1. Introduction
Electrolytic hydrogen water (EHW) contains abundant molecular hydrogen and very small amounts of platinum nanoparticles with alkaline properties [
1,
2]. EHW and hydrogen water (water rich in molecular hydrogen) have been shown to have characteristics that scavenge reactive oxygen species (ROS) [
2]. EHW has also been shown to reduce intracellular ROS levels in the fibrosarcoma cell line, HT1080, when exposed to H
2O
2 as an oxidative stress agent, with stronger activity than hydrogen water containing the same concentration of molecular hydrogen [
2]. These EHW and hydrogen water properties have been verified by animal experiments to contribute to the alleviation of pathological conditions such as diabetes, arteriosclerosis, and neurodegenerative diseases, which are closely related to oxidative stress [
3,
4,
5]. A recent study also showed that in a rat model of sustained stress-loading, pre-treatment with EHW significantly suppressed the elevation of peroxides, prevented the decline in antioxidant capacity in the blood, and significantly suppressed the elevation of inflammatory marker, IL-1β, and stress response hormone, ACTH [
6]. In addition, a non-randomized, non-blinded, prospective observational study reported that hemodialysis using a dialysis solution containing high concentrations of hydrogen produced by electrolysis can reduce the mortality risk and cardiac and cerebrovascular complications in dialysis patients [
7]. EHW also significantly reduces the energy expenditure in triathletes during endurance exercise [
8].
The medical complications associated with alcohol consumption encompass a wide range of areas, among which is alcoholic liver disease (ALD). ALD includes alcoholic steatosis, alcoholic hepatitis, fibrosis, cirrhosis, and hepatocellular carcinoma [
9] and it is a relatively common, life-threatening disease. The liver is the main organ of alcohol metabolism in the human body. In the liver, alcohol is metabolized into highly toxic acetaldehyde, which is then broken down into non-toxic acetic acid. Persistent drinking of alcohol causes acetaldehyde to damage liver cells, leading to serious diseases such as alcoholic cirrhosis. Of note, alcohol (ethanol: CH
3CH
2OH, ethyl alcohol) causes poisoning of the central nervous system, but alcohol itself is not hepatotoxic. The biological response to ethanol is regulated by the processes of acetaldehyde production by alcohol dehydrogenase (ADH), the drug-metabolizing enzymes cytochrome P450 (CYP) 2E1 and catalase, and its metabolism to acetic acid by aldehyde dehydrogenase (ALDH). The mechanism of alcohol-induced hepatocellular damage is thought to involve the redox shift associated with ethanol metabolism, toxicity of acetaldehyde, increased oxidative stress due to reactive oxygen species (ROS) production, as well as microcirculatory disturbances and activation of the inflammation system. In addition, acetaldehyde is a highly reactive and unstable substance that binds to proteins and DNA to form adducts, resulting in glutathione depletion, lipid peroxidation, and mitochondrial damage [
10].
In the past, there have been many studies on ALD. To date, there is no FDA-approved nutritional therapy for ALD, and liver transplantation is the ultimate treatment option for patients with advanced alcoholic cirrhosis [
11]. Pre-drinking hydrogen water containing high concentrations of hydrogen has been reported to reduce the development of fatty liver (early lesions of ALD) in mice and the production of pro-inflammatory cytokines (such as IL6 and TNFα) in response to alcohol administration [
12]. It has also been shown that the addition of hydrogen to acetaldehyde solution decreases the aldehyde concentration [
13]. These results suggest that hydrogen has a detoxifying effect on acetaldehyde and may contribute to alleviating the pathology of alcoholic diseases. However, the mechanisms are largely unknown.
Although the antioxidant properties of EHW are well known, the effects of EHW on ethanol-induced hepatic cell damages have not been evaluated so far. In this study, we found that EHW suppressed cell death and ROS accumulation in the ethanol-treated hepatocyte cell line, HepG2, by decreasing intracellular acetaldehyde, a toxic substance produced by ethanol degradation. Analysis of the mechanism of action revealed that EHW inhibits the metabolism of ethanol to acetaldehyde by suppressing alcohol dehydrogenase. EHW also promotes the metabolism of acetaldehyde to acetic acid by activating aldehyde dehydrogenase (ALDH), which reduces intracellular toxic aldehyde levels in HepG2 cells. These functions were correlated with the concentration of molecular hydrogen in EHW, and were abolished by degassing treatment, suggesting that molecular hydrogen may contribute as a functional factor in the suppression of ethanol-induced hepatocellular damage.
2. Materials and Methods
2.1. Different Types of Water
EHW was obtained from a previously reported apparatus, TRIM ION GRACE (Nihon Trim Co., Osaka, Japan) [
6]. In brief, tap water is first passed through an activated charcoal filter for removal of bacteria and other microscopic impurities, then subject to electrolysis for enrichment in hydrogen. The apparatus was used to generate 4 kinds of EHW (LV1-4) by varying the electric current. Filtered water (FW) that was not subjected electrolysis was used as a control. Additionally, EHW was subjected to different treatments to obtain EHW with specific physical and chemical compositions; EHW with neutral pH, high concentration of dissolved hydrogen, and a small amount of dissolved oxygen were prepared [
3,
14,
15]. EHW (pH) was neutralized with HEPES buffer to adjust the pH. EHW (LV4) was autoclaved twice to generate autoclave water (AC). Hydrogen water (HW) with high dissolved hydrogen was obtained using a kit from TRIM SEVEN WATER (Nihon Trim). HW was prepared at the same hydrogen concentration as the EHW LV4. The concentration of dissolved hydrogen in fresh EHW was measured using a flow cell type hydrogen sensor (DH-35A, TOADKK, Tokyo, Japan). The dissolved hydrogen concentration of this study is as follows; LV1: 780–830 ppb, LV2: 850–880 ppb, LV3: 1060–1140 ppb, LV4: 1260–1350 ppb, and HW: 1300 ppb. The pH of FW and EHW (LV1-4) was measured using LAQUAact D-71 pH meter (HORIBA Advanced Techno, Co., Ltd., Kyoto, Japan), value of which is as follow; FW: 8.05 ± 0.02, LV1: 8.94 ± 0.04, LV2: 9.20 ± 0.04, LV3: 9.57 ± 0.12, LV4: 10.09 ± 0.02.
2.2. Cell Culture
HepG2 cells (RIKEN BRC), a human liver cancer cell line, were cultured in Dulbecco’s Modified Eagle Medium (DMEM) (Wako, 044–29765) containing 10% fetal bovine serum (FBS; Sigma-Aldrich, F7524) and 1% penicillin-streptomycin (Wako, 168–23191) at 37 °C in a 5% CO2 atmosphere. Experimental 5× DMEM medium was prepared by D-MEM powder (Wako, 049-33561) and ultrapure water from a Milli-Q synthesis system (Millipore, Tokyo, Japan), which was diluted with FW, EHW, EHW (pH), AC, or HW, to prepare the treatment medium (20%, 5× DMEM, 80%, FW, EHW, EHW (pH), AC, or HW).
2.3. Measurement of the Inhibitory Effect on Cell Proliferation
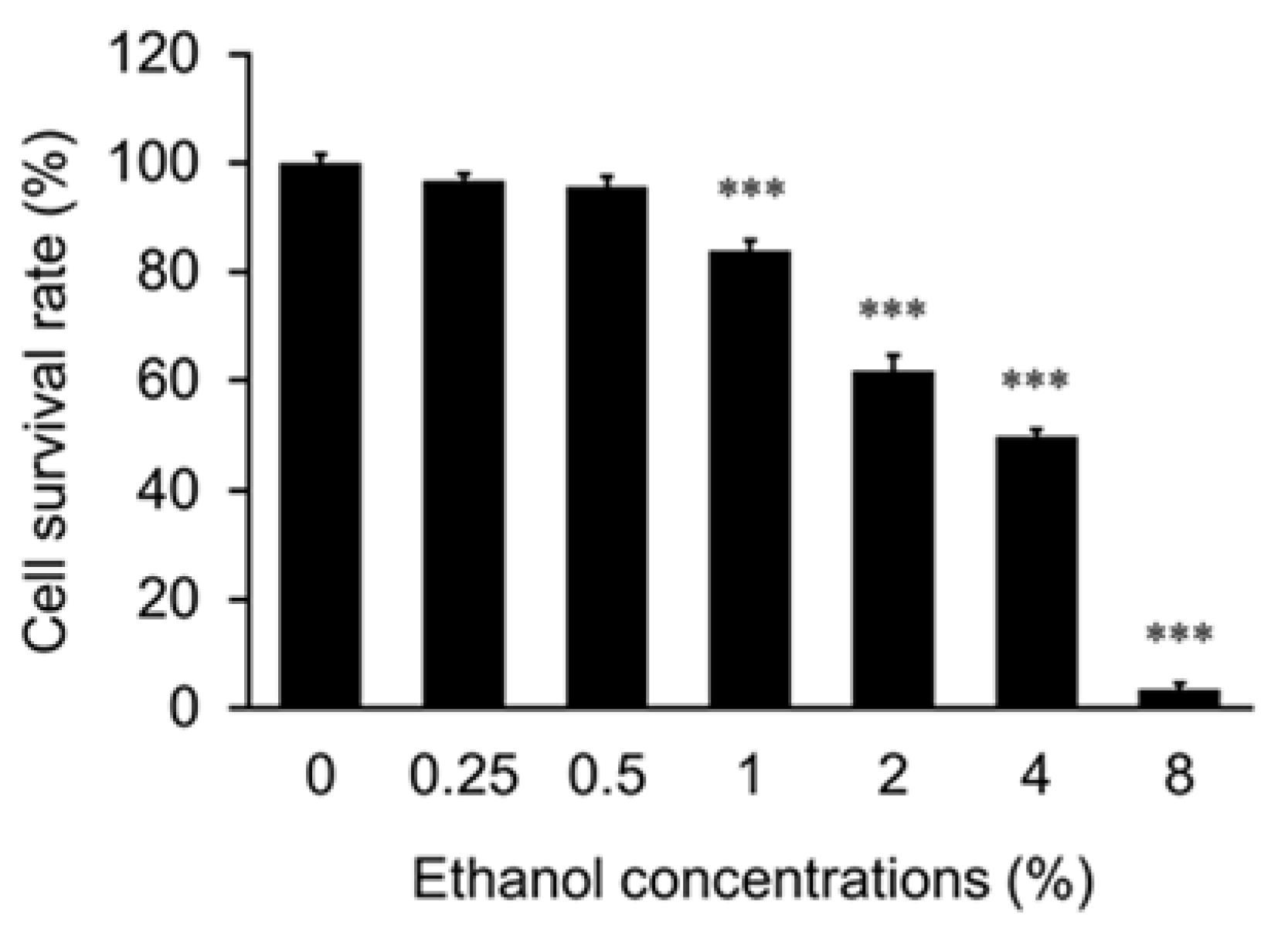
HepG2 cells were seeded in a 24-well plate at a density of 5.0 × 104 cells in DMEM and cultured for 24 h in the CO2 incubator. The cells were treated with medium containing FW, EHW, EHW (pH), AC, or HW spiked with ethanol or acetaldehyde. The cells were incubated for 24 h. Then, the cells were stained with trypan blue (Wako, 207–17081), and unstained cells were counted as alive under an inverted microscope, and the cell survival rate was calculated.
2.4. Measurement of Intracellular ROS
HepG2 cells were seeded in a 12-well plate at a density of 1.0 × 105 cells/well for 24 h, treated with ethanol or acetaldehyde in a medium prepared with FW, EHW, EHW (pH), AC or HW for 6 h. Then, the cells were treated with 5 μM intracellular ROS fluorescence detection reagent CM-H2DCFDA (Thermo Fisher Scientific, C6827) for 30 min. The fluorescence in the cells was visualized by fluorescence microscope (KEYENCE BZ-9000), and the fluorescence intensity was measured using Cellometer®Vision (Nexcelom Bioscience LLC) after harvest. FCS Express4 (De Novo software) was used for quantitative analysis.
2.5. Measurement of the Concentration of Ethanol and Acetaldehyde
Ethanol or acetaldehyde were added to FW, EHW (LV4), AC, or HW to prepare 4% ethanol and 1 mM acetaldehyde solutions in 50 mL tube, respectively. The solutions were stored at room temperature for 24 h. Then, the ethanol concentration was measured using the QuantiChrom™ Ethanol Assay Kit (BioAssay Systems, DIET-500), according to the manufacturer’s protocol. Briefly, 100 µL of these waters that were mixed with 4% ethanol and standard were added to each well at 96-well plate, followed by addition of 100 µL of Reagent A, colorimetric assay buffer. After incubation at room temperature for 8 min, the absorbance was determined by measuring at 570 nm with Multiskan™ FC (Thermo Scientific™, MA, USA). Additionally, the acetaldehyde concentration was measured using the EnzyChrom™ Acetaldehyde Assay Kit (BioAssay Systems, EACT-100) according to the manufacturer’s protocol. Briefly, 20 µL of these waters that were mixed with 1 mM acetaldehyde and standard were added to each well at 96-well plate, followed by addition of 80 µL of Warking Reagent, including Assay buffer, NAD/MTT solution, Enzyme A, and Enzyme B. After incubation at room temperature for 30 min, the absorbance was determined by measuring at 570 nm with Multiskan™ FC.
2.6. Measurement of the Ethanol Concentration in Culture Medium
HepG2 cells were seeded in a 6-well plate at a density of 2.0 × 10
5 cells/well for 24 h. Then, the cells were treated for 24 h with medium that was mixed with 4% ethanol and FW, EHW (LV4), AC, or HW. Ethanol concentration in the culture medium was measured using the QuantiChrom™ Ethanol Assay Kit (BioAssay Systems, DIET-500) according to the manufacturer’s instruction. Briefly, the culture medium was deproteinated by adding 1 volume of these medium to 2 volumes of 10% TCA, followed by centrifuge at 14,000 rpm for 5 min, and their supernatant was used for the assay. Colorimetric quantification was performed in the same way as in
Section 2.5.
2.7. Measurement of Acetaldehyde Concentration in Cells
HepG2 cells were seeded in a 10 cm dish at a density of 1.0 × 10
6 cells/well for 24 h. Cells were incubated for 24 h with medium that was mixed with 1 mM acetaldehyde and FW, EHW (LV4), AC, or HW. The intracellular acetaldehyde concentration was measured using the EnzyChrom™ Acetaldehyde Assay Kit (BioAssay Systems, EACT-100) according to the manufacturer’s instruction. Briefly, the cells were lysed and sonicated, followed by centrifuge at 12,000 rpm, for 5 min, and their supernatant was used for the assay. Colorimetric quantification was performed in the same way as in
Section 2.5.
2.8. Measurement of Cellular ADH Activity
HepG2 cells were seeded in a 6-well plate at a density of 2.0 × 10
5 cells/well for 24 h. Then, the cells were incubated for 24 h with medium that was mixed with 4% ethanol and FW, EHW (LV4), AC, or HW. The cellular ADH activity was measured using the Alcohol Dehydrogenase Activity Colorimetric Assay Kit (BioVision, K787-100) according to the manufacturer’s instruction. Briefly, the cells were lysed with ADH Assay Buffer, 50 µL of which were mixed with 100 µL of Reaction Mix, including ADH Assay Buffer, Developer, and Substrate, to each well at 96-well plate. The mix was incubated for 3 min at 37 °C, then the absorbance was measured at 450 nm with Multiskan™ FC, which was expressed as Abs
0min, and was incubated for another 60 min at 37 °C and the absorbance was measured at 450 nm again, which was expressed as Abs
60min. The ∆Abs (= Abs
60min − Abs
0min) was applied to the NADH standard curve to get X nmol of NADH generated by ADH during the reaction time (60 min). The ADH activity was calculated from the following equation:
Then, the cell lysate volume was converted to the number of cells and counted by trypan blue method, and the ADH activity was recalculated (µU/1.0 × 106 cells).
2.9. Measurement of Cellular ALDH Activity
HepG2 cells were seeded in a 6-well plate at a density of 2.0 × 10
5 cells/well for 24 h. Then, the cells were incubated for 24 h with medium that was mixed with 4% ethanol and FW, EHW (LV4), AC, or HW. The cellular ALDH activity was measured using the Aldehyde Dehydrogenase Activity Colorimetric Assay Kit (BioVision, K731-100) according to the manufacturer’s instruction. Briefly, the cells were lysed with ALDH Assay Buffer, 50 µL of which were mixed with 50 µL of ALDH Measurement, including ALDH Assay Buffer, ALDH Substrate Mix, and Acetaldehyde, to each well at 96-well plate. The mix was incubated for 5 min at room temperature, then the absorbance was measured at 450 nm with Multiskan™ FC, which was expressed as Abs
0min, and was incubated for another 60 min at room temperature and the absorbance was measured at 450 nm again, which was expressed as Abs
60min. The ∆Abs (= Abs
60min − Abs
0min) was applied to the NADH standard curve to get X nmol of NADH generated during the reaction time (60 min). The ALDH activity was calculated from the following equation:
Then, the cell lysate volume was converted to the number of cells and counted by trypan blue method, and the ALDH activity was recalculated (µU/1.0 × 106 cells).
2.10. Statistical Analysis
The data represent the mean ± SEM. Statistical analysis was performed by two-tailed Student’s t-test, or one-way analysis of variance (ANOVA), and then, Dunnett’s test and Tukey’s test were used as a multiple comparison test. A probability of p < 0.05 was considered as significant.
4. Discussion
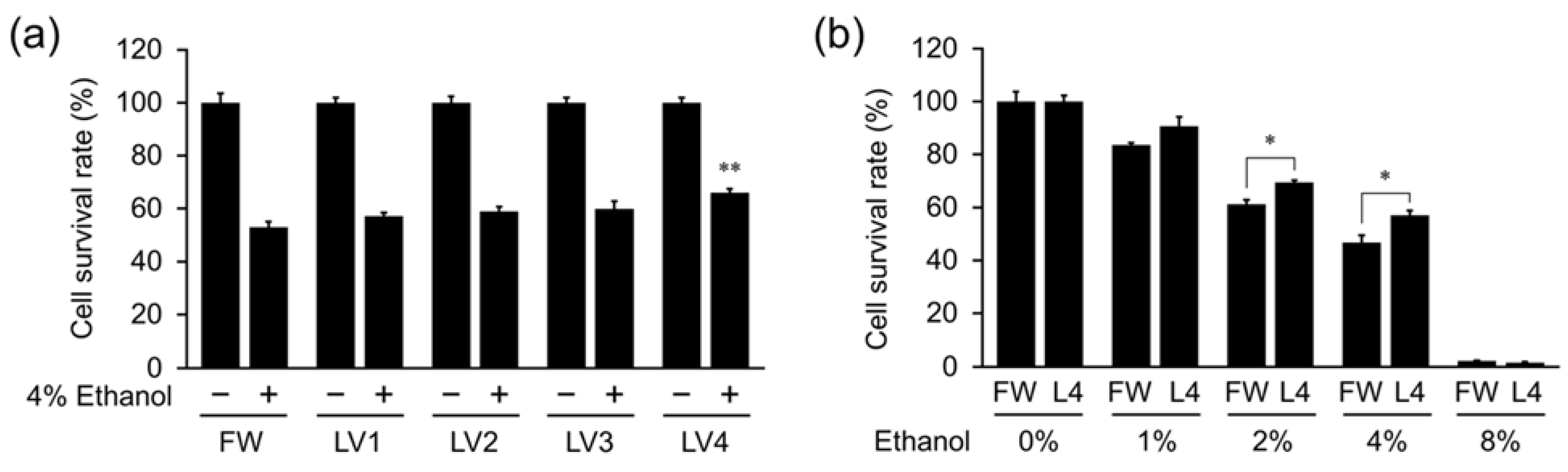
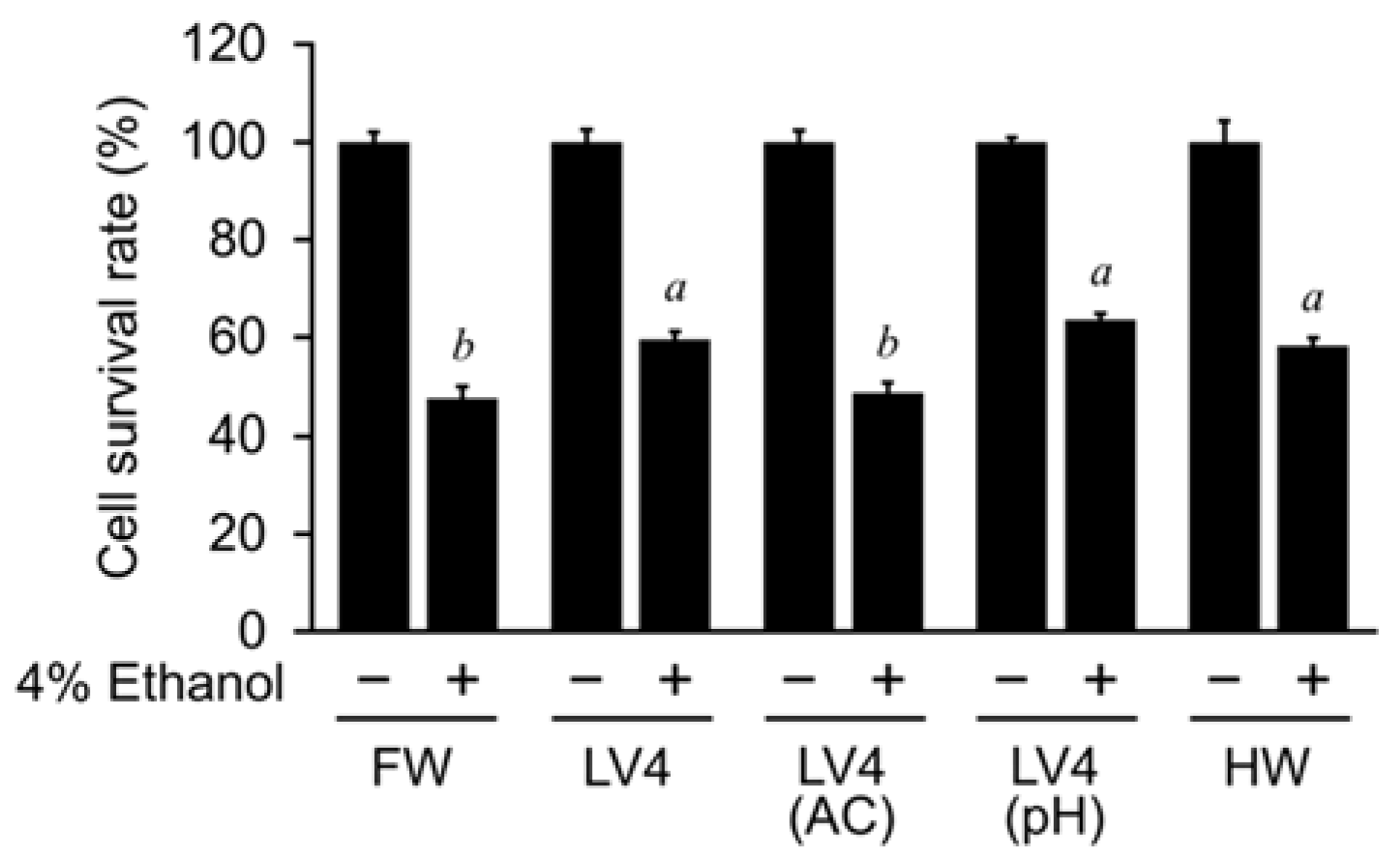
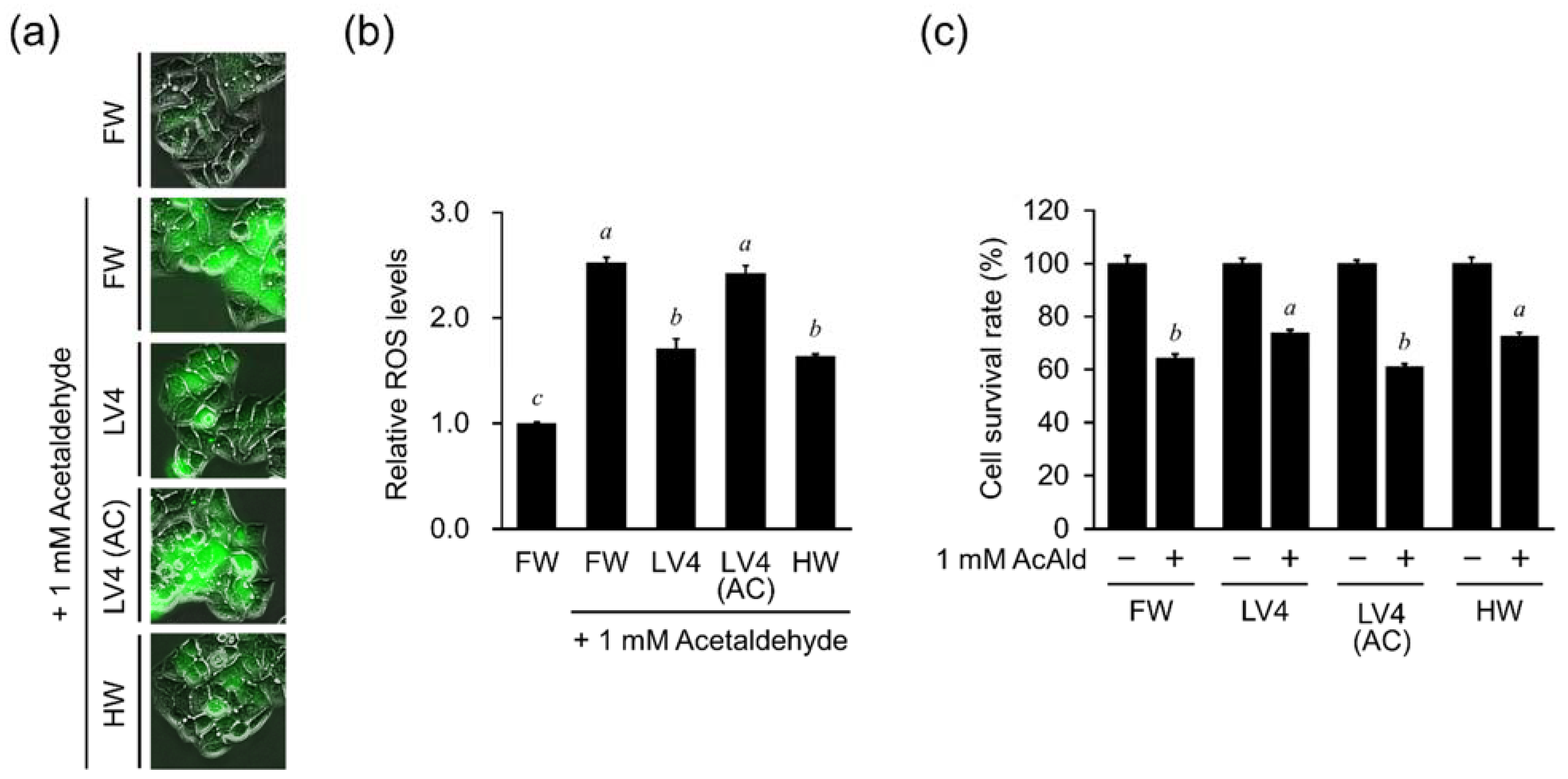
In this study, we analyzed the protective effect of EHW against ethanol-induced hepatocellular damage and its mechanism using the hepatocyte cell line HepG2. We found that EHW can inhibit the increase in intracellular ROS levels caused by ethanol and aldehyde treatment, and prevent cell death, which is related to the electrolytic intensity of EHW. We also revealed that EHW decreases the highly toxic acetaldehyde in ethanol treated cells by inhibiting the activity of ADH, which is responsible for the production of acetaldehyde, and increasing the activity of ALDH, which is responsible for the degradation of acetaldehyde.
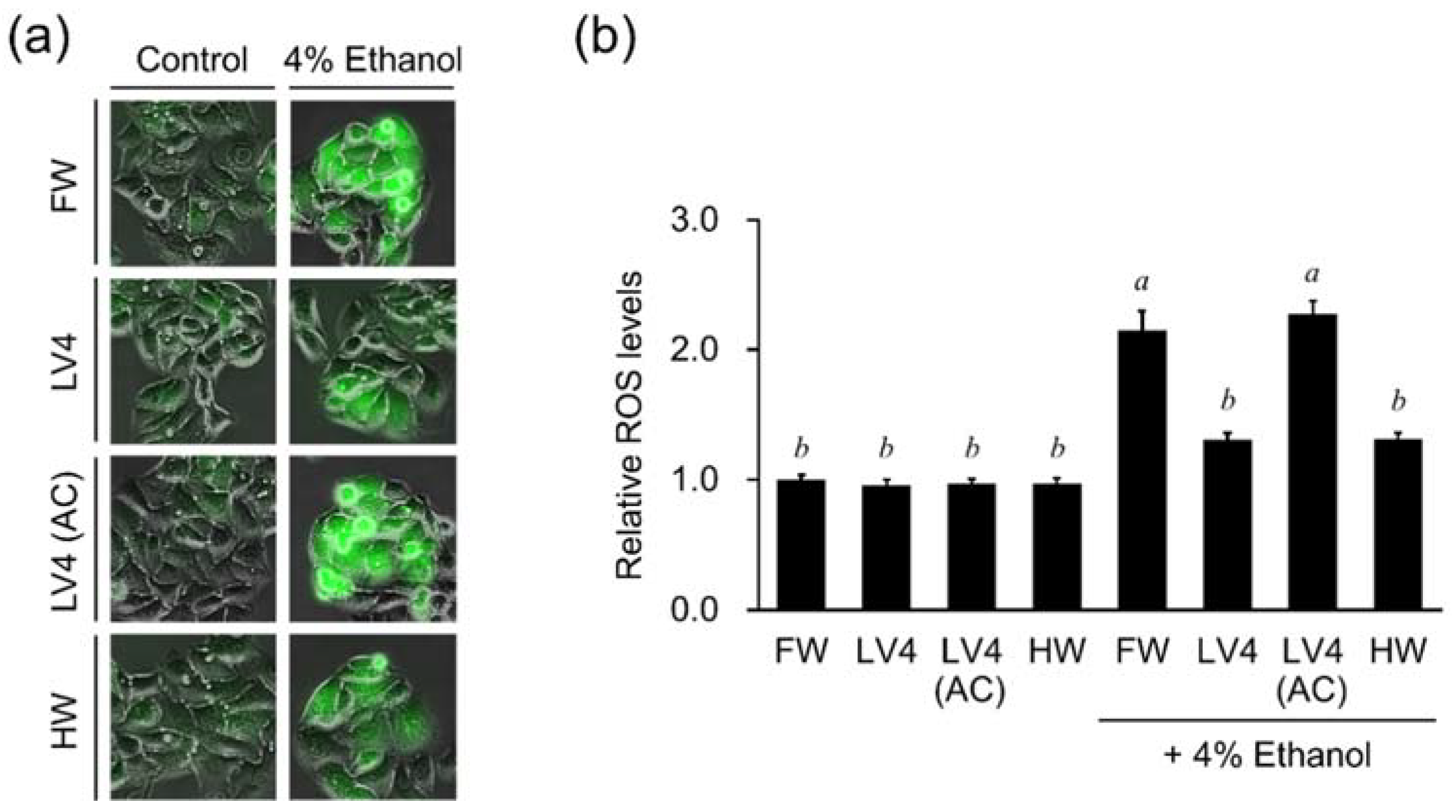
Unlike tap water, EHW has alkaline properties, including high concentrations of molecular hydrogen and trace amounts of platinum nanoparticles generated at the cathode by electrolysis of water. Previous reports have shown that EHW significantly inhibits the production of ROS in H
2O
2-treated HT1080 cells. This activity was found to be five times stronger than that of bubbled hydrogen water containing the same concentration of molecular hydrogen. In addition, about 60% of the activity of EHW remained even after degassing to remove hydrogen gas, speculating that a small amount of platinum nanocolloid in the electrolytic hydrogen water may contribute to the intracellular ROS scavenging ability by H
2O
2 treatment [
2]. Similarly, the production of ROS is increased in HepG2 cells treated with ethanol and aldehyde, which inhibited by treatment of the cells with EHW or hydrogen water (
Figure 4 and
Figure 5). However, the cytoprotective and ROS scavenging effects of EHW against ethanol and aldehyde were abolished by degassing treatment but not by neutralization (
Figure 3,
Figure 4 and
Figure 5). The same activity was observed in hydrogen rich water (
Figure 3,
Figure 4 and
Figure 5), suggesting that the cytoprotective ability is related to the large amount of dissolved hydrogen molecules in the EHW. Whereas, since EHW and hydrogen water are also characterized by low dissolved oxygen [
2], we cannot deny the possibility that low dissolved oxygen may have an effect on the increase in intracellular ROS and cytoprotective effects of ethanol and aldehydes. The production of ROS by ethanol and aldehydes occurs not only as a result of mitochondrial damage, but also as a major product or byproduct of various enzymatic reactions associated with alcohol degradation. In addition, it is known to induce disruption of the antioxidant system [
19], suggesting that H
2 molecules suppress ROS generation by acting on the intracellular ROS production mechanism caused by ethanol metabolism and aldehyde toxicity. On the other hand, since H
2O
2 is a precursor of hydroxyl radicals, components other than molecular hydrogen in EHW may act more strongly to scavenge intracellular reactive oxygen species caused by hydroxyl radicals [
20]. Alternatively, the fibrosarcoma cell line, HT1080, and the hepatocyte cell line, HepG2, may differ in their responses to components in EHW in the production and scavenging mechanism of ROS, depending on the cell type. Further elucidation of the mechanism of action of EHW on intracellular ROS scavenging activity will be necessary to clarify these differences.
Oxidative stress has been shown to be deeply involved in the development of chronic liver diseases, including ALD [
21]. The process of alcohol metabolism in cells causes the generation of ROS and also impairs the antioxidant system, resulting in oxidative stress that induces cellular damage [
22]. EHW and hydrogen water markedly inhibit alcohol- and aldehyde-induced increases in ROS, suggesting that suppression of ROS production by molecular hydrogen may function as one of the mechanisms of action for cytoprotection (
Figure 4 and
Figure 5). However, aldehydes can react with various macromolecules such as lipids, proteins, and nucleic acids, impairing their functions and inducing strong cytotoxicity [
23]. Thus, they may be considered a major factor in alcohol toxicity, affecting not only in the production and scavenging of ROS, but also in cellular homeostasis, producing a wide range of cytotoxic effects. In the present study, EHW and hydrogen water suppressed the production of intracellular aldehyde (
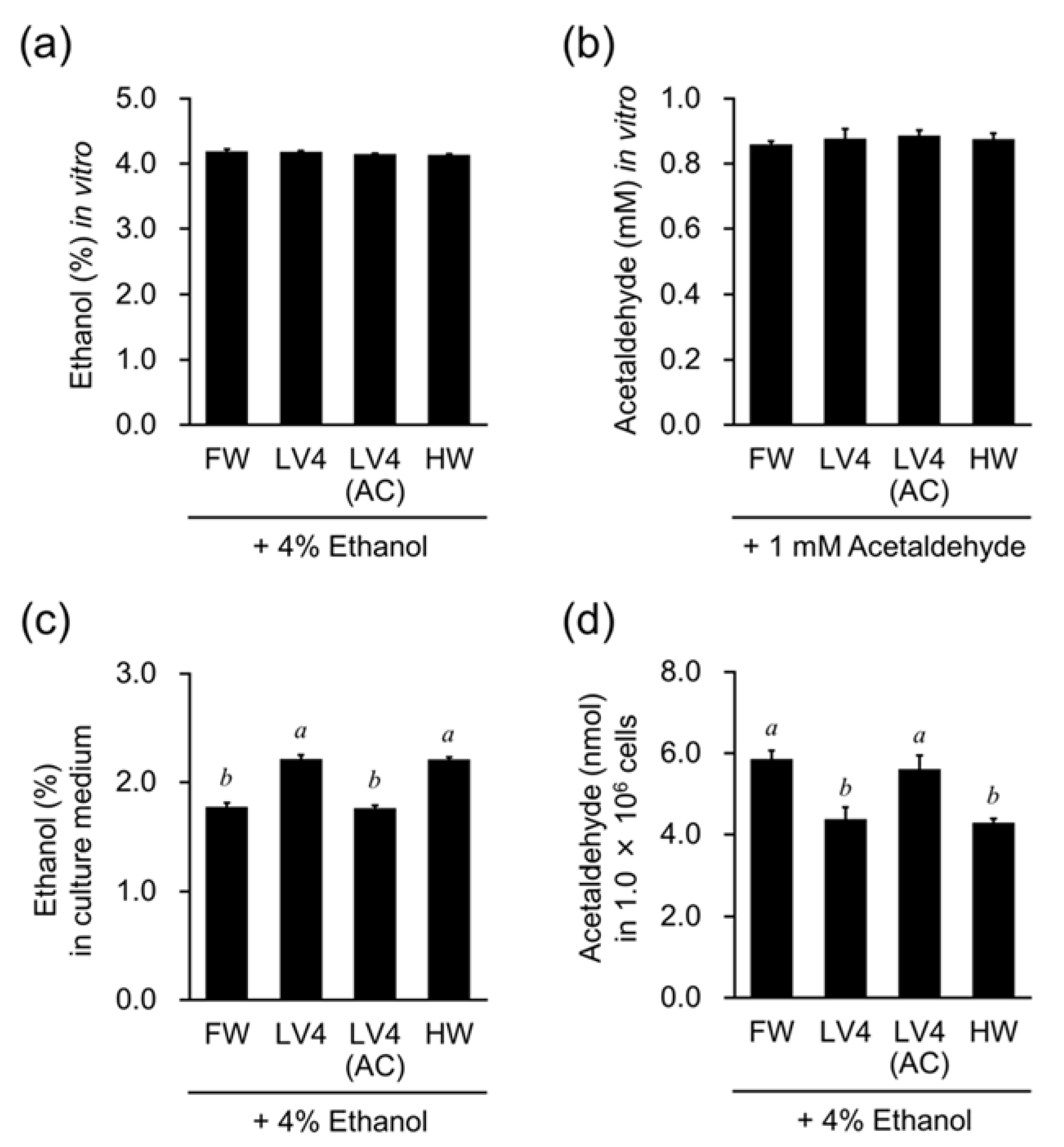
Figure 6d). We suspect that the decrease in intracellular aldehydes may play a greater role in the cytoprotective effect than the inhibition of ROS production. Microcluster, a hydrogen-absorbing microporous silica, has been reported to reduce acetaldehyde in distilled spirits [
13]. On the other hand, our in vitro analysis showed that the addition of ethanol or acetaldehyde to EHW or hydrogen water did not make any difference in their concentrations in the 24-h reaction (
Figure 6a,b). There was no significant difference in the concentration of aldehydes used compared to previous reports (30 or 50 ppm vs. 44 ppm in this experiment). Since the microcluster continuously generates hydrogen gas in the aldehyde solution, the aldehyde concentration may be reduced by the continuous reaction of fresh hydrogen with the aldehyde. In fact, EHW or hydrogen water suppressed activity of ADH, the enzyme related to ethanol metabolism in ethanol-treated cells (
Figure 7a), and increased the activity of ALDH, the enzyme responsible for the degradation of aldehydes (
Figure 7b), suggesting that the regulation of alcohol metabolism plays an important role in reducing the amount of intracellular aldehyde. Therefore, although EHW and hydrogen water inhibit ROS production and cell death in aldehyde-treated cells (
Figure 5), the degradation of aldehydes via activation of ALDH may contribute to these protective effects as a more fundamental mechanism of protection.
It is an important issue whether the inhibitory effect of molecular hydrogen on alcohol- and aldehyde-induced hepatocyte cytotoxicity, which was revealed in this study, can be observed in vivo. However, it has been reported that the development of fatty liver, which is an initial of alcoholic liver disease, and inflammatory reactions are suppressed in mice pretreated with hydrogen water [
12]. In the future, it will be important to examine the effects of hydrogen water on the activity of ADH and ALDH in the liver in this model. Interestingly, although ADH and ALDH are different enzymes involved in the production and degradation of aldehydes, EHW acts positively and negatively on their respective activities to reduce the amount of aldehydes. Notably, electrolytic hydrogen water and hydrogen water reduced ADH activity only in the ethanol-treated HepG2 cells (
Figure 7a), whereas ALDH activity was activated with or without ethanol treatment (
Figure 7b), suggesting that molecular hydrogen may regulate the activity of each enzyme through indirect and/or direct mechanisms. However, the mechanism has not been clarified in this study. It has been reported that not only the activity of ADH but also mRNA levels and protein levels are altered in ethanol feeding experiments in rats [
24]. In addition, an analysis using
Aldh2-/- mice, in which human ALDH2 was overexpressed, reported that ethanol administration decreased the amount of acetaldehyde in the blood [
25]. Whether EHW and hydrogen water directly regulate ADH and ALDH enzymatic activities, as well as gene expression and/or protein levels of ADH and ALDH, will need to be clarified in the future.
Moderate drinking of alcohol has been reported to be beneficial in reducing the risk of cardiovascular and all-cause mortality, which is widely accepted as a J-shaped curve association [
26]. However, recent reports indicate that the increased risk of other diseases, including cancer, has been reported even for low levels of alcohol intake, indicating that there are no safe levels of alcohol consumption for improving health [
27]. Thus, a two-sided effect of alcohol consumption on health has been reported, and this difference may be due to the balance between effects such as stress relief and platelet inhibition [
28,
29] by ethanol and cytotoxicity by aldehydes. The ability of EHW and hydrogen water to alter the balance between alcohol and toxic aldehyde levels by regulating ethanol metabolism may be a new preventive strategy to address health problems caused by alcohol consumption. In addition, it has been suggested that ALDH is involved in the metabolism of environmental toxins other than aldehydes [
10], and if EHW and hydrogen water can increase ALDH activity in the body, it may also be effective in mitigating the toxicity of environmental factors other than alcohol.