The Use of the Coenzyme Q10 as a Food Supplement in the Management of Fibromyalgia: A Critical Review
Abstract
1. Introduction
2. Fibromyalgia: Key Characteristics and Therapeutic Approaches
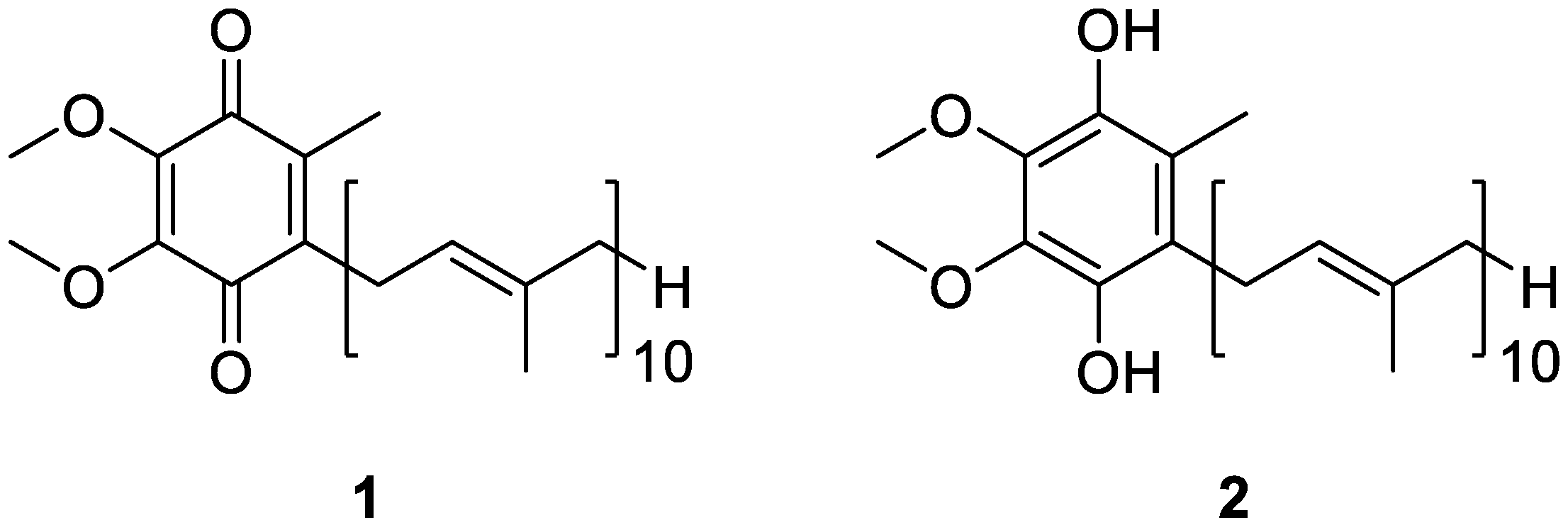
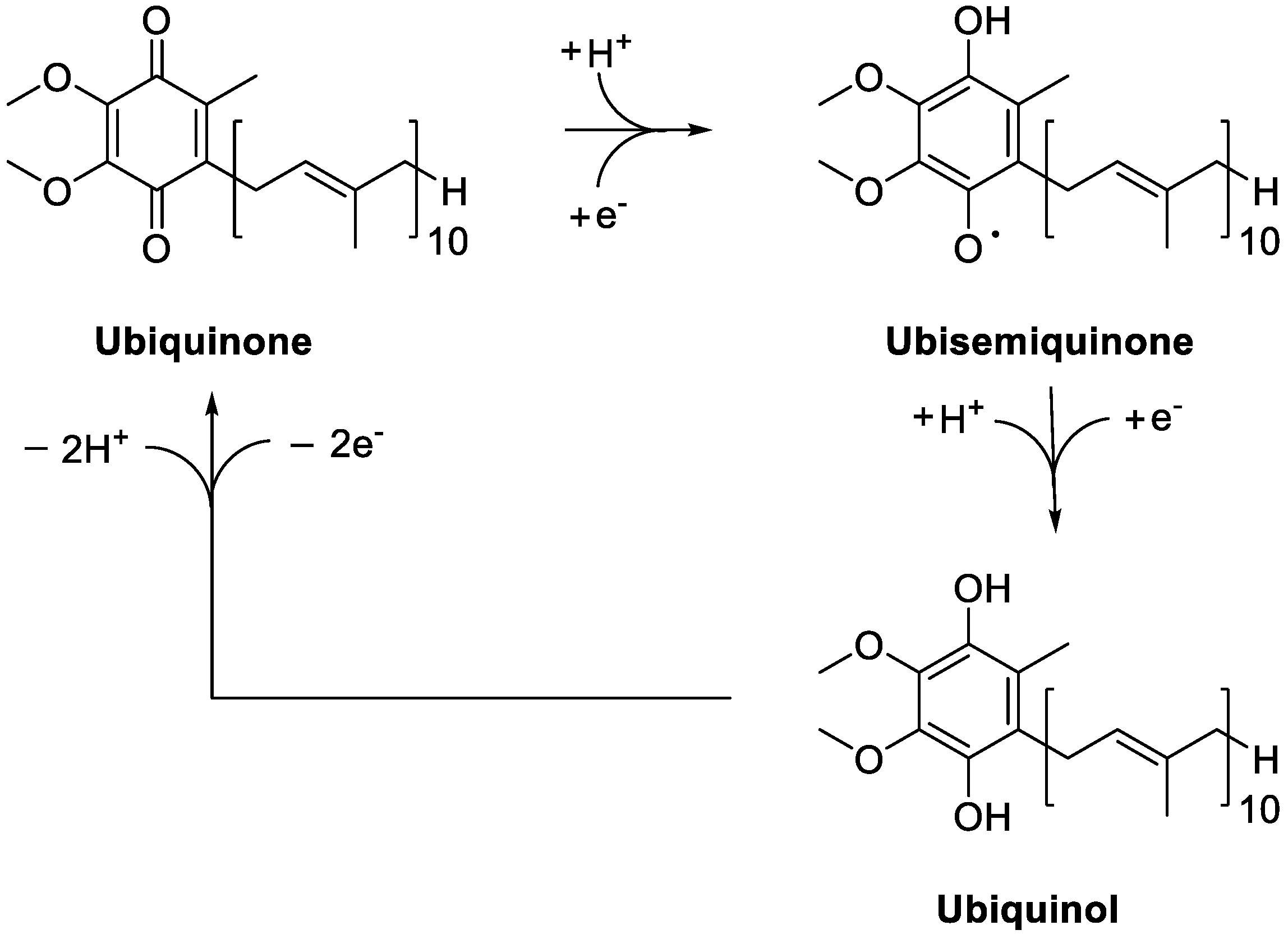
3. Coenzyme Q10 as a Key Functional Derivative
4. Sources of Coenzyme Q10 and Metabolic Fate
4.1. Food Matrices
4.2. Food Supplements
| Delivery System | Emulsifier and Additives | Study Findings | Reference |
|---|---|---|---|
| Nanoparticle | Polylactic-co-glycolitic acid | Administered as a single oral dose to Sprague Dawley rats, nanoparticles allowed for a sustained CoQ10 plasma level for at least 72 h. An almost four-fold improvement in oral bioavailability of CoQ10 was obtained, if compared to non-nanoparticulate formulation. | Swarnakar et al. [72] |
| Liposome | Cholesterol Soya bean phosphatidylcholine | The effects of temperature, pressure, and components on CoQ10 loading in liposomes were investigated and optimized. The optimized conditions allowed for a 82.28% entrapment efficiency of CoQ10. | Xia et al. [73] |
| Liposome | Cholesterol Phosphatidylcholine | The Rapid Expansion of Supercritical Solutions (RESS) technique was used to obtain highly dispersed, nanoscaled CoQ10 liposomes. Their CoQ10 entrapment efficiency was up to 90%. | Xu et al. [74] |
| Liposome | Cholesterol Egg phosphatidylcholine 1,2-Distearoyl-sn-glycero-3-phosphoethanolamine-N-[methoxy(polyethyleneglycol)-2000] | Administered by intracoronary infusion to rabbits, with an acute experimental myocardial infarction, they proved to effectively protect the myocardium from ischemia/reperfusion damage by diminishing the size of the irreversibly damaged zone. | Verma et al. [75] |
| Nanocapsule | Lecithin Caprylic/capric triglycerides Glycerol | Nanocapsules were prepared by high-pressure homogenization. Administered orally in mice, they increased the bioavailability by 176.6% for the coenzyme after 24 h. | Zhou et al. [76] |
| Solid Dispersion | Poloxamer-188 Water | A simple, rapid, cost effective, uncomplicated, and potentially scalable method was developed, to obtain a binary solid dispersion. It exhibited a remarkably improved aqueous solubility and dissolution of CoQ10. | Bhandari et al. [78] |
| Solid Dispersion | Poloxamer 407 Aerosil® 200 | The solid dispersion made from CoQ10, poloxamer 407, and Aerosil® 200, used in the weight ratio of 1:5:6, allowed for an increase in both the solubility and dissolution of the coenzyme. | Nepal et al. [79] |
| Capsule | γ-Cyclodextrin | Administered to healthy adult volunteers as a single dose, capsules containing CoQ10 complexed with γ-cyclodextrin allowed for a significant increase in the absorption and bioavailability of the coenzyme. | Terao et al. [80] |
| Nanoemulsion | Rice bran oil Octenyl succinic anhydride modified starch | A stable nanoemulsion was obtained, showing a CoQ10 encapsulation efficiency higher than 98%. | Cheuk et al. [81] |
| Nanoemulsion | d-α-Tocopheryl polyethylene glycol 1000 succinate Lecithin | Two different nanoemulsions were obtained, using either the water-soluble alpha-tocopheryl polyethylene glycol 1000 succinate (CoQ10-NE-TPGS) or lecithin (CoQ10-NE-LC) as the key lipid solubilizers. After intravenous administration to Sprague Dawley rats, CoQ10-NE-TPGS delivered CoQ10 to heart tissue more effectively than CoQ10-NE-LC. | Zhou et al. [82] |
4.3. Metabolic Fate
5. Coenzyme Q10 as Food Supplement in Fibromyalgia Patients
| Year of Study | Number of FM Subjects | Study Findings | Reference |
|---|---|---|---|
| 2012 | 20 | Decreased levels of CoQ10 in SCs and BMCs were observed in FM patients when compared to healthy subjects. 10 patients were supplemented with 300 mg/day CoQ10 for 3 months. An enhancement in CoQ10 levels in both SCs and BMCs was observed in the supplemented patients, accompanied by a concomitant improvement of symptoms. | Cordero et al. [92] |
| 2012 | 20 | Decreased levels of CoQ10, catalase, and ATP, and increased levels of LPO in BMCs were observed in FM patients when compared to healthy subjects. 10 patients were supplemented with 300 mg/day CoQ10 for 3 months. Oral supplementation restored biochemical parameters and induced a significant improvement in clinical and headache symptoms | Cordero et al. [93] |
| 2012 | 1 | Decreased expression levels of mitochondrial chain proteins (complex I, 39-kDa subunit; complex III, core 1 subunit; and complex IV, cyclooxygenase-2) and decreased levels of CoQ10 were observed in the BMCs of the patient. Instead, LPO and expression levels of OGG-1 turned out to be increased in BMCs. Supplementation of 300 mg/day CoQ10 for 3 months restored the mitochondrial proteins expression levels and CoQ10 levels, decreasing also LPO and OGG-1 expression levels in BMCs. A significant improvement of clinical symptoms was also observed. | Cordero et al. [94] |
| 2013 | 20 | 40 days of CoQ10 supplementation, 300 mg/day, allowed for a reduction in pain, fatigue, and morning tiredness in FM patients. Furthermore, a recovery of inflammation, antioxidant enzymes, mitochondrial biogenesis, and AMPK gene expression levels, associated with phosphorylation of the AMPK activity, were also observed. | Cordero et al. [95] |
| 2013 | 4 | 9 months of CoQ10 supplementation, 300 mg/day, allowed for an improvement in clinical symptoms, including pain, fatigue, sleep, and tender points. Somatization and anxiety were also significantly reduced. | Alcocer-Gómez et al. [96] |
| 2017 | 22 | DDM Chinone®, containing 200 mg/dose of CoQ10 enriched with vitamins E, B2, B6, B12 and folic acid, was administered twice a day for three months. Supplementation exerted beneficial effects in counteracting pain, fatigue, sleep disturbance, and mental difficulties in the treated patients. | Di Pierro et al. [97] |
| 2022 | 23 | Supplementation with CoQ10, tryptophan, and magnesium for 3 months proved to be well tolerated by the administered patients. Moreover, it improved physical symptoms such as fatigue, sleep quality and functional capacity, as well as the global well-being. | Gómez-Centeno et al. [98] |
| 2013 | 10 | Patients with juvenile FM displayed significantly increased levels of free cholesterol, cholesterol esters and free fatty acids, accompanied by CoQ10 deficiency and increased oxidative stress. Supplementation of 100 mg/day of ubiquinol-10 for 12 weeks increased CoQ10 levels and decreased plasma levels of free cholesterol and cholesterol esters. Moreover, chronic fatigue was also improved. | Miyamae et al. [99] |
| 2019 | 11 | Administration of CoQ10 to pregabalin-treated FM patients reduced pain, anxiety, brain activity, mitochondrial oxidative stress, and inflammation. Moreover, it also increased the levels of reduced glutathione and superoxide dismutase. | Sawaddiruk et al. [100] |
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Pelton, R. Coenzyme Q10: A miracle nutrient advances in understanding. Integr. Med. 2020, 19, 16–20. [Google Scholar]
- Jafari, M.; Masood Mousavi, S.; Asgharzadeh, A.; Yazdanie, N. Coenzyme Q10 in the treatment of heart failure: A systematic review of systematic reviews. Indian Heart J. 2018, 70, S111–S117. [Google Scholar] [CrossRef] [PubMed]
- Littarru, G.P.; Tiano, L. Clinical aspects of coenzyme Q10: An update. Nutrition 2010, 26, 250–254. [Google Scholar] [CrossRef] [PubMed]
- Cordero, M.D.; de Miguel, M.; Carmona-López, I.; Bonal, P.; Campa, F.; Moreno-Fernández, A.M. Oxidative stress and mitochondrial dysfunction in fibromyalgia. Neuro Endocrinol. Lett. 2010, 31, 169–173. [Google Scholar]
- Häuser, W.; Ablin, J.; Fitzcharles, M.-A.; Littlejohn, G.; Luciano, J.V.; Usui, C.; Walitt, B. Fibromyalgia. Nat. Rev. Dis. Prim. 2015, 13, 15022. [Google Scholar] [CrossRef]
- Marques, A.P.; de Sousa do Espirito Santo, A.; Berssaneti, A.A.; Matsutani, L.A.; Yuan, S.L.K. Prevalence of fibromyalgia: Literature review update. Rev. Bras. Reumatol. Engl. Ed. 2017, 57, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Creed, F. A review of the incidence and risk factors for fibromyalgia and chronic widespread pain in population-based studies. PAIN 2020, 161, 1169–1176. [Google Scholar] [CrossRef] [PubMed]
- Kashikar-Zuck, S.; Ting, T.V. Juvenile fibromyalgia: Current status of research and future developments. Nat. Rev. Rheumatol. 2014, 10, 89–96. [Google Scholar] [CrossRef]
- Jacobson, S.A.; Simpson, R.G.; Lubahn, C.; Hu, C.; Belden, C.M.; Davis, K.J.; Nicholson, L.R.; Long, K.E.; Osredkar, T.; Lorton, D. Characterization of fibromyalgia symptoms in patients 55 to 95 years old: A longitudinal study showing symptom persistence with suboptimal treatment. Aging Clin. Exp. Res. 2015, 27, 75–82. [Google Scholar] [CrossRef]
- Ursini, F.; Naty, S.; Grembiale, R.D. Fibromyalgia and obesity: The hidden link. Rheumatol. Int. 2011, 31, 1403–1408. [Google Scholar] [CrossRef]
- Vincent, A.; Clauw, D.; Oh, T.H.; Whipple, M.O.; Toussaint, L.L. Decreased physical activity attributable to higher body mass index influences fibromyalgia symptoms. PM R 2014, 6, 802–807. [Google Scholar] [CrossRef] [PubMed]
- Salaffi, F.; Di Carlo, M.; Farah, S.; Atzeni, F.; Buskila, D.; Ablin, J.N.; Häuser, W.; Sarzi-Puttini, W. Diagnosis of fibromyalgia: Comparison of the 2011/2016 ACR and AAPT criteria and validation of the modified Fibromyalgia Assessment Status. Rheumatology 2020, 59, 3042–3049. [Google Scholar] [CrossRef] [PubMed]
- Ablin, J.N.; Buskila, D.; Clauw, D.J. Biomarkers in fibromyalgia. Curr. Pain Headache Rep. 2009, 13, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Harris, R.E.; Clauw, D.J.; Scott, D.J.; McLean, S.A.; Gracely, R.H.; Zubieta, J.K. Decreased central mu-opioid receptor availability in fibromyalgia. J. Neurosci. 2007, 27, 10000–10006. [Google Scholar] [CrossRef]
- Watkins, L.R.; Hutchinson, M.R.; Rice, K.C.; Maier, S.F. The “toll” of opioid-induced glial activation: Improving the clinical efficacy of opioids by targeting glia. Trends Pharmacol. Sci. 2009, 30, 581–591. [Google Scholar] [CrossRef]
- Harris, R.E.; Sundgren, P.C.; Craig, A.D.; Kirshenbaum, E.; Sen, A.; Napadow, V.; Clauw, D.J. Elevated insular glutamate (Glu) in fibromyalgia (FM) is associated with experimental pain. Arthritis Rheum. 2009, 60, 3146–3152. [Google Scholar] [CrossRef]
- Foerster, B.R.; Petrou, M.; Edden, R.A.; Sundgren, P.C.; Schmidt-Wilcke, T.; Lowe, S.E.; Harte, S.E.; Clauw, D.J.; Harris, R.E. Reduced insular gamma-aminobutyric acid in fibromyalgia. Arthritis Rheum. 2012, 64, 579–583. [Google Scholar] [CrossRef]
- Petrovic, P.; Kalso, E.; Petersson, K.M.; Ingvar, M. Placebo and opioid analgesia-- imaging a shared neuronal network. Science 2002, 295, 1737–1740. [Google Scholar] [CrossRef]
- Littlejohn, G. Neurogenic neuroinflammation in fibromyalgia and complex regional pain syndrome. Nat. Rev. Rheumatol. 2015, 11, 639–648. [Google Scholar] [CrossRef]
- Guedj, E.; Taieb, D.; Cammilleri, S.; Lussato, D.; de Laforte, C.; Niboyet, J.; Mundler, O. 99mTc-ECD brain perfusion SPECT in hyperalgesic fibromyalgia. Eur. J. Nucl. Med. Mol. Imaging 2007, 34, 130–134. [Google Scholar] [CrossRef]
- Gracely, R.H.; Petzke, F.; Wolf, J.M.; Clauw, D.J. Functional magnetic resonance imaging evidence of augmented pain processing in fibromyalgia. Arthritis Rheum. 2002, 46, 1333–1343. [Google Scholar] [CrossRef] [PubMed]
- Feraco, P.; Bacci, A.; Pedrabissi, F.; Passamonti, L.; Zampogna, G.; Pedrabissi, F.; Malavolta, N.; Leonardi, M. Metabolic abnormalities in pain-processing regions of patients with fibromyalgia: A 3T MR spectroscopy study. Am. J. Neuroradiol. 2011, 32, 1585–1590. [Google Scholar] [CrossRef] [PubMed]
- Üçeyler, N.; Zeller, D.; Kahn, A.-K.; Kewenig, S.; Kittel-Schneider, S.; Schmid, A.; Casanova-Molla, J.; Reiners, K.; Sommer, C. Small fibre pathology in patients with fibromyalgia syndrome. Brain 2013, 136, 1857–1867. [Google Scholar] [CrossRef] [PubMed]
- Castro-Marrero, J.; Cordero, M.D.; Saez-Francas, N.; Jimenez-Gutierrez, C.; Auilar-Montilla, F.J.; Aliste, L.; Alegre-Martin, J. Could mitochondrial dysfunction be a differentiating marker between chronic fatigue syndrome and fibromyalgia? Antioxid. Redox Signal 2013, 19, 1855–1860. [Google Scholar] [CrossRef]
- Sprott, H.; Salemi, S.M.; Gay, R.E.; Bradley, L.A.; Alarcon, G.S.; Oh, S.J.; Michel, B.A.; Gay, S. Increased DNA fragmentation and ultrastructural changes in fibromyalgic muscle fibres. Ann. Rheum. Dis. 2004, 63, 245–251. [Google Scholar] [CrossRef]
- Cordero, M.D.; De Miguel, M.; Moreno Fernández, A.M.; Carmona López, I.M.; Maraver, J.G.; Cotán, D.; Gómez Izquierdo, L.; Bonal, P.; Campa, F.; Bullon, P.; et al. Mitochondrial dysfunction and mitophagy activation in blood mononuclear cells of fibromyalgia patients: Implications in the pathogenesis of the disease. Arthritis Res. Ther. 2010, 12, R17. [Google Scholar] [CrossRef]
- Bullon, P.; Alcocer-Gómez, E.; Carrión, A.M.; Marín-Aguilar, F.; Garrido-Maraver, J.; Román-Malo, L.; Ruiz-Cabello, J.; Culic, O.; Ryffel, B.; Apetoh, L.; et al. AMPK phosphorylation modulates pain by activation of NLRP3 inflammasome. Antioxid. Redox Signal 2016, 24, 157–170. [Google Scholar] [CrossRef]
- Alcocer-Gómez, E.; Sánchez-Alcazar, J.A.; Battino, M.; Bullón, P.; Cordero, M.D. Aging-related changes in inflammatory and LKB1/AMPK gene expression in fibromyalgia patients. CNS Neurosci. Ther. 2014, 20, 476–478. [Google Scholar] [CrossRef]
- Macfarlane, G.J.; Kronisch, C.; Dean, L.E.; Atzeni, F.; Hauser, W.; Flu, E.; Cho, E.; Kosek, E.; Amris, K.; Branco, J.; et al. EULAR revised recommendations for the management of fibromyalgia. Ann. Rheum. Dis. 2017, 76, 318–328. [Google Scholar] [CrossRef]
- Hauser, W.; Klose, P.; Langhorst, J.; Moradi, B.; Steinbach, M.; Schiltenwolf, M.; Busch, A. Efficacy of different types of aerobic exercise in fibromyalgia syndrome: A systematic review and meta-analysis of randomised controlled trials. Arthritis Res. Ther. 2010, 12, R79. [Google Scholar] [CrossRef]
- Cazzola, M.; Atzeni, F.; Salaffi, F.; Stisi, S.; Cassisi, G.; Sarzi-Puttini, P. Which kind of exercise is best in fibromyalgia therapeutic programmes? A practical review. Clin. Exp. Rheumatol. 2010, 28, S117–S124. [Google Scholar] [PubMed]
- Larsson, A.; Palstam, A.; Löfgren, M.; Ernberg, M.; Bjersing, J.; Bileviciute-Ljungar, I.; Gerdle, B.; Kosek, E.; Mannerkorpi, K. Resistance exercise improves muscle strength, health status and pain intensity in fibromyalgia--a randomized controlled trial. Arthritis Res. Ther. 2015, 17, 161. [Google Scholar] [CrossRef] [PubMed]
- Pagliai, G.; Giangrandi, I.; Dinu, M.; Sofi, F.; Colombini, B. Nutritional interventions in the management of fibromyalgia syndrome. Nutrients 2020, 12, 2525. [Google Scholar] [CrossRef] [PubMed]
- Lowry, E.; Marley, J.; McVeigh, J.G.; Mc Sorley, E.; Allsopp, P.; Kerr, D. Dietary interventions in the management of fibromyalgia: A systematic review and best-evidence synthesis. Nutrients 2020, 12, 2664. [Google Scholar] [CrossRef] [PubMed]
- Kadayifci, F.Z.; Bradley, M.J.; Onat, A.M.; Shi, H.N.; Zheng, S. Review of nutritional approaches to fibromyalgia. Nutr. Rev. 2022, nuac036. [Google Scholar] [CrossRef]
- Fernández-Araque, A.; Verde, Z.; Torres-Ortega, C.; Sainz-Gil, M.; Velasco-Gonzalez, V.; González-Bernal, J.J.; Mielgo-Ayuso, J. Effects of antioxidants on pain perception in patients with fibromyalgia-A systematic review. J. Clin. Med. 2022, 11, 2462. [Google Scholar] [CrossRef]
- Haddad, H.W.; Mallepalli, N.R.; Scheinuk, J.E.; Bhargava, P.; Cornett, E.M.; Urits, I.; Kaye, A.D. The Role of nutrient supplementation in the management of chronic pain in fibromyalgia: A narrative review. Pain Ther. 2021, 10, 827–848. [Google Scholar] [CrossRef]
- Crane, F.L.; Hatefi, Y.; Lester, R.L.; Widmer, C. Isolation of a quinone from beef heart mitochondria. Biochim. Biophys. Acta 1957, 25, 220–221. [Google Scholar] [CrossRef]
- Morton, R.A. Ubiquinone. Nature 1958, 182, 1764–1767. [Google Scholar] [CrossRef]
- Ernster, L.; Dallner, G. Biochemical, physiological and medical aspects of ubiquinone function. Biochim. Biophys. Acta 1995, 1271, 195–204. [Google Scholar] [CrossRef]
- Frederick, L.; Crane, F.L. Biochemical functions of Coenzyme Q10. J. Am. Coll. Nutr. 2001, 20, 591–598. [Google Scholar]
- Turunen, M.; Sindelar, P.; Dallner, G. Induction of endogenous coenzyme Q biosynthesis by administration of peroxisomal inducers. Biofactors 1999, 9, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Arroyo, A.; Kagan, V.E.; Tyurin, V.A.; Burgess, J.R.; de Cabo, R.; Navas, P.; Villalba, J.M. NADH and NADPH dependent reduction of coenzyme Q at the plasma membrane. Antioxid. Redox Signal 2000, 2, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.R.; Witting, P.K.; Stocker, R. A role for reduced coenzyme Q in atherosclerosis. Biofactors 1999, 9, 207–224. [Google Scholar] [CrossRef] [PubMed]
- Schneider, D.; Elstner, E.F. Coenzyme Q10, vitamin E and dihydrothioctic acid cooperatively prevent diene conjugation in isolated low density lipoprotein. Antioxid. Redox Signal 2000, 2, 327–333. [Google Scholar] [CrossRef]
- Takahashi, T.; Okamoto, T.; Mori, K.; Sayo, H.; Kishi, T. Distribution of ubiquinone and ubiquinol homologues in rat tissues and subcellular fraction. Lipids 1993, 28, 803–809. [Google Scholar] [CrossRef]
- Quinn, P.J.; Fabisiak, J.P.; Kagan, V.E. Expansion of antioxidant function of vitamin E by coenzyme Q. Biofactors 1999, 9, 149–154. [Google Scholar] [CrossRef]
- Crane, F.L. New functions for coenzyme Q. Protoplasma 2000, 213, 127–133. [Google Scholar] [CrossRef]
- Villalba, J.M.; Navas, P. Plasma membrane redox system in the control of stress-induced apoptosis. Antioxid. Redox Signal 2000, 2, 213–230. [Google Scholar] [CrossRef]
- Sun, I.L.; Sun, E.E.; Crane, F.L.; Morré, D.J.; Lindgren, A.; Löw, H. A requirement for coenzyme Q in plasma membrane electron transport. Proc. Nat. Acad. Sci. USA 1992, 89, 11126–11130. [Google Scholar] [CrossRef]
- Takahashi, T.; Okamoto, T.; Kishi, T. Characterization of NADPH dependent ubiquinone reductase activity in rat liver cytosol. J. Biochem. 1996, 119, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Navarro, F.; Arroyo, A.; Martin, S.F.; Bello, R.I.; de Cabo, R.; Burgess, J.R.; Navas, P.; Villalba, J.M. Protective role of ubiquinone in vitamin E and selenium deficient plasma membranes. Biofactors 1999, 9, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Gorelick, C.; Lopez-Jones, M.; Goldberg, G.L.; Romney, S.L.; Khabele, D. Coenzyme Q10 and lipid-related gene induction in HeLa cells. Am. J. Obstet. Gynecol. 2004, 190, 1432–1434. [Google Scholar] [CrossRef] [PubMed]
- Groneberg, D.A.; Kindermann, B.; Althammer, M.; Klapper, M.; Vormann, J.; Littarru, G.P.; Döring, F. Coenzyme Q10 affects expression of genes involved in cell signalling, metabolism and transport in human CaCo-2 cells. Int. J. Biochem. Cell Biol. 2005, 37, 1208–1218. [Google Scholar] [CrossRef] [PubMed]
- Schmelzer, C.; Lindner, I.; Rimbach, G.; Niklowitz, P.; Menke, T.; Döring, F. Functions of coenzyme Q10 in inflammation and gene expression. Biofactors 2008, 32, 179–183. [Google Scholar] [CrossRef]
- Åberg, F.; Appelkvist, E.L.; Dallner, G.; Ernster, L. Distribution and redox state of ubiquinones in rat and human tissues. Archiv. Biochem. Biophys. 1992, 295, 230–234. [Google Scholar] [CrossRef]
- Kawamukai, M. Biosynthesis, bioproduction and novel roles of ubiquinone. J. Biosci. Bioeng. 2002, 94, 511–517. [Google Scholar] [CrossRef]
- Bonakdar, R.A.; Guarneri, E. Coenzyme Q10. Am. Fam. Phys. 2005, 72, 1065–1070. [Google Scholar]
- Hosoe, K.; Kitano, M.; Kishida, H.; Kubo, H.; Fujii, K.; Kitahara, M. Study on safety and bioavailability of ubiquinol (Kaneka QHTM) after single and 4-week multiple oral administration to heathy volunteers. Regul. Toxicol. Pharmacol. 2007, 47, 19–28. [Google Scholar] [CrossRef]
- Ikematsu, H.; Nakamura, K.; Harashima, S.I.; Fujii, K.; Fukutomi, N. Safety assessment of coenzyme Q10 (Kaneka Q10) in healthy subjects: A double-blind, randomized, placebo-control trial. Regul. Toxicol. Pharmacol. 2006, 44, 212–218. [Google Scholar] [CrossRef]
- Kikkawa, K.; Takehara, I.; Miyakoshi, T.; Miyawaki, H. Safety of high dose supplementation of coenzyme Q10 in healthy human adults. Jpn. J. Food Chem. 2007, 14, 76–81. [Google Scholar] [CrossRef]
- Kitano, M.; Watanabe, D.; Oda, S.; Kubo, H.; Kishida, H.; Fujii, K.; Kitahara, M.; Hosoe, K. Subchronic oral toxicity of ubiquinol in rats and dogs. Int. J. Toxicol. 2008, 27, 189–215. [Google Scholar] [CrossRef] [PubMed]
- Kamei, M.; Fujita, T.; Kanbe, T.; Sasaki, K.; Oshiba, K.; Otani, S.; Matsuiyuasa, I.; Morisawa, S. The distribution and content of ubiquinone in foods. Int. J. Vit. Nutr. Res. 1986, 56, 57–63. [Google Scholar]
- Mattila, P.; Kumpulainen, J. Coenzymes Q9 and Q10: Contents in foods and dietary intake. J. Food Compos. Anal. 2001, 14, 409–417. [Google Scholar] [CrossRef]
- Souchet, N.; Laplante, S. Seasonal variation of Co-enzyme Q10 content in pelagic fish tissues from Eastern Quebec. J. Food Compos. Anal. 2007, 20, 403–410. [Google Scholar] [CrossRef]
- Pravst, I.; Zmitek, K.; Zmitek, J. Coenzyme Q10 contents in food and fortification strategy. Crit. Rev. Food Sci. Nutr. 2010, 50, 269–280. [Google Scholar] [CrossRef]
- Weber, C.; Bysted, A.; Hølmer, G. Coenzyme Q10 in the diet--daily intake and relative bioavailability. Mol. Aspects Med. 1997, 18, S251–S254. [Google Scholar] [CrossRef]
- Kubo, H.; Fujii, K.; Kawabe, T.; Matsumoto, S.; Kishida, H.; Hosoe, K. Food content of ubiquinol-10 and ubiquinone-10 in the Japanese diet. J. Food Compos. Anal. 2008, 21, 199–210. [Google Scholar] [CrossRef]
- Cabrini, L.; Barzanti, V.; Cipollone, M.; Fiorentini, D.; Grossi, G.; Tolomelli, B.; Zambonin, L.; Landi, L. Antioxidants and total peroxyl radical-trapping ability of olive and seed oils. J. Agric. Food Chem. 2001, 49, 6026–6032. [Google Scholar] [CrossRef]
- Strazisar, M.; Fir, M.; Golc-Wondra, A.; Milivojevic, L.; Prosek, M.; Abram, V. Quantitative determination of coenzyme Q10 by liquid chromatography and liquid chromatography/mass spectrometry in dairy products. J. AOAC Int. 2005, 88, 1020–1027. [Google Scholar] [CrossRef]
- Ezhilarasi, P.N.; Karthik, P.; Chhanwal, N.; Anandharamakrishnan, C. Nanoencapsulation techniques for food bioactive components: A review. Food Bioprocess Technol. 2012, 6, 628–647. [Google Scholar] [CrossRef]
- Swarnakar, N.K.; Jain, A.K.; Singh, R.P.; Godugu, C.; Das, M.; Jain, S. Oral bioavailability, therapeutic efficacy and reactive oxygen species scavenging properties of coenzyme Q10-loaded polymeric nanoparticles. Biomaterials 2011, 32, 6860–6874. [Google Scholar] [CrossRef] [PubMed]
- Xia, F.; Jin, H.; Zhao, Y.; Guo, X. Preparation of coenzyme Q10 liposomes using supercritical anti-solvent technique. J. Microencapsul. 2012, 29, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Zhao, B.; He, D. Synthesis of highly dispersed nanoscaled CoQ10 liposome by supercritical fluid. Mater. Lett. 2015, 142, 283–286. [Google Scholar] [CrossRef]
- Verma, D.D.; Hartner, W.C.; Thakkar, V.; Levchenko, T.S.; Torchilin, V.P. Protective effect of coenzyme Q10-loaded liposomes on the myocardium in rabbits with an acute experimental myocardial infarction. Pharm. Res. 2007, 24, 2131–2137. [Google Scholar] [CrossRef]
- Zhou, H.; Zhang, J.; Long, Y.; Liu, G.; Duan, M.; Xia, Q. Improvement of the oral bioavailability of coenzyme Q10 with lecithin nanocapsules. J. Nanosci. Nanotechnol. 2013, 13, 706–710. [Google Scholar] [CrossRef]
- Leuner, C.; Dressman, J. Improving drug solubility for oral delivery using solid dispersions. Eur. J. Pharm. Biopharm. 2000, 50, 47–60. [Google Scholar] [CrossRef]
- Bhandari, K.H.; Newa, M.; Kim, J.A.; Yoo, B.K.; Woo, J.S.; Lyoo, W.S.; Lim, H.T.; Choi, H.G.; Yong, C.S. Preparation, characterization and evaluation of coenzyme Q10 binary solid dispersions for enhanced solubility and dissolution. Biol. Pharm. Bull. 2007, 30, 1171–1176. [Google Scholar] [CrossRef]
- Nepal, P.R.; Han, H.K.; Choi, H.K. Enhancement of solubility and dissolution of coenzyme Q10 using solid dispersion formulation. Int. J. Pharm. 2010, 383, 147–153. [Google Scholar] [CrossRef]
- Terao, K.; Nakata, D.; Fukumi, H.; Schmid, G.; Arima, H.; Hirayama, F.; Uekama, K. Enhancement of oral bioavailability of coenzyme Q10 by complexation with cyclodextrin in healthy adults. Nutr. Res. 2006, 26, 503–508. [Google Scholar] [CrossRef]
- Cheuk, S.Y.; Shih, F.F.; Champagne, E.T.; Daigle, K.W.; Patindol, J.A.; Mattison, C.P.; Boue, S.M. Nano-encapsulation of coenzyme Q10 using octenyl succinic anhydride modified starch. Food Chem. 2015, 174, 585–590. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Zhang, J.; Jin, Q.; Liu, G.; Long, Y.; Duan, M.; Xia, Q. Targeting of coenzyme Q10 via d-alpha-tocopheryl polyethylene glycol 1000 succinate-based nanoemulsion to the heart. Mater Lett. 2013, 109, 20–22. [Google Scholar] [CrossRef]
- Molyneux, S.; Florkowski, C.; Lever, M.; George, P. The bioavailability of coenzyme Q10 supplements available in New Zealand differs markedly. N. Z. Med. J. 2004, 117, U1108. [Google Scholar] [PubMed]
- Wang, Q.; Lee, B.L.; Ong, C.N. Automated high-performance liquid chromatographic method with precolumn reduction for the determination of ubiquinol and ubiquinone in human plasma. J. Chromatogr. B Biomed. Sci. Appl. 1999, 726, 297–302. [Google Scholar] [CrossRef]
- Tang, P.H.; Miles, M.V.; DeGrauw, A.; Hershey, A.; Pesce, A. HPLC analysis of reduced and oxidized coenzyme Q10 in human plasma. Clin. Chem. 2001, 47, 56–65. [Google Scholar] [CrossRef]
- Yamashita, S.; Yamamoto, Y. Simultaneous detection of ubiquinol and ubiquinone in human plasma as a marker of oxidative stress. Anal. Biochem. 1997, 250, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Kaikkonen, J.; Nyyssönen, K.; Salonen, J.T. Measurement and stability of plasma reduced, oxidized and total coenzyme Q10 in humans. Scand. J. Clin. Lab. Investig. 1999, 59, 457–466. [Google Scholar] [CrossRef]
- Craft, N.E.; Tucker, R.T.; Chitchumroonchokchai, C.; Failla, M.; Bhagavan, H.N. Assessment of coenzyme Q10 bioavailability using a coupled in vitro digestion/Caco-2 human intestinal cell model. FASEB J. 2005, 19, A449. [Google Scholar] [CrossRef]
- Miles, M.V.; Horn, P.S.; Morrison, J.A.; Tang, P.H.; DeGrauw, T.; Pesce, A.J. Plasma coenzyme Q10 reference intervals, but not redox status, are affected by gender and race in self-reported healthy adults. Clin. Chim. Acta 2003, 332, 123–132. [Google Scholar] [CrossRef]
- Bhagavan, H.N.; Chopra, R.K. Coenzyme Q10: Absorption, tissue uptake, metabolism and pharmacokinetics. Free Radic. Res. 2006, 40, 445–453. [Google Scholar] [CrossRef]
- Hargreaves, I.P.; Mantle, D. Targeted treatment of age-related fibromyalgia with supplemental coenzyme Q10. Adv. Exp. Med. Biol. 2021, 1286, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Cordero, M.D.; Santos-García, R.; Bermejo-Jover, D.; Sánchez-Domínguez, B.; Jaramillo-Santos, M.R.; Bullón, P. Coenzyme Q10 in salivary cells correlate with blood cells in fibromyalgia: Improvement in clinical and biochemical parameter after oral treatment. Clin. Biochem. 2012, 45, 509–511. [Google Scholar] [CrossRef]
- Cordero, M.D.; Cano-García, F.J.; Alcocer-Gómez, E.; De Miguel, M.; Sánchez-Alcázar, J.A. Oxidative stress correlates with headache symptoms in fibromyalgia: Coenzyme Q10 effect on clinical improvement. PLoS ONE 2012, 7, e35677. [Google Scholar] [CrossRef] [PubMed]
- Cordero, M.D.; Cotán, D.; del-Pozo-Martín, Y.; Carrión, A.M.; de Miguel, M.; Bullón, P.; Sánchez-Alcazar, J.A. Oral coenzyme Q10 supplementation improves clinical symptoms and recovers pathologic alterations in blood mononuclear cells in a fibromyalgia patient. Nutrition 2012, 28, 1200–1203. [Google Scholar] [CrossRef] [PubMed]
- Cordero, M.D.; Alcocer-Gómez, E.; de Miguel, M.; Culic, O.; Carrión, A.M.; Alvarez-Suarez, J.M.; Bullón, P.; Battino, M.; Fernández-Rodríguez, A.; Sánchez-Alcazar, J.A. Can coenzyme Q10 improve clinical and molecular parameters in fibromyalgia? Antioxid. Redox Signal 2013, 19, 1356–1361. [Google Scholar] [CrossRef] [PubMed]
- Alcocer-Gómez, E.; Cano-García, F.J.; Cordero, M.D. Effect of coenzyme Q10 evaluated by 1990 and 2010 ACR diagnostic criteria for fibromyalgia and SCL-90-R: Four case reports and literature review. Nutrition 2013, 29, 1422–1425. [Google Scholar] [CrossRef]
- Di Pierro, F.; Rossi, A.; Consensi, A.; Giacomelli, C.; Bazzichi, L. Role for a water-soluble form of CoQ10 in female subjects affected by fibromyalgia. A preliminary study. Clin. Exp. Rheumatol. 2017, 105, 20–27. [Google Scholar]
- Gómez-Centeno, A.; Ramentol, M.; Alegre, C. Nutritional supplementation with coenzyme Q10, tryptophan and magnesium in fibromyalgia treatment: A letter to editor. Reumatol. Clin. 2022, 18, 62–63. [Google Scholar] [CrossRef]
- Miyamae, T.; Seki, M.; Naga, T.; Uchino, S.; Asazuma, H.; Yoshida, T.; Iizuka, Y.; Kikuchi, M.; Imagawa, T.; Natsumeda, Y.; et al. Increased oxidative stress and coenzyme Q10 deficiency in juvenile fibromyalgia: Amelioration of hypercholesterolemia and fatigue by ubiquinol-10 supplementation. Redox Rep. 2013, 18, 12–19. [Google Scholar] [CrossRef]
- Sawaddiruk, P.; Apaijai, N.; Paiboonworachat, S.; Kaewchur, T.; Kasitanon, N.; Jaiwongkam, T.; Kerdphoo, S.; Chattipakorn, N.; Chattipakorn, S.C. Coenzyme Q10 supplementation alleviates pain in pregabalin-treated fibromyalgia patients via reducing brain activity and mitochondrial dysfunction. Free Radic. Res. 2019, 53, 901–909. [Google Scholar] [CrossRef]
- Mizuno, K.; Tanaka, M.; Nozaki, S.; Mizuma, H.; Ataka, S.; Tahara, T.; Sugino, T.; Shirai, T.; Kajimoto, Y.; Kuratsune, H.; et al. Antifatigue effects of coenzyme Q10 during physical fatigue. Nutrition 2008, 24, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-J.; Cho, W.-J.; Kim, J.-K.; Lee, D.-C. Effects of coenzyme Q10 on arterial stiffness, metabolic parameters, and fatigue in obese subjects: A double-blind randomized controlled study. Med. Food 2011, 14, 386–390. [Google Scholar] [CrossRef] [PubMed]
- Gökbel, H.; Gül, I.; Belviranl, M.; Okudan, N. The effects of coenzyme Q10 supplementation on performance during repeated bouts of supramaximal exercise in sedentary men. J. Strength Cond. Res. 2010, 24, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Thorsteinsdóttir, B.; Rafnsdóttir, S.; Geirsson, A.J.; Sigurjónsson, S.V.; Kjeld, M. No difference in ubiquinone concentration of muscles and blood in fibromyalgia patients and healthy controls. Clin. Exp. Rheumatol. 1998, 16, 513–514. [Google Scholar] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Campisi, L.; La Motta, C. The Use of the Coenzyme Q10 as a Food Supplement in the Management of Fibromyalgia: A Critical Review. Antioxidants 2022, 11, 1969. https://doi.org/10.3390/antiox11101969
Campisi L, La Motta C. The Use of the Coenzyme Q10 as a Food Supplement in the Management of Fibromyalgia: A Critical Review. Antioxidants. 2022; 11(10):1969. https://doi.org/10.3390/antiox11101969
Chicago/Turabian StyleCampisi, Luca, and Concettina La Motta. 2022. "The Use of the Coenzyme Q10 as a Food Supplement in the Management of Fibromyalgia: A Critical Review" Antioxidants 11, no. 10: 1969. https://doi.org/10.3390/antiox11101969
APA StyleCampisi, L., & La Motta, C. (2022). The Use of the Coenzyme Q10 as a Food Supplement in the Management of Fibromyalgia: A Critical Review. Antioxidants, 11(10), 1969. https://doi.org/10.3390/antiox11101969








