SPOTing Acetyl-Lysine Dependent Interactions
Abstract
:1. Introduction
1.1. Validation of Commercial Antibodies Using Peptide Arrays
1.2. Identification of Acetyl-Histone Peptide Interactions Using Peptide Arrays
2. Experimental Section
2.1. Membrane Synthesis
2.2. Choice of Membrane Material
2.3. Control of SPOT Density
2.4. Membrane Blocking
2.5. Probing Protein:Peptide Interactions
2.6. Quantification and Visualization
2.7. Membrane Stripping
2.8. Biolayer Interferometry (BLI)
2.9. Experimental Protocol
- His-tag® Antibody HPR conjugated, Novagen, distributed by Merck-Millipore, #71841.
- Pierce® ECL Western blotting Substrate, Thermo Scientific distributed by Fisher Scientific, #32106
- Bovine Serum Albumin Heat Shock Reagent grade powder pH7.0, Fisher Chemical, BPE 1600-100.
- Restore Western Blot Stripping Buffer, Thermo Scientific, #21059.
3. Results and Discussion
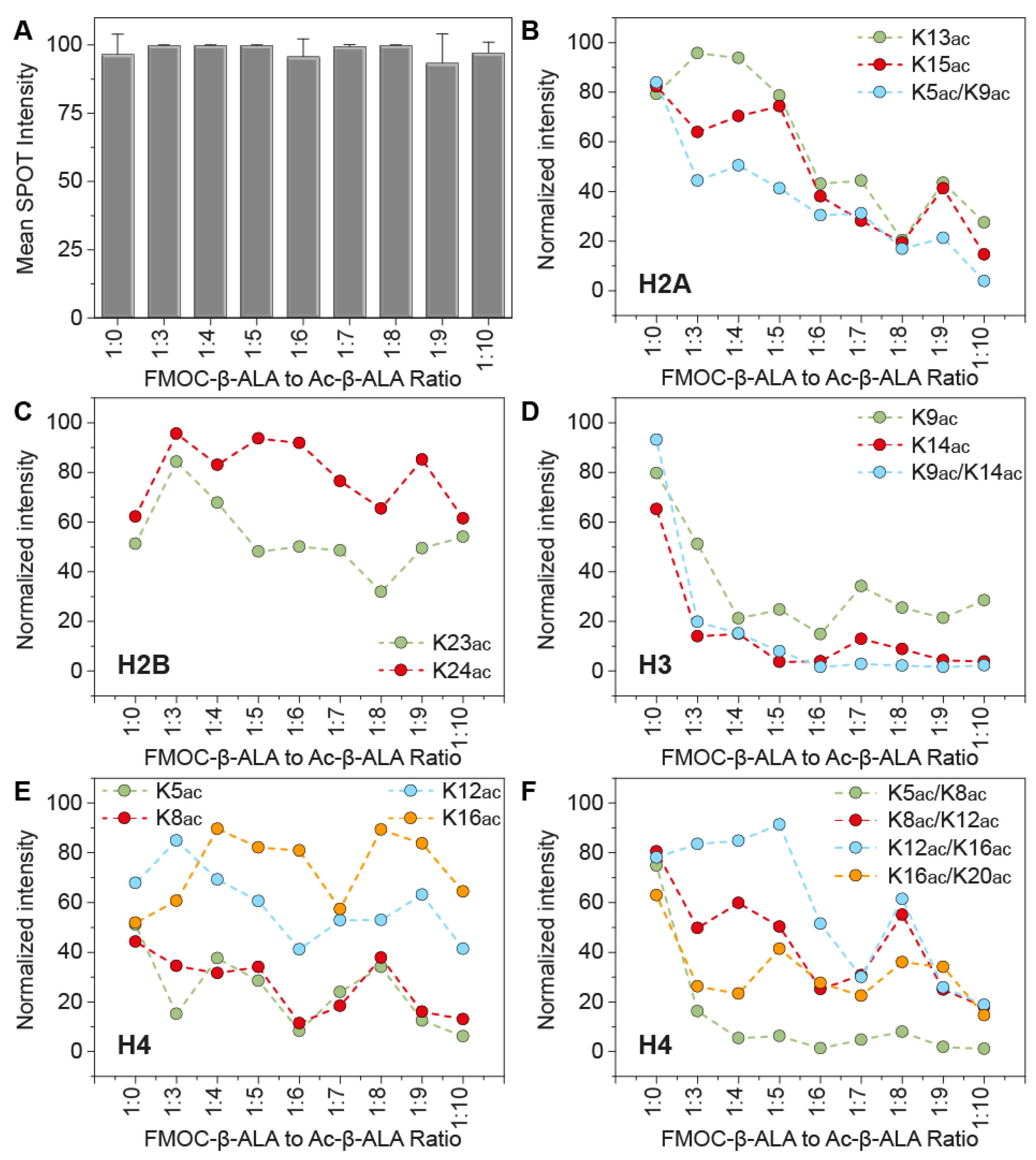
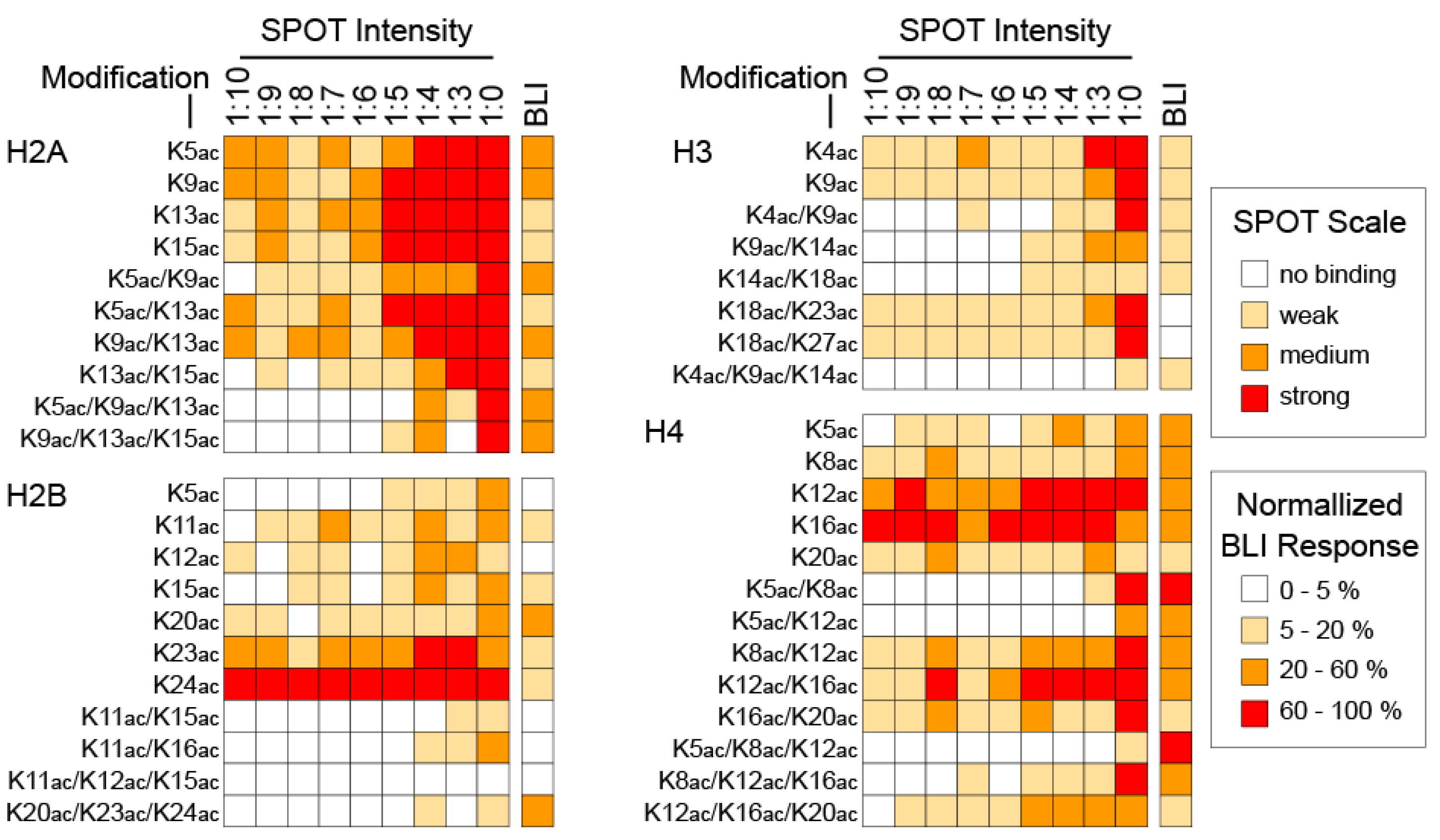
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Choudhary, C.; Kumar, C.; Gnad, F.; Nielsen, M.L.; Rehman, M.; Walther, T.C.; Olsen, J.V.; Mann, M. Lysine acetylation targets protein complexes and co-regulates major cellular functions. Science 2009, 325, 834–840. [Google Scholar] [CrossRef] [PubMed]
- Kouzarides, T. Acetylation: A regulatory modification to rival phosphorylation? EMBO J. 2000, 19, 1176–1179. [Google Scholar] [CrossRef] [PubMed]
- Filippakopoulos, P.; Picaud, S.; Mangos, M.; Keates, T.; Lambert, J.P.; Barsyte-Lovejoy, D.; Felletar, I.; Volkmer, R.; Muller, S.; Pawson, T.; et al. Histone recognition and large-scale structural analysis of the human bromodomain family. Cell 2012, 149, 214–231. [Google Scholar] [CrossRef] [PubMed]
- Frank, R. Spot-Synthesis—An easy technique for the positionally addressable, parallel chemical synthesis on a membrane support. Tetrahedron 1992, 48, 9217–9232. [Google Scholar] [CrossRef]
- Volkmer, R.; Tapia, V.; Landgraf, C. Synthetic peptide arrays for investigating protein interaction domains. FEBS Lett. 2012, 586, 2780–2786. [Google Scholar] [CrossRef] [PubMed]
- Volkmer, R. Synthesis and application of peptide arrays: Quo vadis SPOT technology. Chembiochem 2009, 10, 1431–1442. [Google Scholar] [CrossRef] [PubMed]
- Molina, F.; Laune, D.; Gougat, C.; Pau, B.; Granier, C. Improved performances of spot multiple peptide synthesis. Pept. Res. 1996, 9, 151–155. [Google Scholar] [PubMed]
- Ay, B.; Streitz, M.; Boisguerin, P.; Schlosser, A.; Mahrenholz, C.C.; Schuck, S.D.; Kern, F.; Volkmer, R. Sorting and pooling strategy: A novel tool to map a virus proteome for CD8 T-cell epitopes. Pept. Sci. 2007, 88, 64–75. [Google Scholar] [CrossRef] [PubMed]
- Cushman, I.; Palzkill, T.; Moore, M.S. Using peptide arrays to define nuclear carrier binding sites on nucleoporins. Methods 2006, 39, 329–341. [Google Scholar] [CrossRef] [PubMed]
- Nady, N.; Min, J.; Kareta, M.S.; Chédin, F.; Arrowsmith, C.H. A SPOT on the chromatin landscape? Histone peptide arrays as a tool for epigenetic research. Trends Biochem. Sci. 2008, 33, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Luo, H.; Lee, S.; Jin, F.; Yang, J.S.; Montellier, E.; Buchou, T.; Cheng, Z.; Rousseaux, S.; Rajagopal, N.; et al. Identification of 67 histone marks and histone lysine crotonylation as a new type of histone modification. Cell 2011, 146, 1016–1028. [Google Scholar] [CrossRef] [PubMed]
- Rathert, P.; Dhayalan, A.; Murakami, M.; Zhang, X.; Tamas, R.; Jurkowska, R.; Komatsu, Y.; Shinkai, Y.; Cheng, X.; Jeltsch, A. Protein lysine methyltransferase G9a acts on non-histone targets. Nat. Chem. Biol. 2008, 4, 344–346. [Google Scholar] [CrossRef] [PubMed]
- Tapia, V.; Ay, B.; Triebus, J.; Wolter, E.; Boisguerin, P.; Volkmer, R. Evaluating the coupling efficiency of phosphorylated amino acids for SPOT synthesis. J. Pept. Sci.: Off. Publ. Eur. Pept. Soc. 2008, 14, 1309–1314. [Google Scholar] [CrossRef] [PubMed]
- Winkler, D.F.; Hilpert, K.; Brandt, O.; Hancock, R.E. Synthesis of peptide arrays using SPOT-technology and the CelluSpots-method. Methods Mol. Biol. 2009, 570, 157–174. [Google Scholar] [PubMed]
- Bock, I.; Dhayalan, A.; Kudithipudi, S.; Brandt, O.; Rathert, P.; Jeltsch, A. Detailed specificity analysis of antibodies binding to modified histone tails with peptide arrays. Epigenetics 2011, 6, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Chakravarty, S.; Ghersi, D.; Zeng, L.; Plotnikov, A.N.; Sanchez, R.; Zhou, M.M. Biochemical profiling of histone binding selectivity of the yeast bromodomain family. PLoS ONE 2010, 5, e8903. [Google Scholar] [CrossRef] [PubMed]
- Charlop-Powers, Z.; Zeng, L.; Zhang, Q.; Zhou, M.M. Structural insights into selective histone H3 recognition by the human Polybromo bromodomain 2. Cell Res. 2010, 20, 529–538. [Google Scholar] [CrossRef] [PubMed]
- Ruthenburg, A.J.; Li, H.; Milne, T.A.; Dewell, S.; McGinty, R.K.; Yuen, M.; Ueberheide, B.; Dou, Y.; Muir, T.W.; Patel, D.J.; et al. Recognition of a mononucleosomal histone modification pattern by BPTF via multivalent interactions. Cell 2011, 145, 692–706. [Google Scholar] [CrossRef] [PubMed]
- Delvecchio, M.; Gaucher, J.; Aguilar-Gurrieri, C.; Ortega, E.; Panne, D. Structure of the p300 catalytic core and implications for chromatin targeting and HAT regulation. Nat. Struct. Mol. Biol. 2013, 20, 1040–1046. [Google Scholar] [CrossRef] [PubMed]
- Tsai, W.W.; Wang, Z.; Yiu, T.T.; Akdemir, K.C.; Xia, W.; Winter, S.; Tsai, C.Y.; Shi, X.; Schwarzer, D.; Plunkett, W.; et al. TRIM24 links a non-canonical histone signature to breast cancer. Nature 2010, 468, 927–932. [Google Scholar] [CrossRef] [PubMed]
- Wen, H.; Li, Y.; Xi, Y.; Jiang, S.; Stratton, S.; Peng, D.; Tanaka, K.; Ren, Y.; Xia, Z.; Wu, J.; et al. ZMYND11 links histone H3.3K36me3 to transcription elongation and tumour suppression. Nature 2014, 508, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Qin, S.; Li, F.; Li, S.; Zhang, W.; Peng, J.; Zhang, Z.; Gong, Q.; Wu, J.; Shi, Y. Crystal structure of human BS69 Bromo-ZnF-PWWP reveals its role in H3K36me3 nucleosome binding. Cell Res. 2014, 24, 890–893. [Google Scholar] [CrossRef] [PubMed]
- Gong, F.; Chiu, L.Y.; Cox, B.; Aymard, F.; Clouaire, T.; Leung, J.W.; Cammarata, M.; Perez, M.; Agarwal, P.; Brodbelt, J.S.; et al. Screen identifies bromodomain protein ZMYND8 in chromatin recognition of transcription-associated DNA damage that promotes homologous recombination. Genes Dev. 2015, 29, 197–211. [Google Scholar] [CrossRef] [PubMed]
- Bryson, B.D.; del Rosario, A.M.; Gootenberg, J.S.; Yaffe, M.B.; White, F.M. Engineered bromodomains to explore the acetylproteome. Proteomics 2015, 15, 1470–1475. [Google Scholar] [CrossRef] [PubMed]
- Kramer, A.; Reineke, U.; Dong, L.; Hoffmann, B.; Hoffmüller, U.; Winkler, D.; Volkmer-engert, R.; Schneider-mergener, J. Spot synthesis: Observations and optimizations. J. Pept. Res. 1999, 54, 319–326. [Google Scholar] [CrossRef] [PubMed]
- Otvos, L., Jr.; Pease, A.M.; Bokonyi, K.; Giles-Davis, W.; Rogers, M.E.; Hintz, P.A.; Hoffmann, R.; Ertl, H.C.J. In situ stimulation of a T helper cell hybridoma with a cellulose-bound peptide antigen. J. Immunol. Methods 2000, 233, 95–105. [Google Scholar] [CrossRef]
- Hilpert, K.; Winkler, D.F.; Hancock, R.E. Peptide arrays on cellulose support: SPOT synthesis, a time and cost efficient method for synthesis of large numbers of peptides in a parallel and addressable fashion. Nat. Protoc. 2007, 2, 1333–1349. [Google Scholar] [CrossRef] [PubMed]
- Martens, W.; Greiser-Wilke, I.; Harder, T.C.; Dittmar, K.; Frank, R.; Orvell, C.; Moennig, V.; Liess, B. Spot synthesis of overlapping peptides on paper membrane supports enables the identification of linear monoclonal antibody binding determinants on morbillivirus phosphoproteins. Vet. Microbiol. 1995, 44, 289–298. [Google Scholar] [CrossRef]
- Briant, D.J.; Murphy, J.M.; Leung, G.C.; Sicheri, F. Rapid identification of linear protein domain binding motifs using peptide SPOT arrays. Methods Mol. Biol. 2009, 570, 175–185. [Google Scholar] [PubMed]
- Filippakopoulos, P.; Knapp, S. The bromodomain interaction module. FEBS Lett. 2012, 586, 2692–2704. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Picaud, S.; Filippakopoulos, P. SPOTing Acetyl-Lysine Dependent Interactions. Microarrays 2015, 4, 370-388. https://doi.org/10.3390/microarrays4030370
Picaud S, Filippakopoulos P. SPOTing Acetyl-Lysine Dependent Interactions. Microarrays. 2015; 4(3):370-388. https://doi.org/10.3390/microarrays4030370
Chicago/Turabian StylePicaud, Sarah, and Panagis Filippakopoulos. 2015. "SPOTing Acetyl-Lysine Dependent Interactions" Microarrays 4, no. 3: 370-388. https://doi.org/10.3390/microarrays4030370
APA StylePicaud, S., & Filippakopoulos, P. (2015). SPOTing Acetyl-Lysine Dependent Interactions. Microarrays, 4(3), 370-388. https://doi.org/10.3390/microarrays4030370




