Effect of Experimental Cutaneous Hand Pain on Corticospinal Excitability and Short Afferent Inhibition
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
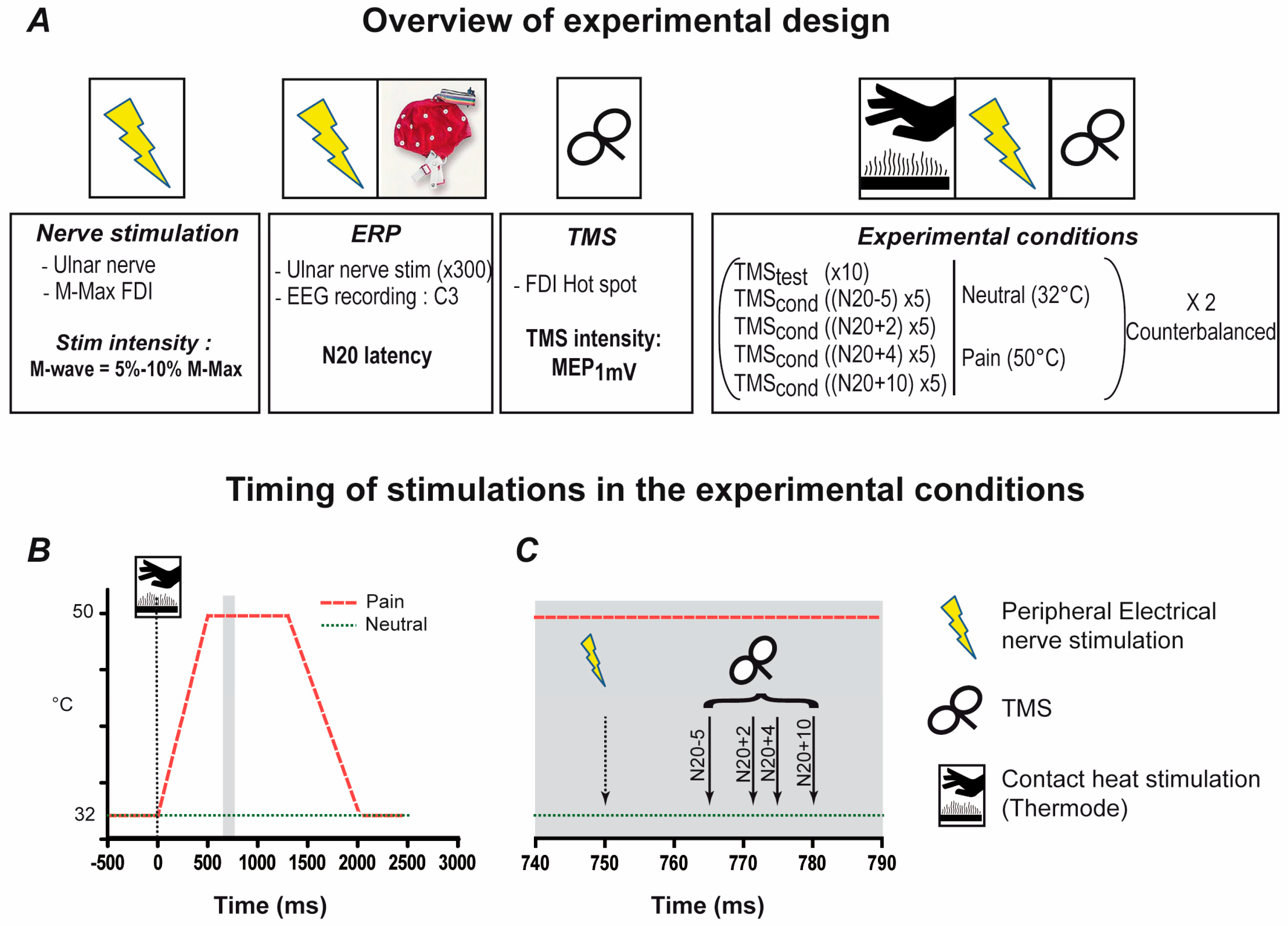
2.2. General Procedure
2.2.1. Preparation Phase
2.2.2. Experimental Phase
2.3. Data Reduction and Statistical Analysis
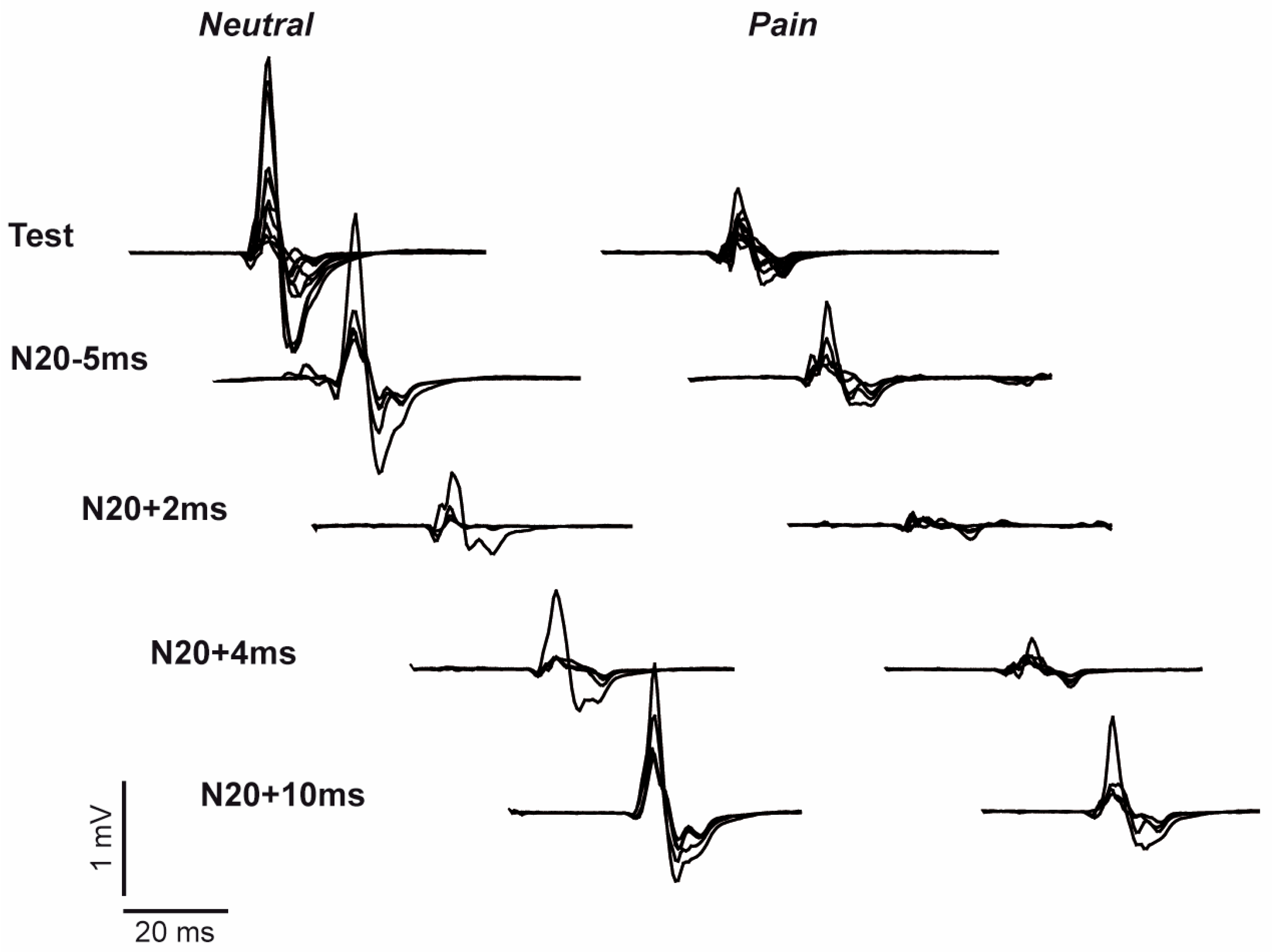
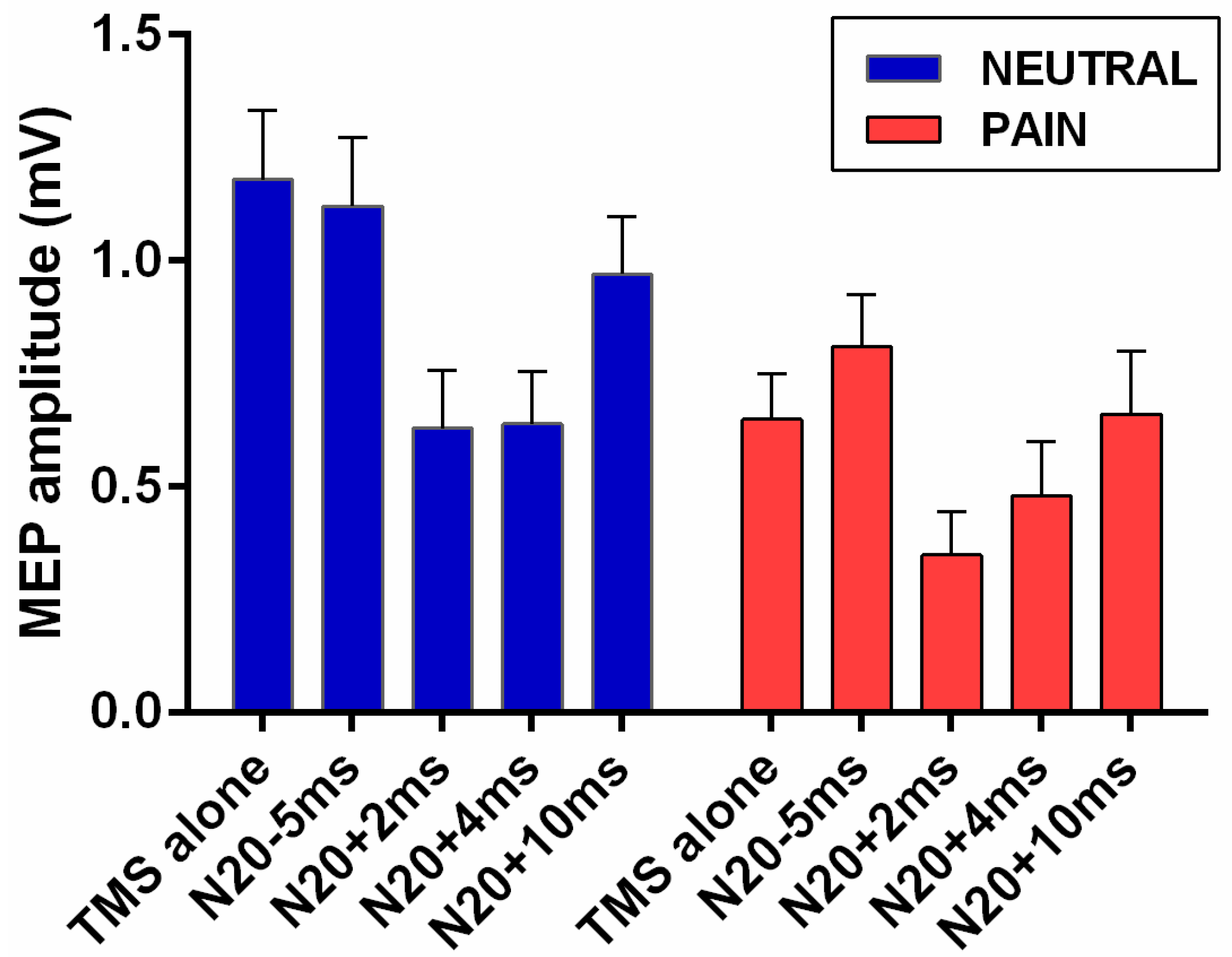
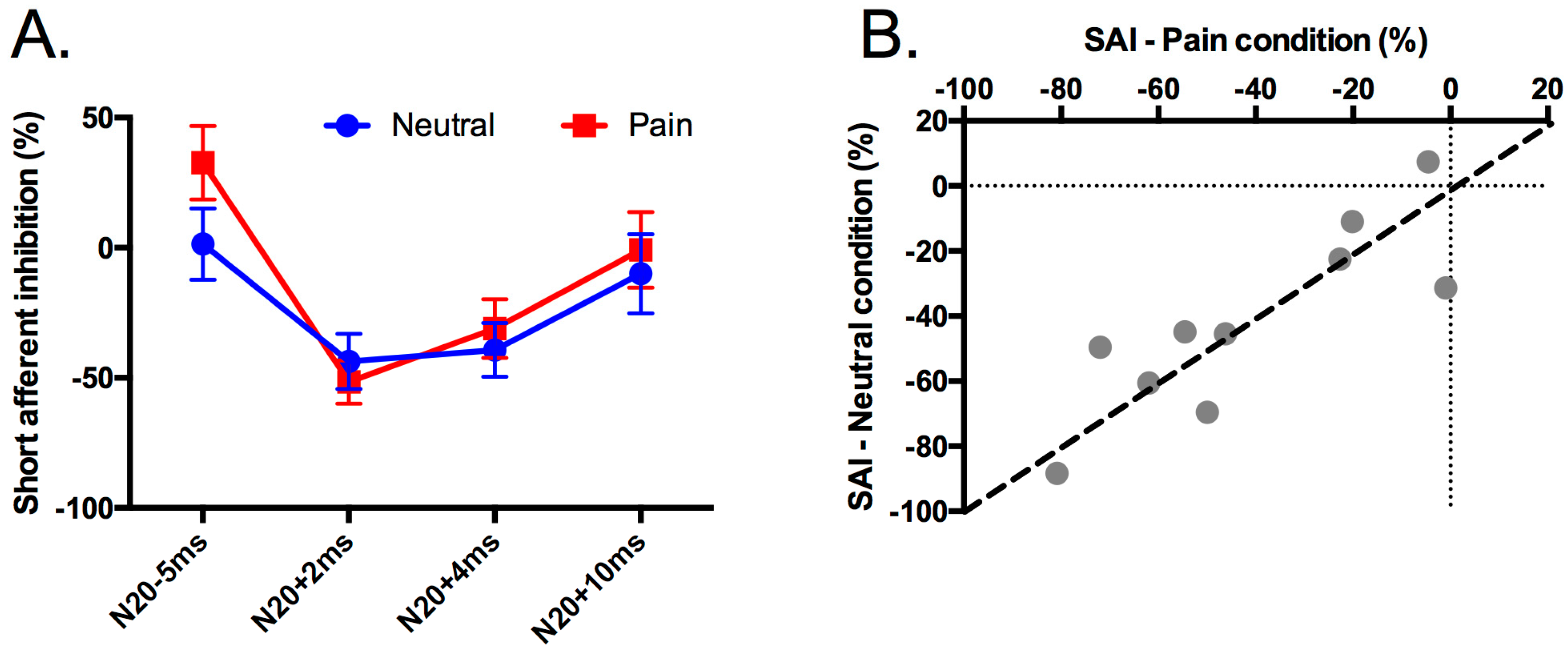
3. Results
3.1. Stimulation Parameters
3.2. Effect of Pain on Short Afferent Inhibition
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gagne, M.; Reilly, K.T.; Hetu, S.; Mercier, C. Motor control over the phantom limb in above-elbow amputees and its relationship with phantom limb pain. Neuroscience 2009, 162, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Reilly, K.T.; Mercier, C.; Schieber, M.H.; Sirigu, A. Persistent hand motor commands in the amputees’ brain. Brain 2006, 129, 2211–2223. [Google Scholar] [CrossRef] [PubMed]
- Raffin, E.; Mattout, J.; Reilly, K.T.; Giraux, P. Disentangling motor execution from motor imagery with the phantom limb. Brain 2012, 135, 582–595. [Google Scholar] [CrossRef] [PubMed]
- Harris, A.J. Cortical origin of pathological pain. Lancet 1999, 354, 1464–1466. [Google Scholar] [CrossRef]
- Daenen, L.; Nijs, J.; Roussel, N.; Wouters, K.; Van Loo, M.; Cras, P. Sensorimotor incongruence exacerbates symptoms in patients with chronic whiplash associated disorders: An experimental study. Rheumatology 2012, 51, 1492–1499. [Google Scholar] [CrossRef] [PubMed]
- McCabe, C.S.; Cohen, H.; Blake, D.R. Somaesthetic disturbances in fibromyalgia are exaggerated by sensory motor conflict: Implications for chronicity of the disease? Rheumatology 2007, 46, 1587–1592. [Google Scholar] [CrossRef] [PubMed]
- Thieme, H.; Morkisch, N.; Rietz, C.; Dohle, C.; Borgetto, B. The efficacy of movement representation techniques for treatment of limb pain—A systematic review and meta-analysis. J. Pain 2016, 17, 167–180. [Google Scholar] [CrossRef] [PubMed]
- Boesch, E.; Bellan, V.; Moseley, G.L.; Stanton, T.R. The effect of bodily illusions on clinical pain: A systematic review and meta-analysis. Pain 2016, 157, 516–529. [Google Scholar] [CrossRef] [PubMed]
- Perry, B.N.; Mercier, C.; Pettifer, S.R.; Cole, J.; Tsao, J.W. Virtual reality therapies for phantom limb pain. Eur. J. Pain 2014, 18, 897–899. [Google Scholar] [CrossRef] [PubMed]
- Di Pietro, F.; McAuley, J.H.; Parkitny, L.; Lotze, M.; Wand, B.M.; Moseley, G.L.; Stanton, T.R. Primary motor cortex function in complex regional pain syndrome: A systematic review and meta-analysis. J. Pain 2013, 14, 1270–1288. [Google Scholar] [CrossRef] [PubMed]
- Di Pietro, F.; McAuley, J.H.; Parkitny, L.; Lotze, M.; Wand, B.M.; Moseley, G.L.; Stanton, T.R. Primary somatosensory cortex function in complex regional pain syndrome: A systematic review and meta-analysis. J. Pain 2013, 14, 1001–1018. [Google Scholar] [CrossRef] [PubMed]
- Henry, D.E.; Chiodo, A.E.; Yang, W. Central nervous system reorganization in a variety of chronic pain states: A review. PM R 2011, 3, 1116–1125. [Google Scholar] [CrossRef] [PubMed]
- Nardone, R.; Holler, Y.; Brigo, F.; Seidl, M.; Christova, M.; Bergmann, J.; Golaszewski, S.; Trinka, E. Functional brain reorganization after spinal cord injury: Systematic review of animal and human studies. Brain Res. 2013, 1504, 58–73. [Google Scholar] [CrossRef] [PubMed]
- Pelletier, R.; Higgins, J.; Bourbonnais, D. Is neuroplasticity in the central nervous system the missing link to our understanding of chronic musculoskeletal disorders? BMC Musculoskelet. Disord. 2015, 16, 25. [Google Scholar] [CrossRef] [PubMed]
- Avendano, C.; Isla, A.J.; Rausell, E. Area 3a in the cat. II. Projections to the motor cortex and their relations to other corticocortical connections. J. Comp. Neurol. 1992, 321, 373–386. [Google Scholar] [CrossRef] [PubMed]
- Widener, G.L.; Cheney, P.D. Effects on muscle activity from microstimuli applied to somatosensory and motor cortex during voluntary movement in the monkey. J. Neurophysiol. 1997, 77, 2446–2465. [Google Scholar] [PubMed]
- Tokimura, H.; Di Lazzaro, V.; Tokimura, Y.; Oliviero, A.; Profice, P.; Insola, A.; Mazzone, P.; Tonali, P.; Rothwell, J.C. Short latency inhibition of human hand motor cortex by somatosensory input from the hand. J. Physiol. 2000, 523 Pt 2, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Bradnam, L.; Shanahan, E.M.; Hendy, K.; Reed, A.; Skipworth, T.; Visser, A.; Lennon, S. Afferent inhibition and cortical silent periods in shoulder primary motor cortex and effect of a suprascapular nerve block in people experiencing chronic shoulder pain. Clin. Neurophysiol. 2016, 127, 769–778. [Google Scholar] [CrossRef] [PubMed]
- Turton, A.J.; McCabe, C.S.; Harris, N.; Filipovic, S.R. Sensorimotor integration in complex regional pain syndrome: A transcranial magnetic stimulation study. Pain 2007, 127, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Fischer, M.; Orth, M. Short-latency sensory afferent inhibition: Conditioning stimulus intensity, recording site, and effects of 1 Hz repetitive TMS. Brain Stimul. 2011, 4, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Dube, J.A.; Mercier, C. Effect of pain and pain expectation on primary motor cortex excitability. Clin. Neurophysiol. 2011, 122, 2318–2323. [Google Scholar] [CrossRef] [PubMed]
- Farina, S.; Valeriani, M.; Rosso, T.; Aglioti, S.; Tamburin, S.; Fiaschi, A.; Tinazzi, M. Transient inhibition of the human motor cortex by capsaicin-induced pain. A study with transcranial magnetic stimulation. Neurosci. Lett. 2001, 314, 97–101. [Google Scholar] [CrossRef]
- Fierro, B.; De Tommaso, M.; Giglia, F.; Giglia, G.; Palermo, A.; Brighina, F. Repetitive transcranial magnetic stimulation (rTMS) of the dorsolateral prefrontal cortex (DLPFC) during capsaicin-induced pain: Modulatory effects on motor cortex excitability. Exp. Brain Res. 2010, 203, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Le Pera, D.; Graven-Nielsen, T.; Valeriani, M.; Oliviero, A.; Di Lazzaro, V.; Tonali, P.A.; Arendt-Nielsen, L. Inhibition of motor system excitability at cortical and spinal level by tonic muscle pain. Clin. Neurophysiol. 2001, 112, 1633–1641. [Google Scholar] [CrossRef]
- Martin, P.G.; Weerakkody, N.; Gandevia, S.C.; Taylor, J.L. Group III and IV muscle afferents differentially affect the motor cortex and motoneurones in humans. J. Physiol. 2008, 586, 1277–1289. [Google Scholar] [CrossRef] [PubMed]
- Valeriani, M.; Restuccia, D.; Di Lazzaro, V.; Oliviero, A.; Le Pera, D.; Profice, P.; Saturno, E.; Tonali, P. Inhibition of biceps brachii muscle motor area by painful heat stimulation of the skin. Exp. Brain Res. 2001, 139, 168–172. [Google Scholar] [CrossRef] [PubMed]
- Valeriani, M.; Restuccia, D.; Di Lazzaro, V.; Oliviero, A.; Profice, P.; Le Pera, D.; Saturno, E.; Tonali, P. Inhibition of the human primary motor area by painful heat stimulation of the skin. Clin. Neurophysiol. 1999, 110, 1475–1480. [Google Scholar] [CrossRef]
- Burns, E.; Chipchase, L.S.; Schabrun, S.M. Reduced short- and long-latency afferent inhibition following acute muscle pain: A potential role in the recovery of motor output. Pain Med. 2016. [Google Scholar] [CrossRef] [PubMed]
- Schabrun, S.M.; Burns, E.; Hodges, P.W. New insight into the time-course of motor and sensory system changes in pain. PLoS ONE 2015, 10, e0142857. [Google Scholar] [CrossRef] [PubMed]
- Rossi, A.; Decchi, B.; Groccia, V.; Della Volpe, R.; Spidalieri, R. Interactions between nociceptive and non-nociceptive afferent projections to cerebral cortex in humans. Neurosci. Lett. 1998, 248, 155–158. [Google Scholar] [CrossRef]
- Rossi, S.; della Volpe, R.; Ginanneschi, F.; Ulivelli, M.; Bartalini, S.; Spidalieri, R.; Rossi, A. Early somatosensory processing during tonic muscle pain in humans: Relation to loss of proprioception and motor ‘defensive’ strategies. Clin. Neurophysiol. 2003, 114, 1351–1358. [Google Scholar] [CrossRef]
- McCabe, C.S.; Haigh, R.C.; Halligan, P.W.; Blake, D.R. Simulating sensory-motor incongruence in healthy volunteers: Implications for a cortical model of pain. Rheumatology 2005, 44, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Di Lazzaro, V.; Pilato, F.; Dileone, M.; Tonali, P.A.; Ziemann, U. Dissociated effects of diazepam and lorazepam on short-latency afferent inhibition. J. Physiol. 2005, 569, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Ni, Z.; Gunraj, C.; Wagle-Shukla, A.; Udupa, K.; Mazzella, F.; Lozano, A.M.; Chen, R. Direct demonstration of inhibitory interactions between long interval intracortical inhibition and short interval intracortical inhibition. J. Physiol. 2011, 589, 2955–2962. [Google Scholar] [CrossRef] [PubMed]
- Udupa, K.; Ni, Z.; Gunraj, C.; Chen, R. Interactions between short latency afferent inhibition and long interval intracortical inhibition. Exp. Brain Res. 2009, 199, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Garry, M.I.; Thomson, R.H. The effect of test tms intensity on short-interval intracortical inhibition in different excitability states. Exp. Brain. Res. 2009, 193, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Ni, Z.; Charab, S.; Gunraj, C.; Nelson, A.J.; Udupa, K.; Yeh, I.J.; Chen, R. Transcranial magnetic stimulation in different current directions activates separate cortical circuits. J. Neurophysiol. 2011, 105, 749–756. [Google Scholar] [CrossRef] [PubMed]
- Schabrun, S.M.; Christensen, S.W.; Mrachacz-Kersting, N.; Graven-Nielsen, T. Motor cortex reorganization and impaired function in the transition to sustained muscle pain. Cereb. Cortex 2016, 26, 1878–1890. [Google Scholar] [CrossRef] [PubMed]
- Suppa, A.; Biasiotta, A.; Belvisi, D.; Marsili, L.; La Cesa, S.; Truini, A.; Cruccu, G.; Berardelli, A. Heat-evoked experimental pain induces long-term potentiation-like plasticity in human primary motor cortex. Cereb. Cortex 2013, 23, 1942–1951. [Google Scholar] [CrossRef] [PubMed]
- Dancey, E.; Murphy, B.; Andrew, D.; Yielder, P. The interactive effect of acute pain and motor learning acquisition on sensorimotor integration and motor learning outcomes. J. Neurophysiol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Dancey, E.; Murphy, B.; Srbely, J.; Yielder, P. The effect of experimental pain on motor training performance and sensorimotor integration. Exp. Brain Res. 2014, 232, 2879–2889. [Google Scholar] [CrossRef] [PubMed]
- Dancey, E.; Murphy, B.A.; Andrew, D.; Yielder, P. The effect of local vs remote experimental pain on motor learning and sensorimotor integration using a complex typing task. Pain 2016, 157, 1682–1695. [Google Scholar] [CrossRef] [PubMed]
- Dowman, R. Laser pain fails to inhibit innocuous-related activity in the central somatosensory pathways. Psychophysiology 1999, 36, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Mouraux, A.; Plaghki, L. Cortical interactions and integration of nociceptive and non-nociceptive somatosensory inputs in humans. Neuroscience 2007, 150, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Burns, E.; Chipchase, L.S.; Schabrun, S.M. Primary sensory and motor cortex function in response to acute muscle pain: A systematic review and meta-analysis. Eur. J. Pain 2016, 20, 1203–1213. [Google Scholar] [CrossRef] [PubMed]
- Rice, D.A.; McNair, P.J. Quadriceps arthrogenic muscle inhibition: Neural mechanisms and treatment perspectives. Semin. Arthritis Rheum. 2010, 40, 250–266. [Google Scholar] [CrossRef] [PubMed]
- Rice, D.A.; McNair, P.J.; Lewis, G.N.; Dalbeth, N. Quadriceps arthrogenic muscle inhibition: The effects of experimental knee joint effusion on motor cortex excitability. Arthritis Res. Ther. 2014, 16, 502. [Google Scholar] [CrossRef] [PubMed]
- Schabrun, S.M.; Hodges, P.W. Muscle pain differentially modulates short interval intracortical inhibition and intracortical facilitation in primary motor cortex. J. Pain 2012, 13, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Nishigami, T.; Nakano, H.; Osumi, M.; Tsujishita, M.; Mibu, A.; Ushida, T. Central neural mechanisms of interindividual difference in discomfort during sensorimotor incongruence in healthy volunteers: An experimental study. Rheumatology 2014, 53, 1194–1199. [Google Scholar] [CrossRef] [PubMed]
- Karabanov, A.; Jin, S.H.; Joutsen, A.; Poston, B.; Aizen, J.; Ellenstein, A.; Hallett, M. Timing-dependent modulation of the posterior parietal cortex-primary motor cortex pathway by sensorimotor training. J. Neurophysiol. 2012, 107, 3190–3199. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mercier, C.; Gagné, M.; Reilly, K.T.; Bouyer, L.J. Effect of Experimental Cutaneous Hand Pain on Corticospinal Excitability and Short Afferent Inhibition. Brain Sci. 2016, 6, 45. https://doi.org/10.3390/brainsci6040045
Mercier C, Gagné M, Reilly KT, Bouyer LJ. Effect of Experimental Cutaneous Hand Pain on Corticospinal Excitability and Short Afferent Inhibition. Brain Sciences. 2016; 6(4):45. https://doi.org/10.3390/brainsci6040045
Chicago/Turabian StyleMercier, Catherine, Martin Gagné, Karen T. Reilly, and Laurent J. Bouyer. 2016. "Effect of Experimental Cutaneous Hand Pain on Corticospinal Excitability and Short Afferent Inhibition" Brain Sciences 6, no. 4: 45. https://doi.org/10.3390/brainsci6040045
APA StyleMercier, C., Gagné, M., Reilly, K. T., & Bouyer, L. J. (2016). Effect of Experimental Cutaneous Hand Pain on Corticospinal Excitability and Short Afferent Inhibition. Brain Sciences, 6(4), 45. https://doi.org/10.3390/brainsci6040045







