Portable Neuroimaging-Guided Noninvasive Brain Stimulation of the Cortico-Cerebello-Thalamo-Cortical Loop—Hypothesis and Theory in Cannabis Use Disorder
Abstract
:1. Introduction
2. Hypothesis 1: Cerebellar Brain Inhibition in a Competing Neurobehavioral Decision Systems Model
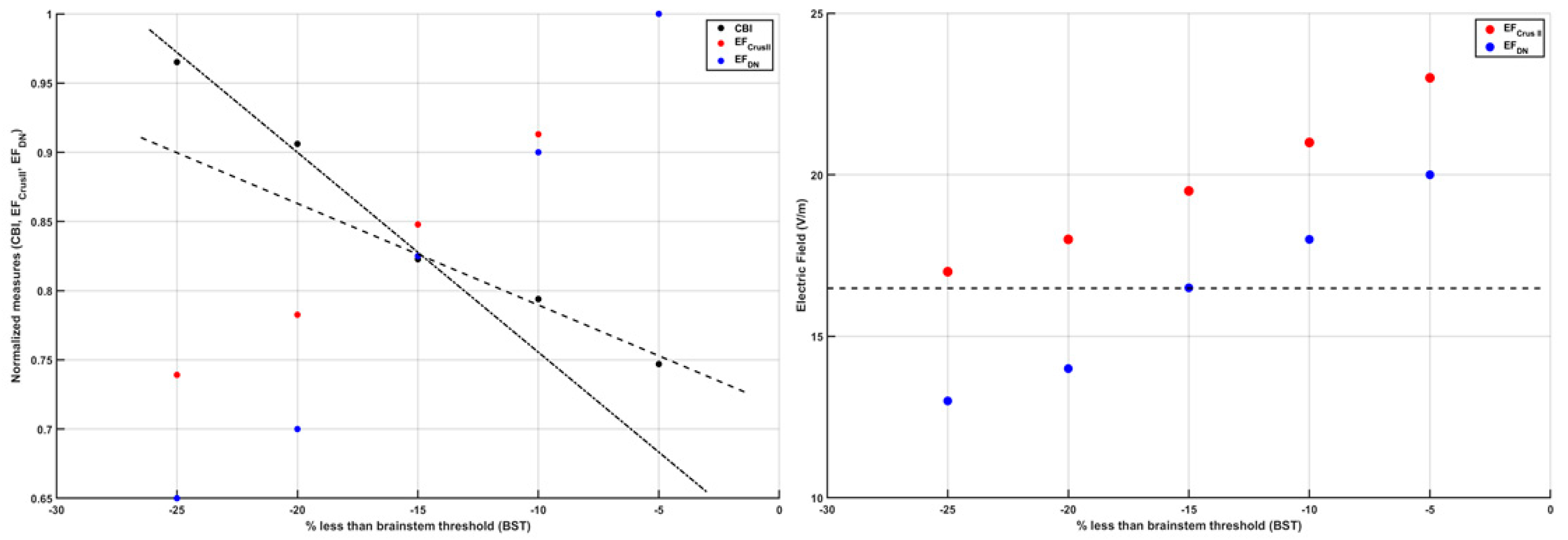
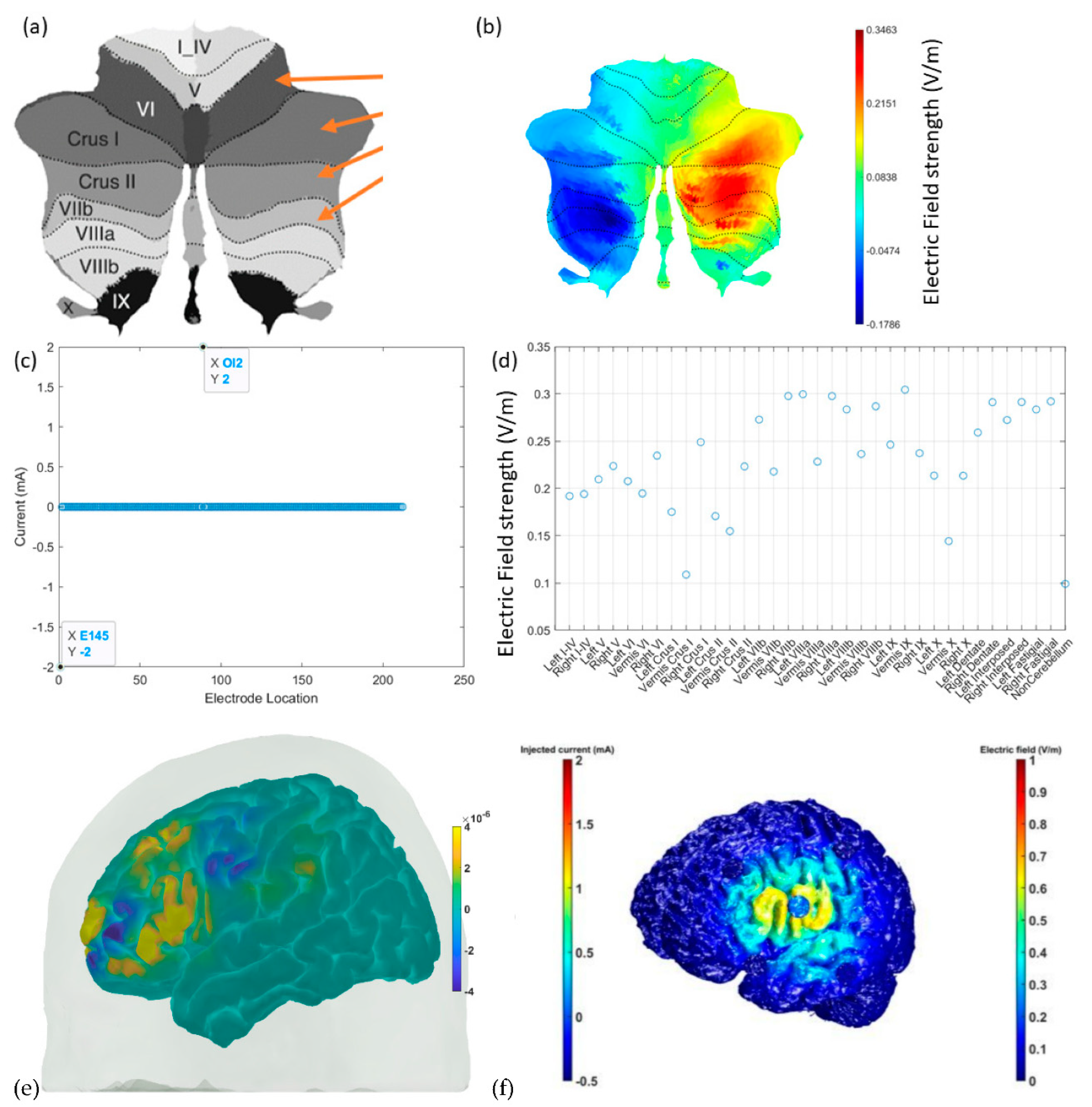
3. Theory 1: Computational Modeling and Simulation of Cerebellar Brain Inhibition Measure
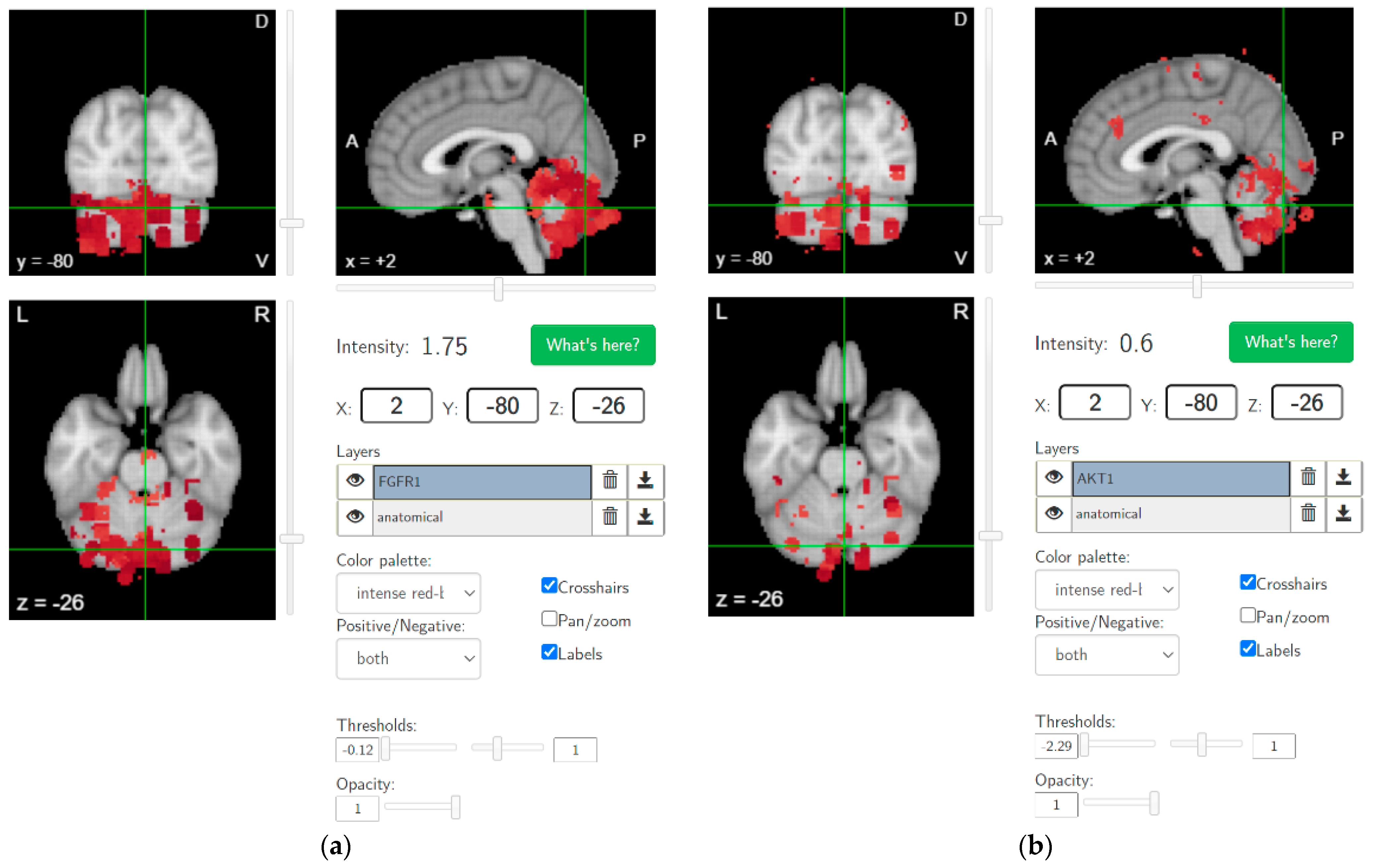
4. Hypothesis 2: Cannabis Use-Related Dysrhythmia in the Cerebellocortical Circuit and Psychotic Disorder
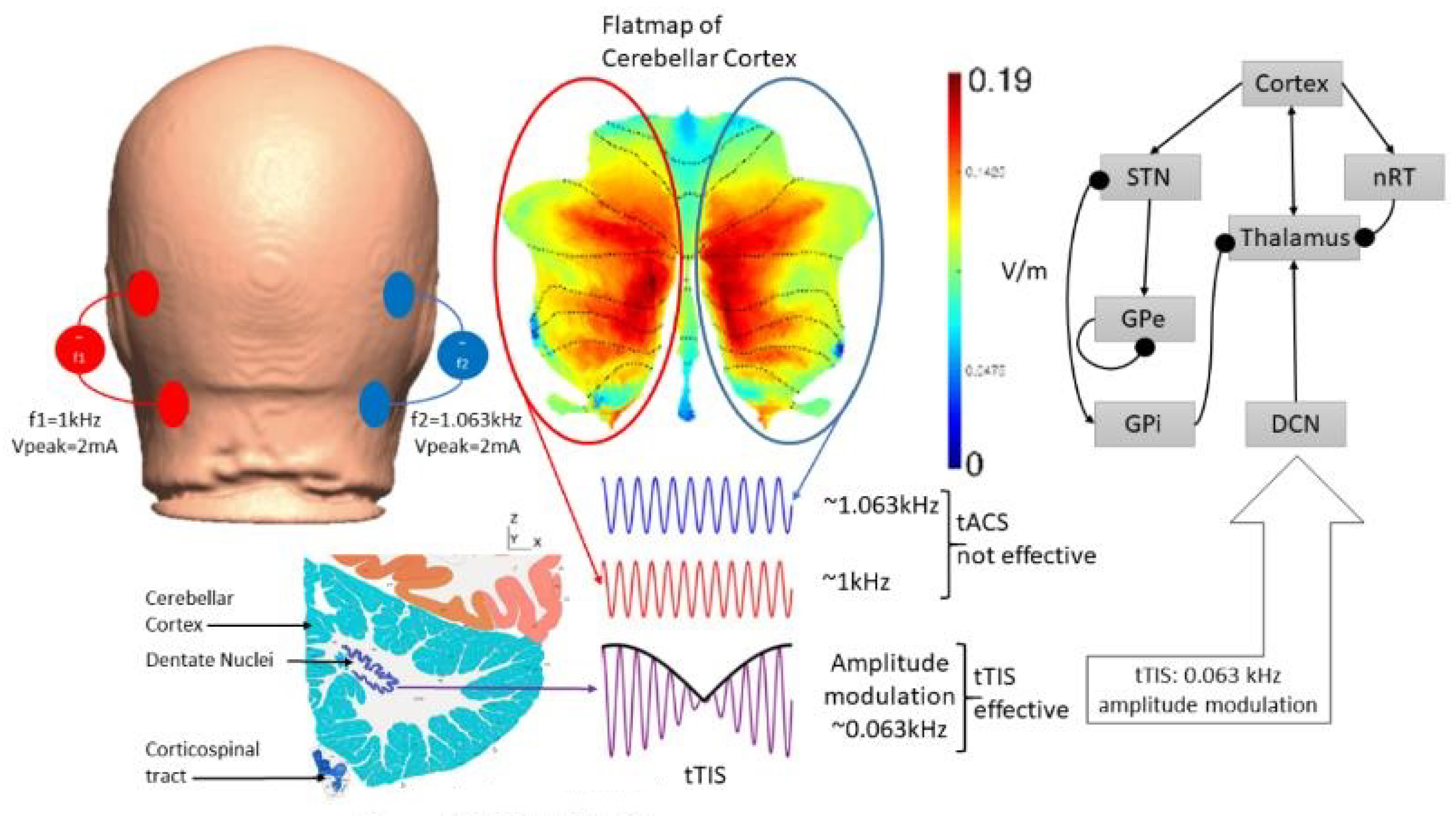
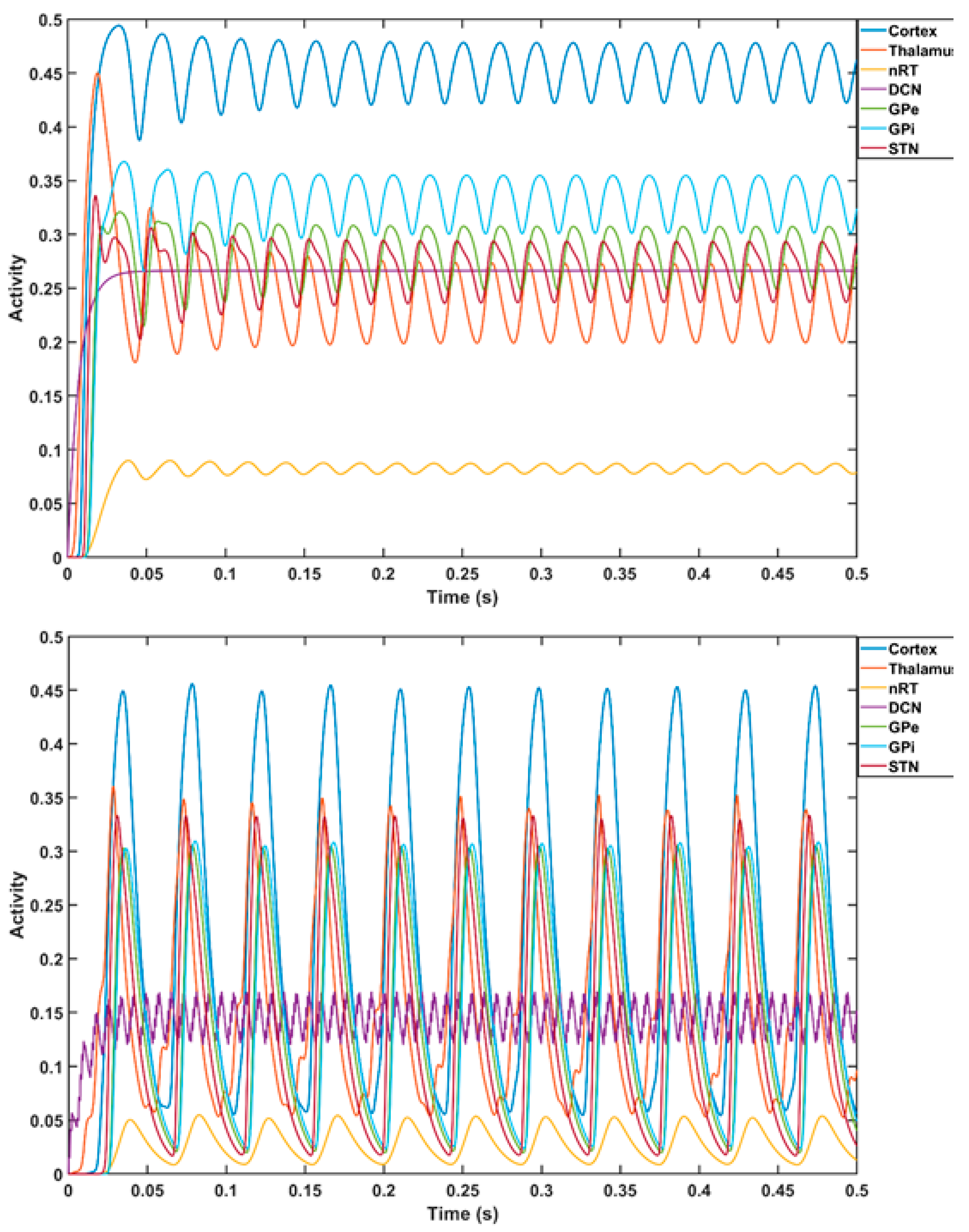
5. Theory 2: Computational Modeling and Simulation of tTIS-Based Amelioration of Dysrhythmia in the Cortico-Cerebello-Thalamo-Cortical Loop
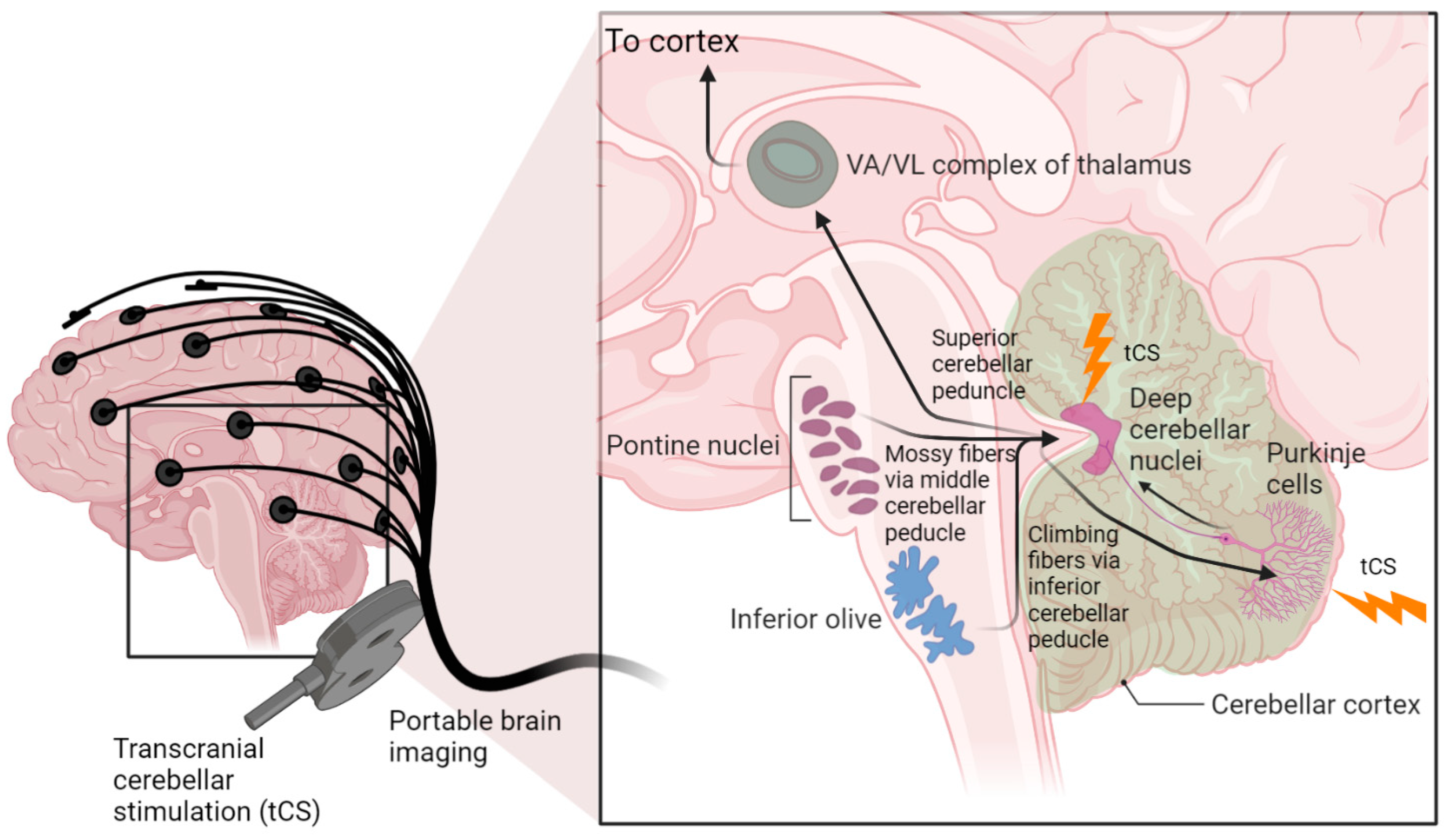
6. Hypothesis 3: Portable Neuroimaging-Guided NIBS to Reduce Inter-Individual Variability
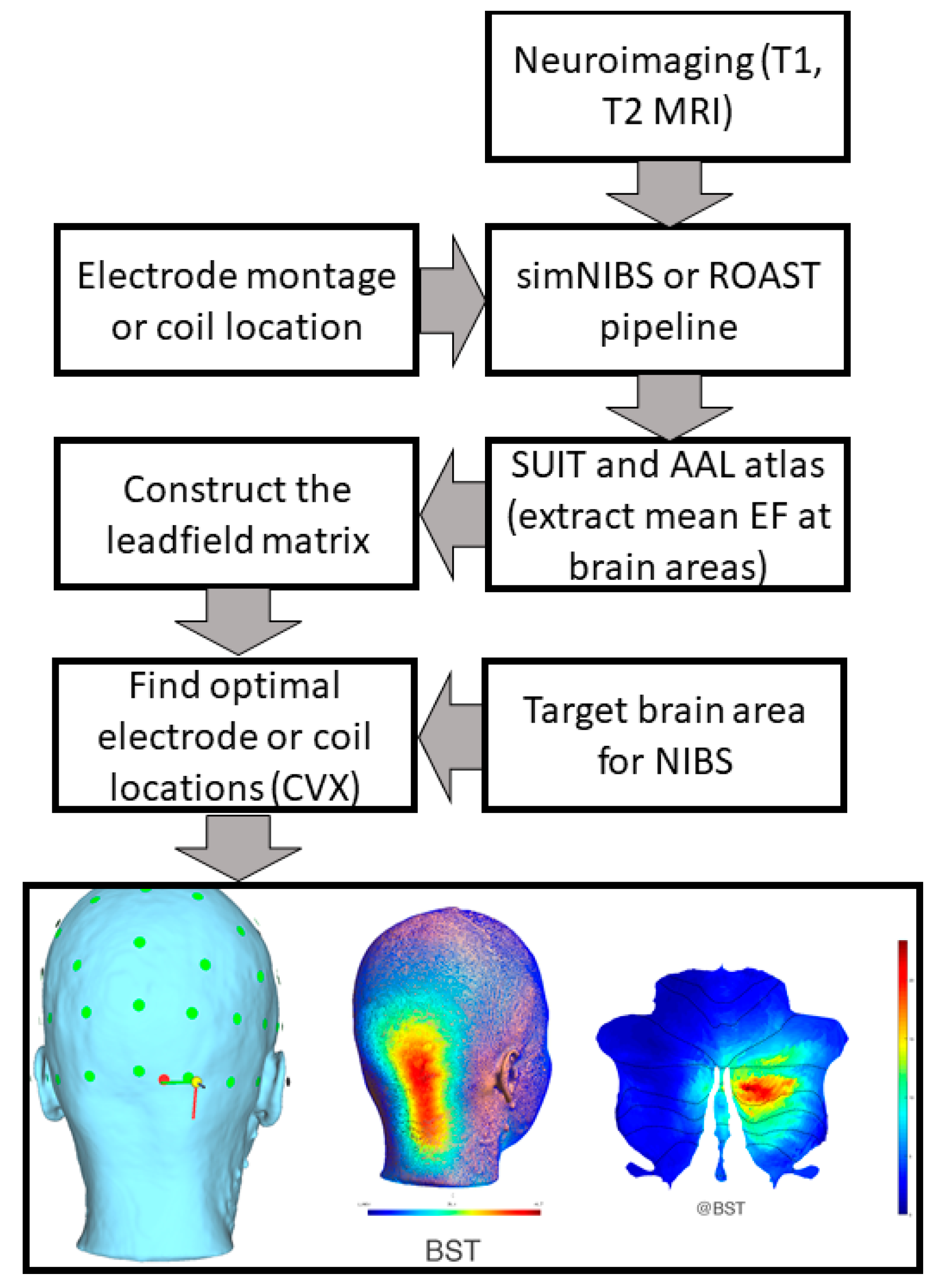
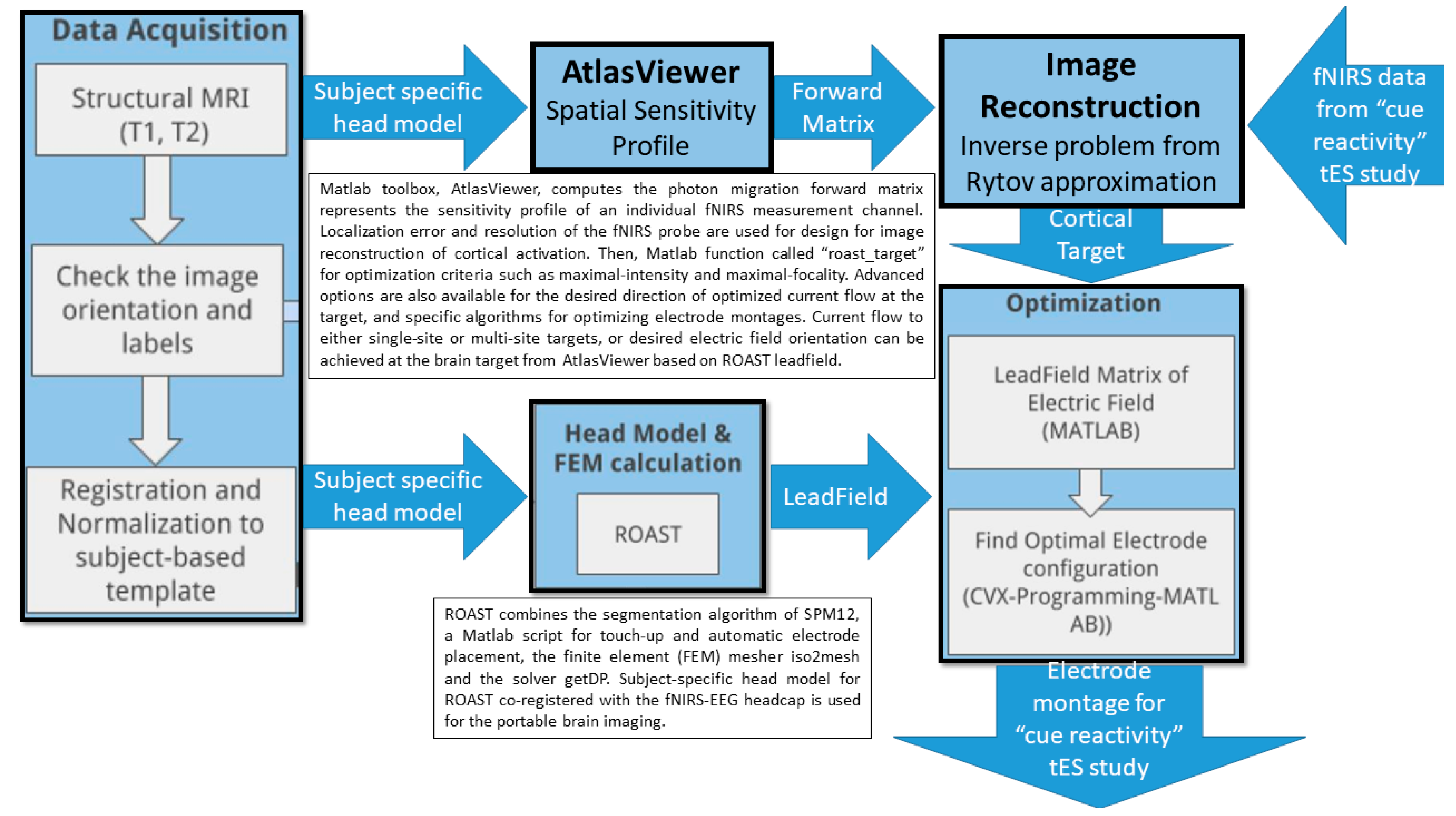
7. Theory 3: Portable Neuroimaging-Guided Subject-Specific NIBS Application
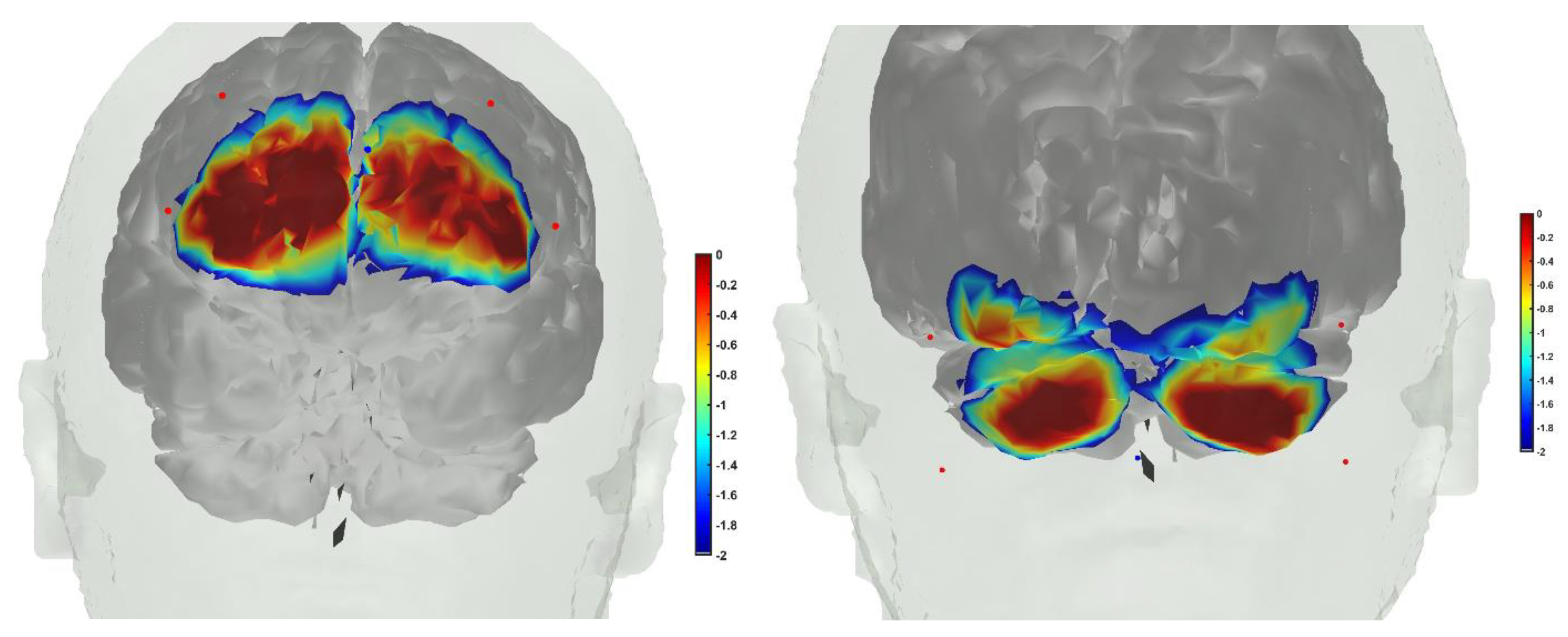
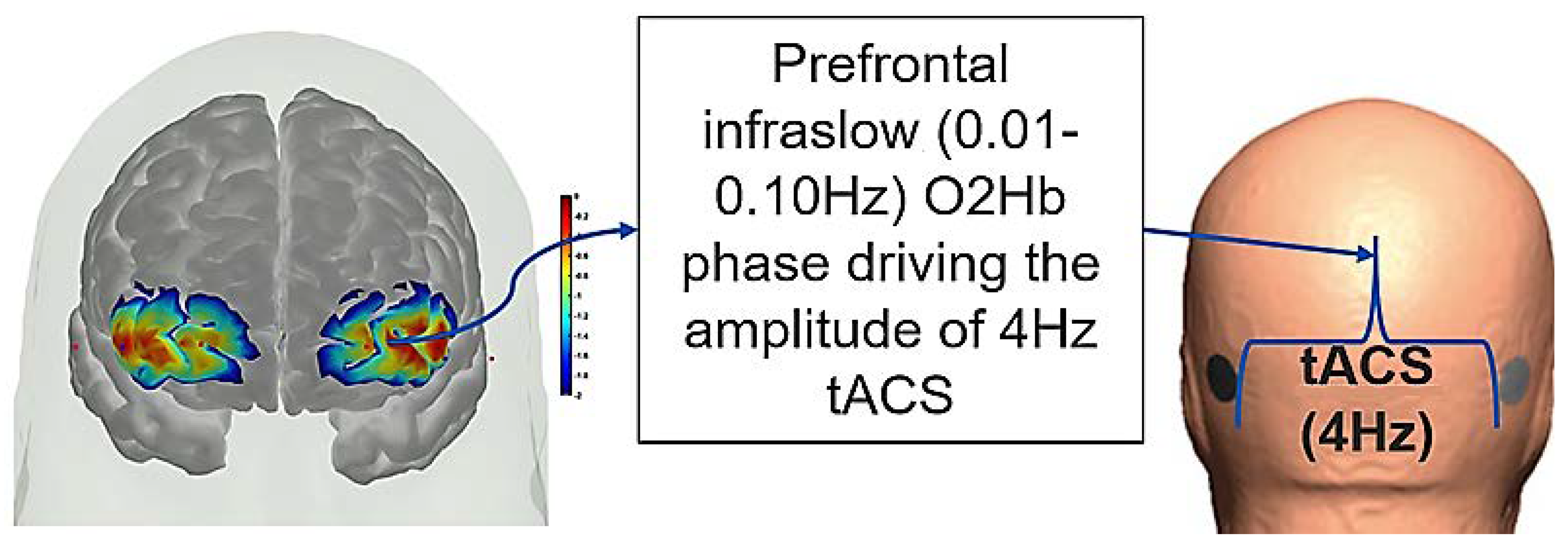
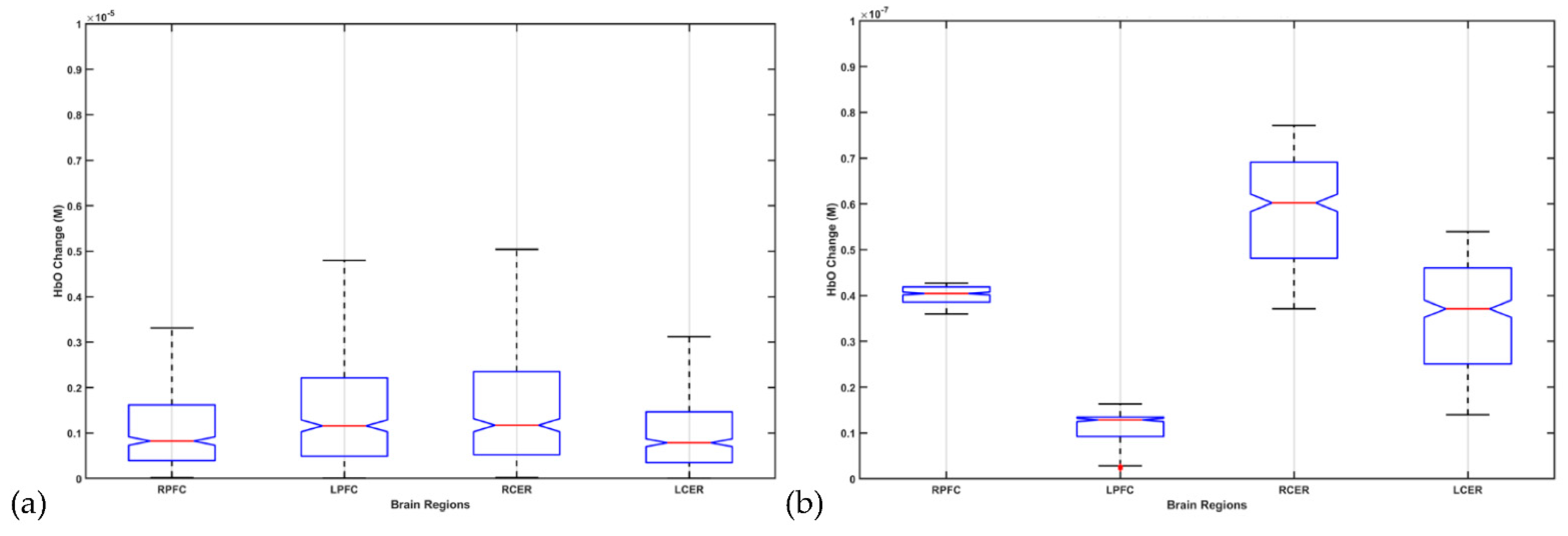
8. Hypothesis 4: Portable Neuroimaging for Online Monitoring and Driving Cerebellar NIBS
9. Theory 4: fNIRS-Driven Cerebellar NIBS
10. Discussion
11. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Substance Abuse and Mental Health Services Administration (US), Office of the Surgeon General (US). The Neurobiology of Substance Use, Misuse, and Addiction. In Facing Addiction in America: The Surgeon General’s Report on Alcohol, Drugs, and Health; US Department of Health and Human Services, Ed.; US Department of Health and Human Services: Washington, DC, USA, 2016. [Google Scholar]
- Windle, M.; Gray, J.C.; Lei, K.M.; Barton, A.W.; Brody, G.; Beach, S.R.H.; Galván, A.; MacKillop, J.; Clark, U.S.; Sweet, L.H. Age Sensitive Associations of Adolescent Substance Use with Amygdalar, Ventral Striatum, and Frontal Volumes in Young Adulthood. Drug Alcohol Depend 2018, 186, 94–101. [Google Scholar] [CrossRef] [PubMed]
- WHO|Cannabis. Available online: https://www.who.int/substance_abuse/facts/cannabis/en/ (accessed on 17 October 2020).
- Degenhardt, L.; Ferrari, A.J.; Calabria, B.; Hall, W.D.; Norman, R.E.; McGrath, J.; Flaxman, A.D.; Engell, R.E.; Freedman, G.D.; Whiteford, H.A.; et al. The Global Epidemiology and Contribution of Cannabis Use and Dependence to the Global Burden of Disease: Results from the GBD 2010 Study. PLoS ONE 2013, 8, e76635. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Degenhardt, L.; Charlson, F.; Ferrari, A.; Santomauro, D.; Erskine, H.; Mantilla-Herrara, A.; Whiteford, H.; Leung, J.; Naghavi, M.; Griswold, M.; et al. The Global Burden of Disease Attributable to Alcohol and Drug Use in 195 Countries and Territories, 1990–2016: A Systematic Analysis for the Global Burden of Disease Study 2016. Lancet Psychiatry 2018, 5, 987–1012. [Google Scholar] [CrossRef] [Green Version]
- Large, M.; Sharma, S.; Compton, M.T.; Slade, T.; Nielssen, O. Cannabis Use and Earlier Onset of Psychosis: A Systematic Meta-Analysis. Arch. Gen. Psychiatry 2011, 68, 555–561. [Google Scholar] [CrossRef] [Green Version]
- Volkow, N.D.; Baler, R.D.; Compton, W.M.; Weiss, S.R.B. Adverse Health Effects of Marijuana Use. N. Engl. J. Med. 2014, 370, 2219–2227. [Google Scholar] [CrossRef] [Green Version]
- Macleod, J.; Oakes, R.; Copello, A.; Crome, I.; Egger, M.; Hickman, M.; Oppenkowski, T.; Stokes-Lampard, H.; Davey Smith, G. Psychological and Social Sequelae of Cannabis and Other Illicit Drug Use by Young People: A Systematic Review of Longitudinal, General Population Studies. Lancet 2004, 363, 1579–1588. [Google Scholar] [CrossRef]
- Cerdá, M.; Mauro, C.; Hamilton, A.; Levy, N.S.; Santaella-Tenorio, J.; Hasin, D.; Wall, M.M.; Keyes, K.M.; Martins, S.S. Association Between Recreational Marijuana Legalization in the United States and Changes in Marijuana Use and Cannabis Use Disorder From 2008 to 2016. JAMA Psychiatry 2020, 77, 165–171. [Google Scholar] [CrossRef]
- 2019 NSDUH Detailed Tables|CBHSQ Data. Available online: https://www.samhsa.gov/data/report/2019-nsduh-detailed-tables (accessed on 18 October 2020).
- Marcaggi, P. Cerebellar Endocannabinoids: Retrograde Signaling from Purkinje Cells. Cerebellum 2015, 14, 341–353. [Google Scholar] [CrossRef]
- Brown, A.M.; Arancillo, M.; Lin, T.; Catt, D.R.; Zhou, J.; Lackey, E.P.; Stay, T.L.; Zuo, Z.; White, J.J.; Sillitoe, R.V. Molecular Layer Interneurons Shape the Spike Activity of Cerebellar Purkinje Cells. Sci. Rep. 2019, 9, 1742. [Google Scholar] [CrossRef] [Green Version]
- Alger, B.E.; Kim, J. Supply and Demand for Endocannabinoids. Trends Neurosci. 2011, 34, 304–315. [Google Scholar] [CrossRef] [Green Version]
- Dorgans, K.; Demais, V.; Bailly, Y.; Poulain, B.; Isope, P.; Doussau, F. Short-Term Plasticity at Cerebellar Granule Cell to Molecular Layer Interneuron Synapses Expands Information Processing. eLife 2019, 8, e41586. [Google Scholar] [CrossRef] [PubMed]
- Di Marzo, V.; Stella, N.; Zimmer, A. Endocannabinoid Signalling and the Deteriorating Brain. Nat. Rev. Neurosci. 2015, 16, 30–42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hirvonen, J.; Goodwin, R.S.; Li, C.-T.; Terry, G.E.; Zoghbi, S.S.; Morse, C.; Pike, V.W.; Volkow, N.D.; Huestis, M.A.; Innis, R.B. Reversible and Regionally Selective Downregulation of Brain Cannabinoid CB 1 Receptors in Chronic Daily Cannabis Smokers. Mol. Psychiatry 2012, 17, 642–649. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carta, I.; Chen, C.H.; Schott, A.L.; Dorizan, S.; Khodakhah, K. Cerebellar Modulation of the Reward Circuitry and Social Behavior. Science 2019, 363, eaav0581. [Google Scholar] [CrossRef]
- Lohani, S.; Martig, A.K.; Deisseroth, K.; Witten, I.B.; Moghaddam, B. Dopamine Modulation of Prefrontal Cortex Activity Is Manifold and Operates at Multiple Temporal and Spatial Scales. Cell Rep. 2019, 27, 99–114.e6. [Google Scholar] [CrossRef] [Green Version]
- Rogers, L.M.; Madhavan, S.; Roth, H.; Stinear, J.W. Transforming Neurorehabilitation of Walking Following Stroke: The Promise of Non-Invasive Brain Stimulation--a Review. Restor. Neurol. Neurosci. 2011, 29, 507–516. [Google Scholar] [CrossRef] [Green Version]
- Dutta, A. Portable Neuroimaging and Computational Modeling Approach to Elucidate Potential Cognitive Confounds in Non-Invasive Stimulation of the Motor Cerebellum. Brain Stimul. Basic Transl. Clin. Res. Neuromodul. 2021, 14, 1133–1134. [Google Scholar] [CrossRef]
- Makani, R.; Pradhan, B.; Shah, U.; Parikh, T. Role of Repetitive Transcranial Magnetic Stimulation (RTMS) in Treatment of Addiction and Related Disorders: A Systematic Review. Curr. Drug. Abuse Rev. 2017, 10, 31–43. [Google Scholar] [CrossRef]
- Ekhtiari, H.; Tavakoli, H.; Addolorato, G.; Baeken, C.; Bonci, A.; Campanella, S.; Castelo-Branco, L.; Challet-Bouju, G.; Clark, V.P.; Claus, E.; et al. Transcranial Electrical and Magnetic Stimulation (TES and TMS) for Addiction Medicine: A Consensus Paper on the Present State of the Science and the Road Ahead. Neurosci. Biobehav. Rev. 2019, 104, 118–140. [Google Scholar] [CrossRef]
- Stein, E.R.; Gibson, B.C.; Votaw, V.R.; Wilson, A.D.; Clark, V.P.; Witkiewitz, K. Non-Invasive Brain Stimulation in Substance Use Disorders: Implications for Dissemination to Clinical Settings. Curr. Opin. Psychol. 2019, 30, 6–10. [Google Scholar] [CrossRef]
- Goldstein, R.Z.; Volkow, N.D. Dysfunction of the Prefrontal Cortex in Addiction: Neuroimaging Findings and Clinical Implications. Nat. Rev. Neurosci. 2011, 12, 652–669. [Google Scholar] [CrossRef] [PubMed]
- Sahlem, G.L.; Baker, N.L.; George, M.S.; Malcolm, R.J.; McRae-Clark, A.L. Repetitive Transcranial Magnetic Stimulation (RTMS) Administration to Heavy Cannabis Users. Am. J. Drug Alcohol Abuse 2018, 44, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Aron, A.R.; Robbins, T.W.; Poldrack, R.A. Inhibition and the Right Inferior Frontal Cortex. Trends Cogn. Sci. 2004, 8, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Claire Wilcox, M.D. A New Treatment for Substance Use Disorders? NEJM J. Watch 2019, 2019. [Google Scholar] [CrossRef]
- Levy, B.J.; Wagner, A.D. Cognitive Control and Right Ventrolateral Prefrontal Cortex: Reflexive Reorienting, Motor Inhibition, and Action Updating. Ann. N. Y. Acad. Sci. 2011, 1224, 40–62. [Google Scholar] [CrossRef]
- Walia, P.; Kumar, K.N.; Dutta, A. Neuroimaging Guided Transcranial Electrical Stimulation in Enhancing Surgical Skill Acquisition. Comment on Hung et al. The Efficacy of Transcranial Direct Current Stimulation in Enhancing Surgical Skill Acquisition: A Preliminary Meta-Analysis of Randomized Controlled Trials. Brain Sci. 2021, 11, 707. Brain Sci. 2021, 11, 1078. [Google Scholar] [CrossRef]
- Badre, D.; Wagner, A.D. Left Ventrolateral Prefrontal Cortex and the Cognitive Control of Memory. Neuropsychologia 2007, 45, 2883–2901. [Google Scholar] [CrossRef]
- Barredo, J.; Verstynen, T.D.; Badre, D. Organization of Cortico-Cortical Pathways Supporting Memory Retrieval across Subregions of the Left Ventrolateral Prefrontal Cortex. J. Neurophysiol. 2016, 116, 920–937. [Google Scholar] [CrossRef] [Green Version]
- Torregrossa, M.M.; Corlett, P.R.; Taylor, J.R. Aberrant Learning and Memory in Addiction. Neurobiol. Learn. Mem. 2011, 96, 609–623. [Google Scholar] [CrossRef] [Green Version]
- Rae, C.L.; Hughes, L.E.; Anderson, M.C.; Rowe, J.B. The Prefrontal Cortex Achieves Inhibitory Control by Facilitating Subcortical Motor Pathway Connectivity. J. Neurosci. 2015, 35, 786–794. [Google Scholar] [CrossRef] [Green Version]
- Chambers, C.D.; Bellgrove, M.A.; Stokes, M.G.; Henderson, T.R.; Garavan, H.; Robertson, I.H.; Morris, A.P.; Mattingley, J.B. Executive “Brake Failure” Following Deactivation of Human Frontal Lobe. J. Cogn. Neurosci. 2006, 18, 444–455. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Balleine, B.W.; O’Doherty, J.P. Human and Rodent Homologies in Action Control: Corticostriatal Determinants of Goal-Directed and Habitual Action. Neuropsychopharmacology 2010, 35, 48–69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vandaele, Y.; Mahajan, N.R.; Ottenheimer, D.J.; Richard, J.M.; Mysore, S.P.; Janak, P.H. Distinct Recruitment of Dorsomedial and Dorsolateral Striatum Erodes with Extended Training. eLife 2019, 8, e49536. [Google Scholar] [CrossRef] [PubMed]
- Haluk, D.M.; Floresco, S.B. Ventral Striatal Dopamine Modulation of Different Forms of Behavioral Flexibility. Neuropsychopharmacology 2009, 34, 2041–2052. [Google Scholar] [CrossRef]
- Myers, K.M.; Carlezon, W.A., Jr. Extinction of Drug- and Withdrawal-Paired Cues in Animal Models: Relevance to the Treatment of Addiction. Neurosci. Biobehav. Rev. 2010, 35, 285–302. [Google Scholar] [CrossRef] [Green Version]
- Leite, J.; Gonçalves, Ó.F.; Pereira, P.; Khadka, N.; Bikson, M.; Fregni, F.; Carvalho, S. The Differential Effects of Unihemispheric and Bihemispheric TDCS over the Inferior Frontal Gyrus on Proactive Control. Neurosci. Res. 2018, 130, 39–46. [Google Scholar] [CrossRef]
- Hone-Blanchet, A.; Wensing, T.; Fecteau, S. The Use of Virtual Reality in Craving Assessment and Cue-Exposure Therapy in Substance Use Disorders. Front. Hum. Neurosci. 2014, 8. [Google Scholar] [CrossRef] [Green Version]
- Schacht, J.P.; Anton, R.F.; Myrick, H. Functional Neuroimaging Studies of Alcohol Cue Reactivity: A Quantitative Meta-Analysis and Systematic Review. Addict. Biol. 2013, 18, 121–133. [Google Scholar] [CrossRef] [Green Version]
- Mannarelli, D.; Pauletti, C.; Currà, A.; Marinelli, L.; Corrado, A.; Delle Chiaie, R.; Fattapposta, F. The Cerebellum Modulates Attention Network Functioning: Evidence from a Cerebellar Transcranial Direct Current Stimulation and Attention Network Test Study. Cerebellum 2019, 18, 457–468. [Google Scholar] [CrossRef]
- Uddin, L.Q.; Kelly, A.M.; Biswal, B.B.; Castellanos, F.X.; Milham, M.P. Functional Connectivity of Default Mode Network Components: Correlation, Anticorrelation, and Causality. Hum. Brain Mapp. 2009, 30, 625–637. [Google Scholar] [CrossRef] [Green Version]
- Alves, P.N.; Foulon, C.; Karolis, V.; Bzdok, D.; Margulies, D.S.; Volle, E.; Thiebaut de Schotten, M. An Improved Neuroanatomical Model of the Default-Mode Network Reconciles Previous Neuroimaging and Neuropathological Findings. Commun. Biol. 2019, 2, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Marek, S.; Siegel, J.S.; Gordon, E.M.; Raut, R.V.; Gratton, C.; Newbold, D.J.; Ortega, M.; Laumann, T.O.; Adeyemo, B.; Miller, D.B.; et al. Spatial and Temporal Organization of the Individual Human Cerebellum. Neuron 2018, 100, 977–993.e7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiu, Z.; Wang, J. Altered Neural Activities during Response Inhibition in Adults with Addiction: A Voxel-Wise Meta-Analysis. Psychol. Med. 2021, 51, 387–399. [Google Scholar] [CrossRef] [PubMed]
- Kumar, D.; Dutta, A.; Das, A.; Lahiri, U. SmartEye: Developing a Novel Eye Tracking System for Quantitative Assessment of Oculomotor Abnormalities. IEEE Trans. Neural Syst. Rehabil. Eng. 2016, 24, 1051–1059. [Google Scholar] [CrossRef]
- Mannarelli, D.; Pauletti, C.; Petritis, A.; Delle Chiaie, R.; Currà, A.; Trompetto, C.; Fattapposta, F. Effects of Cerebellar TDCS on Inhibitory Control: Evidence from a Go/NoGo Task. Cerebellum 2020, 19, 788–798. [Google Scholar] [CrossRef]
- Huhn, A.S.; Sweeney, M.M.; Brooner, R.K.; Kidorf, M.S.; Tompkins, D.A.; Ayaz, H.; Dunn, K.E. Prefrontal Cortex Response to Drug Cues, Craving, and Current Depressive Symptoms Are Associated with Treatment Outcomes in Methadone-Maintained Patients. Neuropsychopharmacology 2019, 44, 826–833. [Google Scholar] [CrossRef] [Green Version]
- Cousijn, J.; Goudriaan, A.E.; Ridderinkhof, K.R.; van den Brink, W.; Veltman, D.J.; Wiers, R.W. Neural Responses Associated with Cue-Reactivity in Frequent Cannabis Users. Addict. Biol. 2013, 18, 570–580. [Google Scholar] [CrossRef]
- Wacker, J.; Dillon, D.G.; Pizzagalli, D.A. The Role of the Nucleus Accumbens and Rostral Anterior Cingulate Cortex in Anhedonia: Integration of Resting EEG, FMRI, and Volumetric Techniques. Neuroimage 2009, 46, 327–337. [Google Scholar] [CrossRef] [Green Version]
- Pripfl, J.; Tomova, L.; Riecansky, I.; Lamm, C. Transcranial Magnetic Stimulation of the Left Dorsolateral Prefrontal Cortex Decreases Cue-Induced Nicotine Craving and EEG Delta Power. Brain Stimul. 2014, 7, 226–233. [Google Scholar] [CrossRef]
- Sood, M.; Besson, P.; Muthalib, M.; Jindal, U.; Perrey, S.; Dutta, A.; Hayashibe, M. NIRS-EEG Joint Imaging during Transcranial Direct Current Stimulation: Online Parameter Estimation with an Autoregressive Model. J. Neurosci. Methods 2016, 274, 71–80. [Google Scholar] [CrossRef]
- Fernandez, L.; Rogasch, N.C.; Do, M.; Clark, G.; Major, B.P.; Teo, W.-P.; Byrne, L.K.; Enticott, P.G. Cerebral Cortical Activity Following Non-Invasive Cerebellar Stimulation—a Systematic Review of Combined TMS and EEG Studies. Cerebellum 2020, 19, 309–335. [Google Scholar] [CrossRef] [PubMed]
- Morellini, N.; Grehl, S.; Tang, A.; Rodger, J.; Mariani, J.; Lohof, A.M.; Sherrard, R.M. What Does Low-Intensity RTMS Do to the Cerebellum? Cerebellum 2015, 14, 23–26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Middleton, F.A.; Strick, P.L. Cerebellar Projections to the Prefrontal Cortex of the Primate. J. Neurosci. 2001, 21, 700–712. [Google Scholar] [CrossRef] [PubMed]
- Koffarnus, M.N.; Jarmolowicz, D.P.; Mueller, E.T.; Bickel, W.K. Changing Delay Discounting in the Light of the Competing Neurobehavioral Decision Systems Theory: A Review. J. Exp. Anal. Behav. 2013, 99, 32–57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hanlon, C.A.; Dowdle, L.T.; Henderson, J.S. Modulating Neural Circuits with Transcranial Magnetic Stimulation: Implications for Addiction Treatment Development. Pharmacol. Rev. 2018, 70, 661–683. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Volkow, N.D. Brain Default-Mode Network Dysfunction in Addiction. NeuroImage 2019, 200, 313–331. [Google Scholar] [CrossRef]
- Moulton, E.A.; Elman, I.; Becerra, L.R.; Goldstein, R.Z.; Borsook, D. The Cerebellum and Addiction: Insights Gained from Neuroimaging Research. Addict. Biol. 2014, 19, 317–331. [Google Scholar] [CrossRef] [Green Version]
- Hu, C.; Zhang, L.-B.; Chen, H.; Xiong, Y.; Hu, B. Neurosubstrates and Mechanisms Underlying the Extinction of Associative Motor Memory. Neurobiol. Learn. Memory 2015, 126, 78–86. [Google Scholar] [CrossRef]
- Kimpel, O.; Hulst, T.; Batsikadze, G.; Ernst, T.M.; Nitsche, M.A.; Timmann, D.; Gerwig, M. Long-Term Effects of Cerebellar Anodal Transcranial Direct Current Stimulation (TDCS) on the Acquisition and Extinction of Conditioned Eyeblink Responses. Sci. Rep. 2020, 10, 22434. [Google Scholar] [CrossRef]
- Walther, S.; Strik, W. Motor Symptoms and Schizophrenia. NPS 2012, 66, 77–92. [Google Scholar] [CrossRef]
- Rezaee, Z.; Dutta, A. A Computational Pipeline to Optimize Lobule-Specific Electric Field Distribution during Cerebellar Transcranial Direct Current Stimulation. Front. Neurosci. 2019, 13. [Google Scholar] [CrossRef] [PubMed]
- Rampersad, S.M.; Janssen, A.M.; Lucka, F.; Aydin, Ü.; Lanfer, B.; Lew, S.; Wolters, C.H.; Stegeman, D.F.; Oostendorp, T.F. Simulating Transcranial Direct Current Stimulation with a Detailed Anisotropic Human Head Model. IEEE Trans. Neural Syst. Rehabil. Eng. 2014, 22, 441–452. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, L.; Major, B.P.; Teo, W.-P.; Byrne, L.K.; Enticott, P.G. Assessing Cerebellar Brain Inhibition (CBI) via Transcranial Magnetic Stimulation (TMS): A Systematic Review. Neurosci. Biobehav. Rev. 2018, 86, 176–206. [Google Scholar] [CrossRef] [PubMed]
- Batsikadze, G.; Rezaee, Z.; Chang, D.-I.; Gerwig, M.; Herlitze, S.; Dutta, A.; Nitsche, M.A.; Timmann, D. Effects of Cerebellar Transcranial Direct Current Stimulation on Cerebellar-Brain Inhibition in Humans: A Systematic Evaluation. Brain Stimul. Basic Transl. Clin. Res. Neuromodul. 2019, 12, 1177–1186. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Datta, A.; Bikson, M.; Parra, L.C. Realistic Volumetric-Approach to Simulate Transcranial Electric Stimulation—ROAST—a Fully Automated Open-Source Pipeline. J. Neural Eng. 2019, 16, 056006. [Google Scholar] [CrossRef]
- Saturnino, G.B.; Siebner, H.R.; Thielscher, A.; Madsen, K.H. Accessibility of Cortical Regions to Focal TES: Dependence on Spatial Position, Safety, and Practical Constraints. NeuroImage 2019, 203, 116183. [Google Scholar] [CrossRef]
- Rezaee, Z.; Dutta, A. Lobule-Specific Dosage Considerations for Cerebellar Transcranial Direct Current Stimulation During Healthy Aging: A Computational Modeling Study Using Age-Specific Magnetic Resonance Imaging Templates. Neuromodulation 2020, 23, 341–365. [Google Scholar] [CrossRef]
- Krienen, F.M.; Buckner, R.L. Segregated Fronto-Cerebellar Circuits Revealed by Intrinsic Functional Connectivity. Cereb. Cortex 2009, 19, 2485–2497. [Google Scholar] [CrossRef] [Green Version]
- Guhathakurta, D.; Dutta, A. Computational Pipeline for NIRS-EEG Joint Imaging of TDCS-Evoked Cerebral Responses—An Application in Ischemic Stroke. Front. Neurosci. 2016, 10. [Google Scholar] [CrossRef] [Green Version]
- Dutta, A.; Jacob, A.; Chowdhury, S.R.; Das, A.; Nitsche, M.A. EEG-NIRS Based Assessment of Neurovascular Coupling during Anodal Transcranial Direct Current Stimulation--a Stroke Case Series. J. Med. Syst. 2015, 39, 205. [Google Scholar] [CrossRef]
- Rezaee, Z.; Ranjan, S.; Solanki, D.; Bhattacharya, M.; Srivastava, M.V.P.; Lahiri, U.; Dutta, A. Feasibility of Combining Functional Near-Infrared Spectroscopy with Electroencephalography to Identify Chronic Stroke Responders to Cerebellar Transcranial Direct Current Stimulation-a Computational Modeling and Portable Neuroimaging Methodological Study. Cerebellum 2021, 20, 853–871. [Google Scholar] [CrossRef] [PubMed]
- Rezaee, Z.; Kaura, S.; Solanki, D.; Dash, A.; Srivastava, M.V.P.; Lahiri, U.; Dutta, A. Deep Cerebellar Transcranial Direct Current Stimulation of the Dentate Nucleus to Facilitate Standing Balance in Chronic Stroke Survivors—A Pilot Study. Brain Sci. 2020, 10, 94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walter, J.T.; Khodakhah, K. The Linear Computational Algorithm of Cerebellar Purkinje Cells. J. Neurosci. 2006, 26, 12861–12872. [Google Scholar] [CrossRef] [Green Version]
- Ishikawa, T.; Tomatsu, S.; Tsunoda, Y.; Lee, J.; Hoffman, D.S.; Kakei, S. Releasing Dentate Nucleus Cells from Purkinje Cell Inhibition Generates Output from the Cerebrocerebellum. PLoS ONE 2014, 9, e108774. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, M.; Lee, T.H.; Park, H.; Moon, S.-Y.; Lho, S.K.; Kwon, J.S. Thalamocortical Dysrhythmia in Patients with Schizophrenia Spectrum Disorder and Individuals at Clinical High Risk for Psychosis. Neuropsychopharmacology 2022, 47, 673–680. [Google Scholar] [CrossRef] [PubMed]
- Burns, J.K. Pathways from Cannabis to Psychosis: A Review of the Evidence. Front. Psychiatry 2013, 4, 128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Forti, M.; Iyegbe, C.; Sallis, H.; Kolliakou, A.; Falcone, M.A.; Paparelli, A.; Sirianni, M.; La Cascia, C.; Stilo, S.A.; Marques, T.R.; et al. Confirmation That the AKT1 (Rs2494732) Genotype Influences the Risk of Psychosis in Cannabis Users. Biol. Psychiatry 2012, 72, 811–816. [Google Scholar] [CrossRef]
- Klejbor, I.; Kucinski, A.; Wersinger, S.R.; Corso, T.; Spodnik, J.H.; Dziewiatkowski, J.; Moryś, J.; Hesse, R.A.; Rice, K.C.; Miletich, R.; et al. Serotonergic Hyperinnervation and Effective Serotonin Blockade in an FGF Receptor Developmental Model of Psychosis. Schizophr. Res. 2009, 113, 308–321. [Google Scholar] [CrossRef] [Green Version]
- Williams, E.-J.; Walsh, F.S.; Doherty, P. The FGF Receptor Uses the Endocannabinoid Signaling System to Couple to an Axonal Growth Response. J. Cell Biol. 2003, 160, 481–486. [Google Scholar] [CrossRef] [Green Version]
- Brewer, J.R.; Mazot, P.; Soriano, P. Genetic Insights into the Mechanisms of Fgf Signaling. Genes Dev. 2016, 30, 751–771. [Google Scholar] [CrossRef] [Green Version]
- Gao, R.; Penzes, P. Common Mechanisms of Excitatory and Inhibitory Imbalance in Schizophrenia and Autism Spectrum Disorders. Curr. Mol. Med. 2015, 15, 146–167. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Wang, H.; Zeng, Z.; Lin, J.; Little, P.J.; Srivastava, L.K.; Quirion, R. The Possible Role of the Akt Signaling Pathway in Schizophrenia. Brain Res. 2012, 1470, 145–158. [Google Scholar] [CrossRef] [PubMed]
- Shumay, E.; Wiers, C.E.; Shokri-Kojori, E.; Kim, S.W.; Hodgkinson, C.A.; Sun, H.; Tomasi, D.; Wong, C.T.; Weinberger, D.R.; Wang, G.-J.; et al. New Repeat Polymorphism in the AKT1 Gene Predicts Striatal Dopamine D2/D3 Receptor Availability and Stimulant-Induced Dopamine Release in the Healthy Human Brain. J. Neurosci. 2017, 37, 4982–4991. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Talukdar, S.; Owen, B.M.; Song, P.; Hernandez, G.; Zhang, Y.; Zhou, Y.; Scott, W.T.; Paratala, B.; Turner, T.; Smith, A.; et al. FGF21 Regulates Sweet and Alcohol Preference. Cell Metab. 2016, 23, 344–349. [Google Scholar] [CrossRef] [Green Version]
- Dutta, A.; Karanth, S.S.; Bhattacharya, M.; Liput, M.; Augustyniak, J.; Cheung, M.; Stachowiak, E.K.; Stachowiak, M.K. A Proof of Concept ‘Phase Zero’ Study of Neurodevelopment Using Brain Organoid Models with Vis/near-Infrared Spectroscopy and Electrophysiology. Sci. Rep. 2020, 10, 20987. [Google Scholar] [CrossRef]
- Page, C.E.; Coutellier, L. Reducing Inhibition: A Promising New Strategy for the Treatment of Schizophrenia. EBioMedicine 2018, 35, 25–26. [Google Scholar] [CrossRef] [Green Version]
- Winklbaur, B.; Ebner, N.; Sachs, G.; Thau, K.; Fischer, G. Substance Abuse in Patients with Schizophrenia. Dialogues Clin. Neurosci. 2006, 8, 37–43. [Google Scholar]
- Hirano, T. GABA and Synaptic Transmission in the Cerebellum. In Handbook of the Cerebellum and Cerebellar Disorders; Manto, M., Schmahmann, J.D., Rossi, F., Gruol, D.L., Koibuchi, N., Eds.; Springer: Dordrecht, The Netherlands, 2013; pp. 881–893. ISBN 978-94-007-1333-8. [Google Scholar]
- Sadanandan, S.M.; Kreko-Pierce, T.; Khatri, S.N.; Pugh, J.R. Cannabinoid Type 2 Receptors Inhibit GABAA Receptor-Mediated Currents in Cerebellar Purkinje Cells of Juvenile Mice. PLOS ONE 2020, 15, e0233020. [Google Scholar] [CrossRef]
- Martin-Rodriguez, J.F.; Ruiz-Veguilla, M.; Alvarez de Toledo, P.; Aizpurua-Olaizola, O.; Zarandona, I.; Canal-Rivero, M.; Rodriguez-Baena, A.; Mir, P. Impaired Motor Cortical Plasticity Associated with Cannabis Use Disorder in Young Adults. Addict. Biol. 2020, e12912. [Google Scholar] [CrossRef]
- Kuepper, R.; van Os, J.; Lieb, R.; Wittchen, H.-U.; Höfler, M.; Henquet, C. Continued Cannabis Use and Risk of Incidence and Persistence of Psychotic Symptoms: 10 Year Follow-up Cohort Study. BMJ 2011, 342, d738. [Google Scholar] [CrossRef] [Green Version]
- Kendler, K.S.; Lönn, S.L.; Sundquist, J.; Sundquist, K. Smoking and Schizophrenia in Population Cohorts of Swedish Women and Men: A Prospective Co-Relative Control Study. Am. J. Psychiatry 2015, 172, 1092–1100. [Google Scholar] [CrossRef] [PubMed]
- Bernard, J.A.; Mittal, V.A. Cerebellar-Motor Dysfunction in Schizophrenia and Psychosis-Risk: The Importance of Regional Cerebellar Analysis Approaches. Front. Psychiatry 2014, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schulman, J.J.; Cancro, R.; Lowe, S.I.; Lu, F.; Walton, K.D.; Llinás, R.R. Imaging of Thalamocortical Dysrhythmia in Neuropsychiatry. Front. Hum. Neurosci. 2011, 5, 69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, D.; Sinha, N.; Dutta, A.; Lahiri, U. Virtual Reality-Based Balance Training System Augmented with Operant Conditioning Paradigm. BioMed. Eng. OnLine 2019, 18, 90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carvalho, F.; Brietzke, A.P.; Gasparin, A.; Dos Santos, F.P.; Vercelino, R.; Ballester, R.F.; Sanches, P.R.S.; da Silva, D.P.; Torres, I.L.S.; Fregni, F.; et al. Home-Based Transcranial Direct Current Stimulation Device Development: An Updated Protocol Used at Home in Healthy Subjects and Fibromyalgia Patients. J. Vis. Exp. 2018, 137, e57614. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.; Lee, C.; Park, J.; Im, C.-H. Individually Customized Transcranial Temporal Interference Stimulation for Focused Modulation of Deep Brain Structures: A Simulation Study with Different Head Models. Sci. Rep. 2020, 10, 11730. [Google Scholar] [CrossRef]
- Zhang, X.; Santaniello, S. Role of Cerebellar GABAergic Dysfunctions in the Origins of Essential Tremor. PNAS 2019, 116, 13592–13601. [Google Scholar] [CrossRef] [Green Version]
- Hong, L.E.; Summerfelt, A.; McMahon, R.P.; Thaker, G.K.; Buchanan, R.W. Gamma/Beta Oscillation and Sensory Gating Deficit in Schizophrenia. Neuroreport 2004, 15, 155–159. [Google Scholar] [CrossRef]
- Haenschel, C.; Baldeweg, T.; Croft, R.J.; Whittington, M.; Gruzelier, J. Gamma and Beta Frequency Oscillations in Response to Novel Auditory Stimuli: A Comparison of Human Electroencephalogram (EEG) Data with in Vitro Models. PNAS 2000, 97, 7645–7650. [Google Scholar] [CrossRef] [Green Version]
- Yousif, N.; Bain, P.G.; Nandi, D.; Borisyuk, R. A Population Model of Deep Brain Stimulation in Movement Disorders From Circuits to Cells. Front. Hum. Neurosci. 2020, 14, 55. [Google Scholar] [CrossRef] [Green Version]
- Grossman, N.; Bono, D.; Dedic, N.; Kodandaramaiah, S.B.; Rudenko, A.; Suk, H.-J.; Cassara, A.M.; Neufeld, E.; Kuster, N.; Tsai, L.-H.; et al. Noninvasive Deep Brain Stimulation via Temporally Interfering Electric Fields. Cell 2017, 169, 1029–1041.e16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, W.; Krook-Magnuson, E. Cognitive Collaborations: Bidirectional Functional Connectivity Between the Cerebellum and the Hippocampus. Front. Syst. Neurosci. 2015, 9, 177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watson, T.C.; Obiang, P.; Torres-Herraez, A.; Watilliaux, A.; Coulon, P.; Rochefort, C.; Rondi-Reig, L. Anatomical and Physiological Foundations of Cerebello-Hippocampal Interaction. eLife 2019, 8, e41896. [Google Scholar] [CrossRef] [PubMed]
- Bohne, P.; Schwarz, M.K.; Herlitze, S.; Mark, M.D. A New Projection From the Deep Cerebellar Nuclei to the Hippocampus via the Ventrolateral and Laterodorsal Thalamus in Mice. Front. Neural Circ. 2019, 13, 51. [Google Scholar] [CrossRef] [Green Version]
- Lindeman, S.; Hong, S.; Kros, L.; Mejias, J.F.; Romano, V.; Oostenveld, R.; Negrello, M.; Bosman, L.W.J.; Zeeuw, C.I.D. Cerebellar Purkinje Cells Can Differentially Modulate Coherence between Sensory and Motor Cortex Depending on Region and Behavior. PNAS 2021, 118. [Google Scholar] [CrossRef]
- Pinti, P.; Merla, A.; Aichelburg, C.; Lind, F.; Power, S.; Swingler, E.; Hamilton, A.; Gilbert, S.; Burgess, P.W.; Tachtsidis, I. A Novel GLM-Based Method for the Automatic IDentification of Functional Events (AIDE) in FNIRS Data Recorded in Naturalistic Environments. Neuroimage 2017, 155, 291–304. [Google Scholar] [CrossRef]
- Mikkonen, M.; Laakso, I.; Tanaka, S.; Hirata, A. Cost of Focality in TDCS: Interindividual Variability in Electric Fields. Brain Stimul. 2020, 13, 117–124. [Google Scholar] [CrossRef] [Green Version]
- Guell, X.; Schmahmann, J. Cerebellar Functional Anatomy: A Didactic Summary Based on Human FMRI Evidence. Cerebellum 2020, 19, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Buckner, R.L.; Krienen, F.M.; Castellanos, A.; Diaz, J.C.; Yeo, B.T.T. The Organization of the Human Cerebellum Estimated by Intrinsic Functional Connectivity. J. Neurophysiol. 2011, 106, 2322–2345. [Google Scholar] [CrossRef]
- Diedrichsen, J. A Spatially Unbiased Atlas Template of the Human Cerebellum. Neuroimage 2006, 33, 127–138. [Google Scholar] [CrossRef]
- Kaji, R. Basal Ganglia as a Sensory Gating Devise for Motor Control. J. Med. Investig. 2001, 48, 142–146. [Google Scholar]
- Arora, Y.; Walia, P.; Hayashibe, M.; Muthalib, M.; Chowdhury, S.R.; Perrey, S.; Dutta, A. Grey-Box Modeling and Hypothesis Testing of Functional near-Infrared Spectroscopy-Based Cerebrovascular Reactivity to Anodal High-Definition TDCS in Healthy Humans. PLoS Comput. Biol. 2021; 17, e1009386. [Google Scholar]
- Aasted, C.M.; Yücel, M.A.; Cooper, R.J.; Dubb, J.; Tsuzuki, D.; Becerra, L.; Petkov, M.P.; Borsook, D.; Dan, I.; Boas, D.A. Anatomical Guidance for Functional Near-Infrared Spectroscopy: AtlasViewer Tutorial. Neurophotonics 2015, 2. [Google Scholar] [CrossRef] [PubMed]
- Kedzior, K.K.; Martin-Iverson, M.T. Chronic Cannabis Use Is Associated with Attention-Modulated Reduction in Prepulse Inhibition of the Startle Reflex in Healthy Humans. J. Psychopharmacol. 2006, 20, 471–484. [Google Scholar] [CrossRef] [PubMed]
- Rohleder, C.; Wiedermann, D.; Neumaier, B.; Drzezga, A.; Timmermann, L.; Graf, R.; Leweke, F.M.; Endepols, H. The Functional Networks of Prepulse Inhibition: Neuronal Connectivity Analysis Based on FDG-PET in Awake and Unrestrained Rats. Front. Behav. Neurosci. 2016, 10, 148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zimeo Morais, G.A.; Balardin, J.B.; Sato, J.R. FNIRS Optodes’ Location Decider (FOLD): A Toolbox for Probe Arrangement Guided by Brain Regions-of-Interest. Sci. Rep. 2018, 8, 3341. [Google Scholar] [CrossRef] [Green Version]
- Spampinato, D.; Avci, E.; Rothwell, J.; Rocchi, L. Frequency-Dependent Modulation of Cerebellar Excitability during the Application of Non-Invasive Alternating Current Stimulation. Brain Stimul. 2021, 14, 277–283. [Google Scholar] [CrossRef]
- Singh, S.M.; Kumar, K.N.; Walia, P.; Ranjan, S.; Rezaee, Z.; Lahiri, U.; Dutta, A. Functional Near-Infrared Spectroscopy (FNIRS) of Posterolateral Cerebellum and Prefrontal Cortex for FNIRS-Driven Cerebellar TES—A Case Report. Brain Stimul. Basic Transl. Clin. Res. Neuromodul. 2021, 14, 1692. [Google Scholar] [CrossRef]
- Huang, Y.; Liu, A.A.; Lafon, B.; Friedman, D.; Dayan, M.; Wang, X.; Bikson, M.; Doyle, W.K.; Devinsky, O.; Parra, L.C. Measurements and Models of Electric Fields in the in Vivo Human Brain during Transcranial Electric Stimulation. eLife 2017, 6, e18834. [Google Scholar] [CrossRef]
- Johnson, L.; Alekseichuk, I.; Krieg, J.; Doyle, A.; Yu, Y.; Vitek, J.; Johnson, M.; Opitz, A. Dose-Dependent Effects of Transcranial Alternating Current Stimulation on Spike Timing in Awake Nonhuman Primates. Sci. Adv. 2020, 6, eaaz2747. [Google Scholar] [CrossRef]
- Nitsche, M.A.; Paulus, W. Excitability Changes Induced in the Human Motor Cortex by Weak Transcranial Direct Current Stimulation. J. Physiol. (Lond.) 2000, 527 Pt 3, 633–639. [Google Scholar] [CrossRef]
- Nitsche, M.A.; Paulus, W. Sustained Excitability Elevations Induced by Transcranial DC Motor Cortex Stimulation in Humans. Neurology 2001, 57, 1899–1901. [Google Scholar] [CrossRef] [PubMed]
- Paquette, C.; Sidel, M.; Radinska, B.A.; Soucy, J.-P.; Thiel, A. Bilateral Transcranial Direct Current Stimulation Modulates Activation-Induced Regional Blood Flow Changes during Voluntary Movement. J. Cereb. Blood Flow Metab. 2011, 31, 2086–2095. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huppert, T.J.; Diamond, S.G.; Franceschini, M.A.; Boas, D.A. HomER: A Review of Time-Series Analysis Methods for near-Infrared Spectroscopy of the Brain. Appl. Opt. 2009, 48, D280–D298. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jahani, S.; Setarehdan, S.K.; Boas, D.A.; Yücel, M.A. Motion Artifact Detection and Correction in Functional Near-Infrared Spectroscopy: A New Hybrid Method Based on Spline Interpolation Method and Savitzky–Golay Filtering. Neurophotonics 2018, 5, 015003. [Google Scholar] [CrossRef] [Green Version]
- Ye, J.C.; Tak, S.; Jang, K.E.; Jung, J.; Jang, J. NIRS-SPM: Statistical Parametric Mapping for near-Infrared Spectroscopy. Neuroimage 2009, 44, 428–447. [Google Scholar] [CrossRef]
- Cortes-Briones, J.; Skosnik, P.D.; Mathalon, D.; Cahill, J.; Pittman, B.; Williams, A.; Sewell, R.A.; Ranganathan, M.; Roach, B.; Ford, J.; et al. Δ9-THC Disrupts Gamma (γ)-Band Neural Oscillations in Humans. Neuropsychopharmacology 2015, 40, 2124–2134. [Google Scholar] [CrossRef] [Green Version]
- Tournier, B.B.; Ginovart, N. Repeated but Not Acute Treatment with ∆9-Tetrahydrocannabinol Disrupts Prepulse Inhibition of the Acoustic Startle: Reversal by the Dopamine D₂/₃ Receptor Antagonist Haloperidol. Eur. Neuropsychopharmacol. 2014, 24, 1415–1423. [Google Scholar] [CrossRef]
- Saletti, P.G.; Tomaz, C. Cannabidiol Effects on Prepulse Inhibition in Nonhuman Primates. Rev. Neurosci. 2019, 30, 95–105. [Google Scholar] [CrossRef]
- Kawamura, Y.; Fukaya, M.; Maejima, T.; Yoshida, T.; Miura, E.; Watanabe, M.; Ohno-Shosaku, T.; Kano, M. The CB1 Cannabinoid Receptor Is the Major Cannabinoid Receptor at Excitatory Presynaptic Sites in the Hippocampus and Cerebellum. J. Neurosci. 2006, 26, 2991–3001. [Google Scholar] [CrossRef] [Green Version]
- Thomas, A.; Baillie, G.L.; Phillips, A.M.; Razdan, R.K.; Ross, R.A.; Pertwee, R.G. Cannabidiol Displays Unexpectedly High Potency as an Antagonist of CB1 and CB2 Receptor Agonists in Vitro. Br. J. Pharmacol. 2007, 150, 613–623. [Google Scholar] [CrossRef] [Green Version]
- Kesby, J.P.; Eyles, D.W.; McGrath, J.J.; Scott, J.G. Dopamine, Psychosis and Schizophrenia: The Widening Gap between Basic and Clinical Neuroscience. Transl. Psychiatry 2018, 8, 30. [Google Scholar] [CrossRef] [PubMed]
- Zachek, M.K.; Takmakov, P.; Park, J.; Wightman, R.M.; McCarty, G.S. Simultaneous Monitoring of Dopamine Concentration at Spatially Different Brain Locations in Vivo. Biosens. Bioelectron. 2010, 25, 1179–1185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, J.; Takmakov, P.; Wightman, R.M. In Vivo Comparison of Norepinephrine and Dopamine Release in Rat Brain by Simultaneous Measurements with Fast-Scan Cyclic Voltammetry. J. Neurochem. 2011, 119, 932–944. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, J.; Wheeler, R.A.; Fontillas, K.; Keithley, R.B.; Carelli, R.M.; Wightman, R.M. Catecholamines in the Bed Nucleus of the Stria Terminalis Reciprocally Respond to Reward and Aversion. Biol. Psychiatry 2012, 71, 327–334. [Google Scholar] [CrossRef] [Green Version]
- Park, J.; Bucher, E.S.; Fontillas, K.; Owesson-White, C.; Ariansen, J.L.; Carelli, R.M.; Wightman, R.M. Opposing Catecholamine Changes in the Bed Nucleus of the Stria Terminalis during Intracranial Self-Stimulation and Its Extinction. Biol. Psychiatry 2013, 74, 69–76. [Google Scholar] [CrossRef] [Green Version]
- Park, J.W.; Bhimani, R.V.; Park, J. Noradrenergic Modulation of Dopamine Transmission Evoked by Electrical Stimulation of the Locus Coeruleus in the Rat Brain. ACS Chem. Neurosci. 2017, 8, 1913–1924. [Google Scholar] [CrossRef]
- Wickham, R.J.; Park, J.; Nunes, E.J.; Addy, N.A. Examination of Rapid Dopamine Dynamics with Fast Scan Cyclic Voltammetry During Intra-Oral Tastant Administration in Awake Rats. J. Vis. Exp. 2015. [Google Scholar] [CrossRef] [Green Version]
- Rodeberg, N.T.; Sandberg, S.G.; Johnson, J.A.; Phillips, P.E.M.; Wightman, R.M. Hitchhiker’s Guide to Voltammetry: Acute and Chronic Electrodes for in Vivo Fast-Scan Cyclic Voltammetry. ACS Chem. Neurosci. 2017, 8, 221–234. [Google Scholar] [CrossRef] [Green Version]
- Koganemaru, S.; Mikami, Y.; Matsuhashi, M.; Truong, D.Q.; Bikson, M.; Kansaku, K.; Mima, T. Cerebellar Transcranial Alternating Current Stimulation Modulates Human Gait Rhythm. Neurosci. Res. 2020, 156, 265–270. [Google Scholar] [CrossRef]
- Lewis, M. Brain Change in Addiction as Learning, Not Disease. N. Engl. J. Med. 2018, 379, 1551–1560. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Walia, P.; Ghosh, A.; Singh, S.; Dutta, A. Portable Neuroimaging-Guided Noninvasive Brain Stimulation of the Cortico-Cerebello-Thalamo-Cortical Loop—Hypothesis and Theory in Cannabis Use Disorder. Brain Sci. 2022, 12, 445. https://doi.org/10.3390/brainsci12040445
Walia P, Ghosh A, Singh S, Dutta A. Portable Neuroimaging-Guided Noninvasive Brain Stimulation of the Cortico-Cerebello-Thalamo-Cortical Loop—Hypothesis and Theory in Cannabis Use Disorder. Brain Sciences. 2022; 12(4):445. https://doi.org/10.3390/brainsci12040445
Chicago/Turabian StyleWalia, Pushpinder, Abhishek Ghosh, Shubhmohan Singh, and Anirban Dutta. 2022. "Portable Neuroimaging-Guided Noninvasive Brain Stimulation of the Cortico-Cerebello-Thalamo-Cortical Loop—Hypothesis and Theory in Cannabis Use Disorder" Brain Sciences 12, no. 4: 445. https://doi.org/10.3390/brainsci12040445
APA StyleWalia, P., Ghosh, A., Singh, S., & Dutta, A. (2022). Portable Neuroimaging-Guided Noninvasive Brain Stimulation of the Cortico-Cerebello-Thalamo-Cortical Loop—Hypothesis and Theory in Cannabis Use Disorder. Brain Sciences, 12(4), 445. https://doi.org/10.3390/brainsci12040445






