Molecular Imaging of Central Dopamine in Obesity: A Qualitative Review across Substrates and Radiotracers
Abstract
1. Introduction
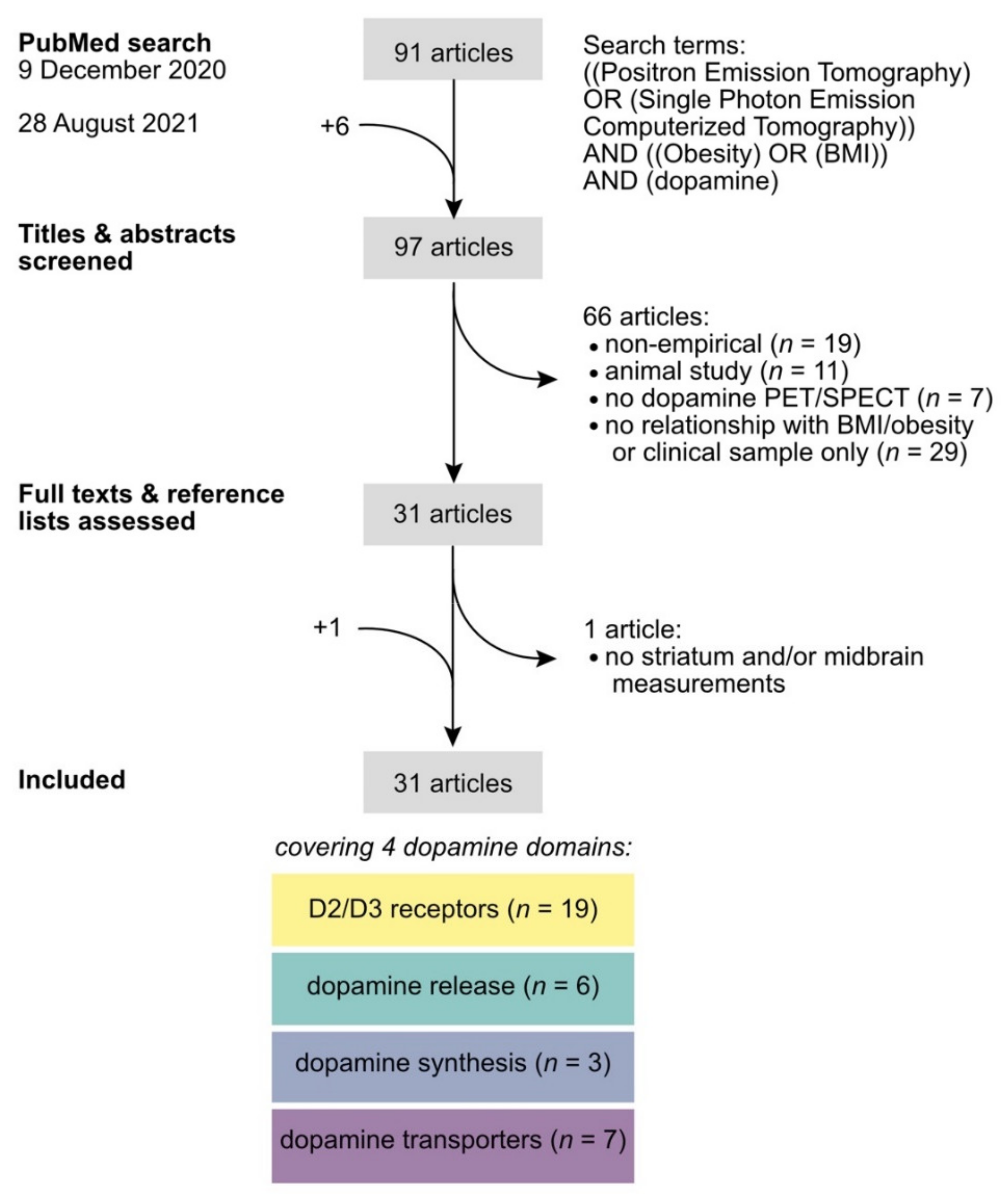
2. Literature Search
3. Summary of the Findings
3.1. D2/D3 Receptors
3.2. Dopamine Release
3.3. Dopamine Synthesis
3.4. Dopamine Transporters
4. Discussion
4.1. Integrating Findings across Domains
4.2. Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Montague, P.R.; Hyman, S.E.; Cohen, J.D. Computational roles for dopamine in behavioural control. Nature 2004, 431, 760–767. [Google Scholar] [CrossRef] [PubMed]
- Fallon, S.J.; van der Schaaf, M.E.; ter Huurne, N.; Cools, R. The Neurocognitive Cost of Enhancing Cognition with Methylphenidate: Improved Distractor Resistance but Impaired Updating. J. Cogn. Neurosci. 2017, 29, 652–663. [Google Scholar] [CrossRef] [PubMed]
- Bloemendaal, M.; van Schouwenburg, M.R.; Miyakawa, A.; Aarts, E.; D’Esposito, M.; Cools, R. Dopaminergic modulation of distracter-resistance and prefrontal delay period signal. Psychopharmacology 2015, 232, 1061–1070. [Google Scholar] [CrossRef] [PubMed]
- Cools, R.; D’Esposito, M. Inverted-U Shaped Dopamine Actions on Human Working Memory and Cognitive Control. Biol. Psychiatry 2011, 69, e113–e125. [Google Scholar] [CrossRef] [PubMed]
- Cools, R.; Gibbs, S.E.; Miyakawa, A.; Jagust, W.; D’Esposito, M. Working Memory Capacity Predicts Dopamine Synthesis Capacity in the Human Striatum. J. Neurosci. 2008, 28, 1208–1212. [Google Scholar] [CrossRef] [PubMed]
- Durstewitz, D.; Seamans, J.K. The Dual-State Theory of Prefrontal Cortex Dopamine Function with Relevance to Catechol-O-Methyltransferase Genotypes and Schizophrenia. Biol. Psychiatry 2008, 64, 739–749. [Google Scholar] [CrossRef] [PubMed]
- Schultz, W.; Dayan, P.; Montague, P.R. A Neural Substrate of Prediction and Reward. Science 1997, 275, 1593–1599. [Google Scholar] [CrossRef]
- Montague, P.R.; Dayan, P.; Sejnowski, T.J. A framework for mesencephalic dopamine systems based on predictive Hebbian learning. J. Neurosci. 1996, 16, 1936–1947. [Google Scholar] [CrossRef]
- Schultz, W. Behavioral theories and the neurophysiology of reward. Annu. Rev. Psychol. 2006, 57, 87–115. [Google Scholar] [CrossRef]
- Robbins, T.W.; Everitt, B.J. A role for mesencephalic dopamine in activation: Commentary on Berridge (2006). Psychopharmacology 2007, 191, 433–437. [Google Scholar] [CrossRef]
- Berridge, K. The debate over dopamine’s role in reward: The case for incentive salience. Psychopharmacology 2007, 191, 391–431. [Google Scholar] [CrossRef] [PubMed]
- Berridge, K.C. From prediction error to incentive salience: Mesolimbic computation of reward motivation. Eur. J. Neurosci. 2012, 35, 1124–1143. [Google Scholar] [CrossRef] [PubMed]
- Salamone, J.D.; Correa, M. Motivational views of reinforcement: Implications for understanding the behavioral functions of nucleus accumbens dopamine. Behav. Brain Res. 2002, 137, 3–25. [Google Scholar] [CrossRef]
- Janssen, L.K.; Herzog, N.; Waltmann, M.; Breuer, N.; Wiencke, K.; Rausch, F.; Hartmann, H.; Poessel, M.; Horstmann, A. Lost in Translation? On the Need for Convergence in Animal and Human Studies on the Role of Dopamine in Diet-Induced Obesity. Curr. Addict. Rep. 2019, 6, 229–257. [Google Scholar] [CrossRef]
- Horstmann, A.; Fenske, W.K.; Hankir, M.K. Argument for a non-linear relationship between severity of human obesity and dopaminergic tone. Obes. Rev. 2015, 16, 821–830. [Google Scholar] [CrossRef]
- Val-Laillet, D.; Aarts, E.; Weber, B.; Ferrari, M.; Quaresima, V.; Stoeckel, L.E.; Alonso-Alonso, M.; Audette, M.; Malbert, C.H.; Stice, E. Neuroimaging and neuromodulation approaches to study eating behavior and prevent and treat eating disorders and obesity. NeuroImage Clin. 2015, 8, 1–31. [Google Scholar] [CrossRef]
- Volkow, N.D.; Wise, R.A.; Baler, R. The dopamine motive system: Implications for drug and food addiction. Nat. Rev. Neurosci. 2017, 18, 741. [Google Scholar] [CrossRef]
- Matikainen-Ankney, B.A.; Kravitz, A.V. Persistent effects of obesity: A neuroplasticity hypothesis. Ann. N. Y. Acad. Sci. 2018, 1428, 221–239. [Google Scholar] [CrossRef]
- Burger, K.S.; Stice, E. Variability in Reward Responsivity and Obesity: Evidence from Brain Imaging Studies. Curr. Drug Abuse Rev. 2011, 4, 182–189. [Google Scholar] [CrossRef]
- Dagher, A. Functional brain imaging of appetite. Trends Endocrinol. Metab. 2012, 23, 250–260. [Google Scholar] [CrossRef]
- Wallace, C.W.; Fordahl, S.C. Obesity and dietary fat influence dopamine neurotransmission: Exploring the convergence of metabolic state, physiological stress, and inflammation on dopaminergic control of food intake. Nutr. Res. Rev. 2021, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Treadway, M.T.; Cooper, J.A.; Miller, A.H. Can’t or Won’t? Immunometabolic Constraints on Dopaminergic Drive. Trends Cogn. Sci. 2019, 23, 435–448. [Google Scholar] [CrossRef] [PubMed]
- Van Galen, K.A.; ter Horst, K.W.; Booij, J.; la Fleur, S.E.; Serlie, M.J. The role of central dopamine and serotonin in human obesity: Lessons learned from molecular neuroimaging studies. Metab. Clin. Exp. 2018, 85, 325–339. [Google Scholar] [CrossRef] [PubMed]
- Bini, J.; Norcross, M.; Cheung, M.; Duffy, A. The Role of Positron Emission Tomography in Bariatric Surgery Research: A Review. Obes. Surg. 2021, 31, 4592–4606. [Google Scholar] [CrossRef]
- Pak, K.; Kim, K.; Kim, I.J. Dopamine transporter in obesity: A meta-analysis. medRxiv 2021. medRxiv:2021.01.05.21249294. [Google Scholar] [CrossRef]
- Van de Giessen, E.; McIlwrick, S.; Veltman, D.; van den Brink, W.; Booij, J. Obesity an addiction? Imaging of neurotransmitter systems in obesity. In PET and SPECT in Psychiatry; Dierckx, R.A.J.O., Otte, A., de Vries, E.F.J., van Waarde, A., den Boer, J.A., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 641–656. ISBN 978-3-642-40384-2. [Google Scholar]
- Volkow, N.D.; Wang, G.-J.; Fowler, J.S.; Telang, F. Overlapping neuronal circuits in addiction and obesity: Evidence of systems pathology. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2008, 363, 3191–3200. [Google Scholar] [CrossRef]
- Topor, M.; Pickering, J.; Mendes, A.B.; Bishop, D.; Büttner, F.C.; Elsherif, M.; Evans, T.R.; Henderson, E.L.; Kalandadze, T.; Nitschke, F.; et al. An integrative framework for planning and conducting Non-Intervention, Reproducible, and Open Systematic Reviews (NIRO-SR). MetaArXiv 2020. [Google Scholar] [CrossRef]
- Yasuno, F.; Suhara, T.; Sudo, Y.; Yamamoto, M.; Inoue, M.; Okubo, Y.; Suzuki, K. Relation among dopamine D(2) receptor binding, obesity and personality in normal human subjects. Neurosci. Lett. 2001, 300, 59–61. [Google Scholar] [CrossRef]
- De Weijer, B.A.; van de Giessen, E.; van Amelsvoort, T.A.; Boot, E.; Braak, B.; Janssen, I.M.; van de Laar, A.; Fliers, E.; Serlie, M.J.; Booij, J. Lower striatal dopamine D2/3 receptor availability in obese compared with non-obese subjects. EJNMMI Res. 2011, 1, 37. [Google Scholar] [CrossRef]
- van de Giessen, E.; Celik, F.; Schweitzer, D.H.; van den Brink, W.; Booij, J. Dopamine D2/3 receptor availability and amphetamine-induced dopamine release in obesity. J. Psychopharmacol. 2014, 28, 866–873. [Google Scholar] [CrossRef]
- Wang, G.-J.; Volkow, N.D.; Logan, J.; Pappas, N.R.; Wong, C.T.; Zhu, W.; Netusll, N.; Fowler, J.S. Brain dopamine and obesity. Lancet 2001, 357, 354–357. [Google Scholar] [CrossRef]
- Haltia, L.T.; Rinne, J.O.; Merisaari, H.; Maguire, R.P.; Savontaus, E.; Helin, S.; Någren, K.; Kaasinen, V. Effects of intravenous glucose on dopaminergic function in the human brain in vivo. Synapse 2007, 61, 748–756. [Google Scholar] [CrossRef] [PubMed]
- Volkow, N.D.; Wang, G.-J.; Telang, F.; Fowler, J.S.; Thanos, P.K.; Logan, J.; Alexoff, D.; Ding, Y.-S.; Wong, C.; Ma, Y.; et al. Low dopamine striatal D2 receptors are associated with prefrontal metabolism in obese subjects: Possible contributing factors. NeuroImage 2008, 42, 1537–1543. [Google Scholar] [CrossRef] [PubMed]
- Steele, K.E.; Prokopowicz, G.P.; Schweitzer, M.A.; Magunsuon, T.H.; Lidor, A.O.; Kuwabawa, H.; Kumar, A.; Brasic, J.; Wong, D.F. Alterations of central dopamine receptors before and after gastric bypass surgery. Obes. Surg. 2010, 20, 369–374. [Google Scholar] [CrossRef]
- Wang, G.-J.; Geliebter, A.; Volkow, N.D.; Telang, F.W.; Logan, J.; Jayne, M.C.; Galanti, K.; Selig, P.A.; Han, H.; Zhu, W.; et al. Enhanced Striatal Dopamine Release During Food Stimulation in Binge Eating Disorder. Obesity 2011, 19, 1601–1608. [Google Scholar] [CrossRef]
- Karlsson, H.K.; Tuominen, L.; Tuulari, J.J.; Hirvonen, J.; Parkkola, R.; Helin, S.; Salminen, P.; Nuutila, P.; Nummenmaa, L. Obesity Is Associated with Decreased -Opioid but Unaltered Dopamine D2 Receptor Availability in the Brain. J. Neurosci. 2015, 35, 3959–3965. [Google Scholar] [CrossRef]
- Tuominen, L.; Tuulari, J.; Karlsson, H.; Hirvonen, J.; Helin, S.; Salminen, P.; Parkkola, R.; Hietala, J.; Nuutila, P.; Nummenmaa, L. Aberrant mesolimbic dopamine–opiate interaction in obesity. NeuroImage 2015, 122, 80–86. [Google Scholar] [CrossRef]
- Cho, S.S.; Yoon, E.J.; Kim, S.E. Asymmetry of Dopamine D2/3 Receptor Availability in Dorsal Putamen and Body Mass Index in Non-obese Healthy Males. Exp. Neurobiol. 2015, 24, 90–94. [Google Scholar] [CrossRef]
- Caravaggio, F.; Raitsin, S.; Gerretsen, P.; Nakajima, S.; Wilson, A.; Graff-Guerrero, A. Ventral Striatum Binding of a Dopamine D2/3 Receptor Agonist but Not Antagonist Predicts Normal Body Mass Index. Biol. Psychiatry 2015, 77, 196–202. [Google Scholar] [CrossRef]
- Cosgrove, K.P.; Veldhuizen, M.G.; Sandiego, C.M.; Morris, E.D.; Small, D.M. Opposing relationships of BMI with BOLD and dopamine D2/3 receptor binding potential in the dorsal striatum. Synapse 2015, 69, 195–202. [Google Scholar] [CrossRef]
- Gaiser, E.C.; Gallezot, J.-D.; Worhunsky, P.D.; Jastreboff, A.M.; Pittman, B.; Kantrovitz, L.; Angarita, G.A.; Cosgrove, K.P.; Potenza, M.N.; Malison, R.T.; et al. Elevated Dopamine D2/3 Receptor Availability in Obese Individuals: A PET Imaging Study with [11C](+)PHNO. Neuropsychopharmacology 2016, 41, 3042–3050. [Google Scholar] [CrossRef] [PubMed]
- Dunn, J.P.; Kessler, R.M.; Feurer, I.D.; Volkow, N.D.; Patterson, B.W.; Ansari, M.S.; Li, R.; Marks-Shulman, P.; Abumrad, N.N. Relationship of dopamine type 2 receptor binding potential with fasting neuroendocrine hormones and insulin sensitivity in human obesity. Diabetes Care 2012, 35, 1105–1111. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Simmons, W.K.; Herscovitch, P.; Martin, A.; Hall, K.D. Striatal dopamine D2-like receptor correlation patterns with human obesity and opportunistic eating behavior. Mol. Psychiatry 2014, 19, 1078–1084. [Google Scholar] [CrossRef] [PubMed]
- Kessler, R.M.; Zald, D.H.; Ansari, M.S.; Li, R.; Cowan, R.L. Changes in dopamine release and dopamine D2/3 receptor levels with the development of mild obesity. Synapse 2014, 68, 317–320. [Google Scholar] [CrossRef] [PubMed]
- Dang, L.C.; Samanez-Larkin, G.R.; Castrellon, J.J.; Perkins, S.F.; Cowan, R.L.; Zald, D.H. Associations between dopamine D2 receptor availability and BMI depend on age. NeuroImage 2017, 138, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Eisenstein, S.A.; Antenor-Dorsey, J.A.V.; Gredysa, D.M.; Koller, J.M.; Bihun, E.C.; Ranck, S.A.; Arbeláez, A.M.; Klein, S.; Perlmutter, J.S.; Moerlein, S.M.; et al. A Comparison of D2 Receptor Specific Binding in Obese and Normal-weight Individuals Using PET with (N-[11C]methyl)benperidol. Synapse 2013, 67, 748–756. [Google Scholar] [CrossRef]
- Eisenstein, S.A.; Bischoff, A.N.; Gredysa, D.M.; Antenor-Dorsey, J.A.V.; Koller, J.M.; Al-Lozi, A.; Pepino, M.Y.; Klein, S.; Perlmutter, J.S.; Moerlein, S.M.; et al. Emotional Eating Phenotype is Associated with Central Dopamine D2 Receptor Binding Independent of Body Mass Index. Sci. Rep. 2015, 5, 11283. [Google Scholar] [CrossRef]
- Haltia, L.T.; Rinne, J.O.; Helin, S.; Parkkola, R.; Någren, K.; Kaasinen, V. Effects of intravenous placebo with glucose expectation on human basal ganglia dopaminergic function. Synapse 2008, 62, 682–688. [Google Scholar] [CrossRef]
- Wang, G.-J.; Tomasi, D.; Convit, A.; Logan, J.; Wong, C.T.; Shumay, E.; Fowler, J.S.; Volkow, N.D. BMI Modulates Calorie-Dependent Dopamine Changes in Accumbens from Glucose Intake. PLoS ONE 2014, 9, e101585. [Google Scholar] [CrossRef]
- Wilcox, C.E.; Braskie, M.N.; Kluth, J.T.; Jagust, W.J. Overeating Behavior and Striatal Dopamine with 6- [ 1 8 F ] -Fluoro-L- 𝑚 -Tyrosine PET. J. Obes. 2010, 2010, e909348. [Google Scholar] [CrossRef]
- Wallace, D.L.; Aarts, E.; Dang, L.C.; Greer, S.M.; Jagust, W.J.; D’Esposito, M. Dorsal Striatal Dopamine, Food Preference and Health Perception in Humans. PLoS ONE 2014, 9, e96319. [Google Scholar] [CrossRef]
- Lee, Y.; Kroemer, N.B.; Oehme, L.; Beuthien-Baumann, B.; Goschke, T.; Smolka, M.N. Lower dopamine tone in the striatum is associated with higher body mass index. Eur. Neuropsychopharmacol. 2018, 28, 719–731. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.S.; Yang, Y.K.; Yeh, T.L.; Lee, I.-H.; Yao, W.J.; Chiu, N.T.; Lu, R.B. Correlation between body mass index and striatal dopamine transporter availability in healthy volunteers—A SPECT study. NeuroImage 2008, 40, 275–279. [Google Scholar] [CrossRef] [PubMed]
- Koskela, A.K.; Kaurijoki, S.; Pietiläinen, K.H.; Karhunen, L.; Pesonen, U.; Kuikka, J.T.; Kaprio, J.; Rissanen, A. Serotonin transporter binding and acquired obesity—An imaging study of monozygotic twin pairs. Physiol. Behav. 2008, 93, 724–732. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, G.; Ziebell, M.; Jensen, P.S.; da Cuhna-Bang, S.; Knudsen, G.M.; Pinborg, L.H. No correlation between body mass index and striatal dopamine transporter availability in healthy volunteers using SPECT and [123I]PE2I. Obesity 2013, 21, 1803–1806. [Google Scholar] [CrossRef]
- Van de Giessen, E.; Hesse, S.; Caan, M.W.A.; Zientek, F.; Dickson, J.C.; Tossici-Bolt, L.; Sera, T.; Asenbaum, S.; Guignard, R.; Akdemir, U.O.; et al. No association between striatal dopamine transporter binding and body mass index: A multi-center European study in healthy volunteers. NeuroImage 2013, 64, 61–67. [Google Scholar] [CrossRef]
- Versteeg, R.I.; Koopman, K.E.; Booij, J.; Ackermans, M.T.; Unmehopa, U.A.; Fliers, E.; la Fleur, S.E.; Serlie, M.J. Serotonin Transporter Binding in the Diencephalon Is Reduced in Insulin-Resistant Obese Humans. Neuroendocrinology 2017, 105, 141–149. [Google Scholar] [CrossRef]
- Nam, S.B.; Kim, K.; Kim, B.S.; Im, H.-J.; Lee, S.H.; Kim, S.-J.; Kim, I.J.; Pak, K. The Effect of Obesity on the Availabilities of Dopamine and Serotonin Transporters. Sci. Rep. 2018, 8, 4924. [Google Scholar] [CrossRef]
- Pak, K.; Seo, S.; Kim, K.; Lee, M.J.; Shin, M.J.; Suh, S.; Im, H.-J.; Park, J.-J.; Kim, S.-J.; Kim, I.J. Striatal dopamine transporter changes after glucose loading in humans. Diabetes Obes. Metab. 2020, 22, 116–122. [Google Scholar] [CrossRef]
- Mukherjee, J.; Yang, Z.-Y.; Brown, T.; Lew, R.; Wernick, M.; Ouyang, X.; Yasillo, N.; Chen, C.-T.; Mintzer, R.; Cooper, M. Preliminary assessment of extrastriatal dopamine d-2 receptor binding in the rodent and nonhuman primate brains using the high affinity radioligand, 18F-fallypride. Nucl. Med. Biol. 1999, 26, 519–527. [Google Scholar] [CrossRef]
- Berry, A.S.; Shah, V.D.; Baker, S.L.; Vogel, J.W.; O’Neil, J.P.; Janabi, M.; Schwimmer, H.D.; Marks, S.M.; Jagust, W.J. Aging Affects Dopaminergic Neural Mechanisms of Cognitive Flexibility. J. Neurosci. Off. J. Soc. Neurosci. 2016, 36, 12559–12569. [Google Scholar] [CrossRef] [PubMed]
- Berry, A.S.; Jagust, W.J.; Hsu, M. Age-related variability in decision-making: Insights from neurochemistry. Cogn. Affect. Behav. Neurosci. 2019, 19, 415–434. [Google Scholar] [CrossRef] [PubMed]
- Rinne, J.O.; Hietala, J.; Ruotsalainen, U.; Säkö, E.; Laihinen, A.; Någren, K.; Lehikoinen, P.; Oikonen, V.; Syvälahti, E. Decrease in human striatal dopamine D2 receptor density with age: A PET study with [11C]raclopride. J. Cereb. Blood Flow Metab. 1993, 13, 310–314. [Google Scholar] [CrossRef]
- Small, D.M.; Jones-Gotman, M.; Dagher, A. Feeding-induced dopamine release in dorsal striatum correlates with meal pleasantness ratings in healthy human volunteers. Neuroimage 2003, 19, 1709–1715. [Google Scholar] [CrossRef]
- Feifel, D.; Shilling, P.D.; Kuczenski, R.; Segal, D.S. Altered extracellular dopamine concentration in the brains of cholecystokinin-A receptor deficient rats. Neurosci. Lett. 2003, 348, 147–150. [Google Scholar] [CrossRef]
- Thanos, P.K.; Michaelides, M.; Piyis, Y.K.; Wang, G.-J.; Volkow, N.D. Food restriction markedly increases dopamine D2 receptor (D2R) in a rat model of obesity as assessed with in-vivo muPET imaging ([11C] raclopride) and in-vitro ([3H] spiperone) autoradiography. Synapse 2008, 62, 50–61. [Google Scholar] [CrossRef] [PubMed]
- Anderzhanova, E.; Covasa, M.; Hajnal, A. Altered basal and stimulated accumbens dopamine release in obese OLETF rats as a function of age and diabetic status. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007, 293, R603–R611. [Google Scholar] [CrossRef]
- Bossert, J.M.; Poles, G.C.; Wihbey, K.A.; Koya, E.; Shaham, Y. Differential effects of blockade of dopamine D1-family receptors in nucleus accumbens core or shell on reinstatement of heroin seeking induced by contextual and discrete cues. J. Neurosci. Off. J. Soc. Neurosci. 2007, 27, 12655–12663. [Google Scholar] [CrossRef]
- Floresco, S.B.; McLaughlin, R.J.; Haluk, D.M. Opposing roles for the nucleus accumbens core and shell in cue-induced reinstatement of food-seeking behavior. Neuroscience 2008, 154, 877–884. [Google Scholar] [CrossRef]
- Ito, R.; Hayen, A. Opposing Roles of Nucleus Accumbens Core and Shell Dopamine in the Modulation of Limbic Information Processing. J. Neurosci. 2011, 31, 6001–6007. [Google Scholar] [CrossRef]
- Sesia, T.; Temel, Y.; Lim, L.W.; Blokland, A.; Steinbusch, H.W.M.; Visser-Vandewalle, V. Deep brain stimulation of the nucleus accumbens core and shell: Opposite effects on impulsive action. Exp. Neurol. 2008, 214, 135–139. [Google Scholar] [CrossRef] [PubMed]
- Berry, A.S.; Shah, V.D.; Furman, D.J.; White, R.L., III; Baker, S.L.; O’Neil, J.P.; Janabi, M.; D’Esposito, M.; Jagust, W.J. Dopamine Synthesis Capacity is Associated with D2/3 Receptor Binding but Not Dopamine Release. Neuropsychopharmacology 2018, 43, 1201–1211. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Garamszegi, S.P.; Xie, X.; Mash, D.C. Altered Dopamine Synaptic Markers in Postmortem Brain of Obese Subjects. Front. Hum. Neurosci. 2017, 11, 386. [Google Scholar] [CrossRef] [PubMed]
- Everitt, B.J.; Belin, D.; Economidou, D.; Pelloux, Y.; Dalley, J.W.; Robbins, T.W. Neural mechanisms underlying the vulnerability to develop compulsive drug-seeking habits and addiction. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2008, 363, 3125–3135. [Google Scholar] [CrossRef]
- Everitt, B.J.; Robbins, T.W. Neural systems of reinforcement for drug addiction: From actions to habits to compulsion. Nat. Neurosci. 2005, 8, 1481–1489. [Google Scholar] [CrossRef]
- Everitt, B.J.; Robbins, T.W. From the ventral to the dorsal striatum: Devolving views of their roles in drug addiction. Neurosci. Biobehav. Rev. 2013, 37, 1946–1954. [Google Scholar] [CrossRef]
- Ziauddeen, H.; Fletcher, P.C. Is food addiction a valid and useful concept? Obes. Rev. 2013, 14, 19–28. [Google Scholar] [CrossRef]
- Ziauddeen, H.; Farooqi, I.S.; Fletcher, P.C. Obesity and the brain: How convincing is the addiction model? Nat. Rev. Neurosci. 2012, 13, 279–286. [Google Scholar] [CrossRef]
- Fletcher, P.C.; Kenny, P.J. Food addiction: A valid concept? Neuropsychopharmacology 2018, 43, 2506–2513. [Google Scholar] [CrossRef]
- Hebebrand, J.; Albayrak, Ö.; Adan, R.; Antel, J.; Dieguez, C.; de Jong, J.; Leng, G.; Menzies, J.; Mercer, J.G.; Murphy, M.; et al. “Eating addiction”, rather than “food addiction”, better captures addictive-like eating behavior. Neurosci. Biobehav. Rev. 2014, 47, 295–306. [Google Scholar] [CrossRef]
- Smeets, P.A.M.; Dagher, A.; Hare, T.A.; Kullmann, S.; van der Laan, L.N.; Poldrack, R.A.; Preissl, H.; Small, D.; Stice, E.; Veldhuizen, M.G. Good practice in food-related neuroimaging. Am. J. Clin. Nutr. 2019, 109, 491–503. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, H.K.; Tuominen, L.; Helin, S.; Salminen, P.; Nuutila, P.; Nummenmaa, L. Mesolimbic opioid-dopamine interaction is disrupted in obesity but recovered by weight loss following bariatric surgery. Transl. Psychiatry 2021, 11, 259. [Google Scholar] [CrossRef] [PubMed]
- Caravaggio, F.; Borlido, C.; Hahn, M.; Feng, Z.; Fervaha, G.; Gerretsen, P.; Nakajima, S.; Plitman, E.; Chung, J.K.; Iwata, Y. Reduced insulin sensitivity is related to less endogenous dopamine at D2/3 receptors in the ventral striatum of healthy nonobese humans. Int. J. Neuropsychopharmacol. 2015, 18, pyv014. [Google Scholar] [CrossRef] [PubMed]
- de Weijer, B.A.; van de Giessen, E.; Janssen, I.; Berends, F.J.; van de Laar, A.; Ackermans, M.T.; Fliers, E.; la Fleur, S.E.; Booij, J.; Serlie, M.J. Striatal dopamine receptor binding in morbidly obese women before and after gastric bypass surgery and its relationship with insulin sensitivity. Diabetologia 2014, 57, 1078–1080. [Google Scholar] [CrossRef]
- Dunn, J.P.; Abumrad, N.N.; Patterson, B.W.; Kessler, R.M.; Tamboli, R.A. Brief communication: β-cell function influences dopamine receptor availability. PLoS ONE 2019, 14, e0212738. [Google Scholar] [CrossRef]
- Eisenstein, S.A.; Gredysa, D.M.; Antenor–Dorsey, J.A.; Green, L.; Arbeláez, A.M.; Koller, J.M.; Black, K.J.; Perlmutter, J.S.; Moerlein, S.M.; Hershey, T. Insulin, Central Dopamine D2 Receptors, and Monetary Reward Discounting in Obesity. PLoS ONE 2015, 10, e0133621. [Google Scholar] [CrossRef]
- Kullmann, S.; Blum, D.; Jaghutriz, B.A.; Gassenmaier, C.; Bender, B.; Häring, H.-U.; Reischl, G.; Preissl, H.; la Fougère, C.; Fritsche, A.; et al. Central insulin modulates dopamine signaling in the human striatum. J. Clin. Endocrinol. Metab. 2021, 106, 2949–2961. [Google Scholar] [CrossRef]
- Suissa, K.; Schneeweiss, S.; Kim, D.W.; Patorno, E. Prescribing trends and clinical characteristics of patients starting antiobesity drugs in the United States. Diabetes Obes. Metab. 2021, 23, 1542–1551. [Google Scholar] [CrossRef]
- Coulter, A.A.; Rebello, C.J.; Greenway, F.L. Centrally Acting Drugs for Obesity: Past, Present, and Future. Drugs 2018, 78, 1113–1132. [Google Scholar] [CrossRef]
- Arumäe, K.; Mõttus, R.; Vainik, U. Beyond BMI: Personality Traits’ Associations with Adiposity and Metabolic Rate. Physiol. Behav. 2022, 246, 113703. [Google Scholar] [CrossRef]
- Nuttall, F.Q. Body Mass Index: Obesity, BMI, and Health: A Critical Review. Nutr. Today 2015, 50, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Sulc, J.; Sonrel, A.; Mounier, N.; Auwerx, C.; Marouli, E.; Darrous, L.; Draganski, B.; Kilpeläinen, T.O.; Joshi, P.; Loos, R.J.F.; et al. Composite trait Mendelian randomization reveals distinct metabolic and lifestyle consequences of differences in body shape. Commun. Biol. 2021, 4, 1064. [Google Scholar] [CrossRef] [PubMed]
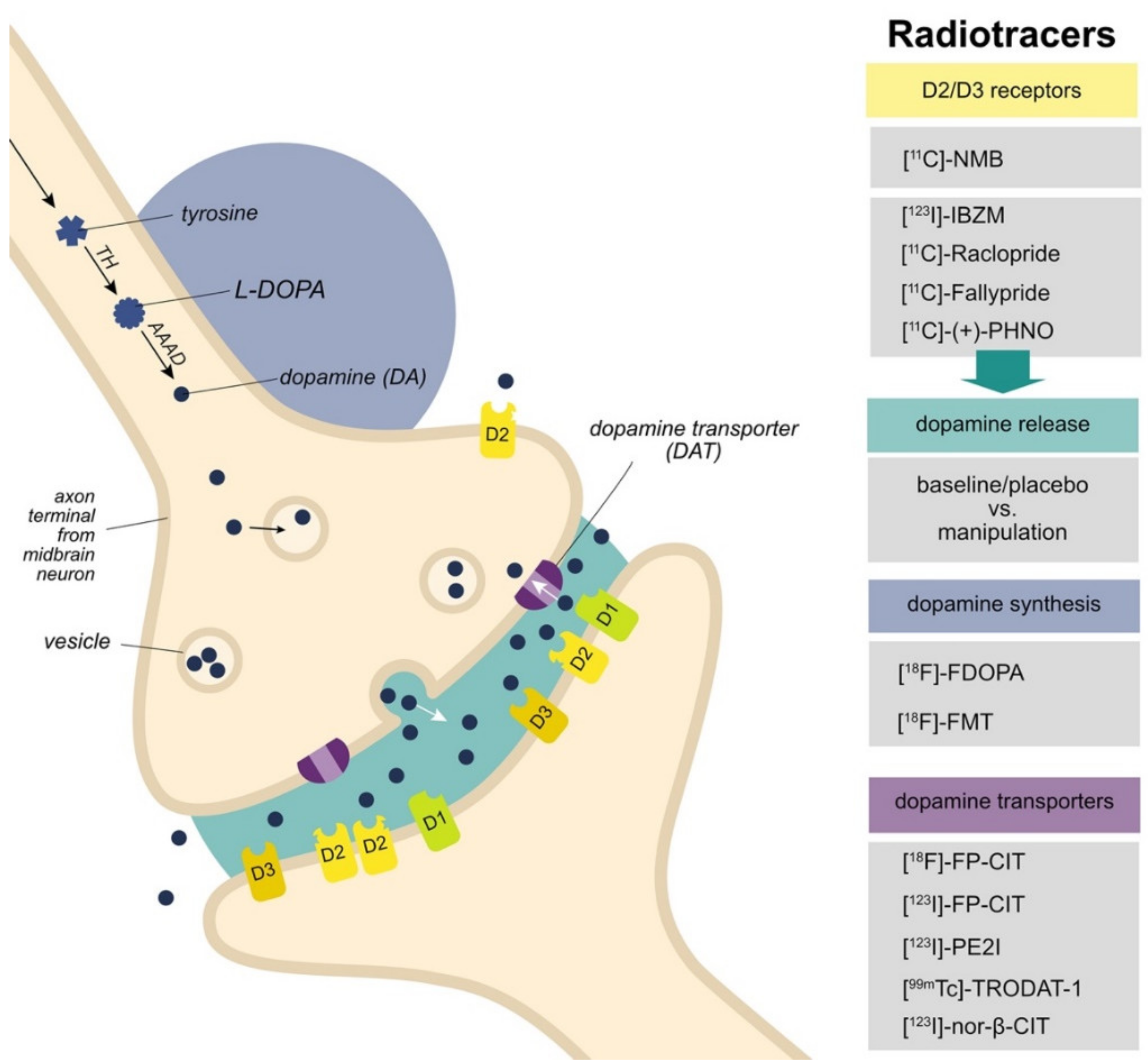
| Reference | Imaging | Sample | Primary/ Secondary Analysis | Findings | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | BMI | Age | Fasting | Smoking | |||||||||
| Radiotracer | DV | WB/ROI | F:M | Groups | M(SD) | Range | M(SD) | Range | |||||
| SPECT studies | |||||||||||||
| De Weijer et al., 2011 [30] | [123I]-IBZM | D2/D3 BPND | STR | 15:0 | NW + OW | 21.7(2.1) | 19.5–27.6 | 28(10.4) | 20–60 | Not fasted, not fed | Primary | OB < NW + OW BMI: N.S. in separate groups | |
| 15:0 | OB | 46.8(6.5) | 38.7–61.3 | 37.8(7) | 26–49 | Fasted (overnight) | |||||||
| van de Giessen et al., 2014 [31] | [123I]-IBZM (+baseline) | D2/D3 BPND | STR | 15:0 | NW | 21.8(1.8) | 18.5–24.9 | 38.5(5.6) | 18–45 | Fed (standard breakfast) | None | Secondary | OB < NW BMI: N.S. in separate groups |
| 15:0 | OB | 42.9(4.9) | 36.3–56.6 | 36.3(4.0) | 18–45 | ||||||||
| PET studies | |||||||||||||
| Wang et al., 2001 [32] | [11C]-Raclopride | D2/D3 DVR = BPND + 1 | STR | 3:7 | NW + OW | 24.7(2.6) | 21–28 | 37.5(5.9) | 25–54 | 1 light smoker | Primary 1 | OB < NW + OW BMI (OB): STR↓ | |
| 5:5 | OB | 51.2(4.8) | 42–60 | 38.9(7.3) | 26–54 | None | |||||||
| Haltia et al., 2007 [33] | [11C]-Raclopride (+placebo) | D2/D3 BPND | WB; CAU, PUT | 6:6 | NW | 22(1) | <24 | ±25.5(4.5) | Fasted (overnight) | No heavy smokers | Secondary | OB < NW in STR (WB only); BMI (OW/OB): PUT↓ | |
| 6:6 | OW + OB | 33(5) | >27 | ±25(2.5) | |||||||||
| Volkow et al., 2008 [34] | [11C]-Raclopride | D2/D3 DVR = BPND + 1 | STR (CAU + PUT) | 6:6 | NW + OW | 25(3) | <30 | 33.2(8) | 20–55 | None | Secondary 1 | OB < NW + OW | |
| 5:5 | OB | 51(5) | >40 | 35.9(10) | 20–55 | ||||||||
| Steele et al., 2010 [35] | [11C]-Raclopride | D2/D3 DVR = BPND + 1 | a, pCAU a, pPUT, vSTR | 5:0 | NW | 21.3 | 21.8 | No tobacco use >once a week | Primary | OB = NW | |||
| 5:0 | OB | 44.8 | 40–53 | 32.2 | 20–38 | ||||||||
| Wang et al., 2011 [36] | [11C]-Raclopride (+placebo) | D2/D3 BPND | CAU, PUT, vSTR | 5:3 | OB | 36.5(9.4) | 31–59 | 41.8(8.9) | 28–56 | Fasted (overnight) | No nicotine use >1 pack/day | Secondary | BMI: N.S. |
| Karlsson et al., 2015 [37] | [11C]-Raclopride | D2/D3 BPND | WB + dCAU, PUT, vSTR | 14:0 | NW | 22.7(2.9) | 44.9(12.9) | Fasted (2 h before scan) | None | Primary 2 | OB = NW | ||
| 13:0 | OB | 41.9(3.9) | 37.1–49.3 | 39.1(10.7) | 5 | ||||||||
| Tuominen et al., 2015 [38] | [11C]-Raclopride | D2/D3 BPND | CAU, PUT, vSTR | 20:0 | NW | 22.4(2.6) | 42.0(13.2) | Fasted (2 h before scan) | None | Secondary 2 | OB = NW | ||
| 25:0 | OB | 41.3(4.1) | 41.2(9.2) | 8 | |||||||||
| Cho et al., 2015 [39] | [11C]-Raclopride | D2/D3 BPND | dPUT, dCAU, vSTR | 0:25 | NW-OW | 22.0(2.5) | 17.6–28.0 | 23.3(2.9) | 18–29 | 5 | Primary | BMI: N.S. (Asymmetry Index in dPUT↑) | |
| Caravaggio et al., 2015 [40] | [11C]-Raclopride | D2/D3 BPND | dCAU, dPUT, vSTR | 14:21 | NW-OW | 23.2(2.7) | 18.6–27.8 | 31.3(9.0) | 20–47 | Not fasted | None | Primary | BMI: N.S. |
| [11C]-(+)-PHNO | D2/D3 BPND | dCAU, dPUT, vSTR | 7:19 | NW-OW | 23.7(3.0) | 18.6–27.8 | 29.9(7.4) | 20–45 | Not fasted | None | Primary | BMI: vSTR↑ | |
| Cosgrove et al., 2015[41] | [11C]-(+)-PHNO | D2/D3 BPND | dCAU | 2:10 | NW-OB | 20.8–36.5 | 19–37 | None | Secondary | BMI: right dCAU↑ | |||
| Gaiser et al., 2016 [42] | [11C]-(+)-PHNO | D2/D3 BPND | CAU, PAL, PUT, MB (SN/ VTA), vSTR | 4:10 | NW | 22.3(1.8) | 18.5–24.9 | 34.9(10.2) | Fasted (overnight) | None | Primary | OB > NW in vSTR, PAL, MB BMI (with OW): vSTR, PAL, MB↑ | |
| 1:13 | OW | 27.2 (1.3) | 25.0–29.9 | 36.7(11.5) | 1 | ||||||||
| 4:10 | OB | 35.5(4.5) | >29.9 | 37.0(10.1) | None | ||||||||
| Dunn et al., 2012 [43] | [18F]-Fallypride | D2/D3 BPND | CAU, PUT, vSTR, SN | 8:0 | NW | 23(2) | <25 | 40(9) | Fasted (8 h before scanning) | None | Secondary | OB > NW in CAU | |
| 14:0 | OB | 40(5) | >30 | 40(8) | |||||||||
| Guo et al., 2014 [44] | [18F]-Fallypride | D2/D3 BPND | WB + CAU, PUT, vmSTR | 11:12 | NW + OW | 22.4 CI (21.3,23.5) | >18 | 28 CI (25.1, 30.4) | 18–45 | Fed (standardized breakfast 2 h before scan) | None | Primary | BMI: dSTR↑; vmSTR↓ |
| 10:10 | OB | 36.1 CI (34.9,38.3) | <45 | 35 CI (31.9, 38.8) | 18–45 | ||||||||
| Kessler et al., 2014 [45] | [18F]-Fallypride (baseline) | D2/D3 BPND | CAU, PUT, vSTR, SN/vMB | 15:18 | NW-OB | 24.8 | 19–35 | 25.8 | 18–35 | Primary | BMI: CAU↓(borderline significant) | ||
| Dang et al., 2017 [46] | [18F]-Fallypride | D2/D3 BPND | WB + CAU, PUT, vSTR, MB | 72:58 | NW-OB | 25.5(4.8) | 18.5–40 | 35.6(18.2) | 18–81 | None | Primary | BMI (controlled for age and gender): PUT↑, (above 30 years old only): all ROI↑ | |
| Eisenstein et al., 2013 [47] | [11C]-NMB | D2 BPND | WB + CAU, PUT, vSTR | 11:4 | NW + OW | 22.6(2.2) | 18.9–27.7 | 29.7(5.6) | 22–40 | Not fasted, not fed | None | Primary 3 | OB = NW + OW, BMI: N.S. |
| 12:3 | OB | 40.3(4.9) | 33.2–47.0 | 32.5(5.9) | 25–41 | ||||||||
| Eisenstein et al., 2015 [48] | [11C]-NMB | D2 BPND | dSTR (CAU + PUT), vSTR (Nac), MB | 13:4 | NW + OW | 22.1(2.0) | 18.7–25.9 | 28.5(5.5) | 21–39 | Not fasted, not fed | None | Secondary 3 | OB = NW + OW, BMI: N.S. |
| 19:3 | OB | 39.6(5.2) | 33.4–51.0 | 31.4(6.3) | 23–40 | ||||||||
| Reference | Imaging | Sample | Primary/Secondary Analysis | Findings | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | BMI | Age | Fasting | Smoking | |||||||||
| Radiotracer | DV + manipulation | ROI | F:M | Groups | M(SD) | Range | M(SD) | Range | |||||
| SPECT studies | |||||||||||||
| van de Giessen et al., 2014 [31] | [123I]-IBZM | ΔD2/D3 BPND 0.3 mg/kg d-amphetamine vs. baseline (oral) | STR | 15:0 | NW | 21.8(1.8) | 18.5–24.9 | 38.5(5.6) | 18–45 | Fed (standard breakfast) | None | Primary | OB = NW |
| 15:0 | OB | 42.9(4.9) | 36.3–56.6 | 36.3(4.0) | 18–45 | ||||||||
| PET studies | |||||||||||||
| Haltia et al., 2007 [33] | [11C]-Raclopride | ΔD2/D3 BPND 300 mg/kg glucose vs. placebo (intravenous) | WB; CAU, PUT | 6:6 | NW | 22(1) | <24 | ±25.5(4.5) | Fasted (overnight) | No heavy smokers | Primary | OW + OB = NW | |
| 6:6 | OW + OB | 33(5) | >27 | ±25(2.5) | |||||||||
| Haltia et al., 2008 [49] | [11C]-Raclopride | ΔD2/D3 BPND expected glucose (placebo) vs. placebo (intravenous) * | WB; CAU, PUT, vSTR | 6:6 | NW | 22(1) | <24 | ±25.5(4.5) | Fasted (overnight) | No heavy smokers | Primary | OW + OB = NW | |
| 6:6 | OW + OB | 33(5) | >27 | ±25(2.5) | |||||||||
| Wang et al., 2011 [36] | [11C]-Raclopride | ΔD2/D3 BPND 20 mg. methylphenidate vs. placebo (oral) | CAU, PUT, vSTR | 5:3 | OB | 35.5(9.4) | 31–59 | 41.8(8.9) | 28–56 | Fasted (overnight) | No nicotine use >1 pack/day | Secondary | BMI: N.S. (but interaction between MPH and food stimulation in BED group) |
| ΔD2/D3 BPND food vs. neutral stimulation (view, smell, taste (not eat)) | |||||||||||||
| Wang et al., 2014 [50] | [11C]-Raclopride | ΔD2/D3 BPND 75 g glucose drink vs. sucralose drink (oral) | WB; vSTR | 19 F + M | NW-OB | 21–35 | 40–60 | Fasted (overnight) | No nicotine dependence | Primary | BMI: vSTR↑, with DA increase (decrease) for lower (higher) BMI | ||
| Kessler et al., 2014 [45] | [18F]-Fallypride | ΔD2/D3 BPND 0.42 mg/kg d-amphetamine vs. baseline (oral) | CAU, PUT, vSTR, SN | 8:8 | NW-OB | 25.2 | 19–35 | 24.3 | 21–32 | BMI: rPUT, lSN↑ | |||
| Reference | Imaging | Sample | Primary/Secondary Analysis | Findings | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | BMI | Age | Fasting | Smoking | |||||||||
| Radiotracer | DV | ROI | F:M | Groups | M(SD) | Range | M(SD) | Range | |||||
| Wilcox et al., 2010 [51] | [18F]-FMT | Ki | dCAU, dPUT, vSTR | 9:6 | NW-OB | 25.3 | ±19.0–33.0 | 22.9 | 20–30 | No regular smokers | Primary | BMI: dCAU↓ | |
| Wallace et al., 2014 [52] | [18F]-FMT | Ki | dSTR (CAU) | 8:8 | NW-OB | 20.2–33.4 | 20–30 | Fed (light meal before scan) | Primary | BMI: rCAU↓ | |||
| Lee et al., 2018 [53] | [18F]-FDOPA | EDVR = kocc/kloss | CAU, PUT, vSTR | 11:49 | NW-OB | 25.2(3.3) | 19.2–36.6 | 36.4(3.8) | 30–43 | Fed (no protein-containing foods on scan day) | n = 18 | Primary | EDVR: BMI↓ (all ROI) kocc: BMI↓ (vSTR + PUT) kloss: BMI ↑(vSTR + PUT) |
| Reference | Imaging | Sample | Primary/Secondary Analysis | Findings | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | BMI | Age | Fasting | Smoking | |||||||||
| Radiotracer | DV | ROI | F:M | Groups | M(SD) | Range | M(SD) | Range | |||||
| SPECT studies | |||||||||||||
| Chen et al., 2008 [54] | [99mTc]- TRODAT-1 | ROI/ REF | STR | 27:23 | NW-OB | 23.0 | 18.7–30.6 | 30.3 | 20–57 | No exclusion criterion | Primary | BMI: STR↓ | |
| Koskela et al., 2008 [55] | [123I]-nor-β-CIT | BP | STR | 15:16 | NW-OB | 25.6(3.5) | 19.1–31.9 | 25.4(1.3) | 24–27 | Fasted (overnight) | 2 | Primary | BMI: N.S. Low vs. high BMI twins: N.S. |
| Thomsen et al., 2013 [56] | [123I]-PE2I | BP | STR (CAU + PUT) | 6:6 | NW | 22.7 (1.4) | 21–24.5 | 48(13.9) | 28–69 | Primary | OB = OW = NW BMI: N.S. | ||
| 4:5 | OW | 26.9 (1.6) | 25.3–29.7 | 59.8(8.2) | 46–71 | ||||||||
| 6:6 | OB | 38.5(5.7) | 30.9–49.5 | 44.3(12.3) | 29–59 | ||||||||
| van de Giessen et al., 2013 [57] | [123I]-FP-CIT | BP | STR (CAU + PUT) | 56:67 | NW-OB | 25.2(3.8) | 18.2–41.1 | 53.3(18.3) | 20–83 | Not fasted, not fed | Primary | BMI: N.S. | |
| Versteeg et al., 2016 [58] | [123I]-FP-CIT | BP | STR | 8:0 | NW | 21.3(1.3) | 30.9 (10.5) | Fasted (overnight) | None | Secondary | OB = NW BMI: N.S. | ||
| 10:2 | OB | 36.6(4.4) | 31.7(8.9) | ||||||||||
| Nam et al., 2018 [59] | [123I]-FP-CIT | BP | STR (CAU + PUT) | 51:91 | NW-OW | 25.1(2.8) | 61.9(11.4) | >30 | Primary | BMI: N.S. | |||
| 13:27 | OB | 32.9(3) | 58.2(10.1) | >30 | |||||||||
| PET studies | |||||||||||||
| Pak et al., 2020 [60] | [18F]-FP-CIT | BPND (placebo) | PUT, CAU, vSTR | 0: 33 | NW-OB | 23.1(2.2) | <35 | 24.5(2.8) | 20–31 | Fasted (overnight) | No heavy smokers | Primary | BMI: N.S. |
| ΔBPND vs. 300 mg/kg glucose | Primary | BMI: N.S. (BMI: glucose BPND in vSTR↓) | |||||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Janssen, L.K.; Horstmann, A. Molecular Imaging of Central Dopamine in Obesity: A Qualitative Review across Substrates and Radiotracers. Brain Sci. 2022, 12, 486. https://doi.org/10.3390/brainsci12040486
Janssen LK, Horstmann A. Molecular Imaging of Central Dopamine in Obesity: A Qualitative Review across Substrates and Radiotracers. Brain Sciences. 2022; 12(4):486. https://doi.org/10.3390/brainsci12040486
Chicago/Turabian StyleJanssen, Lieneke Katharina, and Annette Horstmann. 2022. "Molecular Imaging of Central Dopamine in Obesity: A Qualitative Review across Substrates and Radiotracers" Brain Sciences 12, no. 4: 486. https://doi.org/10.3390/brainsci12040486
APA StyleJanssen, L. K., & Horstmann, A. (2022). Molecular Imaging of Central Dopamine in Obesity: A Qualitative Review across Substrates and Radiotracers. Brain Sciences, 12(4), 486. https://doi.org/10.3390/brainsci12040486






