Behavioral and Cortical Activation Changes in Children Following Auditory Training for Dichotic Deficits
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Baseline Behavioral Tests
2.3. fMRI Scanning
2.4. ARIA Training
2.5. Post-ARIA Behavioral Measures
2.6. Behavioral Data Analysis
2.7. fMRI Analysis
3. Results
3.1. Behavioral Results Pre-ARIA
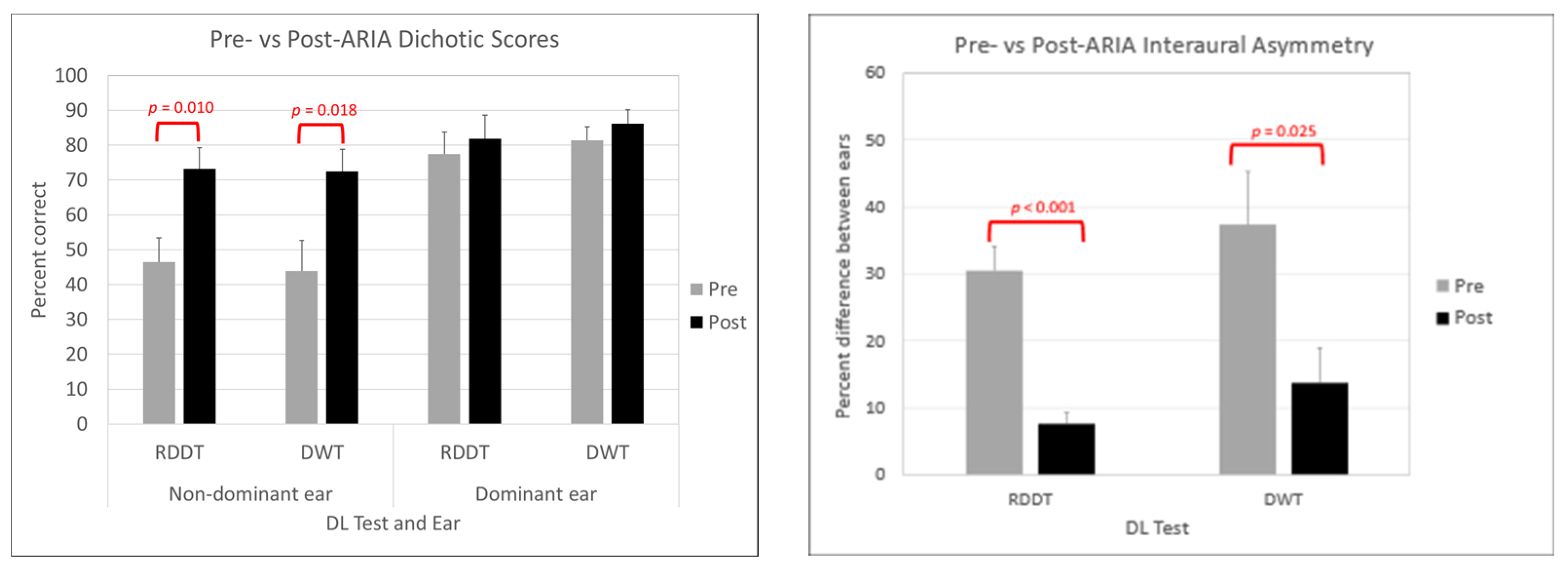
3.2. Behavioral Results Post-ARIA
3.3. fMRI Results Pre- and Post-ARIA
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- American Speech-Language-Hearing Association (ASHA) 2005. (Central) Auditory Processing Disorders [Technical Report]. Available online: www.asha.org/policy (accessed on 13 December 2023).
- American Academy of Audiology (AAA) 2010. Practice Guidelines for the Diagnosis, Treatment, and Management of Children and Adults with Central Auditory Processing Disorder (CAPD). Available online: http://www.audiology.org/publications-resources/document-library/central-auditory-processingdisorder (accessed on 13 December 2023).
- Emanuel, D.C.; Ficca, K.N.; Korczak, P. Survey of the diagnosis and management of auditory processing disorder. Am. J. Audiol. 2011, 20, 48–60. [Google Scholar] [CrossRef]
- Ismen, K.; Emanuel, D.C. Auditory processing disorder: Protocols and controversy. Am. J. Audiol. 2023, 32, 614–639. [Google Scholar] [CrossRef]
- Kimura, D. Some effects of temporal-lobe damage on auditory perception. Can. J. Psychol. 1961, 15, 156–165. [Google Scholar] [CrossRef]
- Musiek, F.E. Assessment of central auditory dysfunction: The dichotic digit test revisited. Ear Hear. 1983, 4, 79–83. [Google Scholar] [CrossRef]
- Damasio, H.; Damasio, A. “Paradoxic” ear extinction in dichotic listening. Possible anatomic significance. Neurology 1979, 29, 644–653. [Google Scholar] [CrossRef]
- Moncrieff, D.; Keith, W.; Abramson, M.; Swann, A. Clinical evidence on the diagnosis of amblyaudia, a binaural integration type of auditory processing disorder. Int. J. Audiol. 2016, 55, 333–345. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, A.B.; Kozin, E.D.; Remenschneider, A.; Eftekhari, K.; Jung, D.H.; Polley, D.B.; Lee, D.J. Amblyaudia: Review of pathophysiology, clinical presentation, and treatment of a new diagnosis. Otolaryngol. Head Neck Surg. 2016, 154, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Moncrieff, D. Amblyaudia: Evidence of indistinct processing of binaural information in children. In Proceedings of the American Auditory Society Annual Meeting, Phoenix, AZ, USA, 1–3 March 2010. [Google Scholar]
- Kimura, D. Functional asymmetry of the brain in dichotic listening. Cortex 1967, 3, 163–168. [Google Scholar] [CrossRef]
- Pujol, J.; Vendrell, P.; Junqué, C.; Marti-Vilalta, J.L.; Capdevila, A. When does human brain development end? Evidence of corpus callosum growth up to adulthood. Ann. Neurol. 1993, 34, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Von Plessen, K.; Lundervold, A.; Duta, N.; Heiervang, E.; Klauschen, F.; Smievoll, A.I.; Ersland, L.; Hugdahl, K. Less developed corpus callosum in dyslexic subjects-a structural MRI study. Neuropsychologia 2002, 40, 1035–1044. [Google Scholar] [CrossRef] [PubMed]
- Choo, A.L.; Kraft, S.J.; Olivero, W.; Ambrose, N.G.; Sharma, H.; Chang, S.-E.; Loucks, T.M. Corpus callosum differences associated with persistent stuttering in adults. J. Commun. Disord. 2011, 44, 470–477. [Google Scholar] [CrossRef]
- Kronfeld-Duenias, V.; Civier, O.; Ezrati-Vinacour, O.A.R.; Ben-Shachar, M. White matter pathways in persistent developmental stuttering: Lessons from tractography. J. Fluency Dis. 2018, 55, 68–83. [Google Scholar] [CrossRef]
- Luders, E.; Kurth, F.; Pigdon, L.; Conti-Ramsden, G.; Reilly, S.; Morgan, A.T. Atypical callosal morphology in children with speech sound disorder. Neuroscience 2017, 367, 211–218. [Google Scholar] [CrossRef]
- Farah, M.J.; Shera, D.M.; Savage, J.H.; Betancourt, L.; Giannetta, J.M.; Brodsky, N.L.; Malmud, E.K.; Hurt, H. The corpus callosum in childhood poverty: Specific associations with neurocognitive development. Brain Res. 2006, 1110, 166–174. [Google Scholar] [CrossRef]
- Bryden, M.P.; Munhall, K.; Allard, F. Attentional biases and the right-ear effect in dichotic listening. Brain Lang. 1983, 18, 236–248. [Google Scholar] [CrossRef]
- Kinsbourne, M. The cerebral basis of lateral asymmetries in attention. Acta Psychol. 1970, 33, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Hiscock, M.; Kinsbourne, M. Attention and the right-ear advantage: What is the connection? Brain Cogn. 2011, 76, 263–275. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Chang, W.-T.; Belliveau, J.W.; Hämäläinen, M.; Ahveninen, J. Lateralized parietotemporal oscillatory phase synchronization during auditory selective attention. Neuroimage 2014, 86, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Payne, L.; Rogers, C.S.; Wingfield, A.; Sekuler, R. A right-ear bias of auditory selective attention is evident in alpha oscillations. Psychophysiology 2017, 54, 528–535. [Google Scholar] [CrossRef] [PubMed]
- Pugh, K.R.; Shaywitz, B.A.; Shaywitz, S.E.; Fulbright, R.K.; Byrd, D.; Skudlarski, P.; Shankweiler, D.P.; Katz, L.; Constable, R.T.; Fletcher, J.; et al. Auditory selective attention: An fMRI investigation. Neuroimage 1996, 4, 159–173. [Google Scholar] [CrossRef] [PubMed]
- Asbjørnsen, A.E.; Hugdahl, K. Attentional effects in dichotic listening. Brain Lang. 1995, 49, 189–201. [Google Scholar] [CrossRef]
- Kershner, J.R.; Morton, L.L. Directed attention dichotic listening in reading disabled children: A test of four models of maladaptive lateralization. Neuropsychologia 1990, 28, 181–198. [Google Scholar] [CrossRef]
- Obrzut, J.E.; Boliek, C.A.; Obrzut, A. The effect of stimulus type and directed attention on dichotic listening with children. J. Exp. Child. Psychol. 1986, 41, 198–209. [Google Scholar] [CrossRef]
- Blauert, J. Spatial Hearing: The Psychophysics of Human Sound Localization; MIT Press: Cambridge, MA, USA, 1997. [Google Scholar]
- Middlebrooks, J.C.; Green, D.M. Sound localization by human listeners. Ann. Rev. Psychol. 1991, 42, 135–159. [Google Scholar] [CrossRef]
- Litovsky, R.Y. Development of binaural and spatial hearing. In Springer Handbook of Auditory Research; Werner, L.A., Popper, A., Fay, R., Eds.; Springer: New York, NY, USA, 2011. [Google Scholar]
- Moncrieff, D.W. Dichotic listening in children: Age-related changes in direction and magnitude of ear advantage. Brain Cogn. 2011, 76, 316–322. [Google Scholar] [CrossRef]
- Moushegian, G.; Rupert, A.; Whitcomb, M.A. Brain-stem neuronal response patterns to monaural and binaural tones. J. Neurophysiol. 1964, 27, 1174–1191. [Google Scholar] [CrossRef]
- Westerhausen, R.; Luders, E.; Specht, K.; Ofte, S.H.; Toga, A.W.; Thompson, P.M.; Helland, T.; Hugdahl, K. Structural and functional reorganization of the corpus callosum between the age of 6 and 8 years. Cereb. Cortex 2011, 21, 1012–1017. [Google Scholar] [CrossRef] [PubMed]
- Polley, D.B.; Thompson, J.H.; Guo, W. Brief hearing loss disrupts binaural integration during two early critical periods of auditory cortex development. Nat. Commun. 2013, 4, 2547. [Google Scholar] [CrossRef] [PubMed]
- Popescu, M.V.; Polley, D.B. Monaural deprivation disrupts development of binaural selectivity in auditory midbrain and cortex. Neuron 2010, 65, 718–731. [Google Scholar] [CrossRef] [PubMed]
- Moncrieff, D.; Mendez, K.R.G. Prevalence and severity of dichotic deficits in adjudicated adolescents: Implications for language skills, educational success and competency within the juvenile justice system. Eur. Soc. Med. 2024; submitted. [Google Scholar]
- Moncrieff, D. Identification and treatment of dichotic listening deficits in children. In Auditory Processing Disorders: Assessment, Management, and Treatment, 4th ed.; Geffner, D., Ross-Swain, D., Eds.; Plural Publishing, Inc.: San Diego, CA, USA, 2024. [Google Scholar]
- Bamiou, D.-E.; Musiek, F.E.; Luxon, L.M. Aetiology and clinical presentations of auditory processing disorders-a review. Arch. Dis. Child. 2001, 85, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.C. Neuroplasticity as a proposed mechanism for the efficacy of optometric vision therapy & rehabilitation. J. Behav. Optometry 2009, 20, 95–99. [Google Scholar]
- Hakkennes, S.; Keating, J. Constraint-induced movement therapy following stroke: A systematic review of randomized controlled trials. Aust. J. Physiother. 2005, 51, 221–231. [Google Scholar] [CrossRef]
- Hsin, Y.; Chen, F.-C.; Lin, K.; Kang, L.; Chen, C.; Chen, C. Efficacy of constraint-induced therapy on functional performance and health-related quality of life for children with cerebral palsy: A randomized controlled trial. J. Child. Neurol. 2012, 27, 992–999. [Google Scholar] [CrossRef]
- Moncrieff, D.W.; Wertz, D. Auditory rehabilitation for interaural asymmetry: Preliminary evidence of improved dichotic listening performance following intensive training. Int. J. Audiol. 2008, 47, 84–97. [Google Scholar] [CrossRef]
- Moncrieff, D.; Keith, W.; Abramson, M.; Swann, A. Evidence of binaural integration benefits following ARIA training in children and adolescents diagnosed with amblyaudia. Int. J. Audiol. 2017, 56, 580–588. [Google Scholar] [CrossRef]
- Hoshino, O. Neuronal bases of perceptual learning revealed by a synaptic balance scheme. Neural Comput. 2004, 16, 563–594. [Google Scholar] [CrossRef] [PubMed]
- Wamsley, E.J. Memory Consolidation during Waking Rest. Trends Cogn. Sci. 2019, 23, 171–173. [Google Scholar] [CrossRef] [PubMed]
- Davis, M.H.; Johnsrude, I.S. Hierarchical processing in spoken language comprehension. J. Neurosci. 2003, 23, 3423–3431. [Google Scholar] [CrossRef]
- Hickok, G.; Poeppel, D. The cortical organization of speech processing. Nat. Rev. Neurosci. 2007, 8, 393–402. [Google Scholar] [CrossRef]
- Obleser, J.; Zimmermann, J.; Van Meter, J.; Rauschecker, J.P. Multiple stages of auditory speech perception reflected in event-related FMRI. Cereb. Cortex 2007, 17, 2251–2257. [Google Scholar] [CrossRef] [PubMed]
- Price, C.J. A review and synthesis of the first 20 years of PET and fMRI studies of heard speech, spoken language, and reading. Neuroimage 2012, 62, 816–847. [Google Scholar] [CrossRef]
- Scott, S.K.; Blank, C.C.; Rosen, S.; Wise, R.J. Identification of a pathway for intelligible speech in the left temporal lobe. Brain 2000, 123, 2400–2406. [Google Scholar] [CrossRef]
- Hugdahl, K.; Brøonnick, K.; Kyllingsbæk, S.; Law, I.; Gade, A.; Paulson, O.B. Brain activation during dichotic presentations of consonant-vowel and musical instrument stimuli: A 15O-PET study. Neuropsychologia 1999, 37, 431–440. [Google Scholar] [CrossRef]
- Stefanatos, G.A.; Joe, W.Q.; Aguirre, G.K.; Detre, J.A.; Wetmore, G. Activation of human auditory cortex during speech perception: Effects of monaural, binaural, and dichotic presentation. Neuropsychologia 2008, 46, 301–315. [Google Scholar] [CrossRef]
- Van den Noort, M.; Specht, K.; Rimol, L.M.; Ersland, L.; Hugdahl, K. A new verbal reports fMRI dichotic listening paradigm for studies of hemispheric asymmetry. Neuroimage 2008, 40, 902–911. [Google Scholar] [CrossRef] [PubMed]
- Westerhausen, R.; Kompus, K.; Hugdahl, K. Mapping hemispheric asymmetries, relative asymmetries, and absolute asymmetries underlying the auditory laterality effect. Neuroimage 2014, 84, 962–970. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.A.; Zatorre, R.J. Neural substrates for dividing and focusing attention between simultaneous auditory and visual events. Neuroimage 2006, 31, 1673–1681. [Google Scholar] [CrossRef] [PubMed]
- Pollmann, S. A unified structural-attentional framework for dichotic listening. In The Two Halves of the Brain: Information Processing in the Cerebral Hemispheres; Hugdahl, K., Westerhausen, R., Eds.; MIT Press: Cambridge, MA, USA, 2010. [Google Scholar]
- Bellis, T.J. Assessment and Management of Central Auditory Processing Disorders in the Educational Setting: From Science to Practice; Thompson Learning: Delmar, NY, USA, 2003. [Google Scholar]
- Bellis, T.J. Interpretation of APD test results. In An Introduction to Auditory Processing Disorders in Children; Parthasarathy, T.K., Ed.; Erlbaum: Mahwah, NJ, USA, 2006; pp. 145–160. [Google Scholar]
- Moncrieff, D.W.; Wilson, R.H. Recognition of one-, two-, and three-pair dichotic digits by children and young adults. J. Am. Acad. Audiol. 2009, 20, 58–70. [Google Scholar]
- Moncrieff, D. Age- and gender-specific normative information from children assessed with a dichotic words test. J. Am. Acad. Audiol. 2015, 26, 632–644. [Google Scholar] [CrossRef]
- Keith, R. SCAN-C: Test for Auditory Processing Disorders in Children-Revised; The Psychological Corporation: San Antonio, TX, USA, 2000. [Google Scholar]
- White, S.; Moncrieff, D. ARIA treatment benefits are related to severity of dichotic listening deficits in children. Int. J. Pediatr. Otorhinolaryngol. 2023, 168, 111551. [Google Scholar] [CrossRef]
- Wilson, R.H.; Farmer, N.M.; Gandhi AShelburne, E.; Weaver, J. Normative data for the words-in-noise test for 6- to 12-year-old children. J. Speech Lang. Hear. Res. 2010, 53, 1111–1121. [Google Scholar] [CrossRef] [PubMed]
- Noffsinger, D.; Wilson, R.H.; Musiek, F.E. Department of Veterans Affairs compact disc recording for auditory perceptual assessment: Background and introduction. J. Am. Acad. Audiol. 1994, 5, 231–235. [Google Scholar] [PubMed]
- Myklebust, H.R. Auditory Disorders in Children; Grune & Stratton: New York, NY, USA, 1954. [Google Scholar]
- Celesia, G.G. Hearing disorders in brainstem lesions. In Handbook of Clinical Neurology, Volume 129: The Human Auditory System: Fundamental Organization and Clinical Disorders; Celesia, G.G., Hickok, G., Eds.; Elsevier: Edinburgh, UK, 2015; p. 531. [Google Scholar]
- Chermak, G.D.; Musiek, F.E. Managing central auditory processing disorders in youth. Am. J. Audiol. 1992, 1, 61–65. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chermak, G.D.; Musiek, F.E. Auditory training: Principles and approaches for managing auditory processing disorders. Semin. Hear. 2002, 23, 297–308. [Google Scholar] [CrossRef]
- Ahmmed, A.U.; Ahmmed, A.A.; Bath, J.R.; Ferguson, M.A.; Plack, C.J.; Moore, D.R. Assessment of children with suspected auditory processing disorder: A factor analysis study. Ear Hear. 2014, 35, 295–305. [Google Scholar] [CrossRef]
- Vermiglio, A.J. On the clinical entity in audiology: (Central) auditory processing and speech recognition in noise disorders. J. Am. Acad. Audiol. 2014, 25, 904–917. [Google Scholar] [CrossRef]
- Loo, J.H.Y.; Bamiou, D.-E.; Campbell, N.; Luxon, L.M. Computer-based auditory training (CBAT): Benefits for children with language- and reading-related learning difficulties. Dev. Med. Child. Neurol. 2010, 52, 708–717. [Google Scholar] [CrossRef]
- Delphi, M.; Abdollahi, F.Z. Dichotic training in children with auditory processing disorder. Int. J. Pediatr. Otorhinolaryngol. 2018, 110, 114–117. [Google Scholar] [CrossRef]
- Helland, T.; Morken, F.; Bless, J.J.; Valderhaug, H.V.; Eiken, M.; Helland, W.A.; Torkildsen, J.V.K. Auditive training effects from a dichotic listening app in children with dyslexia. Dyslexia 2018, 24, 336–356. [Google Scholar] [CrossRef]
- Denman, I.; Banajee, M.; Hurley, A. Dichotic listening training in children with autism spectrum disorder: A single subject design. Int. J. Audiol. 2015, 54, 991–996. [Google Scholar] [CrossRef]
- Kozou, H.; Azouz, H.G.; Abdou, R.M.; Shaltout, A. Evaluation and remediation of central auditory processing disorders in children with autism spectrum disorders. Int. J. Pediatr. Otorhinolaryngol. 2018, 104, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Hawkey, D.J.C.; Amitay, S.; Moore, D. Early and rapid perceptual learning. Nat. Neurosci. 2004, 7, 1055–1056. [Google Scholar] [CrossRef] [PubMed]
- Green, C.S.; Banai, K.; Lu, Z.L.; Bevalier, D. Perceptual learning. In Stevens’ Handbook of Experimental Psychology and Cognitive Neuroscience, Sensation, Perception, and Attention, 4th ed.; Wixted, J.T., Serences, J., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2018; Volume 2, pp. 755–802. [Google Scholar]
- Ortiz, J.A.; Wright, B.A. Differential rates of consolidation of conceptual and stimulus learning following training on an auditory skill. Exp. Brain Res. 2010, 201, 441–451. [Google Scholar] [CrossRef] [PubMed]
- Tzounopoulos, T.; Kraus, N. Learning to encode timing: Mechanisms of plasticity in the auditory brainstem. Neuron 2016, 62, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Ahissar, M.; Nahum, M.; Nelken, I.; Hochstein, S. Reverse hierarchies and sensory learning. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 285–299. [Google Scholar] [CrossRef] [PubMed]
- Pisoni, D. Long-term memory in speech perception: Some new findings on talker variability, speaking rate and perceptual learning. Speech Commun. 1993, 13, 109–125. [Google Scholar] [CrossRef]
- Alain, C.; Snyder, H.J.S.; He, Y.; Reinke, K.S. Changes in auditory cortex parallel rapid perceptual learning. Cereb. Cortex 2007, 17, 1074–1084. [Google Scholar] [CrossRef]
- Carcagno, S.; Plack, C.J. Short-term learning and memory: Training and perceptual learning. In Springer Handbook of Auditory Research Volume 61: The Frequency-Following Response: A Window into Human Communication; Kraus, N., Anderson, S., White-Schwoch, T., Fay, R.R., Popper, A.N., Eds.; ASA Press, Springer: Cham, Switzerland, 2017. [Google Scholar]
- Binder, J.R.; Rao, S.M.; Hammeke, T.A.; Yetkin, F.Z.; Jesmanowicz, A.; Bandettini, P.A.; Wong, E.C.; Estkowski, L.D.; Goldstein, M.D. Functional magnetic resonance imaging of human auditory cortex. Ann. Neurol. 1994, 35, 662–672. [Google Scholar] [CrossRef]
- Jäncke, L.; Wüstenberg, H.; Scheich, H.; Heinze, H.-J. Phonetic perception and the temporal cortex. Neuroimage 2002, 15, 733–746. [Google Scholar] [CrossRef]
- Broyd, S.J.; Demanuele, C.; Debener, S.; Helps, S.K.; James, C.J.; Sonuga-Barke, E.J.S. Default-mode brain dysfunction in mental disorders: A systematic review. Neurosci. Behav. Rev. 2009, 33, 279–296. [Google Scholar] [CrossRef]
- Lawrence, N.S.; Ross, T.J.; Hoffmann, R.; Garavan, H.; Stein, E.A. Multiple neuronal networks mediate sustained attention. J. Cogn. Neurosci. 2003, 15, 1028–1038. [Google Scholar] [CrossRef] [PubMed]
- Pastor, M.A.; Macaluso, E.; Day, B.L.; Frackowiak, R. The neural basis of temporal auditory discrimination. Neuroimage 2006, 30, 512–520. [Google Scholar] [CrossRef] [PubMed]
- Joris, P.X.; van der Heijden, M. Early binaural hearing: The comparison of temporal differences at the two ears. Ann. Rev. Neurosci. 2019, 42, 433–457. [Google Scholar] [CrossRef] [PubMed]
- Jäncke, L.; Buchanan, T.W.; Lutz, K.; Shah, N.J. Focused and non-focused attention in verbal and emotional dichotic listening: An FMRI study. Brain Lang. 2001, 78, 349–363. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, T.; Rimol, L.M.; Ersland, L.; Hugdahl, K. Dichotic listening reveals functional specificity n prefrontal cortex: An fMRI study. Neuroimage 2003, 21, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Jäncke, L.; Specht, K.; Shah, J.N.; Hugdahl, K. Focused attention in a simple dichotic listening task: An fMRI experiment. Cogn. Bran Res. 2003, 16, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Hugdahl, K.; Westerhausen, R.; Alho, K.; Medvedev, S.; Laine, M.; Hämäläinen, H. Attention and cognitive control: Unfolding the dichotic listening story. Scand. J. Psychol. 2009, 50, 11–22. [Google Scholar] [CrossRef]
- Kompus, K.; Specht, K.; Ersland, L.; Juvodden, H.T.; van Wageningen, H.; Hugdahl, K.; Westernausen, R. A forced-attention dichotic listening fMRI study on 113 subjects. Brain Lang. 2012, 121, 240–247. [Google Scholar] [CrossRef]
- Westerhausen, R.; Hugdahl, K. Cognitive control of laterality. In The Two Halves of the Brain; Hugdahl, K., Westerhausen, Eds.; MIT Press: Cambridge, MA, USA, 2010. [Google Scholar]
- Falkenberg, L.E.; Specht, K.; Westerhausen, R. Attention and cognitive control networks assessed in a dichotic listening fMRI study. Brain Cogn. 2011, 76, 276–285. [Google Scholar] [CrossRef]
- Piai, V.; Roelofs, A.; Acheson, D.J.; Takashima, A. Attention for speaking: Domain-general control from the anterior cingulate cortex in spoken word production. Front. Hum. Neurosci. 2013, 7, 832. [Google Scholar] [CrossRef]
- Emch, M.; von Bastian, C.C.; Koch, K. Neural correlates of verbal working memory: An fMRI meta-analysis. Front. Hum. Neurosci. 2019, 13, 180. [Google Scholar] [CrossRef]
- Naeser, M.A.; Martin, P.I.; Theoret, H.; Kobayashi, M.; Fregni, F.; Nicholas, M.; Tormos, J.M.; Steven, M.S.; Baker, E.H.; Pascual-Leone, A. TMS suppression of right pars triangularis, but not pars opercularis, improves naming in aphasia. Brain Lang. 2011, 119, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Gold, B.T.; Buckner, R.L. Common prefrontal regions coactivate with dissociable posterior regions during controlled semantic and phonological tasks. Neuron 2002, 35, 803–812. [Google Scholar] [CrossRef] [PubMed]
- Iacoboni, M. The role of premotor cortex in speech perception: Evidence from fMRI and rTMS. J. Physiol. 2008, 102, 31–34. [Google Scholar] [CrossRef] [PubMed]
- Maess, B.; Herrmann, C.S.; Hahne, A.; Nakamura, A.; Friederici, A.D. Localizing the distributed language network responsible for the N400 measured by MEG during auditory sentence processing. Brain Res. 2006, 1096, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Moncrieff, D.; Jerger, J.; Wambacq, I.; Greenwald, R.; Black, J. ERP evidence of a dichotic left-ear deficit in some dyslexic children. J. Am. Acad. Audiol. 2004, 15, 518–534. [Google Scholar] [CrossRef] [PubMed]
- Plakke, B.; Romanski, L.M. Auditory connections and functions of prefrontal cortex. Front. Neurosci. 2014, 8, 199. [Google Scholar] [CrossRef]
- Knight, R.T.; Staines, W.R.; Swick, D.; Chao, L.L. Prefrontal cortex regulates inhibition and excitation in distributed neural networks. Acta Psychol. 1999, 101, 159–178. [Google Scholar] [CrossRef]
- Scholz, J.; Klein, M.C.; Behrens TE, J.; Johansen-Berg, H. Training induces changes in white-matter architecture. Nat. Neurosci. 2009, 12, 1370–1371. [Google Scholar] [CrossRef]




| Pre-ARIA | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| RDDT | DWT | |||||||||||||
| Code | Age | non | dom | IA | non | dom | IA | WIN | FPT | ID | %ile | |||
| 2112 | 7 | 25 | 67 | 42 | 12 | 80 | 68 | 10 | 50 | 40 | 9.6 | 7 | AMB | 5th |
| 2114 | 9 | 61 | 97 | 36 | 88 | 92 | 4 | 73 | 93 | 20 | 8 | 70 | AMB | 10th |
| 2120 | 10 | 69 | 97 | 28 | 16 | 76 | 60 | 30 | 73 | 43 | 5.6 | 20 | AMB | 5th |
| 2126 | 7 | 27 | 67 | 40 | 28 | 68 | 40 | 30 | 70 | 40 | 9.6 | 0 | AMB | 5th |
| 2128 | 9 | 42 | 86 | 44 | 28 | 92 | 64 | 17 | 67 | 50 | 10.4 | 23 | AMB | 5th |
| 2179 | 8 | 50 | 83 | 33 | 64 | 72 | 8 | 47 | 63 | 16 | 12 | 40 | DD | 10th |
| 2181 | 13 | 86 | 97 | 14 | 72 | 96 | 24 | 4 | 87 | AMB | 10th | |||
| 2183 | 7 | 25 | 47 | 22 | 40 | 64 | 24 | 53 | 77 | 24 | 7.2 | 0 | DD | 5th |
| 2184 | 9 | 36 | 56 | 20 | 48 | 92 | 44 | 7.2 | 27 | MIX | 5th | |||
| Post-ARIA | ||||||||||||||
| RDDT | DWT | |||||||||||||
| non | dom | IA | non | dom | IA | WIN | FPT | |||||||
| 2112 | 7 | 72 | 83 | 11 | 72 | 84 | 12 | 4.4 | 0 | |||||
| 2114 | 10 | 56 | 61 | 5 | 60 | 60 | 0 | 5.2 | 27 | DD | 5th | |||
| 2120 | 10 | 89 | 100 | 11 | 88 | 92 | 4 | 6.0 | 67 | |||||
| 2126 | 7 | 81 | 97 | 16 | 76 | 100 | 24 | 4.4 | 40 | |||||
| 2128 | 9 | 58 | 69 | 11 | 64 | 88 | 24 | 5.6 | 13 | UND | 10th | |||
| 2179 | 8 | 83 | 100 | 7 | 96 | 100 | 4 | 2.4 | 73 | |||||
| 2181 | 14 | 100 | 100 | 0 | 84 | 88 | 4 | 5.8 | 100 | |||||
| 2183 | 7 | 42 | 44 | 2 | 32 | 80 | 48 | 4.4 | 20 | MIX | 5th | |||
| 2184 | 10 | 78 | 83 | 5 | 80 | 84 | 4 | 6.8 | 0 | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moncrieff, D.; Schmithorst, V. Behavioral and Cortical Activation Changes in Children Following Auditory Training for Dichotic Deficits. Brain Sci. 2024, 14, 183. https://doi.org/10.3390/brainsci14020183
Moncrieff D, Schmithorst V. Behavioral and Cortical Activation Changes in Children Following Auditory Training for Dichotic Deficits. Brain Sciences. 2024; 14(2):183. https://doi.org/10.3390/brainsci14020183
Chicago/Turabian StyleMoncrieff, Deborah, and Vanessa Schmithorst. 2024. "Behavioral and Cortical Activation Changes in Children Following Auditory Training for Dichotic Deficits" Brain Sciences 14, no. 2: 183. https://doi.org/10.3390/brainsci14020183
APA StyleMoncrieff, D., & Schmithorst, V. (2024). Behavioral and Cortical Activation Changes in Children Following Auditory Training for Dichotic Deficits. Brain Sciences, 14(2), 183. https://doi.org/10.3390/brainsci14020183





