Postpartum Follow-Up of Women Who Developed Subclinical Hypothyroidism during Pregnancy
Abstract
1. Introduction
2. Materials and Methods
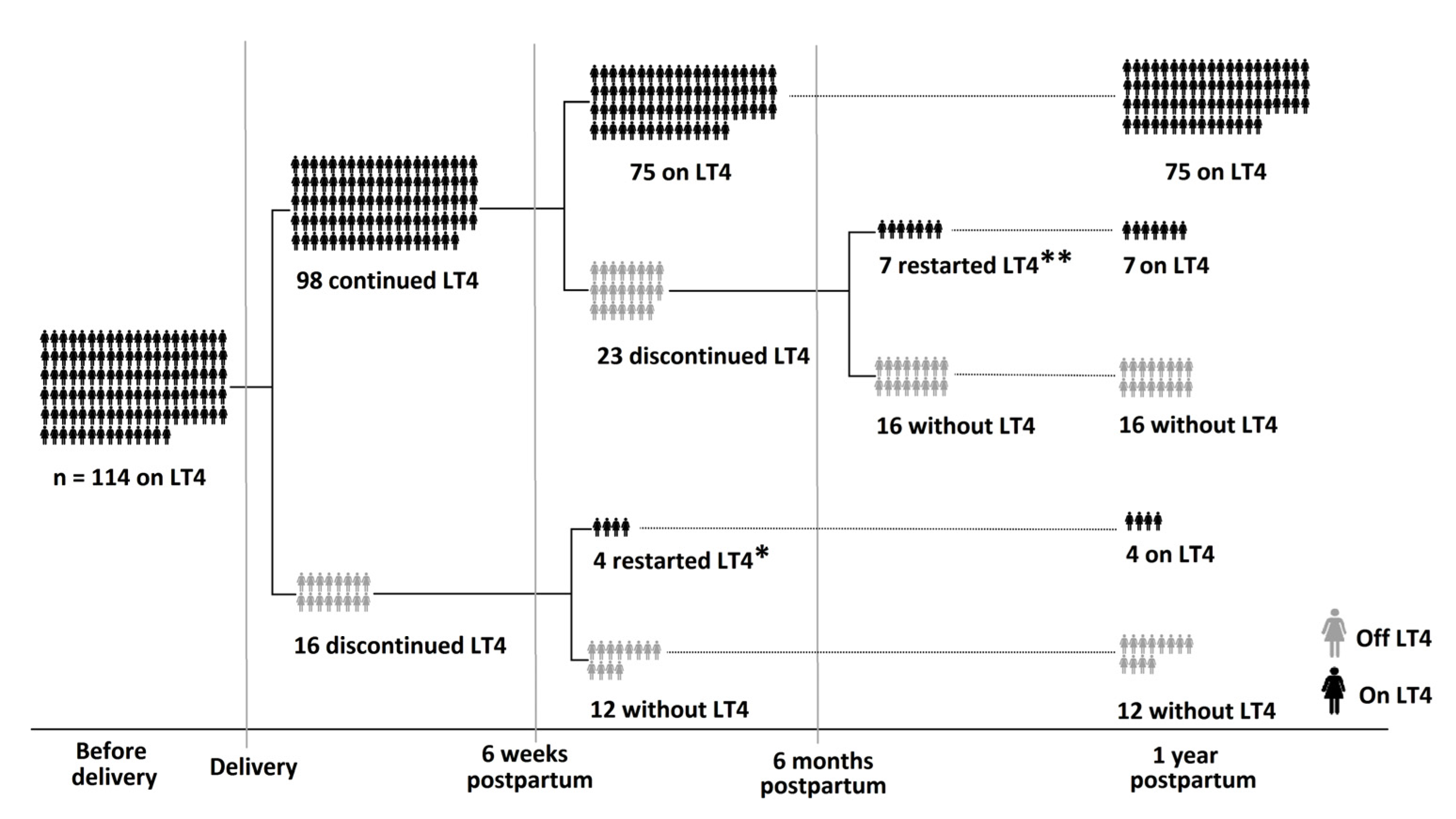
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Stagnaro-Green, A.; Pearce, E. Thyroid disorders in pregnancy. Nat. Rev. Endocrinol. 2012, 8, 650–658. [Google Scholar] [CrossRef] [PubMed]
- Glinoer, D. The regulation of thyroid function in pregnancy: Pathways of endocrine adaptation from physiology to pathology. Endocr. Rev. 1997, 18, 404–433. [Google Scholar] [CrossRef] [PubMed]
- Stagnaro-Green, A. Postpartum Management of Women Begun on Levothyroxine during Pregnancy. Front. Endocrinol. Lausanne 2015, 6, 183. [Google Scholar] [CrossRef]
- Glinoer, D. Pregnancy and iodine. Thyroid 2001, 11, 471–481. [Google Scholar] [CrossRef] [PubMed]
- Alexander, E.K.; Pearce, E.N.; Brent, G.A.; Brown, R.S.; Chen, H.; Dosiou, C.; Grobman, A.W.; Laurberg, P.; Lazarus, H.J.; Mandel, J.S.; et al. Guidelines of the American Thyroid Association for the Diagnosis and Management of Thyroid Disease During Pregnancy and the Postpartum. Thyroid 2017, 27, 315–389. [Google Scholar] [CrossRef] [PubMed]
- Lazarus, H.J. Thyroid Regulation and Dysfunction in the Pregnant Patient. Endotext. Available online: https://www.ncbinlmnihgov/books/NBK279154/ (accessed on 8 July 2019).
- Yarrington, C.; Pearce, E. Iodine and pregnancy. J. Thyroid Res. 2011, 2011, 934104. [Google Scholar] [CrossRef]
- Hammond, K.R.; Cataldo, N.A.; Hubbard, J.A.; Malizia, B.A.; Steinkampf, M.P. Gestational hypothyroidism: Development of mild hypothyroidism in early pregnancy in previously euthyroid women. Fertil. Steril. 2015, 103, 1532–1536. [Google Scholar] [CrossRef]
- Teng, W.; Shan, Z.; Patil-Sisodia, K.; Cooper, D.S. Hypothyroidism in pregnancy. Lancet Diabetes Endocrinol. 2013, 1, 228–237. [Google Scholar] [CrossRef]
- Haddow, J.E.; Palomaki, G.E.; Allan, W.C.; Williams, J.R.; Knight, G.J.; Gagnon, J.; O’Heir, C.E.; Mitchell, L.M.; Hermos, J.R.; Waisbren, E.S.; et al. Maternal thyroid deficiency during pregnancy and subsequent neuropsychological development of the child. N. Engl. J. Med. 1999, 341, 549–555. [Google Scholar] [CrossRef]
- Krassas, G.E.; Poppe, K.; Glinoer, D. Thyroid function and human reproductive health. Endocr. Rev. 2010, 31, 702–755. [Google Scholar] [CrossRef]
- Liu, H.; Shan, Z.; Li, C.; Mao, J.; Xie, X.; Wang, W.; Chenling, F.; Wang, H.; Zhang, H.; Han, C.; et al. Maternal subclinical hypothyroidism, thyroid autoimmunity, and the risk of miscarriage: A prospective cohort study. Thyroid 2014, 24, 1642–1649. [Google Scholar] [CrossRef] [PubMed]
- Tudela, C.M.; Casey, B.M.; McIntire, D.D.; Cunningham, F.G. Relationship of subclinical thyroid disease to the incidence of gestational diabetes. Obstet. Gynecol. 2012, 119, 983–988. [Google Scholar] [CrossRef] [PubMed]
- Van den Boogaard, E.; Vissenberg, R.; Land, J.A.; van Wely, M.; Ven der Post, J.A.; Goddijn, M.; Bisschop, H.P. Significance of (sub)clinical thyroid dysfunction and thyroid autoimmunity before conception and in early pregnancy: A systematic review. Hum. Reprod. Update 2016, 22, 532–533. [Google Scholar] [CrossRef] [PubMed]
- De Groot, L.; Abalovich, M.; Alexander, E.K.; Amino, N.; Barbour, L.; Cobin, R.H.; Eastman, J.C.; Lazarus, H.J.; Luton, D.; Mandel, J.S.; et al. Management of thyroid dysfunction during pregnancy and postpartum: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2012, 97, 2543–2565. [Google Scholar] [CrossRef]
- Stagnaro-Green, A.; Abalovich, M.; Alexander, E.; Azizi, F.; Mestman, J.; Negro, R.; Nixon, A.; Pearce, N.E.; Soldin, P.O.; Sullivan, S.; et al. Guidelines of the American Thyroid Association for the diagnosis and management of thyroid disease during pregnancy and postpartum. Thyroid 2011, 21, 1081–1125. [Google Scholar] [CrossRef] [PubMed]
- Lazarus, J.; Brown, R.S.; Daumerie, C.; Hubalewska-Dydejczyk, A.; Negro, R.; Vaidya, B. European thyroid association guidelines for the management of subclinical hypothyroidism in pregnancy and in children. Eur. Thyroid J. 2014, 3, 76–94. [Google Scholar] [CrossRef]
- Dror, D.K.; Allen, L.H. Iodine in Human Milk: A Systematic Review. Adv. Nutr. 2018, 9, 347S–357S. [Google Scholar] [CrossRef]
- Azizi, F.; Smyth, P. Breastfeeding and maternal and infant iodine nutrition. Clin. Endocrinol. Oxf. 2009, 70, 803–809. [Google Scholar] [CrossRef]
- Delange, F. Iodine requirements during pregnancy, lactation and the neonatal period and indicators of optimal iodine nutrition. Public Health Nutr. 2007, 10, 1571–1580. [Google Scholar] [CrossRef]
- Neelaveni, K.; Kumar, K.; Sahay, R.; Ramesh, J. Postpartum Follow-up in Women Diagnosed with Subclinical Hypothyroidism during Pregnancy. Indian J. Endocrinol. Metab. 2017, 21, 699–702. [Google Scholar] [CrossRef]
- Shields, B.M.; Knight, B.A.; Hill, A.V.; Hattersley, A.T.; Vaidya, B. Five-year follow-up for women with subclinical hypothyroidism in pregnancy. J. Clin. Endocrinol. Metab. 2013, 98, E1941–E1945. [Google Scholar] [CrossRef] [PubMed]
- Boufas, D.; Vryonidou, A.; Mastorakos, G.; Ilias, I. Thyroid Function and Autoimmunity Versus Number of Pregnancies. J. Reprod. Infertil. 2016, 17, 240–242. [Google Scholar] [PubMed]
| N | 114 |
| Mean Age ± SD (years) | 31.0 ± 6.1 |
| Mean gestational age at diagnosis ± SD (weeks) | 18.7 ± 8.6 |
| Mean body weight gain during pregnancy ± SD (kg) | 13.3 ± 5.5 |
| Mean neonatal weight at delivery ± SD (gr) | 3197.4 ± 535.8 |
| Primipara | 60/114 (52.6%) |
| Caesarean section | 62/114 (54.3%) |
| Gestational Diabetes | 32/114 (28.0%) |
| Positive TPOAbs during pregnancy | 17/114 (14.9%) |
| Positive thyroid ultrasound findings * | 21/114 (18.4%) |
| Mean TSH at diagnosis ± SD (mIU/L) | 5.24 ± 2.55 |
| Mean LT4 dose ± SD (μg/day) | 77.9 ± 20.4 |
| Mean TSH before delivery ± SD (mIU/L) | 1.65 ± 1.19 |
| Mean LT4 dose before delivery ± SD (μg/day) | 85.1 ± 23.2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Linardi, A.; Kakoulidis, I.; Ilias, I.; Michou, A.; Pappa, A.; Venaki, E.; Koukkou, E. Postpartum Follow-Up of Women Who Developed Subclinical Hypothyroidism during Pregnancy. Med. Sci. 2020, 8, 29. https://doi.org/10.3390/medsci8030029
Linardi A, Kakoulidis I, Ilias I, Michou A, Pappa A, Venaki E, Koukkou E. Postpartum Follow-Up of Women Who Developed Subclinical Hypothyroidism during Pregnancy. Medical Sciences. 2020; 8(3):29. https://doi.org/10.3390/medsci8030029
Chicago/Turabian StyleLinardi, Anastasia, Ioannis Kakoulidis, Ioannis Ilias, Aikaterini Michou, Athina Pappa, Evangelia Venaki, and Eftychia Koukkou. 2020. "Postpartum Follow-Up of Women Who Developed Subclinical Hypothyroidism during Pregnancy" Medical Sciences 8, no. 3: 29. https://doi.org/10.3390/medsci8030029
APA StyleLinardi, A., Kakoulidis, I., Ilias, I., Michou, A., Pappa, A., Venaki, E., & Koukkou, E. (2020). Postpartum Follow-Up of Women Who Developed Subclinical Hypothyroidism during Pregnancy. Medical Sciences, 8(3), 29. https://doi.org/10.3390/medsci8030029






