Modulating Behavioural and Self-Reported Aggression with Non-Invasive Brain Stimulation: A Literature Review
Abstract
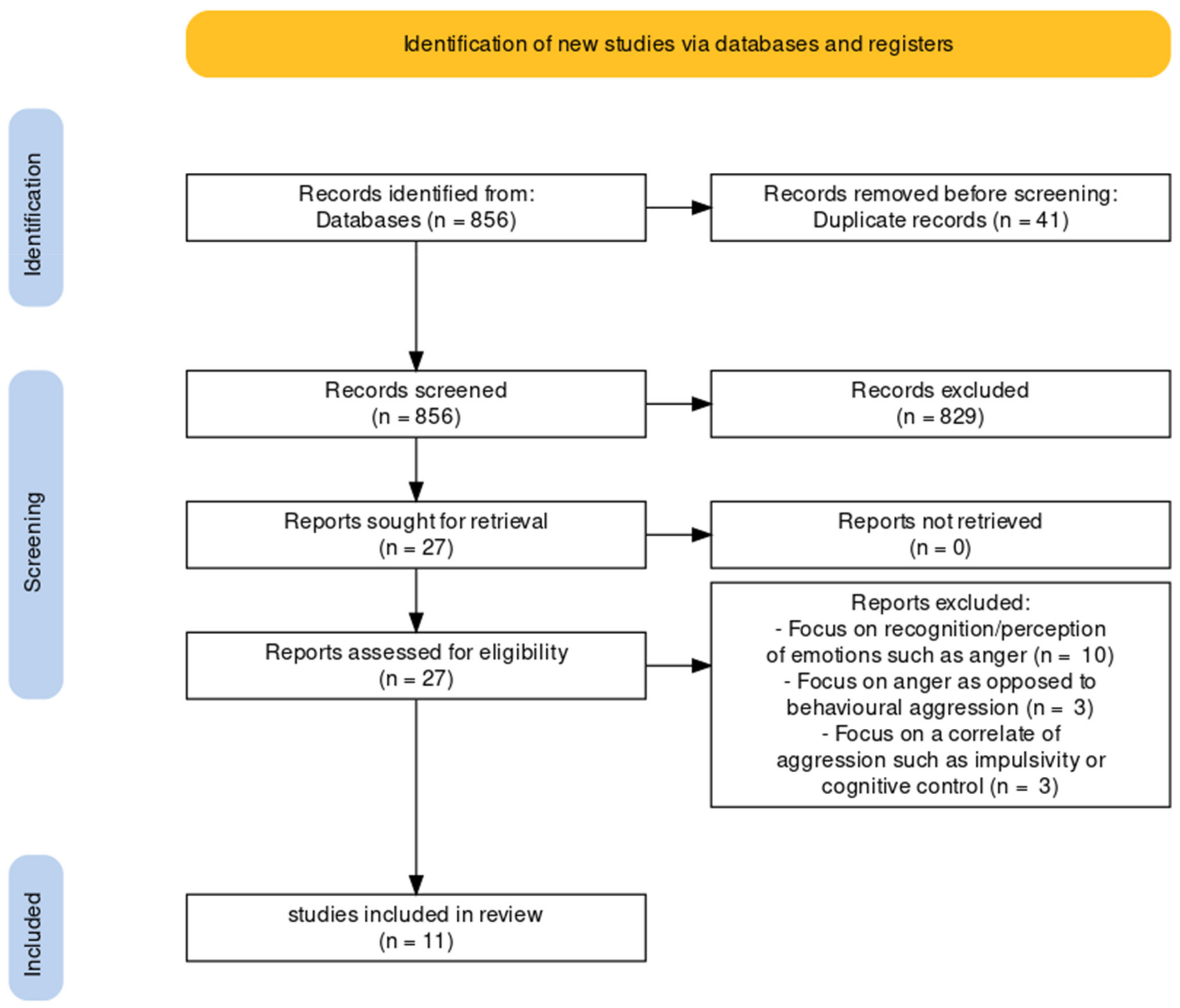
1. Introduction
2. Materials and Methods
2.1. Literature Review
2.2. Measures of Aggression
2.3. Overview of the Study Methodologies
2.3.1. Stimulation Sites
2.3.2. Stimulation Lengths and Intensities
2.3.3. Side Effects
3. Results
3.1. Results for Each Stimulation Site
3.1.1. DLPFC
3.1.2. VLPFC
3.1.3. IFG
3.1.4. VMPFC
3.2. Stimulation Instruments
3.3. Cortical Asymmetry
3.4. Task versus Questionnaire
3.5. Gender Effects
4. Discussion
4.1. Contextual Factors
4.2. Cortical Asymmetry
4.3. Up- or Downregulation
4.4. Gender Differences
4.5. Group Differences
4.6. Consistency of NIBS
4.7. Temporary Effects of NIBS
4.8. Limitations
4.9. Clinical and Forensic Relevance
4.10. Future Research
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Coie, J.D.; Dodge, K.A.; Terry, R.; Wright, V. The role of aggression in peer relations: An analysis of aggression episodes in boys’ play groups. Child Dev. 1991, 62, 812–826. [Google Scholar] [CrossRef] [PubMed]
- Foster, E.M.; Jones, D.E. The high costs of aggression: Public expenditures resulting from conduct disorder. Am. J. Public Health 2005, 95, 1767–1772. [Google Scholar] [CrossRef] [PubMed]
- Gaynes, B.N.; Lloyd, S.W.; Lux, L.; Gartlehner, G.; Hansen, R.A.; Brode, S.; Jonas, D.E.; Swinson Evans, T.; Viswanathan, M.; Lohr, K.N. Repetitive transcranial magnetic stimulation for treatment-resistant depression: A systematic review and meta-analysis. J. Clin. Psychiatry 2014, 75, 477–489. [Google Scholar] [CrossRef] [PubMed]
- Anderson, C.A.; Bushman, B.J. Human aggression. Annu. Rev. Psychol. 2002, 53, 27–51. [Google Scholar] [CrossRef] [PubMed]
- Poulin, F.; Boivin, M. Reactive and proactive aggression: Evidence of a two-factor model. Psychol. Assess. 2000, 12, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Raine, A.; Dodge, K.; Loeber, R.; Gatzke-Kopp, L.; Lynam, D.; Reynolds, C.; Stouthamer-Loeber, M.; Liu, J. The reactive–proactive aggression questionnaire: Differential correlates of reactive and proactive aggression in adolescent boys. Aggress. Behav. 2006, 32, 159–171. [Google Scholar] [CrossRef]
- Lobbestael, J.; Cima, M.; Arntz, A. The relationship between adult reactive and proactive aggression, hostile interpretation bias, and antisocial personality disorder. J. Personal. Disord. 2013, 27, 53–66. [Google Scholar] [CrossRef]
- Brugman, S.; Lobbestael, J.; Arntz, A.; Cima, M.; Schuhmann, T.; Dambacher, F.; Sack, A.T. Identifying cognitive predictors of reactive and proactive aggression. Aggress. Behav. 2014, 41, 51–64. [Google Scholar] [CrossRef]
- Cima, M.; Raine, A. Do distinct characteristics of psychopathy relate to different subtypes of aggression? Personal. Individ. Differ. 2009, 47, 835–840. [Google Scholar] [CrossRef]
- Ritter, D.; Eslea, M. Hot sauce, toy guns, and graffiti: A critical account of current laboratory aggression paradigms. Aggress. Behav. 2005, 31, 407–419. [Google Scholar] [CrossRef]
- Taylor, S.P. Aggressive behavior and physiological arousal as a function of provocation and the tendency to inhibit aggression. J. Personal. 1967, 35, 297–310. [Google Scholar] [CrossRef] [PubMed]
- De Wall, C.N.; Finkel, E.J.; Lambert, N.M.; Slotter, E.B.; Bodenhausen, G.V.; Pond, R.S.; Renzetti, C.M.; Fincham, F.D. The voodoo doll task: Introducing and validating a novel method for studying aggressive inclinations. Aggress. Behav. 2013, 39, 419–439. [Google Scholar]
- Gilam, G.; Abend, R.; Gurevitch, G.; Erdman, A.; Baker, H.; Ben-Zion, Z.; Hendler, T. Attenuating anger and aggression with neuromodulation of the vmPFC: A simultaneous tDCS-fMRI study. Cortex 2018, 109, 156–170. [Google Scholar] [CrossRef] [PubMed]
- Choy, O.; Raine, A.; Hamilton, R.H. Stimulation of the Prefrontal Cortex Reduces Intentions to Commit Aggression: A Randomized, Double-Blind, Placebo-Controlled, Stratified, Parallel-Group Trial. J. Neurosci. 2018, 38, 6505–6512. [Google Scholar] [CrossRef]
- Achterberg, M.; van Duijvenvoorde, A.C.K.; van IJzendoorn, M.H.; Bakermans-Kranenburg, M.J.; Crone, E.A. Longitudinal changes in DLPFC activation during childhood are related to decreased aggression following social rejection. Proc. Natl. Acad. Sci. USA 2020, 117, 8602–8610. [Google Scholar] [CrossRef]
- Carnagey, N.L.; Anderson, C.A.; Bartholow, B.D. Media violence and social neuroscience: New questions and new opportunities. Curr. Dir. Psychol. Sci. 2007, 16, 178–182. [Google Scholar] [CrossRef]
- Ernst, M.; Bolla, K.; Mouratidis, M.; Contoreggi, C.; Matochik, J.A.; Kurian, V.; London, E.D. Decision-making in a risk- taking task: A PET study. Neuropsychopharmacology 2002, 26, 682–691. [Google Scholar] [CrossRef]
- McClure, S.M.; Laibson, D.I.; Loewenstein, G.; Cohen, J.D. Separate neural systems value immediate and delayed monetary rewards. Science 2004, 306, 503–507. [Google Scholar] [CrossRef]
- Kim, S.H.; Hamann, S. Neural correlates of positive and negative emotion regulation. J. Cogn. Neurosci. 2007, 19, 776–798. [Google Scholar] [CrossRef]
- Dambacher, F.; Schuhmann, T.; Lobbestael, J.; Arnzt, A.; Brugman, S.; Sack, A.T. No effects of bilateral tDCS over inferior frontal gyrus on response inhibition and aggression. PLoS ONE 2015, 10, e0132170. [Google Scholar] [CrossRef]
- Harmon-Jones, E.; Gable, P. On the role of asymmetric frontal cortical activity in approach and withdrawal motivation: An updated review of the evidence. Psychophysiology 2017, 55, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Molero-Chamizo, A.; Riquel, R.M.; Moriana, J.A.; Nitsche, M.A.; Rivera-Urbina, G.N. Bilateral Prefrontal Cortex Anodal tDCS Effects on Self-Reported Aggressiveness in Imprisoned Violent Offenders. Neuroscience 2019, 397, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Fertonani, A.; Miniussi, C. Transcranial Electrical Stimulation: What We Know and Do Not Know About Mechanisms. Neuroscientist 2017, 23, 109–123. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, L.; Koslowsky, M.; Lavidor, M. tDCS polarity effects in motor and cognitive domains: A meta-analytical review. Exp. Brain Res. 2012, 216, 1–10. [Google Scholar] [CrossRef] [PubMed]
- George, M.S.; Aston-Jones, G. Noninvasive techniques for probing neurocircuitry and treating illness: Vagus nerve stimulation (VNS), transcranial magnetic stimulation (TMS) and direct current stimulation (tDCS). Neuropsychopharmacol. Rev. 2010, 35, 301–316. [Google Scholar] [CrossRef]
- Post, A.; Keck, M.E. Transcranial magnetic stimulation as a therapeutic tool in psychiatry: What do we know about the neurobiological mechanisms? J. Psychiatr. Res. 2001, 35, 193–215. [Google Scholar] [CrossRef]
- Perach-Barzilay, N.; Tauber, A.; Klein, E.; Chistyakov, A.; Ne’eman, R.; Shamay-Tsoory, S.G. Asymmetry in the dorsolateral prefrontal cortex and aggressive behavior: A continuous theta-burst magnetic stimulation study. Soc. Neurosci. 2013, 8, 178–188. [Google Scholar] [CrossRef]
- Vonck, S.; Patrick Swinnen, S.P.; Wenderoth, N.; Alaerts, K. Effects of transcranial direct current stimulation on the recognition of bodily emotions from point-light displays. Front. Hum. Neurosci. 2015, 9, 438. [Google Scholar] [CrossRef]
- Janik, A.B.; Rezlescu, C.; Banissy, M.J. Enhancing Anger Perception With Transcranial Alternating Current Stimulation Induced Gamma Oscillations. Brain Stimul. 2015, 8, 1138–1143. [Google Scholar] [CrossRef]
- Yang, T.; Banissy, M.J. Enhancing anger perception in older adults by stimulating inferior frontal cortex with high frequency transcranial random noise stimulation. Neuropsychologia 2017, 102, 163–169. [Google Scholar] [CrossRef]
- Ferrucci, R.; Giannicola, G.; Rosa, M.; Fumagalli, M.; Boggio, P.S.; Hallett, M.; Zago, S.; Priori, A. Cerebellum and processing of negative facial emotions: Cerebellar transcranial DC stimulation specifically enhances the emotional recognition of facial anger and sadness. Cogn. Emot. 2012, 5, 786–799. [Google Scholar] [CrossRef] [PubMed]
- Van Honk, J.; Hermans, E.J.; D’alfonso, A.A.; Schutter, D.J.; van Doornen, L.; de Haan, E.H. A left-prefrontal lateralized, sympathetic mechanism directs attention towards social threat in humans: Evidence from repetitive transcranial magnetic stimulation. Neurosci. Lett. 2002, 319, 99–102. [Google Scholar] [CrossRef]
- D’Alfonso, A.A.; Van Honk, J.; Hermans, E.; Postma, A.; de Haan, E.H. Laterality effects in selective attention to threat after repetitive transcranial magnetic stimulation at the prefrontal cortex in female subjects. Neurosci. Lett. 2000, 280, 195–198. [Google Scholar] [CrossRef]
- Donhauser, P.W.; Belin, P.; Grosbras, M. Biasing the perception of ambiguous vocal affect: A TMS study on frontal asymmetry. Soc. Cogn. Affect. Neurosci. 2014, 9, 1046–1051. [Google Scholar] [CrossRef][Green Version]
- Harmer, C.J.; Thilo, K.V.; Rothwell, C.J.; Goodwin, G.M. Transcranial magnetic stimulation of medial-frontal cortex impairs the processing of angry facial expressions. Nat. Neurosci. 2001, 4, 17–18. [Google Scholar] [CrossRef]
- Schutter, D.J.L.G.; Van Honk, J.; Laman, M.; Vergouwen, A.C.; Koerselman, F. Increased sensitivity for angry faces in depressive disorder following 2 weeks of 2-Hz repetitive transcranial magnetic stimulation to the right parietal cortex. Int. J. Neuropsychopharmacol. 2010, 13, 1155–1161. [Google Scholar] [CrossRef]
- Nitsche, M.A.; Koschack, J.; Pohlers, H.; Hullemann, S.; Paulus, W.; Happe, S. Effects of frontal transcranial direct current stimulation on emotional state and processing in healthy humans. Front. Psychiatry 2012, 3, 58. [Google Scholar] [CrossRef]
- Hung, G.C.; Huang, M. Transient anger attacks associated with bifrontal transcranial direct current stimulation. Brain Stimul. 2017, 10, 981–982. [Google Scholar] [CrossRef]
- Kelley, N.J.; Hortensius, R.; Harmon-Jones, E. When anger leads to rumination: Induction of relative right frontal cortical activity with transcranial direct current stimulation increases anger-related rumination. Psychol. Sci. 2013, 24, 475–481. [Google Scholar] [CrossRef]
- Hofman, D.; Schutter, D.J. Inside the wire: Aggression and functional interhemispheric connectivity in the human brain. Psychophysiology 2009, 46, 1054–1058. [Google Scholar] [CrossRef]
- Yang, C.C.; Völlm, B.; Khalifa, N. The Effects of rTMS on Impulsivity in Normal Adults: A Systematic Review and Meta-Analysis. Neuropsychol. Rev. 2018, 28, 377–392. [Google Scholar] [CrossRef] [PubMed]
- Reyes-López, J.; Ricardo-Garcell, J.; Armas-Castañeda, G.; García-Anaya, M.; Arango-De Montis, I.; González-Olvera, J.J.; Pellicer, F. Clinical improvement in patients with borderline personality disorder after treatment with repetitive transcranial magnetic stimulation: Preliminary results. Braz. J. Psychiatry 2018, 40, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Fischer, R.; Ventura-Bort, C.; Hamm, A.; Weymar, M. Transcutaneous vagus nerve stimulation (tVNS) enhances conflict-triggered adjustment of cognitive control. Cogn. Affect. Behav. Neurosci. 2018, 18, 680–693. [Google Scholar] [CrossRef] [PubMed]
- Warburton, W.A.; Bushman, B.J. The competitive reaction time task: The development and scientific utility of a flexible laboratory aggression paradigm. Aggress. Behav. 2019, 45, 389–396. [Google Scholar] [CrossRef] [PubMed]
- Gallucci, A.; Riva, P.; Romero Lauro, L.J.; Bushman, B.J. Stimulating the ventrolateral prefrontal cortex (VLPFC) modulates frustration-induced aggression: A tDCS experiment. Brain Stimul. 2019, 13, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Lieberman, J.D.; Solomon, S.; Greenberg, J.; McGregor, H.A. A hot new way to measure aggression: Hot sauce allocation. Aggress. Behav. 1999, 25, 331–348. [Google Scholar] [CrossRef]
- Buss, A.H.; Perry, M. The aggression questionnaire. J. Personal. Soc. Psychol. 1992, 63, 452–459. [Google Scholar] [CrossRef]
- Chen, C. Right ventrolateral prefrontal cortex involvement in proactive and reactive aggression: A transcranial direct current stimulation study. Clin. Neurosci. 2019, 29, 1509–1515. [Google Scholar] [CrossRef]
- Dambacher, F.; Schuhmann, T.; Lobbestael, J.; Arntz, A.; Brugman, S.; Sack, A.T. Reducing proactive aggression through non-invasive brain stimulation. Soc. Cogn. Affect. Neurosci. 2015, 10, 1303–1309. [Google Scholar] [CrossRef]
- Hortensius, R.; Schutter, D.J.; Harmon-Jones, E. When anger leads to aggression: Induction of relative left frontal cortical activity with transcranial direct current stimulation increases the anger–aggression relationship. Soc. Cogn. Affect. Neurosci. 2012, 7, 342–347. [Google Scholar] [CrossRef]
- Riva, P.; Gabbiadini, A.; Romero Lauro, L.J.; Andrighetto, L.; Volpato, C.; Bushman, B.J. Neuromodulation can reduce aggressive behavior elicited by violent video games. Cogn. Affect. Behav. Neurosci. 2017, 17, 452–459. [Google Scholar] [CrossRef]
- Lobbestael, J.; Emmerling, F.; Brugman, S.; Broers, N.; Sack, A.T.; Schuhmann, T.; Bonnemayer, C.; Benning, R.; Arntz, A. Toward a More Valid Assessment of Behavioral Aggression: An Open Source Platform and an Empirically Derived Scoring Method for Using the Competitive Reaction Time Task (CRTT). Assessment 2021, 28, 1065–1079. [Google Scholar] [CrossRef] [PubMed]
- Chester, D.S.; Lasko, E.N. Validating a standardized approach to the Taylor Aggression Paradigm. Soc. Psychol. Personal. Sci. 2019, 10, 620–631. [Google Scholar] [CrossRef]
- Saleem, M.; Barlett, C.P.; Anderson, C.A.; Hawkins, I. Helping and hurting others: Person and situation effects on aggressive and prosocial behavior as assessed by the Tangram task. Aggress. Behav. 2017, 43, 133–146. [Google Scholar] [CrossRef]
- Pan, W.; Gao, X.; Shi, S.; Liu, F.; Li, C. Spontaneous brain activity did not show the effect of violent video games on aggression: A resting-state fMRI study. Front. Psychol. 2018, 8, 2219. [Google Scholar] [CrossRef]
- Gerevich, J.; Bácskai, E.; Czobor, P. The generalizability of the Buss–Perry Aggression Questionnaire. Int. J. Methods Psychiatr. Res. 2007, 16, 124–136. [Google Scholar] [CrossRef]
- Riva, P.; Romero Lauro, L.J.; DeWall, C.N.; Chester, D.S.; Bushman, B.J. Reducing aggressive responses to social exclusion using transcranial direct current stimulation. Soc. Cogn. Affect. Neurosci. 2015, 10, 352–356. [Google Scholar] [CrossRef] [PubMed]
- Rabipour, S.; Wu, A.D.; Davidson, P.S.R.; Iacoboni, M. Expectations may influence the effects of transcranial direct current stimulation. Neuropsychologia 2018, 119, 524–534. [Google Scholar] [CrossRef]
- Lagerspetz, K.M.J.; Bjorkqvist, K.; Peltonen, T. Is indirect aggression typical of females? Gender differences in aggressiveness in 11 to 12 year old children. Aggress. Behav. 1988, 14, 403–414. [Google Scholar] [CrossRef]
- Bjorkqvist, K. Sex differences in physical, verbal, and indirect aggression: A review of recent research. Sex Roles 1994, 30, 177–188. [Google Scholar] [CrossRef]
- Archer, J. Sex differences in aggression in real-world settings: A meta-analytic review. Rev. Gen. Psychol. 2004, 8, 291–322. [Google Scholar] [CrossRef]
- Li, L.M.; Uehara, K.; Hanakawa, T. The contribution of interindividual factors to variability of response in transcranial direct current stimulation studies. Front. Cell. Neurosci. 2015, 9, 181. [Google Scholar] [CrossRef] [PubMed]
- Chaieb, L.; Antal, A.; Paulus, W. Gender-specific modulation of short-term neuroplasticity in the visual cortex induced by transcranial direct current stimulation. Vis. Neurosci. 2008, 25, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Adenzato, M.; Brambilla, M.; Manenti, R.; De Lucia, L.; Trojano, L.; Garofalo, S. Gender differences in cognitive Theory of Mind revealed by transcranial direct current stimulation on medial prefrontal cortex. Sci. Rep. 2017, 7, 41219. [Google Scholar] [CrossRef]
- Boggio, P.S.; Rocha, R.R.; da Silva, M.T.; Fregni, F. Differential modulatory effects of transcranial direct current stimulation on a facial expression go-no-go task in males and females. Neurosci. Lett. 2008, 447, 101–105. [Google Scholar] [CrossRef]
- Peña-Gómez, C.; Vidal-Piñeiro, D.; Clemente, I.C.; Pascual-Leone, Á.; Bertrés-Faz, D. Down-Regulation of Negative Emotional Processing by Transcranial Direct Current Stimulation: Effects of Personality Characteristics. PLoS ONE 2011, 6, e22812. [Google Scholar] [CrossRef]
- Batsikadze, G.; Moliadze, V.; Paulus, W.; Kuo, M.F.; Nitsche, M.A. Partially non-linear stimulation intensity-dependent effects of direct current stimulation on motor cortex excitability in humans. J. Physiol. 2013, 591, 1987–2000. [Google Scholar] [CrossRef]
- Monte-Silva, K.; Kuo, M.F.; Liebetanz, D.; Paulus, W.; Nitsche, M.A. Shaping the optimal repetition interval for cathodal transcranial direct current stimulation (tDCS). J. Neurophysiol. 2010, 103, 1735–1740. [Google Scholar] [CrossRef]
- Kuo, M.F.; Paulus, W.; Nitsche, M.A. Boosting focally-induced brain plasticity by dopamine. Cerebral Cortex 2008, 18, 648–651. [Google Scholar] [CrossRef]
- Lefaucheur, J.-P.; André-Obadia, N.; Antal, A.; Ayache, S.S.; Baeken, C.; Benninger, D.H.; Garcia-Larrea, L. Evidence-based guidelines on the therapeutic use of repetitive transcranial magnetic stimulation (rTMS). Clin. Neurophysiol. 2014, 125, 2150–2206. [Google Scholar] [CrossRef]
- Lefaucheur, J.-P.; Aleman, A.; Baeken, C.; Benninger, D.H.; Brunelin, J.; Di Lazzaro, V.; Ziemann, U. Evidence-based guidelines on the therapeutic use of repetitive transcranial magnetic stimulation (rTMS): An update (2014–2018). Clin. Neurophysiol. 2020, 131, 474–528. [Google Scholar] [CrossRef] [PubMed]
- Pal, E.; Nagy, F.; Aschermann, Z.; Balazs, E.; Kovacs, N. The impact of left prefrontal repetitive transcranial magnetic stimulation on depression in Parkinson’s disease: A randomized, double-blind, placebo-controlled study. Mov. Disord. 2010, 25, 2311–2317. [Google Scholar] [CrossRef] [PubMed]
| Authors and Publication Year | Participants and Gender Division | Design | NIBS Method | Stimulated Brain Region | NIBS Location | Stimulation Type | Stimulation Length | Stimulation Intensity | Side Effects | Aggression Measures | Sham Condition Total Aggression | Sham Condition Reactive Aggression (M/SD) | Sham Condition Proactive Aggression (M/SD) | Experimental Condition Total Aggression (M/SD) | Experimental Condition Reactive Aggression (M/SD) | Experimental Condition Proactive Aggression (M/SD) | Cohen d |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Chen (2019) [48] | 32 (50% males) | Sham-controlled, RCT, double-blind, between-subject design | tDCS | Right VLPFC | Anode over right VLPFC (F6), cathode over occipital cortex (Oz) | Anodal or sham | 12.5 min (20 s ramp up/down) | 2.0 mA | None reported. | TAP | N/A | M = 5.96 + −1.17 | M = 4.82 + −1.32 | N/A | M = 5.19 + −0.92 * | M = 3.43 + −1.24 * | Reactive aggression d = 0.73 (−) Proactive aggression d = 1.09 (−) |
| Choy et al. (2018) [14] | 81 (44% males) | Sham-controlled, RCT, double-blind, between-subject design | tDCS | Bilateral DLPFC | Anodes bilaterally over left DLPFC (F3) and right DLPFC (F4), cathode: posterior base of the neck | Anodal or sham | 20 min (30 s ramp up and 2 s ramp down) | 2.0 mA | Mild physical side effects reported. | Voodoo Doll Task | N/A | N/A | N/A | F(1,71) = 1.31, p = 0.26 | N/A | N/A | - |
| Dambacher et al. (2015) [49] | 32 (63% males) | Sham-controlled, RCT, between-subject design | tDCS | Right DLPFC | Anode: right DLPFC (F4), cathode: left eyebrow | Anodal or sham | 12.5 min (20 s ramp up/down) | 2.0 mA | None reported. | TAP | 4.00 + −1.33 | M = 4.13 + −1.48 | 3.49 + −1.72 | 3.84 + −1.16 | M = 4.07 + −1.25 | M = 2.93 + −1.25 ** | Proactive aggression d = 1.56 ** (−) |
| Dambacher et al. (2015) [20] | 64 (61% males) | Sham-controlled, RCT, between-subject design, two experimental conditions | tDCS | Right or left IFG | Anode: right IFG (F8), cathode: left IFG (F7). | Anodal and cathodal or sham | 21.75 min (20 s ramp up/down) | 1.5 mA | Mild physical side effects reported. | TAP | 4.53 + −1.09 | 4.83 + −1.24 | 3.36 + −1.43 | 4.14 + −1.57 | 4.4 + −1.65 | 3.10 + −1.69 | - |
| Gallucci et al. (2020) [45] | 90 (50% males) | Sham-controlled, RCT, double-blind, between-subject design, two experimental conditions | tDCS | Right or left VLPFC | Anode: right VLPFC (F6) or anode: left VLPFC (F5); sham, randomized target area (left/right); cathodal reference electrode placed over contralateral supraorbital area | Anodal or sham | 20 min (10 s ramp up/down) | 1.5 mA | Mild physical side effects reported. | CRTT, Sequence choosing task, Tangram Task | M = −0.28 + −0.14 | N/A | N/A | M = 0.24 + −0.14 * | N/A | N/A | Total aggression d = 3.71 (+) |
| Gilam et al. (2018) [13] | 25 (40% males) | Sham-controlled, RCT, within-subject design | tDCS | VMPFC | Anode: placed vertically over forehead (side-edges equidistant from eyes, lower edge at nasion line, fixed with head sweat-band), cathode: extra-cephalically placed on right shoulder and fixed with elastic band-aid | Anodal or sham | 22 min (30 s ramp up/down) | 1.5 mA | Minor increase in stress level in participants who sensed the stimulation. | TAP | M = 1.92 + −2.78 | N/A | N/A | M = −1.00 + −3.38 * | N/A | N/A | Total aggression d = 0.95 (−) |
| Hortensius et al. (2012) [50] | 60 (50% males) | Sham-controlled, RCT, double-blind, between-subject design, two experimental conditions | tDCS | Right or left DLPFC | Bilateral montage: anode left DLPFC (F3) and cathode right DLPFC (F4) and vice-versa | Anodal and cathodal or sham | 15 min (5 s ramp up/down) | 2 mA | None reported. | TAP | N/A | N/A | N/A | F(3, 56) = 6.47, p = 0.001 *** | N/A | N/A | Total aggression d = 1.19 *** (+) |
| Molero-Chamizo et al. (2019) [22] | 41 (100% males) | Sham-controlled, RCT, mixed design (within- and between-subject) | tDCS | Bilateral DLPFC | Anodes: bilaterally over left DLPFC (F3) and right DLPFC (F4), cathodes: supraorbital ridges (Fp2 and Fp1) | Anodal or sham | 15 min (10 s ramp up/down) | 1.5 mA | Mild physical side effects reported. | Buss Perry Aggression Questionnaire | N/A | N/A | N/A | p = 0.001 physical aggression * p = 0.002 verbal aggression * | N/A | N/A | Physical aggression d = 1.24 (−) Verbal aggression d = 1.11 (−) |
| Perach-Barzilay et al. (2013) [27] | 18 (77% males) | Sham-controlled, RCT, within-subject design, two experimental conditions | cTBS | Right or left DLPFC | 5 cm rule first identifying the motor spot (site in which a single TMS produces maximal amplitude of motor response of the APB muscle) and then moving the coil 5 cm to the anterior along mid-sagittal line | Inhibitory cTBS | 20 min | triple-pulse 50 Hz bursts delivered at a rate of 5 Hz (200 ms between bursts) | None reported. | Social orientation Paradigm | M = 3.31 + −1.32 | N/A | N/A | M = 4.69 + −1.58 * | N/A | N/A | Total aggression d = 0.95 (+) |
| Riva P, et al. (2015) [57] | 80 (21% males) | Sham-controlled, RCT, between-subject design | tDCS | Right VLPFC | Anode: right VLPFC (F6), cathode: contralateral supraorbital area | Anodal or sham | 20 min (8 s ramp up and 5 s ramp down) | 1.5 mA | Mild physical side effects reported. | Hot Sauce Paradigm | N/A | N/A | N/A | F(1,76) = 2.30, p < 0.14 **** | N/A | N/A | Total aggression d = 0.62 **** (−) |
| Riva P, et al. (2017) [51] | 79 (53% males) | Sham-controlled, RCT, mixed design (sham-controlled between-subject, aggression type within-subject) | tDCS | Right VLPFC | Anode: right VLPFC (F6), cathode: contralateral supraorbital area | Anodal or sham | 20 min (8 s ramp up/down) | 1.5 mA | Mild physical side effects reported. | TAP | M = 4.63 + −1.31 | N/A | N/A | M = 5.32 + −1.96 * | N/A | N/A | Total aggression d = 0.42 (−) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Knehans, R.; Schuhmann, T.; Roef, D.; Nelen, H.; à Campo, J.; Lobbestael, J. Modulating Behavioural and Self-Reported Aggression with Non-Invasive Brain Stimulation: A Literature Review. Brain Sci. 2022, 12, 200. https://doi.org/10.3390/brainsci12020200
Knehans R, Schuhmann T, Roef D, Nelen H, à Campo J, Lobbestael J. Modulating Behavioural and Self-Reported Aggression with Non-Invasive Brain Stimulation: A Literature Review. Brain Sciences. 2022; 12(2):200. https://doi.org/10.3390/brainsci12020200
Chicago/Turabian StyleKnehans, Ruben, Teresa Schuhmann, David Roef, Hans Nelen, Joost à Campo, and Jill Lobbestael. 2022. "Modulating Behavioural and Self-Reported Aggression with Non-Invasive Brain Stimulation: A Literature Review" Brain Sciences 12, no. 2: 200. https://doi.org/10.3390/brainsci12020200
APA StyleKnehans, R., Schuhmann, T., Roef, D., Nelen, H., à Campo, J., & Lobbestael, J. (2022). Modulating Behavioural and Self-Reported Aggression with Non-Invasive Brain Stimulation: A Literature Review. Brain Sciences, 12(2), 200. https://doi.org/10.3390/brainsci12020200






