Feasibility of an Intelligent Algorithm Based on an Assist-as-Needed Controller for a Robot-Aided Gait Trainer (Lokomat) in Neurological Disorders: A Longitudinal Pilot Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Trial Design
2.2. Participants
2.3. Experimental Apparatus and Control
2.4. Protocol
2.5. Data Analysis
2.5.1. Feasibility
2.5.2. AGS as a Training Tool
2.5.3. AGS as an Assessment Tool
2.6. Statistical Analysis
2.6.1. AGS as a Training Tool
2.6.2. AGS as an Assessment Tool—Validity
2.6.3. Longitudinal Within-Patient Data
2.6.4. Relationship between L-FORCE and AGS
2.6.5. Reliability
3. Results
3.1. Participants
3.2. Primary Aim—Feasibility
3.3. Secondary Aims
3.3.1. AGS as a Training Tool
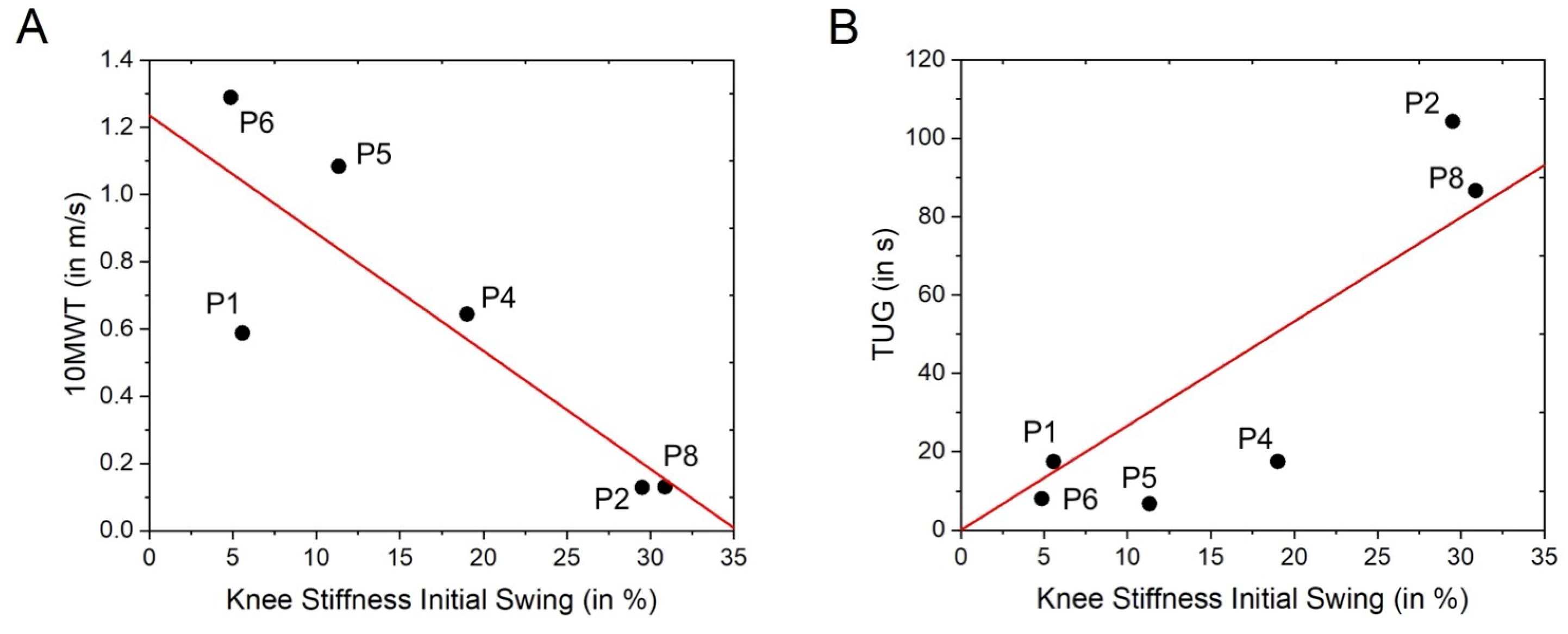
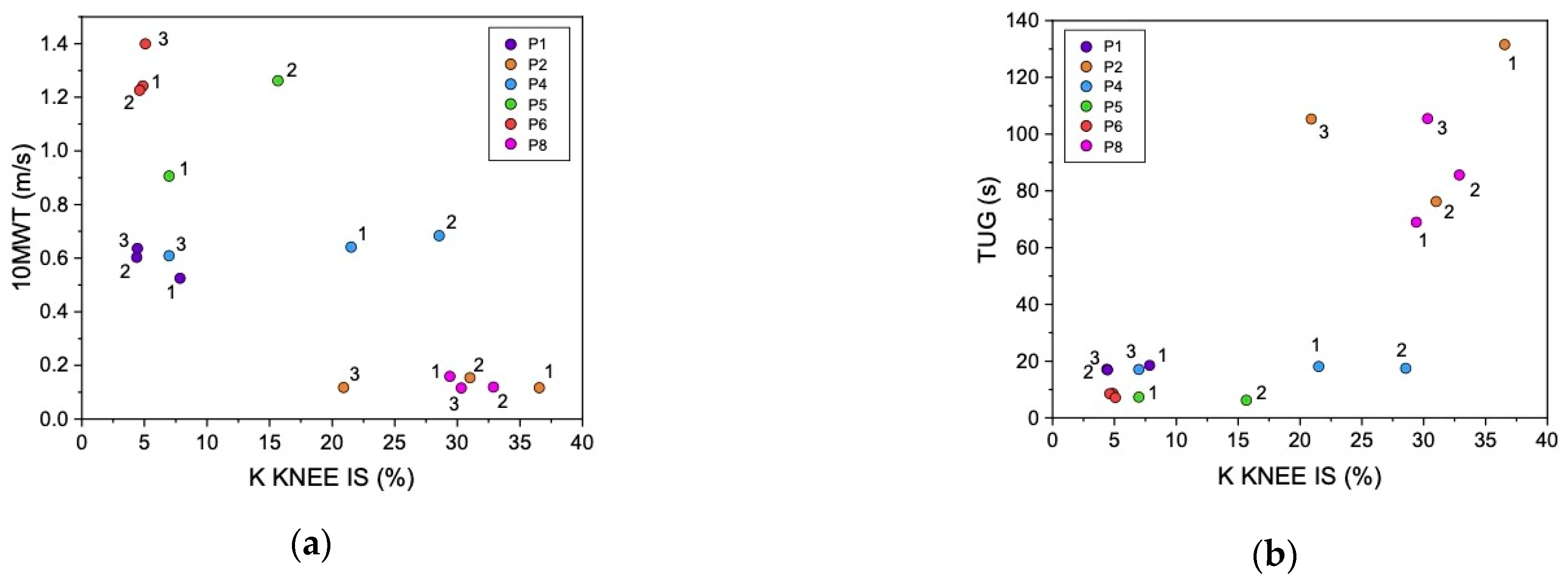
3.3.2. AGS as an Assessment Tool: Validity
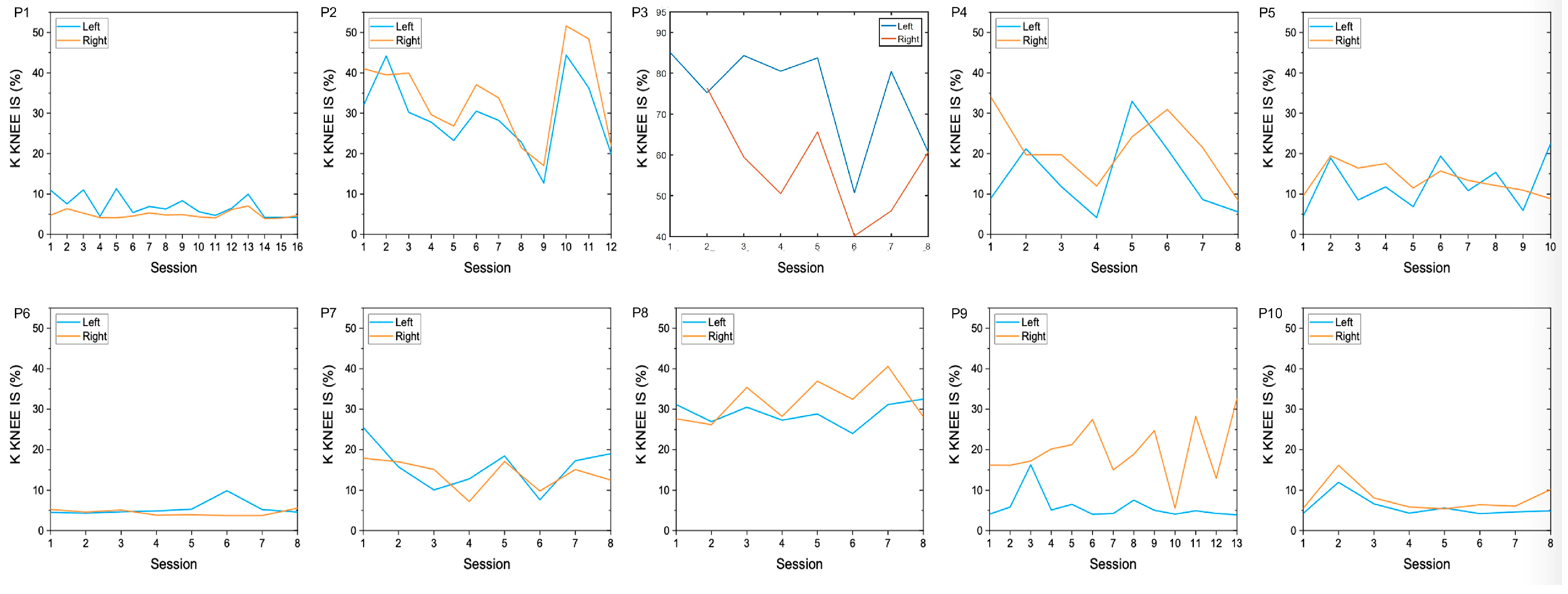
3.3.3. Longitudinal Within-Patient Data
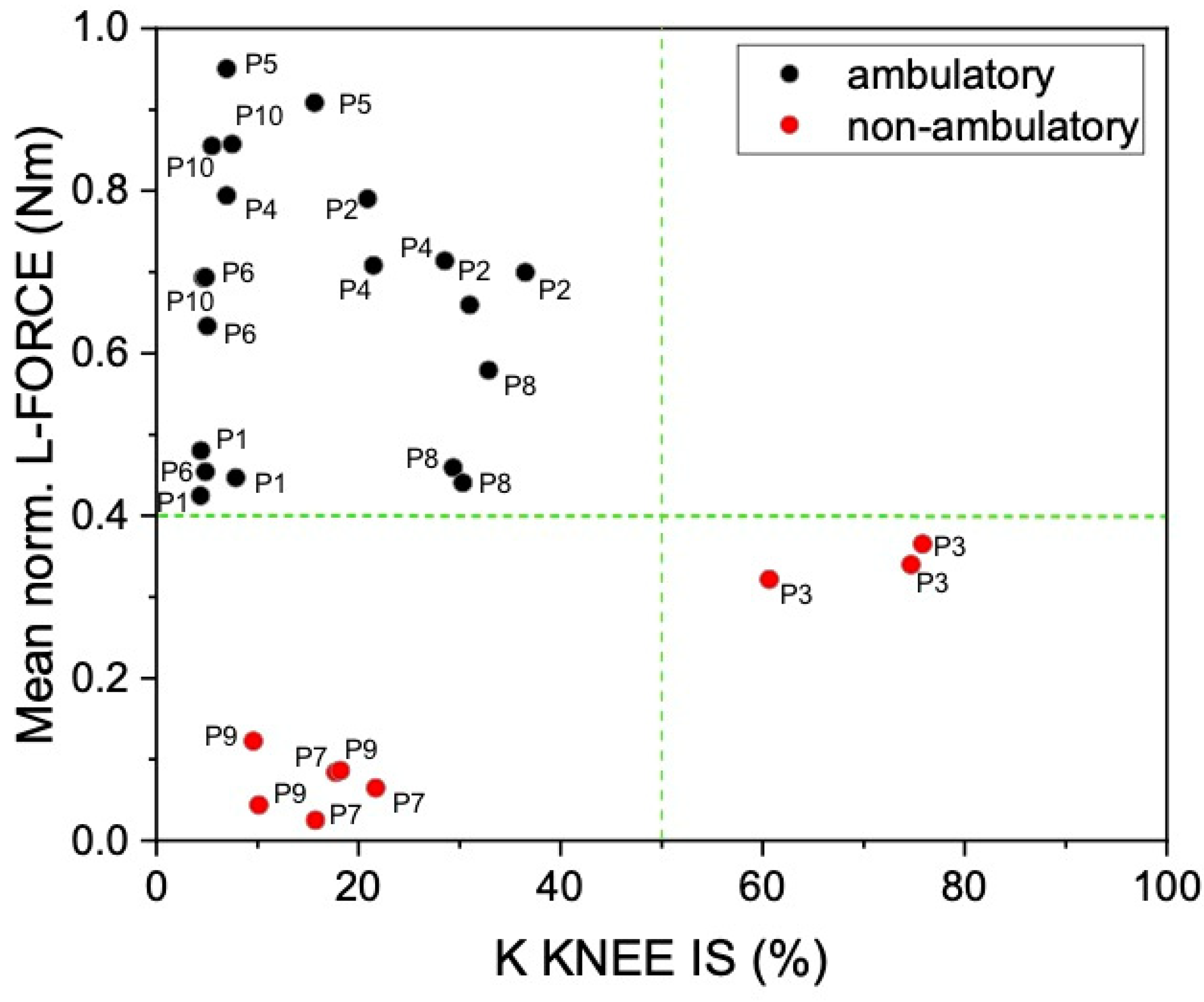
3.3.4. Relationship between L-FORCE and AGS
3.3.5. Reliability
4. Discussion
4.1. Feasibility of AGS
4.2. Adaptive Gait Support as a Training Tool
4.3. Adaptive Gait Support as an Assessment Tool
4.4. Future Directions
4.5. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ditunno, P.L.; Patrick, M.; Stineman, M.; Ditunno, J.F. Who wants to walk? Preferences for recovery after SCI: A longitudinal and cross-sectional study. Spinal Cord 2008, 46, 500–506. [Google Scholar] [CrossRef] [PubMed]
- Mudge, S.; Stott, N.S. Outcome measures to assess walking ability following stroke: A systematic review of the literature. Physiotherapy 2007, 93, 189–200. [Google Scholar] [CrossRef]
- Corbetta, D.; Imeri, F.; Gatti, R. Rehabilitation that incorporates virtual reality is more effective than standard rehabilitation for improving walking speed, balance and mobility after stroke: A systematic review. J. Physiother. 2015, 61, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Poli, P.; Morone, G.; Rosati, G.; Masiero, S. Robotic Technologies and Rehabilitation: New Tools for Stroke Patients’ Therapy. BioMed Res. Int. 2013, 2013, 153872. [Google Scholar] [CrossRef] [PubMed]
- Hobbs, B.; Artemiadis, P. A Review of Robot-Assisted Lower-Limb Stroke Therapy: Unexplored Paths and Future Directions in Gait Rehabilitation. Front. Neurorobot. 2020, 14, 19. [Google Scholar] [CrossRef]
- Lo, K.; Stephenson, M.; Lockwood, C. Effectiveness of robotic assisted rehabilitation for mobility and functional ability in adult stroke patients: A systematic review. JBI Database System. Rev. Implement. Rep. 2017, 15, 3049–3091. [Google Scholar] [CrossRef]
- Clark, W.E.; Sivan, M.; O’Connor, R.J. Evaluating the use of robotic and virtual reality rehabilitation technologies to improve function in stroke survivors: A narrative review. J. Rehabil. Assist. Technol. Eng. 2019, 6, 2055668319863557. [Google Scholar] [CrossRef]
- Riener, R.; Lunenburger, L.; Jezernik, S.; Anderschitz, M.; Colombo, G.; Dietz, V. Patient-Cooperative Strategies for Robot-Aided Treadmill Training: First Experimental Results. IEEE Trans. Neural Syst. Rehabil. Eng. 2005, 13, 380–394. [Google Scholar] [CrossRef]
- Duschau-Wicke, A.; von Zitzewitz, J.; Caprez, A.; Lunenburger, L.; Riener, R. Path control: A method for patient-cooperative robot-aided gait rehabilitation. IEEE Trans. Neural Syst. Rehabil. Eng. 2010, 18, 38–48. [Google Scholar] [CrossRef]
- Marchal-Crespo, L.; Reinkensmeyer, D.J. Review of control strategies for robotic movement training after neurologic injury. J. Neuroeng. Rehabil. 2009, 6, 20. [Google Scholar] [CrossRef]
- Campagnini, S.; Liuzzi, P.; Mannini, A.; Riener, R.; Carrozza, M.C. Effects of control strategies on gait in robot-assisted post-stroke lower limb rehabilitation: A systematic review. J. Neuroeng. Rehabil. 2022, 19, 52. [Google Scholar] [CrossRef]
- Blank, A.A.; French, J.A.; Pehlivan, A.U.; O’Malley, M.K. Current Trends in Robot-Assisted Upper-Limb Stroke Rehabilitation: Promoting Patient Engagement in Therapy. Curr. Phys. Med. Rehabil. Rep. 2014, 2, 184–195. [Google Scholar] [CrossRef]
- de Miguel-Fernández, J.; Lobo-Prat, J.; Prinsen, E.; Font-Llagunes, J.M.; Marchal-Crespo, L. Control strategies used in lower limb exoskeletons for gait rehabilitation after brain injury: A systematic review and analysis of clinical effectiveness. J. Neuroeng. Rehabil. 2023, 20, 23. [Google Scholar] [CrossRef]
- Meuleman, J.; van Asseldonk, E.; van Oort, G.; Rietman, H.; van der Kooij, H. LOPES II--Design and Evaluation of an Admittance Controlled Gait Training Robot With Shadow-Leg Approach. IEEE Trans. Neural Syst. Rehabil. Eng. 2016, 24, 352–363. [Google Scholar] [CrossRef]
- Aoyagi, D.; Ichinose, W.E.; Harkema, S.J.; Reinkensmeyer, D.J.; Bobrow, J.E. A robot and control algorithm that can synchronously assist in naturalistic motion during body-weight-supported gait training following neurologic injury. IEEE Trans. Neural Syst. Rehabil. Eng. 2007, 15, 387–400. [Google Scholar] [CrossRef]
- Banala, S.K.; Agrawal, S.K.; Scholz, J.P. Active Leg Exoskeleton (ALEX) for Gait Rehabilitation of Motor-Impaired Patients. In Proceedings of the 2007 IEEE 10th International Conference on Rehabilitation Robotics, Noordwijk, Netherlands, 13–15 June 2007. [Google Scholar]
- Mehrholz, J.; Thomas, S.; Kugler, J.; Pohl, M.; Elsner, B. Electromechanical-assisted training for walking after stroke. Cochrane Database Syst. Rev. 2020, 10, Cd006185. [Google Scholar] [CrossRef]
- Baronchelli, F.; Zucchella, C.; Serrao, M.; Intiso, D.; Bartolo, M. The Effect of Robotic Assisted Gait Training With Lokomat® on Balance Control After Stroke: Systematic Review and Meta-Analysis. Front. Neurol. 2021, 12, 661815. [Google Scholar] [CrossRef]
- Zhang, L.; Lin, F.; Sun, L.; Chen, C. Comparison of Efficacy of Lokomat and Wearable Exoskeleton-Assisted Gait Training in People With Spinal Cord Injury: A Systematic Review and Network Meta-Analysis. Front. Neurol. 2022, 13, 772660. [Google Scholar] [CrossRef]
- Calabrò, R.S.; Cassio, A.; Mazzoli, D.; Andrenelli, E.; Bizzarini, E.; Campanini, I.; Carmignano, S.M.; Cerulli, S.; Chisari, C.; Colombo, V.; et al. What does evidence tell us about the use of gait robotic devices in patients with multiple sclerosis? A comprehensive systematic review on functional outcomes and clinical recommendations. Eur. J. Phys. Rehabil. Med. 2021, 57, 841–849. [Google Scholar] [CrossRef]
- Hocoma. Lokomat Software. Available online: https://www.hocoma.com/solutions/lokomat/software-2/ (accessed on 22 February 2023).
- Maggioni, S.; Lünenburger, L.; Riener, R.; Curt, A.; Bolliger, M.; Melendez-Calderon, A. Assessing Walking Ability Using a Robotic Gait Trainer: Opportunities and Limitations of Assist-as-Needed Control in Spinal Cord Injury. PREPRINT (Version 1) available at Research Square. Res. Sq. 2022. [Google Scholar] [CrossRef]
- Nasreddine, Z.S.; Phillips, N.A.; Bédirian, V.; Charbonneau, S.; Whitehead, V.; Collin, I.; Cummings, J.L.; Chertkow, H. The Montreal Cognitive Assessment, MoCA: A brief screening tool for mild cognitive impairment. J. Am. Geriatr. Soc. 2005, 53, 695–699. [Google Scholar] [CrossRef] [PubMed]
- Colombo, G.; Joerg, M.; Schreier, R.; Dietz, V. Treadmill Training of Paraplegic Patients Using a Robotic Orthosis. J. Rehabil. Res. Dev. 2000, 37, 693–700. [Google Scholar] [PubMed]
- Maeda, A.; Yuasa, T.; Nakamura, K.; Higuchi, S.; Motohashi, Y. Physical performance tests after stroke: Reliability and validity. Am. J. Phys. Med. Rehabil. 2000, 79, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Podsiadlo, D.; Richardson, S. The timed “Up & Go”: A test of basic functional mobility for frail elderly persons. J. Am. Geriatr. Soc. 1991, 39, 142–148. [Google Scholar] [PubMed]
- Collen, F.M.; Wade, D.T.; Bradshaw, C.M. Mobility after stroke: Reliability of measures of impairment and disability. Int. Disabil. Stud. 1990, 12, 6–9. [Google Scholar] [CrossRef]
- Lunenburger, L.; Colombo, G.; Riener, R.; Dietz, V. Clinical assessments performed during robotic rehabilitation by the gait training robot Lokomat. In Proceedings of the 9th International Conference on Rehabilitation Robotics, 2005. ICORR 2005., Chicago, IL, USA, 28 June–1 July 2005. [Google Scholar]
- Rabin, R.; de Charro, F. EQ-5D: A measure of health status from the EuroQol Group. Ann. Med. 2001, 33, 337–343. [Google Scholar] [CrossRef]
- Federici, S.; Bracalenti, M.; Meloni, F.; Luciano, J.V. World Health Organization disability assessment schedule 2.0: An international systematic review. Disabil. Rehabil. 2017, 39, 2347–2380. [Google Scholar] [CrossRef]
- Davis, F.D. A Technology Acceptance Model for Empirically Testing New End-User Information Systems: Theory and Results. Ph.D. Thesis, Massachusetts Institute of Technology, Sloan School of Management, Cambridge, MA, USA, 1985. [Google Scholar]
- Venkatesh, V.; Davis, F. A Theoretical Extension of the Technology Acceptance Model: Four Longitudinal Field Studies. Manag. Sci. 2000, 46, 186–204. [Google Scholar] [CrossRef]
- Perry, J.; Burnfield, J.M. Gait Analysis. Normal and Pathological Function; SLACK Inc.: Thorofare, NJ, USA, 2010. [Google Scholar]
- Baker, R. Gait Graphs for Beginners. Available online: https://wwrichard.net/tag/gait-graphs/ (accessed on 20 April 2016).
- Zou, H.; Hastie, T. Regularization and variable selection via the elastic net. J. R. Stat. Society 2005, 67, 301–320. [Google Scholar] [CrossRef]
- Atkinson, G.; Nevill, A.M. Statistical methods for assessing measurement error (reliability) in variables relevant to sports medicine. Sport. Med. 1998, 26, 217–238. [Google Scholar] [CrossRef]
- Krishnan, C.; Ranganathan, R.; Dhaher, Y.Y.; Rymer, W.Z. A Pilot Study on the Feasibility of Robot-Aided Leg Motor Training to Facilitate Active Participation. PLoS ONE 2013, 8, e77370. [Google Scholar] [CrossRef]
- Lefeber, N.; De Keersmaecker, E.; Henderix, S.; Michielsen, M.; Tamburella, F.; Tagliamonte, N.L.; Molinari, M.; de Geus, B.; Kerckhofs, E.; Swinnen, E. Physiological responses and perceived exertion during robot-assisted treadmill walking in non-ambulatory stroke survivors. Disabil. Rehabil. 2021, 43, 1576–1584. [Google Scholar] [CrossRef]
- Lee, S.Y.; Han, E.Y.; Kim, B.R.; Chun, M.H.; Lee, Y.K. Can Lowering the Guidance Force of Robot-Assisted Gait Training Induce a Sufficient Metabolic Demand in Subacute Dependent Ambulatory Patients with Stroke? Arch. Phys. Med. Rehabil. 2017, 98, 695–700. [Google Scholar] [CrossRef]
- Krewer, C.; Muller, F.; Husemann, B.; Heller, S.; Quintern, J.; Koenig, E. The Influence of Different Lokomat Walking Conditions on the Energy Expenditure of Hemiparetic Patients and Healthy Subjects. Gait Posture 2007, 26, 372–377. [Google Scholar] [CrossRef]
- Chang, W.H.; Kim, M.S.; Huh, J.P.; Lee, P.K.; Kim, Y.H. Effects of Robot-Assisted Gait Training on Cardiopulmonary Fitness in Subacute Stroke Patients: A Randomized Controlled Study. Neurorehabil. Neural Repair 2012, 26, 318–324. [Google Scholar] [CrossRef]
- Mayr, A.; Kofler, M.; Quirbach, E.; Matzak, H.; Frohlich, K.; Saltuari, L. Prospective, Blinded, Randomized Crossover Study of Gait Rehabilitation in Stroke Patients Using the Lokomat Gait Orthosis. Neurorehabil. Neural Repair 2007, 21, 307–314. [Google Scholar] [CrossRef]
- Kirtley, C. Clinical Gait Analysis: Theory and Practice; Elsevier Health Sciences: Amsterdam, The Netherlands, 2006. [Google Scholar]
- Kim, C.M.; Eng, J.J. The relationship of lower-extremity muscle torque to locomotor performance in people with stroke. Phys. Ther. 2003, 83, 49–57. [Google Scholar] [CrossRef]
- Shrout, P.E. Measurement reliability and agreement in psychiatry. Stat. Methods Med. Res. 1998, 7, 301–317. [Google Scholar] [CrossRef]
- Awai, L.; Curt, A. Comprehensive assessment of walking function after human spinal cord injury. Prog. Brain Res. 2015, 218, 1–14. [Google Scholar] [CrossRef]
- Chaparro-Rico, B.D.M.; Cafolla, D.; Tortola, P.; Galardi, G. Assessing Stiffness, Joint Torque and ROM for Paretic and Non-Paretic Lower Limbs during the Subacute Phase of Stroke Using Lokomat Tools. Appl. Sci. 2020, 10, 6168. [Google Scholar] [CrossRef]



| PID | Diagnosis | Time Since Diagnosis | Gender (m/f) | Age (Years) | BMI (Kg/m2) | FAC (Level) |
|---|---|---|---|---|---|---|
| 1 | Aorta dissection | 10 months | m | 44 | 24.38 | 1 |
| 2 | CLIPPERS | 7 years | m | 45 | 20.57 | 1 |
| 3 | Epidural abscesses | 18 months | f | 40 | 25.54 | 0 |
| 4 | Stroke | 6 years | f | 71 | 28.40 | 4 |
| 5 | Viral infection | 19 years | m | 20 | 18.21 | 4 |
| 6 | Cerebral palsy | 26 years | f | 26 | 20.50 | 5 |
| 7 | Multiple sclerosis | 20 years | f | 58 | 15.63 | 0 |
| 8 | Stroke | 22 months | m | 79 | 30.06 | 1 |
| 9 | Neurosarcoidosis | 7 years | m | 65 | 26.09 | 0 |
| 10 | Friedreich’s ataxia | 13 years | f | 53 | 21.51 | 2 |
| PID | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
|---|---|---|---|---|---|---|---|---|---|---|
| Num. of training sessions | 16 | 12 | 8 | 8 | 10 | 8 | 8 | 8 | 13 | 8 |
| Assessment 1 | Assessment 2 | Assessment 3 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PID | MoCA | 10MWT | TUG | L-FORCE | Quality of Life Questionnaires | 10MWT | TUG | L-FORCE | Quality of Life Questionnaires | 10MWT | TUG | L-FORCE | Quality of Life Questionnaires | TAM |
| 1 | x | x | x | x | x | x | x | x | x | x | x | x | x | x |
| 2 | x | x | x | x | x | x | x | x | x | x | x | x | x | x |
| 3 | x | x | x | x | x | x | x | x | ||||||
| 4 | x | x | x | x | x | x | x | x | x | x | x | x | x | x |
| 5 | x | x | x | x | x | x | x | x | x | x | ||||
| 6 | x | x | x | x | x | x | x | x | x | x | x | x | x | x |
| 7 | x | x | x | x | x | x | x | x | ||||||
| 8 | x | x | x | x | x | x | x | x | x | x | x | x | x | x |
| 9 | x | x | x | x | x | x | x | x | ||||||
| 10 | x | x | x | x | x | x | x | x | ||||||
| 10MWT~1 | 0.0214 | |||||||||||
| Stiffness | Damping | |||||||||||
| Knee | Hip | Knee | Hip | |||||||||
| Initial Swing | Mid Swing | Terminal Swing | Initial Swing | Mid Swing | Terminal Swing | Initial Swing | Mid Swing | Terminal Swing | Initial Swing | Mid Swing | Terminal Swing | |
| 10MW~variable | 0.03429 | 0.907 | 0.1439 | 0.459 | 0.4535 | 0.1258 | 0.9441 | 0.877 | 0.844 | 0.388 | 0.9154 | 0.9397 |
| TUG~1 | 0.0733 | |||||||||||
| Stiffness | Damping | |||||||||||
| Knee | Hip | Knee | Hip | |||||||||
| Initial Swing | Mid Swing | Terminal Swing | Initial Swing | Mid Swing | Terminal Swing | Initial Swing | Mid Swing | Terminal Swing | Initial Swing | Mid Swing | Terminal Swing | |
| TUG~variable | 0.016 | 0.680 | 0.0407 | 0.674 | 0.596 | 0.0353 | 0.694 | 0.782 | 0.444 | 0.3865 | 0.658 | 0.541 |
| PID | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
|---|---|---|---|---|---|---|---|---|---|---|
| change in normalized K KNEE IS (%) | −3.4 | −15.6 | −15.2 | −14.5 | +8.7 | +0.2 | −5.9 | +0.9 | +8.1 | +2.6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laszlo, C.; Munari, D.; Maggioni, S.; Knechtle, D.; Wolf, P.; De Bon, D. Feasibility of an Intelligent Algorithm Based on an Assist-as-Needed Controller for a Robot-Aided Gait Trainer (Lokomat) in Neurological Disorders: A Longitudinal Pilot Study. Brain Sci. 2023, 13, 612. https://doi.org/10.3390/brainsci13040612
Laszlo C, Munari D, Maggioni S, Knechtle D, Wolf P, De Bon D. Feasibility of an Intelligent Algorithm Based on an Assist-as-Needed Controller for a Robot-Aided Gait Trainer (Lokomat) in Neurological Disorders: A Longitudinal Pilot Study. Brain Sciences. 2023; 13(4):612. https://doi.org/10.3390/brainsci13040612
Chicago/Turabian StyleLaszlo, Caroline, Daniele Munari, Serena Maggioni, Deborah Knechtle, Peter Wolf, and Dino De Bon. 2023. "Feasibility of an Intelligent Algorithm Based on an Assist-as-Needed Controller for a Robot-Aided Gait Trainer (Lokomat) in Neurological Disorders: A Longitudinal Pilot Study" Brain Sciences 13, no. 4: 612. https://doi.org/10.3390/brainsci13040612
APA StyleLaszlo, C., Munari, D., Maggioni, S., Knechtle, D., Wolf, P., & De Bon, D. (2023). Feasibility of an Intelligent Algorithm Based on an Assist-as-Needed Controller for a Robot-Aided Gait Trainer (Lokomat) in Neurological Disorders: A Longitudinal Pilot Study. Brain Sciences, 13(4), 612. https://doi.org/10.3390/brainsci13040612








