Compensatory and Lifestyle-Based Brain Health Program for Subjective Cognitive Decline: Self-Implementation versus Coaching
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
- (1)
- Age 50 or older
- (2)
- A positive complaint or concern in response to two questions: “Do you feel like your memory or other aspects of thinking are becoming worse?” and “Does this worry you?”
- (3)
- Normal cognitive performance on the Montreal Cognitive Assessment [32] (adjusted for age and education)
- (4)
- Self-reported independent function in daily life as measured by the Lawton IADL scale (IADL = 8) [33]
- (5)
- English speaking
- (6)
- Approval letter from a physician (due to exercise component)
- (1)
- Known neurological disorder with potential cognitive symptoms (Parkinson’s disease, epilepsy, history of traumatic brain injury, etc.)
- (2)
- Uncontrolled moderate or severe depression (e.g., CES-D > 21)
2.2. Intervention
2.2.1. The Executive and Memory Support (EMS) System
- (2)
- Goal Planning
- (3)
- Organization Systems for the Environment
2.2.2. Training in Healthy Lifestyle
- (1)
- Physical Exercise
- (2)
- Cognitive Stimulation
- (3)
- Stress management
2.3. Outcomes
2.3.1. Primary Outcomes
2.3.2. Secondary Outcomes
2.4. Statistical Analyses
3. Results
3.1. Primary Outcome Measures
3.2. Secondary Outcomes
3.3. Cognitive Outcomes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Klimova, B.; Valis, M.; Kuca, K. Cognitive decline in normal aging and its prevention: A review on non-pharmacological lifestyle strategies. Clin. Interv. Aging 2017, 12, 903–910. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brain Health. Available online: https://www.who.int/health-topics/brain-health#tab=tab_1 (accessed on 16 June 2021).
- Baur, R.; Bilder, R. Disruptive Technologists in Neuropsychological Assessment: How Smart are Smart Things? Invited CE presentation. Annual Conference of the American Academy of Clinical Neuropsychology, Virtual, 9–12 June 2021. [Google Scholar]
- Cheng, Y.W.; Chen, T.F.; Chiu, M.J. From mild cognitive impairment to subjective cognitive decline: Conceptual and methodological evolution. Neuropsychiatr. Dis. Treat. 2017, 13, 491–498. [Google Scholar] [CrossRef] [Green Version]
- Jessen, F.; Amariglio, R.E.; Van Boxtel, M.; Breteler, M.; Ceccaldi, M.; Chételat, G.; Dubois, B.; Dufouil, C.; Ellis, K.A.; Van Der Flier, W.M.; et al. A conceptual framework for research on subjective cognitive decline in preclinical Alzheimer’s disease. Alzheimer Dement. 2014, 10, 844–852. [Google Scholar] [CrossRef] [Green Version]
- Subjective Cognitive Decline—A Public Health Issue. Available online: https://www.cdc.gov/aging/data/subjective-cognitive-decline-brief.html (accessed on 17 June 2021).
- Farias, S.T.; Chou, E.; Harvey, D.J.; Mungas, D.; Reed, B.; DeCarli, C.; Park, L.Q.; Beckett, L. Longitudinal trajectories of everyday function by diagnostic status. Psychol. Aging 2013, 28, 1070–1075. [Google Scholar] [CrossRef] [Green Version]
- Andersen, C.K.; Wittrup-Jensen, K.U.; Lolk, A.; Andersen, K.; Kragh-Sørensen, P. Ability to perform activities of daily living is the main factor affecting quality of life in patients with dementia. Health Qual. Life Outcomes 2004, 2. [Google Scholar] [CrossRef] [Green Version]
- Mitchell, A.J.; Beaumont, H.; Ferguson, D.; Yadegarfar, M.; Stubbs, B. Risk of dementia and mild cognitive impairment in older people with subjective memory complaints: Meta-analysis. Acta Psychiatr. Scand. 2014, 130, 439–451. [Google Scholar] [CrossRef]
- Miebach, L.; Wolfsgruber, S.; Polcher, A.; Peters, O.; Menne, F.; Luther, K.; Incesoy, E.; Priller, J.; Spruth, E.; Altenstein, S.; et al. Which features of subjective cognitive decline are related to amyloid pathology? Findings from the DELCODE study. Alzheimer Res. Ther. 2019, 11, 66. [Google Scholar] [CrossRef] [PubMed]
- Amariglio, R.E.; Mormino, E.C.; Pietras, A.C.; Marshall, G.A.; Vannini, P.; Johnson, K.A.; Sperling, R.A.; Rentz, D.M. Subjective cognitive concerns, amyloid-b, and neurodegeneration in clinically normal elderly. Neurology 2015, 85, 56–62. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.; Denny, K.G.; Harvey, D.; Farias, S.T.; Mungas, D.; DeCarli, C.; Beckett, L. Progression from normal cognition to mild cognitive impairment in a diverse clinic-based and community-based elderly cohort. Alzheimer Dement. 2017, 13, 399–405. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farias, S.T.; Park, L.Q.; Harvey, D.J.; Simon, C.; Reed, B.R.; Carmichael, O.; Mungas, D. Everyday cognition in older adults: Associations with neuropsychological performance and structural brain imaging. J. Int. Neuropsychol. Soc. 2013, 19, 430–441. [Google Scholar] [CrossRef] [Green Version]
- Greenaway, M.C.; Hanna, S.M.; Lepore, S.W.; Smith, G.E. A Behavioral Rehabilitation Intervention for Amnestic Mild Cognitive Impairment. Am. J. Alzheimer Dis. Other Dement. 2008, 23, 451–461. [Google Scholar] [CrossRef] [PubMed]
- Greenaway, M.C.; Duncan, N.L.; Smith, G.E. The memory support system for mild cognitive impairment: Randomized trial of a cognitive rehabilitation intervention. Int. J. Geriatr. Psychiatry 2013, 28, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Levine, B.; Stamenova, V. Goal Management Training. In Encyclopedia of Clinical Neuropsychology; Springer International Publishing: Berlin/Heidelberg, Germany, 2018; pp. 1590–1594. [Google Scholar]
- Levine, B.; Stuss, D.T.; Winocur, G.; Binns, M.A.; Fahy, L.; Mandic, M.; Bridges, K.; Robertson, I.H. Cognitive rehabilitation in the elderly: Effects on strategic behavior in relation to goal management. J. Int. Neuropsychol. Soc. 2007, 13, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Voss, M.W.; Heo, S.; Prakash, R.S.; Erickson, K.I.; Alves, H.; Chaddock, L.; Szabo, A.N.; Mailey, E.L.; Wójcicki, T.R.; White, S.M.; et al. The influence of aerobic fitness on cerebral white matter integrity and cognitive function in older adults: Results of a one-year exercise intervention. Hum. Brain Mapp. 2013, 34, 2972–2985. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Erickson, K.I.; Voss, M.W.; Prakash, R.S.; Basak, C.; Szabo, A.; Chaddock, L.; Kim, J.S.; Heo, S.; Alves, H.; White, S.M.; et al. Exercise training increases size of hippocampus and improves memory. Proc. Natl. Acad. Sci. USA 2011, 108, 3017–3022. [Google Scholar] [CrossRef] [Green Version]
- Northey, J.M.; Cherbuin, N.; Pumpa, K.L.; Smee, D.J.; Rattray, B. Exercise interventions for cognitive function in adults older than 50: A systematic review with meta-Analysis. Br. J. Sports Med. 2018, 52, 154–160. [Google Scholar] [CrossRef]
- Reed, B.R.; Dowling, M.; Tomaszewski Farias, S.; Sonnen, J.; Strauss, M.; Schneider, J.A.; Bennett, D.A.; Mungas, D. Cognitive activities during adulthood are more important than education in building reserve. J. Int. Neuropsychol. Soc. 2011, 17, 615–624. [Google Scholar] [CrossRef] [Green Version]
- Wilson, R.S.; Boyle, P.A.; Yu, L.; Barnes, L.L.; Schneider, J.A.; Bennett, D.A. Life-span cognitive activity, neuropathologic burden, and cognitive aging. Neurology 2013, 81, 314–321. [Google Scholar] [CrossRef] [Green Version]
- Smith, G.E.; Housen, P.; Yaffe, K.; Ruff, R.; Kennison, R.F.; Mahncke, H.W.; Zelinski, E.M. A cognitive training program based on principles of brain plasticity: Results from the improvement in memory with plasticity-based adaptive cognitive training (IMPACT) study. J. Am. Geriatr. Soc. 2009, 57, 594–603. [Google Scholar] [CrossRef] [Green Version]
- McEwen, B.S.; Sapolsky, R.M. Stress and cognitive function. Curr. Opin. Neurobiol. 1995, 5, 205–216. [Google Scholar] [CrossRef]
- Shields, G.S.; Slavich, G.M. Lifetime stress exposure and health: A review of contemporary assessment methods and biological mechanisms. Soc. Personal. Psychol. Compass 2017, 11. [Google Scholar] [CrossRef] [PubMed]
- Gard, T.; Hölzel, B.K.; Lazar, S.W. The potential effects of meditation on age-related cognitive decline: A systematic review. Ann. N. Y. Acad. Sci. 2014, 1307, 89–103. [Google Scholar] [CrossRef] [Green Version]
- Ngandu, T.; Lehtisalo, J.; Solomon, A.; Levälahti, E.; Ahtiluoto, S.; Antikainen, R.; Bäckman, L.; Hänninen, T.; Jula, A.; Laatikainen, T.; et al. A 2 year multidomain intervention of diet, exercise, cognitive training, and vascular risk monitoring versus control to prevent cognitive decline in at-risk elderly people (FINGER): A randomised controlled trial. Lancet 2015, 385, 2255–2263. [Google Scholar] [CrossRef]
- Denny, K.G.; Harvey, D.; Meyer, O.L.; Chan, M.L.; Barba, C.; Farias, S.T. A Combined Treatment Approach to Support Everyday Function and Promote Brain Health in Older Adults: A Pilot Study. Clin. Gerontol. 2020, 43, 209–220. [Google Scholar] [CrossRef]
- Mark, R.E.; Sitskoorn, M.M. Are subjective cognitive complaints relevant in preclinical Alzheimer’s disease? A review and guidelines for healthcare professionals. Rev. Clin. Gerontol. 2013, 23, 61–74. [Google Scholar] [CrossRef] [Green Version]
- Robinson, J.H.; Callister, L.C.; Berry, J.A.; Dearing, K.A. Patient-centered care and adherence: Definitions and applications to improve outcomes. J. Am. Acad. Nurse Pract. 2008, 20, 600–607. [Google Scholar] [CrossRef]
- Martins, R.K.; McNeil, D.W. Review of Motivational Interviewing in promoting health behaviors. Clin. Psychol. Rev. 2009, 29, 283–293. [Google Scholar] [CrossRef]
- Smith, T.; Gildeh, N.; Holmes, C. The Montreal cognitive assessment: Validity and utility in a memory clinic setting. Can. J. Psychiatry 2007, 52, 329–332. [Google Scholar] [CrossRef]
- Instrumental Activities of Daily Living Scale. Available online: https://www.apa.org/pi/about/publications/caregivers/practice-settings/assessment/tools/daily-activities (accessed on 19 June 2021).
- Rabin, L.A.; Smart, C.M.; Crane, P.K.; Amariglio, R.E.; Berman, L.M.; Boada, M.; Buckley, R.F.; Chételat, G.; Dubois, B.; Ellis, K.A.; et al. Subjective Cognitive Decline in Older Adults: An Overview of Self-Report Measures Used Across 19 International Research Studies. J. Alzheimer Dis. 2015, 48, S63–S86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peter, J.; Scheef, L.; Abdulkadir, A.; Boecker, H.; Heneka, M.; Wagner, M.; Koppara, A.; Klöppel, S.; Jessen, F. Gray matter atrophy pattern in elderly with subjective memory impairment. Alzheimer Dement. 2014, 10, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Sohlberg, M.M.; Mateer, C.A. Training use of compensatory memory books: A three stage behavioral approach. J. Clin. Exp. Neuropsychol. 1989, 11, 871–891. [Google Scholar] [CrossRef]
- Halloway, S.; Wilbur, J.E.; Braun, L.T.; Schoeny, M.E.; Volgman, A.S. The feasibility of a combined lifestyle physical activity and cognitive training intervention to prevent cognitive impairment in older women with cardiovascular disease. J. Phys. Act. Health 2021, 18, 70–75. [Google Scholar] [CrossRef]
- McMaster, M.; Kim, S.; Clare, L.; Torres, S.J.; D’este, C.; Anstey, K.J. Body, brain, life for cognitive decline (BBL-CD): Protocol for a multidomain dementia risk reduction randomized controlled trial for subjective cognitive decline and mild cognitive impairment. Clin. Interv. Aging 2018, 13, 2397–2406. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sauer-Zavala, S.E.; Walsh, E.C.; Eisenlohr-Moul, T.A.; Lykins, E.L.B. Comparing Mindfulness-Based Intervention Strategies: Differential Effects of Sitting Meditation, Body Scan, and Mindful Yoga. Mindfulness 2013, 4, 383–388. [Google Scholar] [CrossRef]
- Crane-Okada, R.; Kiger, H.; Sugerman, F.; Uman, G.C.; Shapiro, S.L.; Wyman-McGinty, W.; Anderson, N.L.R. Mindful movement program for older breast cancer survivors: A pilot study. Cancer Nurs. 2012, 35. [Google Scholar] [CrossRef]
- Tomaszewski Farias, S.; Schmitter-Edgecombe, M.; Weakley, A.; Harvey, D.; Denny, K.G.; Barba, C.; Gravano, J.T.; Giovannetti, T.; Willis, S. Compensation Strategies in Older Adults: Association With Cognition and Everyday Function. Am. J. Alzheimers. Dis. Other Demen. 2018, 33, 184–191. [Google Scholar] [CrossRef]
- Carlson, M.C.; Parisi, J.M.; Xia, J.; Xue, Q.L.; Rebok, G.W.; Bandeen-Roche, K.; Fried, L.P. Lifestyle activities and memory: Variety may be the spice of life. the women’s health and aging study II. J. Int. Neuropsychol. Soc. 2012, 18, 286–294. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stewart, A.L.; Mills, K.M.; King, A.C.; Haskell, W.L.; Gillis, D.; Ritter, P.L. CHAMPS physical activity questionnaire for older adults: Outcomes for interventions. Med. Sci. Sports Exerc. 2001, 33, 1126–1141. [Google Scholar] [CrossRef]
- Resnicow, K.; McCarty, F.; Blissett, D.; Wang, T.; Heitzler, C.; Lee, R.E. Validity of a modified CHAMPS physical activity questionnaire among African-Americans. Med. Sci. Sports Exerc. 2003, 35, 1537–1545. [Google Scholar] [CrossRef] [Green Version]
- Brown, K.W.; Ryan, R.M. The Benefits of Being Present: Mindfulness and Its Role in Psychological Well-Being. J. Pers. Soc. Psychol. 2003, 84, 822–848. [Google Scholar] [CrossRef] [Green Version]
- Farias, S.T.; Mungas, D.; Reed, B.R.; Cahn-Weiner, D.; Jagust, W.; Baynes, K.; DeCarli, C. The Measurement of Everyday Cognition (ECog): Scale Development and Psychometric Properties. Neuropsychology 2008, 22, 531–544. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Logsdon, R.G.; Gibbons, L.E.; McCurry, S.M.; Teri, L. Assessing quality of life in older adults with cognitive impairment. Psychosom. Med. 2002, 64, 510–519. [Google Scholar] [CrossRef] [PubMed]
- Ryff, C.D. Happiness is everything, or is it? Explorations on the meaning of psychological well-being. J. Pers. Soc. Psychol. 1989, 57, 1069–1081. [Google Scholar] [CrossRef]
- Lewinsohn, P.M.; Seeley, J.R.; Roberts, R.E.; Allen, N.B. Center for Epidemiologic Studies Depression Scale (CES-D) as a screening instrument for depression among community-residing older adults. Psychol. Aging 1997, 12, 277–287. [Google Scholar] [CrossRef] [PubMed]
- Wisniewski, S.R.; Belle, S.H.; Marcus, S.M.; Burgio, L.D.; Coon, D.W.; Ory, M.G.; Burns, R.; Schulz, R. The Resources for Enhancing Alzheimer’s Caregiver Health (REACH): Project design and baseline characteristics. Psychol. Aging 2003, 18, 375–384. [Google Scholar] [CrossRef] [Green Version]
- Benedict, R.H.B.; Schretlen, D.; Groninger, L.; Brandt, J. Hopkins verbal learning test - Revised: Normative data and analysis of inter-form and test-retest reliability. Clin. Neuropsychol. 1998, 12, 43–55. [Google Scholar] [CrossRef]
- Ruff, R.M.; Light, R.H.; Parker, S.B.; Levin, H.S. Benton controlled Oral Word Association Test: Reliability and updated norms. Arch. Clin. Neuropsychol. 1996, 11, 329–338. [Google Scholar] [CrossRef]
- Sheridan, L.K.; Fitzgerald, H.E.; Adams, K.M.; Nigg, J.T.; Martel, M.M.; Puttler, L.I.; Wong, M.M.; Zucker, R.A. Normative Symbol Digit Modalities Test performance in a community-based sample. Arch. Clin. Neuropsychol. 2006, 21, 23–28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khayoun, R.; Devick, K.L.; Chandler, M.J.; Shandera-Ochsner, A.L.; De Wit, L.; Cuc, A.; Smith, G.E.; Locke, D.E.C. The impact of patient and partner personality traits on learning success for a cognitive rehabilitation intervention for patients with MCI. Neuropsychol. Rehabil. 2021, 1–13. [Google Scholar] [CrossRef]
- De Wit, L.; Chandler, M.; Amofa, P.; DeFeis, B.; Mejia, A.; O’Shea, D.; Locke, D.E.C.; Fields, J.A.; Smith, G.E. Memory Support System training in mild cognitive impairment: Predictors of learning and adherence. Neuropsychol. Rehabil. 2021, 31, 92–104. [Google Scholar] [CrossRef]
- McMaster, M.; Kim, S.; Clare, L.; Torres, S.J.; Cherbuin, N.; D’Este, C.; Anstey, K.J. Lifestyle Risk Factors and Cognitive Outcomes from the Multidomain Dementia Risk Reduction Randomized Controlled Trial, Body Brain Life for Cognitive Decline (BBL-CD). J. Am. Geriatr. Soc. 2020, 68, 2629–2637. [Google Scholar] [CrossRef]
- Anstey, K.J.; Cherbuin, N.; Kim, S.; McMaster, M.; D’Este, C.; Lautenschlager, N.; Rebok, G.; McRae, I.; Torres, S.J.; Cox, K.L.; et al. An internet-based intervention augmented with a diet and physical activity consultation to decrease the risk of dementia in at-risk adults in a primary care setting: Pragmatic randomized controlled trial. J. Med. Internet Res. 2020, 22, e19431. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, H.E.M.; Bay, C.P.; McFeeley, B.M.; Krivanek, T.J.; Daffner, K.R.; Gale, S.A. The Brain Health Champion study: Health coaching changes behaviors in patients with cognitive impairment. Alzheimer Dement. Transl. Res. Clin. Interv. 2019, 5, 771–779. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.K.; Lee, W.J.; Hwang, A.C.; Lin, C.S.; Chou, M.Y.; Peng, L.N.; Lin, M.H.; Chen, L.K. Efficacy of Multidomain Intervention Against Physio-cognitive Decline Syndrome: A Cluster-randomized Trial. Arch. Gerontol. Geriatr. 2021, 95, 104392. [Google Scholar] [CrossRef] [PubMed]
- Lehtisalo, J.; Levälahti, E.; Lindström, J.; Hänninen, T.; Paajanen, T.; Peltonen, M.; Antikainen, R.; Laatikainen, T.; Strandberg, T.; Soininen, H.; et al. Dietary changes and cognition over 2 years within a multidomain intervention trial—The Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability (FINGER). Alzheimer Dement. 2019, 15, 410–417. [Google Scholar] [CrossRef]
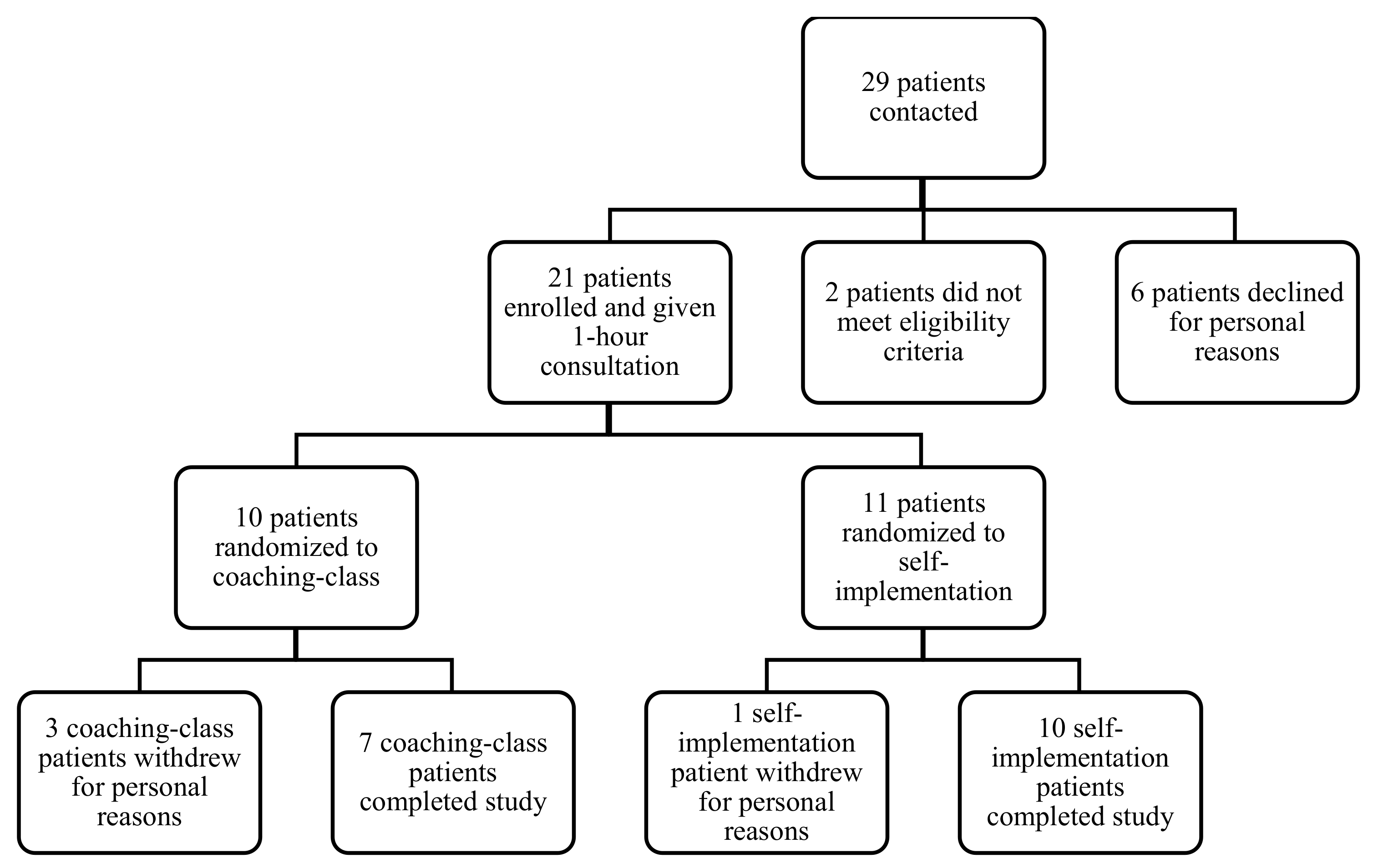
| Coaching-Class (N = 7) | Self-Implementation (N = 10) | Total (N = 17) | |
|---|---|---|---|
| Age | |||
| Median (Q1, Q3) | 63.5 (62.1, 75.0) | 68.8 (63.2, 73.3) | 67.6 (62.6, 74.2) |
| Range | 51.5–88.4 | 57.0–75.9 | 51.5–88.4 |
| Gender | |||
| Male | 2 (28.6%) | 4 (40.0%) | 6 (35.3%) |
| Female | 5 (71.4%) | 6 (60.0%) | 11 (64.7%) |
| Number of Years of Education | |||
| Median (Q1, Q3) | 16.0 (14.0, 18.0) | 16.0 (16.0, 17.0) | 16.0 (14.0, 18.0) |
| Range | 14.0–20.0 | 12.0–20.0 | 12.0–20.0 |
| Current Employment Status | |||
| Employed full-time | 4 (57.1%) | 2 (20.0%) | 6 (35.3%) |
| Employed part-time | 0 (0.0%) | 2 (20.0%) | 2 (11.8%) |
| Unemployed due to disability | 0 (0.0%) | 1 (10.0%) | 1 (5.9%) |
| Retired | 3 (42.9%) | 5 (50.0%) | 8 (47.1%) |
| Race/Ethnicity | |||
| Hispanic or Latino | 0 (0.0%) | 1 (10.0%) | 1 (5.9%) |
| White/Caucasian | 7 (100.0%) | 9 (90.0%) | 16 (94.1%) |
| Marital status | |||
| Married | 5 (71.4%) | 8 (80.0%) | 13 (76.5%) |
| Living with someone in a committed relationship | 0 (0.0%) | 1 (10.0%) | 1 (5.9%) |
| Divorced | 2 (28.6%) | 0 (0.0%) | 2 (11.8%) |
| Never married | 0 (0.0%) | 1 (10.0%) | 1 (5.9%) |
| Baseline | Post-Intervention | 3-Month Follow-Up | 6-Month Follow-Up | |
|---|---|---|---|---|
| Physical Activity (Total Minutes) | ||||
| Mean (SD) | 193.44 (171.36) | 263.50 (234.83) | 291.71 (378.76) | 132.12 (187.28) |
| Difference from Baseline (95% CI) | - | 73.73 (−2.03, 149.50) | 112.75 (−38.00, 263.50) | −37.73 (−137.08, 61.61) |
| p-value | - | 0.0556 | 0.1318 | 0.4289 |
| Effect Size | - | 0.54 | 0.4 | 0.21 |
| MAAS Score | ||||
| Mean (SD) | 4.52 (0.82) | 4.61 (0.54) | 4.55 (0.71) | 4.38 (0.72) |
| Difference from Baseline (95% CI) | - | 0.09 (−0.19, 0.37) | 0.04 (−0.20, 0.27) | −0.11 (−0.43, 0.21) |
| p-value | - | 0.5084 | 0.7501 | 0.4667 |
| Effect Size | - | 0.16 | 0.08 | 0.19 |
| Self-report EComp | ||||
| Mean (SD) | 2.05 (0.55) | 2.31 (0.37) | 2.21 (0.49) | 2.24 (0.45) |
| Difference from Baseline (95% CI) | - | 0.27 (0.07, 0.46) | 0.16 (0.00, 0.32) | 0.20 (0.00, 0.40) |
| p-value | - | 0.0096 | 0.051 | 0.0521 |
| Effect Size | - | 0.71 | 0.51 | 0.51 |
| LAQ Score | ||||
| Mean (SD) | 39.88 (8.42) | 39.88 (9.77) | 40.00 (6.87) | 40.19 (7.56) |
| Difference from Baseline (95% CI) | - | 0.00 (−3.82, 3.82) | 0.12 (−2.67, 2.91) | 1.25 (−2.18, 4.68) |
| p-value | - | 1 | 0.9299 | 0.4499 |
| Effect Size | - | 0 | 0.02 | 0.19 |
| Informant EComp (N = 11) | ||||
| Mean (SD) | 1.90 (0.60) | 2.43 (0.44) | 2.39 (0.67) | 2.39 (0.64) |
| Difference from Baseline (95% CI) | - | 0.47 (0.08, 0.86) | 0.43 (−0.05, 0.91) | 0.40 (0.02, 0.79) |
| p-value | - | 0.0227 | 0.0709 | 0.0393 |
| Effect Size | - | 0.87 | 0.65 | 0.76 |
| Baseline | Post-Intervention | 3-Month Follow-Up | 6-Month Follow-Up | |
|---|---|---|---|---|
| CES-D Score | ||||
| Mean (SD) | 8.82 (8.59) | 8.76 (6.35) | 6.38 (5.41) | 7.12 (4.47) |
| Mean Difference from Baseline (95% CI) | - | −0.06 (−4.03, 3.91) | −2.81 (−6.95, 1.33) | −2.06 (−6.17, 2.05) |
| p-value | - | 0.9753 | 0.168 | 0.3019 |
| Effect Size | - | 0.01 | 0.36 | 0.27 |
| REACH | ||||
| Mean (SD) | 15.59 (5.79) | 14.35 (4.14) | 14.75 (4.06) | 14.06 (5.52) |
| Mean Difference from Baseline (95% CI) | - | −1.24 (−4.12, 1.65) | −1.00 (−3.45, 1.45) | −1.75 (−3.25, −0.25) |
| p-value | - | 0.3781 | 0.3972 | 0.0252 |
| Effect Size | - | 0.22 | 0.22 | 0.62 |
| QOL-AD Score | ||||
| Mean (SD) | 39.12 (6.53) | 39.71 (5.83) | 40.50 (6.83) | 39.94 (5.46) |
| Mean Difference from Baseline (95% CI) | - | 0.59 (−1.44, 2.61) | 0.94 (−1.33, 3.20) | 1.56 (−0.52, 3.65) |
| p-value | - | 0.5466 | 0.3915 | 0.1313 |
| Effect Size | - | 0.15 | 0.22 | 0.4 |
| Ryff Total | ||||
| Mean (SD) | 253.12 (23.16) | 256.29 (19.82) | 256.44 (24.81) | 255.81 (22.51) |
| Median (Q1, Q3) | 256.00 (230.00, 267.00) | 260.00 (250.00, 268.00) | 262.50 (237.75, 272.75) | 255.50 (242.00, 271.75) |
| Mean Difference from Baseline (95% CI) | - | 3.18 (−4.47, 10.82) | 4.00 (−4.27, 12.27) | 4.81 (−4.58, 14.20) |
| p-value | - | 0.3914 | 0.3191 | 0.2919 |
| Effect Size | - | 0.21 | 0.26 | 0.27 |
| Informant ECog (N = 11) | ||||
| Mean (SD) | 53.10 (11.73) | 53.09 (7.82) | 51.82 (11.57) | 50.91 (8.51) |
| Mean Difference from Baseline (95% CI) | - | −0.60 (−5.00, 3.80) | −1.50 (−5.32, 2.32) | −2.90 (−5.46, −0.34) |
| p-value | - | 0.7647 | 0.3974 | 0.0304 |
| Effect Size | - | 0.1 | 0.28 | 0.81 |
| All Active Subjects Outcomes | ||||
|---|---|---|---|---|
| Baseline | Post-Intervention | 3-Month Follow-Up | 6-Month Follow-Up | |
| HVLT Total Recall | ||||
| N | 17 | 7 | 14 | 13 |
| Mean (SD) | 47.47 (9.15) | 48.14 (7.56) | 49.50 (9.77) | 51.31 (9.30) |
| Median (Q1, Q3) | 49.00 (40.00, 52.00) | 50.00 (47.00, 52.50) | 47.00 (43.25, 53.25) | 51.00 (42.00, 60.00) |
| Mean Difference from Baseline (95% CI) | - | −0.43 (−9.55, 8.69) | 3.71 (−0.63, 8.06) | 5.00 (−0.10, 10.10) |
| p-value | - | 0.9122 | 0.0878 | 0.0539 |
| Effect Size | - | 0.04 | 0.49 | 0.59 |
| COWAT Total Items | ||||
| N | 17 | 7 | 14 | 13 |
| Mean (SD) | 11.94 (2.56) | 12.14 (2.41) | 11.36 (2.59) | 12.31 (2.98) |
| Median (Q1, Q3) | 12.00 (11.00, 14.00) | 12.00 (10.50, 12.50) | 11.00 (10.00, 13.00) | 13.00 (12.00, 13.00) |
| Mean Difference from Baseline (95% CI) | - | −0.57 (−1.86, 0.72) | −0.29 (−1.26, 0.69) | 0.38 (−0.49, 1.26) |
| p-value | - | 0.3208 | 0.5365 | 0.3563 |
| Effect Size | - | 0.41 | 0.17 | 0.27 |
| Symbol Digit Total Items | ||||
| N | 17 | 7 | 14 | 13 |
| Mean (SD) | 49.76 (8.70) | 49.43 (9.78) | 54.07 (9.38) | 54.54 (10.45) |
| Median (Q1, Q3) | 52.00 (49.00, 53.00) | 52.00 (44.50, 53.00) | 52.50 (49.25, 59.00) | 56.00 (49.00, 58.00) |
| Mean Difference from Baseline (95% CI) | - | 2.14 (−0.85, 5.14) | 5.43 (1.12, 9.74) | 5.92 (0.96, 10.88) |
| p-value | - | 0.1304 | 0.0175 | 0.0232 |
| Effect Size | - | 0.66 | 0.73 | 0.72 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liou, H.; Stonnington, C.M.; Shah, A.A.; Buckner-Petty, S.A.; Locke, D.E.C. Compensatory and Lifestyle-Based Brain Health Program for Subjective Cognitive Decline: Self-Implementation versus Coaching. Brain Sci. 2021, 11, 1306. https://doi.org/10.3390/brainsci11101306
Liou H, Stonnington CM, Shah AA, Buckner-Petty SA, Locke DEC. Compensatory and Lifestyle-Based Brain Health Program for Subjective Cognitive Decline: Self-Implementation versus Coaching. Brain Sciences. 2021; 11(10):1306. https://doi.org/10.3390/brainsci11101306
Chicago/Turabian StyleLiou, Harris, Cynthia M. Stonnington, Amit A. Shah, Skye A. Buckner-Petty, and Dona E. C. Locke. 2021. "Compensatory and Lifestyle-Based Brain Health Program for Subjective Cognitive Decline: Self-Implementation versus Coaching" Brain Sciences 11, no. 10: 1306. https://doi.org/10.3390/brainsci11101306
APA StyleLiou, H., Stonnington, C. M., Shah, A. A., Buckner-Petty, S. A., & Locke, D. E. C. (2021). Compensatory and Lifestyle-Based Brain Health Program for Subjective Cognitive Decline: Self-Implementation versus Coaching. Brain Sciences, 11(10), 1306. https://doi.org/10.3390/brainsci11101306







