- Poznyak, A.; Grecho, A.V.; Orekhova, V.A.; Khotina, V.; Ivanova, E.A.; Orekhov, A.N. NADPH oxidases and their role in atherosclerosis. Biomedicines 2020, 8, 206–218. [Google Scholar] [CrossRef] [PubMed]
- Iademarco, M.F.; Barks, J.L.; Dean, D.C. Regulation of vascular cell adhesion molecule-1 expression by IL-4 and TNF-alpha in cultured endothelial cells. J. Clin. Investig. 1995, 95, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Andresen, B.T.; Hill, M.; Zhang, J.; Booth, F.; Zhang, C. Role of Reactive Oxygen Species in Tumor Necrosis Factor-alpha Induced Endothelial Dysfunction. Curr. Hypertens. Rev. 2008, 4, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Urschel, K.; Cicha, I. TNF-α in the cardiovascular system: From physiology to therapy. Int. J. Interferon, Cytokine Mediat. Res. 2015, 7, 9–25. [Google Scholar]
- Dibattista, M.; Pifferi, S.; Menini, A.; Reisert, J. Alzheimer’s Disease: What Can We Learn From the Peripheral Olfactory System? Front. Neurosci. 2020, 14, 440. [Google Scholar] [CrossRef] [PubMed]
- Attems, J.; Walker, L.; Jellinger, K.A. Olfactory bulb involvement in neurodegenerative diseases. Acta Neuropathol. 2014, 127, 459–475. [Google Scholar]
- Gandy, S. The role of cerebral amyloid β accumulation in common forms of Alzheimer disease. J. Clin. Investig. 2005, 115, 1121–1129. [Google Scholar]
- De Paula, V.J.; Radanovic, M.; Diniz, B.S.; Forlenza, O.V. Alzheimer’s disease. Subcell. Biochem. 2012, 65, 329–352. [Google Scholar]
- Singh, G.; Zhang, J.; Ma, Y.; Cerione, R.A.; Antonyak, M.A. The Different Conformational States of Tissue Transglutaminase Have Opposing Affects on Cell Viability. J. Biol. Chem. 2016, 291, 9119–9132. [Google Scholar] [CrossRef]
- Min, B.; Chung, K.C. New insight into transglutaminase 2 and link to neurodegenerative diseases. BMB. Rep. 2018, 51, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Campisi, A.; Spatuzza, M.; Russo, A.; Raciti, G.; Vanella, A.; Stanzani, S.; Pellitteri, R. Expression of tissue transglutaminase on primary olfactory ensheathing cells cultures exposed to stress conditions. Neurosci. Res. 2012, 72, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Pellitteri, R.; Bonfanti, R.; Spatuzza, M.; Cambria, M.T.; Ferrara, M.; Raciti, G.; Campisi, A. Effect of some growth factors on tissue transglutaminase overexpression induced by β-amyloid in olfactory ensheathing cells. Mol. Neurobiol. 2017, 54, 6785–6794. [Google Scholar] [CrossRef] [PubMed]
- Caccamo, D.; Currò, M.; Ferlazzo, N.; Condello, S.; Ientile, R. Monitoring of transglutaminase 2 under different oxidative stress conditions. Amino Acids 2012, 42, 1037–1043. [Google Scholar] [CrossRef]
- Shin, D.-M.; Jeon, J.H.; Kim, C.W.; Cho, S.Y.; Kwon, J.C.; Lee, H.J.; Choi, K.H.; Park, S.C.; Kim, I.G. Cell type-specific activation of intracellular transglutaminase 2 by oxidative stress or ultraviolet irradiation: Implications of transglutaminase 2 in age-related cataractogenesis. J. Biol. Chem. 2004, 279, 15032–15039. [Google Scholar] [CrossRef]
- Shin, D.; Jeon, J.; Kim, C.; Cho, S.; Lee, H.; Jang, G.; Jeong, E.M.; Lee, D.; Kang, J.; Melino, G.; et al. TGFβ mediates activation of transglutaminase 2 in response to oxidative stress that leads to protein aggregation. FASEB J. 2008, 22, 2498–2507. [Google Scholar] [CrossRef]
- Jeong, E.M.; Kim, I.G. Regulation of Transglutaminase 2 by Oxidative Stress. In Transglutaminases; Hitomi, K., Kojima, S., Fesus, L., Eds.; Springer: Tokyo, Japan, 2015. [Google Scholar]
- Bianca, V.D.; Dusi, S.; Bianchini, E.; Dal Prà, I.; Rossi, F. beta-amyloid activates the O-2 forming NADPH oxidase in microglia, monocytes, and neutrophils. A possible inflammatory mechanism of neuronal damage in Alzheimer’s disease. J. Biol. Chem. 1999, 274, 15493–15499. [Google Scholar] [CrossRef]
- Geng. L.; Fan, L.M.; Liu, F.; Smith, C.; Li, J. Nox2 dependent redox-regulation of microglial response to amyloid-β stimulation and microgliosis in aging. Sci Rep. 2020, 10, 1582–1592. [Google Scholar] [CrossRef]
- Niu, Z.; Zhang, Z.; Zhao, W.; Yang, I. Interaction between amyloid-β peptide and lipid membrane. Biochim. Biophys. Acta Biomembr. 2018, 1860, 1663–1669. [Google Scholar] [CrossRef]
- Evangelisti, E.; Wright, D.; Zampagni, M.; Cascella, R.; Fiorillo, C.; Bagnoli, S.; Relini, A.; Nichino, D.; Scartabelli, T.; Nacmias, B.; et al. Lipid rafts mediate amyloid-induced calcium dyshomeostasis and oxidative stress in Alzheimer’s disease. Curr. Alzheimer Res. 2013, 10, 143–153. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rushworth, J.V.; Hooper, N.M. Lipid Rafts: Linking Alzheimer’s Amyloid-β Production, Aggregation, and Toxicity at Neuronal Membranes. Int. J. Alzheimer’s Dis. 2010, 2011, 603052. [Google Scholar] [CrossRef] [PubMed]
- Konaté, M.M.; Antony, S.; Doroshow, J.H. Inhibiting the Activity of NADPH Oxidase in Cancer. Antioxid. Redox. Signal. 2020, 33, 435–454. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, S.; Meijles, D.N.; Pagano, P.J. NADPH oxidase: Key modulators in aging and age-related cardiovascular disease? Clin. Sci. 2016, 130, 317–335. [Google Scholar]
- Ewald, C.Y. Redox Signaling of NADPH Oxidases Regulates Oxidative Stress Responses, Immunity and Aging. Antioxidants 2018, 7, 130. [Google Scholar] [CrossRef]
- Maraldi, T. Natural compounds as modulators of NADPH oxidases. Oxid. Med. Cell. Longev. 2013, 2013, 271602. [Google Scholar] [CrossRef]
- Moßhammer, M.R.; Rohe, M.; Stintzing, F.C.; Carle, R. Stability of yellow-orange cactus pear (Opuntia ficus-indica (L.) Mill. cv.’Gialla’) betalains as affected by the juice matrix and selected food additives. Eur. Food Res. Technol. 2007, 225, 21–32. [Google Scholar] [CrossRef]
- Diamond, J. Role of cyclic nucleotides in control of smooth muscle contraction. In Advances in Cyclic Nucleotide Research; George, W.G., Ignarro, L.J., Eds.; Raven Press: New York, NY, USA, 1978; Volume 9, pp. 327–340. [Google Scholar]
- Makhlouf, G.M.; Murthy, K.S. Signal transduction in gastrointestinal smooth muscle. Cell. Signal. 1997, 9, 269–276. [Google Scholar] [CrossRef]
- Ke, H.; Wang, H. Crystal structures of phosphodiesterases and implications on substrate specificity and inhibitor selectivity. Curr. Top. Med. Chem. 2007, 7, 391–403. [Google Scholar] [CrossRef]
- Choudhari, A.S.; Mandave, P.C.; Deshpande, M.; Ranjekar, P.; Prakash, O. Phytochemicals in Cancer Treatment: From Preclinical Studies to Clinical Practice. Front. Pharmacol. 2020, 10, 1614. [Google Scholar] [CrossRef]
- Mollakhalili-Meybodi, N.; Mortazavian, A.M.; Bahadori-Monfared, A.; Sohrabvandi, S.; Aghaei-Meybodi, F. Phytochemicals in Cancer Prevention: A Review of the Evidence. Int. J. Cancer Manag. 2017, 10, 7219. [Google Scholar] [CrossRef]
- Aggarwal, V.; Tuli, H.S.; Varol, A.; Thakral, F.; Yerer, M.B.; Sak, K.; Varol, M.; Jain, A.; Khan, M.A.; Sethi, G. Role of Reactive Oxygen Species in Cancer Progression: Molecular Mechanisms and Recent Advancements. Biomolecules 2019, 9, 735. [Google Scholar] [CrossRef] [PubMed]
- Schieber, M.; Chandel, N.S. ROS function in redox signaling and oxidative stress. Curr. Biol. 2014, 24, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Waris, G.; Ahsan, H. Reactive oxygen species: Role in the development of cancer and various chronic conditions. J. Carcinog. 2006, 5, 14. [Google Scholar] [CrossRef] [PubMed]
- Ozben, T. Oxidative stress and apoptosis: Impact on cancer therapy. J. Pharm. Sci. 2007, 96, 2181–2196. [Google Scholar] [CrossRef] [PubMed]
- Hegedűs, C.; Kovács, K.; Polgár, Z.; Regdon, Z.; Szabó, É.; Robaszkiewicz, A.; Forman, H.J.; Martner, A.; Virág, L. Redox control of cancer cell destruction. Redox Biol. 2018, 16, 59–74. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Lee, Y.F.; Yeo, X.H.; Loo, S.Y.; Tam, W.L. Shifting the gears of metabolic plasticity to drive cell state transition in cancer. Cancers 2021, 13, 1316–1338. [Google Scholar] [CrossRef]
- Gaikwad, S.; Srivastava, S.K. Role of phytochemicals in perturbation of redox homeostasis in cancer. Antioxidants 2021, 10, 83–110. [Google Scholar] [CrossRef]
- Assi, M. The differential role of reactive oxygen species in early and late stages of cancer. Am. J. Physiol. Integr. Comp. Physiol. 2017, 313, 646–653. [Google Scholar] [CrossRef] [PubMed]
- Trachootham, D.; Alexandre, J.; Huang, P. Targeting cancer cells by ROS-mediated mechanisms: A radical therapeutic approach? Nat. Rev. Drug. Discov. 2009, 8, 579–591. [Google Scholar] [CrossRef]
- Kaur, V.; Kumar, M.; Kumar, A.; Kaur, K.; Dhillon, V.S.; Kaur, S. Pharmacotherapeutic potential of phytochemicals: Implications in cancer chemoprevention and future perspectives. Biomed. Pharmacother. 2018, 97, 564–586. [Google Scholar]
- El Omari, N.; Bakrim, S.; Bakha, M.; Lorenzo, J.M.; Rebezov, M.; Shariati, M.A.; Aboulaghras, S.; Balahbib, A.; Khayrullin, M.; Bouyahya, A. Natural Bioactive Compounds Targeting Epigenetic Pathways in Cancer: A Review on Alkaloids, Terpenoids, Quinones, and Isothiocyanates. Nutrients 2021, 13, 3714. [Google Scholar] [CrossRef] [PubMed]
- Juhasz, A.; Markel, S.; Gaur, S.; Liu, H.; Lu, J.; Jiang, G.; Wu, X.; Antony, S.; Wu, Y.; Melillo, G.; et al. NADPH oxidase 1 supports proliferation of colon cancer cells by modulating reactive oxygen species-dependent signal transduction. J. Biol. Chem. 2017, 292, 7866–7887. [Google Scholar] [CrossRef] [PubMed]
- Szlasa, W.; Zendran, I.; Zalesińska, A.; Tarek, M.; Kulbacka, J. Lipid composition of the cancer cell membrane. J. Bioenerg. Biomembr. 2020, 52, 321–342. [Google Scholar] [CrossRef]
- Jones, P.A.; Laird, P.W. Cancer epigenetics comes of age. Nat. Genet. 1999, 21, 163–167. [Google Scholar] [CrossRef] [PubMed]
- Frizelle, S.P.; Grim, J.; Zhou, J.; Gupta, P.; Curiel, D.T.; Geradts, J.; Kratzke, R.A. Re-expression of p16INK4a in mesothelioma cells results in cell cycle arrest, cell death, tumor suppression and tumor regression. Oncogene 1998, 16, 3087–3095. [Google Scholar] [CrossRef]
- Hingeldorf, K.I.; Leshchiner, E.S.; Nedelcu, S.; Maynard, M.A.; Calo, E.; Ianari, A.; Walensky, L.D.; Lees, J.A. The retinoblastoma proteininduces apoptosis directly at the mitochondria. Genes Dev. 2013, 27, 1003–1015. [Google Scholar]
- Knudsen, K.E.; Weber, E.; Arden, K.C.; Cavenee, W.K.; Feramisco, J.R.; Knudsen, E.S. The retinoblastoma tumor suppressor inhibits cellular proliferation through two distinct mechanisms: Inhibition of cell cycle progression and induction of cell death. Oncogene 1999, 18, 5239–5245. [Google Scholar] [CrossRef][Green Version]
- Kohli, R.M.; Zhang, Y. TET enzymes, TDG and the dynamics of DNA demethylation. Nature 2013, 502, 472–509. [Google Scholar] [CrossRef]
- Wu, R.; Li, S.; Hudlikar, R.; Wang, L.; Shannar, A.; Peter, R.; Chou, P.J.; Kuo, H.D.; Liu, Z.; Kong, A.N. Redox signaling, mitochondrial metabolism, epigenetics and redox active phytochemicals. Free Radic. Biol. Med. 2022, 179, 328–336. [Google Scholar] [CrossRef]
- Choi, S.H.; Heo, K.; Byun, H.M.; An, W.; Lu, W.; Yang, A.S. Identification of preferential target sites for human DNA methyltransferases. Nucleic Acids Res. 2011, 39, 104–118. [Google Scholar] [CrossRef]
- Mahé, S.; Huneau, J.F.; Marteau, P.; Thuillier, F.; Tomé, D. Gastroileal nitrogen and electrolyte movements after bovine milk ingestion in humans. Am. J. Clin. Nutr. 1992, 56, 410–416. [Google Scholar] [CrossRef] [PubMed]
- Cullen, S.; Ponnappan, S.; Ponnappan, U. Redox-regulated pathway of tyrosine phosphorylation underlies NF-κB induction by an atypical pathway independent of the 26S proteasome. Biomolecules 2015, 5, 95–112. [Google Scholar] [CrossRef] [PubMed]
- Flohé, L.; Brigelius-Flohé, R.; Saliou, C.; Traber, M.G.; Packer, L. Redox regulation of NF-kappa B activation. Free Radic. Biol. Med. 1997, 22, 1115–1126. [Google Scholar] [CrossRef] [PubMed]
- Oliveira-Marques, V.; Marinho, H.S.; Cyrne, L.; Antunes, F. Role of hydrogen peroxide in NF-kappaB activation: From inducer to modulator. Antioxid. Redox Signal. 2009, 11, 2223–2243. [Google Scholar] [CrossRef]
- Morgan, M.J.; Liu, Z.G. Crosstalk of reactive oxygen species and NF-κB signaling. Cell Res. 2011, 21, 103–115. [Google Scholar] [CrossRef]
- Yee, Y.H.; Chong, S.J.F.; Kong, L.R.; Goh, B.C.; Pervaiz, S. Sustained IKKβ phosphorylation and NF-κB activation by superoxide-induced peroxynitrite-mediated nitrotyrosine modification of B56γ3 and PP2A inactivation. Redox Biol. 2021, 41, 101834. [Google Scholar] [CrossRef]
- Taniguchi, K.; Karin, M. NF-κB, inflammation, immunity and cancer: Coming of age. Nat. Rev. Immunol. 2018, 18, 309–324. [Google Scholar]
- Mirzaei, S.; Hushmandi, K.; Zabolian, A.; Saleki, H.; Torabi, S.M.R.; Ranjbar, A.; SeyedSaleh, S.; Sharifzadeh, S.O.; Khan, H.; Ashrafizadeh, M.; et al. Elucidating Role of Reactive Oxygen Species (ROS) in Cisplatin Chemotherapy: A Focus on Molecular Pathways and Possible Therapeutic Strategies. Molecules 2021, 26, 2382. [Google Scholar] [CrossRef]
- Chou, T.C. Theoretical basis, experimental design, and computerized simulation of synergism and antagonism in drug combination studies. Pharmacol. Rev. 2006, 58, 621–681. [Google Scholar] [CrossRef]
- Marullo, R.; Werner, E.; Degtyareva, N.; Moore, B.; Altavilla, G.; Ramalingam, S.S.; Doetsch, P.W. Cisplatin induces a mitochondrial-ROS response that contributes to cytotoxicity depending on mitochondrial redox status and bioenergetic functions. PloS ONE 2013, 8, e81162. [Google Scholar] [CrossRef]
- Kleih, M.; Böpple, K.; Dong, M.; Gaißler, A.; Heine, S.; Olayioye, M.A.; Aulitzky, W.E.; Essmann, F. Direct impact of cisplatin on mitochondria induces ROS production that dictates cell fate of ovarian cancer cells. Cell Death Dis. 2019, 10, 851. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.M.; Kim, H.K.; Shim, W.; Anwar, M.A.; Kwon, J.W.; Kwon, H.K.; Kim, H.J.; Jeong, H.; Kim, H.M.; Hwang, D.; et al. Mechanism of Cisplatin-Induced cytotoxicity is correlated to impaired metabolism due to mitochondrial ROS generation. PLoS ONE 2015, 10, e0135083. [Google Scholar] [CrossRef] [PubMed]
- Galadari, S.; Rahman, A.; Pallichankandy, S.; Thayyullathil, F. Reactive oxygen species and cancer paradox: To promote or to suppress? Free Radic. Biol. Med. 2017, 104, 144–164. [Google Scholar] [PubMed]
- Yordi, E.G.; Pérez, E.M.; Matos, M.J.; Villares, E.U. Antioxidant and pro-oxidant effects of polyphenolic compounds and structure-activity relationship evidence. Nutr. Well-Being Health 2012, 2, 23–48. [Google Scholar]
- Barrera, G.; Gentile, F.; Pizzimenti, S.; Canuto, R.A.; Daga, M.; Arcaro, A.; Cetrangolo, G.P.; Lepore, A.; Ferretti, C.; Dianzani, C.; et al. Mitochondrial dysfunction in cancer and neurodegenerative diseases: Spotlight on fatty acid oxidation and lipoperoxidation products. Antioxidants 2016, 5, 7. [Google Scholar] [CrossRef]
- Gentile, F.; Arcaro, A.; Pizzimenti, S.; Daga, M.; Cetrangolo, G.P.; Dianzani, C.; Lepore, A.; Graf, M.; Ames, P.R.J.; Barrera, G. DNA damage by lipid peroxidation products: Implications in cancer, inflammation and autoimmunity. AIMS Genet. 2017, 4, 103–137. [Google Scholar]
- Kreuzer, T.; Zarkovic, N.; Grube, R.; Schaur, R.J. Inhibition of HeLa cell proliferation by 4-hydroxynonenal is associated with enhanced expression of the c-fos oncogene. Cancer Biother. Radiopharmaceut. 1997, 12, 131–136. [Google Scholar] [CrossRef]
- Kreuzer, T.; Grube, R.; Wutte, A.; Zarkovic, N.; Schaur, R.J. 4-Hydroxynonenal Modifies the Effects of Serum Growth Factors on the Expression of the c-fos Proto-Oncogene and the Proliferation of HeLa Carcinoma Cells. Free Radic. Biol. Med. 1998, 25, 42–49. [Google Scholar] [CrossRef]
- Martinho, N.; Santos, T.C.B.; Florindo, H.F.; Silva, L.C. Cisplatin-membrane interactions and their influence on platinum complexes activity and toxicity. Front. Physiol. 2019, 9, 1898. [Google Scholar] [CrossRef]
- Reynoso, R.C.; Giner, T.V.; de Mejia, E.G. Safety of a filtrate of fermented garambullo fruit: Biotransformation and toxicity studies. Food Chem. Toxicol. 1999, 37, 825–830. [Google Scholar] [CrossRef]
- Sarkar, F.H.; Li, Y. Using chemopreventive agents to enhance the efficacy of cancer therapy. Cancer Res. 2006, 66, 3347–3355. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Ma, L.; Weng, D.; Yao, J.; Liu, X.; Jin, F. Gallic acid induces apoptosis and enhances the anticancer effects of cisplatin in human small cell lung cancer H446 cell line via the ROS-dependent mitochondrial apoptotic pathway. Oncol. Rep. 2016, 35, 3075–3083. [Google Scholar] [CrossRef] [PubMed]
- Tan, B.L.; Norhaizan, M.E. Curcumin combination chemotherapy: The implication and efficacy in cancer. Molecules 2019, 24, 2527. [Google Scholar] [CrossRef] [PubMed]
- Yi, J.L.; Shi, S.; Shen, Y.L.; Wang, L.; Chen, H.Y.; Zhu, J.; Ding, Y. Myricetin and methyl eugenol combination enhances the anticancer activity, cell cycle arrest and apoptosis induction of cis-platin against HeLa cervical cancer cell lines. Int. J. Clin. Exp. Pathol. 2015, 8, 1116–1127. [Google Scholar]
- Sun, C.Y.; Zhang, Q.Y.; Zheng, G.J.; Feng, B. Phytochemicals: Current strategy to sensitize cancer cells to cisplatin. Biomed. Pharmacother. 2019, 110, 518–527. [Google Scholar] [CrossRef]
- Corsi, A.K.; Wightman, B.; Chalfie, M. A Transparent Window into Biology: A Primer on Caenorhabditis elegans. Genetics 2015, 200, 387–407. [Google Scholar] [CrossRef]
- Zhang, S.; Li, F.; Zhou, T.; Wang, G.; Li, Z. Caenorhabditis elegans as a Useful Model for Studying Aging Mutations. Front. Endocrinol. 2020, 11, 554994. [Google Scholar] [CrossRef]
- McCormick, M.; Chen, K.; Ramaswamy, P.; Kenyon, C. New genes that extend Caenorhabditis elegans’ lifespan in response to reproductive signals. Aging Cell 2012, 11, 192–202. [Google Scholar] [CrossRef]
- Uno, M.; Nishida, E. Lifespan-regulating genes in C. elegans. NPJ Aging Mech. Dis. 2016, 2, 16010. [Google Scholar] [CrossRef]
- Halliwell, B. Reactive species and antioxidants. Redox biology is a fundamental theme of aerobic life. Plant Physiol. 2006, 141, 312–322. [Google Scholar] [CrossRef]
- Ahmad, T.; Suzuki, Y.J. Juglone in Oxidative Stress and Cell Signaling. Antioxidants 2019, 8, 91. [Google Scholar] [CrossRef] [PubMed]
- Chobot, V.; Hadacek, F. Milieu-dependent pro- and antioxidant activity of juglone may explain linear and nonlinear effects on seedling development. J. Chem. Ecol. 2009, 35, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Szyller, J.; Bil-Lula, I. Heat shock proteins in oxidative stress and ischemia/reperfusion injury and benefits from physical exercises: A review to the current knowledge. Oxid. Med. Cell. Longev. 2021, 2021, 6678457. [Google Scholar] [CrossRef] [PubMed]
- Vigh, L.; Nakamoto, H.; Landry, J.; Gomez-Munoz, A.; Harwood, J.L.; Horvath, I. Membrane regulation of the stress response from prokaryotic models to mammalian cells. Ann. N. Y. Acad. Sci. 2007, 1113, 40–51. [Google Scholar] [CrossRef] [PubMed]
- Balogh, G.; Péter, M.; Glatz, A.; Gombos, I.; Török, Z.; Horváth, I.; Harwood, J.L.; Vígh, L. Key role of lipids in heat stress management. FEBS Lett. 2013, 587, 1970–1980. [Google Scholar] [CrossRef] [PubMed]
- Murakami, K.; Haneda, M.; Iwata, S.; Yoshino, M. Effect of hydroxy substituent on the prooxidant action of naphthoquinone compounds. Toxicol. In Vitro 2010, 24, 905–909. [Google Scholar] [CrossRef] [PubMed]
- Dayalan Naidu, S.; Dinkova-Kostova, A.T. Regulation of the mammalian heat shock factor 1. FEBS J. 2017, 284, 1606–1627. [Google Scholar] [CrossRef]
- Gems, D.; Partridge, L. Genetics of longevity in model organisms: Debates and paradigm shifts. Annu. Rev. Physiol. 2013, 75, 621–644. [Google Scholar] [CrossRef]
- Tullet, J.M.A.; Green, J.W.; Au, C.; Benedetto, A.; Thompson, M.A.; Clark, E.; Gilliat, A.F.; Young, A.; Schmeisser, K.; Gems, D. The SKN-1/Nrf2 transcription factor can protect against oxidative stress and increase lifespan in C. elegans by distinct mechanisms. Aging Cell 2017, 16, 1191–1194. [Google Scholar] [CrossRef]
- Meng, J.; Lv, Z.; Qiao, X.; Li, X.; Li, Y.; Zhang, Y.; Chen, C. The decay of Redox-stress Response Capacity is a substantive characteristic of aging: Revising the redox theory of aging. Redox Biol. 2017, 11, 365–374. [Google Scholar] [CrossRef]
- Ewald, C.Y.; Hourihan, J.M.; Bland, M.S.; Obieglo, C.; Katic, I.; Moronetti Mazzeo, L.E.; Alcedo, J.; Blackwell, T.K.; Hynes, N.E. NADPH oxidase-mediated redox signaling promotes oxidative stress resistance and longevity through memo-1 in C. elegans. eLife 2017, 6, e19493. [Google Scholar] [CrossRef] [PubMed]
- Heidler, T.; Hartwig, K.; Daniel, H.; Wenzel, U. Caenorhabditis elegans lifespan extension caused by treatment with an orally active ROS-generator is dependent on DAF-16 and SIR-2.1. Biogerontology 2010, 11, 183–195. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Rezaizadehnajafi, L.; Wink, M. Influence of resveratrol on oxidative stress resistance and life span in Caenorhabditis elegans. J. Pharm. Pharmacol. 2013, 65, 682–688. [Google Scholar] [CrossRef] [PubMed]
- Ayuda-Durán, B.; González-Manzano, S.; Miranda-Vizuete, A.; Dueñas, M.; Santos-Buelga, C.; González-Paramás, A.M. Epicatechin modulates stress-resistance in C. elegans via insulin/IGF-1 signaling pathway. PLoS ONE 2019, 14, e0199483. [Google Scholar] [CrossRef] [PubMed]
- Galam, L.; Failla, A.; Soundararajan, R.; Lockey, R.F.; Kolliputi, N. 4-hydroxynonenal regulates mitochondrial function in human small airway epithelial cells. Oncotarget 2015, 6, 41508–41521. [Google Scholar] [CrossRef] [PubMed]
- Senchuk, M.M.; Dues, D.J.; Schaar, C.E.; Johnson, B.K.; Madaj, Z.B.; Bowman, M.J.; Winn, M.E.; Van Raamsdonk, J.M. Activation of DAF-16/FOXO by reactive oxygen species contributes to longevity in long-lived mitochondrial mutants in Caenorhabditis elegans. PLoS Genet. 2018, 14, e1007268. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Hekimi, S. A mitochondrial superoxide signal triggers increased longevity in Caenorhabditis elegans. PLoS Biol. 2010, 8, e1000556. [Google Scholar] [CrossRef]
- Tesoriere, L.; Allegra, M.; Gentile, C.; Livrea, M.A. Betacyanins as phenol antioxidants. Chemistry and mechanistic aspects of the lipoperoxyl radical-scavenging activity in solution and liposomes. Free Radic. Res. 2009, 43, 706–717. [Google Scholar] [CrossRef]
- Dues, D.J.; Schaar, C.E.; Johnson, B.K.; Bowman, M.J.; Winn, M.E.; Senchuk, M.M.; Van Raamsdonk, J.M. Uncoupling of oxidative stress resistance and lifespan in long-lived isp-1 mitochondrial mutants in Caenorhabditis elegans. Free Radic. Biol. Med. 2017, 108, 362–373. [Google Scholar] [CrossRef]
- Zečić, A.; Braeckman, B.P. DAF-16/FoxO in Caenorhabditis elegans and Its Role in Metabolic Remodeling. Cells 2020, 9, 109. [Google Scholar] [CrossRef]
- Sun, X.; Chen, W.D.; Wang, Y.D. DAF-16/FOXO transcription factor in aging and longevity. Front. Pharmacol. 2017, 8, 548. [Google Scholar] [CrossRef]
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2008, 454, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Furman, D.; Campisi, J.; Verdin, E.; Carrera-Bastos, P.; Targ, S.; Franceschi, C.; Ferrucci, L.; Gilroy, D.W.; Fasano, A.; Miller, G.W.; et al. Chronic inflammation in the etiology of disease across the life span. Nat. Med. 2019, 25, 1822–1832. [Google Scholar] [CrossRef] [PubMed]
- Di Rosa, M.; Willoughby, D.A. Screens for anti-inflammatory drugs. J. Pharm. Pharmacol. 1971, 23, 297–298. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-κB signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef] [PubMed]
- Tak, P.P.; Firestein, G.S. NF-kappaB: A key role in inflammatory diseases. J. Clin. Investig. 2001, 107, 7–11. [Google Scholar] [CrossRef]
- Mantovani, A.; Allavena, P.; Sica, A.; Balkwill, F. Cancer-related inflammation. Nature 2008, 454, 436–444. [Google Scholar] [CrossRef]
- Bald, T.; Quast, T.; Landsberg, J.; Rogava, M.; Glodde, N.; Lopez-Ramos, D.; Kohlmeyer, J.; Riesenberg, S.; van den Boorn-Konijnenberg, D.; Hömig-Hölzel, C.; et al. Ultraviolet-radiation-induced inflammation promotes angiotropism and metastasis in melanoma. Nature 2014, 507, 109–113. [Google Scholar] [CrossRef]
- Meyer, C.; Sevko, A.; Ramacher, M.; Bazhin, A.V.; Falk, C.S.; Osen, W.; Borrello, I.; Kato, M.; Schadendorf, D.; Baniyash, M.; et al. Chronic inflammation promotes myeloid-derived suppressor cell activation blocking antitumor immunity in transgenic mouse melanoma model. Proc. Natl. Acad. Sci. USA 2011, 108, 17111–17116. [Google Scholar] [CrossRef]
- Reinhardt, J.; Landsberg, J.; Schmid-Burgk, J.L.; Ramis, B.B.; Bald, T.; Glodde, N.; Lopez-Ramos, D.; Young, A.; Ngiow, S.F.; Nettersheim, D.; et al. MAPK signaling and inflammation link melanoma phenotype switching to induction of CD73 during immunotherapy. Cancer Res. 2017, 77, 4697–4709. [Google Scholar] [CrossRef]
- Soudja, S.M.; Wehbe, M.; Mas, A.; Chasson, L.; de Tenbossche, C.P.; Huijbers, I.; Van den Eynde, B.; Schmitt-Verhulst, A.M. Tumor-initiated inflammation overrides protective adaptive immunity in an induced melanoma model in mice. Cancer Res. 2010, 70, 3515–3525. [Google Scholar] [CrossRef] [PubMed]
- Landsberg, J.; Kohlmeyer, J.; Renn, M.; Bald, T.; Rogava, M.; Cron, M.; Fatho, M.; Lennerz, V.; Wolfel, T.; Holzel, M.; et al. Melanomas resist T-cell therapy through inflammation-induced reversible dedifferentiation. Nature 2012, 490, 412–416. [Google Scholar] [CrossRef] [PubMed]
- Pal, H.C.; Hunt, K.M.; Diamond, A.; Elmets, C.A.; Afaq, F. Phytochemicals for the Management of Melanoma. Mini-Rev. Med. Chem. 2016, 16, 953–979. [Google Scholar] [PubMed]
- Samson, S.L.; Garber, A.J. Metabolic syndrome. Endocrinol. Metab. Clin. N. Am. 2014, 43, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Bentham, J.; Di Cesare, M.; Bilano, B.; Bixbi, H.; Zhou, B.; Stevens, G.A.; Riley, L.M.; Taddei, C.; Hajifathalian, K.; Lu, Y.; et al. Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: A pooled analysis of 2416 population-based measurement studies in 128·9 million children, adolescents, and adults. Lancet 2017, 390, 2627–2642. [Google Scholar]
- Maury, E.; Brichard, S.M. Adipokine dysregulation, adipose tissue inflammation and metabolic syndrome. Mol. Cell. Endocrinol. 2010, 314, 1–16. [Google Scholar] [CrossRef]
- Kwon, H.; Pessin, J.E. Adipokines mediate inflammation and insulin resistance. Front. Endocrinol. 2013, 4, 71. [Google Scholar] [CrossRef]
- Boden, G.; Shulman, G.I. Free fatty acids in obesity and type 2 diabetes: Defining their role in the development of insulin resistance and beta-cell dysfunction. Eur. J. Clin. Investig. 2002, 32, 14–23. [Google Scholar] [CrossRef]
- Samuel, V.T.; Shulman, G.I. The pathogenesis of insulin resistance: Integrating signaling pathways and substrate flux. J. Clin. Investig. 2016, 126, 12–22. [Google Scholar] [CrossRef]
- Lionetti, L.; Mollica, M.P.; Lombardi, A.; Cavaliere, G.; Gifuni, G.; Barletta, A. From chronic overnutrition to insulin resistance: The role of fat-storing capacity and inflammation. Nutr. Metab. Cardiovasc. Dis. 2009, 19, 146–152. [Google Scholar] [CrossRef]
- Vekic, J.; Zeljkovic, A.; Stefanovic, A.; Jelic-Ivanovic, Z.; Spasojevic-Kalimanovska, V. Obesity and dyslipidemia. Metabolism 2019, 92, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, S.; Fujita, T.; Shimabukuro, M.; Iwaki, M.; Yamada, Y.; Nakajima, Y.; Nakayama, O.; Makishima, M.; Matsuda, M.; Shimomura, I. Increased oxidative stress in obesity and its impact on metabolic syndrome. J. Clin. Investig. 2004, 114, 1752–1761. [Google Scholar] [CrossRef] [PubMed]
- Kitade, H.; Chen, G.; Ni, Y.; Ota, T. Nonalcoholic Fatty Liver Disease and Insulin Resistance: New Insights and Potential New Treatments. Nutrients 2017, 9, 387. [Google Scholar] [CrossRef] [PubMed]
- Vatner, D.F.; Majumdar, S.K.; Kumashiro, N.; Petersen, M.C.; Rahimi, Y.; Gattu, A.K.; Bears, M.; Camporez, J.P.; Cline, G.W.; Jurczak, M.J.; et al. Insulin-independent regulation of hepatic triglyceride synthesis by fatty acids. Proc. Natl. Acad. Sci. USA 2015, 112, 1143–1148. [Google Scholar] [CrossRef] [PubMed]
- Terzo, S.; Caldara, G.F.; Ferrantelli, V.; Puleio, R.; Cassata, G.; Mulè, F.; Amato, A. Pistachio consumption prevents and improves lipid dysmetabolism by reducing the lipid metabolizing gene expression in diet-induced obese mice. Nutrients 2018, 10, 1857. [Google Scholar] [CrossRef]
- Toma, L.; Sanda, G.M.; Niculescu, L.S.; Deleanu, M.; Sima, A.V.; Stancu, C.S. Phenolic compounds exerting lipid-regulatory, anti-inflammatory and epigenetic effects as complementary treatments in cardiovascular diseases. Biomolecules 2020, 10, 641. [Google Scholar] [CrossRef]
- Islam, S.; Ahmed, M.; Ahsan, H.; Lee, Y.-S. Recent molecular mechanisms and beneficial effects of phytochemicals and plant-based whole foods in reducing LDL-C and preventing cardiovascular disease. Antioxidants 2021, 10, 784. [Google Scholar] [CrossRef]
- Cong, W.; Golden, E.; Pantaleo, N.; White, C.M.; Maudsley, S.; Martin, B. Ghrelin receptor signaling: A promising therapeutic target for metabolic syndrome and cognitive dysfunction. CNS Neurol. Disord. Drug. Targets 2010, 9, 557–563. [Google Scholar] [CrossRef]
- Milbury, P.E.; Kalt, W. Xenobiotic metabolism and berry flavonoid transport across the blood-brain barrier. J. Agric. Food Chem. 2010, 58, 3950–3956. [Google Scholar] [CrossRef] [PubMed]
- Vingtdeux, V.; Giliberto, L.; Zhao, H.; Chandakkar, P.; Wu, Q.; Simon, J.E.; Janle, E.M.; Lobo, J.; Ferruzzi, M.G.; Davies, P.; et al. AMP-activated protein kinase signaling activation by resveratrol modulates amyloid-beta peptide metabolism. J. Biol. Chem. 2010, 285, 9100–9113. [Google Scholar] [CrossRef]
- Pandareesh, M.D.; Mythri, R.B.; Srinivas-Bharath, M.M. Bioavailability of dietary polyphenols: Factors contributing to their clinical application in CNS diseases. Neurochem. Int. 2015, 89, 198–208. [Google Scholar] [CrossRef] [PubMed]
- Andres-Lacueva, C.; Shukitt-Hale, B.; Galli, R.L.; Jauregui, O.; Lamuela-Raventos, R.M.; Joseph, J.A. Anthocyanins in aged blueberry-fed rats are found centrally and may enhance memory. Nutr. Neurosci. 2005, 8, 111–120. [Google Scholar] [CrossRef] [PubMed]
| Radical | Activity [Ref.] |
|---|---|
| ABTS●+ a | 1.76 TE b [56]; 4.0 TE [48] |
| ROO● c | 5.0 TE [30]; 6.0 TE [48] |
| DPPH● d | 0.06 TE [30] |
| NO● e | 0.28 TE [30] |
| LOO● f | 3.6 × 105 M−1 s−1 [25] |
| ● Hb[FeIV=O] g | 660 nMs−1 [29] |
| MPO[FeIV=O]● h MPO[FeIV=O] i O2●− j | 1.1 × 106 M−1 s−1 [26] 2.9 × 105 M−1 s−1 [26] none [59] |
| MW a | Log P b | Log D c | PSA d (Å2) | NPSA e (Å2) | pKa COOH f | Ref. |
|---|---|---|---|---|---|---|
| 309 309.3 | 0.362 −1.12 | −7.25 | 126.92 126.94 | 161.11 | 5.0(2), 3.7(11); 2.6(13) | [60] [59] |
| Cells | Experimental Design | Effect | Key Molecular Mechanisms | Ref. |
|---|---|---|---|---|
| RBCs (human) | Exposure to cumene hydroperoxide Exposure to toxic oxysterols | Antioxidant Antioxidant Anti-eriptotic | Radical-scavenging Resistance to oxidative hemolysis Resistance to oxidative hemolysis ROS production, glutathione depletion, PGE2 release, and Ca2+ entry ROS production, glutathione depletion, PGE2 release, and Ca2+ entry | [28] [38] |
| β–Thalassemia RBCs (from patients) | Exposure to cumene hydroperoxide | Antioxidant | Perferryl-Hb reduction Resistance to oxidative hemolysis Resistance to oxidative hemolysis Vit E and GSH depletion Vit E and GSH depletion Lipid and hemoglobin oxidation Lipid and hemoglobin oxidation | [29] |
| HUVEC (human umbilical vein endothelial) | Exposure to oxidized human LDL Exposure to TNFα | Anti-inflammatory Anti-inflammatory |  ROS formation ROS formation NF-κB transcriptional activity NF-κB transcriptional activity expression of adhesion molecules (ICAM-1; VCAM-1; ELAM-1) expression of adhesion molecules (ICAM-1; VCAM-1; ELAM-1)Preserved activity of cholesterol efflux system ABC-A1  expression of ICAM-1 expression of ICAM-1 | [39] [40] |
| RAW 264.7 (murine macrophages) | Exposure to LPS | Anti-inflammatory Pro-oxidant |  NF-κB activation NF-κB activation lipid hydroperoxides lipid hydroperoxides HNE formation HNE formationModulation of prostaglandin biosynthetic pathway:  mPGES-1 expression mPGES-1 expression COX2 and MPGDS expression COX2 and MPGDS expression Synthesis of proresolvin cyclopentenone Synthesis of proresolvin cyclopentenone | [41] |
| Differentiated human Caco-2 (colorectal adenocarcinoma) | Exposure to IL-1β Exposure to a mixture of TNFα, IL-1 β, IFNγ | Anti-inflammatory Anti-inflammatory |  active NOX-1 assembly active NOX-1 assembly ROS generation ROS generation NF-κB activation NF-κB activation COX2 and NOS expression COX2 and NOS expression release of IL6, IL8, PGE2, NO release of IL6, IL8, PGE2, NO expression of inflammatory expression of inflammatoryIL6, IL8, COX2, NOS, NOX-1  expression of glutamate- expression of glutamate-cysteine ligase catalytic subunit (GCLC) and glutathione peroxidase-1 (GPX-1) | [32] [59] |
| THP-1 (human monocyte/macrophages) | exposure to 7-keto-cholesterol | Anti-apoptotic |  NOX-4 basal activity NOX-4 basal activity NOX-4 over-expression NOX-4 over-expression NF-κB activation NF-κB activation intracellular Ca2+ intracellular Ca2+ mitochondrial apoptotic pathway mitochondrial apoptotic pathway | [33] |
| OECS (olfactory ensheating nerve) | Exposure to amyloid beta | Anti-apoptotic and cell regenerating |  O2•− and ROS production O2•− and ROS production transglutaminase 2 expression transglutaminase 2 expression caspase 3 expression caspase 3 expression nestin and cyclin-D1 nestin and cyclin-D1 | [35] |
| Tissues | ||||
| Mouse ileum muscle | Recording of spontaneous or carbachol- or KCl-evoked mechanical activity in organ bath | Spasmolytic | Interference with pathways regulating intracellular Ca2+ release Phosphodiesterase activity Phosphodiesterase activity cAMP cAMPVitamin C potentiated the Indicaxanthin effect | [34] [45] |
 increase;
increase;  decrease with respect to relevant experiment in the absence of Indicaxanthin.
decrease with respect to relevant experiment in the absence of Indicaxanthin.| Cell Lines | Effect | Key Results | Ref. |
|---|---|---|---|
| Caco-2 (colon adeno-carcinoma) | Anti-proliferative Pro-apoptotic Epigenetic modification | IC50 115 μM Exposure to 115 μM did not affect ROS formation nor total thiols. Exposure to 10–50 μM resulted in: cell cycle arrest at G2/M phase  de-methylated p16INK4a gene promoter de-methylated p16INK4a gene promoter  expression and accumulation of the p16INK4a expression and accumulation of the p16INK4a onco-suppressor protein  hypo-phospho-RB hypo-phospho-RB | [42] |
| Caco-2 LOVO-1 HCT-116 DLD-1 (colon adeno-carcinoma) | Anti-proliferative (varied with different cell lines) Epigenetic modifications (different sensitivity to Indicaxanthin) | Exposure to 50–100 μM resulted in: global DNA methylation global DNA methylation de-methylated onco-suppressor p16INK4a, GATA4 de-methylated onco-suppressor p16INK4a, GATA4ESR1 gene promoters unaffected hypermethylation of SFRP1 and HPP1 gene promoters  expression of DNA methylating enzymes expression of DNA methylating enzymes(DNMTs), enzymes transforming (TET2), and enzymes removing (MBD4, GADD45A) methyl groups  total DNMT activity. total DNMT activity.In silico computational modeling showed stable binding of Indicaxanthin at the DNMT1 catalytic site. | [46] |
| HT-29 (colon adeno-carcinoma) | No anti-proliferative effect | [46] | |
| A375 (melanoma) | Anti-proliferative Pro-apoptotic | GI50 100 μM Exposure to 100 μM resulted in:  cell growth by 56% after 72 h incubation cell growth by 56% after 72 h incubation PS externalization PS externalization IKB-α degradation IKB-α degradation nuclear p65 level nuclear p65 level expression of anti-apoptotic Bcl-2 and c-FLIP expression of anti-apoptotic Bcl-2 and c-FLIP cell invasiveness by 71% cell invasiveness by 71% | [44] |
| HeLa (cervical cancer) | Anti-proliferative Pro-apoptotic Synergistic cytotoxic activity in combination with cisplatin (CDDP) Potentiating effect on CDDP toxicity | IC50 149 μM Exposure to 60 μM resulted in:  MMP and MMP and  PS externalization PS externalizationarrest of cell cycle at G0/G1 and reduction at G2/M phase unaffected expression of apoptosis- or cell cycle- related proteins unaffected ROS formation or GSH content Indicaxanthin/CDDP (60/10 μM) combination resulted in:  ROS production ROS production cell cycle arrest (increase of the cells in G0/G1, absence of cells in G2/M, increase of cells in sub G0/G1 phase)  MMP MMP expression of Bax, cyt C, p53, p21waf1 and expression of Bax, cyt C, p53, p21waf1 anddown-regulation of Bcl-2, apoptosis-related proteins NAC decreased the synergistic effects by 60% Non-toxic Indicaxanthin (2–10 μM) potentiated the CDDP toxicity up to three-fold | [43] |
 increase;
increase;  decrease with respect to relevant experiment in the absence of Indicaxanthin.
decrease with respect to relevant experiment in the absence of Indicaxanthin.| Strain | Effect | Data Evidence | Ref. |
|---|---|---|---|
| TJ375 (hsp-16.2:GFP) submitted to pro-oxidant juglone | Antioxidant | Incubation (10–100 μM) for 48 h resulted in: juglone-induced oxidative stress- juglone-induced oxidative stress-dependent expression of fluorescent GFP | [47] |
| Wild type N2 | Extension of life-span | Incubation (25 μM) resulted in: maximum life-span by 3.9 days maximum life-span by 3.9 days  mean survival by 34.34%. mean survival by 34.34%. Indicaxanthin did not accumulate in the worm | [47] |
| Wild type N2 | Extension of life-span Gene activation | Incubation (25 μM) resulted in: life-span by 16.82%. life-span by 16.82%.Incubation (100 μM) resulted in:  hsp-12.1 and lips-17 gene expression hsp-12.1 and lips-17 gene expression | [48] |
| TJ356 (daf16p:daf-16a/b:GFP + rol-6(su1006) | Activation of DAF-16/FOXO transcription factor | Incubation (25 μM) resulted in: nuclear translocation of DAF-16 nuclear translocation of DAF-16 | [48] |
| CF1038 (daf16(mu86) |  extension of life-span extension of life-span | [48] | |
| LD1 (ldIs7 [skn-1b/c:GFP + rol-6(su1006)] | Unaffected SKN/Nrf2 transcription factor No extension of life-span | [48] |
 increase;
increase;  decrease with respect to relevant experiment in the absence of Indicaxanthin.
decrease with respect to relevant experiment in the absence of Indicaxanthin.| Experimental Set-up | Effect | Data Evidence | Ref. | |
|---|---|---|---|---|
| Rat | λ-Carrageenin-induced pleurisy | Anti-inflammatory | Pre-treatment (0.5–2.0 μmol/kg) orally, every 8 h up to 40 h, resulted in: exudate volume exudate volume number of leukocytes in pleural cavity number of leukocytes in pleural cavity release of PGE2, NO, IL-1β, TNF-α in the release of PGE2, NO, IL-1β, TNF-α in the exudate  IL-1β, TNF-α, iNOS, COX-2 mRNA and i-NOS IL-1β, TNF-α, iNOS, COX-2 mRNA and i-NOSand COX2 protein expression in leukocytes  activation of NF-κΒ in leukocytes activation of NF-κΒ in leukocytesPeak plasma concentration of Indicaxanthin 0.22 μmol/L, half-life 1.5 h | [49] |
| Mouse | Cutaneous melanoma | Antiproliferative Impairment of tumor progression | Treatment (3.2 mg/kg) orally, every 8 h for 14 days, resulted in: tumor volume (86%) and weight (83%) tumor volume (86%) and weight (83%) CXCL1 chemochine plasma level CXCL1 chemochine plasma level | [44] |
| Mouse | High-fat diet (HFD)-induced obesity (metabolic syndrome model) | Anti-obesity Counteracting of insulin resistance Anti-inflammatory | Treatment (1.3 μmol/kg) orally, every 12 h for 4 weeks, resulted in: food intake, body weight, visceral fat, food intake, body weight, visceral fat, subcutaneous adipose tissue mass  fasting glycemia and insulinemia fasting glycemia and insulinemia IPGTT and ITT tolerance test IPGTT and ITT tolerance testrestored HOMA index unaffected circulating lipid profile and liver steatosis  HFD-induced oxidative stress HFD-induced oxidative stressrestored RONS, MDA, NO in liver and adipose tissue restored TNF-α, CCL-2, F4-80 gene expression and p65, p-JNK, COX-2, i-NOS protein levels in liver and adipose tissue | [50] |
| Rat | Pharmacokinetics in brain and neuronal excitability microiontophoretic model | Crossing of BBB Modulatory of neuronal excitability | Treatment (2.0 μmol/kg) orally, single administration, resulted in: accumulation in brain (peak: 20 ng/g fresh tissue at 2.5 h) and disappearance at 4 h with first order kinetics Application (0.085 ng and 0.34 ng per hippocampal neuron) resulted in:  bioelectric activity of hippocampal neurons bioelectric activity of hippocampal neuronsthrough glutamatergic synapses | [36] |
| Rat | Brain distribution and neuronal excitability microiontophoretic model | Neuromodulatory | Treatment (2.0 μmol/kg) orally, single administration, resulted in: selected access to specific brain areas (cerebellum, hippocampus, cortex, diencephalon, brainstem), absence in striatum/ pallidum Application (0.085, 0.17, and 0.34 ng per neuron) resulted in: different effects on neuronal firing rate in different brain areas | [37] |
 increase;
increase;  decrease with respect to relevant experiment in the absence of Indicaxanthin
decrease with respect to relevant experiment in the absence of IndicaxanthinPublisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Attanzio, A.; Restivo, I.; Tutone, M.; Tesoriere, L.; Allegra, M.; Livrea, M.A. Redox Properties, Bioactivity and Health Effects of Indicaxanthin, a Bioavailable Phytochemical from Opuntia ficus indica, L.: A Critical Review of Accumulated Evidence and Perspectives. Antioxidants 2022, 11, 2364. https://doi.org/10.3390/antiox11122364
Attanzio A, Restivo I, Tutone M, Tesoriere L, Allegra M, Livrea MA. Redox Properties, Bioactivity and Health Effects of Indicaxanthin, a Bioavailable Phytochemical from Opuntia ficus indica, L.: A Critical Review of Accumulated Evidence and Perspectives. Antioxidants. 2022; 11(12):2364. https://doi.org/10.3390/antiox11122364
Chicago/Turabian StyleAttanzio, Alessandro, Ignazio Restivo, Marco Tutone, Luisa Tesoriere, Mario Allegra, and Maria A. Livrea. 2022. "Redox Properties, Bioactivity and Health Effects of Indicaxanthin, a Bioavailable Phytochemical from Opuntia ficus indica, L.: A Critical Review of Accumulated Evidence and Perspectives" Antioxidants 11, no. 12: 2364. https://doi.org/10.3390/antiox11122364
APA StyleAttanzio, A., Restivo, I., Tutone, M., Tesoriere, L., Allegra, M., & Livrea, M. A. (2022). Redox Properties, Bioactivity and Health Effects of Indicaxanthin, a Bioavailable Phytochemical from Opuntia ficus indica, L.: A Critical Review of Accumulated Evidence and Perspectives. Antioxidants, 11(12), 2364. https://doi.org/10.3390/antiox11122364











