The Differences in the Whole-Brain Functional Network between Cantonese-Mandarin Bilinguals and Mandarin Monolinguals
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Data Acquisition
2.3. Data Preprocessing
2.4. Brain Network Construction
2.5. Network-Based Statistic (NBS) Analysis
2.6. Graph Analysis
2.7. Statistical Analysis of Network Properties
3. Results
3.1. Demographic Characteristics
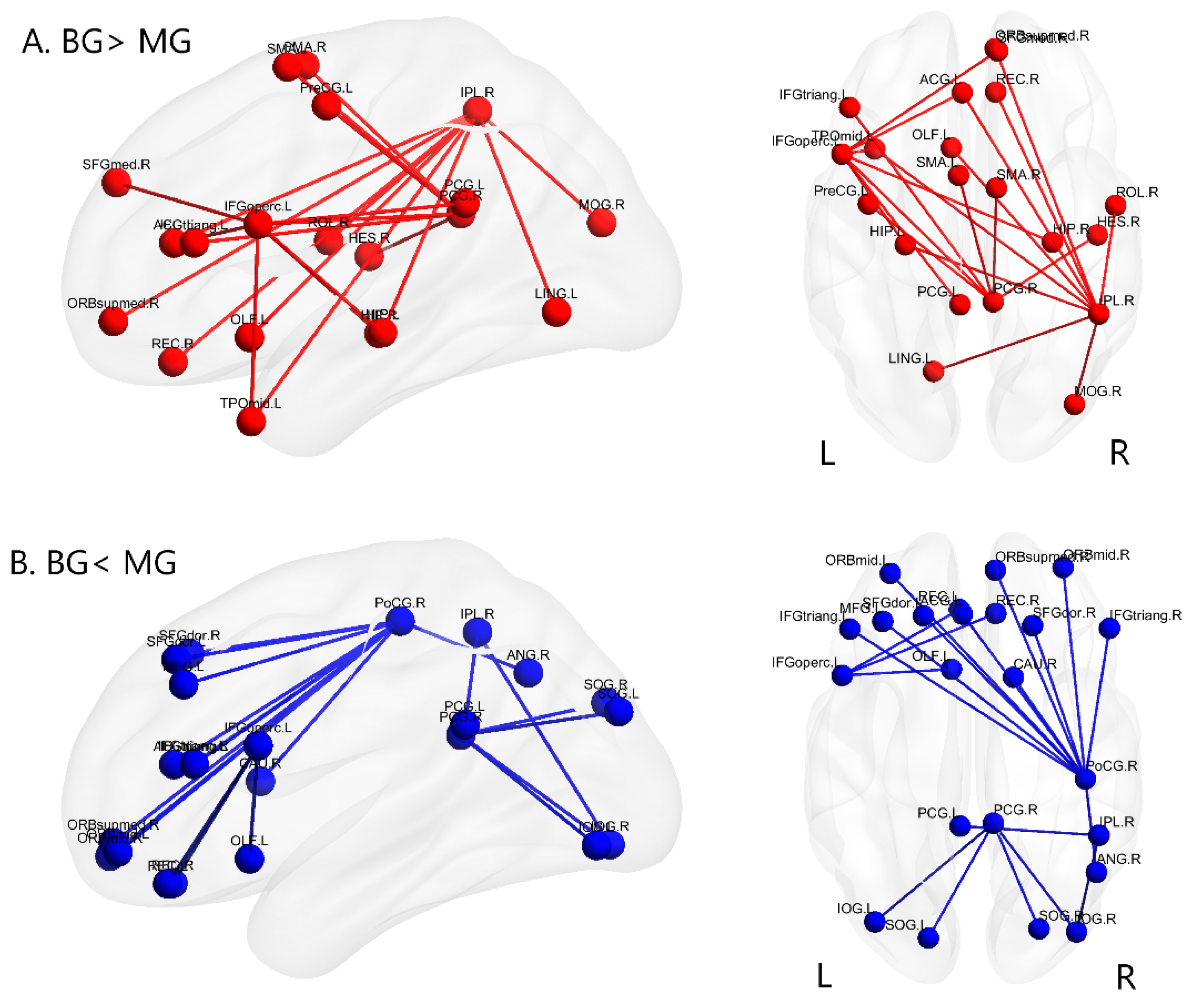
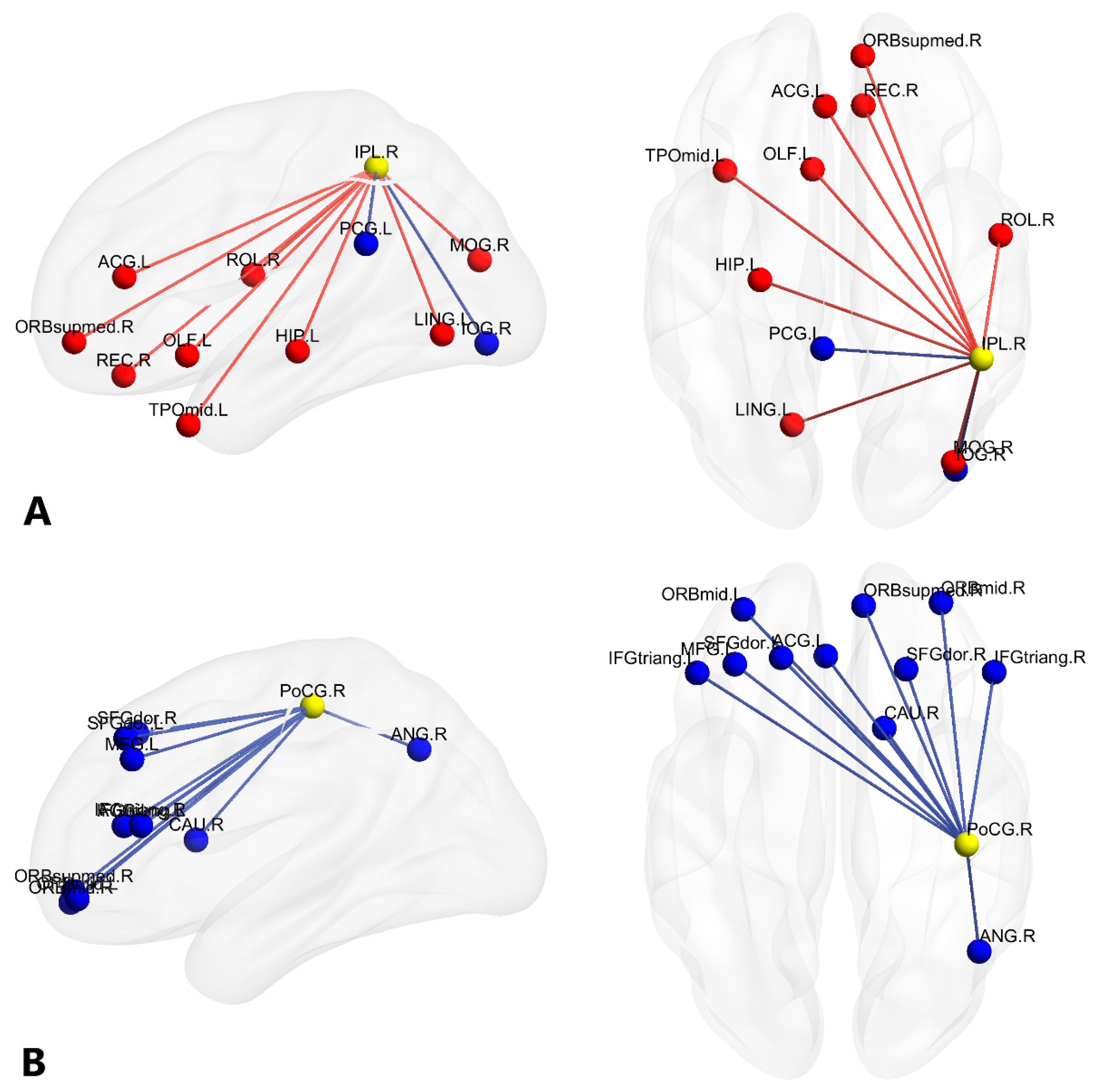
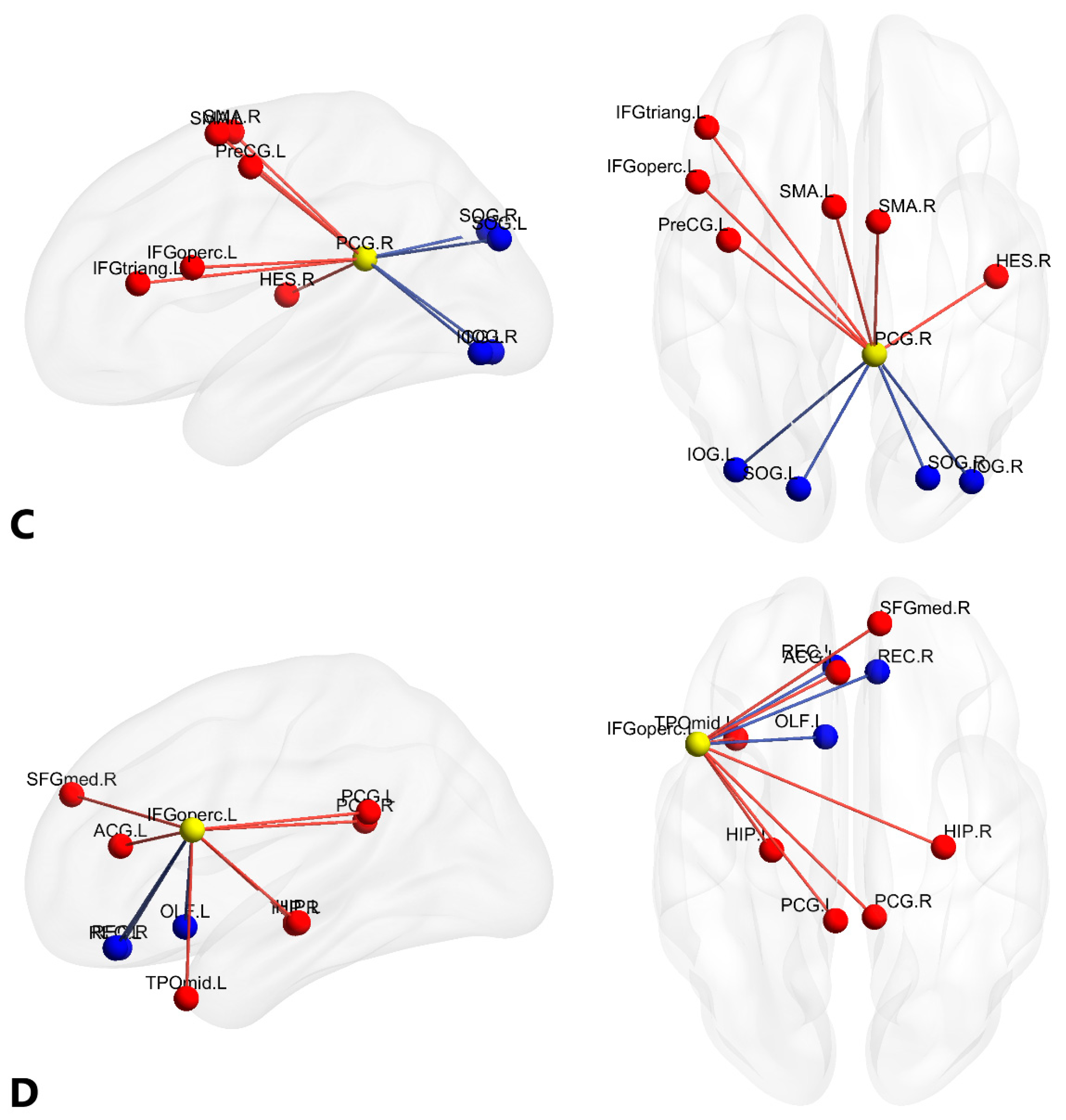
3.2. Alterations of Functional Brain Connectivity in Bilinguals
3.3. Small-Worldness and Efficiency of Brain Functional Networks in Bilinguals
4. Discussion
4.1. Alterations of Functional Network Connectivity in the Cantonese-Mandarin Bilinguals
4.1.1. Brain Networks with Stronger Functional Connectivity in Cantonese-Mandarin Bilinguals
4.1.2. Brain Networks with Weaker Functional Connectivity in Cantonese-Mandarin Bilinguals
4.2. Small-Worldness and Efficiency of the Brain Functional Network in Bilinguals
5. Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bialystok, E.; Craik, F.I.; Luk, G. Bilingualism: Consequences for mind and brain. Trends Cogn. Sci. 2012, 16, 240–250. [Google Scholar] [CrossRef]
- Costa, A.; Sebastián-Gallés, N. How does the bilingual experience sculpt the brain? Nat. Rev. Neurosci. 2014, 15, 336–345. [Google Scholar] [CrossRef]
- Wong, B.; Yin, B.; O’Brien, B. Neurolinguistics: Structure, Function, and Connectivity in the Bilingual Brain. BioMed Res. Int. 2016, 2016, 7069274. [Google Scholar] [CrossRef]
- Grundy, J.G.; Anderson, J.A.; Bialystok, E. Neural correlates of cognitive processing in monolinguals and bilinguals. Ann. N. Y. Acad. Sci. 2017, 1396, 183–201. [Google Scholar] [CrossRef]
- Vinals, L. Second-Language Learning Ability Revealed by Resting-State Functional Connectivity. J. Neurosci. 2016, 36, 6141–6143. [Google Scholar] [CrossRef] [PubMed]
- van den Heuvel, M.P.; Hulshoff Pol, H.E. Exploring the brain network: A review on resting-state fMRI functional connectivity. Eur. Neuropsychopharmacol. 2010, 20, 519–534. [Google Scholar] [CrossRef] [PubMed]
- Lewis, C.M.; Baldassarre, A.; Committeri, G.; Romani, G.L.; Corbetta, M. Learning sculpts the spontaneous activity of the resting human brain. Proc. Natl. Acad. Sci. USA 2009, 106, 17558–17563. [Google Scholar] [CrossRef] [PubMed]
- Biswal, B.B.; Mennes, M.; Zuo, X.-N.; Gohel, S.; Kelly, C.; Smith, S.M.; Beckmann, C.F.; Adelstein, J.S.; Buckner, R.L.; Colcombe, S.; et al. Toward discovery science of human brain function. Proc. Natl. Acad. Sci. USA 2010, 107, 4734–4739. [Google Scholar] [CrossRef] [PubMed]
- Luk, G.; Bialystok, E.; Craik, F.I.M.; Grady, C.L. Lifelong Bilingualism Maintains White Matter Integrity in Older Adults. J. Neurosci. 2011, 31, 16808–16813. [Google Scholar] [CrossRef] [PubMed]
- Grady, C.L.; Luk, G.; Craik, F.I.; Bialystok, E. Brain network activity in monolingual and bilingual older adults. Neuropsychology 2015, 66, 170–181. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Abutalebi, J.; Zou, L.; Yan, X.; Liu, L.; Feng, X.; Wang, R.; Guo, T.; Ding, G. Bilingualism alters brain functional connectivity between “control” regions and “language” regions: Evidence from bimodal bilinguals. Neuropsychology 2015, 71, 236–247. [Google Scholar] [CrossRef]
- Buchweitz, A.; Prat, C. The bilingual brain: Flexibility and control in the human cortex. Phys. Life Rev. 2013, 10, 428–443. [Google Scholar] [CrossRef] [PubMed]
- Bialystok, E. The bilingual adaptation: How minds accommodate experience. Psychol. Bull. 2017, 143, 233–262. [Google Scholar] [CrossRef]
- Friederici, A.D. The Brain Basis of Language Processing: From Structure to Function. Physiol. Rev. 2011, 91, 1357–1392. [Google Scholar] [CrossRef] [PubMed]
- Zalesky, A.; Fornito, A.; Bullmore, E.T. Network-based statistic: Identifying differences in brain networks. NeuroImage 2010, 53, 1197–1207. [Google Scholar] [CrossRef] [PubMed]
- García-Pentón, L.; Fernández, A.P.; Iturria-Medina, Y.; Gillon-Dowens, M.; Carreiras, M. Anatomical connectivity changes in the bilingual brain. NeuroImage 2014, 84, 495–504. [Google Scholar] [CrossRef]
- Zhang, Z.; Liao, W.; Chen, H.; Mantini, D.; Ding, J.-R.; Xu, Q.; Wang, Z.; Yuan, C.; Chen, G.; Jiao, Q.; et al. Altered functional–structural coupling of large-scale brain networks in idiopathic generalized epilepsy. Brain 2011, 134, 2912–2928. [Google Scholar] [CrossRef]
- Heuvel, M.V.D.; Mandl, R.; Luigjes, J.; Pol, H.H. Microstructural Organization of the Cingulum Tract and the Level of Default Mode Functional Connectivity. J. Neurosci. 2008, 28, 10844–10851. [Google Scholar] [CrossRef]
- Greicius, M.D.; Supekar, K.; Menon, V.; Dougherty, R.F. Resting-State Functional Connectivity Reflects Structural Connectivity in the Default Mode Network. Cereb. Cortex 2008, 19, 72–78. [Google Scholar] [CrossRef]
- Skudlarski, P.; Jagannathan, K.; Anderson, K.; Stevens, M.C.; Calhoun, V.D.; Skudlarska, B.A.; Pearlson, G. Brain Connectivity Is Not Only Lower but Different in Schizophrenia: A Combined Anatomical and Functional Approach. Biol. Psychiatry 2010, 68, 61–69. [Google Scholar] [CrossRef]
- Zhu, L.; Nie, Y.; Chang, C.; Gao, J.-H.; Niu, Z. Different patterns and development characteristics of processing written logographic characters and alphabetic words: An ALE meta-analysis. Hum. Brain Mapp. 2013, 35, 2607–2618. [Google Scholar] [CrossRef] [PubMed]
- Bolger, D.J.; Perfetti, C.A.; Schneider, W. Cross-cultural effect on the brain revisited: Universal structures plus writing system variation. Hum. Brain Mapp. 2005, 25, 92–104. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.H.; Laird, A.R.; Li, K.; Fox, P.T. Neuroanatomical correlates of phonological processing of Chinese characters and alphabetic words: A meta-analysis. Hum. Brain Mapp. 2005, 25, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-Y.; Ho, M.-H.R.; Chen, S.-H.A. A meta-analysis of fMRI studies on Chinese orthographic, phonological, and semantic processing. NeuroImage 2012, 63, 381–391. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Wu, Y.; Sun, T.; Cai, L.; Fan, X.; Li, X. Neural substrates of bilingual processing in a logographic writing system: An fMRI study in Chinese Cantonese-Mandarin bilinguals. Brain Res. 2020, 1738, 146794. [Google Scholar] [CrossRef]
- Cai, Z.G.; Pickering, M.J.; Yan, H.; Branigan, H.P. Lexical and syntactic representations in closely related languages: Evidence from Cantonese–Mandarin bilinguals. J. Mem. Lang. 2011, 65, 431–445. [Google Scholar] [CrossRef]
- Tu, L.; Ruiwang, H.; Abutalebi, J.; Jiang, B.; Pan, X.; Li, M.; Gao, W.; Yang, Y.; Liang, B.; Lu, Z.; et al. Language exposure induced neuroplasticity in the bilingual brain: A follow-up fMRI study. Cortex 2015, 64, 8–19. [Google Scholar] [CrossRef] [PubMed]
- Newman, M.E.J. Communities, modules and large-scale structure in networks. Nat. Phys. 2011, 8, 25–31. [Google Scholar] [CrossRef]
- Sporns, O. Network attributes for segregation and integration in the human brain. Curr. Opin. Neurobiol. 2013, 23, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.A.E.; Mak, L.; Keyvani Chahi, A.; Bialystok, E. The language and social background questionnaire: Assessing degree of bilingualism in a diverse population. Behav. Res. Methods 2018, 50, 250–263. [Google Scholar] [CrossRef] [PubMed]
- Morton, J.B.; Harper, S.N. Bilinguals show an advantage in cognitive control—The question is why. Dev. Sci. 2009, 12, 502–503. [Google Scholar] [CrossRef]
- Zhang, M.; Li, J.; Chen, C.; Xue, G.; Lu, Z.; Mei, L.; Xue, H.; Xue, F.; He, Q.; Chen, C.; et al. Resting-state functional connectivity and reading abilities in first and second languages. NeuroImage 2014, 84, 546–553. [Google Scholar] [CrossRef]
- Sun, X.; Li, L.; Ding, G.; Wang, R.; Li, P. Effects of language proficiency on cognitive control: Evidence from resting-state functional connectivity. Neuropsychologia 2019, 129, 263–275. [Google Scholar] [CrossRef]
- Goksan, S.; Argyri, F.; Clayden, J.D.; Liegeois, F.; Wei, L. Early childhood bilingualism: Effects on brain structure and function. F1000Research 2020, 9, 370. [Google Scholar] [CrossRef]
- Oldfield, R.C. The assessment and analysis of handedness: The Edinburgh inventory. Neuropsychologia 1971, 9, 97–113. [Google Scholar] [CrossRef]
- Fox, M.D.; Zhang, D.; Snyder, A.Z.; Raichle, M.E. The Global Signal and Observed Anticorrelated Resting State Brain Networks. J. Neurophysiol. 2009, 101, 3270–3283. [Google Scholar] [CrossRef] [PubMed]
- Murphy, K.; Birn, R.M.; Handwerker, D.A.; Jones, T.B.; Bandettini, P.A. The impact of global signal regression on resting state correlations: Are anti-correlated networks introduced? NeuroImage 2009, 44, 893–905. [Google Scholar] [CrossRef] [PubMed]
- Tzourio-Mazoyer, N.; Landeau, B.; Papathanassiou, D.; Crivello, F.; Etard, O.; Delcroix, N.; Mazoyer, B.; Joliot, M. Automated Anatomical Labeling of Activations in SPM Using a Macroscopic Anatomical Parcellation of the MNI MRI Single-Subject Brain. NeuroImage 2002, 15, 273–289. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, J.; Wu, Q.; Kuang, W.; Huang, X.; He, Y.; Gong, Q. Disrupted Brain Connectivity Networks in Drug-Naive, First-Episode Major Depressive Disorder. Biol. Psychiatry 2011, 70, 334–342. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Bai, L.; Liang, P.; Kang, S.; Gao, H.; Yang, H. Disrupted brain connectivity networks in acute ischemic stroke patients. Brain Imaging Behav. 2017, 11, 444–453. [Google Scholar] [CrossRef]
- Zhan, C.; Chen, H.-J.; Gao, Y.-Q.; Zou, T.-X. Functional Network-Based Statistics Reveal Abnormal Resting-State Functional Connectivity in Minimal Hepatic Encephalopathy. Front. Neurol. 2019, 10, 33. [Google Scholar] [CrossRef]
- Zalesky, A.; Fornito, A.; Harding, I.H.; Cocchi, L.; Yücel, M.; Pantelis, C.; Bullmore, E.T. Whole-brain anatomical networks: Does the choice of nodes matter? NeuroImage 2010, 50, 970–983. [Google Scholar] [CrossRef]
- Cohen, J.; Cohen, P. Chapter 2: Bivariate Correlation and Regression. In Applied Multiple Regression/Correlation Analysis for the Behavioral Sciences, 3rd ed.; Lawrence Erlbaum Associates: Mahwah, NJ, USA, 2003; pp. 23–28. [Google Scholar]
- Wang, J.; Zuo, X.; Dai, Z.; Xia, M.; Zhao, Z.; Zhao, X.; Jia, J.; Han, Y.; He, Y. Disrupted Functional Brain Connectome in Individuals at Risk for Alzheimer’s Disease. Biol. Psychiatry 2013, 73, 472–481. [Google Scholar] [CrossRef]
- Yang, X.; Liu, J.; Meng, Y.; Xia, M.; Cui, Z.; Wu, X.; Hu, X.; Zhang, W.; Gong, G.; Gong, Q.; et al. Network analysis reveals disrupted functional brain circuitry in drug-naive social anxiety disorder. NeuroImage 2019, 190, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, T.; Wang, N.; Xian, J.; He, H. Graph theoretical analysis reveals the reorganization of the brain network pattern in primary open angle glaucoma patients. Eur. Radiol. 2016, 26, 3957–3967. [Google Scholar] [CrossRef] [PubMed]
- Bullmore, E.T.; Sporns, O. Complex brain networks: Graph theoretical analysis of structural and functional systems. Nat. Rev. Neurosci. 2009, 10, 186–198. [Google Scholar] [CrossRef]
- Watts, D.J.; Strogatz, S.H. Collective dynamics of ‘small-world’ networks. Nat. Cell Biol. 1998, 393, 440–442. [Google Scholar] [CrossRef] [PubMed]
- Bassett, D.S.; Bullmore, E. Small-World Brain Networks. Neuroscientist 2006, 12, 512–523. [Google Scholar] [CrossRef] [PubMed]
- Sporns, O.; Honey, C.J. Small worlds inside big brains. Proc. Natl. Acad. Sci. USA 2006, 103, 19219–19220. [Google Scholar] [CrossRef]
- Humphries, M.D.; Gurney, K. Network ‘Small-World-Ness’: A Quantitative Method for Determining Canonical Network Equivalence. PLoS ONE 2008, 3, e0002051. [Google Scholar] [CrossRef]
- Latora, V.; Marchiori, M. Efficient Behavior of Small-World Networks. Phys. Rev. Lett. 2001, 87, 198701. [Google Scholar] [CrossRef]
- Nichols, T.E.; Holmes, A.P. Nonparametric permutation tests for functional neuroimaging: A primer with examples. Hum. Brain Mapp. 2002, 15, 1–25. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate—A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Groppe, D.M.; Urbach, T.P.; Kutas, M. Mass univariate analysis of event-related brain potentials/fields I: A critical tutorial review. Psychophysiology 2011, 48, 1711–1725. [Google Scholar] [CrossRef] [PubMed]
- Finn, E.S.; Shen, X.; Holahan, J.M.; Scheinost, D.; Lacadie, C.; Papademetris, X.; Shaywitz, S.E.; Shaywitz, B.A.; Constable, R.T. Disruption of Functional Networks in Dyslexia: A Whole-Brain, Data-Driven Analysis of Connectivity. Biol. Psychiatry 2014, 76, 397–404. [Google Scholar] [CrossRef] [PubMed]
- Wen, T.; Hsieh, S. Network-Based Analysis Reveals Functional Connectivity Related to Internet Addiction Tendency. Front. Hum. Neurosci. 2016, 10. [Google Scholar] [CrossRef]
- Vigneau, M.; Beaucousin, V.; Hervé, P.-Y.; Jobard, G.; Petit, L.; Crivello, F.; Mellet, E.; Zago, L.; Mazoyer, B.; Tzouriomazoyer, N. What is right-hemisphere contribution to phonological, lexico-semantic, and sentence processing? NeuroImage 2011, 54, 577–593. [Google Scholar] [CrossRef] [PubMed]
- Yang, J. The role of the right hemisphere in metaphor comprehension: A meta-analysis of functional magnetic resonance imaging studies. Hum. Brain Mapp. 2012, 35, 107–122. [Google Scholar] [CrossRef] [PubMed]
- Hull, R.; Vaid, J. Bilingual language lateralization: A meta-analytic tale of two hemispheres. Neuropsychology 2007, 45, 1987–2008. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Tu, L.; Wang, J.; Jiang, B.; Gao, W.; Pan, X.; Li, M.; Zhong, M.; Zhu, Z.; Niu, M.; et al. Onset age of L2 acquisition influences language network in early and late Cantonese-Mandarin bilinguals. Brain Lang. 2017, 174, 16–28. [Google Scholar] [CrossRef]
- Booth, J.R.; Lu, D.; Burman, D.D.; Chou, T.-L.; Jin, Z.; Peng, D.-L.; Zhang, L.; Ding, G.-S.; Deng, Y.; Liu, L. Specialization of phonological and semantic processing in Chinese word reading. Brain Res. 2006, 1071, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Burgess, P.W.; Dumontheil, I.; Gilbert, S.J. The gateway hypothesis of rostral prefrontal cortex (area 10) function. Trends Cogn. Sci. 2007, 11, 290–298. [Google Scholar] [CrossRef]
- Whitfield-Gabrieli, S.; Ford, J.M. Default Mode Network Activity and Connectivity in Psychopathology. Annu. Rev. Clin. Psychol. 2012, 8, 49–76. [Google Scholar] [CrossRef]
- Marcotte, K.; Perlbarg, V.; Marrelec, G.; Benali, H.; Ansaldo, A.I. Default-mode network functional connectivity in aphasia: Therapy-induced neuroplasticity. Brain Lang. 2013, 124, 45–55. [Google Scholar] [CrossRef]
- Geranmayeh, F.; Wise, R.J.; Mehta, A.; Leech, R. Overlapping Networks Engaged during Spoken Language Production and Its Cognitive Control. J. Neurosci. 2014, 34, 8728–8740. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Fornells, A.; van der Lugt, A.; Rotte, M.; Britti, B.; Heinze, H.-J.; Münte, T.F. Second language interferes with word production in fluent bilinguals: Brain potential and functional imaging evidence. J. Cognit. Neurosci. 2005, 17, 422–433. [Google Scholar] [CrossRef]
- Sulpizio, S.; Del Maschio, N.; Fedeli, D.; Abutalebi, J. Bilingual language processing: A meta-analysis of functional neuroimaging studies. Neurosci. Biobehav. Rev. 2020, 108, 834–853. [Google Scholar] [CrossRef]
- Rodriguez-Fornells, A.; Rotte, M.; Heinze, H.-J.; Nösselt, T.; Münte, T.F. Brain potential and functional MRI evidence for how to handle two lan-guages with one brain. Nature 2002, 415, 1026–1029. [Google Scholar] [CrossRef]
- Abutalebi, J.; Canini, M.; Della Rosa, P.A.; Green, D.W.; Weekes, B.S. The neuroprotective effects of bilingualism upon the inferior parietal lobule: A Structural Neuroimaging Study in Aging Chinese Bilinguals. J. Neurolinguistics 2015, 33, 3–13. [Google Scholar] [CrossRef]
- Olulade, O.A.; Jamal, N.I.; Koo, D.S.; Perfetti, C.A.; LaSasso, C.; Eden, G.F. Neuroanatomical Evidence in Support of the Bilingual Advantage Theory. Cereb. Cortex 2015, 26, 3196–3204. [Google Scholar] [CrossRef] [PubMed]
- Abutalebi, J.; Green, D.W. Neuroimaging of language control in bilinguals: Neural adaptation and reserve. Biling. Lang. Cogn. 2016, 19, 689–698. [Google Scholar] [CrossRef]
- Tabu, H.; Mima, T.; Aso, T.; Takahashi, R.; Fukuyama, H. Functional relevance of pre-supplementary motor areas for the choice to stop during Stop signal task. Neurosci. Res. 2011, 70, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Swann, N.C.; Cai, W.; Conner, C.R.; Pieters, T.A.; Claffey, M.P.; George, J.S.; Aron, A.R.; Tandon, N. Roles for the pre-supplementary motor area and the right inferior frontal gyrus in stopping action: Electrophysiological responses and functional and structural connectivity. NeuroImage 2012, 59, 2860–2870. [Google Scholar] [CrossRef]
- Abutalebi, J.; Annoni, J.-M.; Zimine, I.; Pegna, A.J.; Seghier, M.L.; Lee-Jahnke, H.; Lazeyras, F.; Cappa, S.F.; Khateb, A. Language Control and Lexical Competition in Bilinguals: An Event-Related fMRI Study. Cereb. Cortex 2007, 18, 1496–1505. [Google Scholar] [CrossRef] [PubMed]
- Abutalebi, J.; Della Rosa, P.A.; Green, D.W.; Hernandez, M.; Scifo, P.; Keim, R.; Cappa, S.F.; Costa, A. Bilingualism Tunes the Anterior Cingulate Cortex for Conflict Monitoring. Cereb. Cortex 2012, 22, 2076–2086. [Google Scholar] [CrossRef]
- Rodríguez-Pujadas, A.; Sanjuán, A.; Fuentes, P.; Ventura-Campos, N.; Barrós-Loscertales, A.; Ávila, C. Differential neural control in early bilinguals and monolinguals during response inhibition. Brain Lang. 2014, 132, 43–51. [Google Scholar] [CrossRef]
- Gratton, G.; Cooper, P.; Fabiani, M.; Carter, C.S.; Karayanidis, F. Dynamics of cognitive control: Theoretical bases, paradigms, and a view for the future. Psychophysiology 2018, 55, e13016. [Google Scholar] [CrossRef]
- Price, C.J. The anatomy of language: A review of 100 fMRI studies published in 2009. Ann. N. Y. Acad. Sci. 2010, 1191, 62–88. [Google Scholar] [CrossRef] [PubMed]
- Hickok, G.; Poeppel, D. Dorsal and ventral streams: A framework for understanding aspects of the functional anatomy of language. Cognition 2004, 92, 67–99. [Google Scholar] [CrossRef]
- Hickok, G.; Poeppel, D. The cortical organization of speech processing. Nat. Rev. Neurosci. 2007, 8, 393–402. [Google Scholar] [CrossRef]
- Yang, J.; Small, S.L. Language Processing, Functional Magnetic Resonance Imaging of. In International Encyclopedia of the Social & Behavioral Sciences, 2nd ed.; Wright, J.D., Ed.; Elsevier: Oxford, UK, 2015; pp. 368–380. [Google Scholar]
- Jacquemot, C.; Pallier, C.; LeBihan, D.; Dehaene, S.; Dupoux, E. Phonological Grammar Shapes the Auditory Cortex: A Functional Magnetic Resonance Imaging Study. J. Neurosci. 2003, 23, 9541–9546. [Google Scholar] [CrossRef] [PubMed]
- Golestani, N.; Molko, N.; Dehaene, S.; LeBihan, D.; Pallier, C. Brain Structure Predicts the Learning of Foreign Speech Sounds. Cereb. Cortex 2006, 17, 575–582. [Google Scholar] [CrossRef] [PubMed]
- Klooster, N.B.; Tranel, D.; Duff, M.C. The hippocampus and semantic memory over time. Brain Lang. 2020, 201, 104711. [Google Scholar] [CrossRef] [PubMed]
- Parker Jones, Ō.; Green, D.W.; Grogan, A.; Pliatsikas, C.; Filippopolitis, K.; Ali, N.; Lee, H.L.; Ramsden, S.; Gazarian, K.; Prejawa, S.; et al. Where, When and Why Brain Activation Differs for Bilinguals and Monolinguals during Picture Naming and Reading Aloud. Cereb. Cortex 2011, 22, 892–902. [Google Scholar] [CrossRef] [PubMed]
- Román, P.; González, J.; Campos, N.V.; Pujadas, A.R.; Sanjuán, A.; Ávila, C. Neural differences between monolinguals and early bilinguals in their native language during comprehension. Brain Lang. 2015, 150, 80–89. [Google Scholar] [CrossRef]
- Li, P.; Legault, J.; Litcofsky, K.A. Neuroplasticity as a function of second language learning: Anatomical changes in the human brain. Cortex 2014, 58, 301–324. [Google Scholar] [CrossRef]
- Li, L.; Abutalebi, J.; Emmorey, K.; Gong, G.; Yan, X.; Feng, X.; Zou, L.; Ding, G. How bilingualism protects the brain from aging: Insights from bimodal bilinguals. Hum. Brain Mapp. 2017, 38, 4109–4124. [Google Scholar] [CrossRef]
- Ressel, V.; Pallier, C.; Ventura-Campos, N.; Díaz, B.; Roessler, A.; Ávila, C.; Sebastián-Gallés, N. An Effect of Bilingualism on the Auditory Cortex. J. Neurosci. 2012, 32, 16597–16601. [Google Scholar] [CrossRef]
- Green, D.W. Mental control of the bilingual lexico-semantic system. Biling. Lang. Cogn. 1998, 1, 67–81. [Google Scholar] [CrossRef]
- Thierry, G.; Wu, Y.J. Brain potentials reveal unconscious translation during foreign-language comprehension. Proc. Natl. Acad. Sci. USA 2007, 104, 12530–12535. [Google Scholar] [CrossRef]
- Wu, Y.J.; Thierry, G. Chinese-English Bilinguals Reading English Hear Chinese. J. Neurosci. 2010, 30, 7646–7651. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.J.; Thierry, G. Brain potentials predict language selection before speech onset in bilinguals. Brain Lang. 2017, 171, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Carreiras, M.; Seghier, M.L.; Baquero, S.; Estévez, A.; Lozano, A.; Devlin, J.T.; Price, C.J. An anatomical signature for literacy. Nat. Cell Biol. 2009, 461, 983–986. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.H.; Spinks, J.A.; Gao, J.-H.; Liu, H.-L.; Perfetti, C.A.; Xiong, J.; Stofer, K.A.; Pu, Y.; Liu, Y.; Fox, P.T. Brain activation in the processing of Chinese characters and words: A functional MRI study. Hum. Brain Mapp. 2000, 10, 16–27. [Google Scholar] [CrossRef]
- Siok, W.T.; Perfetti, C.A.; Jin, Z.; Tan, L.H. Biological abnormality of impaired reading is constrained by culture. Nature 2004, 431, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Cao, F.; Lee, R.; Shu, H.; Yang, Y.; Xu, G.; Li, K.; Booth, J.R. Cultural Constraints on Brain Development: Evidence from a Developmental Study of Visual Word Processing in Mandarin Chinese. Cereb. Cortex 2009, 20, 1223–1233. [Google Scholar] [CrossRef]
- Cao, F.; Khalid, K.; Lee, R.; Brennan, C.; Yang, Y.; Li, K.; Bolger, D.J.; Booth, J.R. Development of brain networks involved in spoken word processing of Mandarin Chinese. NeuroImage 2011, 57, 750–759. [Google Scholar] [CrossRef]
- Cao, F.; Vu, M.; Chan, D.H.L.; Lawrence, J.M.; Harris, L.N.; Guan, Q.; Xu, Y.; Perfetti, C.A. Writing affects the brain network of reading in Chinese: A functional magnetic resonance imaging study. Hum. Brain Mapp. 2012, 34, 1670–1684. [Google Scholar] [CrossRef]
- Lagarrigue, A.; Longcamp, M.; Anton, J.L.; Nazarian, B.; Prévot, L.; Velay, J.-L.; Cao, F.; Frenck-Mestre, C. Activation of writing-specific brain regions when reading Chinese as a second language. Effects of training modality and transfer to novel characters. Neuropsychology 2017, 97, 83–97. [Google Scholar] [CrossRef]
- Tan, L.H.; Feng, C.M.; Fox, P.T.; Gao, J.H. An fMRI study with written Chinese. Neuroreport 2001, 12, 83–88. [Google Scholar] [CrossRef]
- Seghier, M.L. The Angular Gyrus. Neuroscientist 2012, 19, 43–61. [Google Scholar] [CrossRef]
- Siok, W.T.; Jin, Z.; Fletcher, P.; Tan, L.H. Distinct brain regions associated with syllable and phoneme. Hum. Brain Mapp. 2003, 18, 201–207. [Google Scholar] [CrossRef]
- Pulvermüller, F.; Huss, M.; Kherif, F.; Martin, F.M.D.P.; Hauk, O.; Shtyrov, Y. Motor cortex maps articulatory features of speech sounds. Proc. Natl. Acad. Sci. USA 2006, 103, 7865–7870. [Google Scholar] [CrossRef]
- Grabski, K.; Lamalle, L.; Vilain, C.; Schwartz, J.-L.; Vallée, N.; Tropres, I.; Baciu, M.V.; Le Bas, J.-F.; Sato, M. Functional MRI assessment of orofacial articulators: Neural correlates of lip, jaw, larynx, and tongue movements. Hum. Brain Mapp. 2011, 33, 2306–2321. [Google Scholar] [CrossRef] [PubMed]
- Bouchard, K.E.; Mesgarani, N.; Johnson, K.A.; Chang, E.F. Functional organization of human sensorimotor cortex for speech articulation. Nat. Cell Biol. 2013, 495, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Elmer, S.; Hänggi, J.; Jäncke, L. Processing demands upon cognitive, linguistic, and articulatory functions promote grey matter plasticity in the adult multilingual brain: Insights from simultaneous interpreters. Cortex 2014, 54, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Hebb, D.O. The Organization of Behavior: A Neuropsychological Theory; Wiley: New York, NY, USA, 1949. [Google Scholar]
- Paulsen, O.; Sejnowski, T.J. Natural patterns of activity and long-term synaptic plasticity. Curr. Opin. Neurobiol. 2000, 10, 172–179. [Google Scholar] [CrossRef]
- Song, S.; Miller, K.D.; Abbott, L.F. Competitive Hebbian learning through spike-timing-dependent synaptic plasticity. Nat. Neurosci. 2000, 3, 919–926. [Google Scholar] [CrossRef]
- Gollan, T.H.; Acenas, L.-A.R. What Is a TOT? Cognate and Translation Effects on Tip-of-the-Tongue States in Spanish-English and Tagalog-English Bilinguals. J. Exp. Psychol. Learn. Mem. Cogn. 2004, 30, 246–269. [Google Scholar] [CrossRef]
- Gollan, T.H.; Montoya, R.I.; Cera, C.; Sandoval, T.C. More use almost always a means a smaller frequency effect: Aging, bilin-gualism, and the weaker links hypothesis. J. Memory Language 2008, 58, 787–814. [Google Scholar] [CrossRef]
- Pyers, J.E.; Gollan, T.H.; Emmorey, K. Bimodal bilinguals reveal the source of tip-of-the-tongue states. Cognition 2009, 112, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, L.; Zang, Y.; Yang, H.; Tang, H.; Gong, Q.; Chen, Z.; Zhu, C.; He, Y. Parcellation-dependent small-world brain functional networks: A resting-state fMRI study. Hum. Brain Mapp. 2008, 30, 1511–1523. [Google Scholar] [CrossRef] [PubMed]

| BG | MG | p Value | |
|---|---|---|---|
| N | 30 | 30 | -- |
| Gender (male/female) | 7/23 | 9/21 | 0.559 a |
| Age (years, M ± SD) | 21.00 ± 1.93 | 21.43 ± 2.05 | 0.402 b |
| IQ (M ± SD) | 122.50 ± 11.92 | 124.07 ± 14.10 | 0.644 b |
| Education (undergraduate/postgraduate) | 22/8 | 20/10 | 0.573 a |
| District of residence (city/suburb) | 20/10 | 19/11 | 0.787 a |
| Father’s education | |||
| Junior high school or below | 13 | 12 | 0.957 a |
| Senior high school or technical secondary school | 7 | 7 | |
| College or above | 10 | 11 | |
| Mother’s education | |||
| Junior high school or below | 11 | 14 | 0.542 a |
| Senior high school or technical secondary school | 7 | 8 | |
| College or above | 12 | 8 | |
| Cantonese proficiency (M ± SD) | |||
| Speakingc | 9.40 ± 0.77 | - | |
| Writingc | 5.77 ± 2.13 | - | |
| Understandingc | 8.87 ± 0.94 | - | |
| Readingc | 7.87 ± 1.33 | - | |
| Mandarin proficiency (M ± SD) | |||
| Speakingc | 8.53 ± 1.17 | 8.83 ± 1.18 | 0.326 b |
| Writingc | 8.67 ± 1.12 | 8.73 ± 1.26 | 0.829 b |
| Understandingc | 8.83 ± 1.12 | 8.77 ± 1.17 | 0.822 b |
| Readingc | 8.93 ± 0.98 | 9.00 ± 0.91 | 0.786 b |
| English level (CET-4/CET-6) | 14/16 | 13/17 | 0.795 a |
| Node | Region | Coordinate (x, y, z) | BG > MG Edges | MG > BG Edges | Sum of Edges |
|---|---|---|---|---|---|
| A a | IPL.R | (46.46, −46.29, 49.54) | 9 | 2 | 11 |
| B a | PoCG.R | (41.43, −25.49, 52.55) | 0 | 11 | 11 |
| C a | PCG.R | (7.44, −41.81, 21.87) | 6 | 4 | 10 |
| D a | IFGoperc.L | (−48.43, 12.73, 19.02) | 7 | 3 | 10 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fan, X.; Wu, Y.; Cai, L.; Ma, J.; Pan, N.; Xu, X.; Sun, T.; Jing, J.; Li, X. The Differences in the Whole-Brain Functional Network between Cantonese-Mandarin Bilinguals and Mandarin Monolinguals. Brain Sci. 2021, 11, 310. https://doi.org/10.3390/brainsci11030310
Fan X, Wu Y, Cai L, Ma J, Pan N, Xu X, Sun T, Jing J, Li X. The Differences in the Whole-Brain Functional Network between Cantonese-Mandarin Bilinguals and Mandarin Monolinguals. Brain Sciences. 2021; 11(3):310. https://doi.org/10.3390/brainsci11030310
Chicago/Turabian StyleFan, Xiaoxuan, Yujia Wu, Lei Cai, Jingwen Ma, Ning Pan, Xiaoyu Xu, Tao Sun, Jin Jing, and Xiuhong Li. 2021. "The Differences in the Whole-Brain Functional Network between Cantonese-Mandarin Bilinguals and Mandarin Monolinguals" Brain Sciences 11, no. 3: 310. https://doi.org/10.3390/brainsci11030310
APA StyleFan, X., Wu, Y., Cai, L., Ma, J., Pan, N., Xu, X., Sun, T., Jing, J., & Li, X. (2021). The Differences in the Whole-Brain Functional Network between Cantonese-Mandarin Bilinguals and Mandarin Monolinguals. Brain Sciences, 11(3), 310. https://doi.org/10.3390/brainsci11030310






