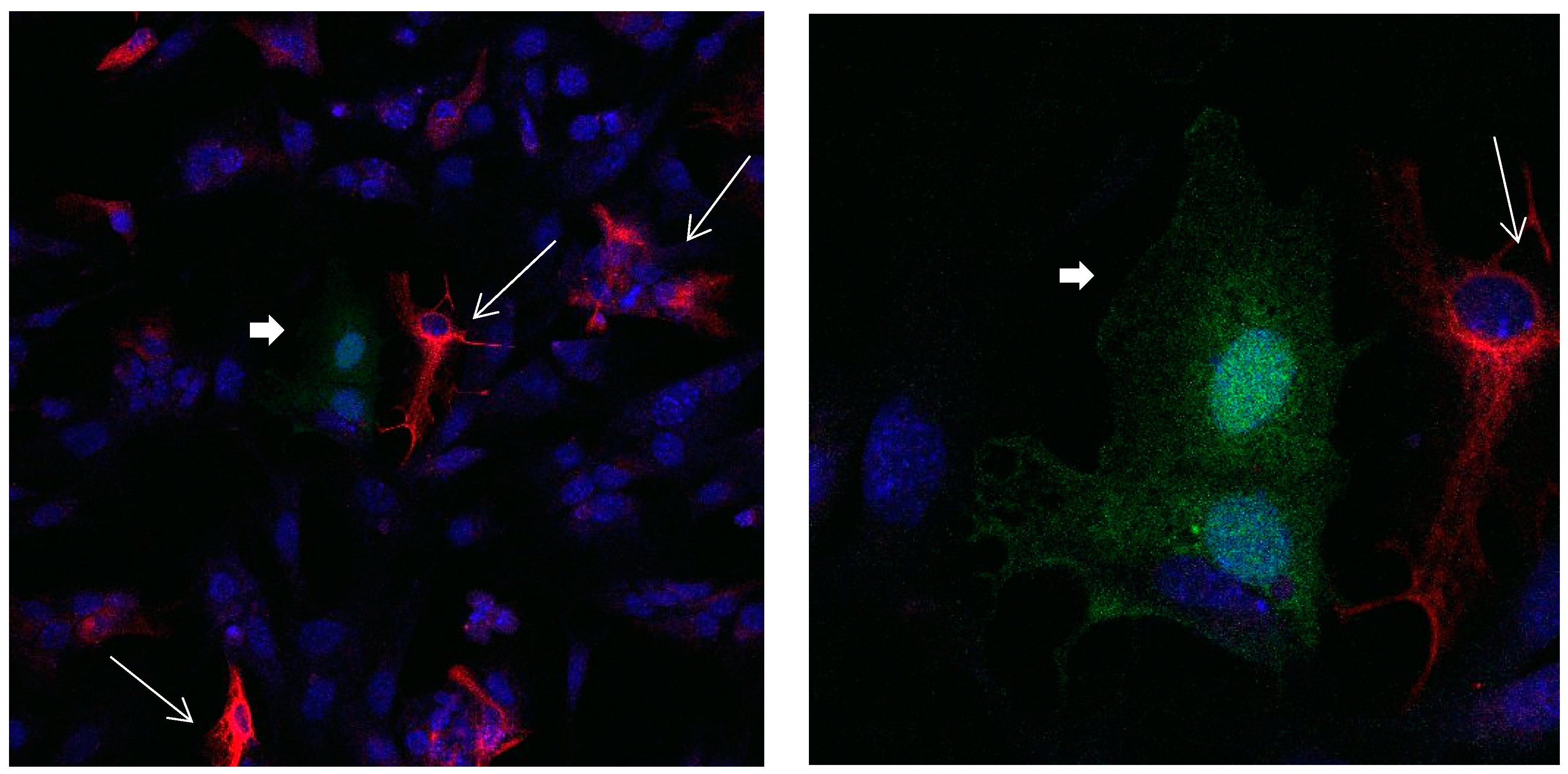
Figure 1.
Cultures in a serum-containing medium, 1 day after passage: Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488); Nuclear staining with DAPI (4′,6-diamidino-2-phenylindole dihydrochloride) (blue). Confocal microscopy. Left: zoom = 1, numerous GFAP-positive astrocytes (long arrows), in between 2 Calbindin D28k positive and GFAP-negative astrocytes (short arrows). Right: zoom = 4, higher magnification of the 2 Calbindin D28k positive astrocytes (short arrows).
Figure 1.
Cultures in a serum-containing medium, 1 day after passage: Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488); Nuclear staining with DAPI (4′,6-diamidino-2-phenylindole dihydrochloride) (blue). Confocal microscopy. Left: zoom = 1, numerous GFAP-positive astrocytes (long arrows), in between 2 Calbindin D28k positive and GFAP-negative astrocytes (short arrows). Right: zoom = 4, higher magnification of the 2 Calbindin D28k positive astrocytes (short arrows).
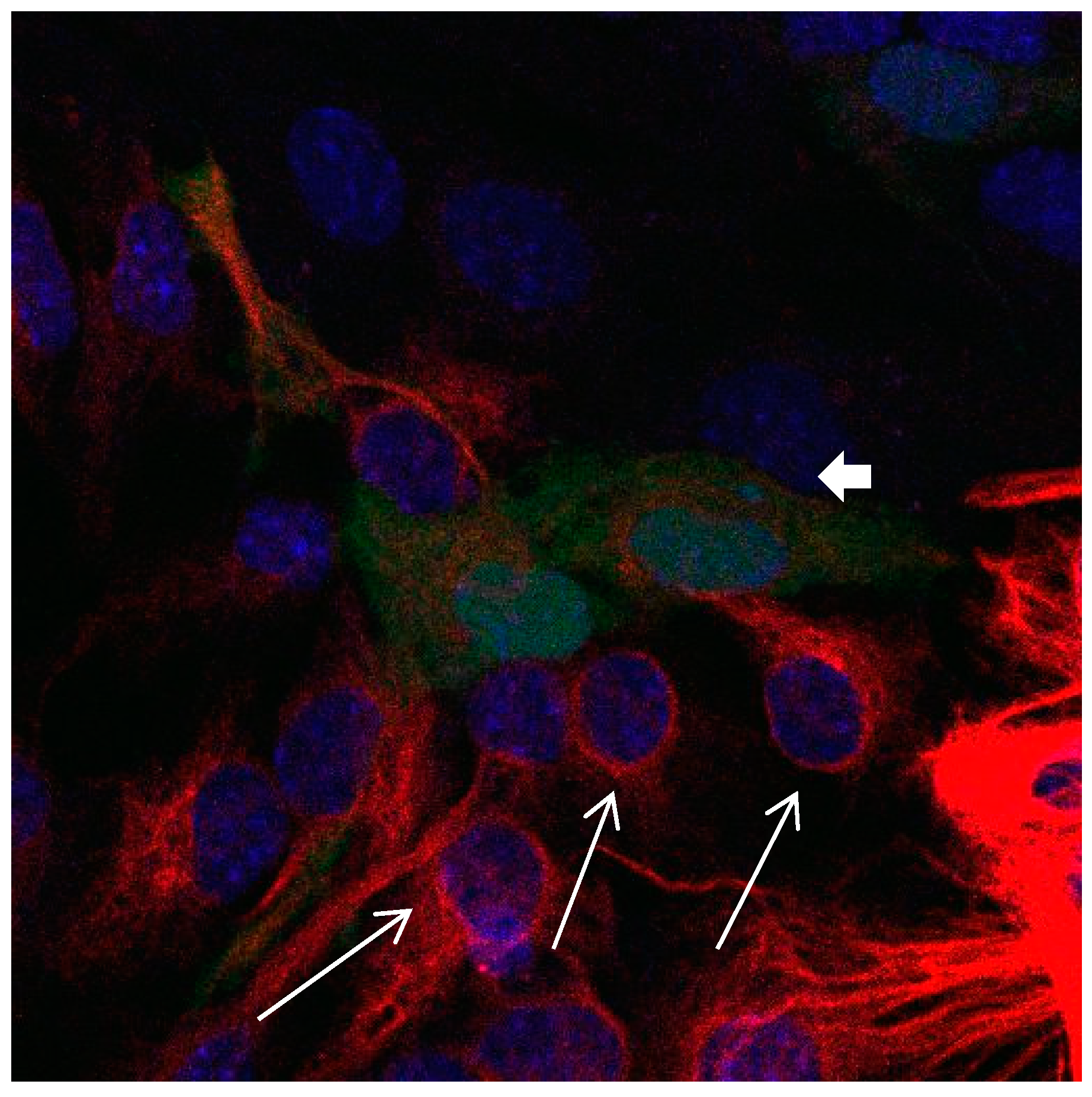
Figure 2.
Cultures in a serum-containing medium, 5 days after passage: Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488); Nuclear staining with DAPI (4′,6-diamidino-2-phenylindole dihydrochloride) (blue). Confocal microscopy. Zoom = 4, numerous GFAP-positive and Calbindin D28k negative astrocytes (long arrows), in between 2 Calbindin D28k positive and GFAP-positive astrocytes (short arrows).
Figure 2.
Cultures in a serum-containing medium, 5 days after passage: Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488); Nuclear staining with DAPI (4′,6-diamidino-2-phenylindole dihydrochloride) (blue). Confocal microscopy. Zoom = 4, numerous GFAP-positive and Calbindin D28k negative astrocytes (long arrows), in between 2 Calbindin D28k positive and GFAP-positive astrocytes (short arrows).
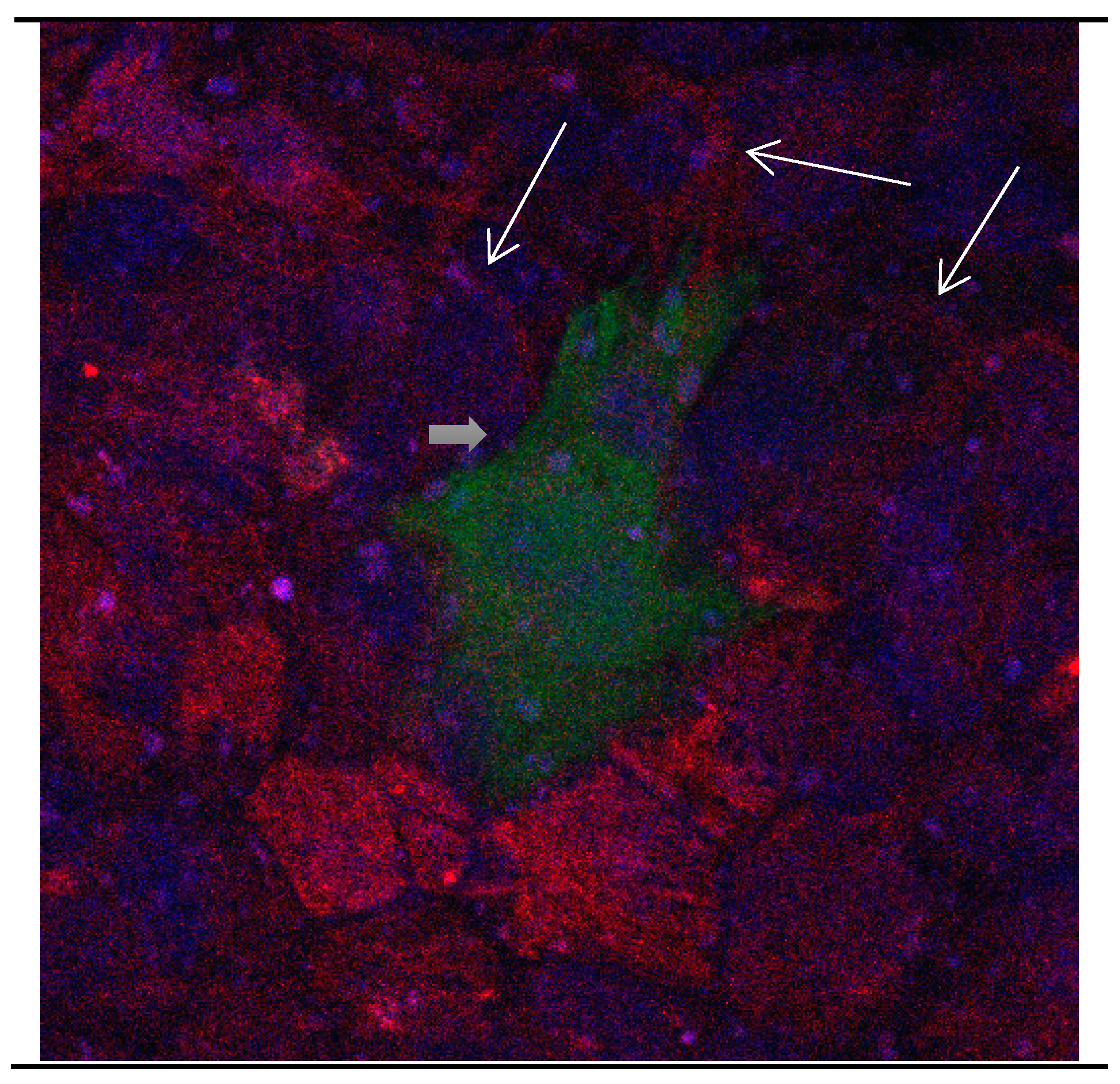
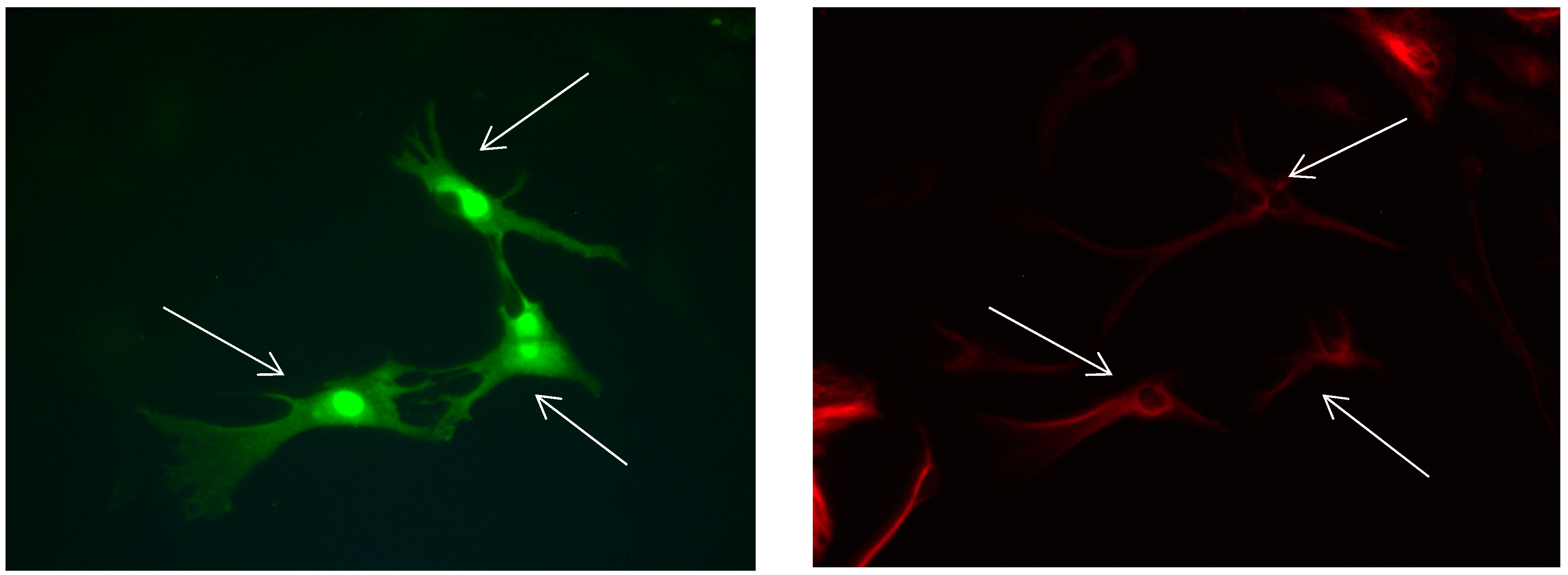
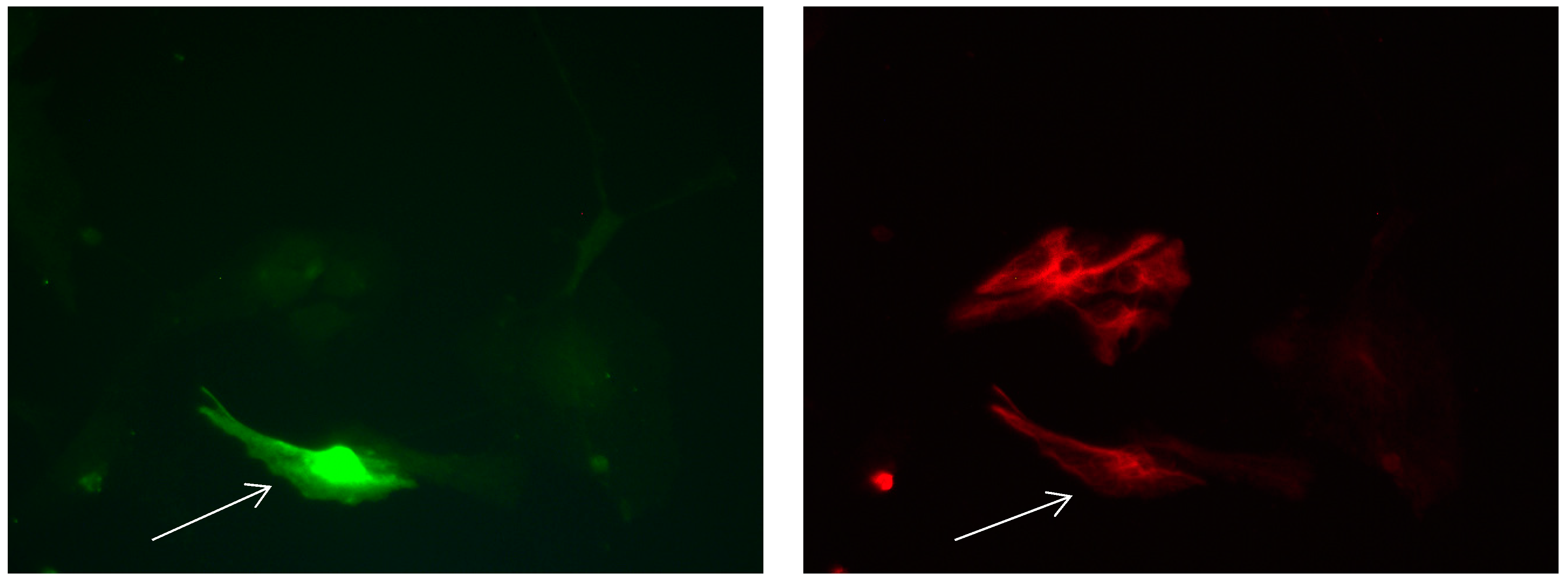
Figure 3.
Cultures in a serum-containing medium, 9 days after passage: Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488); Nuclear staining with DAPI (4′,6-diamidino-2-phenylindole dihydrochloride) (blue). Confocal microscopy. Zoom = 4, numerous GFAP-positive and Calbindin D28k negative astrocytes (long arrows), in between 1 Calbindin D28k positive and GFAP-negative astrocyte (short arrows).
Figure 3.
Cultures in a serum-containing medium, 9 days after passage: Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488); Nuclear staining with DAPI (4′,6-diamidino-2-phenylindole dihydrochloride) (blue). Confocal microscopy. Zoom = 4, numerous GFAP-positive and Calbindin D28k negative astrocytes (long arrows), in between 1 Calbindin D28k positive and GFAP-negative astrocyte (short arrows).
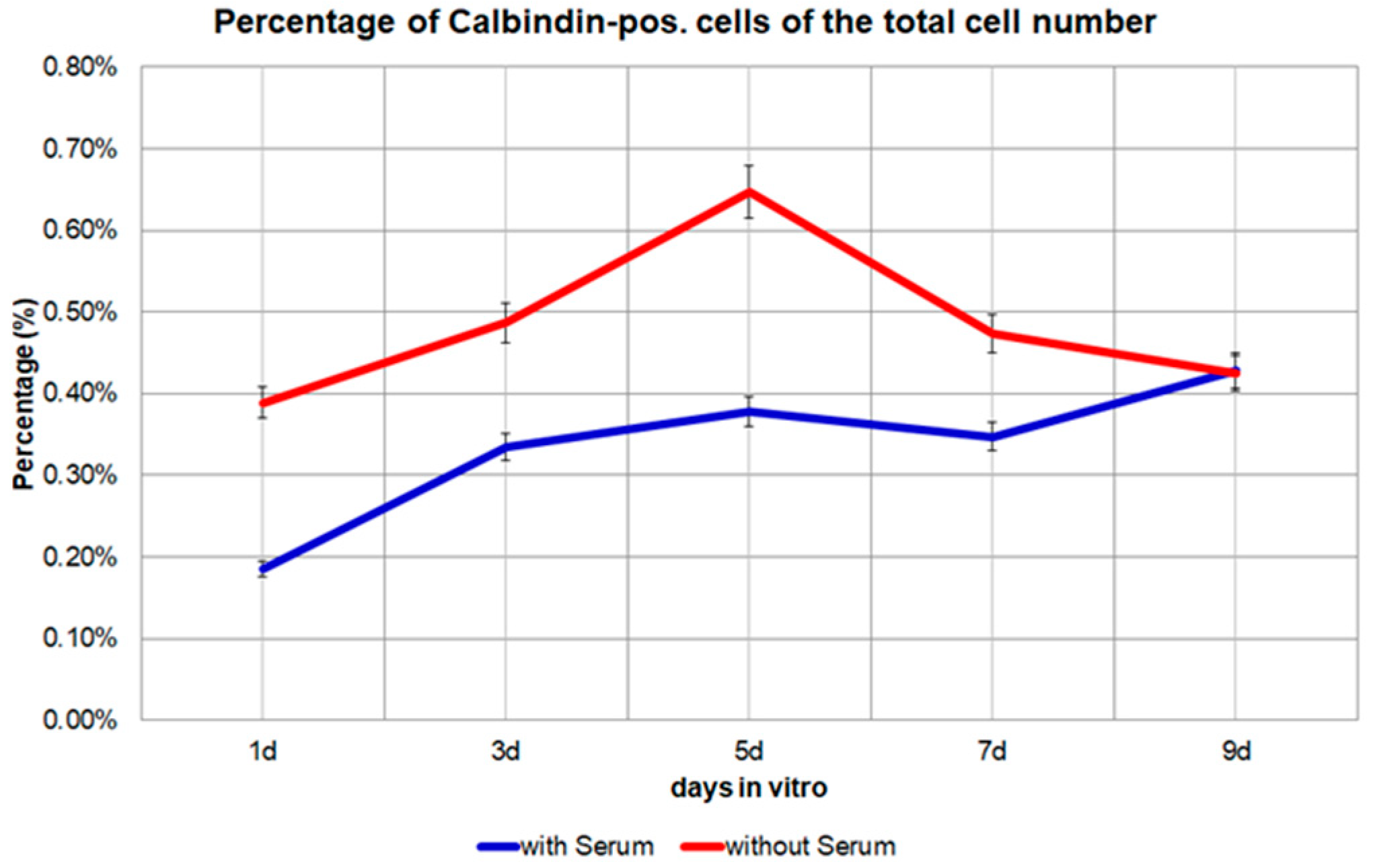
Figure 4.
The percentage of Calbindin D28k positive cells of the total cell number from day 1 to day 9 in the medium with 10% FBS or in the medium without FBS. p = 0.0187, significant.
Figure 4.
The percentage of Calbindin D28k positive cells of the total cell number from day 1 to day 9 in the medium with 10% FBS or in the medium without FBS. p = 0.0187, significant.
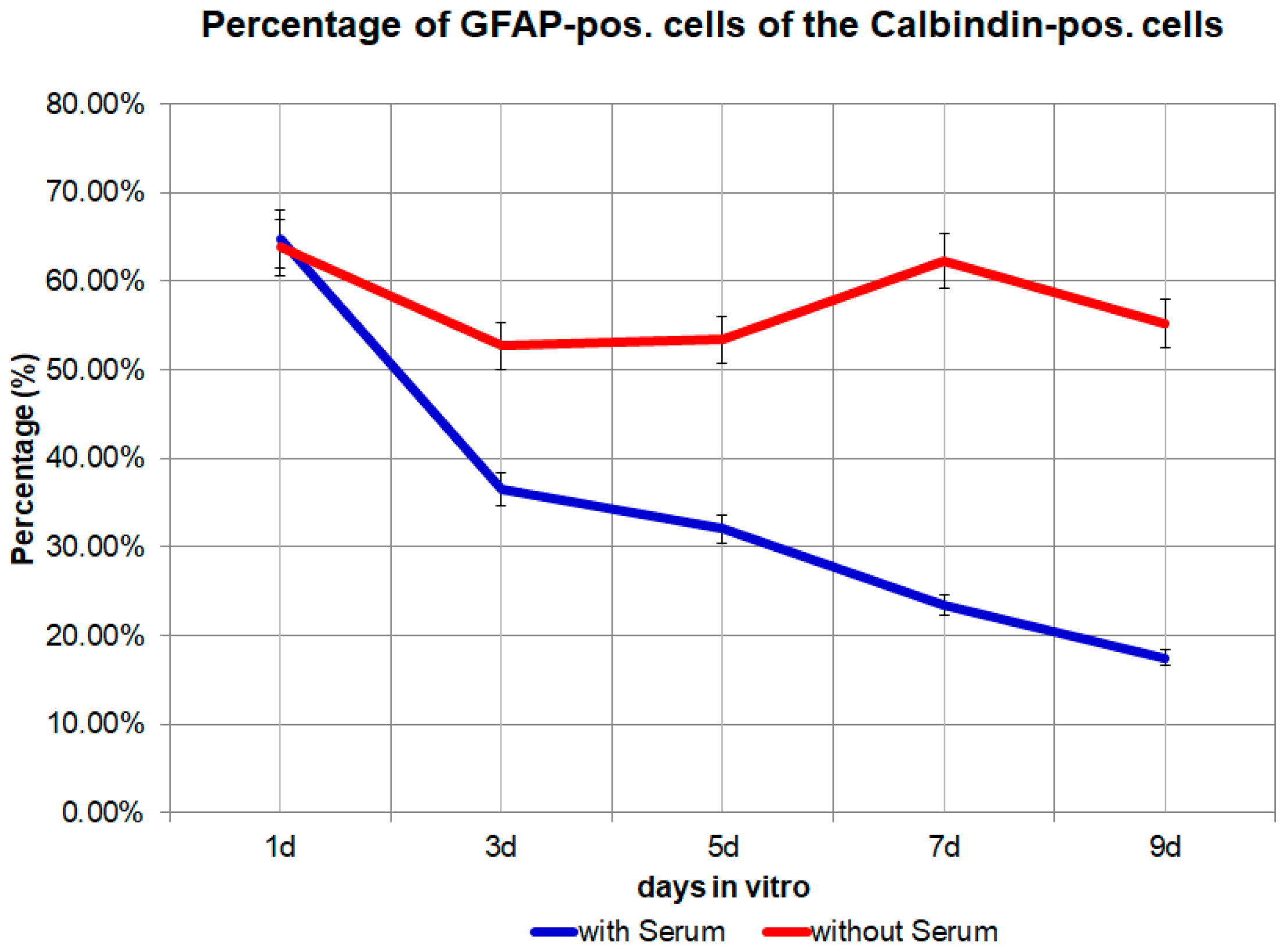
Figure 5.
The percentage of GFAP (glial fibrillary acidic protein) positive cells from day 1 to day 9 in the medium with 10% FBS (fetal bovine serum) or in the medium without FBS. p = 0.0142, significant.
Figure 5.
The percentage of GFAP (glial fibrillary acidic protein) positive cells from day 1 to day 9 in the medium with 10% FBS (fetal bovine serum) or in the medium without FBS. p = 0.0142, significant.
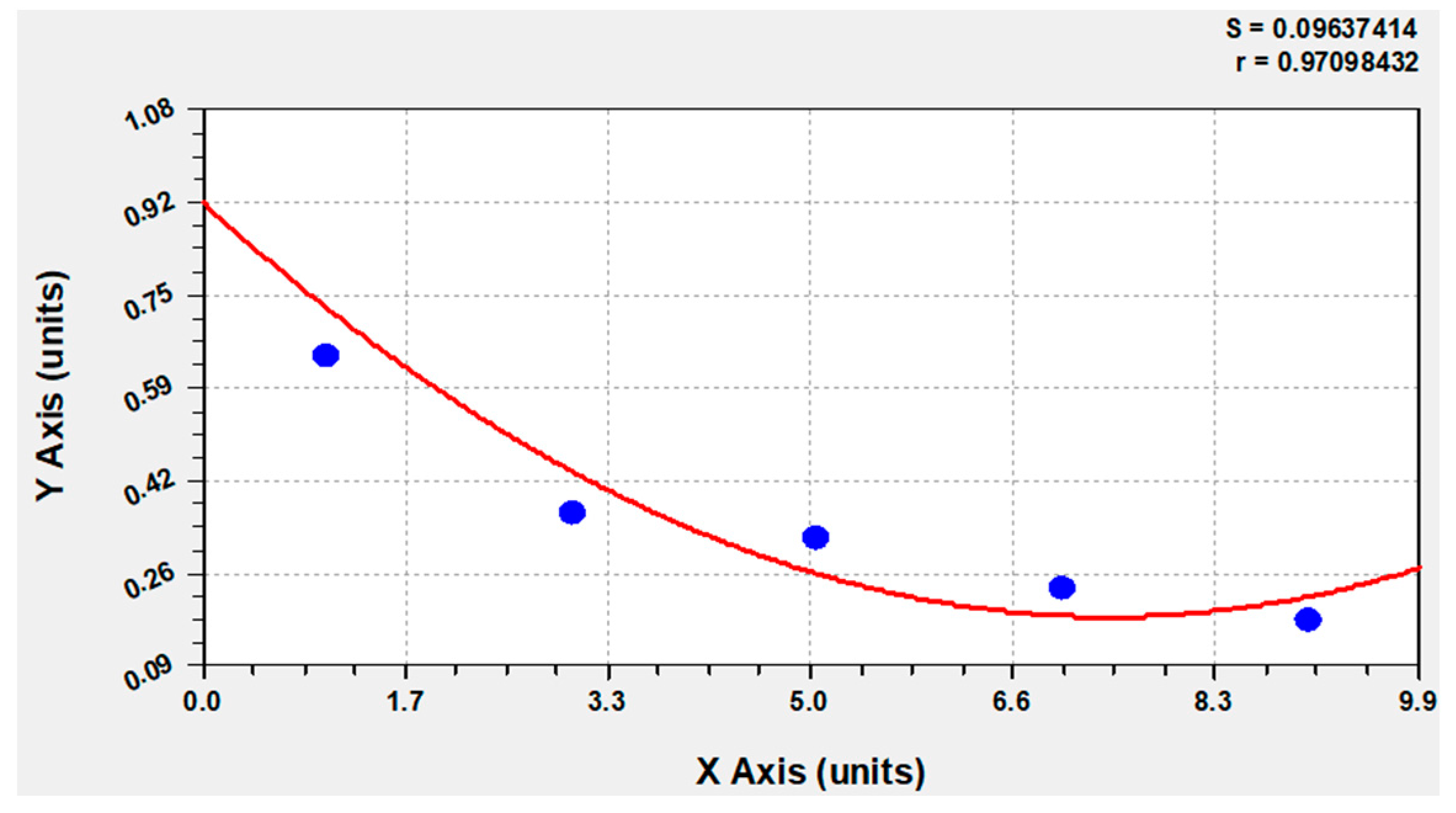
Figure 6.
The computer-based calculation showed at the outset of the investigation 92% that Calbindin D28k positive glial cells are also GFAP-positive. The blue circle: the percentage of GFAP positive cells from day 1 to day 9 in the medium with 10% FBS gradually decreased from 64.69% to −36.54% to −32.07% to −23.38% and finally to −17.47% of the total number of Calbindin D28k positive glial cells. The red line: adjustment of the data with Curve expert 1.4.
Figure 6.
The computer-based calculation showed at the outset of the investigation 92% that Calbindin D28k positive glial cells are also GFAP-positive. The blue circle: the percentage of GFAP positive cells from day 1 to day 9 in the medium with 10% FBS gradually decreased from 64.69% to −36.54% to −32.07% to −23.38% and finally to −17.47% of the total number of Calbindin D28k positive glial cells. The red line: adjustment of the data with Curve expert 1.4.
Figure 7.
Cultures in a serum-free medium, 5 days (6 days after the passage): Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488); Nuclear staining with DAPI (4′,6-diamidino-2-phenylindole dihydrochloride) (blue). Confocal microscopy. Zoom = 4, 2 Calbindin D28k positive and GFAP-positive astrocytes (long arrows) with processes (short arrows).
Figure 7.
Cultures in a serum-free medium, 5 days (6 days after the passage): Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488); Nuclear staining with DAPI (4′,6-diamidino-2-phenylindole dihydrochloride) (blue). Confocal microscopy. Zoom = 4, 2 Calbindin D28k positive and GFAP-positive astrocytes (long arrows) with processes (short arrows).
Figure 8.
Cultures in a serum-free medium, 5 days (6 days after the passage): Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488). Epifluorescence microscopy. One giant Calbindin D28k positive and GFAP-positive astrocyte (long arrows) with processes.
Figure 8.
Cultures in a serum-free medium, 5 days (6 days after the passage): Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488). Epifluorescence microscopy. One giant Calbindin D28k positive and GFAP-positive astrocyte (long arrows) with processes.
Figure 9.
Cultures in a serum-free medium, 5 days (6 after the passage): Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488). Epifluorescence microscopy. One giant Calbindin D28k positive and GFAP-positive astrocyte (long arrows) with processes.
Figure 9.
Cultures in a serum-free medium, 5 days (6 after the passage): Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488). Epifluorescence microscopy. One giant Calbindin D28k positive and GFAP-positive astrocyte (long arrows) with processes.
Figure 10.
Cultures in a serum-free medium, 5 days (6 days after the passage): Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488). Epifluorescence microscopy. Three Calbindin D28k positive and GFAP-positive astrocytes (long arrows) with processes.
Figure 10.
Cultures in a serum-free medium, 5 days (6 days after the passage): Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488). Epifluorescence microscopy. Three Calbindin D28k positive and GFAP-positive astrocytes (long arrows) with processes.
Figure 11.
Cultures in a serum-free medium, 5 days (6 days after the passage): Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488). Epifluorescence microscopy. Three Calbindin D28k positive and GFAP-positive astrocytes (long arrows) with processes.
Figure 11.
Cultures in a serum-free medium, 5 days (6 days after the passage): Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488). Epifluorescence microscopy. Three Calbindin D28k positive and GFAP-positive astrocytes (long arrows) with processes.
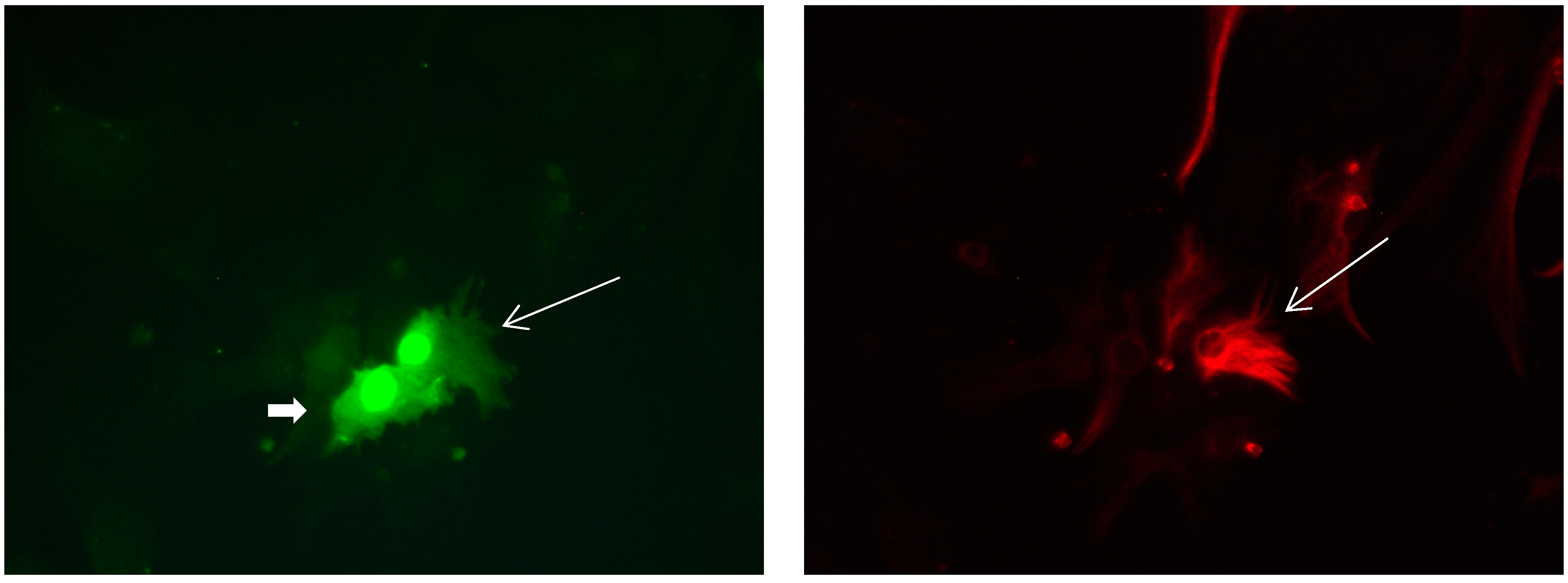
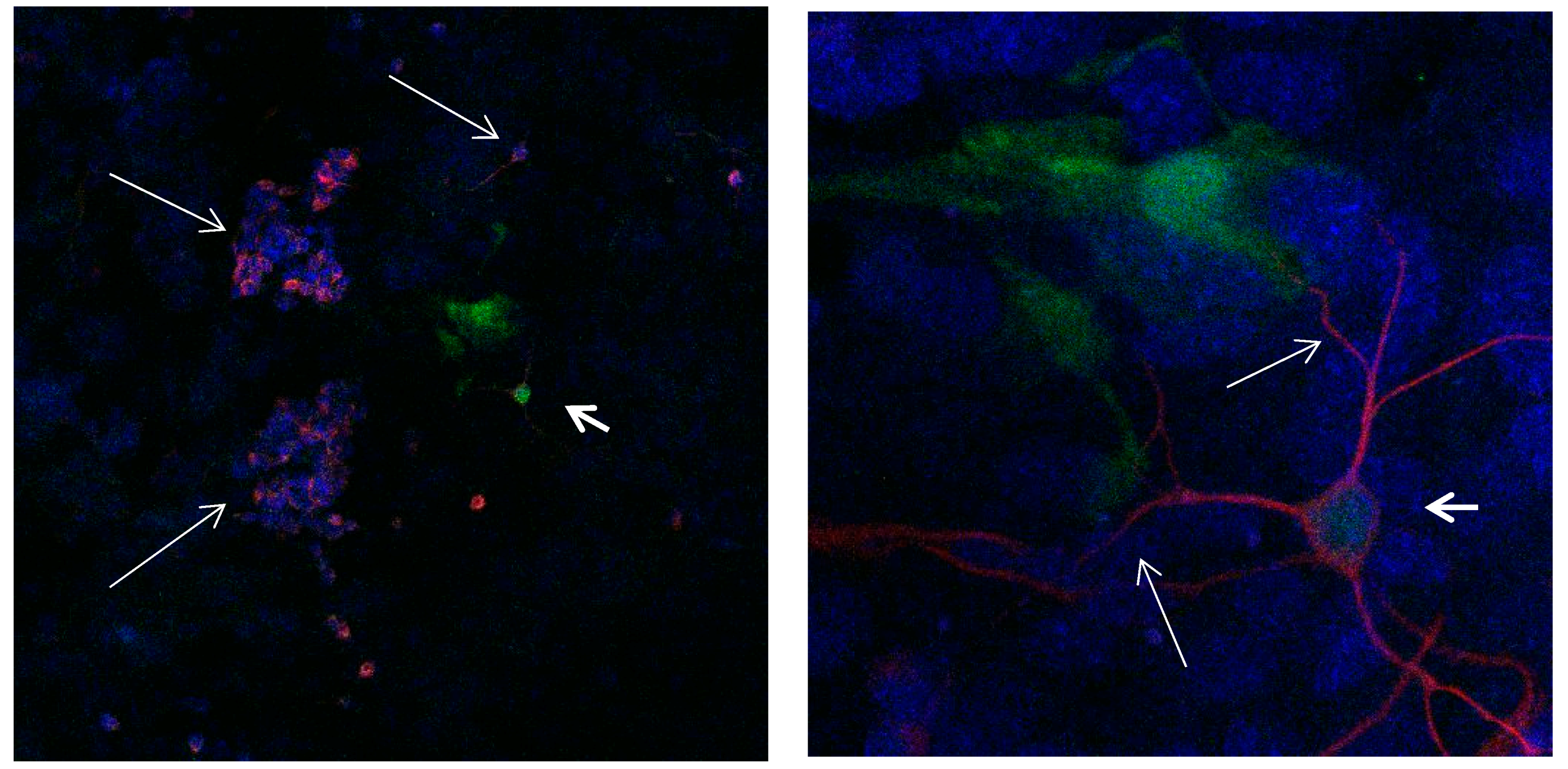
Figure 12.
Cultures in a serum-free medium, 3 days (4 days after the passage): Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488). Epifluorescence microscopy. One Calbindin D28k positive and GFAP-positive astrocyte (long arrows), besides 1 Calbindin D28k positive and GFAP-negative astrocyte (short arrows).
Figure 12.
Cultures in a serum-free medium, 3 days (4 days after the passage): Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488). Epifluorescence microscopy. One Calbindin D28k positive and GFAP-positive astrocyte (long arrows), besides 1 Calbindin D28k positive and GFAP-negative astrocyte (short arrows).
Figure 13.
Cultures in a serum-free medium, 3 days (4 days after the passage): Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488). Epifluorescence microscopy. One Calbindin D28k positive and GFAP-positive astrocyte (long arrows), besides, 1 Calbindin D28k positive and GFAP-weak-positive astrocyte (short arrows).
Figure 13.
Cultures in a serum-free medium, 3 days (4 days after the passage): Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488). Epifluorescence microscopy. One Calbindin D28k positive and GFAP-positive astrocyte (long arrows), besides, 1 Calbindin D28k positive and GFAP-weak-positive astrocyte (short arrows).
Figure 14.
Cultures in a serum-free medium, 1 day (2 days after the passage): Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488). Epifluorescence microscopy. One Calbindin D28k positive and GFAP-positive astrocyte (long arrows).
Figure 14.
Cultures in a serum-free medium, 1 day (2 days after the passage): Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488). Epifluorescence microscopy. One Calbindin D28k positive and GFAP-positive astrocyte (long arrows).
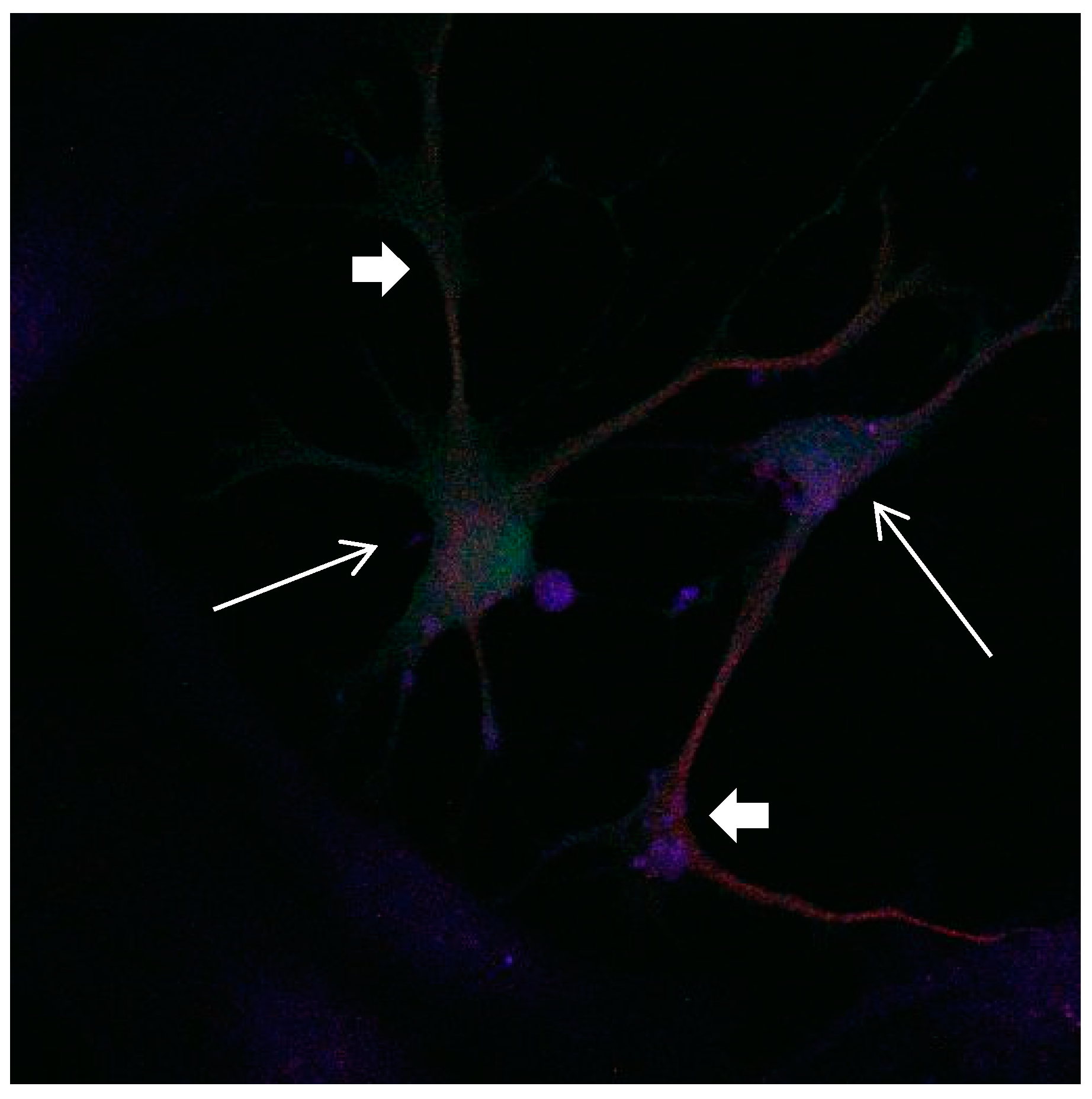
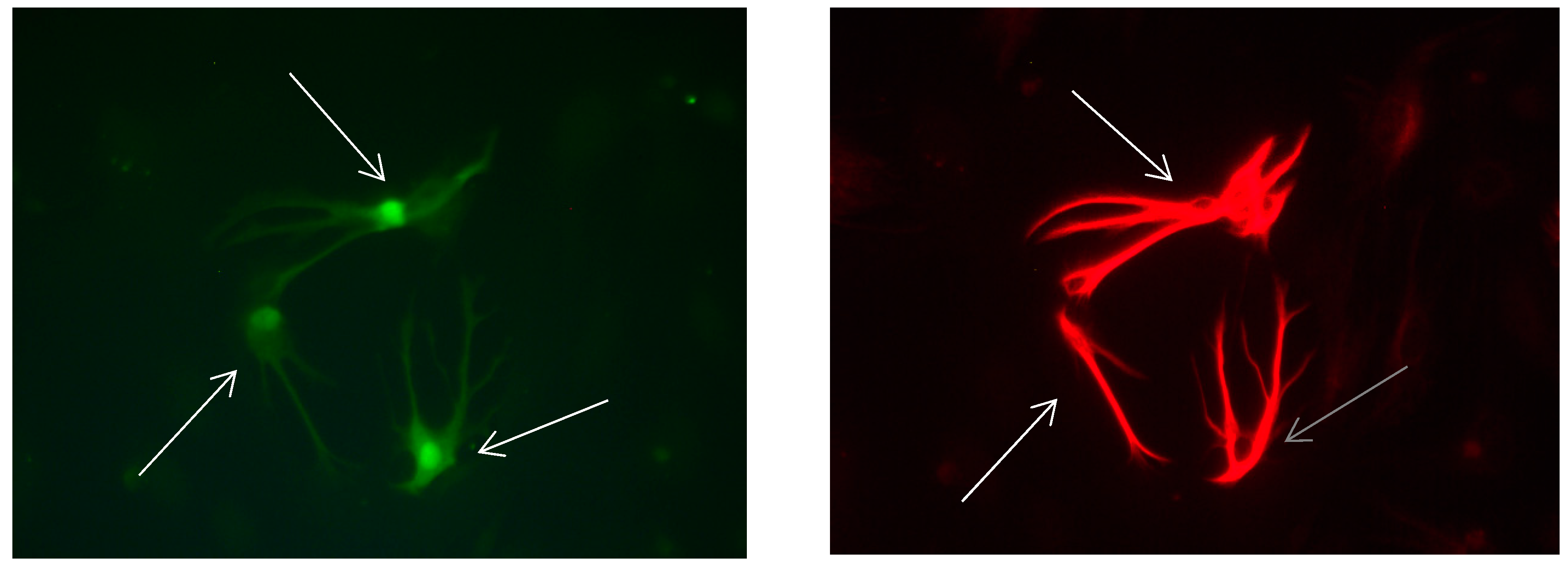
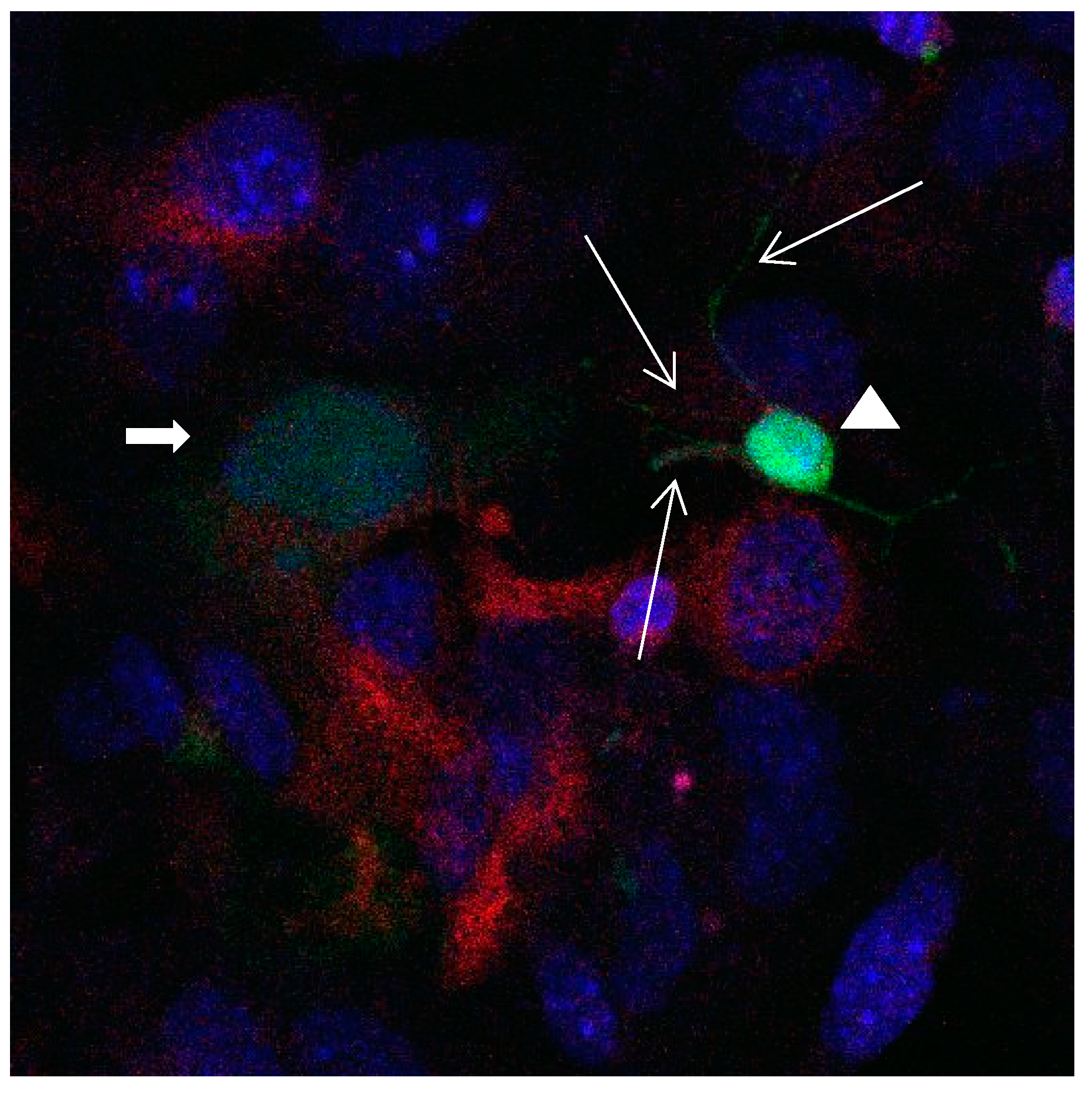
Figure 15.
Cultures in a serum-containing medium, 1 day after the passage: Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488); Nuclear staining with DAPI (4′,6-diamidino-2-phenylindole dihydrochloride) (blue). Confocal microscopy. Zoom = 4. Left: a Calbindin D28k positive and GFAP positive astrocyte (short arrows). Besides: a Calbindin D28k positive and Calbindin D28k expressing neuron (triangle) with extensions (long arrows).
Figure 15.
Cultures in a serum-containing medium, 1 day after the passage: Immunofluorescence with anti-GFAP (glial fibrillary acidic protein) (red, secondary antibody: donkey anti-goat IgG Alexa Fluor 594), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488); Nuclear staining with DAPI (4′,6-diamidino-2-phenylindole dihydrochloride) (blue). Confocal microscopy. Zoom = 4. Left: a Calbindin D28k positive and GFAP positive astrocyte (short arrows). Besides: a Calbindin D28k positive and Calbindin D28k expressing neuron (triangle) with extensions (long arrows).
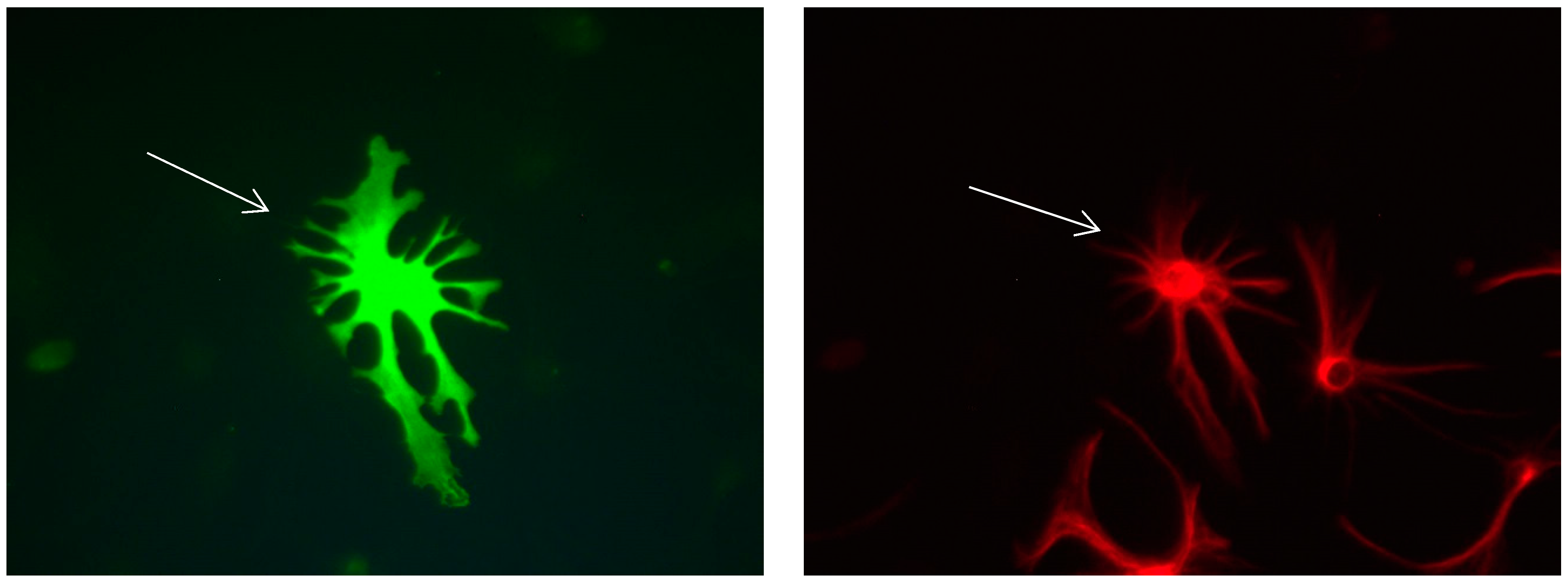
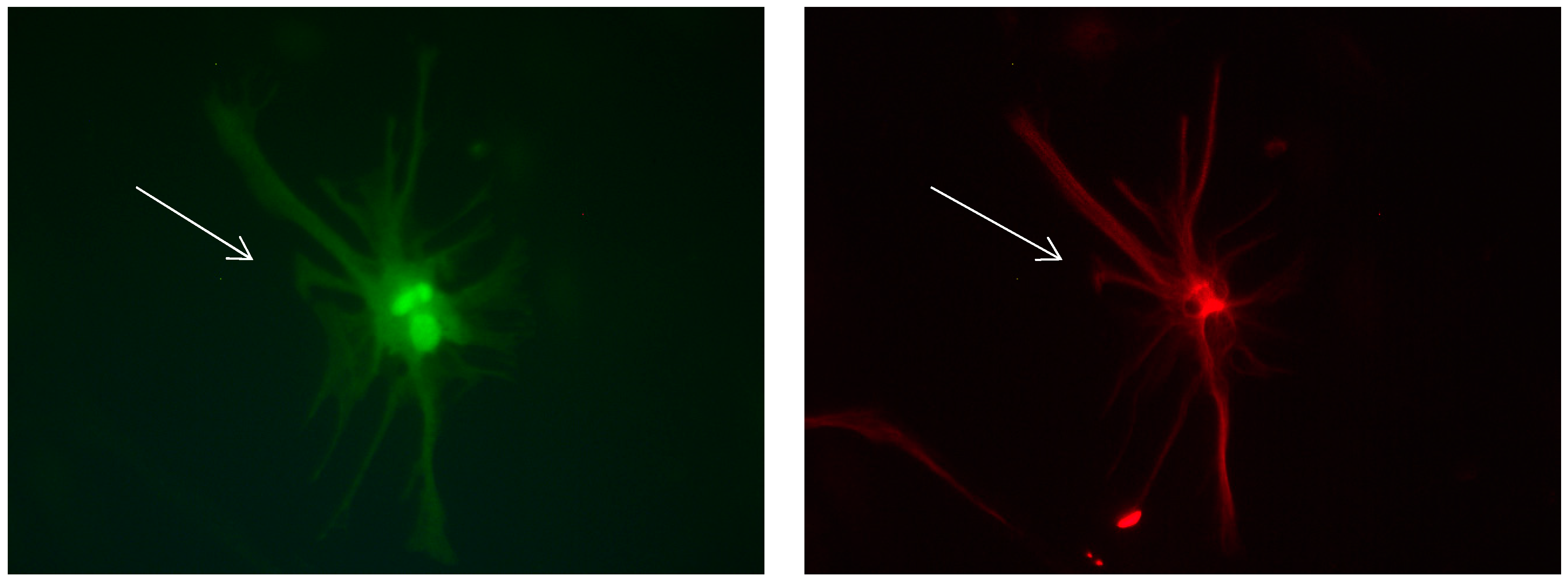
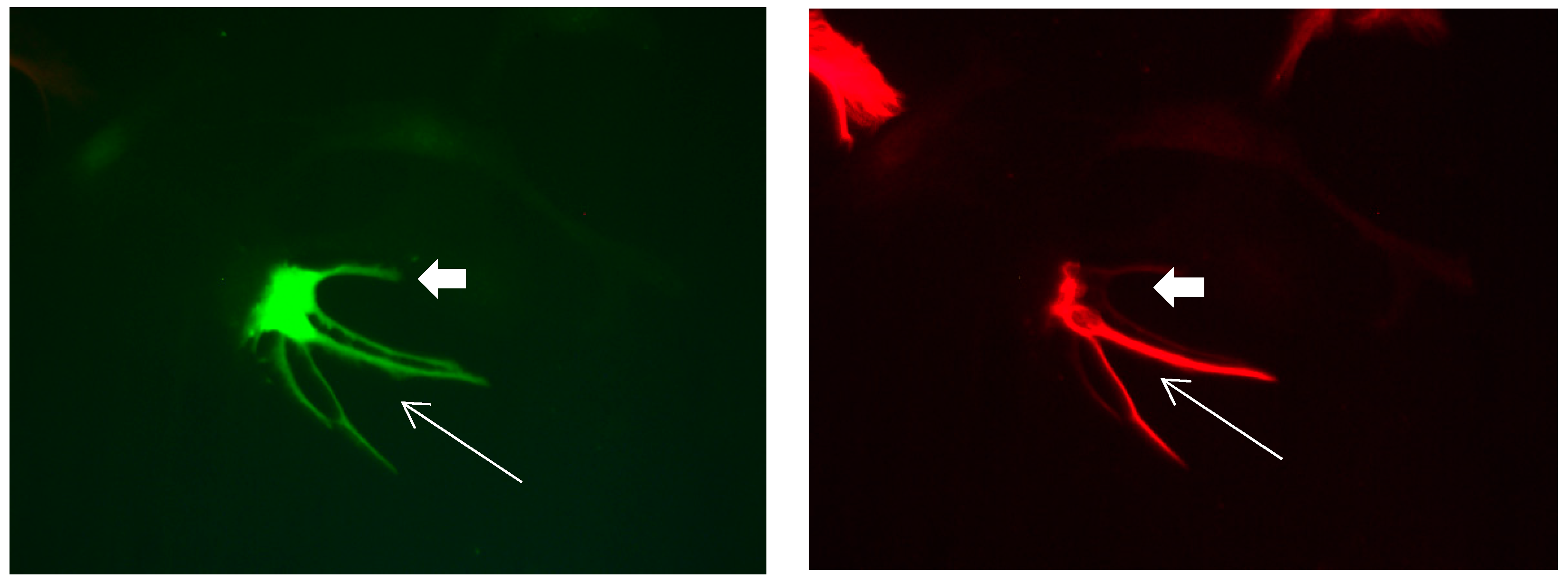
Figure 16.
Cultures in a serum-containing medium, 5 days after the passage: Immunofluorescence with anti-ß-tubulin-III (red, secondary antibody: goat anti-mouse IgG Alexa Fluor 555), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488); Nuclear staining with DAPI (4′,6-diamidino-2-phenylindole dihydrochloride) (blue). Confocal microscopy. Left: zoom = 1, numerous ß-tubulin-III-positive neurons (long arrows), on the right 1 Calbindin D28k positive and tubulin-III-positive neuron (short arrows). Right: zoom = 4, higher magnification of the Calbindin D28k positive and tubulin-III-positive neuron (short arrows) with extensions (long arrows). Above some Calbindin D28k positive astrocytes.
Figure 16.
Cultures in a serum-containing medium, 5 days after the passage: Immunofluorescence with anti-ß-tubulin-III (red, secondary antibody: goat anti-mouse IgG Alexa Fluor 555), and with anti-Calbindin D28k (polyclonal antibody from the company SWANT) (green, secondary antibody: donkey anti-rabbit IgG Alexa Fluor 488); Nuclear staining with DAPI (4′,6-diamidino-2-phenylindole dihydrochloride) (blue). Confocal microscopy. Left: zoom = 1, numerous ß-tubulin-III-positive neurons (long arrows), on the right 1 Calbindin D28k positive and tubulin-III-positive neuron (short arrows). Right: zoom = 4, higher magnification of the Calbindin D28k positive and tubulin-III-positive neuron (short arrows) with extensions (long arrows). Above some Calbindin D28k positive astrocytes.