The Wine Industry By-Products: Applications for Food Industry and Health Benefits
Abstract
1. Introduction
2. Food Industry Applications
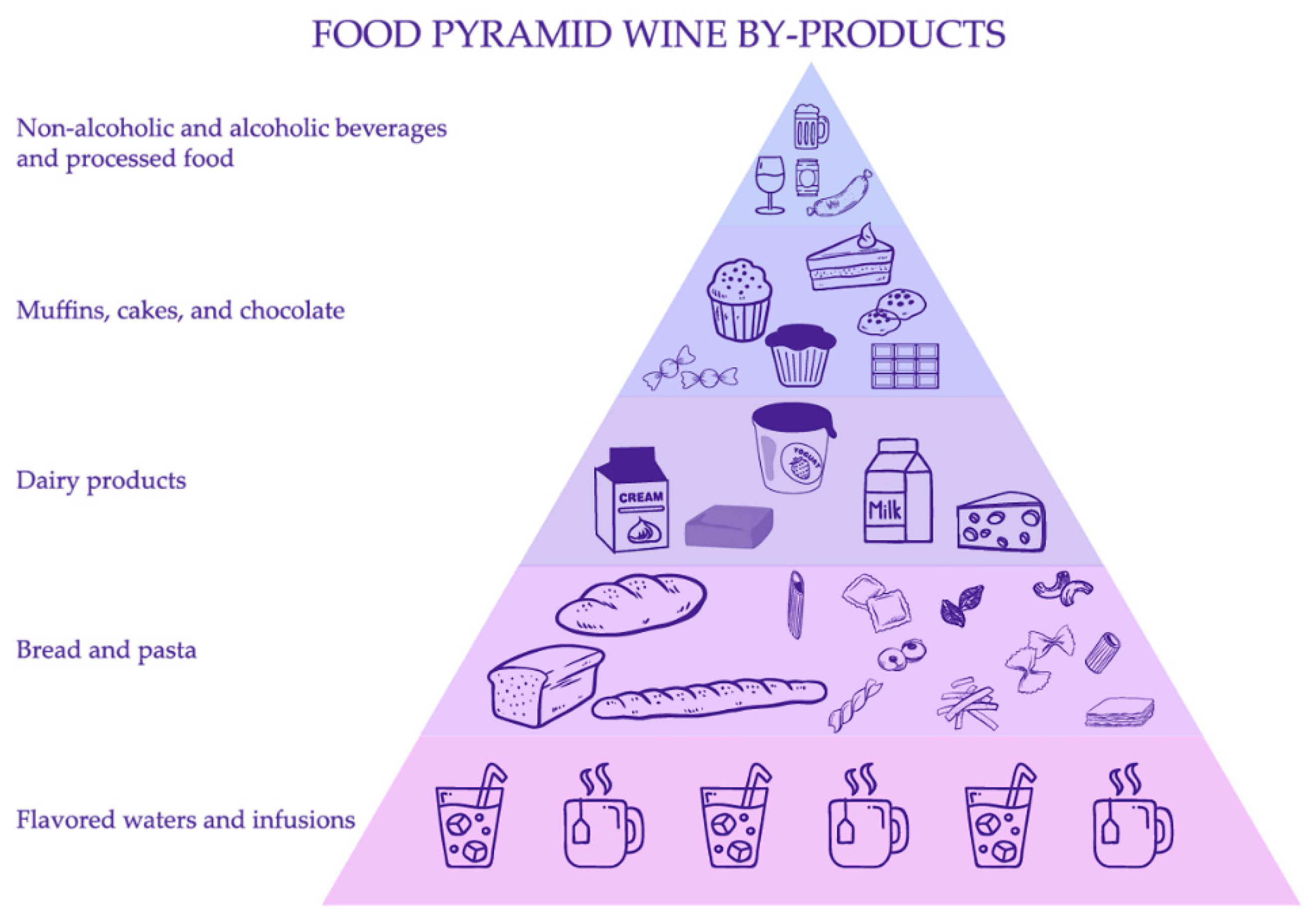
2.1. Food Pyramid of Wine-By Products
2.2. Flavored Waters and Infusions
2.3. Bread and Pasta
2.4. Dairy Products
2.5. Muffins, Cakes and Chocolate
2.6. Non-Alcoholic and Alcoholic Beverages and Processed Food
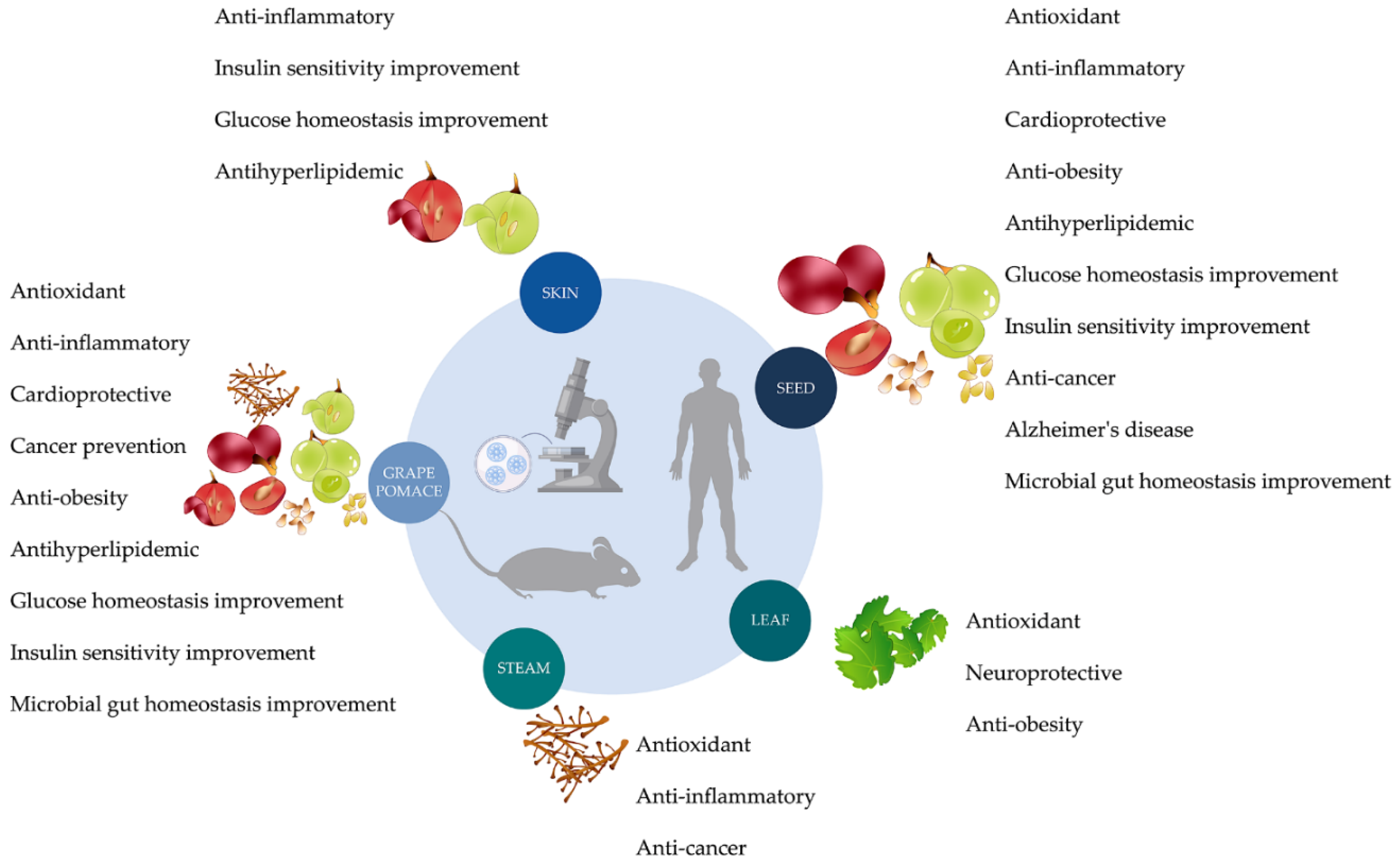
3. Health Benefits
3.1. Grape Pomace
3.2. Skins
3.3. Seeds
3.4. Leaves
3.5. Stems
3.6. Grapevine Shoots
| By-Product | By-Product Health Benefits | Reference |
|---|---|---|
| Grape pomace | Microbial gut homeostasis improvement (non-beneficial bacteria inhibition and stimulation of probiotic bacteria growth) | In vivo: [89,90,91,92,93,94] |
| Anti-inflammatory | In vitro: [96] In vivo: [91,97,101,102,103,104] | |
| Cardioprotective | In vivo: [98,99,100,101] | |
| Skins | Antioxidant | In vitro: [107,109] |
| Anti-inflammatory | In vitro: [107,109] | |
| Seeds | Microbial gut homeostasis improvement (non-beneficial bacteria inhibition and stimulation of probiotic bacteria growth) | In vivo: [110] |
| Antioxidant | In vivo: [111,114,121,125,126,128,129,130] In vitro: [127] | |
| Anti-inflammatory | In vivo: [111,112,113,124,125,126,128,129,130] Clinical trial: [117,122,123] | |
| Anti-cancer | In vivo: [111,112] In vitro: [116] | |
| Leaves | Neuroprotective | In vivo: [132] |
| Antioxidant | In vivo: [132] | |
| Stems | Brain dysfunctions | In vivo: [134,136] |
| Anti-inflammatory | In vivo: [136] | |
| Antioxidant | In vitro: [137] | |
| Anti-cancer | In vitro: [137] | |
| Grapevine shoots | Anti-cancer | In vivo: [138] In vitro: [134] |
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- OIV. Available online: https://www.oiv.int/en/statistiques/ (accessed on 13 July 2021).
- Aliaño-González, M.J.; Gabaston, J.; Ortiz-Somovilla, V.; Cantos-Villar, E. Wood waste from fruit trees: Biomolecules and their applications in agri-food industry. Biomolecules 2022, 12, 238. [Google Scholar] [CrossRef]
- Câmara, J.S.; Lourenço, S.; Silva, C.; Lopes, A.; Andrade, C.; Perestrelo, R. Exploring the potential of wine industry by-products as source of additives to improve the quality of aquafeed. Microchem. J. 2020, 155, 104758. [Google Scholar] [CrossRef]
- Galanakis, C.M. Handbook of Grape Processing By-Products: Sustainable Solutions; Academic Press: New York, NY, USA, 2017. [Google Scholar]
- Pedroza, M.A.; Salinas, M.R.; Alonso, G.L.; Zalacain, A. Oenological applications of winemaking by-products. In Handbook of Grape Processing By-Products: Sustainable Solutions; Galanakis, C., Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2017; Chapter 9; pp. 215–232. [Google Scholar]
- Shrikhande, A.J. Wine by-products with health benefits. Food Res. Int. 2000, 33, 469–474. [Google Scholar] [CrossRef]
- Ferreira, S.M.; Santos, L. A Potential valorization strategy of wine industry by-products and their application in cosmetics—Case study: Grape pomace and grapeseed. Molecules 2022, 27, 969. [Google Scholar] [CrossRef] [PubMed]
- Hassan, Y.I.; Kosir, V.; Yin, X.; Ross, K.; Diarra, M.S. Grape pomace as a promising antimicrobial alternative in feed: A critical review. J. Agric. Food Chem. 2019, 67, 9705–9718. [Google Scholar] [CrossRef] [PubMed]
- Tapia-Quirós, P.; Montenegro-Landívar, M.F.; Reig, M.; Vecino, X.; Cortina, J.L.; Saurina, J.; Granados, M. Recovery of polyphenols from agri-food by-products: The olive oil and winery industries cases. Foods 2022, 11, 362. [Google Scholar] [CrossRef]
- Beres, C.; Costa, G.N.S.; Cabezudo, I.; da Silva-James, N.K.; Teles, A.S.C.; Cruz, A.P.G.; Mellinger-Silva, C.; Tonon, R.V.; Cabral, L.M.C.; Freitas, S.P. Towards integral utilization of grape pomace from winemaking process: A review. Waste Manag. 2017, 68, 581–594. [Google Scholar] [CrossRef] [PubMed]
- Chowdhary, P.; Gupta, A.; Gnansounou, E.; Pandey, A.; Chaturvedi, P. Current trends and possibilities for exploitation of Grape pomace as a potential source for value addition. Environ. Pollut. 2021, 278, 116796. [Google Scholar] [CrossRef]
- Pérez-Bibbins, B.; Torrado-Agrasar, A.; Salgado, J.M.; Oliveira, R.P.D.S.; Domínguez, J.M. Potential of lees from wine, beer and cider manufacturing as a source of economic nutrients: An overview. Waste Manag. 2015, 40, 72–81. [Google Scholar] [CrossRef]
- Jara-Palacios, M.J. Wine lees as a source of antioxidant compounds. Antioxidants 2019, 8, 45. [Google Scholar] [CrossRef]
- Tilman, D.; Clark, M. Global diets link environmental sustainability and human health. Nature 2014, 515, 518–522. [Google Scholar] [CrossRef] [PubMed]
- da Rocha, B.R.S.; Rico-Campà, A.; Romanos-Nanclares, A.; Ciriza, E.; Barbosa, K.B.F.; Martínez-González, M.Á.; Martín-Calvo, N. Adherence to Mediterranean diet is inversely associated with the consumption of ultra-processed foods among Spanish children: The SENDO project. Public Health Nutr. 2021, 24, 3294–3303. [Google Scholar] [CrossRef] [PubMed]
- Tosti, V.; Bertozzi, B.; Fontana, L. Health benefits of the mediterranean diet: Metabolic and molecular mechanisms. J. Gerontol. A Biol. Sci. Med. Sci. 2018, 73, 318–326. [Google Scholar] [CrossRef] [PubMed]
- Antonić, B.; Jančíková, S.; Dordević, D.; Tremlová, B. Grape pomace valorization: A systematic review and meta-analysis. Foods 2020, 9, 1627. [Google Scholar] [CrossRef] [PubMed]
- Pereira, P.; Palma, C.; Ferreira-Pêgo, C.; Amaral, O.; Amaral, A.; Rijo, P.; Gregório, J.; Palma, L.; Nicolai, M. Grape pomace: A potential ingredient for the human diet. Foods 2020, 9, 1772. [Google Scholar] [CrossRef] [PubMed]
- Market, F.W. Global Flavored Water Market By Product Type (Flavored Water And Functional Water) and by Regional Analysis (North America, Europe, Asia Pacific, Latin America, and Middle East & Africa)—Global Industry Analysis, Size, Share, Growth, Trends, and Forecast (2022–2027). Available online: https://www.marketdataforecast.com/market-reports/flavored-water-market (accessed on 13 July 2022).
- Alibade, A.; Lakka, A.; Bozinou, E.; Lalas, S.I.; Chatzilazarou, A.; Makris, D.P. Development of a green methodology for simultaneous extraction of polyphenols and pigments from red winemaking solid wastes (Pomace) using a novel glycerol-sodium benzoate deep eutectic solvent and ultrasonication pretreatment. Environments 2021, 8, 90. [Google Scholar] [CrossRef]
- Rubio, F.T.V.; Haminiuk, C.W.I.; Dos Santos, M.M.; Thomazini, M.; Moraes, I.C.F.; Martelli-Tosi, M.; Fávaro-Trindade, C.S. Development of natural pigments microencapsulated in waste yeast: Saccharomyces cerevisiae using spray drying technology and their application in yogurt. Food Funct. 2021, 12, 8946–8959. [Google Scholar] [CrossRef] [PubMed]
- Pazir, F.; Koçak, E.; Turan, F.; Ova, G. Extraction of anthocyanins from grape pomace by using supercritical carbon dioxide. J. Food Process. Preserv. 2021, 45, e14950. [Google Scholar] [CrossRef]
- Lima, Á.S.; Oliveira, B.S.D.; Shabudin, S.V.; Almeida, M.; Freire, M.G.; Bica, K. Purification of anthocyanins from grape pomace by centrifugal partition chromatography. J. Mol. Liq. 2021, 326, 115324. [Google Scholar] [CrossRef]
- Kammerer, D.; Claus, A.; Carle, R.; Schieber, A. Polyphenol screening of pomace from red and white grape varieties (Vitis vinifera L.) by HPLC-DAD-MS/MS. J. Agric. Food Chem. 2004, 52, 4360–4367. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.; Pai, A.; Liu, D.; Luo, J.; Wu, J.; Fang, Z.; Zhang, P. Optimizing extraction method of aroma compounds from grape pomace. J. Food Sci. 2020, 85, 4225–4240. [Google Scholar] [CrossRef]
- Muñoz-González, C.; Rodríguez-Bencomo, J.J.; Martín-Álvarez, P.J.; Moreno-Arribas, M.V.; Pozo-Bayón, M.Á. Recovery of aromatic aglycones from grape pomace winemaking by-products by using liquid-liquid and pressurized-liquid extraction. Food Anal. Methods 2014, 7, 47–57. [Google Scholar] [CrossRef]
- Vasserot, Y.; Arnaud, A.; Galzy, P. Evidence for muscat marc monoterpenol glucosides hydrolysis by free or immobilized yeast β-glucosidase. Bioresour. Technol. 1993, 43, 269–271. [Google Scholar] [CrossRef]
- Ruberto, G.; Renda, A.; Amico, V.; Tringali, C. Volatile components of grape pomaces from different cultivars of Sicilian Vitis vinifera L. Bioresour. Technol. 2008, 99, 260–268. [Google Scholar] [CrossRef]
- Lindsay, M.A.; Granucci, N.; Greenwood, D.R.; Villas-Boas, S.G. Identification of new natural sources of flavour and aroma metabolites from solid-state fermentation of agro-industrial by-products. Metabolites 2022, 12, 157. [Google Scholar] [CrossRef]
- Costa, J.R.; Monteiro, M.J.; Tonon, R.V.; Cabral, L.M.C.; Pastrana, L.; Pintado, M.E. Fortification of coconut water with microencapsulated grape pomace extract towards a novel electrolyte beverage: Biological, sensorial and quality aspects. Future Foods 2021, 4, 100079. [Google Scholar] [CrossRef]
- Ali, S.M.; Imran, A.; Arshad, M.U.; Ahmed, R.S.; Imran, M. Physicochemical, antioxidant and enzymes activities of grape fruit peel and pomace enriched functional drinks. Cell Mol. Biol. 2021, 67, 125–131. [Google Scholar] [CrossRef]
- Goulas, V.; Stavrou, K.; Michael, C.; Botsaris, G.; Barbouti, A. The potential of sun-dried grape pomace as a multi-functional ingredient for herbal infusion: Effects of brewing parameters on composition and bioactivity. Antioxidants 2021, 10, 586. [Google Scholar] [CrossRef]
- Parisi, V.; Vassallo, A.; Pisano, C.; Signorino, G.; Cardile, F.; Sorrentino, M.; Colelli, F.; Fucci, A.; D’Andrea, E.L.; De Tommasi, N.; et al. A herbal mixture from propolis, pomegranate, and grape pomace endowed with anti-inflammatory activity in an in vivo rheumatoid arthritis model. Molecules 2020, 25, 2255. [Google Scholar] [CrossRef]
- Gerardi, C.; D’Amico, L.; Migoni, D.; Santino, A.; Salomone, A.; Carluccio, M.A.; Giovinazzo, G. Strategies for reuse of skins separated from grape pomace as ingredient of functional beverages. Front. Bioeng. Biotechnol. 2020, 8, 645. [Google Scholar] [CrossRef]
- Tolve, R.; Simonato, B.; Rainero, G.; Bianchi, F.; Rizzi, C.; Cervini, M.; Giuberti, G. Wheat bread fortification by grape pomace powder: Nutritional, technological, antioxidant, and sensory properties. Foods 2021, 10, 75. [Google Scholar] [CrossRef] [PubMed]
- Carpentieri, S.; Larrea-Wachtendorff, D.; Donsì, F.; Ferrari, G. Functionalization of pasta through the incorporation of bioactive compounds from agri-food by-products: Fundamentals, opportunities, and drawbacks. Trends Food Sci. Technol. 2022, 122, 49–65. [Google Scholar] [CrossRef]
- Monteiro, G.C.; Minatel, I.O.; Junior, A.P.; Gomez-Gomez, H.A.; de Camargo, J.P.C.; Diamante, M.S.; Pereira Basílio, L.S.; Tecchio, M.A.; Pereira Lima, G.P. Bioactive compounds and antioxidant capacity of grape pomace flours. LWT 2021, 135, 110053. [Google Scholar] [CrossRef]
- Rosales Soto, M.U.; Brown, K.; Ross, C.F. Antioxidant activity and consumer acceptance of grape seed flour-containing food products. Int. J. Food Sci. 2012, 47, 592–602. [Google Scholar] [CrossRef]
- Lou, W.; Li, B.; Nataliya, G. The influence of Cabernet Sauvignon wine grape pomace powder addition on the rheological and microstructural properties of wheat dough. CYTA—J. Food 2021, 19, 751–761. [Google Scholar] [CrossRef]
- Balli, D.; Cecchi, L.; Innocenti, M.; Bellumori, M.; Mulinacci, N. Food by-products valorisation: Grape pomace and olive pomace (pâté) as sources of phenolic compounds and fiber for enrichment of tagliatelle pasta. Food Chem. 2021, 355, 129642. [Google Scholar] [CrossRef]
- Sant’Anna, V.; Christiano, F.D.P.; Marczak, L.D.F.; Tessaro, I.C.; Thys, R.C.S. The effect of the incorporation of grape marc powder in fettuccini pasta properties. LWT 2014, 58, 497–501. [Google Scholar] [CrossRef]
- Marinelli, V.; Padalino, L.; Nardiello, D.; Del Nobile, M.A.; Conte, A. New approach to enrich pasta with polyphenols from grape marc. J. Chem. 2015, 2015, 734578. [Google Scholar] [CrossRef]
- Chouchouli, V.; Kalogeropoulos, N.; Konteles, S.J.; Karvela, E.; Makris, D.P.; Karathanos, V.T. Fortification of yoghurts with grape (Vitis vinifera) seed extracts. LWT 2013, 53, 522–529. [Google Scholar] [CrossRef]
- Pavlou, A.; Melikidou, I.; Petridis, D.; Panayiotou, C.; Ritzoulis, C. Winery by-product hydrocolloids as texture modifiers in yogurt formulations. J. Culin. Sci. Technol. 2021, 19, 352–371. [Google Scholar] [CrossRef]
- Tseng, A.; Zhao, Y. Wine grape pomace as antioxidant dietary fibre for enhancing nutritional value and improving storability of yogurt and salad dressing. Food Chem. 2013, 138, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Kandylis, P.; Dimitrellou, D.; Moschakis, T. Recent applications of grapes and their derivatives in dairy products. Trends Food Sci. Technol. 2021, 114, 696–711. [Google Scholar] [CrossRef]
- Gaglio, R.; Barbaccia, P.; Barbera, M.; Restivo, I.; Attanzio, A.; Maniaci, G.; Di Grigoli, A.; Francesca, N.; Tesoriere, L.; Bonanno, A.; et al. The use of winery by-products to enhance the functional aspects of the fresh ovine “primosale” cheese. Foods 2021, 10, 461. [Google Scholar] [CrossRef] [PubMed]
- Barbaccia, P.; Busetta, G.; Barbera, M.; Alfonzo, A.; Garofalo, G.; Francesca, N.; Moscarelli, A.; Moschetti, G.; Settanni, L.; Gaglio, R. Effect of grape pomace from red cultivar ‘Nero d’Avola’ on the microbiological, physicochemical, phenolic profile and sensory aspects of ovine Vastedda-like stretched cheese. J. Appl. Microbiol. 2022, 133, 130–144. [Google Scholar] [CrossRef] [PubMed]
- Zeppa, G.; Tedesco, M.; Bertolino, M.; Çilek Tatar, B. Grape pomace as a new coagulant for tofu production: Physicochemical and sensory effects. Foods 2021, 10, 1857. [Google Scholar] [CrossRef] [PubMed]
- dos Santos, K.M.O.; de Oliveira, I.C.; Lopes, M.A.C.; Cruz, A.P.G.; Buriti, F.C.A.; Cabral, L.M. Addition of grape pomace extract to probiotic fermented goat milk: The effect on phenolic content, probiotic viability and sensory acceptability. J. Sci. Food Agric. 2017, 97, 1108–1115. [Google Scholar] [CrossRef] [PubMed]
- Karnopp, A.R.; Oliveira, K.G.; de Andrade, E.F.; Postingher, B.M.; Granato, D. Optimization of an organic yogurt based on sensorial, nutritional, and functional perspectives. Food Chem. 2017, 233, 401–411. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.Y.; Shyu, Y.S.; Hsu, C.K. Grape wine lees improves the rheological and adds antioxidant properties to ice cream. LWT 2009, 42, 312–318. [Google Scholar] [CrossRef]
- Aksoylu, Z.; Çağindi, O.; Köse, E. Effects of blueberry, grape seed powder and poppy seed incorporation on physicochemical and sensory properties of biscuit. J. Food Qual. 2015, 38, 164–174. [Google Scholar] [CrossRef]
- Mildner-Szkudlarz, S.; Bajerska, J.; Zawirska-Wojtasiak, R.; Górecka, D. White grape pomace as a source of dietary fibre and polyphenols and its effect on physical and nutraceutical characteristics of wheat biscuits. J. Sci. Food Agric. 2013, 93, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Karnopp, A.R.; Figueroa, A.M.; Los, P.R.; Teles, J.C.; Simões, D.R.S.; Barana, A.C.; Kubiaki, F.T.; Oliveira, J.G.B.d.; Granato, D. Effects of whole-wheat flour and bordeaux grape pomace (Vitis labrusca L.) on the sensory, physicochemical and functional properties of cookies. Food Sci. Technol. 2015, 35, 750–756. [Google Scholar] [CrossRef]
- Portillo, M.C.; Franquès, J.; Araque, I.; Reguant, C.; Bordons, A. Bacterial diversity of Grenache and Carignan grape surface from different vineyards at Priorat wine region (Catalonia, Spain). Int. J. Food Microbiol. 2016, 219, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Nakov, G.; Brandolini, A.; Hidalgo, A.; Ivanova, N.; Stamatovska, V.; Dimov, I. Effect of grape pomace powder addition on chemical, nutritional and technological properties of cakes. LWT 2020, 134, 109950. [Google Scholar] [CrossRef]
- Baldán, Y.; Riveros, M.; Fabani, M.P.; Rodriguez, R. Grape pomace powder valorization: A novel ingredient to improve the nutritional quality of gluten-free muffins. Biomass Convers. Biorefin. 2021. [Google Scholar] [CrossRef]
- Spigno, G.; Donsì, F.; Amendola, D.; Sessa, M.; Ferrari, G.; De Faveri, D.M. Nanoencapsulation systems to improve solubility and antioxidant efficiency of a grape marc extract into hazelnut paste. J. Food Eng. 2013, 114, 207–214. [Google Scholar] [CrossRef]
- Bolenz, S.; Glöde, L. Technological and nutritional aspects of milk chocolate enriched with grape pomace products. Eur. Food Res. Technol. 2021, 247, 623–636. [Google Scholar] [CrossRef]
- Acan, B.G.; Kilicli, M.; Bursa, K.; Toker, O.S.; Palabiyik, I.; Gulcu, M.; Yaman, M.; Gunes, R.; Konar, N. Effect of grape pomace usage in chocolate spread formulation on textural, rheological and digestibility properties. LWT 2021, 138, 110451. [Google Scholar] [CrossRef]
- Jara-Palacios, M.; Hernanz, D.; Escudero-Gilete, M.; Heredia, F. The use of grape seed byproducts rich in flavonoids to improve the antioxidant potential of red wines. Molecules 2016, 21, 1526. [Google Scholar] [CrossRef]
- Gasiński, A.; Kawa-Rygielska, J.; Mikulski, D.; Kłosowski, G.; Głowacki, A. Application of white grape pomace in the brewing technology and its impact on the concentration of esters and alcohols, physicochemical parameteres and antioxidative properties of the beer. Food Chem. 2022, 367, 130646. [Google Scholar] [CrossRef]
- García-Lomillo, J.; González-Sanjosé, M.L.; Del Pino-García, R.; Rivero-Pérez, M.D.; Muñiz-Rodríguez, P. Antioxidant and antimicrobial properties of wine byproducts and their potential uses in the food industry. J. Agric. Food Chem. 2014, 62, 12595–12602. [Google Scholar] [CrossRef] [PubMed]
- Pazos, M.; Gallardo, J.M.; Torres, J.L.; Medina, I. Activity of grape polyphenols as inhibitors of the oxidation of fish lipids and frozen fish muscle. Food Chem. 2005, 92, 547–557. [Google Scholar] [CrossRef]
- Lavelli, V.; Kerr, W.L.; García-Lomillo, J.; González-San José, M.L. Applications of recovered bioactive compounds in food products. In Handbook of Grape Processing By-Products: Sustainable Solutions; Galanakis, C., Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2017; Chapter 10; pp. 233–266. [Google Scholar]
- Ryu, K.S.; Shim, K.S.; Shin, D. Effect of grape pomace powder addition on TBARS and color of cooked pork sausages during storage. Korean J. Food Sci. Anim. Resour. 2014, 34, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Tremlova, B.; Havlova, L.; Benes, P.; Zemancova, J.; Buchtova, H.; Tesikova, K.; Dordevic, S.; Dordevic, D. Vegetarian “Sausages” with the Addition of Grape Flour. Appl. Sci. 2022, 12, 2189. [Google Scholar] [CrossRef]
- Iuga, M.; Mironeasa, S. Potential of grape byproducts as functional ingredients in baked goods and pasta. Compr. Rev. Food Sci. Food Saf. 2020, 19, 2473–2505. [Google Scholar] [CrossRef]
- Ky, I.; Crozier, A.; Cros, G.; Teissedre, P.-l. Polyphenols composition of wine and grape sub-products and potential effects on chronic diseases. Nutr. Aging 2014, 2, 165–177. [Google Scholar] [CrossRef]
- Mazza, G.; Francis, F.J. Anthocyanins in grapes and grape products. Crit. Rev. Food Sci. Nutr. 1995, 35, 341–371. [Google Scholar] [CrossRef]
- Lee, C.Y.; Jaworski, A.W. Identification of some phenolics in white grapes. Am. J. Enol. Vitic. 1990, 41, 87–89. [Google Scholar]
- Hernandez-Jimenez, A.; Gomez-Plaza, E.; Martinez-Cutillas, A.; Kennedy, J.A. Grape skin and seed proanthocyanidins from Monastrell x Syrah grapes. J. Agric. Food Chem. 2009, 57, 10798–10803. [Google Scholar] [CrossRef] [PubMed]
- De Freitas, V.A.P.; Glories, Y. Concentration and compositional changes of procyanidins in grape seeds and skin of white Vitis vinifera varieties. J. Sci. Food Agric. 1999, 79, 1601–1606. [Google Scholar] [CrossRef]
- Ky, I.; Lorrain, B.; Kolbas, N.; Crozier, A.; Teissedre, P.L. Wine by-products: Phenolic characterization and antioxidant activity evaluation of grapes and grape pomaces from six different French grape varieties. Molecules 2014, 19, 482–506. [Google Scholar] [CrossRef] [PubMed]
- Labarbe, B.; Cheynier, V.; Brossaud, F.; Souquet, J.M.; Moutonnee, M. Quantitative fractionation of grape proanthocyanidins according to their degree of polymerization. J. Agric. Food Chem. 1999, 47, 2719–2723. [Google Scholar] [CrossRef] [PubMed]
- Prieur, C.; Rigaud, J.; Cheynier, V.; Moutounet, M. Oligomeric and polymeric procyanidins from grape seeds. Phytochemistry 1994, 36, 781–784. [Google Scholar] [CrossRef]
- Souquet, J.M.; Cheynier, V.; Brossaud, F.; Moutounet, M. Polymeric proanthocyanidins from grape skins. Phytochemistry 1996, 43, 509–512. [Google Scholar] [CrossRef]
- Pinelo-Jiménez, M.; Arnous, A.; Meyer, A.B.S. Upgrading of grape skins: Significance of plant cell-wall structural components and extraction techniques for phenol release. Trends Food Sci. Technol. 2006, 17, 579–590. [Google Scholar] [CrossRef]
- Mattivi, F.; Guzzon, R.; Vrhovsek, U.; Stefanini, M.; Velasco, R. Metabolite profiling of grape: Flavonols and anthocyanins. J. Agric. Food Chem. 2006, 54, 7692–7702. [Google Scholar] [CrossRef]
- Castillo-Muñoz, N.; Gómez-Alonso, S.; García-Romero, E.; Hermosín-Gutiérrez, I. Flavonol profiles of Vitis vinifera red grapes and their single-cultivar wines. J. Agric. Food Chem. 2007, 55, 992–1002. [Google Scholar] [CrossRef]
- Boido, E.; García-Marino, M.; Dellacassa, E.; Carrau, F.; Rivas-Gonzalo, J.C.; Escribano-Bailón, M.T. Characterisation and evolution of grape polyphenol profiles of Vitis vinifera L. cv. Tannat during ripening and vinification. Aust. J. Grape Wine Res. 2011, 17, 383–393. [Google Scholar] [CrossRef]
- Yilmaz, Y.; Toledo, R.T. Major flavonoids in grape seeds and skins: Antioxidant capacity of catechin, epicatechin, and gallic acid. J. Agric. Food Chem. 2004, 52, 255–260. [Google Scholar] [CrossRef]
- Zhu, L.; Zhang, Y.; Lu, J. Phenolic contents and compositions in skins of red wine grape cultivars among various genetic backgrounds and originations. Int. J. Mol. Sci. 2012, 13, 3492–3510. [Google Scholar] [CrossRef]
- Németh, G.; Hegyi, O.; Dunai, A.; Kocsis, L. Stilbenes in the different organs of Vitis vinifera cv. Merlot grafted on TK5BB rootstock. OENO One 2017, 51, 323–328. [Google Scholar] [CrossRef]
- Guerrero, R.F.; Puertas, B.; Fernández, M.I.; Palma, M.; Cantos-Villar, E. Induction of stilbenes in grapes by UV-C: Comparison of different subspecies of Vitis. Innov. Food Sci. Emerg. Technol. 2010, 11, 231–238. [Google Scholar] [CrossRef]
- Rodríguez-Montealegre, R.; Peces, R.; Vozmediano, J.; Gascueña, J.; Romero, E. Phenolic compounds in skins and seeds of ten grape Vitis vinifera varieties grown in a warm climate. J. Food Compos. Anal. 2006, 19, 687–693. [Google Scholar] [CrossRef]
- Cardona, F.; Andrés-Lacueva, C.; Tulipani, S.; Tinahones, F.J.; Queipo-Ortuño, M.I. Benefits of polyphenols on gut microbiota and implications in human health. J. Nutr. Biochem. 2013, 24, 1415–1422. [Google Scholar] [CrossRef] [PubMed]
- Chacar, S.; Itani, T.; Hajal, J.; Saliba, Y.; Louka, N.; Faivre, J.-F.; Maroun, R.; Fares, N. The impact of long-term intake of phenolic compounds-rich grape pomace on rat gut microbiota. J. Food Sci. 2018, 83, 246–251. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.R.; Krych, L.; Fauzan Ahmad, H.; Nejsum, P.; Skovgaard, K.; Nielsen, D.S.; Thamsborg, S.M. A polyphenol-enriched diet and Ascaris suum infection modulate mucosal immune responses and gut microbiota composition in pigs. PLoS ONE 2017, 12, e0186546. [Google Scholar] [CrossRef] [PubMed]
- Van Hul, M.; Geurts, L.; Plovier, H.; Druart, C.; Everard, A.; Ståhlman, M.; Rhimi, M.; Chira, K.; Teissedre, P.L.; Delzenne, N.M.; et al. Reduced obesity, diabetes, and steatosis upon cinnamon and grape pomace are associated with changes in gut microbiota and markers of gut barrier. Am. J. Physiol. Endocrinol. Metab. 2018, 314, E334–E352. [Google Scholar] [CrossRef]
- Lu, F.; Liu, F.; Zhou, Q.; Hu, X.; Zhang, Y. Effects of grape pomace and seed polyphenol extracts on the recovery of gut microbiota after antibiotic treatment in high-fat diet-fed mice. Food Sci. Nutr. 2019, 7, 2897–2906. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Romero, S.; Martínez-Maqueda, D.; Hereu, M.; Amézqueta, S.; Torres, J.L.; Pérez-Jiménez, J. Modifications of gut microbiota after grape pomace supplementation in subjects at cardiometabolic risk: A randomized cross-over controlled clinical trial. Foods 2020, 9, 1279. [Google Scholar] [CrossRef]
- Taladrid, D.; De Celis, M.; Belda, I.; Bartolomé, B.; Moreno-Arribas, M.V. Hypertension- and glycaemia-lowering effects of a grape-pomace-derived seasoning in high-cardiovascular risk and healthy subjects. Interplay with the gut microbiome. Food Funct. 2022, 13, 2068–2082. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Romero, S.; Léniz, A.; Martínez-Maqueda, D.; Amézqueta, S.; Fernández-Quintela, A.; Hereu, M.; Torres, J.L.; Portillo, M.P.; Pérez-Jiménez, J. Inter-individual variability in insulin response after grape pomace supplementation in subjects at high cardiometabolic risk: Role of microbiota and miRNA. Mol. Nutr. Food Res. 2021, 65, 2000113. [Google Scholar] [CrossRef] [PubMed]
- Calabriso, N.; Massaro, M.; Scoditti, E.; Verri, T.; Barca, A.; Gerardi, C.; Giovinazzo, G.; Carluccio, M.A. Grape pomace extract attenuates inflammatory response in intestinal epithelial and endothelial cells: Potential health-promoting properties in bowel inflammation. Nutrients 2022, 14, 1175. [Google Scholar] [CrossRef]
- Boussenna, A.; Joubert-Zakeyh, J.; Fraisse, D.; Pereira, B.; Vasson, M.P.; Texier, O.; Felgines, C. Dietary supplementation with a low dose of polyphenol-rich grape pomace extract prevents dextran sulfate sodium-induced colitis in rats. J. Med. Food 2016, 19, 755–758. [Google Scholar] [CrossRef] [PubMed]
- Perdicaro, D.J.; Rodriguez Lanzi, C.; Fontana, A.R.; Antoniolli, A.; Piccoli, P.; Miatello, R.M.; Diez, E.R.; Vazquez Prieto, M.A. Grape pomace reduced reperfusion arrhythmias in rats with a high-fat-fructose diet. Food Funct. 2017, 8, 3501–3509. [Google Scholar] [CrossRef] [PubMed]
- Balea, Ş.S.; Parvu, A.E.; Pop, N.; Marin, F.Z.; Andreicuț, A.; Parvu, M. Phytochemical profiling, antioxidant and cardioprotective properties of Pinot Noir cultivar pomace extracts. Farmacia 2018, 66, 432–441. [Google Scholar] [CrossRef]
- Rasines-Perea, Z.; Ky, I.; Cros, G.; Crozier, A.; Teissedre, P.-L. Grape pomace: Antioxidant activity, potential effect against hypertension and metabolites characterization after intake. Diseases 2018, 6, 60. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez Lanzi, C.; Perdicaro, D.J.; Antoniolli, A.; Fontana, A.R.; Miatello, R.M.; Bottini, R.; Vazquez Prieto, M.A. Grape pomace and grape pomace extract improve insulin signaling in high-fat-fructose fed rat-induced metabolic syndrome. Food Funct. 2016, 7, 1544–1553. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Parry, J.; Weeda, S.; Ren, S.; Castonguay, T.W.; Guo, T.L. Grape pomace aqueous extract (GPE) prevents high fat diet-induced diabetes and attenuates systemic inflammation. Food Sci. Nutr. 2016, 7, 647–660. [Google Scholar] [CrossRef]
- Daniel, T.; Ben-Shachar, M.; Drori, E.; Hamad, S.; Permyakova, A.; Ben-Cnaan, E.; Tam, J.; Kerem, Z.; Rosenzweig, T. Grape pomace reduces the severity of non-alcoholic hepatic steatosis and the development of steatohepatitis by improving insulin sensitivity and reducing ectopic fat deposition in mice. J. Nutr. Biochem. 2021, 98, 108867. [Google Scholar] [CrossRef] [PubMed]
- Harikrishnan, R.; Devi, G.; Van Doan, H.; Balasundaram, C.; Esteban, M.Á.; Abdel-Tawwab, M. Impact of grape pomace flour (GPF) on immunity and immune-antioxidant-anti-inflammatory genes expression in Labeo rohita against Flavobacterium columnaris. Fish Shellfish Immunol. 2021, 111, 69–82. [Google Scholar] [CrossRef] [PubMed]
- Del Pino-García, R.; Rivero-Pérez, M.D.; González-Sanjosé, M.L.; Ortega-Heras, M.; García Lomillo, J.; Muñiz, P. Chemopreventive potential of powdered red wine pomace seasonings against colorectal cancer in HT-29 cells. J. Agric. Food Chem. 2017, 65, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Mokni, M.; Hamlaoui, S.; Kadri, S.; Limam, F.; Amri, M.; Marzouki, L.; Aouani, E. Grape seed and skin extract protects kidney from doxorubicin-induced oxidative injury. Pak. J. Pharm. Sci. 2016, 29, 961–968. [Google Scholar]
- Fernández-Fernández, A.M.; Dellacassa, E.; Nardin, T.; Larcher, R.; Ibañez, C.; Terán, D.; Gámbaro, A.; Medrano-Fernandez, A.; Del Castillo, M.D. Tannat grape skin: A feasible ingredient for the formulation of snacks with potential for reducing the risk of diabetes. Nutrients 2022, 14, 419. [Google Scholar] [CrossRef]
- Doshi, P.; Adsule, P.; Banerjee, K.; Oulkar, D. Phenolic compounds, antioxidant activity and insulinotropic effect of extracts prepared from grape (Vitis vinifera L.) byproducts. J. Food Sci. Technol. 2015, 52, 181–190. [Google Scholar] [CrossRef]
- da Costa, G.F.; Santos, I.B.; De Bem, G.F.; Cordeiro, V.S.C.; Da Costa, C.A.; De Carvalho, L.C.R.M.; Ognibene, D.T.; Resende, A.C.; De Moura, R.S. The beneficial effect of anthocyanidin-rich Vitis vinifera L. grape skin extract on metabolic changes induced by high-fat diet in mice involves antiinflammatory and antioxidant actions. Phytother. Res. 2017, 31, 1621–1632. [Google Scholar] [CrossRef] [PubMed]
- Casanova-Martí, À.; Serrano, J.; Portune, K.J.; Sanz, Y.; Blay, M.T.; Terra, X.; Ardévol, A.; Pinent, M. Grape seed proanthocyanidins influence gut microbiota and enteroendocrine secretions in female rats. Food Funct. 2018, 9, 1672–1682. [Google Scholar] [CrossRef]
- Yang, G.; Xue, Y.; Zhang, H.; Du, M.; Zhu, M.-J. Favourable effects of grape seed extract on intestinal epithelial differentiation and barrier function in IL10-deficient mice. Br. J. Nutr. 2015, 114, 15–23. [Google Scholar] [CrossRef][Green Version]
- Yang, G.; Wang, H.; Kang, Y.; Zhu, M.J. Grape seed extract improves epithelial structure and suppresses inflammation in ileum of IL-10-deficient mice. Food Funct. 2014, 5, 2558–2563. [Google Scholar] [CrossRef]
- Saadoune, Z.; Laribi, H.; Benali, Y.; Brahimi, A.; Bennani, R.; El-Hadi, D. Valorization of algerian grape pomace seeds: Extraction of bioactive compounds, prevention and treatment of experimental inflammatory bowel diseases. Waste Biomass Valorization 2021, 12, 5401–5412. [Google Scholar] [CrossRef]
- Mohamed, H.K.; El-Fattah, H.M.A.; Barakat, H.; Ahmed, K.A.; Galal, S.M. Effect of grape seeds (Vitis vinifera L.) and mandarin peels (Citrus reticulate L.) extracts on the cardiotoxicity induced by cyclophosphamide in rats. Annu. Res. Rev. Biol. 2021, 36, 31–53. [Google Scholar] [CrossRef]
- Odai, T.; Terauchi, M.; Kato, K.; Hirose, A.; Miyasaka, N. Effects of grape seed proanthocyanidin extract on vascular endothelial function in participants with prehypertension: A randomized, double-blind, placebo-controlled study. Nutrients 2019, 11, 2844. [Google Scholar] [CrossRef]
- Leone, A.; Longo, C.; Gerardi, C.; Trosko, J.E. Pro-apoptotic effect of grape seed extract on MCF-7 involves transient increase of gap junction intercellular communication and Cx43 up-regulation: A mechanism of chemoprevention. Int. J. Mol. Sci. 2019, 20, 3244. [Google Scholar] [CrossRef]
- Irandoost, P.; Ebrahimi-Mameghani, M.; Pirouzpanah, S. Does grape seed oil improve inflammation and insulin resistance in overweight or obese women? Int. J. Food Sci. Nutr. 2013, 64, 706–710. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Yagiz, Y.; Xu, C.; Lu, J.; Chung, S.; Marshall, M.R. Muscadine grape seed oil as a novel source of tocotrienols to reduce adipogenesis and adipocyte inflammation. Food Funct. 2015, 6, 2293–2302. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Shen, Y.; Zhu, J.; Xiao, J.; Cong, R.; Zhang, H.; Wu, G.; Qi, X. Virgin grape seed oil alleviates insulin resistance and energy metabolism disorder in mice fed a high-fat diet. Eur. J. Lipid Sci. Technol. 2020, 122, 1900158. [Google Scholar] [CrossRef]
- Pazos-Tomas, C.C.; García-Montalvo, I.A. Consumption of vegetable oils of Persea americana L., and Vitis vinifera L., as part of nutritional support in non-communicable diseases in the population of the State of Oaxaca, Mexico: Pretest–postest intervention S. J. Nutr. Sci. Vitaminol. 2021, 67, 135–138. [Google Scholar] [CrossRef]
- Wall-Medrano, A.; De La Rosa, L.A.; Vázquez-Flores, A.A.; Mercado-Mercado, G.; González-Arellanes, R.; López-Díaz, J.A.; González-Córdova, A.F.; González-Aguilar, G.A.; Vallejo-Cordoba, B.; Molina-Corral, F.J. Lipidomic and antioxidant response to grape seed, corn and coconut oils in healthy wistar rats. Nutrients 2017, 9, 82. [Google Scholar] [CrossRef]
- Ebrahimi-Mameghani, M.; Irandoost, P.; Pourmoradian, S. The effects of grape seed oil on the cardiovascular risk factors in overweight and obese women: A double-blind randomized clinical trial. Curr. Top. Nutraceutical. Res. 2019, 18, 221–226. [Google Scholar]
- Kaseb, F.; Biregani, A.N. Effects of olive oil and grape seed oil on lipid profile and blood pressure in patients with hyperlipidemia: A randomized clinical trial. Food Sci. Nutr. 2016, 7, 682–688. [Google Scholar] [CrossRef]
- Bocsan, I.C.; Pop, R.M.; Sabin, O.; Sarkandy, E.; Boarescu, P.M.; Roşian, Ş.H.; Leru, P.M.; Chedea, V.S.; Socaci, S.A.; Buzoianu, A.D. Comparative protective effect of Nigella sativa oil and Vitis vinifera seed oil in an experimental model of isoproterenol-induced acute myocardial ischemia in rats. Molecules 2021, 26, 3221. [Google Scholar] [CrossRef] [PubMed]
- Ismail, A.F.M.; Moawed, F.S.M.; Mohamed, M.A. Protective mechanism of grape seed oil on carbon tetrachloride-induced brain damage in γ-irradiated rats. J. Photochem. Photobiol. 2015, 153, 317–323. [Google Scholar] [CrossRef]
- Ismail, A.F.M.; Salem, A.A.M.; Eassawy, M.M.T. Hepatoprotective effect of grape seed oil against carbon tetrachloride induced oxidative stress in liver of γ-irradiated rat. J. Photochem. Photobiol. 2016, 160, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Millan-Linares, M.C.; Bermudez, B.; Martin, M.E.; Muñoz, E.; Abia, R.; Millan, F.; Muriana, F.J.G.; Montserrat-De La Paz, S. Unsaponifiable fraction isolated from grape (Vitis vinifera L.) seed oil attenuates oxidative and inflammatory responses in human primary monocytes. Food Funct. 2018, 9, 2517–2523. [Google Scholar] [CrossRef] [PubMed]
- Orabi, S.H.; Shawky, S.M. Ameliorative effects of grape seed oil on chromium-induced nephrotoxicity and oxidative stress in rats. Slov. Vet. Zb. 2020, 57, 123–131. [Google Scholar]
- Tanideh, N.; Ashkani-Esfahani, S.; Sadeghi, F.; Koohi-Hosseinabadi, O.; Irajie, C.; Iraji, A.; Lubberts, B.; Mohammadi Samani, S. The protective effects of grape seed oil on induced osteoarthritis of the knee in male rat models. J. Orthop. Surg. Res. 2020, 15, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Niknami, E.; Sajjadi, S.-E.; Talebi, A.; Minaiyan, M. Protective effect of Vitis vinifera (black grape) seed extract and oil on acetic acid-induced colitis in rats. Int. J. Prev. Med. 2020, 11, 102. [Google Scholar]
- Berahmand, F.; Anoush, G.; Hosseini, M.-J.; Anoush, M. Grape seed oil as a natural therapy in male rats with Alzheimer’s diseases. Adv. Pharm. Bull. 2020, 10, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Dani, C.; Oliboni, L.S.; Agostini, F.; Funchal, C.; Serafini, L.; Henriques, J.A.; Salvador, M. Phenolic content of grapevine leaves (Vitis labrusca var. Bordo) and its neuroprotective effect against peroxide damage. Toxicol. Vitro 2010, 24, 148–153. [Google Scholar] [CrossRef]
- Meng, L.; Jiao, Y.; Zhou, X.; Liang, C.; Yan, K.; Zhao, Y.; Deng, X.; Han, X.; Yang, Y.; Liu, H.; et al. Leaf extract from Vitis vinifera L. reduces high fat diet-induced obesity in mice. Food Funct. 2021, 12, 6452–6463. [Google Scholar] [CrossRef]
- Zga, N.; Papastamoulis, Y.; Toribio, A.; Richard, T.; Delaunay, J.C.; Jeandet, P.; Renault, J.H.; Monti, J.P.; Mérillon, J.M.; Waffo-Téguo, P. Preparative purification of antiamyloidogenic stilbenoids from Vitis vinifera (Chardonnay) stems by centrifugal partition chromatography. J. Chromatogr. B 2009, 877, 1000–1004. [Google Scholar] [CrossRef]
- Biais, B.; Krisa, S.; Cluzet, S.; Da Costa, G.; Waffo-Teguo, P.; Mérillon, J.-M.; Richard, T. Antioxidant and cytoprotective activities of grapevine stilbenes. J. Agric. Food Chem. 2017, 65, 4952–4960. [Google Scholar] [CrossRef] [PubMed]
- Nassra, M.; Krisa, S.; Papastamoulis, Y.; Kapche, G.D.; Bisson, J.; André, C.; Konsman, J.P.; Schmitter, J.M.; Mérillon, J.M.; Waffo-Téguo, P. Inhibitory activity of plant stilbenoids against nitric oxide production by lipopolysaccharide-activated microglia. Planta Med. 2013, 79, 966–970. [Google Scholar] [CrossRef] [PubMed]
- Quero, J.; Jiménez-Moreno, N.; Esparza, I.; Osada, J.; Cerrada, E.; Ancín-Azpilicueta, C.; Rodríguez-Yoldi, M.J. Grape stem extracts with potential anticancer and antioxidant properties. Antioxidants 2021, 10, 243. [Google Scholar] [CrossRef] [PubMed]
- Empl, M.T.; Cai, H.; Wang, S.; Junginger, J.; Kostka, T.; Hewicker-Trautwein, M.; Brown, K.; Gescher, A.J.; Steinberg, P. Effects of a grapevine shoot extract containing resveratrol and resveratrol oligomers on intestinal adenoma development in mice: In vitro and in vivo studies. Mol. Nutr. Food Res. 2018, 62, 1700450. [Google Scholar] [CrossRef] [PubMed]

| Chemical Structure | Phenolic Compounds | Grape Pomace | References |
|---|---|---|---|
| Flavonoid | Procyanidins (polymers of catechin), flavan-3-ol monomers; catechin, epicatechin and oligomers (B1, B2, B3, B4 and the trimer C1) | Seeds and skins | [70,71,72,73,74,75,76,77] |
| Prodelphinidins (polymers of gallocatechin), gallocatechin, epigallocatechin, epigallocatechin gallate | Skins | [74,76,77,78] | |
| Anthocyanins; malvidin-3-O-glucoside, cyanidin-3-O-glucoside, delphinidin-3-O-glucoside, peonidin-3-O-glucoside, and petunidin-3-O-glucoside | Skins and pulp | [24,71,73,75,79] | |
| Flavonols (quercetin 3-O-glucoside, quercetin 3-glucuronide, myricetin 3-O-glucoside, kaempferol 3-O-glucoside, laricitrin 3-O-glucoside, isorhamnetin 3-O-glucoside, and syringetin3-O-glucoside) | Seeds and skins | [80,81,82] | |
| Non-Flavonoid | Hydroxybenzoic acids (gallic acid, protocatechuic acid, vanillic acid) | Seeds, skins and stems | [83,84] |
| Stilbenes (glucosides piceid, resveratrol, astringin, viniferin) | Stems, seeds skins | [73,85,86] | |
| Hydroxycinnamic acids (caffeic acid, caftaric acid, p-coumaric acid, fertaric acid) | Skins, seeds and stems | [85,87] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferrer-Gallego, R.; Silva, P. The Wine Industry By-Products: Applications for Food Industry and Health Benefits. Antioxidants 2022, 11, 2025. https://doi.org/10.3390/antiox11102025
Ferrer-Gallego R, Silva P. The Wine Industry By-Products: Applications for Food Industry and Health Benefits. Antioxidants. 2022; 11(10):2025. https://doi.org/10.3390/antiox11102025
Chicago/Turabian StyleFerrer-Gallego, Raúl, and Paula Silva. 2022. "The Wine Industry By-Products: Applications for Food Industry and Health Benefits" Antioxidants 11, no. 10: 2025. https://doi.org/10.3390/antiox11102025
APA StyleFerrer-Gallego, R., & Silva, P. (2022). The Wine Industry By-Products: Applications for Food Industry and Health Benefits. Antioxidants, 11(10), 2025. https://doi.org/10.3390/antiox11102025








