Fate of β-Carotene within Loaded Delivery Systems in Food: State of Knowledge
Abstract
1. Introduction
1.1. Oxidative Stress and Antioxidants
1.2. Mode of Action of β-Carotene against Oxidative Stress
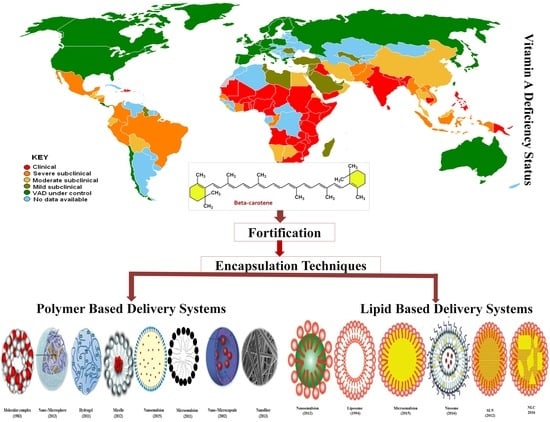
1.3. Challenges Associated with β-Carotene Food Fortification
2. Methodology
3. β-Carotene Metabolism
3.1. Release of β-Carotene from the Food Matrix
3.2. Mass Transfer to Oil Phase
3.3. Micelle Generation
3.4. Absorption
4. Bioavailability Assessment
Improving Bioavailability of β-Carotene by Encapsulation
5. Delivery Systems for β-Carotene
5.1. Polymer-Based Delivery Systems
5.1.1. Inclusion Complexes
5.1.2. Micro-/Nanospheres
5.1.3. Nanohydrogels
5.1.4. Micro-/Nanocapsules
5.1.5. Nanofibers
5.2. Lipid-Based Delivery Systems
5.2.1. Micelles
5.2.2. Micro/Nanoemulsions
5.2.3. Liposomes
5.2.4. Niosomes
5.2.5. Solid Lipid Nanoparticles
5.2.6. Nanostructured Lipid Carriers (NLCs)
6. Safety Compliance and Risks of β-Carotene Nanoparticles
7. Fate of β-Carotene-Loaded Nanodelivery Systems
8. Conclusions
9. Future Prospects and Research Gaps
- There is not a single report on the comparative assessment of the bioavailability of β-carotene EMS to the above mentioned nanodelivery systems. The data produced by devoted studies on bioavailability and health risks comparing various β-carotene-loaded delivery systems (LBDSs and PBDSs), particularly PBDSs, will aid in a better understanding and designing of suitable delivery systems for β-carotene.
- The nature of the carrier oil (fatty acid chain length and degree of saturation) can also affect the biological fate of the lipid-derived delivery systems [366]. Nevertheless, data are too scarce with respect to LBDSs to draw a firm conclusion.
- Although β-carotene-loaded delivery systems display a high bioavailability, other lipophilic compounds and related carotenoids may manipulate the bioavailability of β-carotene. More studies demonstrating the influences of lipophilic compounds present in the food matrix on the bioavailability of β-carotene-loaded delivery systems are needed. This will be aid in a better understanding in designing optimized delivery systems for β-carotene.
- Many researchers have argued that nanoparticles may enhance the bioavailability of β-carotene due to the transfer of intact nanoparticles across enterocytes. Nevertheless, no single study witnessed the penetration of food grade nanoparticles containing β-carotene across intestinal walls in the available literature.
- Most of the delivery systems are fabricated based on extrapolated in vitro and in vivo pharmaceutical data. This cannot be applied to food grade nanoparticles—in particular, polymer-based delivery systems.
- Certain ingredients (EDTA, chitosan, fatty acid, etc.) can manipulate the structure and integrity of the cell membrane. This is perhaps the least explored field and data generated on the effect of these ingredients on the cell membrane are necessary for better understanding and designing efficient β-carotene delivery systems.
- Various research studies displayed the improved permeability of cell membranes for certain kind of nanoparticles. Most of these are coupled with pharmaceutical formulations, containing to some extent certain nonfood grade materials. The same conclusion cannot be drawn for food grade nanoparticles. Thus, more devoted and rigorous investigations are needed to evaluate the impact of food grade nanoparticles on the penetration of cell membranes.
- There is ambiguity on interactions between GIT fluids and nanoparticles encapsulating β-carotene. It is sensible to debate how the bioavailability of β-carotene is influenced when it is encapsulated in available delivery systems.
- The perceived risks endorsed within the transfer of intact particles across the intestinal walls into the systemic circulation and buildup of particles or β-carotene in organs and the incidence of very high peak concentrations of β-carotene in the blood is poorly understood. Since reliable data signifying toxicity or risks are not present in the current literature, this should spark a debate on the various unknown factors.
- The role of digestive enzymes in the release of β-carotene from delivery system as well as on its bioavailability is not fully known. The assessment addressing the effects of enzymes individually or in arrays and their concentration on the bioavailability of β-carotene from delivery systems will aid in better knowledge for designing suitable delivery system.
- There is ambiguity regarding the kinetics of nanoparticle transfer from food matrix GIT fluid as well as from GIT fluid to enterocytes. More data need to be generated to understand the transfer kinetic of nanoparticles, which will result in a better understanding toward the realization of better delivery systems of β-carotene for food applications.
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| Cremophor | 40 PEG hydroxylated castor oil |
| EC | Ethylcellulose |
| GA | Gum arabic |
| LCT | Long-chain triglyceride |
| MCT | Medium-chain triacylglyceride |
| NaCMC | Sodium carboxymethyl cellulose |
| OSA | N-octenyl succinic anhydride |
| PC | Phosphatidylcholine |
| PEA | Phosphatidylethanolamine |
| PHBV | Poly(hydroxybutirate-co-hydroxyvalerate) |
| PLA | Poly(lactic) acid |
| PS | Phosphatidylserine |
| PVA | Polyvinyl alcohol |
| SA | Sodium alginate |
| SC | Sodium caseinate |
| SGF | Simulated gastric fluid |
| SIF | Simulated intestinal fluid |
| SPI | Soy protein isolate |
| SSF | Simulated saliva fluid |
| SSPS | Soybean-soluble polysaccharides |
| Tween | Polyoxyethylenesorbitan monolaurate |
| WPC | Whey protein concentrate |
| WPI | Whey protein isolate |
References
- McLaren, D.S.; Kraemer, K. Manual on Vitamin A Deficiency Disorders (VADD); Karger Medical and Scientific Publishers: Basel, Switzerland, 2012. [Google Scholar] [CrossRef]
- Unit, N.; World Health Organization. Global Prevalence of Vitamin A Deficiency; World Health Organization: Geneva, Switzerland, 1995. [Google Scholar]
- Rodriguez-Concepcion, M.; Avalos, J.; Bonet, M.L.; Boronat, A.; Gomez-Gomez, L.; Hornero-Mendez, D.; Limon, M.C.; Meléndez-Martínez, A.J.; Olmedilla-Alonso, B.; Palou, A. A global perspective on carotenoids: Metabolism, biotechnology, and benefits for nutrition and health. Prog. Lipid Res. 2018, 70, 62–93. [Google Scholar] [CrossRef]
- Shibata, M.; Sato, H.; Shimizu, T.; Shibata, S.; Toriumi, H.; Kuroi, T.; Ebine, T.; Iwashita, T.; Funakubo, M.; Akazawa, C. Differential cellular localization of antioxidant enzymes in the trigeminal ganglion. J. Headache Pain 2013, 14, P83. [Google Scholar] [CrossRef][Green Version]
- Adwas, A.A.; Elsayed, A.; Azab, A.E.; Quwaydir, F.A. Oxidative stress and antioxidant mechanisms in human body. J. Appl. Biotechnol. Bioeng. 2019, 6, 43–47. [Google Scholar] [CrossRef]
- Giustarini, D.; Dalle-Donne, I.; Tsikas, D.; Rossi, R. Oxidative stress and human diseases: Origin, link, measurement, mechanisms, and biomarkers. Crit. Rev. Clin. 2009, 46, 241–281. [Google Scholar] [CrossRef] [PubMed]
- Adam-Vizi, V.; Chinopoulos, C. Bioenergetics and the formation of mitochondrial reactive oxygen species. Trends Pharmacol. Sci. 2006, 27, 639–645. [Google Scholar] [CrossRef]
- Lenaz, G. Mitochondria and reactive oxygen species. Which role in physiology and pathology? Adv. Mitochondrial. Med. 2012, 93–136. [Google Scholar] [CrossRef]
- Auten, R.L.; Davis, J.M. Oxygen toxicity and reactive oxygen species: The devil is in the details. Pediatr. Res. 2009, 66, 121–127. [Google Scholar] [CrossRef]
- Sharifi-Rad, M.; Anil Kumar, N.V.; Zucca, P.; Varoni, E.M.; Dini, L.; Panzarini, E.; Rajkovic, J.; Tsouh Fokou, P.V.; Azzini, E.; Peluso, I. Lifestyle, oxidative stress, and antioxidants: Back and forth in the pathophysiology of chronic diseases. Front. Physiol. 2020, 11, 694. [Google Scholar] [CrossRef] [PubMed]
- Engwa, G.A.; Ferdinand, P.U.; Nwalo, F.N.; Unachukwu, M.N. Mechanism and health effects of heavy metal toxicity in humans. In Poisoning in the Modern World-New Tricks for an Old Dog? IntechOpen: London, UK, 2019; pp. 77–100. [Google Scholar] [CrossRef]
- Leni, Z.; Künzi, L.; Geiser, M. Air pollution causing oxidative stress. Curr. Opin. Toxicol. 2020, 20, 1–8. [Google Scholar] [CrossRef]
- Klawitter, J.; Klawitter, J.; Pennington, A.; Kirkpatrick, B.; Roda, G.; Kotecha, N.C.; Thurman, J.M.; Christians, U. Cyclophilin D knockout protects the mouse kidney against cyclosporin A-induced oxidative stress. Am. J. Physiol. Ren. Physiol. 2019, 317, F683–F694. [Google Scholar] [CrossRef]
- Wu, S.-C.; Yen, G.-C. Effects of cooking oil fumes on the genotoxicity and oxidative stress in human lung carcinoma (A-549) cells. Toxicol. In Vitro 2004, 18, 571–580. [Google Scholar] [CrossRef] [PubMed]
- Bardaweel, S.K.; Gul, M.; Alzweiri, M.; Ishaqat, A.; Alsalamat, H.A.; Bashatwah, R.M. Reactive oxygen species: The dual role in physiological and pathological conditions of the human body. Eurasian J. Med. 2018, 50, 193. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Wang, Q.; Zhu, J.; Xiao, Q.; Zhang, L. Reactive oxygen species: Key regulators in vascular health and diseases. Br. J. Pharmacol. 2018, 175, 1279–1292. [Google Scholar] [CrossRef]
- Franchina, D.G.; Dostert, C.; Brenner, D. Reactive oxygen species: Involvement in T cell signaling and metabolism. Trends Immunol. 2018, 39, 489–502. [Google Scholar] [CrossRef]
- Martina, A.; Jana, P.; Anna, S.; Tomas, B. Nitric oxide—Important messenger in human body. J. Mol. Integr. Physiol. 2012, 2, 98–106. [Google Scholar] [CrossRef]
- Hezel, M.P.; Weitzberg, E. The oral microbiome and nitric oxide homoeostasis. Oral Dis. 2015, 21, 7–16. [Google Scholar] [CrossRef]
- Belhadj Slimen, I.; Najar, T.; Ghram, A.; Dabbebi, H.; Ben Mrad, M.; Abdrabbah, M. Reactive oxygen species, heat stress and oxidative-induced mitochondrial damage. A review. Int. J. Hyperth. 2014, 30, 513–523. [Google Scholar] [CrossRef] [PubMed]
- Kaulmann, A.; Bohn, T. Carotenoids, inflammation, and oxidative stress—implications of cellular signaling pathways and relation to chronic disease prevention. Nutr. Res. 2014, 34, 907–929. [Google Scholar] [CrossRef]
- Kawata, A.; Murakami, Y.; Suzuki, S.; Fujisawa, S. Anti-inflammatory activity of β-carotene, lycopene and tri-n-butylborane, a scavenger of reactive oxygen species. In Vivo 2018, 32, 255–264. [Google Scholar] [CrossRef] [PubMed]
- Evans, R.M.; Mangelsdorf, D.J. Nuclear receptors, RXR, and the big bang. Cell 2014, 157, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Palczewski, G.; Widjaja-Adhi, M.A.; Amengual, J.; Golczak, M.; Von Lintig, J. Genetic dissection in a mouse model reveals interactions between carotenoids and lipid metabolism. J. Lipid Res. 2016, 57, 1684–1695. [Google Scholar] [CrossRef] [PubMed]
- Krinsky, N.I.; Johnson, E.J. Carotenoid actions and their relation to health and disease. Mol. Aspects Med. 2005, 26, 459–516. [Google Scholar] [CrossRef]
- Czarnewski, P.; Das, S.; Parigi, S.M.; Villablanca, E.J. Retinoic acid and its role in modulating intestinal innate immunity. Nutrients 2017, 9, 68. [Google Scholar] [CrossRef]
- Al Binali, H.A.H. Night blindness and ancient remedy. Heart Views 2014, 15, 136. [Google Scholar] [CrossRef] [PubMed]
- Biradar, S.T.; Ankitha, C.S.; Pasha, S.M. An Ayurvedic insight to keratoconus-A case report. J. Ayurveda Integr. Med. 2020, 5, 412–416. Available online: https://www.jaims.in/index.php/jaims/article/view/1312 (accessed on 26 February 2021).
- Balaji, S.; Roy, A. Beta-carotene--A versatile antioxidant in oral cancer: A review. Drug Invent. Today 2020, 13, 398–403. [Google Scholar]
- Miller, A.P.; Coronel, J.; Amengual, J. The role of β-carotene and vitamin A in atherogenesis: Evidences from preclinical and clinical studies. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2020, 158635. [Google Scholar] [CrossRef]
- Csepanyi, E.; Czompa, A.; Szabados-Furjesi, P.; Lekli, I.; Balla, J.; Balla, G.; Tosaki, A.; Bak, I. The effects of long-term, low-and high-dose beta-carotene treatment in Zucker diabetic fatty rats: The role of HO-1. Int. J. Mol. Sci. 2018, 19, 1132. [Google Scholar] [CrossRef]
- Stevens, G.A.; Bennett, J.E.; Hennocq, Q.; Lu, Y.; De-Regil, L.M.; Rogers, L.; Danaei, G.; Li, G.; White, R.A.; Flaxman, S.R. Trends and mortality effects of vitamin A deficiency in children in 138 low-income and middle-income countries between 1991 and 2013: A pooled analysis of population-based surveys. Lancet Glob. Health 2015, 3, e528–e536. [Google Scholar] [CrossRef]
- Imsic, M.; Winkler, S.; Tomkins, B.; Jones, R. Effect of storage and cooking on β-carotene isomers in carrots (Daucus carota L. cv. ‘Stefano’). J. Agric. Food Chem. 2010, 58, 5109–5113. [Google Scholar] [CrossRef]
- Knockaert, G.; Lemmens, L.; Van Buggenhout, S.; Hendrickx, M.; Van Loey, A. Changes in β-carotene bioaccessibility and concentration during processing of carrot puree. Food Chem. 2012, 133, 60–67. [Google Scholar] [CrossRef]
- Schieber, A.; Carle, R. Occurrence of carotenoid cis-isomers in food: Technological, analytical, and nutritional implications. Trends Food Sci. Technol. 2005, 16, 416–422. [Google Scholar] [CrossRef]
- Rodriguez-Amaya, D.B. Food Carotenoids: Chemistry, Biology and Technology; John Wiley & Sons: Hoboken, NJ, USA, 2015. [Google Scholar]
- Erdman, J.W., Jr.; Bierer, T.L.; Gugger, E.T. Absorption and transport of carotenoids. Ann. N. Y. Acad. Sci. 1993, 691, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Nagarajaiah, S.B.; Prakash, J. Nutritional composition, acceptability, and shelf stability of carrot pomace-incorporated cookies with special reference to total and β-carotene retention. Cogent Food Agric. 2015, 1, 1039886. [Google Scholar] [CrossRef]
- Kaya-Celiker, H.; Mallikarjunan, K. Better nutrients and therapeutics delivery in food through nanotechnology. Food Eng. Rev. 2012, 4, 114–123. [Google Scholar] [CrossRef]
- Gul, K.; Tak, A.; Singh, A.; Singh, P.; Yousuf, B.; Wani, A.A. Chemistry, encapsulation, and health benefits of β-carotene-A review. Cogent Food Agric. 2015, 1, 1018696. [Google Scholar] [CrossRef]
- Teng, Z.; Xu, R.; Wang, Q. Beta-lactoglobulin-based encapsulating systems as emerging bioavailability enhancers for nutraceuticals: A review. RSC Adv. 2015, 5, 35138–35154. [Google Scholar] [CrossRef]
- Chen, L.; Bai, G.; Yang, R.; Zang, J.; Zhou, T.; Zhao, G. Encapsulation of β-carotene within ferritin nanocages greatly increases its water-solubility and thermal stability. Food Chem. 2014, 149, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, F.J.; Albillos, S.M.; Casas-Sanz, E.; Cruz, Z.; García-Estrada, C.; García-Guerra, A.; García-Reverter, J.; García-Suárez, M.; Gatón, P.; González-Ferrero, C.; et al. Methods for the nanoencapsulation of β-carotene in the food sector. Trends Food Sci. Technol. 2013, 32, 73–83. [Google Scholar] [CrossRef]
- Zhu, F.; Du, B.; Xu, B. Anti-inflammatory effects of phytochemicals from fruits, vegetables, and food legumes: A review. Crit. Rev. Food Sci. Nutr. 2018, 58, 1260–1270. [Google Scholar] [CrossRef] [PubMed]
- Boon, C.S.; McClements, D.J.; Weiss, J.; Decker, E.A. Factors influencing the chemical stability of carotenoids in foods. Crit. Rev. Food Sci. Nutr. 2010, 50, 515–532. [Google Scholar] [CrossRef]
- Dos Santos, P.P.; de Aguiar Andrade, L.; Flôres, S.H.; de Oliveira Rios, A. Nanoencapsulation of carotenoids: A focus on different delivery systems and evaluation parameters. J. Food Sci. Technol. 2018, 55, 3851–3860. [Google Scholar] [CrossRef] [PubMed]
- Rehman, A.; Tong, Q.; Jafari, S.M.; Assadpour, E.; Shehzad, Q.; Aadil, R.M.; Iqbal, M.W.; Rashed, M.M.A.; Mushtaq, B.S.; Ashraf, W. Carotenoid-loaded nanocarriers: A comprehensive review. Adv. Colloid Interface Sci. 2020, 275, 102048. [Google Scholar] [CrossRef] [PubMed]
- Mao, L.; Wang, D.; Liu, F.; Gao, Y. Emulsion design for the delivery of β-carotene in complex food systems. Crit. Rev. Food Sci. Nutr. 2018, 58, 770–784. [Google Scholar] [CrossRef]
- Maurya, V.K.; Singh, J.; Ranjan, V.; Gothandam, K.M.; Bohn, T.; Pareek, S. Factors affecting the fate of β-carotene in the human gastrointestinal tract: A narrative review. Int. J. Vitam. Nutr. Res. 2020, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Parker, R.S. Absorption, metabolism, and transport of carotenoids. FASEB J. 1996, 10, 542–551. [Google Scholar] [CrossRef] [PubMed]
- Donhowe, E.G.; Kong, F. Beta-carotene: Digestion, microencapsulation, and in vitro bioavailability. Food Bioprocess Technol. 2014, 7, 338–354. [Google Scholar] [CrossRef]
- Van het Hof, K.H.; West, C.E.; Weststrate, J.A.; Hautvast, J.G.A.J. Dietary factors that affect the bioavailability of carotenoids. J. Nutr. 2000, 130, 503–506. [Google Scholar] [CrossRef] [PubMed]
- Iddir, M.; Degerli, C.; Dingeo, G.; Desmarchelier, C.; Schleeh, T.; Borel, P.; Larondelle, Y.; Bohn, T. Whey protein isolate modulates beta-carotene bioaccessibility depending on gastro-intestinal digestion conditions. Food Chem. 2019, 291, 157–166. [Google Scholar] [CrossRef]
- Schweiggert, R.M.; Mezger, D.; Schimpf, F.; Steingass, C.B.; Carle, R. Influence of chromoplast morphology on carotenoid bioaccessibility of carrot, mango, papaya, and tomato. Food Chem. 2012, 135, 2736–2742. [Google Scholar] [CrossRef]
- Hedren, E.; Diaz, V.; Svanberg, U. Estimation of carotenoid accessibility from carrots determined by an in vitro digestion method. Eur. J. Clin. Nutr. 2002, 56, 425–430. [Google Scholar] [CrossRef]
- Rock, C.L.; Lovalvo, J.L.; Emenhiser, C.; Ruffin, M.T.; Flatt, S.W.; Schwartz, S.J. Bioavailability of β-carotene is lower in raw than in processed carrots and spinach in women. J. Nutr. 1998, 128, 913–916. [Google Scholar] [CrossRef] [PubMed]
- Edwards, A.J.; Nguyen, C.H.; You, C.-S.; Swanson, J.E.; Emenhiser, C.; Parker, R.S. α-and β-Carotene from a commercial carrot puree are more bioavailable to humans than from boiled-mashed carrots, as determined using an extrinsic stable isotope reference method. J. Nutr. 2002, 132, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Hornero-Méndez, D.; Mínguez-Mosquera, M.I. Bioaccessibility of carotenes from carrots: Effect of cooking and addition of oil. Innov. Food Sci. Emerg. Technol. 2007, 8, 407–412. [Google Scholar] [CrossRef]
- Hof, K.H.V.H.; de Boer, B.C.; Tijburg, L.B.; Lucius, B.R.; Zijp, I.; West, C.E.; Hautvast, J.G.; Weststrate, J.A. Carotenoid bioavailability in humans from tomatoes processed in different ways determined from the carotenoid response in the triglyceride-rich lipoprotein fraction of plasma after a single consumption and in plasma after four days of consumption. J. Nutr. 2000, 130, 1189–1196. [Google Scholar] [CrossRef] [PubMed]
- Lemmens, L.; Van Buggenhout, S.; Van Loey, A.M.; Hendrickx, M.E. Particle size reduction leading to cell wall rupture is more important for the β-carotene bioaccessibility of raw compared to thermally processed carrots. J. Agric. Food Chem. 2010, 58, 12769–12776. [Google Scholar] [CrossRef]
- Yonekura, L.; Nagao, A. Intestinal absorption of dietary carotenoids. Mol. Nutr. Food Res. 2007, 51, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Faulks, R.M.; Southon, S. Challenges to understanding and measuring carotenoid bioavailability. Biochim. Biophys. Acta Mol. Basis Dis. 2005, 1740, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Rich, G.T.; Faulks, R.M.; Wickham, M.S.J.; Fillery-Travis, A. Solubilization of carotenoids from carrot juice and spinach in lipid phases: II. Modeling the duodenal environment. Lipids 2003, 38, 947–956. [Google Scholar] [CrossRef] [PubMed]
- Rich, G.T.; Fillery-Travis, A.; Parker, M.L. Low pH enhances the transfer of carotene from carrot juice to olive oil. Lipids 1998, 33, 985–992. [Google Scholar] [CrossRef] [PubMed]
- Iddir, M.; Dingeo, G.; Yaruro, J.F.P.; Hammaz, F.; Borel, P.; Schleeh, T.; Desmarchelier, C.; Larondelle, Y.; Bohn, T. Influence of soy and whey protein, gelatin and sodium caseinate on carotenoid bioaccessibility. Food Funct. 2020, 11, 5446–5459. [Google Scholar] [CrossRef] [PubMed]
- Bohn, T. Bioavailability of non-provitamin A carotenoids. Curr. Nutr. Food Sci. 2008, 4, 240–258. [Google Scholar] [CrossRef]
- Yeum, K.-J.; Russell, R.M. Carotenoid bioavailability and bioconversion. Annu. Rev. Nutr. 2002, 22, 483–504. [Google Scholar] [CrossRef] [PubMed]
- Fernández-García, E.; Carvajal-Lérida, I.; Jarén-Galán, M.; Garrido-Fernández, J.; Pérez-Gálvez, A.; Hornero-Méndez, D. Carotenoids bioavailability from foods: From plant pigments to efficient biological activities. Food Res. Int. 2012, 46, 438–450. [Google Scholar] [CrossRef]
- Roodenburg, A.J.C.; Leenen, R.; van het Hof, K.H.; Weststrate, J.A.; Tijburg, L.B.M. Amount of fat in the diet affects bioavailability of lutein esters but not of α-carotene, β-carotene, and vitamin E in humans. Am. J. Clin. Nutr. 2000, 71, 1187–1193. [Google Scholar] [CrossRef]
- Castenmiller, J.J.M.; West, C.E.; Linssen, J.P.H.; van het Hof, K.H.; Voragen, A.G.J. The food matrix of spinach is a limiting factor in determining the bioavailability of β-carotene and to a lesser extent of lutein in humans. J. Nutr. 1999, 129, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Hollander, D.; Ruble, P.E., Jr. beta-carotene intestinal absorption: Bile, fatty acid, pH, and flow rate effects on transport. Am. J. Physiol. Endoc. Metab. 1978, 235, E686. [Google Scholar] [CrossRef] [PubMed]
- Corte-Real, J.; Iddir, M.; Soukoulis, C.; Richling, E.; Hoffmann, L.; Bohn, T. Effect of divalent minerals on the bioaccessibility of pure carotenoids and on physical properties of gastro-intestinal fluids. Food Chem. 2016, 197, 546–553. [Google Scholar] [CrossRef]
- Corte-Real, J.; Bertucci, M.; Soukoulis, C.; Desmarchelier, C.; Borel, P.; Richling, E.; Hoffmann, L.; Bohn, T. Negative effects of divalent mineral cations on the bioaccessibility of carotenoids from plant food matrices and related physical properties of gastro-intestinal fluids. Food Funct. 2017, 8, 1008–1019. [Google Scholar] [CrossRef]
- Prince, M.R.; Frisoli, J.K. Beta-carotene accumulation in serum and skin. Am. J. Clin. Nutr. 1993, 57, 175–181. [Google Scholar] [CrossRef]
- Unlu, N.Z.; Bohn, T.; Clinton, S.K.; Schwartz, S.J. Carotenoid absorption from salad and salsa by humans is enhanced by the addition of avocado or avocado oil. J. Nutr. 2005, 135, 431–436. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, J.R.; Fitch, M.D.; Mortensen, P.B.; Fleming, S.E. In vivo absorption of medium-chain fatty acids by the rat colon exceeds that of short-chain fatty acids. Gastroenterology 2001, 120, 1152–1161. [Google Scholar] [CrossRef] [PubMed]
- Huo, T.; Ferruzzi, M.G.; Schwartz, S.J.; Failla, M.L. Impact of fatty acyl composition and quantity of triglycerides on bioaccessibility of dietary carotenoids. J. Agric. Food Chem. 2007, 55, 8950–8957. [Google Scholar] [CrossRef]
- Yonekura, L.; Nagao, A. Soluble fibers inhibit carotenoid micellization in vitro and uptake by Caco-2 cells. Biosci. Biotechnol. Biochem. 2009, 73, 196–199. [Google Scholar] [CrossRef] [PubMed]
- Rock, C.L.; Swendseid, M.E. Plasma β-carotene response in humans after meals supplemented with dietary pectin. Am. J. Clin. Nutr. 1992, 55, 96–99. [Google Scholar] [CrossRef]
- Riedl, J.; Linseisen, J.; Hoffmann, J.R.; Wolfram, G.N. Some dietary fibers reduce the absorption of carotenoids in women. J. Nutr. 1999, 129, 2170–2176. [Google Scholar] [CrossRef]
- Van Bennekum, A.; Werder, M.; Thuahnai, S.T.; Han, C.-H.; Duong, P.; Williams, D.L.; Wettstein, P.; Schulthess, G.; Phillips, M.C.; Hauser, H. Class B scavenger receptor-mediated intestinal absorption of dietary β-carotene and cholesterol. Biochemistry 2005, 44, 4517–4525. [Google Scholar] [CrossRef]
- Cervantes-Paz, B.; de Jesús Ornelas-Paz, J.; Ruiz-Cruz, S.; Rios-Velasco, C.; Ibarra-Junquera, V.; Yahia, E.M.; Gardea-Béjar, A.A. Effects of pectin on lipid digestion and possible implications for carotenoid bioavailability during pre-absorptive stages: A review. Food Res. Int. 2017, 99, 917–927. [Google Scholar] [CrossRef]
- Bohn, T.; Desmarchelier, C.; Dragsted, L.O.; Nielsen, C.S.; Stahl, W.; Ruhl, R.; Keijer, J.; Borel, P. Host-related factors explaining interindividual variability of carotenoid bioavailability and tissue concentrations in humans. Mol. Nutr. Food Res. 2017, 61, 1600685. [Google Scholar] [CrossRef]
- Reboul, E. Absorption of vitamin A and carotenoids by the enterocyte: Focus on transport proteins. Nutrients 2013, 5, 3563. [Google Scholar] [CrossRef]
- Böhm, V.; Lietz, G.; Olmedilla-Alonso, B.; Phelan, D.; Reboul, E.; Bánati, D.; Borel, P.; Corte-Real, J.; de Lera, A.R.; Desmarchelier, C.; et al. From carotenoid intake to carotenoid blood and tissue concentrations—Implications for dietary intake recommendations. Nutr. Rev. 2020, 1–30. [Google Scholar] [CrossRef]
- Mapelli-Brahm, P.; Margier, M.; Desmarchelier, C.; Halimi, C.; Nowicki, M.; Borel, P.; Meléndez-Martínez, A.J.; Reboul, E. Comparison of the bioavailability and intestinal absorption sites of phytoene, phytofluene, lycopene and β-carotene. Food Chem. 2019, 300, 125232. [Google Scholar] [CrossRef] [PubMed]
- Roman, M.J.; Burri, B.J.; Singh, R.P. Release and bioaccessibility of β-carotene from fortified almond butter during in vitro digestion. J. Agric. Food Chem. 2012, 60, 9659–9666. [Google Scholar] [CrossRef] [PubMed]
- Garrett, D.A.; Failla, M.L.; Sarama, R.J. Development of an in vitro digestion model for estimating the bioavailability of carotenoids from meals. J. Agric. Food Chem. 1999, 47, 4301–4309. [Google Scholar] [CrossRef] [PubMed]
- Minekus, M.; Alminger, M.; Alvito, P.; Ballance, S.; Bohn, T.; Bourlieu, C.; Carriere, F.; Boutrou, R.; Corredig, M.; Dupont, D. A standardised static in vitro digestion method suitable for food–an international consensus. Food Funct. 2014, 5, 1113–1124. [Google Scholar] [CrossRef] [PubMed]
- Brodkorb, A.; Egger, L.; Alminger, M.; Alvito, P.; Assunção, R.; Ballance, S.; Bohn, T.; Bourlieu-Lacanal, C.; Boutrou, R.; Carrière, F. INFOGEST static in vitro simulation of gastrointestinal food digestion. Nat. Protoc. 2019, 14, 991–1014. [Google Scholar] [CrossRef] [PubMed]
- Rao, J.; Decker, E.A.; Xiao, H.; McClements, D.J. Nutraceutical nanoemulsions: Influence of carrier oil composition (digestible versus indigestible oil) on β-carotene bioavailability. J. Sci. Food Agric. 2013, 93, 3175–3183. [Google Scholar] [CrossRef] [PubMed]
- Qian, C.; Decker, E.A.; Xiao, H.; McClements, D.J. Nanoemulsion delivery systems: Influence of carrier oil on β-carotene bioaccessibility. Food Chem. 2012, 135, 1440–1447. [Google Scholar] [CrossRef]
- Grune, T.; Lietz, G.; Palou, A.; Ross, A.C.; Stahl, W.; Tang, G.; Thurnham, D.; Yin, S.A.; Biesalski, H.K. β-Carotene is an important vitamin A source for humans. J. Nutr. 2010, 140, 2268S–2285S. [Google Scholar] [CrossRef]
- Fiedor, J.; Burda, K. Potential role of carotenoids as antioxidants in human health and disease. Nutrients 2014, 6, 466. [Google Scholar] [CrossRef] [PubMed]
- Borba, C.M.; Tavares, M.N.; Macedo, L.P.; Araújo, G.S.; Furlong, E.B.; Dora, C.L.; Burkert, J.F.M. Physical and chemical stability of β-carotene nanoemulsions during storage and thermal process. Food Res. Int. 2019, 121, 229–237. [Google Scholar] [CrossRef] [PubMed]
- Borel, P.; Desmarchelier, C. Genetic variations associated with vitamin A status and vitamin A bioavailability. Nutrients 2017, 9, 246. [Google Scholar] [CrossRef] [PubMed]
- Maurya, V.K.; Aggarwal, M. Factors influencing the absorption of vitamin D in GIT: An overview. J. Food Sci. Technol. 2017, 54, 3753–3765. [Google Scholar] [CrossRef] [PubMed]
- Aguilar-Pérez, K.M.; Avilés-Castrillo, J.I.; Medina, D.I.; Parra-Saldivar, R.; Iqbal, H. Insight into nanoliposomes as smart nanocarriers for greening the twenty-first century biomedical settings. Front. Bioeng. Biotechnol. 2020, 8, 1441. [Google Scholar] [CrossRef] [PubMed]
- Soukoulis, C.; Bohn, T. A comprehensive overview on the micro- and nano-technological encapsulation advances for enhancing the chemical stability and bioavailability of carotenoids. Crit. Rev. Food Sci. Nutr. 2018, 58, 1–36. [Google Scholar] [CrossRef]
- Maurya, V.K.; Aggarwal, M. Enhancing bio-availability of vitamin D by nano-engineered based delivery systems-An overview. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 340–353. [Google Scholar] [CrossRef]
- Maurya, V.K.; Bashir, K.; Aggarwal, M. Vitamin D microencapsulation and fortification: Trends and technologies. J. Steroid Biochem. Mol. Biol. 2020, 196, 105489. [Google Scholar] [CrossRef] [PubMed]
- Rostamabadi, H.; Falsafi, S.R.; Jafari, S.M. Nanoencapsulation of carotenoids within lipid-based nanocarriers. J. Control. Release 2019, 298, 38–67. [Google Scholar] [CrossRef] [PubMed]
- Maurya, V.K.; Aggarwal, M.; Ranjan, V.; Gothandam, K.M. Improving bioavailability of vitamin A in food by encapsulation: An update. In Nanoscience in Medicine Vol. 1; Springer: Cham, Switzerland, 2020; pp. 117–145. [Google Scholar] [CrossRef]
- Matencio, A.; Navarro-Orcajada, S.; García-Carmona, F.; López-Nicolás, J.M. Applications of cyclodextrins in food science. A review. Trends Food Sci. Technol. 2020, 104, 132–143. [Google Scholar] [CrossRef]
- Crini, G.; Fourmentin, S.; Fenyvesi, É.; Torri, G.; Fourmentin, M.; Morin-Crini, N. Fundamentals and applications of cyclodextrins. In Cyclodextrin Fundamentals, Reactivity and Analysis; Springer: Cham, Switzerland, 2018; pp. 1–55. [Google Scholar] [CrossRef]
- Arima, H.; Motoyama, K.; Higashi, T. Potential use of cyclodextrins as drug carriers and active pharmaceutical ingredients. Chem. Pharm. Bull. 2017, 65, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Adeoye, O.; Cabral-Marques, H. Cyclodextrin nanosystems in oral drug delivery: A mini review. Int. J. Pharm. 2017, 531, 521–531. [Google Scholar] [CrossRef]
- Chilajwar, S.V.; Pednekar, P.P.; Jadhav, K.R.; Gupta, G.J.; Kadam, V.J. Cyclodextrin-based nanosponges: A propitious platform for enhancing drug delivery. Expert Opin. Drug Deliv. 2014, 11, 111–120. [Google Scholar] [CrossRef]
- Shafaei, Z.; Ghalandari, B.; Vaseghi, A.; Divsalar, A.; Haertlé, T.; Saboury, A.A.; Sawyer, L. β-Lactoglobulin: An efficient nanocarrier for advanced delivery systems. Nanomed. NBM 2017, 13, 1685–1692. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Ma, C.; Gao, Y.; McClements, D.J. Food-grade covalent complexes and their application as nutraceutical delivery systems: A review. Compr. Rev. Food Sci. 2017, 16, 76–95. [Google Scholar] [CrossRef]
- Chanphai, P.; Vesper, A.; Bariyanga, J.; Bérubé, G.; Tajmir-Riahi, H. Review on the delivery of steroids by carrier proteins. J. Photochem. Photobiol. B Biol. 2016, 161, 184–191. [Google Scholar] [CrossRef]
- Astray, G.; Mejuto, J.C.; Simal-Gandara, J. Latest developments in the application of cyclodextrin host-guest complexes in beverage technology processes. Food Hydrocoll. 2020, 106, 105882. [Google Scholar] [CrossRef]
- Zaibunnisa, A.H.; Aini Marhanna, M.N.A.; AinunAtirah, M. Characterisation and solubility study of γ-cyclodextrin and beta-carotene complex. Int. Food Res. J. 2011, 18, 1061–1065. [Google Scholar]
- Loftsson, T.; Brewster, M.E.; Masson, M. Role of cyclodextrins in improving oral drug delivery. Am. J. Drug Deliv. 2004, 2, 261–275. [Google Scholar] [CrossRef]
- Ozcelik, B.; Karadag, A.; Ersen, S. Bioencapsulation of beta-carotene in three different methods. In Proceedings of the XVIIth International Conference on Bioencapsulation, Groningen, The Netherlands, 24–26 September 2009; pp. 24–26. [Google Scholar] [CrossRef]
- Letona, C.A.M.; Park, C.S.; Kim, Y.R. Amylosucrase-mediated β-carotene encapsulation in amylose microparticles. Biotechnol. Prog. 2017, 33, 1640–1646. [Google Scholar] [CrossRef] [PubMed]
- De Freitas Zômpero, R.H.; López-Rubio, A.; de Pinho, S.C.; Lagaron, J.M.; de la Torre, L.G. Hybrid encapsulation structures based on β-carotene-loaded nanoliposomes within electrospun fibers. Colloids Surf. B 2015, 134, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Liebler, D.C.; Stratton, S.P.; Kaysen, K.L. Antioxidant actions of β-carotene in liposomal and microsomal membranes: Role of carotenoid-membrane incorporation and α-tocopherol. Arch. Biochem. Biophys. 1997, 338, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, J.M.P.; Toniazzo, T.; Cavalcanti, L.P.; Moraes, I.C.F.; Oliveira, C.L.P.; Pinho, S.C. Physico-chemical stability and structural characterization of thickened multilamellar beta-carotene-loaded liposome dispersions produced using a proliposome method. Colloid Polym. Sci. 2015, 293, 2171–2179. [Google Scholar] [CrossRef]
- Bashiri, S.; Ghanbarzadeh, B.; Hamishehkar, H.; Dehghannia, J. β-carotene loaded nanoliposome: Effects of gama–oryzanol on particle size stability and encapsulation. 2015, 4, pp. 365–382. Available online: https://www.sid.ir/en/journal/ViewPaper.aspx?ID=502779 (accessed on 25 December 2020).
- Schwartz, J.; Flynn, E.; Trickier, D.; Shklar, G. Directed lysis of experimental cancer by β-carotene in liposomes. Nutr. Cancer 1991, 16, 107–124. [Google Scholar] [CrossRef]
- Silva, C.R.; Moraes, M.; Carvalho, J.M.P.; Pinho, S.C. Characterization of Spray-Dried Phospholipid Particles for the Production of Beta-Carotene-Loaded Liposomes. 2011. Available online: https://www.semanticscholar.org/paper/Characterization-of-spray-dried-phospholipid-for-of-Silva-Moraes/a6bf4d0de1b7a9ec75b887a8d44b71480199a5ca (accessed on 25 December 2020).
- Tan, C.; Zhang, Y.; Abbas, S.; Feng, B.; Zhang, X.; Xia, S. Modulation of the carotenoid bioaccessibility through liposomal encapsulation. Colloids Surf. B 2014, 123, 692–700. [Google Scholar] [CrossRef]
- Palozza, P.; Muzzalupo, R.; Trombino, S.; Valdannini, A.; Picci, N. Solubilization and stabilization of β-carotene in niosomes: Delivery to cultured cells. Chem. Phys. Lipids 2006, 139, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Nik, A.M.; Langmaid, S.; Wright, A.J. Nonionic surfactant and interfacial structure impact crystallinity and stability of β-carotene loaded lipid nanodispersions. J. Agric. Food Chem. 2012, 60, 4126–4135. [Google Scholar] [CrossRef] [PubMed]
- Yi, J.; Lam, T.I.; Yokoyama, W.; Cheng, L.W.; Zhong, F. Cellular uptake of β-carotene from protein stabilized solid lipid nanoparticles prepared by homogenization–evaporation method. J. Agric. Food Chem. 2014, 62, 1096–1104. [Google Scholar] [CrossRef]
- Helgason, T.; Awad, T.S.; Kristbergsson, K.; Decker, E.A.; McClements, D.J.; Weiss, J. Impact of surfactant properties on oxidative stability of β-carotene encapsulated within solid lipid nanoparticles. J. Agric. Food Chem. 2009, 57, 8033–8040. [Google Scholar] [CrossRef] [PubMed]
- Mehrad, B.; Ravanfar, R.; Licker, J.; Regenstein, J.M.; Abbaspourrad, A. Enhancing the physicochemical stability of β-carotene solid lipid nanoparticle (SLNP) using whey protein isolate. Food Res. Int. 2018, 105, 962–969. [Google Scholar] [CrossRef] [PubMed]
- Lima, J.G.D.; Brito-Oliveira, T.C.; Pinho, S.C.D. Characterization and evaluation of sensory acceptability of ice creams incorporated with beta-carotene encapsulated in solid lipid microparticles. Food Sci. Technol. 2016, 36, 664–671. [Google Scholar] [CrossRef]
- Gao, S.; McClements, D.J. Formation and stability of solid lipid nanoparticles fabricated using phase inversion temperature method. Colloids Surf. A Physicochem. Eng. Asp. 2016, 499, 79–87. [Google Scholar] [CrossRef]
- Gomes, G.V.L.; Simplício, I.A.S.; Souto, E.B.; Cardoso, L.P.; Pinho, S.C. Development of a lipid particle for β-carotene encapsulation using a blend of tristearin and sunflower oil: Choice of lipid matrix and evaluation of shelf life of dispersions. Food Technol. Biotechnol. 2012, 51, 383. [Google Scholar]
- Qian, C.; Decker, E.A.; Xiao, H.; McClements, D.J. Impact of lipid nanoparticle physical state on particle aggregation and β-carotene degradation: Potential limitations of solid lipid nanoparticles. Food Res. Int. 2013, 52, 342–349. [Google Scholar] [CrossRef]
- Zhang, L. Transparent Dispersions of Milk Fat-Based Solid Lipid Nanoparticles for Delivery of Beta-Carotene. Master’s Thesis, University of Tennessee, Knoxville, TN, USA, 2013. Available online: https://trace.tennessee.edu/utk_gradthes/2483 (accessed on 15 December 2020).
- Gomes, G.V.D.L.; Borrin, T.R.; Cardoso, L.P.; Souto, E.; Pinho, S.C.D. Characterization and shelf life of β-carotene loaded solid lipid microparticles produced with stearic acid and sunflower oil. Braz. Arch. Biol. Technol. 2013, 56, 663–671. [Google Scholar] [CrossRef]
- Oliveira, D.R.B.; Michelon, M.; de Figueiredo Furtado, G.; Sinigaglia-Coimbra, R.; Cunha, R.L. β-Carotene-loaded nanostructured lipid carriers produced by solvent displacement method. Food Res. Int. 2016, 90, 139–146. [Google Scholar] [CrossRef]
- Hejri, A.; Khosravi, A.; Gharanjig, K.; Hejazi, M. Optimisation of the formulation of β-carotene loaded nanostructured lipid carriers prepared by solvent diffusion method. Food Chem. 2013, 141, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Lacatusu, I.; Badea, N.; Ovidiu, O.; Bojin, D.; Meghea, A. Highly antioxidant carotene-lipid nanocarriers: Synthesis and antibacterial activity. J. Nanopart. Res. 2012, 14, 902. [Google Scholar] [CrossRef]
- Gomes, G.V.L.; Sola, M.R.; Marostegan, L.F.P.; Jange, C.G.; Cazado, C.P.S.; Pinheiro, A.C.; Vicente, A.A.; Pinho, S.C. Physico-chemical stability and in vitro digestibility of beta-carotene-loaded lipid nanoparticles of cupuacu butter (Theobroma grandiflorum) produced by the phase inversion temperature (PIT) method. J. Food Eng. 2017, 192, 93–102. [Google Scholar] [CrossRef]
- Ariviani, S.; Anggrahini, S.; Naruki, S.; Raharjo, S. Characterization and chemical stability evaluation of β-carotene microemulsions prepared by spontaneous emulsification method using VCO and palm oil as oil phase. Int. Food Res. J. 2015, 22, 2432–2439. [Google Scholar]
- Mao, Y.; Dubot, M.; Xiao, H.; McClements, D.J. Interfacial engineering using mixed protein systems: Emulsion-based delivery systems for encapsulation and stabilization of β-carotene. J. Agric. Food Chem. 2013, 61, 5163–5169. [Google Scholar] [CrossRef]
- Neves, M.A.; Ribeiro, H.S.; Fujiu, K.B.; Kobayashi, I.; Nakajima, M. Formulation of controlled size PUFA-loaded oil-in-water emulsions by microchannel emulsification using β-carotene-rich palm oil. Ind. Eng. Chem. Res. 2008, 47, 6405–6411. [Google Scholar] [CrossRef]
- Salvia-Trujillo, L.; Qian, C.; Martín-Belloso, O.; McClements, D.J. Influence of particle size on lipid digestion and β-carotene bioaccessibility in emulsions and nanoemulsions. Food Chem. 2013, 141, 1472–1480. [Google Scholar] [CrossRef] [PubMed]
- Yao, M.; McClements, D.J.; Xiao, H. Improving oral bioavailability of nutraceuticals by engineered nanoparticle-based delivery systems. Curr. Opin. Food Sci. 2015, 2, 14–19. [Google Scholar] [CrossRef]
- Jo, Y.-J.; Kwon, Y.-J. Characterization of β-carotene nanoemulsions prepared by microfluidization technique. Food Sci. Biotechnol. 2014, 23, 107–113. [Google Scholar] [CrossRef]
- Campardelli, R.; Adami, R.; Reverchon, E. Preparation of stable aqueous nanodispersions of β-carotene by supercritical assisted injection in a liquid antisolvent. Procedia Eng. 2012, 42, 1493–1501. [Google Scholar] [CrossRef]
- Meroni, E.; Raikos, V. Physicochemical stability, antioxidant properties and bioaccessibility of β-carotene in orange oil-in-water beverage emulsions: Influence of carrier oil types. Food Funct. 2018, 9, 320–330. [Google Scholar] [CrossRef] [PubMed]
- Teixé-Roig, J.; Oms-Oliu, G.; Ballesté-Muñoz, S.; Odriozola-Serrano, I.; Martín-Belloso, O. Improving the in vitro bioaccessibility of β-carotene using pectin added nanoemulsions. Foods 2020, 9, 447. [Google Scholar] [CrossRef] [PubMed]
- Zirak, M.B.; Pezeshki, A. Effect of surfactant concentration on the particle size, stability and potential zeta of beta carotene nano lipid carrier. Int. J. Curr. Microbiol. App. Sci. 2015, 4, 924–932. [Google Scholar]
- Salvia-Trujillo, L.; Qian, C.; Martín-Belloso, O.; McClements, D.J. Modulating β-carotene bioaccessibility by controlling oil composition and concentration in edible nanoemulsions. Food Chem. 2013, 139, 878–884. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Zhong, Q. Thermal and UV stability of β-carotene dissolved in peppermint oil microemulsified by sunflower lecithin and Tween 20 blend. Food Chem. 2015, 174, 630–636. [Google Scholar] [CrossRef] [PubMed]
- Qian, C.; Decker, E.A.; Xiao, H.; McClements, D.J. Physical and chemical stability of β-carotene-enriched nanoemulsions: Influence of pH, ionic strength, temperature, and emulsifier type. Food Chem. 2012, 132, 1221–1229. [Google Scholar] [CrossRef] [PubMed]
- Hasani, F.; Pezeshki, A.; Hamishehkar, H. Effect of surfactant and oil type on size droplets of β-carotene-bearing nanoemulsions. Int. J. Curr. Microbiol. Appl. Sci. 2015, 4, 146–155. [Google Scholar]
- Muhamad, I.I.; Fen, L.S.; Hui, N.H.; Mustapha, N.A. Genipin-cross-linked kappa-carrageenan/carboxymethyl cellulose beads and effects on beta-carotene release. Carbohydr. Polym. 2011, 83, 1207–1212. [Google Scholar] [CrossRef]
- Liang, R.; Huang, Q.; Ma, J.; Shoemaker, C.F.; Zhong, F. Effect of relative humidity on the store stability of spray-dried beta-carotene nanoemulsions. Food Hydrocoll. 2013, 33, 225–233. [Google Scholar] [CrossRef]
- Liang, R.; Shoemaker, C.F.; Yang, X.; Zhong, F.; Huang, Q. Stability and bioaccessibility of β-carotene in nanoemulsions stabilized by modified starches. J. Agric. Food Chem. 2013, 61, 1249–1257. [Google Scholar] [CrossRef] [PubMed]
- Barragán, P. Studies of Process Intensification for the Development of Hydrophilic and Hydrophobic β-Carotene Formulations. 2013. Available online: http://uvadoc.uva.es/handle/10324/4076 (accessed on 4 March 2021). [CrossRef]
- Liu, F.; Wang, D.; Sun, C.; Gao, Y. Influence of polysaccharides on the physicochemical properties of lactoferrin–polyphenol conjugates coated β-carotene emulsions. Food Hydrocoll. 2016, 52, 661–669. [Google Scholar] [CrossRef]
- Lin, Q.; Liang, R.; Ye, A.; Singh, H.; Zhong, F. Effects of calcium on lipid digestion in nanoemulsions stabilized by modified starch: Implications for bioaccessibility of β-carotene. Food Hydrocoll. 2017, 73, 184–193. [Google Scholar] [CrossRef]
- Chu, B.S.; Ichikawa, S.; Kanafusa, S.; Nakajima, M. Stability of protein-stabilised β-carotene nanodispersions against heating, salts and pH. J. Sci. Food Agric. 2008, 88, 1764–1769. [Google Scholar] [CrossRef]
- Sáiz-Abajo, M.-J.; González-Ferrero, C.; Moreno-Ruiz, A.; Romo-Hualde, A.; González-Navarro, C.J. Thermal protection of β-carotene in re-assembled casein micelles during different processing technologies applied in food industry. Food Chem. 2013, 138, 1581–1587. [Google Scholar] [CrossRef]
- Yi, J.; Fan, Y.; Zhang, Y.; Zhao, L. Characterization of catechin-α-lactalbumin conjugates and the improvement in β-carotene retention in an oil-in-water nanoemulsion. Food Chem. 2016, 205, 73–80. [Google Scholar] [CrossRef]
- Cornacchia, L.; Roos, Y.H. State of dispersed lipid carrier and interface composition as determinants of beta-carotene stability in oil-in-water emulsions. J. Food Sci. 2011, 76, C1211–C1218. [Google Scholar] [CrossRef] [PubMed]
- Cornacchia, L.; Roos, Y.H. Stability of β-carotene in protein-stabilized oil-in-water delivery systems. J. Agric. Food Chem. 2011, 59, 7013–7020. [Google Scholar] [CrossRef] [PubMed]
- Salem, A. Use of Whey Protein Nanoparticles for the Encapsulation and Sustained Delivery of Beta-Carotene and Zinc Micronutrient. Master’s Thesis, American University in Cairo, Cairo, Egypt, 2015. [Google Scholar]
- Yi, J.; Zhang, Y.; Liang, R.; Zhong, F.; Ma, J. Beta-carotene chemical stability in nanoemulsions was improved by stabilized with beta-lactoglobulin–catechin conjugates through free radical method. J. Agric. Food Chem. 2014, 63, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Caliskan, G.; Lim, A.S.L.; Roos, Y.H. Beta-carotene stability in extruded snacks produced using interface engineered emulsions. Int. J. Food Prop. 2015, 18, 2256–2267. [Google Scholar] [CrossRef][Green Version]
- Yi, J.; Li, Y.; Zhong, F.; Yokoyama, W. The physicochemical stability and in vitro bioaccessibility of beta-carotene in oil-in-water sodium caseinate emulsions. Food Hydrocoll. 2014, 35, 19–27. [Google Scholar] [CrossRef]
- Yin, L.-J.; Chu, B.-S.; Kobayashi, I.; Nakajima, M. Performance of selected emulsifiers and their combinations in the preparation of β-carotene nanodispersions. Food Hydrocoll. 2009, 23, 1617–1622. [Google Scholar] [CrossRef]
- Liu, F.; Wang, D.; Sun, C.; McClements, D.J.; Gao, Y. Utilization of interfacial engineering to improve physicochemical stability of β-carotene emulsions: Multilayer coatings formed using protein and protein–polyphenol conjugates. Food Chem. 2016, 205, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Wang, J.; McClements, D.J.; Zou, L. Encapsulation of β-carotene-loaded oil droplets in caseinate/alginate microparticles: Enhancement of carotenoid stability and bioaccessibility. J. Funct. Foods 2018, 40, 527–535. [Google Scholar] [CrossRef]
- Yi, J.; Zhong, F.; Zhang, Y.; Yokoyama, W.; Zhao, L. Effects of lipids on in vitro release and cellular uptake of β-carotene in nanoemulsion-based delivery systems. J. Agric. Food Chem. 2015, 63, 10831–10837. [Google Scholar] [CrossRef]
- Salvia Trujillo, L. Nanoemulsions as Delivery Systems of Food Ingredients: Improving Food Safety and Functionality. Ph.D. Thesis, Universitat de Lleida, Lleida, Spain, 2014. [Google Scholar]
- Sutter, S.C.; Buera, M.P.; Elizalde, B.E. β-Carotene encapsulation in a mannitol matrix as affected by divalent cations and phosphate anion. Int. J. Pharm. 2007, 332, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Chu, B.-S.; Ichikawa, S.; Kanafusa, S.; Nakajima, M. Preparation and characterization of β-carotene nanodispersions prepared by solvent displacement technique. J. Agric. Food Chem. 2007, 55, 6754–6760. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Guo, Y.; Sun, R.; Wang, X. Self-assembly and β-carotene loading capacity of hydroxyethyl cellulose-graft-linoleic acid nanomicelles. Carbohydr. Polym. 2016, 145, 56–63. [Google Scholar] [CrossRef]
- Moeller, H.; Martin, D.; Schrader, K.; Hoffmann, W.; Lorenzen, P.C. Native casein micelles as nanocarriers for β-carotene: pH-and temperature-induced opening of the micellar structure. Int. J. Food Sci. 2017, 52, 1122–1130. [Google Scholar] [CrossRef]
- Ge, W.; Li, D.; Chen, M.; Wang, X.; Liu, S.; Sun, R. Characterization and antioxidant activity of β-carotene loaded chitosan-graft-poly(lactide) nanomicelles. Carbohydr. Polym. 2015, 117, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Roohinejad, S.; Oey, I.; Wen, J.; Lee, S.J.; Everett, D.W.; Burritt, D.J. Formulation of oil-in-water β-carotene microemulsions: Effect of oil type and fatty acid chain length. Food Chem. 2015, 174, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Cao-Hoang, L.; Fougère, R.; Waché, Y. Increase in stability and change in supramolecular structure of β-carotene through encapsulation into polylactic acid nanoparticles. Food Chem. 2011, 124, 42–49. [Google Scholar] [CrossRef]
- Jain, A.; Sharma, G.; Kushwah, V.; Garg, N.K.; Kesharwani, P.; Ghoshal, G.; Singh, B.; Shivhare, U.S.; Jain, S.; Katare, O.P. Methotrexate and beta-carotene loaded-lipid polymer hybrid nanoparticles: A preclinical study for breast cancer. Nanomedicine 2017, 12, 1851–1872. [Google Scholar] [CrossRef]
- Wang, T.; Wang, R.; Chen, Z.; Zhong, Q. Coating oil droplets with rice proteins to control the release rate of encapsulated beta-carotene during in vitro digestion. RSC Adv. 2016, 6, 73627–73635. [Google Scholar] [CrossRef]
- Chuacharoen, T.; Sabliov, C.M. The potential of zein nanoparticles to protect entrapped β-carotene in the presence of milk under simulated gastrointestinal (GI) conditions. LWT—Food Sci. Technol. 2016, 72, 302–309. [Google Scholar] [CrossRef]
- Lim, A.S.L.; Burdikova, Z.; Sheehan, J.J.; Roos, Y.H. Carotenoid stability in high total solid spray dried emulsions with gum Arabic layered interface and trehalose–WPI composites as wall materials. Innov. Food Sci. Emerg. Technol. 2016, 34, 310–319. [Google Scholar] [CrossRef]
- Kim, J.-Y.; Huber, K.C. Preparation and characterization of corn starch-β-carotene composites. Carbohydr. Polym. 2016, 136, 394–401. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.B.; Shakil, N.A.; Kumar, J.; Walia, S.; Kar, A. Development of slow release formulations of β-carotene employing amphiphilic polymers and their release kinetics study in water and different pH conditions. J. Food Sci. Technol. 2015, 52, 8068–8076. [Google Scholar] [CrossRef][Green Version]
- de Paz, E.; Martín, Á.; Bartolomé, A.; Largo, M.; Cocero, M.J. Development of water-soluble β-carotene formulations by high-temperature, high-pressure emulsification and antisolvent precipitation. Food Hydrocoll. 2014, 37, 14–24. [Google Scholar] [CrossRef]
- Yi, J.; Lam, T.I.; Yokoyama, W.; Cheng, L.W.; Zhong, F. Beta-carotene encapsulated in food protein nanoparticles reduces peroxyl radical oxidation in Caco-2 cells. Food Hydrocoll. 2015, 43, 31–40. [Google Scholar] [CrossRef]
- De Paz, E.; Martín, Á.; Cocero, M.J. Production of water-soluble β-carotene formulations by high pressure processes. In Proceedings of the III Iberoamerican Conference on Supercritical Fluids, Cartagena de Indias, Colombia, 1–5 April 2013; pp. 1–6. [Google Scholar]
- Soukoulis, C.; Tsevdou, M.; Andre, C.M.; Cambier, S.; Yonekura, L.; Taoukis, P.S.; Hoffmann, L. Modulation of chemical stability and in vitro bioaccessibility of beta-carotene loaded in kappa-carrageenan oil-in-gel emulsions. Food Chem. 2017, 220, 208–218. [Google Scholar] [CrossRef]
- Fan, Y.; Yi, J.; Zhang, Y.; Wen, Z.; Zhao, L. Physicochemical stability and in vitro bioaccessibility of β-carotene nanoemulsions stabilized with whey protein-dextran conjugates. Food Hydrocoll. 2017, 63, 256–264. [Google Scholar] [CrossRef]
- Akinosho, H.O.; Wicker, L. Stability of β-carotene loaded emulsions vary by viscosity of hydroxypropyl methylcellulose dispersions. LWT-Food Sci. Technol. 2015, 63, 582–589. [Google Scholar] [CrossRef]
- Akrami, M. Gum Arabic-Caseinate Nanocomplexes Bearing Beta-Carotene (1): Studying of Complex Formation by FTIR, DSC, Turbidity and Rheology. 2016. Available online: https://www.sid.ir/en/journal/ViewPaper.aspx?id=553536 (accessed on 26 February 2021).
- Chen, D.; Zhao, C.-X.; Lagoin, C.; Hai, M.; Arriaga, L.R.; Koehler, S.; Abbaspourrad, A.; Weitz, D.A. Dispersing hydrophobic natural colourant β-carotene in shellac particles for enhanced stability and tunable colour. R. Soc. Open Sci. 2017, 4, 170919. [Google Scholar] [CrossRef]
- Chen, J.; Li, F.; Li, Z.; McClements, D.J.; Xiao, H. Encapsulation of carotenoids in emulsion-based delivery systems: Enhancement of β-carotene water-dispersibility and chemical stability. Food Hydrocoll. 2017, 69, 49–55. [Google Scholar] [CrossRef]
- Chloe, M.O.; Davidovich-Pinhas, M.; Wright, A.J.; Barbut, S.; Marangoni, A.G. Ethylcelluloseoleogels for lipophilic bioactive delivery–effect of oleogelation on in vitro bioaccessibility and stability of beta-carotene. Food Funct. 2017, 8, 1438–1451. [Google Scholar] [CrossRef]
- Deng, X.X.; Zhang, N.; Tang, C.H. Soy protein isolate as a nanocarrier for enhanced water dispersibility, stability and bioaccessibility of β-carotene. J. Sci. Food Agric. 2017, 97, 2230–2237. [Google Scholar] [CrossRef] [PubMed]
- Dobrzanski, C.D. Comparison of Non-Toxic Methods for Creating Beta-Carotene Encapsulated in Pmmananoparticles. 2016. Available online: https://rucore.libraries.rutgers.edu/rutgers-lib/51269/ (accessed on 4 March 2021).
- Fernández, A.; Torres-Giner, S.; Lagarón, J.M. Encapsulation of the functional ingredient beta-carotene in electrospun PLA fibers of interest in bioactive food packaging and food processing applications. In Proceedings of the 23rd IAPRI Symposium of Packaging, Windsor, UK, 3–5 September 2007. [Google Scholar]
- Mahfoudhi, N.; Hamdi, S. Kinetic degradation and storage stability of β-carotene encapsulated by freeze-drying using almond gum and gum Arabic as wall materials. J. Food Process. Preserv. 2015, 39, 896–906. [Google Scholar] [CrossRef]
- Mahfoudhi, N.; Hamdi, S. Kinetic degradation and storage stability of β-carotene encapsulated by spray drying using almond gum and gum arabic as wall materials. Polym. Eng. Sci. 2014, 34, 683–693. [Google Scholar] [CrossRef]
- Hejri, A.; Gharanjig, K.; Khosravi, A.; Hejazi, M. Effect of surfactants on kinetics of β-carotene photodegradation in emulsions. Chem. Eng. Commun. 2013, 200, 437–447. [Google Scholar] [CrossRef]
- Sharif, H.R.; Goff, H.D.; Majeed, H.; Shamoon, M.; Liu, F.; Nsor-Atindana, J.; Haider, J.; Liang, R.; Zhong, F. Physicochemical properties of β-carotene and eugenol co-encapsulated flax seed oil powders using OSA starches as wall material. Food Hydrocoll. 2017, 73, 274–283. [Google Scholar] [CrossRef]
- Meng, J.; Kang, T.-T.; Wang, H.-F.; Zhao, B.-B.; Lu, R.-R. Physicochemical properties of casein-dextran nanoparticles prepared by controlled dry and wet heating. Int. J. Biol. Macromol. 2018, 107, 2604–2610. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Zhou, Y.; Bai, L.; Liu, F.; Deng, Y.; McClements, D.J. Fabrication of β-carotene nanoemulsion-based delivery systems using dual-channel microfluidization: Physical and chemical stability. J. Colloid Interface Sci. 2017, 490, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Gao, L.; Yi, J.; Zhang, Y.; Yokoyama, W. Development of β-carotene loaded organogel based nanoemulsion with improved invitro and in vivo bioaccessibility. J. Agric. Food Chem. 2017, 65, 6188–6194. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Kelly, A.; Miao, S. Bioaccessibility and cellular uptake of β-carotene encapsulated in model O/W emulsions: Influence of initial droplet size and emulsifiers. Nanomaterials 2017, 7, 282. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, X.; Wang, X.; Huang, Y.; Yang, B.; Pan, X.; Wu, C. The influence of maltodextrin on the physicochemical properties and stabilization of beta-carotene emulsions. AAPS Pharm. Sci. Tech. 2017, 18, 821–828. [Google Scholar] [CrossRef]
- Thais, C.; Oliveira, B.; Camila, V.; Samantha, C. Encapsulation of beta carotene in lipid microparticles stabilized with hydrolyzed soy protein isolate: Production parameters, alphaâ tocopherol coencapsulation and stability under stress conditions. J. Food Sci. 2017, 82, 659–669. [Google Scholar] [CrossRef]
- Ozvural, E.B.; Huang, Q. Quality differences of hamburger patties incorporated with encapsulated β-carotene both as an additive and edible coating. J. Food Process. Preserv. 2017, 42, e13353. [Google Scholar] [CrossRef]
- Shao, P.; Qiu, Q.; Xiao, J.; Zhu, Y.; Sun, P. Chemical stability and in vitro release properties of β-carotene in emulsions stabilized by Ulvafasciata polysaccharide. Int. J. Biol. Macromol. 2017, 102, 225–231. [Google Scholar] [CrossRef]
- Wang, M.; Fu, Y.; Chen, G.; Shi, Y.; Li, X.; Zhang, H.; Shen, Y. Fabrication and characterization of carboxymethyl chitosan and tea polyphenols coating on zein nanoparticles to encapsulate β-carotene by anti-solvent precipitation method. Food Hydrocoll. 2018, 77, 577–587. [Google Scholar] [CrossRef]
- Sharif, H.R.; Goff, H.D.; Majeed, H.; Liu, F.; Nsor-Atindana, J.; Haider, J.; Liang, R.; Zhong, F. Physicochemical stability of β-carotene and α-tocopherol enriched nanoemulsions: Influence of carrier oil, emulsifier and antioxidant. Colloids Surf. A Physicochem. Eng. Asp. 2017, 529, 550–559. [Google Scholar] [CrossRef]
- Soukoulis, C.; Tsevdou, M.; Yonekura, L.; Cambier, S.; Taoukis, P.S.; Hoffmann, L. Does kappa-carrageenan thermoreversible gelation affect β-carotene oxidative degradation and bioaccessibility in o/w emulsions? Carbohydr. Polym. 2017, 167, 259–269. [Google Scholar] [CrossRef] [PubMed]
- Thakur, D.; Jain, A.; Ghoshal, G.; Shivhare, U.S.; Katare, O.P. Microencapsulation of β-carotene based on casein/guar gum blend using zeta potential-yield stress phenomenon: An approach to enhance photo-stability and retention of functionality. AAPS Pharm. Sci. Technol. 2017, 18, 1447–1459. [Google Scholar] [CrossRef]
- Gu, L.; Su, Y.; Zhang, M.; Chang, C.; Li, J.; McClements, D.J.; Yang, Y. Protection of β-carotene from chemical degradation in emulsion-based delivery systems using antioxidant interfacial complexes: Catechin-egg white protein conjugates. Food Res. Int. 2017, 96, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Xia, Z.; McClements, D.J.; Xiao, H. Influence of lipid content in a corn oil preparation on the bioaccessibility of β-carotene: A comparison of low-fat and high-fat samples. J. Food Sci. 2017, 82, 373–379. [Google Scholar] [CrossRef]
- Mun, S.; McClements, D.J. Influence of simulated in-mouth processing (size reduction and alpha-amylase addition) on lipid digestion and β-carotene bioaccessibility in starch-based filled hydrogels. LWT-Food Sci. Technol. 2017, 80, 113–120. [Google Scholar] [CrossRef]
- Morowvat, M.H.; Ghasemi, Y. Spray-drying microencapsulation of β-carotene contents in powdered Dunaliella salina biomass. Int. J. Pharm. Clin. Res. 2016, 8, 1533–1536. [Google Scholar] [CrossRef]
- Miyazawa, T.; Nakagawa, K.; Harigae, T.; Onuma, R.; Kimura, F.; Fujii, T.; Miyazawa, T. Distribution of β-carotene-encapsulated polysorbate 80-coated poly (D, L-lactide-co-glycolide) nanoparticles in rodent tissues following intravenous administration. Int. J. Nanomed. 2015, 10, 7223. [Google Scholar] [CrossRef][Green Version]
- Gupta, S.S.; Ghosh, M. In vitro study of anti-oxidative effects of β-carotene and α-lipoic acid for nanocapsulated lipids. LWT-Food Sci. Technol. 2012, 49, 131–138. [Google Scholar] [CrossRef]
- Hou, Z.; Zhang, M.; Liu, B.; Yan, Q.; Yuan, F.; Xu, D.; Gao, Y. Effect of chitosan molecular weight on the stability and rheological properties of β-carotene emulsions stabilized by soybean soluble polysaccharides. Food Hydrocoll. 2012, 26, 205–211. [Google Scholar] [CrossRef]
- Tavares, J.K.; de Souza, A.A.U.; de Oliveira, J.V.; Priamo, W.L.; de Souza, S.M.A.G.U. Modeling of the controlled release of beta carotene into anhydrous ethanol from microcapsules. Open Nano 2016, 1, 25–35. [Google Scholar] [CrossRef]
- Wagner, L.A.; Warthesen, J.J. Stability of spray-dried encapsulated carrot carotenes. J. Food Sci. 1995, 60, 1048–1053. [Google Scholar] [CrossRef]
- Dluzewska, E.; Florowska, A.; Jasiorowska, E. Effect of carrier type of stability of β-carotene micro-encapsulated using spray-drying method. Zywnosc Nauka Technol. Jakosc 2011, 18, 140–151. [Google Scholar] [CrossRef]
- Aissa, A.F.; Bianchi, M.L.P.; Ribeiro, J.C.; Hernandes, L.C.; de Faria, A.F.; Mercadante, A.Z.; Antunes, L.M.G. Comparative study of β-carotene and microencapsulated β-carotene: Evaluation of their genotoxic and antigenotoxic effects. Food Chem. Toxicol. 2012, 50, 1418–1424. [Google Scholar] [CrossRef]
- González-Reza, R.M.; Quintanar-Guerrero, D.; Flores-Minutti, J.J.; Gutiérrez-Cortez, E.; Zambrano-Zaragoza, M.L. Nanocapsules of β-carotene: Thermal degradation kinetics in a scraped surface heat exchanger (SSHE). LWT—Food Sci. Technol. 2015, 60, 124–130. [Google Scholar] [CrossRef]
- Jain, A.; Thakur, D.; Ghoshal, G.; Katare, O.P.; Shivhare, U.S. Characterization of microcapsulated β-carotene formed by complex coacervation using casein and gum tragacanth. Int. J. Biol. Macromol. 2016, 87, 101–113. [Google Scholar] [CrossRef]
- Donhowe, E.G. Microencapsulation of β-Carotene: Characterization, In Vitro Release, and Bioavailability. Master’s Thesis, University of Georgia, Athens, Greece, 2013. [Google Scholar]
- Nik, A.M.; Wright, A.J.; Corredig, M. Micellization of beta-carotene from soy-protein stabilized oil-in-water emulsions under in vitro conditions of lipolysis. J. Am. Oil Chem. Soc. 2011, 88, 1397–1407. [Google Scholar] [CrossRef]
- Franceschi, E.; De Cezaro, A.; Ferreira, S.R.S.; Kunita, M.H.; Muniz, E.C.; Rubira, A.F.; Oliveira, J. Co-precipitation of beta-carotene and bio-polymer using supercritical carbon dioxide as antisolvent. Open Chem. Eng. J. 2010, 4, 11–20. [Google Scholar] [CrossRef]
- Priamo, W.L.; de Cezaro, A.M.; Ferreira, S.R.S.; Oliveira, J.V. Precipitation and encapsulation of β-carotene in PHBV using carbon dioxide as anti-solvent. J. Supercrit. Fluids 2010, 54, 103–109. [Google Scholar] [CrossRef]
- Divya, P.; Anbarasan, B.; Ramaprabhu, S. Preparation and characterization of beta-carotene encapsulated chitosan, oleic acid coated Fe3O4nanoparticles. In Proceedings of the International Conference on Nanoscience, Technology and Societal Implications (NSTSI), Bhubaneswar, India, 8–10 December 2011; pp. 1–4. [Google Scholar]
- Kim, J.-Y.; Seo, T.-R.; Lim, S.-T. Preparation of aqueous dispersion of β-carotene nano-composites through complex formation with starch dextrin. Food Hydrocoll. 2013, 33, 256–263. [Google Scholar] [CrossRef]
- Ramoneda, X.A.; Ponce-Cevallos, P.A.; Buera, M.D.P.; Elizalde, B.E. Degradation of β-carotene in amorphous polymer matrices. Effect of water sorption properties and physical state. J. Sci. Food Agric. 2011, 91, 2587–2593. [Google Scholar] [CrossRef] [PubMed]
- Silva, H.D.; Cerqueira, M.A.; Souza, B.W.S.; Ribeiro, C.; Avides, M.C.; Quintas, M.A.C.; Coimbra, J.S.R.; Carneiro-da-Cunha, M.G.; Vicente, A.A. Nanoemulsions of β-carotene using a high-energy emulsification–evaporation technique. J. Food Eng. 2011, 102, 130–135. [Google Scholar] [CrossRef]
- Trentin, A.; De Lamo, S.; Güell, C.; López, F.; Ferrando, M. Protein-stabilized emulsions containing beta-carotene produced by premix membrane emulsification. J. Food Eng. 2011, 106, 267–274. [Google Scholar] [CrossRef]
- Qi, J. Microencapsulation of Beta-Carotene in Pea Protein Wall System; Department of Food Science, University of Manitoba: Winnipeg, MB, Canada, 2004. [Google Scholar]
- Barreto, H.R.D. Physical Stability of a Lactose-Trehalose Matrix for Nano-Encapsulation of Beta-Carotene by Spray Drying; Instituto Superior de Agronomia: Lisboa, Portugal, 2008. [Google Scholar]
- Silva, M.M.; Nora, L.; Cantillano, R.F.F.; Paese, K.; Guterres, S.S.; Pohlmann, A.R.; Costa, T.M.H.; de Oliveira Rios, A. The production, characterization, and the stability of carotenoids loaded in lipid-core nanocapsules. Food Bioprocess Technol. 2016, 9, 1148–1158. [Google Scholar] [CrossRef][Green Version]
- Gupta, S.S.; Ghosh, M. Synthesis, characterization, stability evaluation and release kinetics of fiber-encapsulated carotene nano-capsules. Grasas Aceites 2015, 66, e104. [Google Scholar] [CrossRef][Green Version]
- Peinado, I.; Mason, M.; Romano, A.; Biasioli, F.; Scampicchio, M. Stability of β-carotene in polyethylene oxide electrospun nanofibers. Appl. Surf. Sci. 2016, 370, 111–116. [Google Scholar] [CrossRef]
- Donhowe, E.G.; Flores, F.P.; Kerr, W.L.; Wicker, L.; Kong, F. Characterization and in vitro bioavailability of β-carotene: Effects of microencapsulation method and food matrix. LWT—Food Sci. Technol. 2014, 57, 42–48. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, R.; McClements, D.J. Encapsulation of β-carotene in alginate-based hydrogel beads: Impact on physicochemical stability and bioaccessibility. Food Hydrocoll. 2016, 61, 1–10. [Google Scholar] [CrossRef]
- Park, S.; Mun, S.; Kim, Y.-R. Effect of xanthan gum on lipid digestion and bioaccessibility of β-carotene-loaded rice starch-based filled hydrogels. Food Res. Int. 2018, 105, 440–445. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, C. In-Vitro Bioaccessibility and Stability of Beta-Carotene in Ethylcelluloseoleogels. 2016. Available online: http://hdl.handle.net/10214/9716 (accessed on 16 December 2020).
- Shao, Y.; Tang, C.-H. Gel-like pea protein pickering emulsions at pH3.0 as a potential intestine-targeted and sustained-release delivery system for β-carotene. Food Res. Int. 2016, 79, 64–72. [Google Scholar] [CrossRef]
- Soukoulis, C.; Cambier, S.; Hoffmann, L.; Bohn, T. Chemical stability and bioaccessibility of β-carotene encapsulated in sodium alginate o/w emulsions: Impact of Ca2+ mediated gelation. Food Hydrocoll. 2016, 57, 301–310. [Google Scholar] [CrossRef]
- Wang, S.; Chen, X.; Shi, M.; Zhao, L.; Li, W.; Chen, Y.; Lu, M.; Wu, J.; Yuan, Q.; Li, Y. Absorption of whey protein isolated (WPI)-stabilized β-carotene emulsions by oppositely charged oxidized starch microgels. Food Res. Int. 2015, 67, 315–322. [Google Scholar] [CrossRef]
- Liu, F.; Tang, C.-H. Soy glycinin as food-grade pickering stabilizers: Part. III. Fabrication of gel-like emulsions and their potential as sustained-release delivery systems for β-carotene. Food Hydrocoll. 2016, 56, 434–444. [Google Scholar] [CrossRef]
- Mun, S.; Kim, Y.-R.; McClements, D.J. Control of β-carotene bioaccessibility using starch-based filled hydrogels. Food Chem. 2015, 173, 454–461. [Google Scholar] [CrossRef]
- Kong, L.; Bhosale, R.; Ziegler, G.R. Encapsulation and stabilization of β-carotene by amylose inclusion complexes. Food Res. Int. 2018, 105, 446–452. [Google Scholar] [CrossRef]
- Ramezanli, T.; Kilfoyle, B.E.; Zhang, Z.; Michniak-Kohn, B.B. Polymeric nanospheres for topical delivery of vitamin D3. Int. J. Pharm. 2017, 516, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Uyen, N.T.T.; Hamid, Z.A.A.; Tram, N.X.T.; Ahmad, N. Fabrication of alginate microspheres for drug delivery: A review. Int. J. Biol. Macromol. 2020, 153, 1035–1046. [Google Scholar] [CrossRef] [PubMed]
- Lengyel, M.; Kállai-Szabó, N.; Antal, V.; Laki, A.J.; Antal, I. Microparticles, microspheres, and microcapsules for advanced drug delivery. Sci. Pharm. 2019, 87, 20. [Google Scholar] [CrossRef]
- Cascone, S.; Lamberti, G. Hydrogel-based commercial products for biomedical applications: A review. Int. J. Pharm. 2020, 573, 118803. [Google Scholar] [CrossRef] [PubMed]
- Batista, R.A.; Espitia, P.J.P.; Quintans, J.D.S.S.; Freitas, M.M.; Cerqueira, M.Â.; Teixeira, J.A.; Cardoso, J.C. Hydrogel as an alternative structure for food packaging systems. Carbohydr. Polym. 2019, 205, 106–116. [Google Scholar] [CrossRef] [PubMed]
- Qu, B.; Luo, Y. Chitosan-based hydrogel beads: Preparations, modifications and applications in food and agriculture sectors–A review. Int. J. Biol. Macromol. 2020, 152, 437–448. [Google Scholar] [CrossRef] [PubMed]
- Huynh, P.T. Solvent-Free Beta-Carotene Nanoparticles for Food Fortification; Rutgers the State University of New Jersey-New Brunswick: New Brunswick, NJ, USA, 2012; Available online: https://rucore.libraries.rutgers.edu/rutgers-lib/38798/ (accessed on 4 March 2021).
- Nascimento, L.G.L.; Casanova, F.; Silva, N.F.N.; de Carvalho Teixeira, A.V.N.; de Carvalho, A.F. Casein-based hydrogels: A mini-review. Food Chem. 2020, 314, 126063. [Google Scholar] [CrossRef]
- Klein, M.; Poverenov, E. Natural biopolymer-based hydrogels for use in food and agriculture. J. Sci. Food Agric. 2020, 100, 2337–2347. [Google Scholar] [CrossRef]
- Zheng, H.; Mao, L.; Cui, M.; Liu, J.; Gao, Y. Development of food-grade bigels based on κ-carrageenan hydrogel and monoglyceride oleogels as carriers for β-carotene: Roles of oleogel fraction. Food Hydrocoll. 2020, 105, 105855. [Google Scholar] [CrossRef]
- Li, J.; Jia, X.; Yin, L. Hydrogel: Diversity of structures and applications in food science. Food Rev. Int. 2021, 1–59. [Google Scholar] [CrossRef]
- Chen, X.; Liang, R.; Zhong, F.; Yokoyama, W.H. Effect of β-carotene status in microcapsules on its in vivo bioefficacy and in vitro bioaccessibility. Food Hydrocoll. 2020, 106, 105848. [Google Scholar] [CrossRef]
- Chen, X.; Liang, R.; Zhong, F.; Ma, J.; John, N.-A.; Goff, H.D.; Yokoyama, W.H. Effect of high concentrated sucrose on the stability of OSA-starch-based β-carotene microcapsules. Food Hydrocoll. 2019, 113, 105472. [Google Scholar] [CrossRef]
- Erdoğar, N.; Akkın, S.; Bilensoy, E. Nanocapsules for drug delivery: An updated review of the last decade. Recent Pat. Drug Deliv. Formul. 2018, 12, 252–266. [Google Scholar] [CrossRef]
- Vijeth, S.; Heggannavar, G.B.; Kariduraganavar, M.Y. Encapsulating wall materials for micro-/nanocapsules. In Microencapsulation-Processes, Technologies and Industrial Applications; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Amgoth, C.; Kumar, K.; Medhi, H.; Paik, P. Polymeric nanocapsules for drug delivery applications. Nanotechnol. Anim. Health Prod. 2014, 99–123. [Google Scholar] [CrossRef]
- Zhao, L.; Duan, G.; Zhang, G.; Yang, H.; He, S.; Jiang, S. Electrospun functional materials toward food packaging applications: A review. Nanomaterials 2020, 10, 150. [Google Scholar] [CrossRef] [PubMed]
- De Farias, B.S.; Junior, T.R.S.A.C.; de Almeida Pinto, L.A. Chitosan-functionalized nanofibers: A comprehensive review on challenges and prospects for food applications. Int. J. Biol. Macromol. 2019, 123, 210–220. [Google Scholar] [CrossRef] [PubMed]
- Kumar, T.S.M.; Kumar, K.S.; Rajini, N.; Siengchin, S.; Ayrilmis, N.; Rajulu, A.V. A comprehensive review of electrospun nanofibers: Food and packaging perspective. Compos. B Eng. 2019, 175, 107074. [Google Scholar] [CrossRef]
- Zhang, C.; Li, Y.; Wang, P.; Zhang, H. Electrospinning of nanofibers: Potentials and perspectives for active food packaging. Compr. Rev. Food Sci. 2020, 19, 479–502. [Google Scholar] [CrossRef]
- Rezaei, A.; Nasirpour, A.; Fathi, M. Application of cellulosic nanofibers in food science using electrospinning and its potential risk. Compr. Rev. Food Sci. 2015, 14, 269–284. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Lin, L.; Zhu, Y.; Liu, W.; Yu, T.; Ge, M. Preparation of ultrafine fast-dissolving cholecalciferol-loaded poly (vinyl pyrrolidone) fiber mats via electrospinning. Polym. Compos. 2013, 34, 282–287. [Google Scholar] [CrossRef]
- Barroso, L.; Viegas, C.; Vieira, J.; Pego, C.; Costa, J.; Fonte, P. Lipid-based carriers for food ingredients delivery. J. Food. Eng. 2020, 295, 110451. [Google Scholar] [CrossRef]
- Chaudhari, V.S.; Murty, U.S.; Banerjee, S. Lipidic nanomaterials to deliver natural compounds against cancer: A review. Environ. Chem. Lett. 2020, 18, 1–10. [Google Scholar] [CrossRef]
- Sarkar, A.; Mackie, A.R. Engineering oral delivery of hydrophobic bioactives in real world scenarios. Curr. Opin. Colloid Interface Sci. 2020, 48, 40–52. [Google Scholar] [CrossRef]
- Shukla, D.; Chakraborty, S.; Singh, S.; Mishra, B. Lipid-based oral multiparticulate formulations–advantages, technological advances and industrial applications. Expert Opin. Drug Deliv. 2011, 8, 207–224. [Google Scholar] [CrossRef] [PubMed]
- Mozafari, M.R.; Flanagan, J.; Matia-Merino, L.; Awati, A.; Omri, A.; Suntres, Z.E.; Singh, H. Recent trends in the lipid-based nanoencapsulation of antioxidants and their role in foods. J. Sci. Food Agric. 2006, 86, 2038–2045. [Google Scholar] [CrossRef]
- Akhavan, S.; Assadpour, E.; Katouzian, I.; Jafari, S.M. Lipid nano scale cargos for the protection and delivery of food bioactive ingredients and nutraceuticals. Trends Food Sci. Technol. 2018, 74, 132–146. [Google Scholar] [CrossRef]
- de Souza Simões, L.; Madalena, D.A.; Pinheiro, A.C.; Teixeira, J.A.; Vicente, A.A.; Ramos, O.L. Micro-and nano bio-based delivery systems for food applications: In vitro behavior. Adv. Colloid Interface Sci. 2017, 243, 23–45. [Google Scholar] [CrossRef] [PubMed]
- Fathi, M.; Mozafari, M.R.; Mohebbi, M. Nanoencapsulation of food ingredients using lipid based delivery systems. Trends Food Sci. Technol. 2012, 23, 13–27. [Google Scholar] [CrossRef]
- Sun, X.; Bandara, N. Applications of reverse micelles technique in food science: A comprehensive review. Trends Food Sci. Technol. 2019, 91, 106–115. [Google Scholar] [CrossRef]
- Kiss, É. Nanotechnology in food systems: A review. Acta Aliment. 2020, 49, 460–474. [Google Scholar] [CrossRef]
- Wakaskar, R.R. General overview of lipid-polymer hybrid nanoparticles, dendrimers, micelles, liposomes, spongosomes and cubosomes. J. Drug Target. 2017, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Keskin, D.; Tezcaner, A. Micelles as delivery system for cancer treatment. Curr. Pharm. Des. 2017, 25, 5230–5241. [Google Scholar] [CrossRef] [PubMed]
- Tanbour, R.; Martins, A.M.; Pitt, W.G.; Husseini, G.A. Drug delivery systems based on polymeric micelles and ultrasound: A review. Curr. Pharm. Des. 2016, 22, 2796–2807. [Google Scholar] [CrossRef] [PubMed]
- Gothwal, A.; Khan, I.; Gupta, U. Polymeric micelles: Recent advancements in the delivery of anticancer drugs. Pharm. Res. 2016, 33, 18–39. [Google Scholar] [CrossRef] [PubMed]
- Ranadheera, C.S.; Liyanaarachchi, W.S.; Chandrapala, J.; Dissanayake, M.; Vasiljevic, T. Utilizing unique properties of caseins and the casein micelle for delivery of sensitive food ingredients and bioactives. Trends Food Sci. Technol. 2016, 57, 178–187. [Google Scholar] [CrossRef]
- Kim, S.; Shi, Y.; Kim, J.Y.; Park, K.; Cheng, J.-X. Overcoming the barriers in micellar drug delivery: Loading efficiency, in vivo stability, and micelle–cell interaction. Expert Opin. Drug Deliv. 2010, 7, 49–62. [Google Scholar] [CrossRef]
- McClements, D.J.; Xiao, H. Is nano safe in foods? Establishing the factors impacting the gastrointestinal fate and toxicity of organic and inorganic food-grade nanoparticles. NPJ Sci. Food 2017, 1, 1–13. [Google Scholar] [CrossRef]
- Du, Y.; Bao, C.; Huang, J.; Jiang, P.; Jiao, L.; Ren, F.; Li, Y. Improved stability, epithelial permeability and cellular antioxidant activity of β-carotene via encapsulation by self-assembled α-lactalbumin micelles. Food Chem. 2019, 271, 707–714. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, P.; André, V.; Rieger, J.; Kühnle, A. Nano-emulsion formation by emulsion phase inversion. Colloids Surf. A Physico Chem. 2004, 251, 53–58. [Google Scholar] [CrossRef]
- Flores-Miranda, G.A.; del Toro, G.V.; Yánez-Fernández, J. Stability evaluation of β-carotene nanoemulsions prepared by homogenization-emulsification process using stearic acid as oil phase. Rev. Mex. Ing. Quim. 2015, 14, 667–680. [Google Scholar] [CrossRef]
- Xia, Z.; Han, Y.; Du, H.; McClements, D.J.; Tang, Z.; Xiao, H. Exploring the effects of carrier oil type on in vitro bioavailability of β-carotene: A cell culture study of carotenoid-enriched nanoemulsions. LWT-Food Sci. Technol. 2020, 134, 110224. [Google Scholar] [CrossRef]
- Gomes, M.; Santos, D.T.; Meireles, M.A.A. Trends in particle formation of bioactive compounds using supercritical fluids and nanoemulsions. Food Public Health 2012, 2, 142–152. [Google Scholar] [CrossRef]
- Zhang, R.; Zhang, Z.; Kumosani, T.; Khoja, S.; Abualnaja, K.O.; McClements, D.J. Encapsulation of β-carotene in nanoemulsion-based delivery systems formed by spontaneous emulsification: Influence of lipid composition on stability and bioaccessibility. Food Biophys. 2016, 11, 154–164. [Google Scholar] [CrossRef]
- Rao, J.; McClements, D.J. Stabilization of phase inversion temperature nanoemulsions by surfactant displacement. J. Agric. Food Chem. 2010, 58, 7059–7066. [Google Scholar] [CrossRef]
- McClements, D.J. Nanoemulsions versus microemulsions: Terminology, differences, and similarities. Soft Matter 2012, 8, 1719–1729. [Google Scholar] [CrossRef]
- Meng, Q.; Long, P.; Zhou, J.; Ho, C.-T.; Zou, X.; Chen, B.; Zhang, L. Improved absorption of β-carotene by encapsulation in an oil-in-water nanoemulsion containing tea polyphenols in the aqueous phase. Food Res. Int. 2019, 116, 731–736. [Google Scholar] [CrossRef] [PubMed]
- NishitaniYukuyama, M.; Tomiko Myiake Kato, E.; Lobenberg, R.; AraciBou-Chacra, N. Challenges and future prospects of nanoemulsion as a drug delivery system. Curr. Pharm. Des. 2017, 23, 495–508. [Google Scholar] [CrossRef]
- Choi, S.J.; McClements, D.J. Nanoemulsions as delivery systems for lipophilic nutraceuticals: Strategies for improving their formulation, stability, functionality and bioavailability. Food Sci. Biotechnol. 2020, 29, 149–168. [Google Scholar] [CrossRef] [PubMed]
- Daeihamed, M.; Dadashzadeh, S.; Haeri, A.; Faghih Akhlaghi, M. Potential of liposomes for enhancement of oral drug absorption. Curr. Drug Deliv. 2017, 14, 289–303. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Li, X.; Zhang, T.; Song, Y.; She, Z.; Li, J.; Deng, Y. Progress involving new techniques for liposome preparation. Asian J. Pharm. Sci. 2014, 9, 176–182. [Google Scholar] [CrossRef]
- Bozzuto, G.; Molinari, A. Liposomes as nanomedical devices. Int. J. Nanomed. 2015, 10, 975–999. [Google Scholar] [CrossRef] [PubMed]
- Nekkanti, V.; Venkatesan, N.; Betageri, G. Proliposomes for oral delivery: Progress and challenges. Curr. Pharm. Biotechnol. 2015, 16, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Ye, A.; Singh, H. Progress in applications of liposomes in food systems. In Microencapsulation and Microspheres for Food Applications; Sagis, L.M.C., Ed.; Academic Press: New York, NY, USA, 2015; pp. 151–170. [Google Scholar] [CrossRef]
- Kim, J.-S. Liposomal drug delivery system. J. Pharm. Sci. 2016, 46, 387–392. [Google Scholar] [CrossRef]
- Mozafari, M.R.; Khosravi-Darani, K.; Borazan, G.G.; Cui, J.; Pardakhty, A.; Yurdugul, S. Encapsulation of food ingredients using nanoliposome technology. Int. J. Food Prop. 2008, 11, 833–844. [Google Scholar] [CrossRef]
- Amoabediny, G.; Haghiralsadat, F.; Naderinezhad, S.; Helder, M.N.; AkhoundiKharanaghi, E.; MohammadnejadArough, J.; Zandieh-Doulabi, B. Overview of preparation methods of polymeric and lipid-based (noisome, solid lipid, liposome) nanoparticles: A comprehensive review. Int. J. Polym. Mater. 2018, 67, 383–400. [Google Scholar] [CrossRef]
- Filipczak, N.; Pan, J.; Yalamarty, S.S.K.; Torchilin, V.P. Recent advancements in liposome technology. Adv. Drug Deliv. Rev. 2020, 152, 4–22. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, V.; Chavan, S.; Jain, U.; Tarwadi, K. Liposomes for Nanodelivery Systems in Food Products. In Nanoscience for Sustainable Agriculture; Springer: Cham, Switzerland, 2019; pp. 627–638. [Google Scholar] [CrossRef]
- Liu, X.; Wang, P.; Zou, Y.-X.; Luo, Z.-G.; Tamer, T.M. Co-encapsulation of vitamin C and β-carotene in liposomes: Storage stability, antioxidant activity, and in vitro gastrointestinal digestion. Food Res. Int. 2020, 136, 109587. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.; Xue, J.; Abbas, S.; Feng, B.; Zhang, X.; Xia, S. Liposome as a delivery system for carotenoids: Comparative antioxidant activity of carotenoids as measured by ferric reducing antioxidant power, DPPH assay and lipid peroxidation. J. Agric. Food Chem. 2014, 62, 6726–6735. [Google Scholar] [CrossRef] [PubMed]
- Rashidinejad, A.; Birch, E.J.; Sun-Waterhouse, D.; Everett, D.W. Delivery of green tea catechin and epigallocatechin gallate in liposomes incorporated into low-fat hard cheese. Food Chem. 2014, 156, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Elkholy, N.S.; Shafaa, M.W.; Mohammed, H.S. Biophysical characterization of lutein or beta carotene-loaded cationic liposomes. RSC Adv. 2020, 10, 32409–32422. [Google Scholar] [CrossRef]
- Payne, N.I.; Ambrose, C.V.; Timmins, P.; Ward, M.D.; Ridgway, F. Proliposomes: A novel solution to an old problem. J. Pharm. Sci. 1986, 75, 325–329. [Google Scholar] [CrossRef] [PubMed]
- Chordiya, D.; Shilpi, S.; Choudhary, D.; Saraogi, G.K.; Sharma, M.; Kalyane, D.; Tekade, R.K. Proliposomes: A potential colloidal carrier for drug delivery applications. In The Future of Pharmaceutical Product Development and Research; Elsevier: Amsterdam, The Netherlands, 2020; pp. 581–608. [Google Scholar] [CrossRef]
- Khorasani, S.; Danaei, M.; Mozafari, M.R. Nanoliposome technology for the food and nutraceutical industries. Trends Food Sci. Technol. 2018, 79, 106–115. [Google Scholar] [CrossRef]
- Matos, M.; Pando, D.; Gutiérrez, G. Nanoencapsulation of food ingredients by niosomes. In Lipid-Based Nanostructures for Food Encapsulation Purposes; Elsevier: Amsterdam, The Netherlands, 2019; pp. 447–481. [Google Scholar] [CrossRef]
- Wen, J.; Al Gailani, M.; Yin, N.; Rashidinejad, A. Liposomes and niosomes. In Liposomes and Niosomes Emulsion-Based Systems for Delivery of Food Active Compounds; Wiley: Hoboken, NJ, USA, 2018; pp. 263–292. [Google Scholar] [CrossRef]
- Roohinejad, S.; Greiner, R.; Oey, I.; Wen, J. Emulsion-Based Systems for Delivery of Food Active Compounds: Formation, Application, Health and Safety; John Wiley & Sons: Hoboken, NJ, USA, 2018. [Google Scholar]
- Moghassemi, S.; Hadjizadeh, A. Nano-niosomes as nanoscale drug delivery systems: An illustrated review. J. Control. Release 2014, 185, 22–36. [Google Scholar] [CrossRef]
- Arora, R. Advances in niosome as a drug carrier: A review. Asian J. Pharm. 2007, 1, 29–39. [Google Scholar]
- Yasam, V.R.; Jakki, S.L.; Natarajan, J.; Kuppusamy, G. A review on novel vesicular drug delivery: Proniosomes. Drug Deliv. 2014, 21, 243–249. [Google Scholar] [CrossRef]
- Paliwal, R.; Paliwal, S.R.; Kenwat, R.; Kurmi, B.D.; Sahu, M.K. Solid lipid nanoparticles: A review on recent perspectives and patents. Expert Opin. Ther. Pat. 2020, 30, 179–194. [Google Scholar] [CrossRef] [PubMed]
- Ghanbarzadeh, B.; Keivani, F.; Mohammadi, M. Encapsulation of food ingredients by solid lipid nanoparticles (SLNs). In Lipid-Based Nanostructures for Food Encapsulation Purposes; Elsevier: Gorgan, Iran, 2019; pp. 179–216. [Google Scholar] [CrossRef]
- Duong, V.A.; Nguyen, T.T.L.; Maeng, H.J. Preparation of solid lipid nanoparticles and nanostructured lipid carriers for drug delivery and the effects of preparation parameters of solvent injection method. Molecules 2020, 25, 4781. [Google Scholar] [CrossRef] [PubMed]
- Aditya, N.P.; Ko, S. Solid lipid nanoparticles (SLNs): Delivery vehicles for food bioactives. RSC Adv. 2015, 5, 30902–30911. [Google Scholar] [CrossRef]
- Trotta, M.; Debernardi, F.; Caputo, O. Preparation of solid lipid nanoparticles by a solvent emulsification–diffusion technique. Int. J. Pharm. 2003, 257, 153–160. [Google Scholar] [CrossRef]
- Trucillo, P.; Campardelli, R. Production of solid lipid nanoparticles with a supercritical fluid assisted process. J. Supercrit. Fluids 2019, 143, 16–23. [Google Scholar] [CrossRef]
- Zardini, A.A.; Mohebbi, M.; Farhoosh, R.; Bolurian, S. Production and characterization of nanostructured lipid carriers and solid lipid nanoparticles containing lycopene for food fortification. J. Food Sci. Technol. 2018, 55, 287–298. [Google Scholar] [CrossRef] [PubMed]
- Salminen, H.; Ankenbrand, J.; Zeeb, B.; Bönisch, G.B.; Schäfer, C.; Kohlus, R.; Weiss, J. Influence of spray drying on the stability of food-grade solid lipid nanoparticles. Food Res. Int. 2019, 119, 741–750. [Google Scholar] [CrossRef]
- Da Silva Santos, V.; Ribeiro, A.P.B.; Santana, M.H.A. Solid lipid nanoparticles as carriers for lipophilic compounds for applications in foods. Food Res. Int. 2019, 122, 610–626. [Google Scholar] [CrossRef] [PubMed]
- Tamjidi, F.; Shahedi, M.; Varshosaz, J.; Nasirpour, A. Nanostructured lipid carriers (NLC): A potential delivery system for bioactive food molecules. Innov. Food Sci. Emerg. Technol. 2013, 19, 29–43. [Google Scholar] [CrossRef]
- Haider, M.; Abdin, S.M.; Kamal, L.; Orive, G. Nanostructured lipid carriers for delivery of chemotherapeutics: A review. Pharmaceutics 2020, 12, 288. [Google Scholar] [CrossRef] [PubMed]
- Can, Q.; Johan, C.; Charlotta, T. Carotenoids particle formation by supercritical fluid technologies. Chin. J. Chem. Eng. 2009, 17, 344–349. [Google Scholar] [CrossRef]
- Subramaniam, B.; Siddik, Z.H.; Nagoor, N.H. Optimization of nanostructured lipid carriers: understanding the types, designs, and parameters in the process of formulations. J. Nanopart. Res. 2020, 22, 1–29. [Google Scholar] [CrossRef]
- Gomes, G.V.L.; Sola, M.R.; Rochetti, A.L.; Fukumasu, H.; Vicente, A.A.; Pinho, S.C. β-carotene and α-tocopherol coencapsulated in nanostructured lipid carriers of murumuru (Astrocaryum murumuru) butter produced by phase inversio n temperature method: Characterisation, dynamic in vitro digestion and cell viability study. J. Microencapsul. 2019, 36, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Maurya, V.K.; Aggarwal, M. A phase inversion based nanoemulsion fabrication process to encapsulate vitamin D3 for food applications. J. Steroid Biochem. 2019, 190, 88–98. [Google Scholar] [CrossRef]
- Hentschel, A.; Gramdorf, S.; Müller, R.H.; Kurz, T. β-Carotene-loaded nanostructured lipid carriers. J. Food Sci. 2008, 73, N1–N6. [Google Scholar] [CrossRef] [PubMed]
- Pezeshki, A.; Hamishehkar, H.; Ghanbarzadeh, B.; Fathollahy, I.; Nahr, F.K.; Heshmati, M.K.; Mohammadi, M. Nanostructured lipid carriers as a favorable delivery system for β-carotene. Food Biosci. 2019, 27, 11–17. [Google Scholar] [CrossRef]
- Azar, F.A.N.; Pezeshki, A.; Ghanbarzadeh, B.; Hamishehkar, H.; Mohammadi, M. Nanostructured lipid carriers: Promising delivery systems for encapsulation of food ingredients. J. Sci. Food Agric. 2020, 2, 100084. [Google Scholar] [CrossRef]
- Gasa-Falcon, A.; Odriozola-Serrano, I.; Oms-Oliu, G.; Martín-Belloso, O. Nanostructured lipid-based delivery systems as a strategy to increase functionality of bioactive compounds. Foods 2020, 9, 325. [Google Scholar] [CrossRef]
- Tamjidi, F.; Shahedi, M.; Varshosaz, J.; Nasirpour, A. EDTA and α-tocopherol improve the chemical stability of astaxanthin loaded into nanostructured lipid carriers. Eur. J. Lipid Sci. Technol. 2014, 116, 968–977. [Google Scholar] [CrossRef]
- Salminen, H.; Aulbach, S.; Leuenberger, B.H.; Tedeschi, C.; Weiss, J. Influence of surfactant composition on physical and oxidative stability of quillaja saponin-stabilized lipid particles with encapsulated ω-3 fish oil. Colloids Surf. B 2014, 122, 46–55. [Google Scholar] [CrossRef]
- Yang, C.; Yan, H.; Jiang, X.; Xu, H.; Tsao, R.; Zhang, L. Preparation of 9 Z-β-carotene and 9 Z-β-carotene high-loaded nanostructured lipid carriers: Characterization and storage stability. J. Agric. Food Chem. 2020, 68, 13844–13853. [Google Scholar] [CrossRef]
- Dima, C.; Assadpour, E.; Dima, S.; Jafari, S.M. Bioavailability of nutraceuticals: Role of the food matrix, processing conditions, the gastrointestinal tract, and nanodelivery systems. Compr. Rev. Food Sci. 2020, 19, 954–994. [Google Scholar] [CrossRef] [PubMed]
- Dowling, A.P. Development of nanotechnologies. Mater. Today 2004, 7, 30–35. [Google Scholar] [CrossRef]
- Chau, C.-F.; Wu, S.-H.; Yen, G.-C. The development of regulations for food nanotechnology. Trends Food Sci. Technol. 2007, 18, 269–280. [Google Scholar] [CrossRef]
- Amenta, V.; Aschberger, K.; Arena, M.; Bouwmeester, H.; Moniz, F.B.; Brandhoff, P.; Gottardo, S.; Marvin, H.J.; Mech, A.; Pesudo, L.Q. Regulatory aspects of nanotechnology in the agri/feed/food sector in EU and non-EU countries. Regul. Toxicol. Pharmacol. 2015, 73, 463–476. [Google Scholar] [CrossRef]
- Committee, E.S. Guidance on the risk assessment of the application of nanoscience and nanotechnologies in the food and feed chain. EFSA J. 2011, 9, 2140. [Google Scholar] [CrossRef]
- Livney, Y.D. Nanostructured delivery systems in food: Latest developments and potential future directions. Curr. Opin. Food Sci. 2015, 3, 125–135. [Google Scholar] [CrossRef]
- Hardy, A.; Benford, D.; Halldorsson, T.; Jeger, M.J.; Knutsen, H.K.; More, S.; Naegeli, H.; Noteborn, H.; Ockleford, C.; Ricci, A. Guidance on risk assessment of the application of nanoscience and nanotechnologies in the food and feed chain: Part 1, human and animal health. EFSA J. 2018, 16. [Google Scholar] [CrossRef]
- Akbari-Alavijeh, S.; Shaddel, R.; Jafari, S.M. In vivo assays for evaluating the release of nanoencapsulated food ingredients. In Release and Bioavailability of Nanoencapsulated Food Ingredients; Elsevier: Gorgan, Iran, 2020; pp. 179–207. [Google Scholar]
- Luo, Y.; Wang, Q.; Zhang, Y. Biopolymer-based nanotechnology approaches to deliver bioactive compounds for food applications: A perspective on the past, present, and future. J. Agric. Food Chem. 2020, 68, 12993–13000. [Google Scholar] [CrossRef]
- Müllertz, A.; Ogbonna, A.; Ren, S.; Rades, T. New perspectives on lipid and surfactant based drug delivery systems for oral delivery of poorly soluble drugs. J. Pharm. Pharmacol. 2010, 62, 1622–1636. [Google Scholar] [CrossRef]
- Santos, D.T.; Meireles, M.A. Carotenoid pigments encapsulation: Fundamentals, techniques and recent trends. Open Chem. Eng. J. 2010, 4, 42–50. [Google Scholar] [CrossRef]
- Salvia-Trujillo, L.; Verkempinck, S.; Rijal, S.K.; Van Loey, A.; Grauwet, T.; Hendrickx, M. Lipid nanoparticles with fats or oils containing β-carotene: Storage stability and in vitro digestibility kinetics. Food Chem. 2019, 278, 396–405. [Google Scholar] [CrossRef]
- Zambrano-Zaragoza, M.L.; Quintanar-Guerrero, D.; Del Real, A.; Piñon-Segundo, E.; Zambrano-Zaragoza, J.F. The release kinetics of β-carotene nanocapsules/xanthan gum coating and quality changes in fresh-cut melon (cantaloupe). Carbohydr. Polym. 2017, 157, 1874–1882. [Google Scholar] [CrossRef]
- Yang, Y.; McClements, D.J. Vitamin E bioaccessibility: Influence of carrier oil type on digestion and release of emulsified α-tocopherol acetate. Food Chem. 2013, 141, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Pouton, C.W.; Porter, C.J. Formulation of lipid-based delivery systems for oral administration: Materials, methods and strategies. Adv. Drug Deliv. Rev. 2008, 60, 625–637. [Google Scholar] [CrossRef] [PubMed]
- Yáñez, J.A.; Wang, S.W.; Knemeyer, I.W.; Wirth, M.A.; Alton, K.B. Intestinal lymphatic transport for drug delivery. Adv. Drug Deliv. Rev. 2011, 63, 923–942. [Google Scholar] [CrossRef] [PubMed]
- Yao, M.; Chen, J.; Zheng, J.; Song, M.; McClements, D.J.; Xiao, H. Enhanced lymphatic transport of bioactive lipids: Cell culture study of polymethoxyflavone incorporation into chylomicrons. Food Funct. 2013, 4, 1662–1667. [Google Scholar] [CrossRef] [PubMed]
- McClements, D.J. Edible lipid nanoparticles: Digestion, absorption, and potential toxicity. Prog. Lipid Res. 2013, 52, 409–423. [Google Scholar] [CrossRef] [PubMed]
- Borel, T.; Sabliov, C.M. Nanodelivery of bioactive components for food applications: Types of delivery systems, properties, and their effect on ADME profiles and toxicity of nanoparticles. Annu. Rev. Food Sci. Technol. 2014, 5, 197–213. [Google Scholar] [CrossRef] [PubMed]
- Ozturk, B.; Argin, S.; Ozilgen, M.; McClements, D.J. Nanoemulsion delivery systems for oil-soluble vitamins: Influence of carrier oil type on lipid digestion and vitamin D3 bioaccessibility. Food Chem. 2015, 187, 499–506. [Google Scholar] [CrossRef] [PubMed]
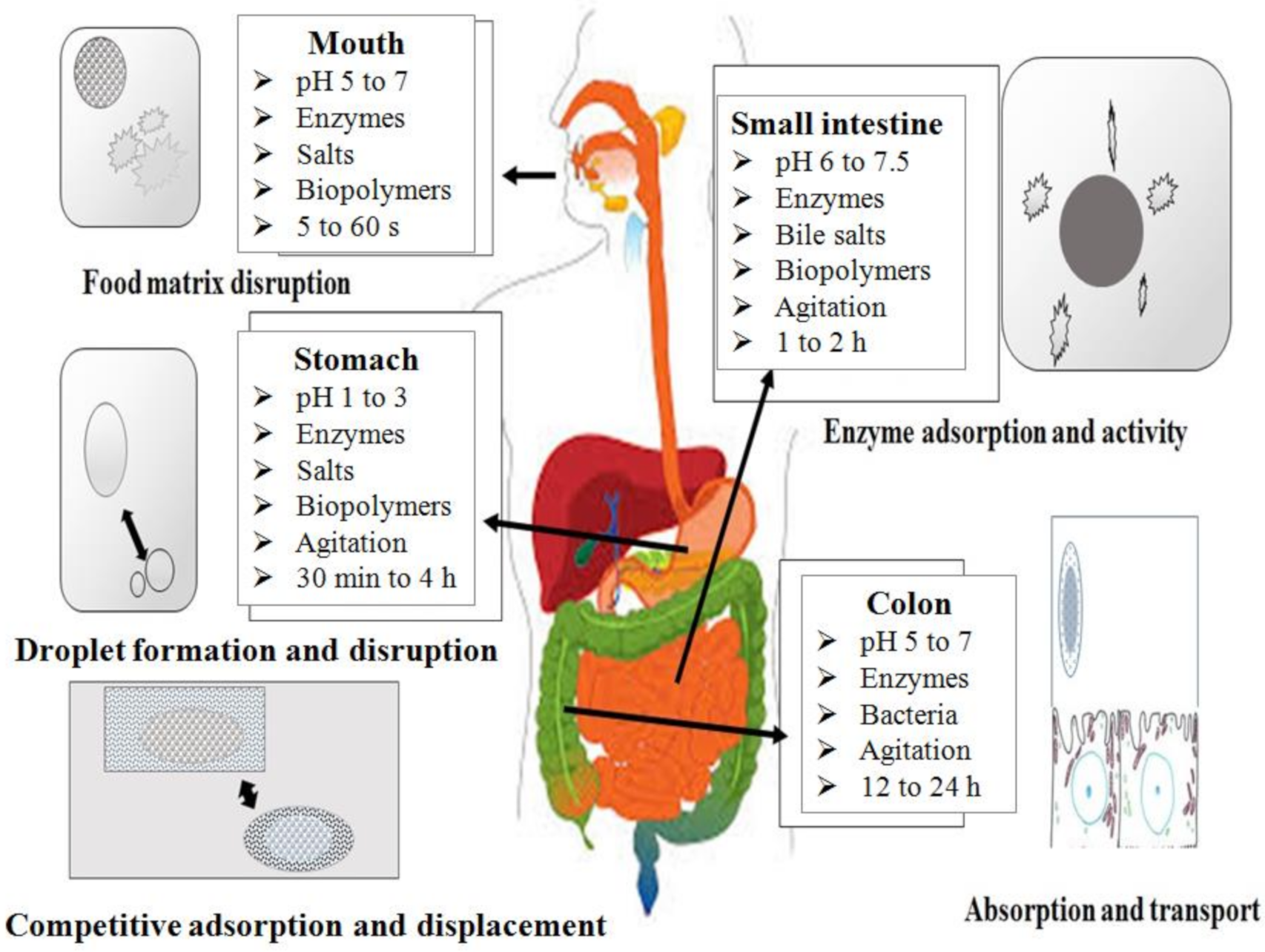

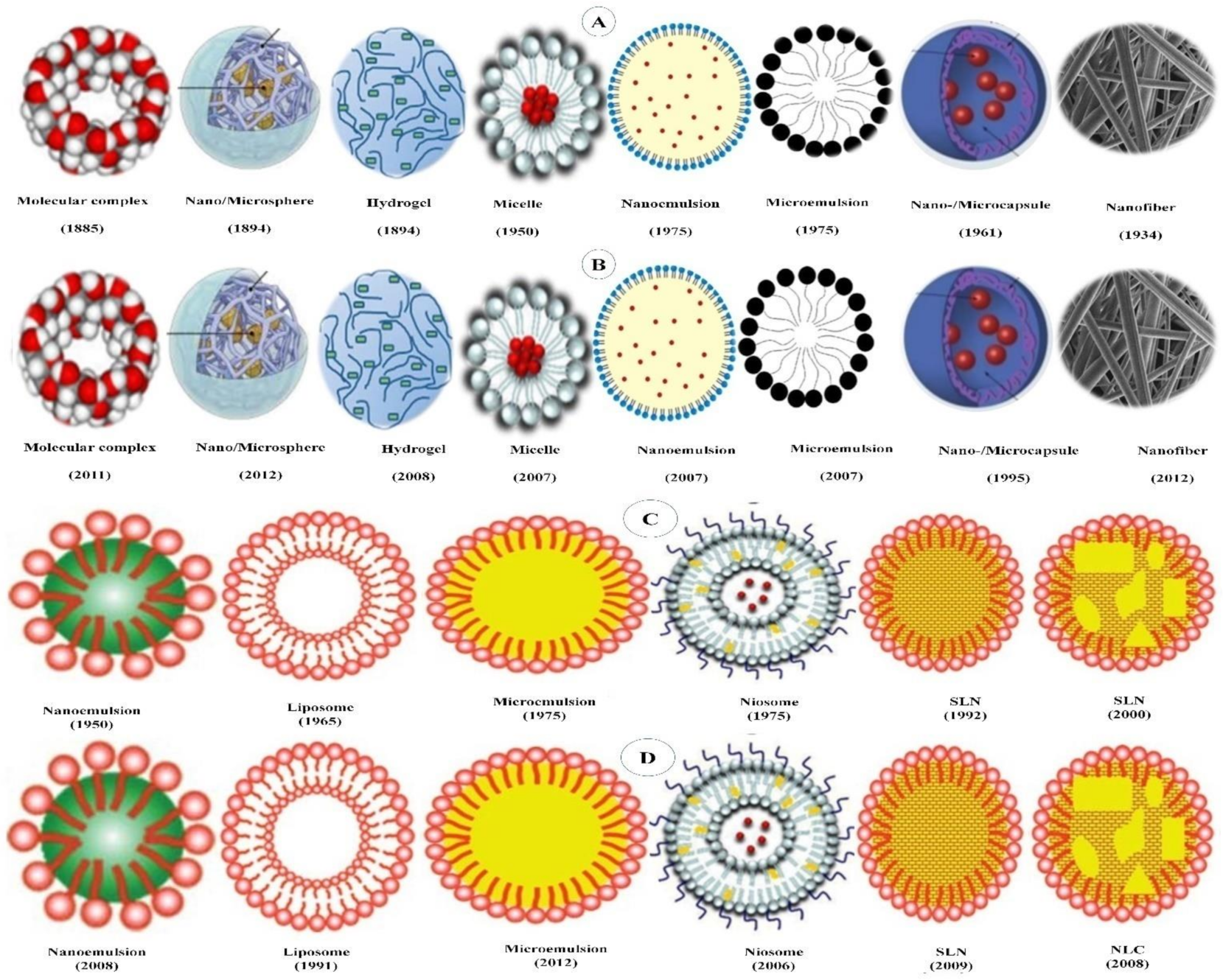
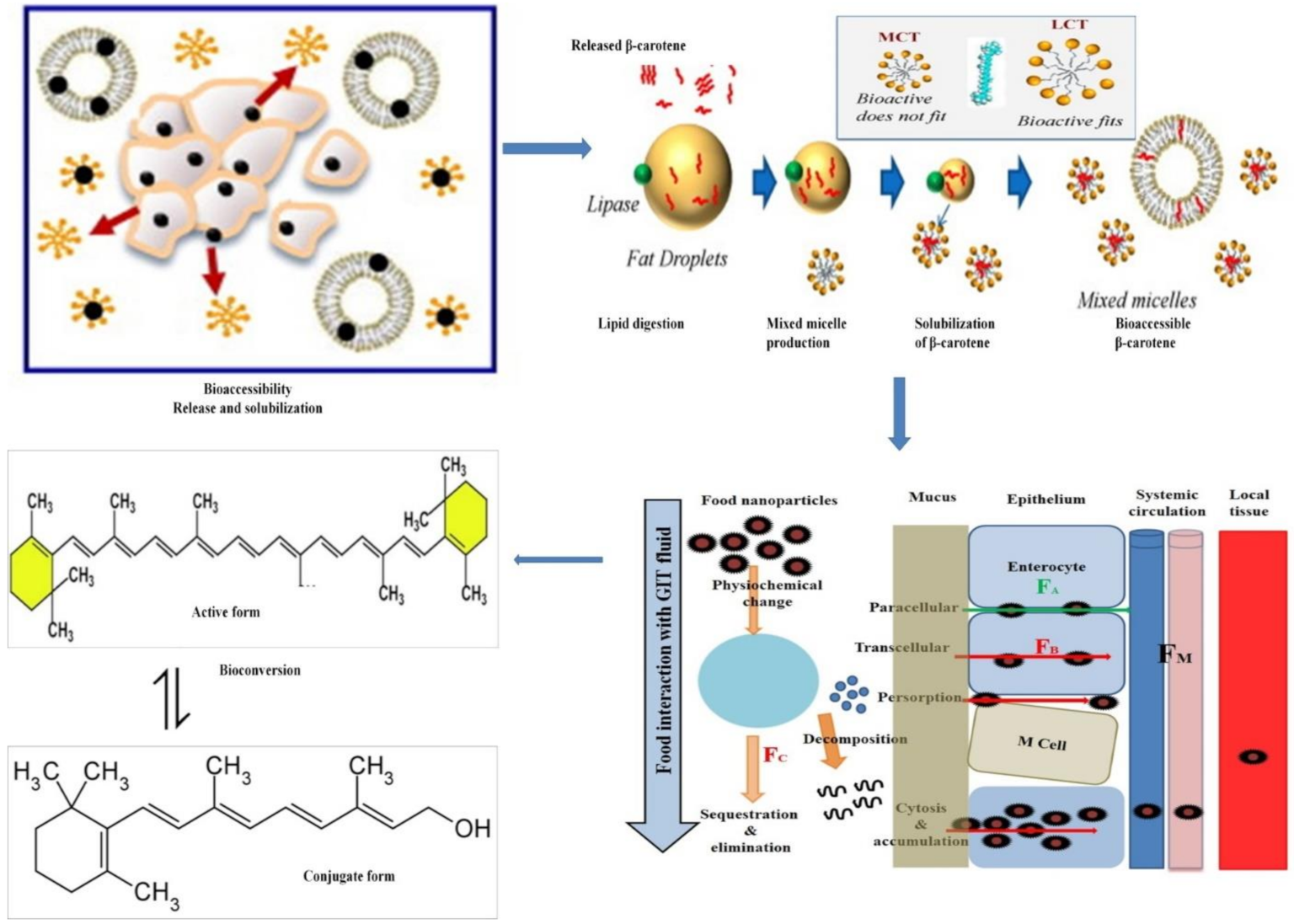
| Class of Delivery Systems | Subclass of Delivery System | Delivery System | Ingredients | Technique/Preparation Method | Physiochemical Studies | Encapsulation Efficiency | Release Studies | Particle Size | Cellular/Animal Studies | Applications | References |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Lipid-derived delivery systems | Self-assembled delivery system | Liposome |
| Ethanol injection method | FTIR, SEM, Raman microspectroscopy, UV-vis irradiation | NA | NA | NA | NA | NA | [117] |
| Dehydration/rehydration method | NA | NA | NA | NA | Microsomes/Rat | pharmaceutical | [118] | |||
| Spray-drying | DSC, small-angle X-ray scattering (SAXS), TEM, DLS, ELS | NA | NA | 700–3000 nm | NA | NA | [119] | |||
| Modified thermal method | FTIR | NA | NA | 64–500 nm | NA | NA | [120] | |||
| Dehydration/rehydration method | NA | NA | NA | NA | Hamster | pharmaceutical | [121] | |||
| Spray-drying | DLS, ELS, XRD, SEM | NA | NA | 285–1695 nm | NA | NA | [122] | |||
| Thin-film evaporation method | DLS, AFM | NA | SGF, SIF | 600 nm | NA | NA | [123] | |||
| Niosome |
| Dehydration/rehydration method | DLS, EE, TEM | 16.0–51% | NA | 273.2–367.9 nm | RAT-1 immortalized fibroblasts | pharmaceutical | [124] | ||
| Particulate delivery systems | Solid lipid nanoparticles |
| Hot homogenization | DLS, DSC, ELS, Cryo TEM, NMR, XRD | NA | NA | 111.7–170.8 nm | NA | NA | [125] | |
| Microfluidization | DLS, TEM | 99.1%,98.8%, | NA | 77.8–190.9 nm | Caco-2 cells | NA | [126] | |||
| Hot high-pressure homogenization | DLS, ELS, DSC | NA | NA | 0.16–0.27µm | NA | NA | [127] | |||
| Homogenization | DLS, ELS, TEM, SEM | NA | NA | <200 µm | NA | NA | [128] | |||
| Hot homogenization | DLS | NA | NA | 1.20–1.70 µm | NA | Food application (ice creams) | [129] | |||
| Phase-inversion temperature | DLS, DSC | NA | NA | 109 and 128 nm | NA | NA | [130] | |||
| Hot pressure homogenization | DLS, ELS, DSC | NA | NA | NA | NA | NA | [131] | |||
| Hot high-pressure homogenization method | DLS, DCS, NMR | NA | NA | 168–227 nm | NA | NA | [132] | |||
| Phase-inversion temperature | AFM, DLS, DSC, XRD | NA | NA | <400 nm | NA | NA | [133] | |||
| Hot agitation | DLS, DSC, XRD | NA | NA | <5 µm | NA | NA | [134] | |||
| Nanostructured lipid carriers |
| Solvent displacement technique | DLS, DSC | NA | NA | 500 nm | NA | NA | [135] | ||
| Hot homogenization | DLS, DSC | NA | NA | 82–217 nm. | NA | NA | [136] | |||
| Hot homogenization | DLS, ELS, DSC, TEM | 65.26–74.35% | NA | 85.2–129.2 nm | NA | NA | [137] | |||
| Phase-inversion temperature | DLS, DSC, TEM | NA | Gastric fluid, Duodenal fluid, Jejunal fluid, Ileal fluid | 31.6–34.08 nm | NA | NA | [138] | |||
| Microemulsion |
| Spontaneous emulsification method | DLS, ELS | NA | NA | 20–22.60 nm | NA | NA | [139] | ||
| Microfluidization | DLS, ELS | NA | NA | <250 nm | NA | NA | [140] | |||
| Microchannel Device | DLS | NA | na | 27.9 µm | NA | NA | [141] | |||
| Hot homogenization | DLS, DSC, ELS, Cryo TEM, NMR, XRD | NA | na | 115 nm | NA | NA | [125] | |||
| Microfluidization | DLS, CFFM | NA | SSF, SGF | 0.21–23 µm | NA | NA | [142] | |||
| Nano emulsion |
| Microfluidization | DLS | NA | SSF, SGF, SIF | <150 nm | NA | NA | [91] | ||
| Microfluidization | DLS | NA | SSF, SGF, SIF | 140–170 nm | NA | NA | [92] | |||
| Hot homogenization | DLS | NA | SSF, SGF, SIF | <200 nm | NA | NA | [143] | |||
| Microfluidization | DLS, ELS | NA | NA | 97.2–416.0 nm | NA | NA | [144] | |||
| Supercritical fluid | DLS | NA | NA | 50–150 nm | NA | NA | [145] | |||
| Homogenization | DLS | NA | SSF, SGF, SIF | 1.25–1.34 µm | NA | NA | [146] | |||
| High-speed homogenization | DLS | NA | NA | 418.8–1689.0 nm | NA | NA | [147] | |||
| Microfluidization | DLS, DSC | NA | SSF, SGF, SIF | 0.2–23 µm | NA | NA | [142] | |||
| Hot-high shear homogenization | DLS, ELS | NA | NA | 79–115 nm | NA | NA | [148] | |||
| Microfluidization | DLS, ELS, DSC | NA | SSF, SGF, SIF | 146 to 415 nm, | NA | NA | [149] | |||
| Heating and stirring | DLS | NA | NA | <10 nm | NA | NA | [150] | |||
| Microfluidization | DLS | NA | NA | <100 nm | NA | NA | [151] | |||
| Spontaneous emulsionfication method | DLS, SEM | NA | NA | 100–300 nm | NA | NA | [152] | |||
| Polymer-derived delivery systems | Self-assembled polymer-derived delivery systems | Starch-based emulsion |
| Cross-linking | SEM | NA | NA | 700 nm | NA | NA | [153] |
| Spray-drying | DLS, ELS, SEM | NA | NA | 114–118 nm, | NA | NA | [154] | |||
| Microfluidization | DLS, ELS | NA | NA | 208–385 nm | NA | NA | [140] | |||
| High-pressure homogenization | DLS | NA | SGF, SIF | 17 nm | NA | NA | [155] | |||
| Ultrasound emulsification | SEM | NA | NA | 300–600 nm | NA | NA | [156] | |||
| Layer-by-layer electrostatic deposition method | DLS, ELS | NA | NA | 250.0–306.3 nm 304.5–466.6 nm | NA | NA | [157] | |||
| Microfluidization | DLS | NA | SGF, SIF | 80.0 ± 1.3 nm | NA | NA | [158] | |||
| Protein-based emulsion |
| Solvent-displacement method | DLS, ELS | NA | NA | 45–127 nm | NA | NA | [159] | ||
| Spontaneous emulsification | DLS, SEM | 100 ± 1% | NA | 50–500 nm | NA | NA | [160] | |||
| Microfluidization | CD, DLS, ELS | NA | NA | 158.8 and 162.7 nm | NA | NA | [161] | |||
| Homogenization | DLS, DSC | NA | NA | 0.48–0.66 µm | NA | NA | [162] | |||
| High speed homogenization | DLS, XRD | NA | NA | 0.46–0.50 µm | NA | NA | [163] | |||
| pH-cycling method | DLS, ELS, FTIR, SEM | NA | SGF, SIF | 409.7 nm | NA | NA | [164] | |||
| Microfluidization | DLS, ELS | NA | NA | 160–170 nm | NA | NA | [165] | |||
| Layer-by-layer electrodeposition technique | DSC, Dynamic Mechanical Analyses (DMA) | NA | NA | NA | NA | NA | [166] | |||
| Microfluidization | DLS | NA | NA | 124–368 nm | NA | NA | [167] | |||
| Solvent displacement technique | DLS, ELS | NA | NA | 30–206 nm | NA | NA | [168] | |||
| Homogenization | CD, DLS, ELS, FSS (Fluorescence spectroscopy) | NA | NA | 302–583 nm | NA | NA | [169] | |||
| Microfluidization | DLS, ELS, FSS | NA | SGF, SIF | 0.48–1.87 µm | NA | NA | [170] | |||
| Microfluidization | DLS | 70.9% | SGF, SIF | 167.4–178.8 nm | Caco-2,Cell toxicity | Pharmaceutical | [171] | |||
| Carbohydrate-based emulsion |
| Sonication and hot homogenization | DLS, ELS, CFSM | NA | SSF, SGF, SIF | 0.2–23 µm | NA | NA | [172] | ||
| Freeze-dryer | DSC | NA | NA | NA | NA | NA | [173] | |||
| Micelle |
| Solvent displacement | DLS, ELS, FSEM | NA | NA | 13–171 nm | NA | NA | [174] | ||
| Spontaneous emulsification | DLS, SEM | 100 ± 1% | NA | 50–500 nm | NA | NA | [160] | |||
| Sonication | DLS, FTIR, NMR, SEM, TEM | 84.67% | SSF, SGF, SIF | 20–50 nm | NA | NA | [175] | |||
| Microfiltration | DLS, FTIR, TEM | NA | NA | 0.04–0.4 µm | NA | NA | [176] | |||
| Polymerization | DLS, FTIR, NMR, XRD, TEM | NA | NA | 14 nm | NA | NA | [177] | |||
| Homogenization | DLS, ELS, TEM | NA | NA | 12–100 nm. | Caco-2, Cell toxicity study | Food application | [178] | |||
| Solvent displacement method | DLS, ELS | NA | NA | 0.087–1.158 µm | NA | NA | [179] | |||
| Particulate nanoparticles | Molecular complex |
| Co-precipitation and physical mixture techniques | FTIR, FESEM | NA | NA | NA | NA | NA | [113] | |
| Spray-drying | SEM | NA | PBS | NA | NA | NA | [115] | |||
| Sonication | DLS, TEM, SEM, XRD | 65% | NA | 12 ± 3 nm | NA | NA | [116] | |||
| Nanosphere |
| Nanoprecipitation method | DLS, ELS | NA | PBS | 117.1 ± 4.6 nm | MCF-7 breast cancer cells, Cell toxicity studies | Pharmaceutical | [180] | ||
| Homogenization | CD, DLS, FTIR, CLSM | NA | SGF, SIF | 300−400 nm | NA | NA | [181] | |||
| Microfluidization | DLS, ELS, TEM | NA | SGF, SIF | 32.44 ± 0.87–168.17 ± 22.36 nm | NA | Food application (milk) | [182] | |||
| Microfluidization | DLS, ELS, Raman-FIB-SEM | NA | NA | 46.77 ± 0.17–48.23 ± 0.13 µm | NA | NA | [183] | |||
| Nanoprecipitation method | DLS, DSC, XRD | NA | SIF | 0.77–0.89 µm | NA | NA | [184] | |||
| Homogenization | DLS, SEM, TEM, NMR | 22.60–28.08% | Water, Buffer | <100 nm | NA | NA | [185] | |||
| High-temperature, high-pressure emulsification and antisolvent precipitation | DLS | 70–80% | NA | 137–135,900 nm | NA | NA | [186] | |||
| Homogenization-evaporation method | DLS, DSC, ELS, FTIR, XRD | NA | SGF, SIF | NA | Caco-2 cells | NA | [187] | |||
| Microsphere |
| Precipitation | DLS, SEM | 65–90% | NA | 300–600 nm | NA | NA | [188] | ||
| Ionic gelation | DLS | NA | SGF, SIF | 80–94 nm, 91–106 nm, 128–134 nm | NA | NA | [189] | |||
| Glycosylation conjugation | CD, DLS, ELS | NA | SGF, SIF | 165.6–176.0 nm | NA | NA | [190] | |||
| High pressure homogenization | DLS | NA | NA | 1.38–1.96 mm. | NA | NA | [191] | |||
| Electrostatic complexation | DSC, FTIR | NA | NA | NA | NA | NA | [192] | |||
| Syringe microfluidization | SEM | NA | NA | 19–84 µm | NA | NA | [193] | |||
| Microfluidization and Spray-drying | DLS, ELS | NA | NA | 230–277 nm | NA | NA | [194] | |||
| Ionic gelation | Lipid lipolysis | NA | NA | NA | NA | NA | [195] | |||
| Freeze-drying | AFM DLS, ELS | NA | SGF, SIF | 55 nm | NA | NA | [196] | |||
| Spontaneous emulsification | DLS | 14.18–64.39% | NA | 655–3418 nm | NA | NA | [197] | |||
| Electrospinning | SEM | NA | NA | NA | NA | NA | [198] | |||
| Microfiltration | DLS, FTIR, TEM | NA | NA | 0.04–0.4 µm | NA | NA | [176] | |||
| Spray-drying and freeze-drying | DLS | 66–70% | Sunflower oil | 1.20–2.30 µm | NA | NA | [199,200] | |||
| Freeze-drying | DLS | 66–70% | Sunflower oil | 2.10–3.2 µm | NA | NA | [200] | |||
| Spray-drying | Photodegradation study | NA | NA | NA | NA | NA | [201] | |||
| Microfluidization | DLS, ELS, FESEM | 90% | NA | 165.0–129.1 nm | NA | NA | [202] | |||
| Dry heating method | DLS, DSC, FSM | 73.64–74.53 | SGF, SIF | 111.1–127.3 nm | NA | NA | [203] | |||
| Microfluidization | DLS, ELS | NA | NA | 0.14–0.16 μm | NA | NA | [204] | |||
| High pressure homogenization | DLS | NA | NA | 176.3–228 nm | CACO-2 CELLS, RATS | PHARMACEUTICAL AND FOOD | [205] | |||
| High-pressure homogenization | DLS, ELS | NA | SGF, SIF | 142 ± 6–160 ± 10 nm | CACO-2 CELLS | NA | [206] | |||
| High-pressure homogenization | DLS, LD, TEM | NA | NA | 262.8 ± 4.10–307.1 ± 5.40 nm | NA | NA | [207] | |||
| Homogenization | DLS, DSC, ELS, FFS | NA | NA | 1–1.5 µm | NA | NA | [208] | |||
| Cross-linking and sonication | DLS, SEM | NA | NA | 1570.0 nm. | NA | Food application (hamburger patties) | [209] | |||
| Microfluidization | DLS | NA | SSF, SGF, SIF | 0.82 µm | NA | NA | [210] | |||
| Rotating evaporation | DLS, DSC, ELS, FTIR, SEM | 56.5–92.7% | SGF, SIF | 70.41 ± 0.67–420.9 ± 2.34 nm | NA | NA | [211] | |||
| Microfluidization | DLS, ELS | NA | NA | 123.9–207.2 nm | NA | NA | [212] | |||
| Microfluidization | DLS, ELS | NA | NA | 123.9–207.2 nm | NA | NA | [212] | |||
| Polymerization | DLS | NA | SGF, SIF | 127–149 nm | NA | NA | [213] | |||
| Homogenization and coacervation pr | DLS, ELS, FTIR, SEM | 65.95 ± 5.33% | SGF, SIF | 176.47± 4.65 µm | NA | NA | [214] | |||
| High-pressure homogenization | DLS | NA | NA | 10.1 ± 0.7–14.5 ± 0.6 nm | NA | NA | [215] | |||
| High-pressure homogenization | DLS | NA | SGF, SIF | 170 nm | NA | NA | [216] | |||
| High pressure homogenization | Effect of digestion on particle size | NA | SSF, SGF, SIF | NA | NA | NA | [217] | |||
| Spray-drying | Stability of carotene in powder | NA | NA | NA | NA | Food application | [218] | |||
| Solvent evaporation | DLS | 14% | NA | 260 nm | NA | Pharmaceutical | [219] | |||
| Homogenization and sonication | DLS, SEM | 79.63 ±1.41–84.32 ± 1.08% | SGF, | 210.5 1.23 nm | NA | NA | [220] | |||
| Homogenization | DLS, ELS | NA | NA | 0.52 µm. | NA | NA | [221] | |||
| Supercritical carbon dioxide micronization technique | NA | NA | organic solvent | NA | NA | NA | [222] | |||
| Capsular nanoparticles | Microcapsule |
| Freeze-drying | CFLM, DLS, ELS | NA | SGF, SIF | 0.23 ± 0.02–0.24 ± 0.01 µm | NA | NA | [115] | |
| Homogenization | Stabilitystudy | NA | NA | NA | NA | NA | [223] | |||
| Spray-drying | NA | NA | NA | NA | NA | NA | [224] | |||
| High speed homogenization, spray-drying | DLS, SEM | NA | NA | 114–159 nm | NA | NA | [154] | |||
| Spray-drying | DLS, SEM | NA | NA | 19.69–20.98 µm | mouse bone marrow and peripheral blood cells/Wistar albino rats, | Pharmaceuticals | [225] | |||
| Emulsification–diffusion method | DLS, ELS, SEM | NA | NA | 250–650 nm. | NA | NA | [226] | |||
| Complex coacervation | CLSM, DLS, FTIR, SEM, TGA, XRD | 79.36±0.541% | SGF | 159.71±2.16 µm | NA | NA | [227] | |||
| Spray-drying | DLS | 34–55% | SIF | 852–958 μm | NA | NA | [228] | |||
| Homogenization | CLSM | NA | NA | 0.23 ± 0.02–6.68 ± 0.65 lm | NA | NA | [229] | |||
| Supercritical fluid | SEM | NA | NA | NA | NA | NA | [230] | |||
| Supercritical fluid | SEM | 0.95–55.54% | NA | 1.3–51.9 µm | NA | NA | [231] | |||
| Solvent displacement technique | SEM, XRD | 78.74–81.2% | PBS | NA | NA | NA | [232] | |||
| Precipitation | DLS, DSC, TEM, XRD | NA | SGF | 16–30 nm | NA | NA | [233] | |||
| Freeze-dryer | DSC | NA | NA | NA | NA | NA | [234] | |||
| Homogenization and evaporation | CFLS, DLS | NA | NA | 161.98 ± 17.19–189.45 ± 22.69 nm | NA | NA | [235] | |||
| Membrane emulsification | DLS | NA | NA | 1.28 ± 0.02–1.69 ± 0.49 µm | NA | NA | [236] | |||
| Spray-drying | DLS, SEM | NA | Water | 4.9 + 2.4–6.0 + 3.0 µm | NA | NA | [237] | |||
| Spray-drying | DLS, DSC | NA | NA | 0.2–0.8 µm | NA | NA | [238] | |||
| Nanocapsule |
| Emulsification–diffusion method | DLS, ELS, SEM | NA | NA | 250–650 nm | NA | NA | [226] | ||
| polymer method (Nanoinjection and stirring) | DSC, ELS, TEM | 99.65–99.75% | NA | 142.33–190.33 nm | NA | NA | [239] | |||
| Homogenization and ultrasonication | DLS, DSC, SEM, XRD | 2.23±1.42% | PBS | 255.9±1.63 nm | NA | NA | [240] | |||
| Fibrous nanoparticles | Nanofiber |
| Electrospinning | DSC, FTIR, SEM | NA | NA | NA | NA | MA | [241] | |
| Spray-drying | DLS, SEM | NA | SSF, SGF, SIF | 10.5–942.8 µm | NA | Food application | [242] | |||
| Electrospinning | SEM | NA | NA | NA | NA | MA | [198] | |||
| Nanotube |
| Electrospinning | FTIR, SEM, RSM | NA | NA | 250 nm | NA | NA | [117] | ||
| Gelatinous nanoparticles | Hydrogel |
| Freeze-drying | SEM | NA | PBS | NA | NA | NA | [115] | |
| Cross-linking | SEM | NA | SGF | NA | NA | NA | [153] | |||
| Solvent-displacement method | DLS, ELS | NA | NA | 45–127 nm | NA | NA | [159] | |||
| Microfluidization | CFLS, DLS, ELS | NA | SSF, SGF, SIF | 285–660 mm | NA | NA | [243] | |||
| Microfluidization | CFSL | NA | SSF, SGF, SIF | 450 nm | NA | NA | [244] | |||
| Heating and stirring | Bioaccessibility | NA | SSF, SGF, SIF | NA | NA | NA | [245] | |||
| Microfluidization | DLS | NA | SSF, SGF, SIF | 3.16–22.1 µm | NA | NA | [246] | |||
| Spontaneous emulsification | Bioaccessibility, DLS | NA | SSF, SGF, SIF | 79–138 nm | NA | NA | [247] | |||
| Ultrasonic emulsification | CFSL, DLS, ELS | NA | SGF | 78–252 nm | NA | NA | [248] | |||
| Microfluidization | CFLS, DLS | NA | NA | 1.5–9.7 µm | NA | NA | [249] | |||
| Hot homogenization | CFSL, Bioaccessibility | NA | SSF, SGF, SIF | NA | NA | NA | [250] |
| ENMS | Class of DeliverySystem | Subclass of Delivery System | Ability to Deliver Lipophilic and Lipophobic BA | Physical Stability | Biological Stability | Biocompatibility | Drug Targeting | Drug Loading | Feasibility to be Delivery System for β-Carotene |
|---|---|---|---|---|---|---|---|---|---|
| Lipid-derived delivery system | Self-assembled delivery system | Liposome | Yes | poor | Poor | Good | Moderate | Low to moderate | Poor |
| Niosome | Yes | moderate | Poor | Moderate | Moderate | Moderate | Poor | ||
| Particulate | Solid lipid nanoparticles | Only lipophilic | Good | Moderate | Good | Moderate | Moderate | Moderate | |
| Nanostructured lipid carriers | Only lipophilic | Good | High | Good | Moderate | High | Good | ||
| Emulsion | Microemulsion | Yes | Moderate | Moderate | Good | Poor | High | Good | |
| Nanoemulsion | Yes | poor | Moderate | Good | Poor | High | Poor | ||
| Polymer-derived delivery system | Self-assembled delivery system | Starch-based Micelle | Yes | Good | Good | Moderate | Poor | Poor | Good |
| Protein-based micelles | Yes | Poor | Good | Moderate | Moderate | Poor | Good | ||
| Carbohydrate | Poor | ||||||||
| Hydrogel | Yes | Good | Good | Poor | Poor | Poor | Good | ||
| Colloidal nanoemulsion | Yes | Moderate | Moderate | Good | Poor | High | moderate | ||
| Nanoemulsion | Yes | poor | Moderate | Good | Poor | High | Poor | ||
| Molecular complexes | Only lipophilic | Good | Moderate | Poor | Poor | Low | Poor | ||
| Particulate | Protein inclusion complexes | Yes | Good | Moderate | Moderate | Moderate | Low | Poor | |
| Nanosphere | Yes | Good | Moderate | Moderate | Moderate | Moderate | Poor | ||
| Microsphere | Yes | Good | Moderate | Moderate | Moderate | Low | Moderate | ||
| Fibrous | Nanofiber | Yes | Good | Moderate | Moderate | Moderate | Low | Poor | |
| Capsular | Microcapsule | Yes | Good | Moderate | Moderate | Moderate | Low | Poor | |
| Nanosphere | Yes | Good | Moderate | Moderate | Moderate | Moderate | Poor |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maurya, V.K.; Shakya, A.; Aggarwal, M.; Gothandam, K.M.; Bohn, T.; Pareek, S. Fate of β-Carotene within Loaded Delivery Systems in Food: State of Knowledge. Antioxidants 2021, 10, 426. https://doi.org/10.3390/antiox10030426
Maurya VK, Shakya A, Aggarwal M, Gothandam KM, Bohn T, Pareek S. Fate of β-Carotene within Loaded Delivery Systems in Food: State of Knowledge. Antioxidants. 2021; 10(3):426. https://doi.org/10.3390/antiox10030426
Chicago/Turabian StyleMaurya, Vaibhav Kumar, Amita Shakya, Manjeet Aggarwal, Kodiveri Muthukaliannan Gothandam, Torsten Bohn, and Sunil Pareek. 2021. "Fate of β-Carotene within Loaded Delivery Systems in Food: State of Knowledge" Antioxidants 10, no. 3: 426. https://doi.org/10.3390/antiox10030426
APA StyleMaurya, V. K., Shakya, A., Aggarwal, M., Gothandam, K. M., Bohn, T., & Pareek, S. (2021). Fate of β-Carotene within Loaded Delivery Systems in Food: State of Knowledge. Antioxidants, 10(3), 426. https://doi.org/10.3390/antiox10030426