Personalized Frequency Modulated Transcranial Electrical Stimulation for Associative Memory Enhancement
Abstract
:1. Introduction
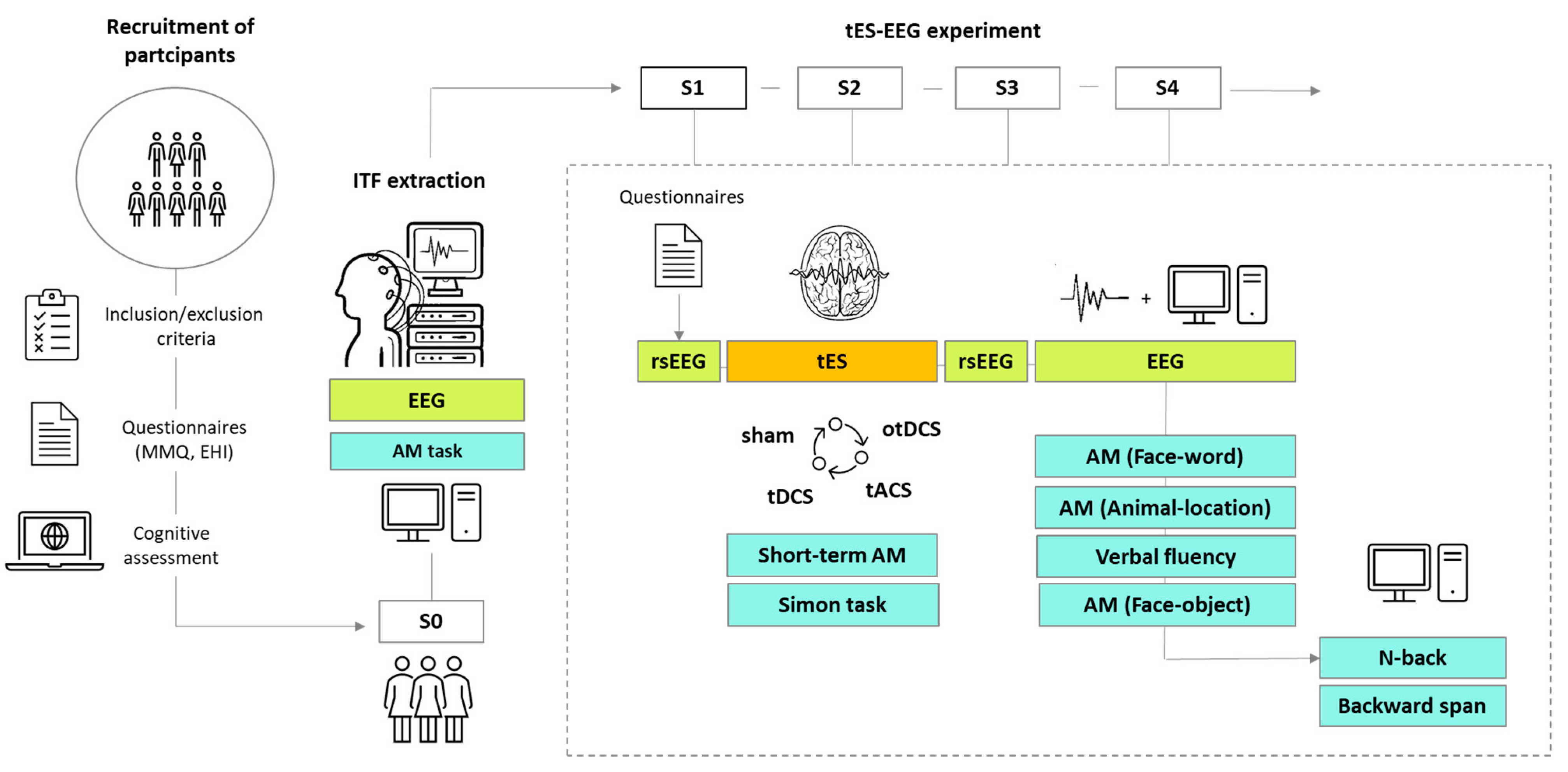
2. Materials and Methods
2.1. Selection and Recruitment of Participants
2.2. Experimental Set-Up and Equipment
2.3. S0: EEG Recording for Initial AM Assessment and ITF Extraction
2.4. S1–S4: The Assessment of Neurophysiological and Behavioral Effects of tES
2.4.1. Counterbalancing
2.4.2. Procedure
2.4.3. Questionnaires
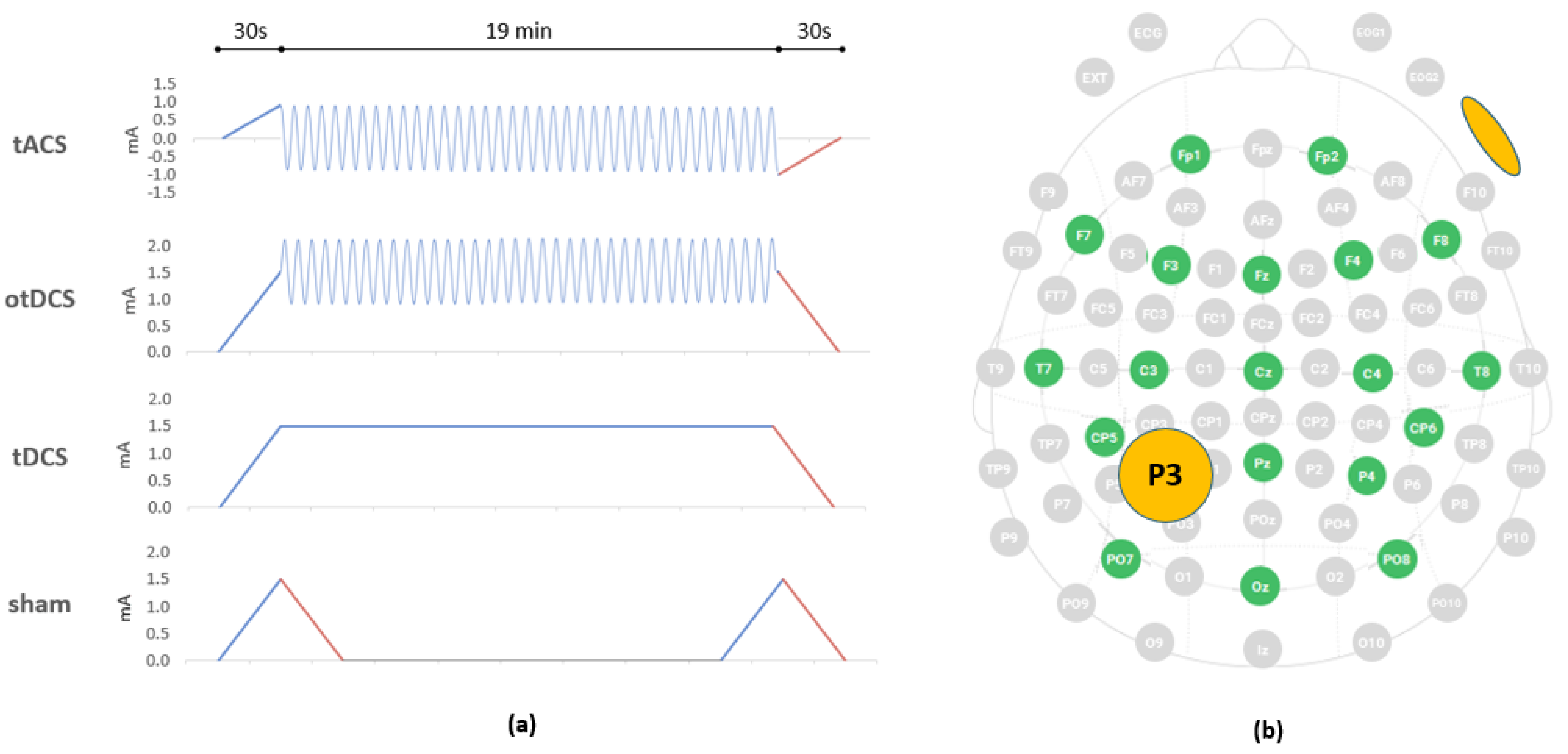
2.4.4. Stimulation Protocols: tACS, otDCS, tDCS and Sham
2.4.5. EEG-tES Electrode Montage
2.4.6. EEG Acquisition Protocols
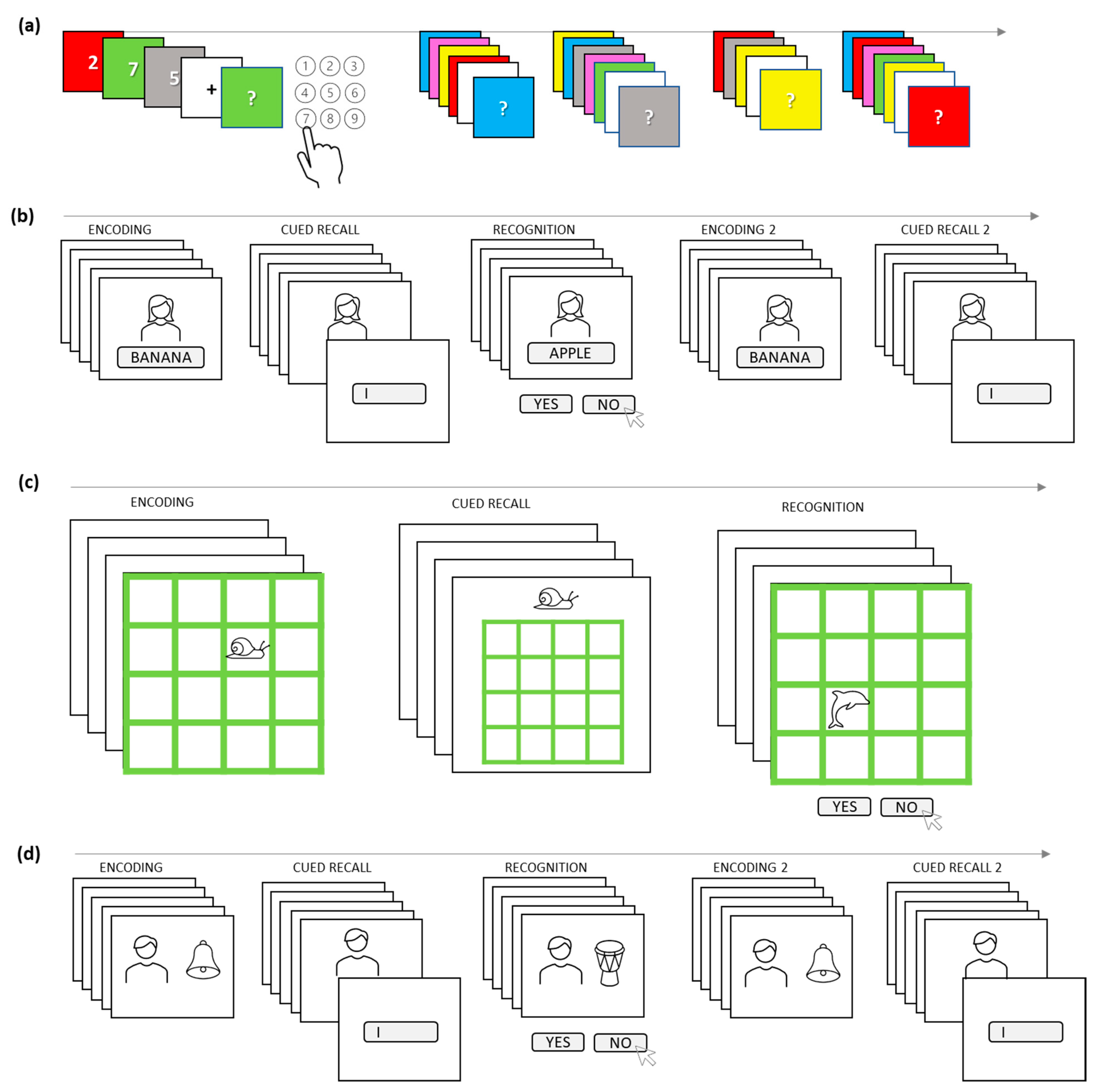
2.4.7. Associative Memory (AM) Tasks
2.4.8. Other Cognitive Tasks
2.4.9. Parallel Forms of the Tasks
2.4.10. Successfulness of Blinding Assessment
2.5. Data and Analytical Approach
- This experiment will generate a relatively large set of neurophysiological and behavioral data, including: Baseline cognitive measures of fluid intelligence (Gf), crystalized intelligence (Gc), visual processing (Gv), and processing speed (Gs);
- Self-report memory assessment (scores for memory satisfaction, everyday memory mistakes, and memory strategies);
- Resting state EEG (eyes-closed and eyes-open from S0, pre-tES and post-tES in S1–S4);
- Short-term AM measures during each tES condition (overall accuracy, success rate for different sequence lengths, recall times);
- Face–word AM measures following each tES condition (associative recognition–overall success rate, hit/miss scores and indexes such as d’, cued recall success rate, false recall, learning slope between two cued-recall blocks, reaction times);
- Face–object AM measures following each tES condition (associative recognition success rates and indices, cued recall success rates, reaction times);
- Animal–location AM measures following each tES condition (cued location memory success rate, assistive recognition rates and indices, reaction times);
- Task-evoked EEG data following each tES condition for three AM tasks (EEG activity during encoding, recall, recognition, repeated encoding);
- Measures of WM following each tES condition–3-back task (success rates and indices, reaction times) and Backward span (WM span and total score);
- Verbal fluency following each tES condition (number of produced words);
- Simon task-measures during each tES condition (reaction speed, error rate, congruency effect)
- pre-tES measures of depression, anxiety, stress, and overall participants’ state.
3. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Disclaimer
References
- Suzuki, W.A. Making New Memories: The Role of the Hippocampus in New Associative Learning. Ann. N. Y. Acad. Sci. 2007, 1097, 1–11. [Google Scholar] [CrossRef]
- Staresina, B.P. Differential Encoding Mechanisms for Subsequent Associative Recognition and Free Recall. J. Neurosci. 2006, 26, 9162–9172. [Google Scholar] [CrossRef] [Green Version]
- Staresina, B.P.; Davachi, L. Selective and Shared Contributions of the Hippocampus and Perirhinal Cortex to Episodic Item and Associative Encoding. J. Cogn. Neurosci. 2008, 20, 1478–1489. [Google Scholar] [CrossRef] [Green Version]
- Herweg, N.A.; Solomon, E.A.; Kahana, M.J. Theta Oscillations in Human Memory. Trends Cogn. Sci. 2020, 24, 208–227. [Google Scholar] [CrossRef]
- Old, S.R.; Naveh-Benjamin, M. Differential Effects of Age on Item and Associative Measures of Memory: A Meta-Analysis. Psychol. Aging 2008, 23, 104–118. [Google Scholar] [CrossRef]
- Chen, P.-C.; Chang, Y.-L. Associative Memory and Underlying Brain Correlates in Older Adults with Mild Cognitive Impairment. Neuropsychologia 2016, 85, 216–225. [Google Scholar] [CrossRef]
- McKhann, G.M.; Knopman, D.S.; Chertkow, H.; Hyman, B.T.; Jack, C.R.; Kawas, C.H.; Klunk, W.E.; Koroshetz, W.J.; Manly, J.J.; Mayeux, R.; et al. The Diagnosis of Dementia Due to Alzheimer’s Disease: Recommendations from the National Institute on Aging-Alzheimer’s Association Workgroups on Diagnostic Guidelines for Alzheimer’s Disease. Alzheimers Dement. 2011, 7, 263–269. [Google Scholar] [CrossRef] [Green Version]
- Kormas, C.; Zalonis, I.; Evdokimidis, I.; Kapaki, E.; Potagas, C. Face–Name Associative Memory Performance Among Cognitively Healthy Individuals, Individuals with Subjective Memory Complaints, and Patients With a Diagnosis of AMCI. Front. Psychol. 2020, 11, 2173. [Google Scholar] [CrossRef]
- Rubiño, J.; Andrés, P. The Face-Name Associative Memory Test as a Tool for Early Diagnosis of Alzheimer’s Disease. Front. Psychol. 2018, 9, 1464. [Google Scholar] [CrossRef] [Green Version]
- Cummings, J.L.; Tong, G.; Ballard, C. Treatment Combinations for Alzheimer’s Disease: Current and Future Pharmacotherapy Options. J. Alzheimers Dis. 2019, 67, 779–794. [Google Scholar] [CrossRef] [Green Version]
- Russ, T.C.; Morling, J.R. Cholinesterase Inhibitors for Mild Cognitive Impairment. Cochrane Database Syst. Rev. 2012, CD009132. [Google Scholar] [CrossRef]
- Phipps, C.J.; Murman, D.L.; Warren, D.E. Stimulating Memory: Reviewing Interventions Using Repetitive Transcranial Magnetic Stimulation to Enhance or Restore Memory Abilities. Brain Sci. 2021, 11, 1283. [Google Scholar] [CrossRef]
- Freitas, C.; Mondragón-Llorca, H.; Pascual-Leone, A. Noninvasive Brain Stimulation in Alzheimer’s Disease: Systematic Review and Perspectives for the Future. Exp. Gerontol. 2011, 46, 611–627. [Google Scholar] [CrossRef] [Green Version]
- Sandrini, M.; Manenti, R.; Sahin, H.; Cotelli, M. Effects of Transcranial Electrical Stimulation on Episodic Memory in Physiological and Pathological Ageing. Ageing Res. Rev. 2020, 61, 101065. [Google Scholar] [CrossRef]
- Fregni, F.; El-Hagrassy, M.M.; Pacheco-Barrios, K.; Carvalho, S.; Leite, J.; Simis, M.; Brunelin, J.; Nakamura-Palacios, E.M.; Marangolo, P.; Venkatasubramanian, G.; et al. Evidence-Based Guidelines and Secondary Meta-Analysis for the Use of Transcranial Direct Current Stimulation in Neurological and Psychiatric Disorders. Int. J. Neuropsychopharmacol. 2021, 24, 256–313. [Google Scholar] [CrossRef] [PubMed]
- Nitsche, M.A.; Paulus, W. Transcranial Direct Current Stimulation—Update 2011. Restor. Neurol. Neurosci. 2011, 29, 463–492. [Google Scholar] [CrossRef] [PubMed]
- Lefaucheur, J.-P.; Aleman, A.; Baeken, C.; Benninger, D.H.; Brunelin, J.; Di Lazzaro, V.; Filipović, S.R.; Grefkes, C.; Hasan, A.; Hummel, F.C.; et al. Evidence-Based Guidelines on the Therapeutic Use of Repetitive Transcranial Magnetic Stimulation (RTMS): An Update (2014–2018). Clin. Neurophysiol. 2020, 131, 474–528. [Google Scholar] [CrossRef]
- Lefaucheur, J.-P.; Antal, A.; Ayache, S.S.; Benninger, D.H.; Brunelin, J.; Cogiamanian, F.; Cotelli, M.; De Ridder, D.; Ferrucci, R.; Langguth, B.; et al. Evidence-Based Guidelines on the Therapeutic Use of Transcranial Direct Current Stimulation (TDCS). Clin. Neurophysiol. 2017, 128, 56–92. [Google Scholar] [CrossRef]
- Giovanello, K.S.; Schnyer, D.M.; Verfaellie, M. A Critical Role for the Anterior Hippocampus in Relational Memory: Evidence from an FMRI Study Comparing Associative and Item Recognition. Hippocampus 2004, 14, 5–8. [Google Scholar] [CrossRef]
- Eichenbaum, H. The Hippocampus and Declarative Memory: Cognitive Mechanisms and Neural Codes. Behav. Brain Res. 2001, 127, 199–207. [Google Scholar] [CrossRef]
- Mayes, A.; Montaldi, D.; Migo, E. Associative Memory and the Medial Temporal Lobes. Trends Cogn. Sci. 2007, 11, 126–135. [Google Scholar] [CrossRef]
- Eichenbaum, H. A Cortical-Hippocampal System for Declarative Memory. Nat. Rev. Neurosci. 2000, 1, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Galli, G.; Vadillo, M.A.; Sirota, M.; Feurra, M.; Medvedeva, A. A Systematic Review and Meta-Analysis of the Effects of Transcranial Direct Current Stimulation (TDCS) on Episodic Memory. Brain Stimulat. 2019, 12, 231–241. [Google Scholar] [CrossRef]
- Kim, K.; Ekstrom, A.D.; Tandon, N. A Network Approach for Modulating Memory Processes via Direct and Indirect Brain Stimulation: Toward a Causal Approach for the Neural Basis of Memory. Neurobiol. Learn. Mem. 2016, 134, 162–177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manenti, R.; Cotelli, M.; Robertson, I.H.; Miniussi, C. Transcranial Brain Stimulation Studies of Episodic Memory in Young Adults, Elderly Adults and Individuals with Memory Dysfunction: A Review. Brain Stimulat. 2012, 5, 103–109. [Google Scholar] [CrossRef] [Green Version]
- Karabanov, A.N.; Saturnino, G.B.; Thielscher, A.; Siebner, H.R. Can Transcranial Electrical Stimulation Localize Brain Function? Front. Psychol. 2019, 10, 213. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.X.; Rogers, L.M.; Gross, E.Z.; Ryals, A.J.; Dokucu, M.E.; Brandstatt, K.L.; Hermiller, M.S.; Voss, J.L. Targeted Enhancement of Cortical-Hippocampal Brain Networks and Associative Memory. Science 2014, 345, 1054–1057. [Google Scholar] [CrossRef] [Green Version]
- Tambini, A.; Nee, D.E.; D’Esposito, M. Hippocampal-Targeted Theta-Burst Stimulation Enhances Associative Memory Formation. J. Cogn. Neurosci. 2018, 30, 1452–1472. [Google Scholar] [CrossRef] [PubMed]
- Hermiller, M.S.; VanHaerents, S.; Raij, T.; Voss, J.L. Frequency-specific Noninvasive Modulation of Memory Retrieval and Its Relationship with Hippocampal Network Connectivity. Hippocampus 2018, 29, 595–609. [Google Scholar] [CrossRef]
- Nilakantan, A.S.; Bridge, D.J.; Gagnon, E.P.; VanHaerents, S.A.; Voss, J.L. Stimulation of the Posterior Cortical-Hippocampal Network Enhances Precision of Memory Recollection. Curr. Biol. 2017, 27, 465–470. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.X.; Voss, J.L. Long-Lasting Enhancements of Memory and Hippocampal-Cortical Functional Connectivity Following Multiple-Day Targeted Noninvasive Stimulation: Long-Lasting Memory and Hippocampal-Network Enhancements. Hippocampus 2015, 25, 877–883. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hermiller, M.S.; Dave, S.; Wert, S.L.; VanHaerents, S.; Riley, M.; Weintraub, S.; Mesulam, M.M.; Voss, J.L. Evidence from Theta-Burst Stimulation That Age-Related de-Differentiation of the Hippocampal Network Is Functional for Episodic Memory. Neurobiol. Aging 2022, 109, 145–157. [Google Scholar] [CrossRef]
- Freedberg, M.; Reeves, J.A.; Toader, A.C.; Hermiller, M.S.; Voss, J.L.; Wassermann, E.M. Persistent Enhancement of Hippocampal Network Connectivity by Parietal rTMS Is Reproducible. eNeuro 2019, 6, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Berryhill, M.E. Insights from Neuropsychology: Pinpointing the Role of the Posterior Parietal Cortex in Episodic and Working Memory. Front. Integr. Neurosci. 2012, 6, 31. [Google Scholar] [CrossRef] [Green Version]
- Levy, D.A. Towards an Understanding of Parietal Mnemonic Processes: Some Conceptual Guideposts. Front. Integr. Neurosci. 2012, 6, 41. [Google Scholar] [CrossRef] [Green Version]
- Wagner, A.D.; Shannon, B.J.; Kahn, I.; Buckner, R.L. Parietal Lobe Contributions to Episodic Memory Retrieval. Trends Cogn. Sci. 2005, 9, 445–453. [Google Scholar] [CrossRef]
- Gilmore, A.W.; Nelson, S.M.; McDermott, K.B. A Parietal Memory Network Revealed by Multiple MRI Methods. Trends Cogn. Sci. 2015, 19, 534–543. [Google Scholar] [CrossRef] [PubMed]
- Uncapher, M.R.; Wagner, A.D. Posterior Parietal Cortex and Episodic Encoding: Insights from FMRI Subsequent Memory Effects and Dual-Attention Theory. Neurobiol. Learn. Mem. 2009, 91, 139–154. [Google Scholar] [CrossRef] [Green Version]
- Bjekić, J.; Čolić, M.V.; Živanović, M.; Milanović, S.D.; Filipović, S.R. Transcranial Direct Current Stimulation (TDCS) over Parietal Cortex Improves Associative Memory. Neurobiol. Learn. Mem. 2019, 157, 114–120. [Google Scholar] [CrossRef]
- Bjekić, J.; Vulić, K.; Živanović, M.; Vujičić, J.; Ljubisavljević, M.; Filipović, S.R. The Immediate and Delayed Effects of Single TDCS Session over Posterior Parietal Cortex on Face-Word Associative Memory. Behav. Brain Res. 2019, 366, 88–95. [Google Scholar] [CrossRef]
- Bjekić, J.; Čolić, M.; Živanović, M.; Milanović, S.; Filipović, S. P302 Right Parietal TDCS and Object-Location Memory. Clin. Neurophysiol. 2017, 128, e275. [Google Scholar] [CrossRef]
- Dubravac, M.; Meier, B. Institute of Psychology, University of Bern, Bern, Switzerland Stimulating the Parietal Cortex by Transcranial Direct Current Stimulation (TDCS): No Effects on Attention and Memory. AIMS Neurosci. 2020, 8, 33–46. [Google Scholar] [CrossRef] [PubMed]
- Čolić, V.M.; Konstantinović, U.; Bjekić, J.; Filipović, R.S. Preliminary Findings of Single Session of Noninvansive Brain Stimulation over Parietal Lobe and Performance on Spatial Memory Task. bioRxiv 2019, 736892. [Google Scholar] [CrossRef]
- Crossman, M.; Bartl, G.; Soerum, R.; Sandrini, M. Effects of Transcranial Direct Current Stimulation over the Posterior Parietal Cortex on Episodic Memory Reconsolidation. Cortex 2019, 121, 78–88. [Google Scholar] [CrossRef]
- Nitsche, M.A.; Paulus, W. Excitability Changes Induced in the Human Motor Cortex by Weak Transcranial Direct Current Stimulation. J. Physiol. 2000, 527, 633–639. [Google Scholar] [CrossRef] [PubMed]
- Antal, A.; Boros, K.; Poreisz, C.; Chaieb, L.; Terney, D.; Paulus, W. Comparatively Weak After-Effects of Transcranial Alternating Current Stimulation (TACS) on Cortical Excitability in Humans. Brain Stimulat. 2008, 1, 97–105. [Google Scholar] [CrossRef]
- Antal, A.; Herrmann, C.S. Transcranial Alternating Current and Random Noise Stimulation: Possible Mechanisms. Neural Plast. 2016, 2016, 3616807. [Google Scholar] [CrossRef] [Green Version]
- Elyamany, O.; Leicht, G.; Herrmann, C.S.; Mulert, C. Transcranial Alternating Current Stimulation (TACS): From Basic Mechanisms towards First Applications in Psychiatry. Eur. Arch. Psychiatry Clin. Neurosci. 2021, 271, 135–156. [Google Scholar] [CrossRef]
- Herrmann, C.S.; Rach, S.; Neuling, T.; Strüber, D. Transcranial Alternating Current Stimulation: A Review of the Underlying Mechanisms and Modulation of Cognitive Processes. Front. Hum. Neurosci. 2013, 7, 279. [Google Scholar] [CrossRef] [Green Version]
- Vulić, K.; Bjekić, J.; Paunović, D.; Jovanović, M.; Milanović, S.; Filipović, S.R. Theta-Modulated Oscillatory Transcranial Direct Current Stimulation over Posterior Parietal Cortex Improves Associative Memory. Sci. Rep. 2021, 11, 3013. [Google Scholar] [CrossRef]
- Klink, K.; Paßmann, S.; Kasten, F.H.; Peter, J. The Modulation of Cognitive Performance with Transcranial Alternating Current Stimulation: A Systematic Review of Frequency-Specific Effects. Brain Sci. 2020, 10, 932. [Google Scholar] [CrossRef]
- Booth, S.J.; Taylor, J.R.; Brown, L.J.E.; Pobric, G. The Effects of Transcranial Alternating Current Stimulation on Memory Performance in Healthy Adults: A Systematic Review. Cortex 2022, 147, 112–139. [Google Scholar] [CrossRef] [PubMed]
- Lang, S.; Gan, L.S.; Alrazi, T.; Monchi, O. Theta Band High Definition Transcranial Alternating Current Stimulation, but Not Transcranial Direct Current Stimulation, Improves Associative Memory Performance. Sci. Rep. 2019, 9, 8562. [Google Scholar] [CrossRef]
- Meng, A.; Kaiser, M.; de Graaf, T.A.; Dücker, F.; Sack, A.T.; De Weerd, P.; van de Ven, V. Transcranial Alternating Current Stimulation at Theta Frequency to Left Parietal Cortex Impairs Associative, but Not Perceptual, Memory Encoding. Neurobiol. Learn. Mem. 2021, 182, 107444. [Google Scholar] [CrossRef] [PubMed]
- Harty, S. Tackling Inter-Individual Variability in Responsiveness to Transcranial Electrical Stimulation. Brain Stimulat. 2019, 12, 391. [Google Scholar] [CrossRef]
- Krause, B.; Cohen Kadosh, R. Not All Brains Are Created Equal: The Relevance of Individual Differences in Responsiveness to Transcranial Electrical Stimulation. Front. Syst. Neurosci. 2014, 8, 25. [Google Scholar] [CrossRef]
- Horvath, J.C.; Vogrin, S.J.; Carter, O.; Cook, M.J.; Forte, J.D. Effects of a Common Transcranial Direct Current Stimulation (TDCS) Protocol on Motor Evoked Potentials Found to Be Highly Variable within Individuals over 9 Testing Sessions. Exp. Brain Res. 2016, 234, 2629–2642. [Google Scholar] [CrossRef] [PubMed]
- Horvath, J.C.; Carter, O.; Forte, J.D. Transcranial Direct Current Stimulation: Five Important Issues We Aren’t Discussing (but Probably Should Be). Front. Syst. Neurosci. 2014, 8, 2. [Google Scholar] [CrossRef] [PubMed]
- Figee, M.; Mayberg, H. The Future of Personalized Brain Stimulation. Nat. Med. 2021, 27, 196–197. [Google Scholar] [CrossRef]
- Lu, H.; Chiu Wa Lam, L.; Ning, Y. Toward Personalized Brain Stimulation: Advances and Challenges. CNS Neurosci. Ther. 2019, 25, 1219–1221. [Google Scholar] [CrossRef] [PubMed]
- van Bueren, N.E.R.; Reed, T.L.; Nguyen, V.; Sheffield, J.G.; van der Ven, S.H.G.; Osborne, M.A.; Kroesbergen, E.H.; Cohen Kadosh, R. Personalized Brain Stimulation for Effective Neurointervention across Participants. PLOS Comput. Biol. 2021, 17, e1008886. [Google Scholar] [CrossRef] [PubMed]
- Thut, G.; Bergmann, T.O.; Fröhlich, F.; Soekadar, S.R.; Brittain, J.-S.; Valero-Cabré, A.; Sack, A.T.; Miniussi, C.; Antal, A.; Siebner, H.R.; et al. Guiding Transcranial Brain Stimulation by EEG/MEG to Interact with Ongoing Brain Activity and Associated Functions: A Position Paper. Clin. Neurophysiol. 2017, 128, 843–857. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mioni, G.; Shelp, A.; Stanfield-Wiswell, C.T.; Gladhill, K.A.; Bader, F.; Wiener, M. Modulation of Individual Alpha Frequency with TACS Shifts Time Perception. Cereb. Cortex Commun. 2020, 1. [Google Scholar] [CrossRef] [PubMed]
- Riddle, J.; Alexander, M.L.; Schiller, C.E.; Rubinow, D.R.; Frohlich, F. Reduction in Left Frontal Alpha Oscillations by Transcranial Alternating Current Stimulation in Major Depressive Disorder Is Context Dependent in a Randomized Clinical Trial. Biol. Psychiatry Cogn. Neurosci. Neuroimagin. 2022, 7, 302–311. [Google Scholar] [CrossRef] [PubMed]
- Pahor, A.; Jaušovec, N. The Effects of Theta and Gamma TACS on Working Memory and Electrophysiology. Front. Hum. Neurosci. 2018, 11, 651. [Google Scholar] [CrossRef] [Green Version]
- Kudo, D.; Koseki, T.; Katagiri, N.; Yoshida, K.; Takano, K.; Jin, M.; Nito, M.; Tanabe, S.; Yamaguchi, T. Individualized Beta-Band Oscillatory Transcranial Direct Current Stimulation over the Primary Motor Cortex Enhances Corticomuscular Coherence and Corticospinal Excitability in Healthy Individuals. Brain Stimulat. 2022, 15, 46–52. [Google Scholar] [CrossRef]
- Jaušovec, N.; Jaušovec, K.; Pahor, A. The Influence of Theta Transcranial Alternating Current Stimulation (TACS) on Working Memory Storage and Processing Functions. Acta Psychol. 2014, 146, 1–6. [Google Scholar] [CrossRef]
- Pahor, A.; Jaušovec, N. The Effects of Theta Transcranial Alternating Current Stimulation (TACS) on Fluid Intelligence. Int. J. Psychophysiol. 2014, 93, 322–331. [Google Scholar] [CrossRef]
- van Driel, J.; Sligte, I.G.; Linders, J.; Elport, D.; Cohen, M.X. Frequency Band-Specific Electrical Brain Stimulation Modulates Cognitive Control Processes. PLoS ONE 2015, 10, e0138984. [Google Scholar] [CrossRef] [Green Version]
- Faul, F.; Erdfelder, E.; Lang, A.-G.; Buchner, A. G*Power 3: A Flexible Statistical Power Analysis Program for the Social, Behavioral, and Biomedical Sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef]
- Cohen, J. A Power Primer. Psychol. Bull. 1992, 112, 155–159. [Google Scholar] [CrossRef] [PubMed]
- McGrew, K.S. CHC Theory and the Human Cognitive Abilities Project: Standing on the Shoulders of the Giants of Psychometric Intelligence Research. Intelligence 2009, 37, 1–10. [Google Scholar] [CrossRef]
- Pallier, G.; Wilkinson, R.; Danthiir, V.; Kleitman, S.; Knezevic, G.; Stankov, L.; Roberts, R.D. The Role of Individual Differences in the Accuracy of Confidence Judgments. J. Gen. Psychol. 2002, 129, 257–299. [Google Scholar] [CrossRef]
- Wolf, B.; Momirović, K.; Džamonja, Z. KOG 3–Baterija Testova Inteligencije [KOG 3–Battery of Intelligence Tests]; Centar za Primenjenu Psihologiju: Beograd, Srbija, 1992. [Google Scholar]
- Živanović, M.; Bjekić, J.; Opačić, G. Multiple Solutions Test—Part II: Evidence on Construct and Predictive Validity. Psihologija 2018, 51, 377–396. [Google Scholar] [CrossRef] [Green Version]
- Troyer, A.K.; Rich, J.B. Psychometric Properties of a New Metamemory Questionnaire for Older Adults. J. Gerontol. Ser. B Psychol. Sci. Soc. Sci. 2002, 57, P19–P27. [Google Scholar] [CrossRef] [Green Version]
- Shaikh, K.T.; Tatham, E.L.; Rich, J.B.; Troyer, A.K. Examining the Factor Structure of the Multifactorial Memory Questionnaire. Mem. Hove Engl. 2021, 29, 255–260. [Google Scholar] [CrossRef]
- Oldfield, R.C. The Assessment and Analysis of Handedness: The Edinburgh Inventory. Neuropsychologia 1971, 9, 97–113. [Google Scholar] [CrossRef]
- Mathôt, S.; Schreij, D.; Theeuwes, J. OpenSesame: An Open-Source, Graphical Experiment Builder for the Social Sciences. Behav. Res. Methods 2012, 44, 314–324. [Google Scholar] [CrossRef] [Green Version]
- Delorme, A.; Makeig, S. EEGLAB: An Open Source Toolbox for Analysis of Single-Trial EEG Dynamics Including Independent Component Analysis. J. Neurosci. Methods 2004, 134, 9–21. [Google Scholar] [CrossRef] [Green Version]
- Mørup, M.; Hansen, L.K.; Arnfred, S.M. ERPWAVELAB: A Toolbox for Multi-Channel Analysis of Time–Frequency Transformed Event Related Potentials. J. Neurosci. Methods 2007, 161, 361–368. [Google Scholar] [CrossRef]
- Bjekić, J.; Paunovic, D.; Živanović, M.; Stanković, M.; Griskova-Bulanova, I.; Filipović, S.R. Extraction of Individual Associative Memory Related Dominant Theta Frequency for Personalized Transcranial Brain Stimulation. BioRxiv Neurosci. 2022. [Google Scholar] [CrossRef]
- Bjekić, J.; Živanović, M.; Filipović, S.R. Transcranial Direct Current Stimulation (TDCS) for Memory Enhancement. J. Vis. Exp. 2021, 62681. [Google Scholar] [CrossRef] [PubMed]
- Lovibond, P.F.; Lovibond, S.H. Manual for the Depression Anxiety Stress Scales; Psychology Foundation: Sydney, Australia, 1995. [Google Scholar]
- Antal, A.; Alekseichuk, I.; Bikson, M.; Brockmöller, J.; Brunoni, A.R.; Chen, R.; Cohen, L.G.; Dowthwaite, G.; Ellrich, J.; Flöel, A.; et al. Low Intensity Transcranial Electric Stimulation: Safety, Ethical, Legal Regulatory and Application Guidelines. Clin. Neurophysiol. 2017, 128, 1774–1809. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sternberg, S. High-Speed Scanning in Human Memory. Science 1966, 153, 652–654. [Google Scholar] [CrossRef] [Green Version]
- Ma, D.S.; Correll, J.; Wittenbrink, B. The Chicago Face Database: A Free Stimulus Set of Faces and Norming Data. Behav. Res. Methods 2015, 47, 1122–1135. [Google Scholar] [CrossRef] [Green Version]
- Ebner, N.C.; Riediger, M.; Lindenberger, U. FACES—A Database of Facial Expressions in Young, Middle-Aged, and Older Women and Men: Development and Validation. Behav. Res. Methods 2010, 42, 351–362. [Google Scholar] [CrossRef]
- Hommel, B. The Simon Effect as Tool and Heuristic. Acta Psychol. 2011, 136, 189–202. [Google Scholar] [CrossRef]
- Živanović, M.; Paunović, D.; Konstantinović, U.; Vulić, K.; Bjekić, J.; Filipović, S.R. The Effects of Offline and Online Prefrontal vs Parietal Transcranial Direct Current Stimulation (tDCS) on Verbal and Spatial Working Memory. Neurobiol. Learn. Mem. 2021, 179, 107398. [Google Scholar] [CrossRef]
- Hill, A.T.; Fitzgerald, P.B.; Hoy, K.E. Effects of Anodal Transcranial Direct Current Stimulation on Working Memory: A Systematic Review and Meta-Analysis of Findings from Healthy and Neuropsychiatric Populations. Brain Stimulat. 2016, 9, 197–208. [Google Scholar] [CrossRef]
- Climie, E.A.; Rostad, K. Test Review: Wechsler Adult Intelligence Scale. J. Psychoeduc. Assess. 2011, 29, 581–586. [Google Scholar] [CrossRef]
- Bjekić, J.; Živanović, M.; Vulić, K.; Paunovic, D.; Konstantinović, U.; Filipović, S. From Brain Waves to Memory Boost: Memory Enhancement by Personalized Frequency-Modulated Noninvasive Brain Stimulation (MEMORYST). JJJJ 2020. [Google Scholar] [CrossRef]
- Alekseichuk, I.; Turi, Z.; Veit, S.; Paulus, W. Model-Driven Neuromodulation of the Right Posterior Region Promotes Encoding of Long-Term Memories. Brain Stimulat. 2020, 13, 474–483. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frohlich, F.; Riddle, J. Conducting Double-Blind Placebo-Controlled Clinical Trials of Transcranial Alternating Current Stimulation (TACS). Transl. Psychiatry 2021, 11, 284. [Google Scholar] [CrossRef] [PubMed]
- Wallace, D.; Cooper, N.R.; Paulmann, S.; Fitzgerald, P.B.; Russo, R. Perceived Comfort and Blinding Efficacy in Randomised Sham-Controlled Transcranial Direct Current Stimulation (TDCS) Trials at 2 MA in Young and Older Healthy Adults. PLoS ONE 2016, 11, e0149703. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stanković, M.; Živanović, M.; Bjekić, J.; Filipović, S.R. Blinding in TDCS Studies: Correct End-of-Study Guess Does Not Moderate the Effects on Associative and Working Memory. Brain Sci. 2021, 12, 58. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bjekić, J.; Živanović, M.; Paunović, D.; Vulić, K.; Konstantinović, U.; Filipović, S.R. Personalized Frequency Modulated Transcranial Electrical Stimulation for Associative Memory Enhancement. Brain Sci. 2022, 12, 472. https://doi.org/10.3390/brainsci12040472
Bjekić J, Živanović M, Paunović D, Vulić K, Konstantinović U, Filipović SR. Personalized Frequency Modulated Transcranial Electrical Stimulation for Associative Memory Enhancement. Brain Sciences. 2022; 12(4):472. https://doi.org/10.3390/brainsci12040472
Chicago/Turabian StyleBjekić, Jovana, Marko Živanović, Dunja Paunović, Katarina Vulić, Uroš Konstantinović, and Saša R. Filipović. 2022. "Personalized Frequency Modulated Transcranial Electrical Stimulation for Associative Memory Enhancement" Brain Sciences 12, no. 4: 472. https://doi.org/10.3390/brainsci12040472
APA StyleBjekić, J., Živanović, M., Paunović, D., Vulić, K., Konstantinović, U., & Filipović, S. R. (2022). Personalized Frequency Modulated Transcranial Electrical Stimulation for Associative Memory Enhancement. Brain Sciences, 12(4), 472. https://doi.org/10.3390/brainsci12040472





