Subliminal and Supraliminal Processing of Facial Expression of Emotions: Brain Oscillation in the Left/Right Frontal Area
Abstract
:1. Introduction
1.1. Brain Activity and Emotional Facial Expression
1.2. The Role of Consciousness: Subliminal/Supraliminal Stimulation Effect in Face Processing
2. Objectives and Hypotheses
3. Method
3.1. Subject
3.2. Stimulus Material and Procedure
3.3. Backward Masking Procedure
3.4. EEG Acquisition and Data Reduction
4. Results
| Right (Supra-) | center (Supra-) | Right (Subli-) | center (Subli-) | |||||
|---|---|---|---|---|---|---|---|---|
| M | (SD) | M | (SD) | M | (SD) | M | (SD) | |
| anger | 6.58 a | 1.28 | 8.08 | 1.10 | 7.34 | 1.47 | 9.56 | 1.56 |
| fear | 7.05 | 1.30 | 8.05 | 1.24 | 7.60 | 1.27 | 9.77 | 1.09 |
| disgust | 7.09 | 1.05 | 8.20 | 0.89 | 8.73 | 1.40 | 9.46 | 0.98 |
| surprise | 7.10 | 1.27 | 8.43 | 0.43 | 7.35 | 0.83 | 9.30 | 0.97 |
| happiness | 8.53 | 1.87 | 6.70 | 1.66 | 12.10 | 1.05 | 8.15 | 1.53 |
| sadness | 10.98 | 1.50 | 10.89 | 1.32 | 11.43 | 1.22 | 9.87 | 1.23 |
| neutral | 12.30 | 1.33 | 12.78 | 0.72 | 12.77 | 1.19 | 11.02 | 1.90 |
| Source | df | F | p | η2 | |
|---|---|---|---|---|---|
| emotion | 6 | 12.43 | 0.001 | 0.40 | |
| emotion × side | 6 | 9.61 | 0.001 | 0.35 | |
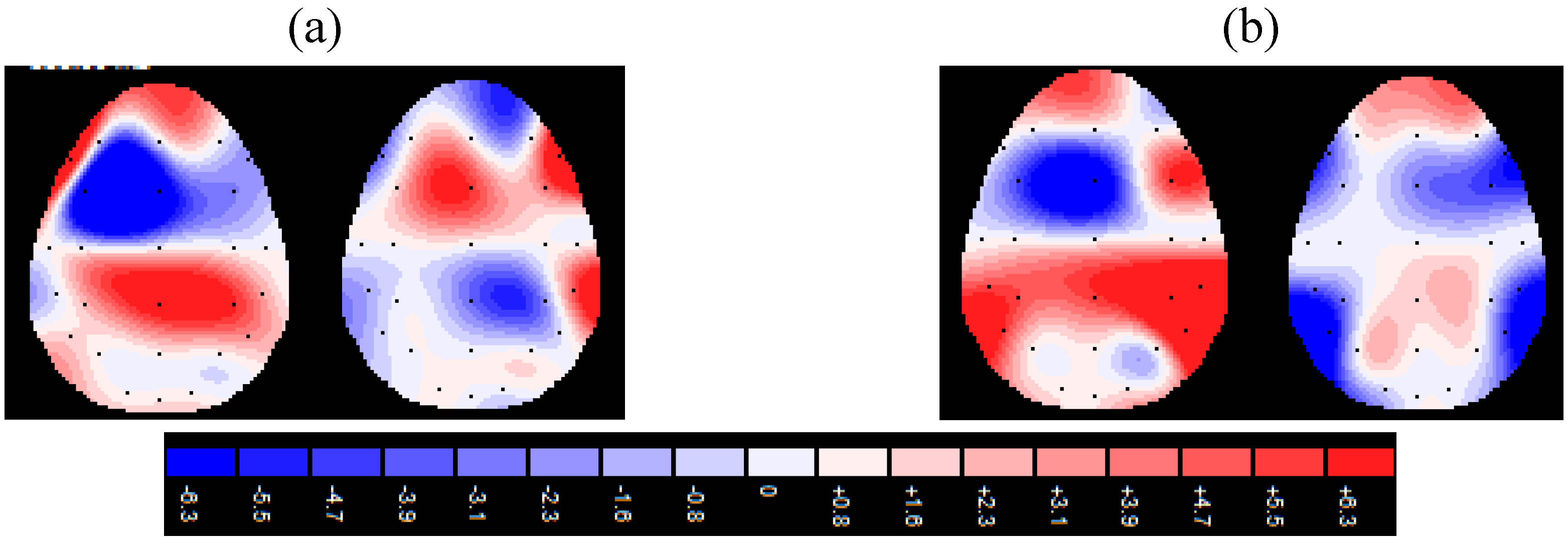
| Source | df | F | p | η2 | |
|---|---|---|---|---|---|
| emotion | 6 | 10.16 | 0.001 | 0.43 | |
| emotion x side | 6 | 9.78 | 0.001 | 0.36 | |
| Source | df | F | p | η2 | |
|---|---|---|---|---|---|
| emotion | 6 | 10.09 | 0.001 | 0.42 | |
| emotion × side | 6 | 9.33 | 0.001 | 0.37 | |
5. Discussion
6. Conclusions
Acknowledgements
References
- Ekman, P. Facial expressions and emotions. Am. Psychol. 1993, 48, 384–392. [Google Scholar] [CrossRef]
- Adolphs, R. Cognitive neuroscience of human social behaviour. Nat. Rev. Neurosci. 2003, 4, 165–178. [Google Scholar] [CrossRef]
- Balconi, M. Neuropsicologia della Comunicazione; Springer-Verlag: New York, NY, USA, 2008. [Google Scholar]
- Eastwood, J.D. From unconscious to conscious perception: Emotionally expressive faces and visual awareness. Int. Brain Sci. Eng. 2003, 64, 988. [Google Scholar]
- Eastwood, J.D.; Smilek, D. Functional consequences of perceiving facial expressions of emotion without awareness. Conscious. Cogn. 2005, 14, 565–584. [Google Scholar] [CrossRef]
- Balconi, M.; Lucchiari, C. Consciousness and emotional facial expression recognition: Subliminal/supraliminal stimulation effect on N200 and P300 ERPs. J. Psychophysiol. 2007, 21, 100–108. [Google Scholar] [CrossRef]
- Balconi, M.; Pozzoli, U. Arousal effect on emotional face comprehension. Frequency band changes in different time intervals. Physiol. Behav. 2009, 97, 455–462. [Google Scholar] [CrossRef]
- Başar, E.; Demiralp, T.; Schürmann, M.; Basar-Eroglu, C.; Ademoglu, A. Oscillatory brain dynamics, wavelet analysis, and cognition. Brain Lang. 1999, 66, 146–183. [Google Scholar] [CrossRef]
- Knyazev, G.G. Motivation, emotion, and their inhibitory control mirrored in brain oscillations. Neurosci. Biobehav. Rev. 2007, 31, 377–395. [Google Scholar] [CrossRef]
- Krause, C.M. Brain electric oscillations and cognitive processes. In Experimental Methods in Neuropsychology; Hugdahl, K., Ed.; Kluwer: New York, NY, USA, 2003; pp. 111–130. [Google Scholar]
- Aftanas, L.I.; Varlamov, A.A.; Pavlov, S.V.; Makhnev, V.P.; Reva, N.V. Time-dependent cortical asymmetries induced by emotional arousal: EEG analysis of event-related synchronization and desynchronization in individually defined frequency bands. Int. J. Psychophysiol. 2002, 44, 67–82. [Google Scholar] [CrossRef]
- Klimesch, W.; Doppelmayer, M.; Russegger, H.; Pachinger, T.; Schwaiger, J. Induced alpha band power changes in the human EEG and attention. Neurosci. Lett. 1998, 244, 73–76. [Google Scholar] [CrossRef]
- Aftanas, L.I.; Koshkarov, V.I.; Pokrovskaja, V.L.; Lotova, N.V.; Mordvintsev, Y. Event-related desynchronization (ERD) patterns to emotion-related feedback stimuli. Int. J. Neurosci. 1996, 87, 151–173. [Google Scholar] [CrossRef]
- Balconi, M.; Mazza, G. Lateralisation effect in comprehension of emotional facial expression: A comparison between EEG alpha band power and behavioural inhibition (BIS) and activation (BAS) systems. Laterality 2010, 3, 1257–1268. [Google Scholar]
- Davidson, R.J. Anterior electrophysiological asymmetries, emotion, and depression: Conceptual and methodological conundrums. Psychophysiology 1998, 35, 607–614. [Google Scholar] [CrossRef]
- Dimberg, U.; Petterson, M. Facial reactions to happy and angry facial expressions: Evidence for right hemisphere dominance. Psychophysiology 2000, 37, 693–696. [Google Scholar] [CrossRef]
- Davidson, R.J.; Henriques, J. Regional brain function in sadness and depression. The neuropsychology of emotion. In The Neuropsychology of Emotion. Series in Affective Science; Joan, C., Ed.; Oxford University Press: New York, NY, USA, 2000; pp. 269–297. [Google Scholar]
- Waldstein, S.R.; Kop, W.J.; Schmidt, L.A.; Haufler, A.J.; Krantz, D.S.; Fox, N.A. Frontal electrocortical and cardiovascular reactivity during happiness and anger. Biol. Psychol. 2000, 55, 3–23. [Google Scholar] [CrossRef]
- Davidson, R.J. Cerebral Asymmetry, Emotion, and Affective Style; The MIT Press: Cambridge, UK, 1995. [Google Scholar]
- Davidson, R.J. Anterior cerebral asymmetry and the nature of emotion. Brain Cogn. 1992, 20, 125–151. [Google Scholar] [CrossRef]
- Wheeler, R.E.; Davidson, R.J.; Tomarken, A.J. Frontal brain asymmetry and emotional reactivity: A biological substrate of affective style. Psychophysiology 1993, 30, 82–89. [Google Scholar]
- Davidson, R.J.; Fox, N.A. Asymmetrical brain activity discriminates between positive and negative affective stimuli in human infants. Science 1982, 218, 1235–1237. [Google Scholar]
- Davidson, R.J.; Schwartz, G.E. Patterns of cerebral lateralization during cardiac biofeedback versus the self-regulation of emotion: Sex differences. Psychophysiology 1976, 13, 62–68. [Google Scholar] [CrossRef]
- Heilman, K.M.; Bowers, D. Neuropsychological Studies of Emotional Changes Induced by Right and Left Hemispheric Lesions; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1990. [Google Scholar]
- Adolphs, R.; Damasio, H.; Tranel, D.; Damasio, A.R. Cortical systems for the recognition of emotion in facial expressions. J. Neurosci. 1996, 16, 7678–7687. [Google Scholar]
- Borod, J.C.; Cicero, B.A.; Obler, L.K.; Welkowitz, J.; Erhan, H.M.; Santschi, C.; Grunwald, I.S.; Agosti, R.M.; Whalen, J.R. Right hemisphere emotional perception: Evidence across multiple channels. Neuropsychology 1998, 12, 446–458. [Google Scholar] [CrossRef]
- Borod, J.C.; Haywood, C.S.; Koff, E. Neuropsychological aspects of facial asymmetry during emotional expression: A review of the normal adult literature. Neuropsychol. Rev. 1997, 7, 41–60. [Google Scholar] [CrossRef]
- Gainotti, G. Emotional behavior and hemispheric side of the lesion. Cortex 1972, 8, 41–55. [Google Scholar] [CrossRef]
- Ahern, G.L.; Schomer, D.L.; Kleefield, J.; Blume, H. Right hemisphere advantage for evaluating emotional facial expressions. Cortex 1991, 27, 193–202. [Google Scholar] [CrossRef]
- Narumoto, J.; Okada, T.; Sadato, N.; Fukui, K.; Yonekura, Y. Attention to emotion modulates fMRI activity in human right superior temporal sulcus. Cogn. Brain Res. 2001, 12, 225–231. [Google Scholar] [CrossRef]
- Sato, W.; Kochiyama, T.; Yoshikawa, S.; Naito, E.; Matsumura, M. Enhanced neural activity in response to dynamic facial expressions of emotion: An fMRI study. Cogn. Brain Res. 2004, 20, 81–91. [Google Scholar] [CrossRef]
- Vanderploeg, R.D.; Brown, W.S.; Marsh, J.T. Judgments of emotion in words and faces: ERP correlates. Int. J. Psychophysiol. 1987, 5, 193–205. [Google Scholar] [CrossRef]
- Everhart, D.E.; Carpenter, M.D.; Carmona, J.E.; Ethridge, A.J.; Demaree, H.A. Adult sex-related P300 differences during the perception of emotional prosody and facial affect. Psychophysilology 2003, 40, S39. [Google Scholar] [CrossRef]
- Silberman, E.K.; Weingartner, H. Hemispheric lateralization of functions related to emotion. Brain Cogn. 1986, 5, 322–353. [Google Scholar] [CrossRef]
- Harmon-Jones, E.; Allen, J.J.B. Behavioral activation sensitivity and resting frontal EEG asymmetry: Covariation of putative indicators related to risk for mood disorders. J. Abnorm. Psychol. 1997, 106, 159–163. [Google Scholar] [CrossRef]
- Sutton, S.K.; Davidson, R.J. Prefrontal brain asymmetry: A biological substrate of the behavioral approach and inhibition systems. Psychol. Sci. 1997, 8, 204–210. [Google Scholar] [CrossRef]
- Balconi, M.; Pozzoli, U. Event-related oscillations (EROs) and event-related potentials (ERPs) comparison in facial expression recognition. J. Neuropsychol. 2007, 1, 283–294. [Google Scholar] [CrossRef]
- Sato, W.; Kochiyama, T.; Yoshikawa, S.; Matsumura, M. Emotional expression boosts early visual processing of the face: ERP recording and its decomposition by independent component analysis. Neuroreport 2001, 12, 709–714. [Google Scholar] [CrossRef]
- Streit, M.; Wolwer, W.; Brinkmeyer, J.; Ihl, R.; Gaebel, W. Electrophysiological correlates of emotional and structural face processing in humans. Neurosci. Lett. 2000, 278, 13–16. [Google Scholar] [CrossRef]
- Frijda, N.H. Emotions are functional, most of the time. In The Nature of Emotion: Fundamental Questions; Ekman, P., Davidson, R.J., Eds.; Oxford University Press: New York, NY, USA, 1994; pp. 112–122. [Google Scholar]
- Frijda, N.H.; Kuipers, P.; Terschure, E. Relations among emotion, appraisal, and emotional action readiness. J. Pers. Soc. Psychol. 1989, 57, 212–228. [Google Scholar] [CrossRef]
- Frijda, N.H. The place of appraisal in emotion. Cogn. Emot. 1993, 7, 357–387. [Google Scholar] [CrossRef]
- Lazarus, R.S. Emotion and Adaptation; Oxford University Press: New York, NY, USA, 1993. [Google Scholar]
- Hewig, J.; Hagemann, D.; Seifert, J.; Naumann, E.; Bartussek, D. On the selective relation of frontal cortical asymmetry and anger-out versus anger-control. J. Pers. Soc. Psychol. 2004, 87, 926–939. [Google Scholar] [CrossRef]
- Balconi, M.; Mazza, G. Consciousness and emotion. ERP modulation and attentive vs. pre-attentive elaboration of emotional facial expressions by backward masking. Motiv. Emot. 2009, 33, 113–124. [Google Scholar]
- Balconi, M.; Mazza, G. Brain oscillations and BIS/BAS (behavioral inhibition/activation system) effects on processing masked emotional cues. ERS/ERD and coherence measures of alpha band. Int. J. Psychophysiol. 2009, 74, 158–165. [Google Scholar] [CrossRef]
- Bunce, V.; Bernat, E.; Wong, P.S.; Shevrin, H. Further evidence for unconscious learning: Preliminary support for the conditioning of facial EMG to subliminal stimuli. J. Psychiatr. Res. 1999, 33, 341–347. [Google Scholar] [CrossRef]
- Dimberg, U.; Öhman, A. Behold the wrath: Psychophysiological responses to facial stimuli. Motiv. Emot. 1996, 20, 49–182. [Google Scholar]
- Posner, M.I. Chronometric Explorations of Mind; Lawrence Erlbaum: Oxford, UK, 1978. [Google Scholar]
- Shiffrin, R.M.; Schneider, W. Controlled and automatic human information processing: II. Perceptual learning, automatic attending and a general theory. Psychol. Rev. 1997, 84, 127–190. [Google Scholar] [CrossRef]
- Wong, P.S.; Shevrin, H.; Williams, W.J. Conscious and nonconscious processes: An ERP index of an anticipatory response in a conditioning paradigm using visual masked stimuli. Psychophysiology 1994, 31, 87–101. [Google Scholar] [CrossRef]
- Dimberg, U.; Elmehed, K.; Thunberg, M. Unconscious facial reactions to emotional facial expressions. Psychol. Sci. 2000, 11, 86–89. [Google Scholar] [CrossRef]
- Wong, P.S.; Root, J.C. Dynamic variations in affective priming. Conscious. Cogn. 2003, 12, 147–168. [Google Scholar] [CrossRef]
- Stone, A.; Valentine, T. The categorical structure of knowledge for famous people (and a novel application of Centre-Surround theory). Cognition 2007, 104, 535–564. [Google Scholar] [CrossRef]
- Esteves, F.; Parra, C.; Dimberg, U.; Öhman, A. Nonconscious associative learning: Pavlovian conditioning of skin conductance responses to masked fear relevant facial stimuli. Psychophysiology 1994, 31, 375–385. [Google Scholar] [CrossRef]
- Morris, J.; Dolan, R. The amygdala and unconscious fear processing. In Out of Mind: Varieties of Unconscious Processes; de Gelder, B., de Haan, E.H.F., Heywood, C.A., Eds.; Oxford University Press: New York, NY, USA, 2001; pp. 185–204. [Google Scholar]
- Whalen, P.J.; Rauch, S.L.; Etcoff, N.L.; McInerney, S.C.; Lee, M.B.; Jenike, M.A. Masked presentations of emotional facial expressions modulate amygdala activity without explicit knowledge. J. Neurosci. 1998, 18, 411–418. [Google Scholar]
- Balconi, M.; Lucchiari, C. Morphed facial expressions elicited a N400 ERP effect. A cerebral domain-specific semantic module. Scand. J. Psychol. 2005, 46, 467–474. [Google Scholar] [CrossRef]
- Kiefer, M.; Spitzer, M. Time course of conscious and unconscious semantic brain activation. Neuroreport 2000, 11, 2401–2407. [Google Scholar] [CrossRef]
- Lang, P.J.; Bradley, M.M.; Cuthbert, B.N. Motivated attention: Affect, activation, and action. In Attention and Orienting: Sensory and Motivational Processes; Lang, P.J., Simons, R.F., Balaban, M., Eds.; Lawrence Erlbaum Associates: Mahwah, NJ, USA, 1997; pp. 97–135. [Google Scholar]
- Batty, M.; Taylor, M.J. Early processing of the six basic facial emotional expressions. Cogn. Brain Res. 2003, 17, 613–620. [Google Scholar] [CrossRef]
- Holmes, A.; Vuilleumier, P.; Eimer, M. The processing of emotional facial expression is gated by spatial attention: Evidence from event-related brain potentials. Cogn. Brain Res. 2003, 16, 174–184. [Google Scholar] [CrossRef]
- Rolls, E.T. Consciousness absent and present: A neurophysiological exploration of masking. In The First Half Second: The Microgenesis and Temporal Dynamics of Unconscious and Conscious Visual Processes; Ögmen, H., Breitmeyer, B.J., Eds.; MIT Press: Cambridge, UK, 2006; pp. 89–108. [Google Scholar]
- Cacioppo, J.T. Feelings and emotions: Roles for electrophysiological markers. Biol. Psychol. 2004, 67, 235–243. [Google Scholar] [CrossRef]
- Ekman, P.; Friesen, W.V. Pictures of Facial Affect; Consulting Psychologist Press: Palo Alto, CA, USA, 1976. [Google Scholar]
- Bernat, E.; Bunce, S.; Shevrin, H. Event-related brain potentials differentiate positive and negative mood adjectives during both supraliminal and subliminal visual processing. Int. J. Psychophysiol. 2001, 42, 11–34. [Google Scholar] [CrossRef]
- Brázdil, M.; Rektor, I.; Dufek, M.; Jurák, P.; Daniel, P. Effect of subthreshold target stimuli on event-related potentials. Electroencephalogr. Clin. Neurophysiol. 1998, 107, 64–68. [Google Scholar] [CrossRef]
- Liddell, B.J.; Williams, L.M.; Rathjen, J.; Shevrin, H.; Gordon, E. A temporal dissociation of subliminal versus supraliminal fear perception: An event-related potential study. J. Cogn. Neurosci. 2004, 16, 479–486. [Google Scholar] [CrossRef]
- Merikle, P.M.; Smilek, D.; Eastwood, J.D. Perception without awareness: Perspectives from cognitive psychology. Cognition 2001, 79, 115–134. [Google Scholar] [CrossRef]
- MacMillan, N. The psychophysics of subliminal perception. Behav. Brain Sci. 1986, 9, 38–39. [Google Scholar] [CrossRef]
- Pfurtscheller, G. Event-related synchronization (ERS): an electrophysiological correlate of cortical areas at rest. Electroencephalogr. Clin. Neurophysiol. 1992, 83, 62–69. [Google Scholar] [CrossRef]
- Kanwisher, N. Domain specificity in face perception. Nat. Neurosci. 2000, 3, 759–763. [Google Scholar] [CrossRef]
- Liu, J.; Harris, A.; Kanwisher, N. Stages of processing in face perception: An EMG study. Nat. Neurosci. 2002, 5, 910–916. [Google Scholar] [CrossRef]
- Reuter-Lorenz, P.A.; Givis, R.P.; Moscovitch, M. Hemispheric specialization and the perception of emotion: Evidence from right-handers and from inverted and non-inverted left-handers. Neuropsychologia 1983, 21, 687–692. [Google Scholar] [CrossRef]
- Junghöfer, M.; Bradley, M.M.; Elbert, T.R.; Lang, P.J. Fleeting images: A new look at early emotion discrimination. Psychophysiology 2011, 38, 175–178. [Google Scholar]
- Morita, Y.; Morita, K.; Yamamoto, M.; Waseda, Y.; Maeda, H. Effects of facial affect recognition on the auditory P300 in healthy subjects. Neurosci. Res. 2001, 41, 89–95. [Google Scholar] [CrossRef]
- Russell, J.A. Core affect and the psychological construction of emotion. Psychol. Rev. 2003, 110, 145–172. [Google Scholar] [CrossRef]
- Russell, J.A.; Bullock, M. Multidimensional scaling of emotional facial expressions: Similarity from preschoolers to adults. J. Pers. Soc. Psychol. 1985, 48, 1290–1298. [Google Scholar] [CrossRef]
- Dehaene, S.; Changeux, J.P.; Naccache, L.; Sackur, J.; Sergent, C. Conscious, preconscious, and subliminal processing: a testable taxonomy. Trends Cogn. Sci. 2006, 10, 204–211. [Google Scholar] [CrossRef]
- Öhman, A. Distinguishing unconscious from conscious emotional processes: Methodological considerations and theoretical implications. In Handbook of Cognition and Emotion; Dalgleish, T., Power, M.J., Eds.; John Wiley: Chichester, NY, USA, 1999; pp. 112–128. [Google Scholar]
- Balconi, M.; Lucchiari, C. Consciousness and arousal effects on emotional face processing as revealed by brain oscillations. A gamma band analysis. Int. J. Psychophysiol. 2008, 67, 41–46. [Google Scholar] [CrossRef]
- Lang, P.J.; Greenwald, M.K.; Bradley, M.M.; Hamm, A.O. Looking at pictures: Affective, facial, visceral, and behavioral reactions. Psychophysiology 1993, 30, 261–273. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Balconi, M.; Ferrari, C. Subliminal and Supraliminal Processing of Facial Expression of Emotions: Brain Oscillation in the Left/Right Frontal Area. Brain Sci. 2012, 2, 85-100. https://doi.org/10.3390/brainsci2020085
Balconi M, Ferrari C. Subliminal and Supraliminal Processing of Facial Expression of Emotions: Brain Oscillation in the Left/Right Frontal Area. Brain Sciences. 2012; 2(2):85-100. https://doi.org/10.3390/brainsci2020085
Chicago/Turabian StyleBalconi, Michela, and Chiara Ferrari. 2012. "Subliminal and Supraliminal Processing of Facial Expression of Emotions: Brain Oscillation in the Left/Right Frontal Area" Brain Sciences 2, no. 2: 85-100. https://doi.org/10.3390/brainsci2020085





