Neurophysiological Characterization of Thalamic Nuclei in Epileptic Anaesthetized Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Surgery
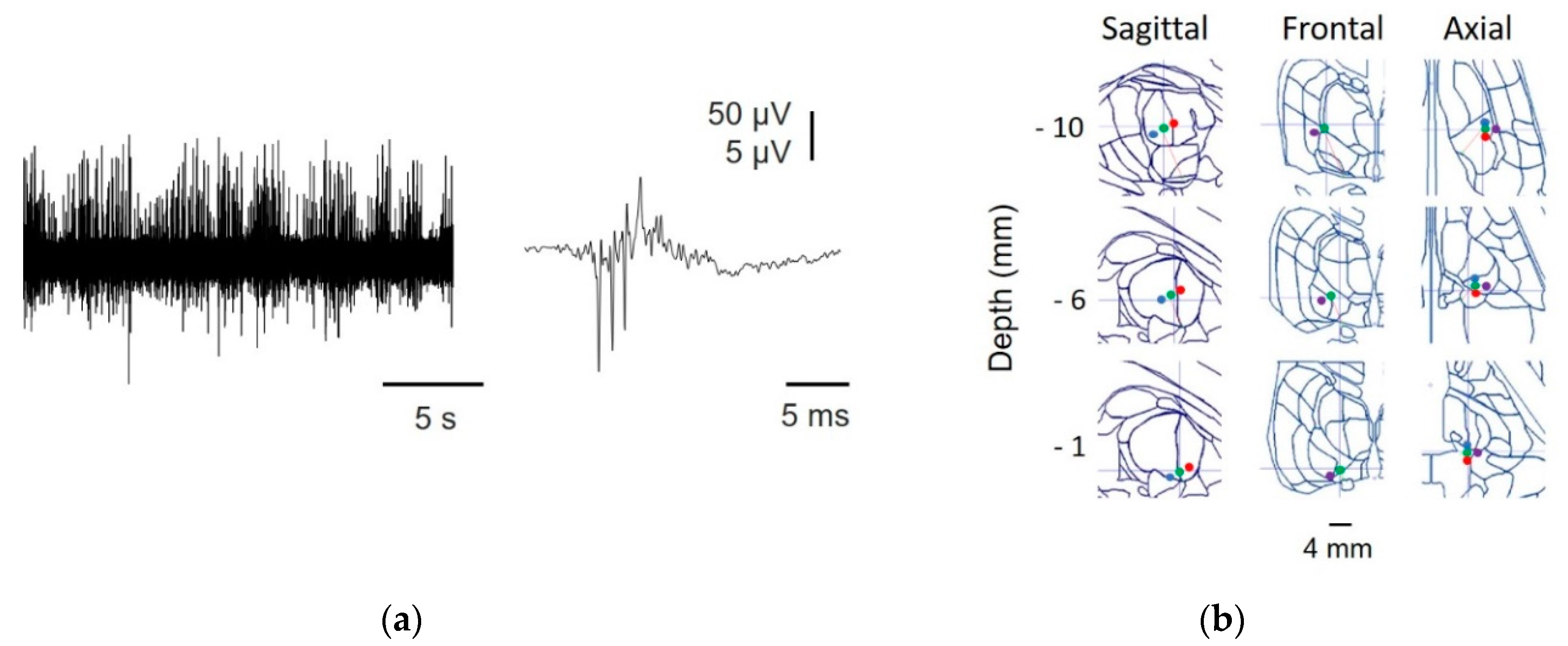
2.2. Somatosensory Evoked Potentials
2.3. Reconstruction of the Trajectory
2.4. Analysis of Discharge Properties
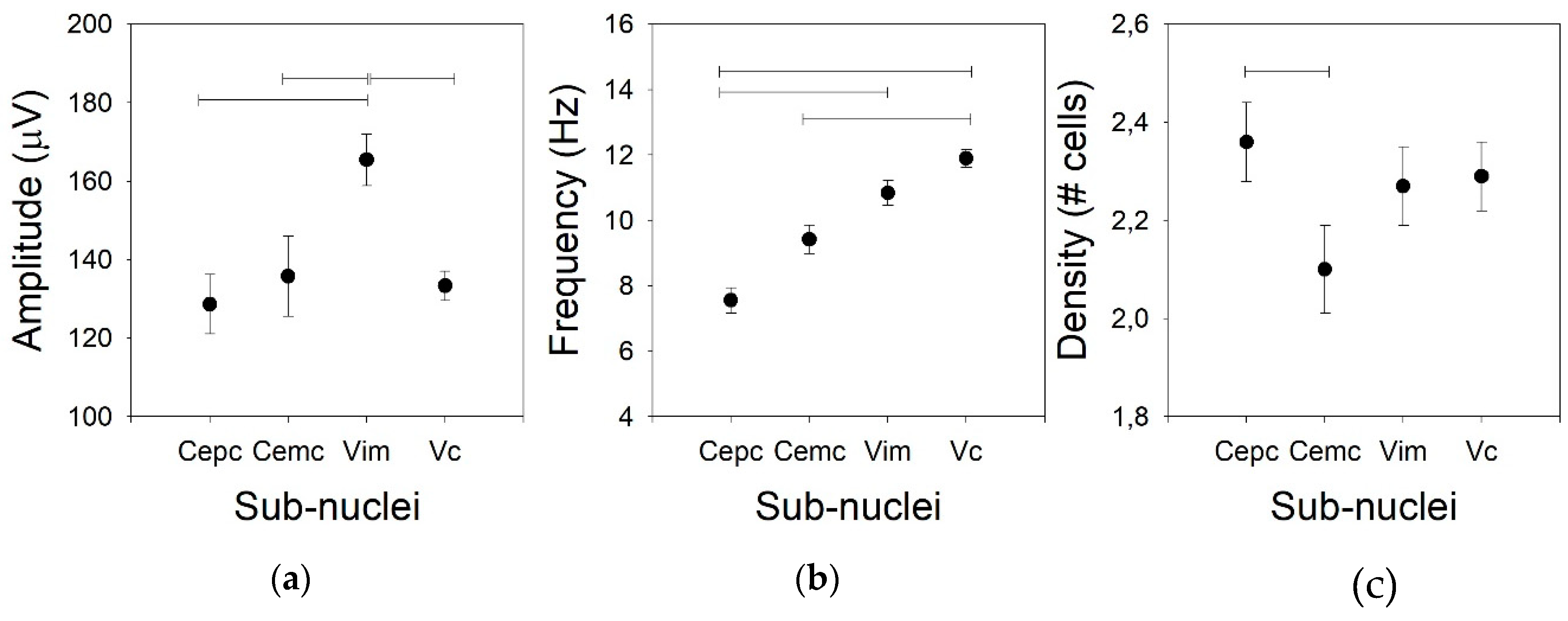
- Amplitude of the AP, measured from peak to peak (in µV). This property is not a tonic property, but is commonly used in clinical practice; therefore, we included it in this group.
- Mean frequency and standard deviation of the raw trace and for every neuron. Both values were obtained from the inverse of the instant frequency.
- Density, defined as the number of cells recorded by every electrode at one position. AP sorting was performed by clustering by using the Mahalanobis distances (see below) of several properties of the AP (e.g., amplitude and duration of positive and negative phases and maximum and minimum value of the first derivative). The maximum number of cells allowed to be identified by this mean was chosen as 5.
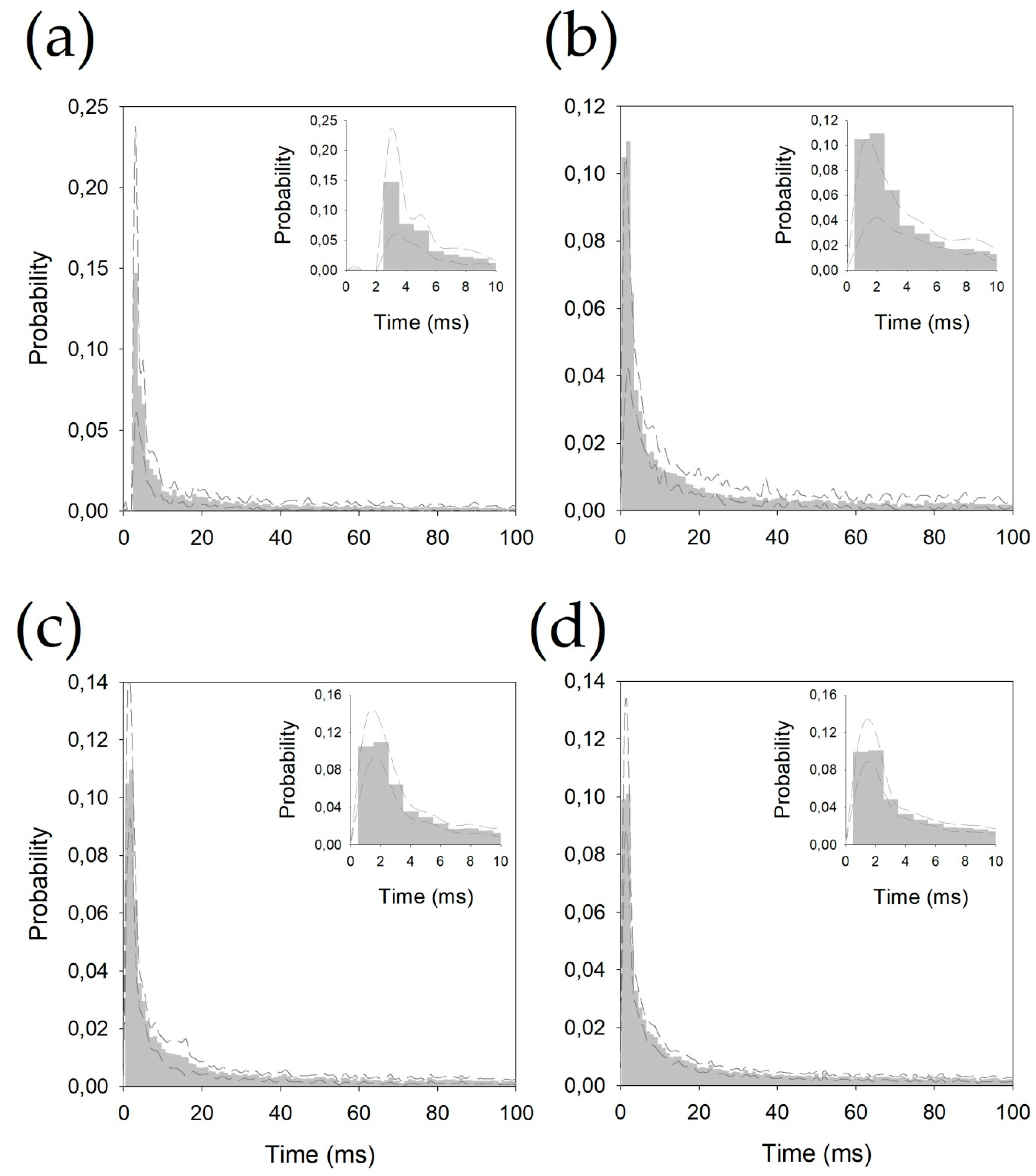
- Probability density functions (pdf) of the inter-spike interval (ISI) for every neuron. Relative frequency was computed for 1 ms bins, and the probability/bin (pi) was calculated with the following expression:where fi is the frequency for the i-bin and N is the total number of bins.
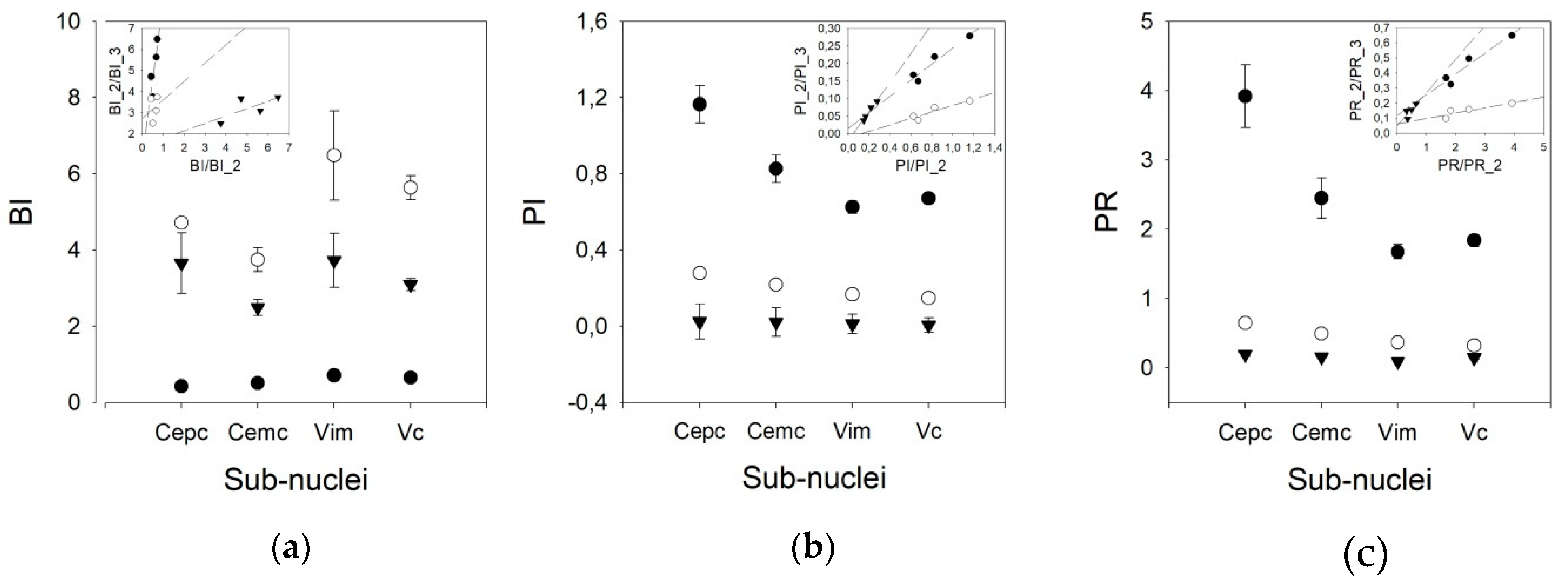
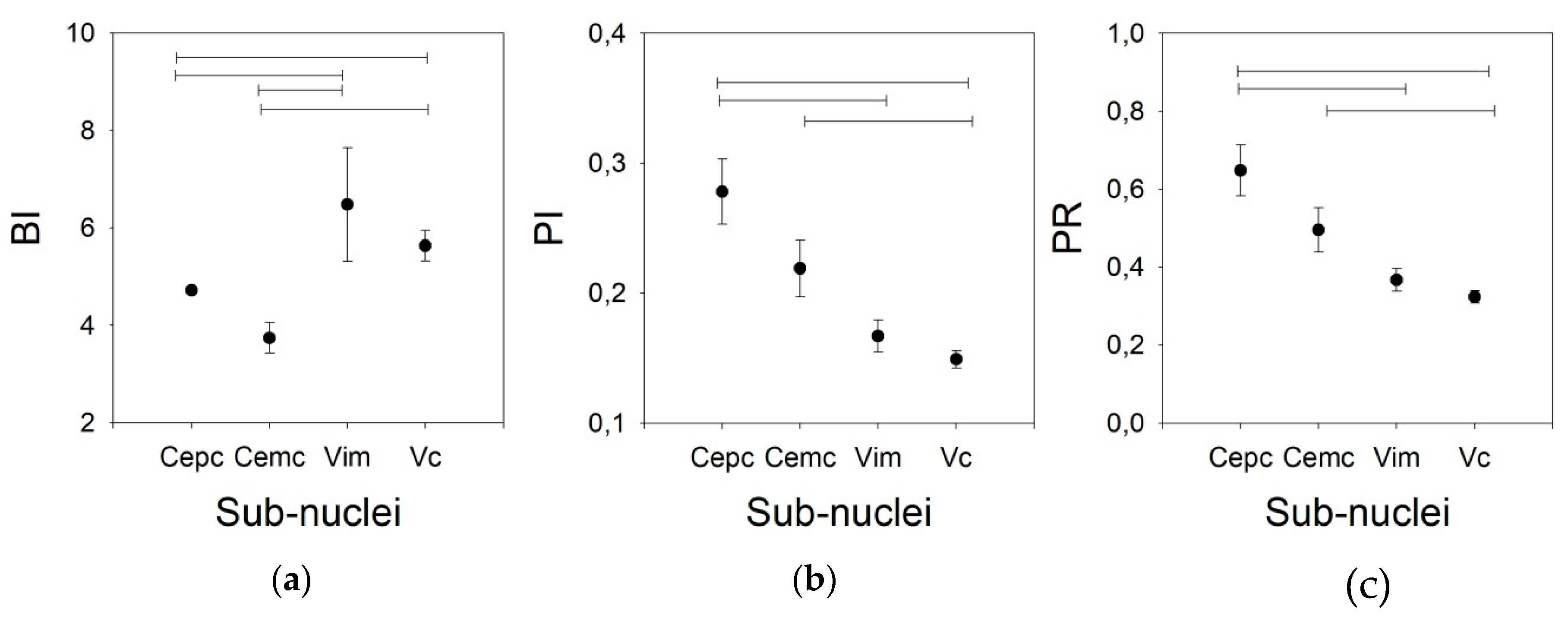
- Burst index (BI) is defined as the ratio between the number of ISI < 10 ms and the number of ISIs > 10 ms. It represents the number of bursts of discharges with respect to individual discharges, and is calculated as follows:
- Pause index (PI), defined as the ratio between the number of ISIs > 50 ms and the number of ISIs < 50 ms, is calculated as follows:
- Pause ratio (PR), defined as the total duration of pauses (ISI > 50 ms) divided by the total duration of no-pauses (ISI < 50 ms). Although similar in name, the information obtained is different from that of the PI, and the PR is calculated as follows:
2.5. Evaluation of Global Similarity
2.6. Statistics
3. Results
Tonic Properties
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A

Appendix B
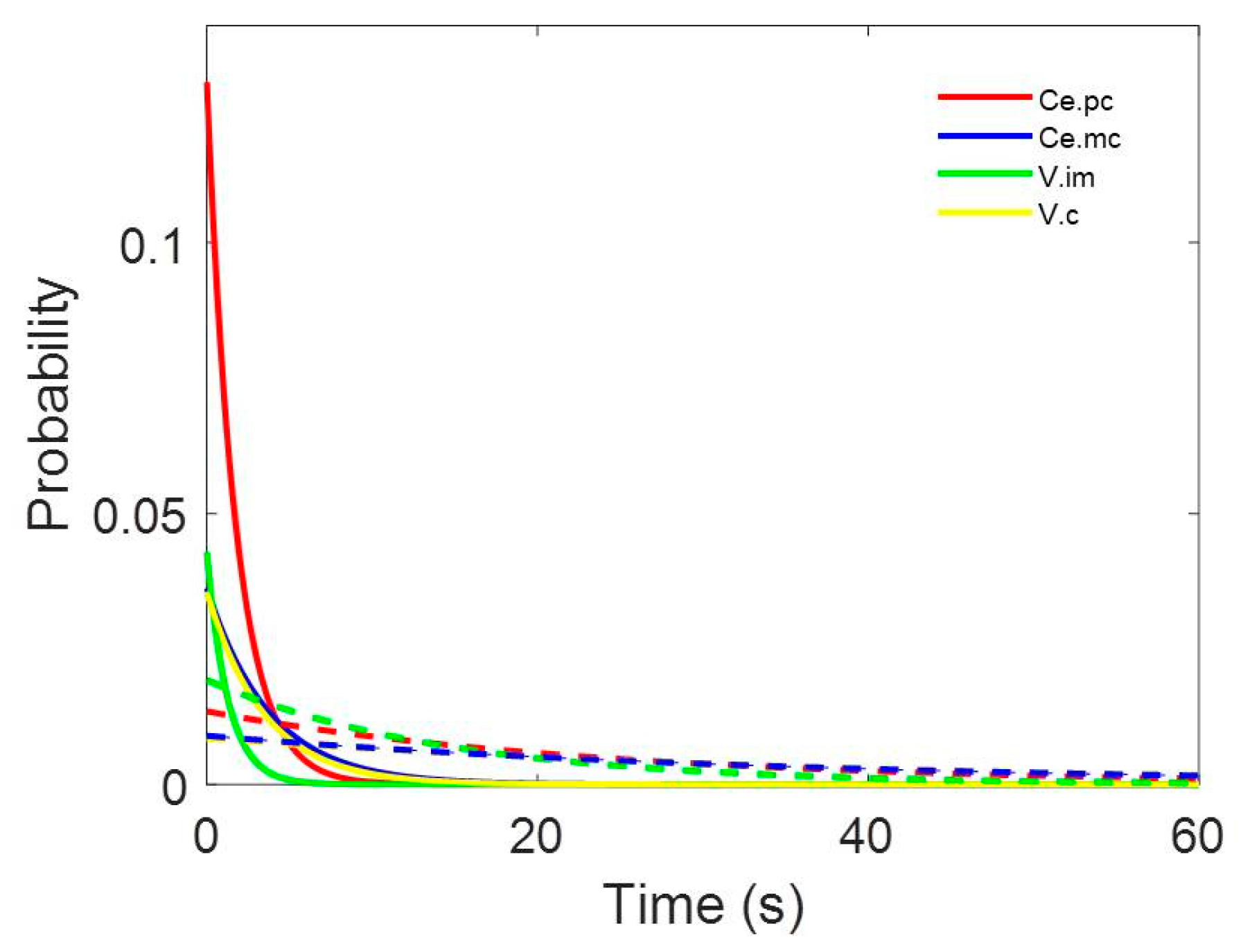
| Nucleus | r | α | β | γ | δ | ε |
|---|---|---|---|---|---|---|
| Ce.pc | 0.9939 | 0.001 | 0.1296 | 0.5683 | 0.0135 | 0.0421 |
| Ce.mc | 0.9904 | 0.001 | 0.0363 | 0.2660 | 0.0090 | 0.0285 |
| V.im | 0.9929 | 0.0013 | 0.0429 | 0.7841 | 0.0193 | 0.0692 |
| V.c | 0.9925 | 0.001 | 0.0356 | 0.2899 | 0.0085 | 0.0262 |
Appendix C
| Index | Pair of Thresholds (ms) | Linear Function [y(x)] | r2 | t | p |
|---|---|---|---|---|---|
| 10/50 | 0.675 | 2.038 | n.s | ||
| BI | 20/100 | 0.040 | 0.289 | n.s | |
| 50/200 | 0.513 | 1.451 | n.s | ||
| 10/50 | 0.932 | 5.215 | <0.01 | ||
| PI | 20/100 | 0.849 | 3.347 | <0.05 | |
| 50/200 | 0.980 | 9.900 | <0.001 | ||
| 10/50 | 0.933 | 5.261 | <0.01 | ||
| PR | 20/100 | 0.757 | 2.496 | n.s | |
| 50/200 | 0.602 | 1.739 | n.s |
References
- Nieuwenhuys, R.; Voogd, J.; van Huijzen, C. (Eds.) Diencephalo: Dorsal Thalamus. In The Human Central Nervous System; Springer: Berlin/Heidelberg, Germany, 2008; pp. 253–279. [Google Scholar]
- Velasco, A.L.; Velasco, F.; Jiménez, F.; Velasco, M.; Castro, G.; Carrillo-Ruiz, J.D.; Fanghänel, G.; Boleaga, B. Neuromodulation of the centromedian thalamic nuclei in the treatment of generalized seizures and the improvement of the quality of life in patients with Lennox-Gastaut syndrome. Epilepsia 2006, 47, 1203–1212. [Google Scholar] [CrossRef]
- Velasco, F.; Velasco, A.L.; Velasco, M.; Jiménez, F.; Carrillo-Ruiz, J.D.; Castro, G. Deep brain stimulation for treatment of the epilepsies: The centromedian thalamic target. Acta Neurochir. Suppl. 2007, 97, 337–342. [Google Scholar]
- Valentín, A.; García-Navarrete, E.; Chelvarajah, R.; Torres, C.; Navas, M.; Vico, L.; Torres, N.; Pastor, J.; Selway, R.; Sola, R.G.; et al. Deep brain stimulation of the centromedian thalamic nucleus for the treatment of generalised and frontal epilepsies. Epilepsia 2013, 54, 1823–1833. [Google Scholar] [CrossRef]
- Torres, C.; Pastor, J.; Navarrete, E.G.; Sola, R.G. Estimulación cerebral profunda talámica para la epilepsia resistente. Rev. Neurol. 2011, 53, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Lim, S.C.; Kim, J.; Son, B.C.; Lee, K.J.; Shon, Y.M. Long-term follow-up of anterior thalamic deep brain stimulation in epilepsy: A 11-year, single center experience. Seizure 2017, 52, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Toyota, E.; Anderson, M.; Abel, T.J.; Donner, E.; Kalia, S.K.; Drake, J.; Rutka, J.T.; Ibrahim, G.M. A systematic review of deep brain stimulation for the treatment of drug-resistant epilepsy in childhood. J. Neurosurg. Pediatr. 2018, 23, 274–284. [Google Scholar] [CrossRef] [PubMed]
- Herrman, H.; Egge, A.; Konglund, A.E.; Ramm-Pettersen, J.; Dietrichs, E.; Taubøll, E. Anterior thalamic deep brain stimulation in refractory epilepsy: A randomized, double-blinded study. Acta Neurol. Scand. 2019, 139, 294–304. [Google Scholar] [CrossRef] [PubMed]
- Koeppen, J.A.; Nahravani, F.; Kramer, M.; Voges, B.; House, P.M.; Gulberti, A.; Moll, C.K.E.; Westphal, M.; Hamel, W. Electrical Stimulation of the Anterior Thalamus for Epilepsy: Clinical Outcome and Analysis of Efficient Target. Neuromodulation 2019, 22, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Obwegeser, A.A.; Uitti, R.J.; Turk, M.F.; Strongosky, A.J.; Wharen, R.E. Thalamic stimulation for the treatment of midline tremors in essential tremor patients. Neurology 2000, 54, 2342–2344. [Google Scholar] [CrossRef]
- Wu, D.; Wang, S.; Stein, J.F.; Aziz, T.Z.; Green, A.L. Reciprocal interactions between the human thalamus and periaqueductal gray may be important for pain perception. Exp. Brain Res. 2014, 232, 527–534. [Google Scholar] [CrossRef]
- Pastor, J.; Vega-Zelaya, L. A new potential specifically marks the sensory thalamus in anaesthetized patients. Clin. Neurophysiol. 2019, 130, 1926–1936. [Google Scholar] [CrossRef] [PubMed]
- Vega-Zelaya, L.; Sola, R.G.; Pulido, P.; Pastor, J. Do we need to awake patients up during cortical surgery? J. Cancer Res. Updates 2018, 7, 84–96. [Google Scholar] [CrossRef]
- Koester, J.; Siegelbaum, S.A. Membrane Potential. In Principles of Neural Science; Kandel, E.R., Schwartz, J.H., Jessell, T.M., Eds.; Elsevier: New York, NY, USA, 2000; pp. 125–139. [Google Scholar]
- Gold, C.; Henze, D.A.; Koch, C.; Buzsáki, G. On the origin of the extracellular action potential waveform: A modeling study. J. Neurophysiol. 2006, 95, 3113–3128. [Google Scholar] [CrossRef] [PubMed]
- Sloan, T.B. Anesthesia and motor evoked potential monitoring. In Neurophysiology in Neurosurgery; Deletis, V., Shils, J.H., Eds.; Academic Press: Amsterdam, The Netherlands, 2002; pp. 451–474. [Google Scholar]
- Voss, L.J.; Sleigh, J.W.; Barnard, J.P.; Kirsch, H.E. The howling cortex: Seizures and general anesthetic drugs. Anesth. Analg. 2008, 107, 1689–1703. [Google Scholar] [CrossRef]
- Hutchison, W.D. Electrophysiological findings in GPe and GPi. In Microelectrode Recording in Movement Disorder Surgery; Zvi, I., Kim, J.B., Eds.; Thieme: New York, NY, USA, 2004. [Google Scholar]
- Vega-Zelaya, L.; Torres, C.; Sola, R.G.; Pastor, J. Characterization of thalamic nuclei and somatosensory evoked potentials in anesthetized humans. Clin. Neurophysiol. 2016, 127, 203–205. [Google Scholar] [CrossRef]
- Hassler, R. Anatomy of the thalamus. In Introduction to Stereotaxis with an Atlas of the Human Brain; Schaltenbrand, G., Bailey, P., Eds.; Thieme: Stuttgart, Germany, 1959; pp. 230–290. [Google Scholar]
- Peppe, A.; Gasbarra, A.; Stefani, A.; Chiavalon, C.; Pierantozzi, M.; Fermi, E.; Stanzione, P.; Caltagirone, C.; Mazzone, P. Deep brain stimulation of CM/PF of thalamus could be the new elective target for tremor in advanced Parkinson’s Disease? Parkinsonism Relat. Disord. 2008, 14, 501–504. [Google Scholar] [CrossRef] [PubMed]
- Semenova, U.; Raeva, S.; Sedov, A. Participation of the thalamic CM-Pf complex in movement performance in patients with dystonia. Mov. Disord. 2016, 31, 1398–1404. [Google Scholar] [CrossRef] [PubMed]
- Owen, S.L.; Green, A.L.; Stein, J.F.; Aziz, T.Z. Deep brain stimulation for the alleviation of post-stroke neuropathic pain. Pain 2006, 120, 202–206. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, J.P.; Nizard, J.; Keravel, Y.; Lefaucheur, J.P. Invasive brain stimulation for the treatment of neuropathic pain. Nat. Rev. Neurol. 2011, 7, 699–709. [Google Scholar] [CrossRef] [PubMed]
- Pastor, J.; Hernando-Requejo, V.; Domínguez-Gadea, L.; de Llano, I.; Meilán-Paz, M.L.; Martínez-Chacón, J.L.; Sola, R.G. Impact of experience on improving the surgical outcome in temporal lobe epilepsy. Rev. Neurol. 2005, 41, 709–716. [Google Scholar] [PubMed]
- Sola, R.G.; Hernando-Requejo, V.; Pastor, J.; García-Navarrete, E.; DeFelipe, J.; Alijarde, M.T.; Sánchez, A.; Domínguez-Gadea, L.; Martín-Plasencia, P.; Maestú, F.; et al. Pharmacoresistant temporal-lobe epilepsy. Exploration with foramen ovale electrodes and surgical outcomes. Rev. Neurol. 2005, 41, 4–16. [Google Scholar] [PubMed]
- Javre, J.; Baumann, T. Signal processing and pattern recognition. In Microelectrode Recording in Movement Disorder Surgery; Zvi, I., Kim, J.B., Eds.; Thieme: New York, NY, USA, 2004. [Google Scholar]
- Favre, J.; Taha, J.M.; Baumann, T.; Burchiel, K.J. Computer analysis of the tonic, phasic, and kinesthetic activity of pallidal discharges in Parkinson patients. Surg. Neurol. 1999, 51, 665–672. [Google Scholar] [CrossRef]
- Mahalanobis, P.C. On the generalized distance in statistics. Proc. Natl. Inst. Sci. India 1936, 2, 49–55. [Google Scholar]
- Peña, D.; Prieto, F.J. The kurtosis coefficient and the linear discriminant function. Stat. Probab. Lett. 2000, 49, 257–261. [Google Scholar] [CrossRef]
- Marceglia, S.; Servello, D.; Foffani, G.; Porta, M.; Sassi, M.; Mrakic-Sposta, S.; Rosa, M.; Barbieri, S.; Priori, A. Thalamic single-unit and local field potential activity in Tourette syndrome. Mov. Disord. 2010, 25, 300–308. [Google Scholar] [CrossRef]
- Zhuang, P.; Li, Y.; Hallett, M. Neuronal activity in the basal ganglia and thalamus in patients with dystonia. Clin. Neurophysiol. 2004, 115, 2542–2557. [Google Scholar] [CrossRef]
- Alam, M.; Schwabe, K.; Lütjens, G.; Capelle, H.H.; Manu, M.; von Wrangel, C.; Müller-Vahl, K.; Schrader, C.; Scheinichen, D.; Blahak, C.; et al. Comparative characterization of single cell activity in the globus pallidus internus of patients with dystonia or Tourette syndrome. J. Neural Transm. (Vienna) 2015, 122, 687–699. [Google Scholar] [CrossRef] [PubMed]
- Malekmohammadi, M.; Sparks, H.; AuYong, N.; Hudson, A.; Pouratian, N. Propofol Anesthesia Precludes LFP-Based Functional Mapping of Pallidum during DBS Implantation. Stereotact. Funct. Neurosurg. 2018, 96, 249–258. [Google Scholar] [CrossRef]
- Tsai, S.T.; Chen, T.Y.; Lin, S.H.; Chen, S.Y. Five-Year Clinical Outcomes of Local versus General Anesthesia Deep Brain Stimulation for Parkinson’s Disease. Parkinsons Dis. 2019, 17. [Google Scholar] [CrossRef]
- Harris, K.D.; Henze, D.A.; Csicsvari, J.; Hirase, H.; Buzsáki, G. Accuracy of tetrode spike separation as determined by simultaneous intracellular and extracellular measurements. J. Neurophysiol. 2000, 84, 401–414. [Google Scholar] [CrossRef]
- Henze, D.A.; Borhegyi, Z.; Csicsvari, J.; Mamiya, A.; Harris, K.D.; Buzsáki, G. Intracellular features predicted by extracellular recordings in the hippocampus in vivo. J. Neurophysiol. 2000, 84, 390–400. [Google Scholar] [CrossRef]
- Marsden, J.E.; Tromba, A.J. Vector Calculus, 5th ed.; Freeman: New York, NY, USA, 2003. [Google Scholar]
- Peña, D. Análisis de Datos Multivariantes; McGraw-Hill/Interamericana de España: Madrid, Spain, 2002. [Google Scholar]
- McLachlan, G. Discriminant Analysis and Statistical Pattern Recognition; Wiley: Hoboken, NJ, USA, 2004. [Google Scholar]
- Seifried, C.; Weise, L.; Hartmann, R.; Gasser, T.; Baudrexel, S.; Szelényi, A.; van de Loo, S.; Steinmetz, H.; Seifert, V.; Roeper, J.; et al. Intraoperative microelectrode recording for the delineation of subthalamic nucleus topography in Parkinson’s disease. Brain Stimul. 2012, 5, 378–387. [Google Scholar] [CrossRef]
- Li, X.; Zhuang, P.; Hallett, M.; Zhang, Y.; Li, J.; Li, Y. Subthalamic oscillatory activity in parkinsonian patients with off-period dystonia. Acta Neurol. Scand. 2016, 134, 327–338. [Google Scholar] [CrossRef]
- Möttönen, T.; Katisko, J.; Haapasalo, J.; Tähtinen, T.; Saastamoinen, A.; Peltola, J.; Öhman, J.; Lehtimäki, K. The Correlation between Intraoperative Microelectrode Recording and 3-Tesla MRI in Patients Undergoing ANT-DBS for Refractory Epilepsy. Stereotact. Funct. Neurosurg. 2016, 94, 86–92. [Google Scholar] [CrossRef]
- Alam, M.; Sanghera, M.K.; Schwabe, K.; Lütjens, G.; Jin, X.; Song, J.; von Wrangel, C.; Stewart, R.M.; Jankovic, J.; Grossman, R.G.; et al. Globus pallidus internus neuronal activity: A comparative study of linear and non-linear features in patients with dystonia or Parkinson’s disease. J. Neural Transm. (Vienna) 2016, 123, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Mirzadeh, Z.; Chapple, K.M.; Lambert, M.; Evidente, V.G.H.; Moguel-Cobos, G.; Oravivattanakul, S.; Mahant, P.; Ponce, F.A. Intraoperative test stimulation versus stereotactic accuracy as a surgical end point: A comparison of essential tremor outcomes after ventral intermediate nucleus deep brain stimulation. J. Neurosurg. 2018, 129, 290–298. [Google Scholar] [CrossRef] [PubMed]
- Brahimaj, B.; Kochanski, R.B.; Sani, S. Microelectrode accuracy in deep brain stimulation surgery. J. Clin. Neurosci. 2018, 50, 58–61. [Google Scholar] [CrossRef] [PubMed]

| Pat. | Sex | Age (years) | History (years) | Etiology | v-EEG | MRI Result | VNS | AED |
|---|---|---|---|---|---|---|---|---|
| 1 | F | 37 | 31 | Genetic 1 | GE | Normal | Yes | PGB, CBZ, CNZ |
| 2 | F | 18 | 12 | LGS | GE | Dysplasia LF | No | RUF, VPT |
| 3 | M | 34 | 27 | Genetic 2 | EG/EE | Normal | Yes | VPT, PGB, LAC, ZNS |
| 4 | M | 27 | 27 | LGS | GE | Normal | No | LVZ, OXC, LAC, CZM |
| 5 | M | 30 | 23 | Structural | GE/EE | Dysplasia biFT | Yes | TPM, VPT |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vega-Zelaya, L.; Torres, C.V.; Navas, M.; Pastor, J. Neurophysiological Characterization of Thalamic Nuclei in Epileptic Anaesthetized Patients. Brain Sci. 2019, 9, 312. https://doi.org/10.3390/brainsci9110312
Vega-Zelaya L, Torres CV, Navas M, Pastor J. Neurophysiological Characterization of Thalamic Nuclei in Epileptic Anaesthetized Patients. Brain Sciences. 2019; 9(11):312. https://doi.org/10.3390/brainsci9110312
Chicago/Turabian StyleVega-Zelaya, Lorena, Cristina V. Torres, Marta Navas, and Jesús Pastor. 2019. "Neurophysiological Characterization of Thalamic Nuclei in Epileptic Anaesthetized Patients" Brain Sciences 9, no. 11: 312. https://doi.org/10.3390/brainsci9110312
APA StyleVega-Zelaya, L., Torres, C. V., Navas, M., & Pastor, J. (2019). Neurophysiological Characterization of Thalamic Nuclei in Epileptic Anaesthetized Patients. Brain Sciences, 9(11), 312. https://doi.org/10.3390/brainsci9110312






