Archaeometric Characterisation of Decorated Pottery from the Archaeological Site of Villa dei Quintili (Rome, Italy): Preliminary Study
Abstract
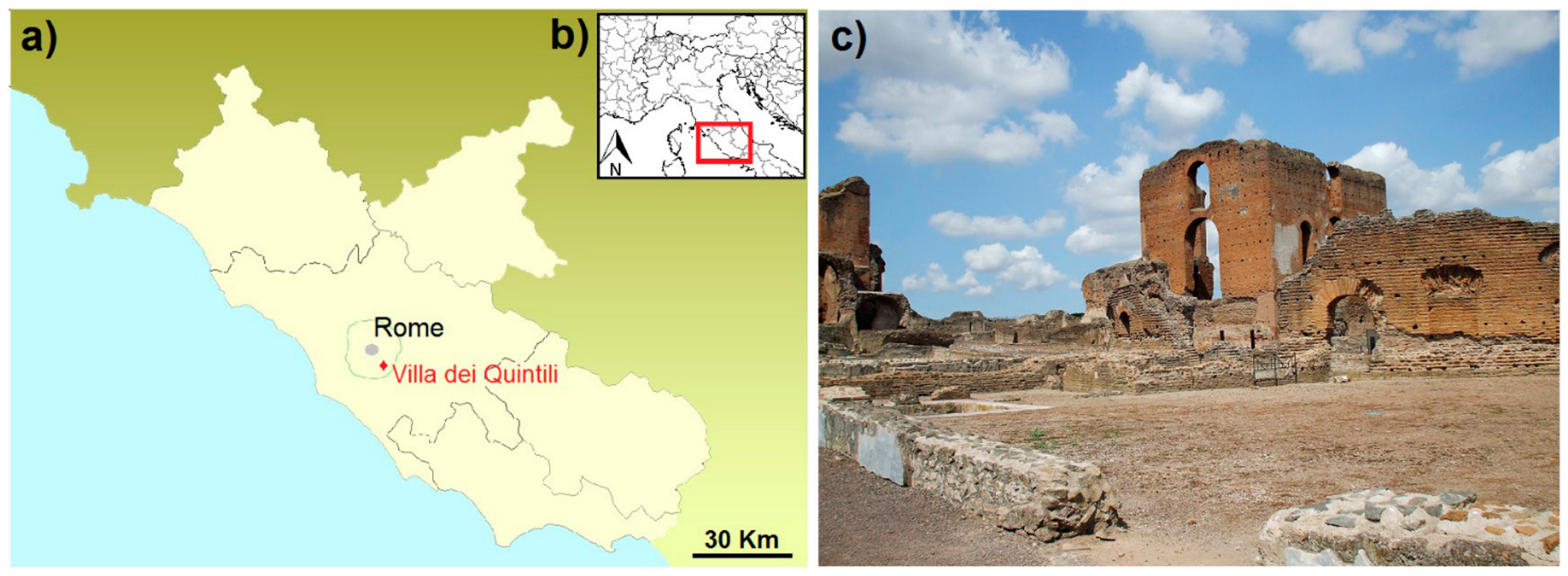
:1. Introduction and Archaeological Setting
2. Analytical Methods
3. Results and Discussion
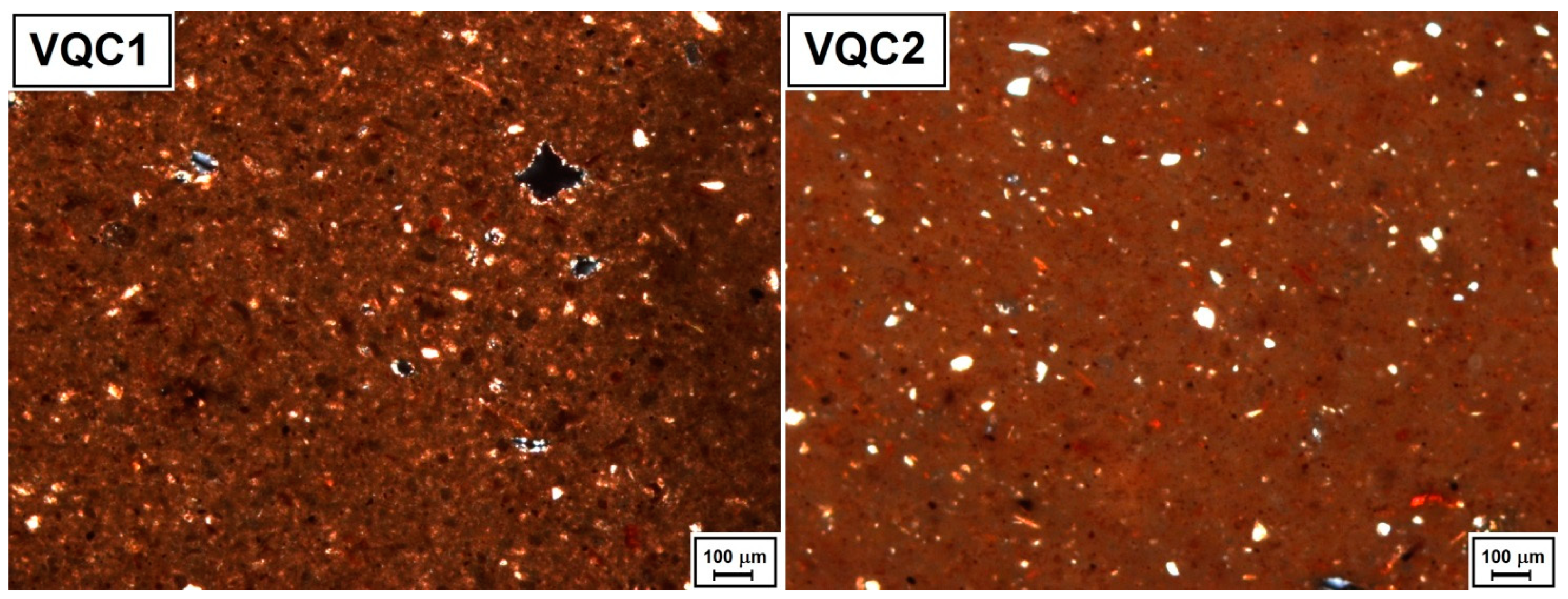
3.1. Polarising Optical Microscopy and X-ray Diffraction Analysis
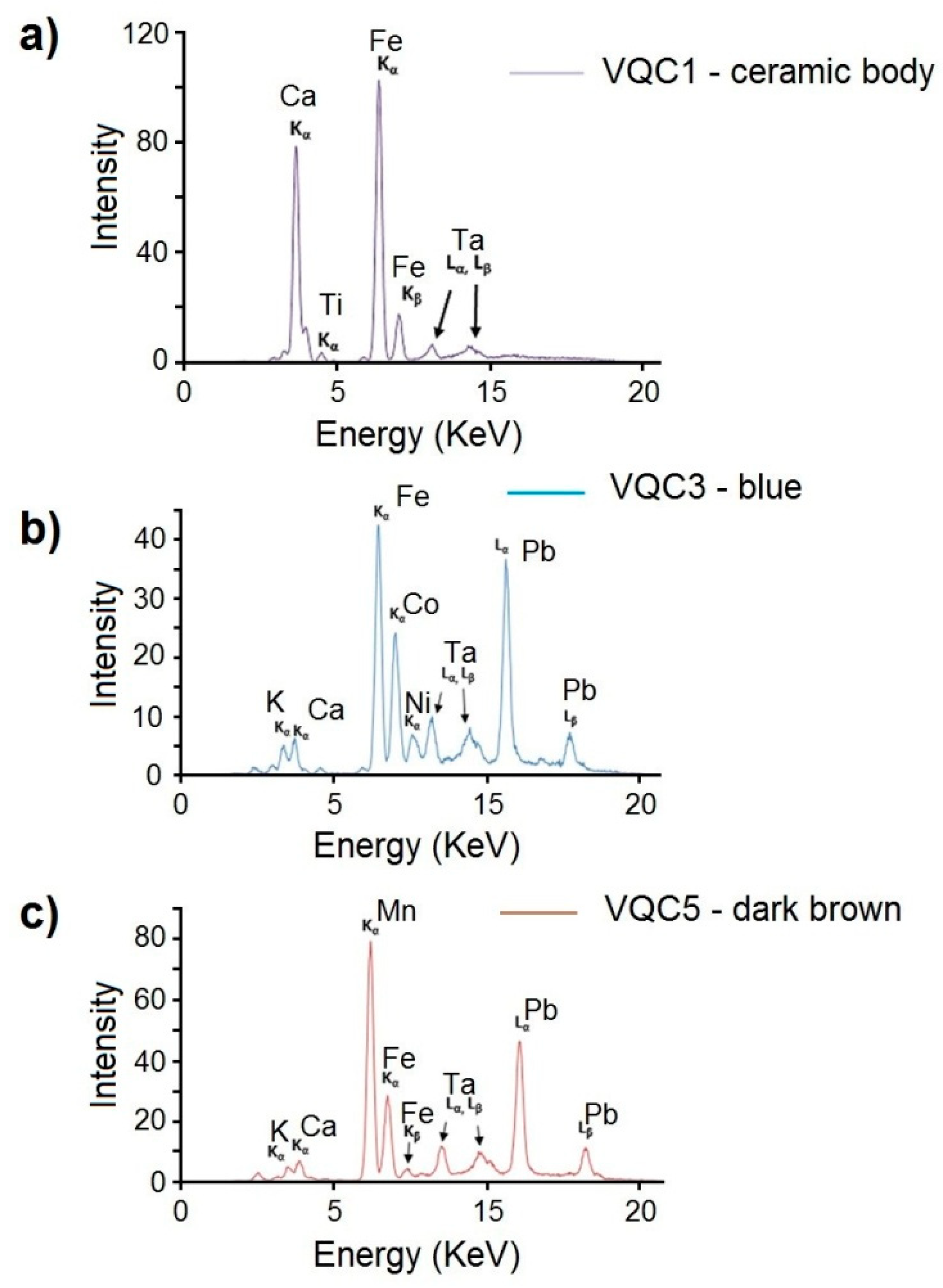
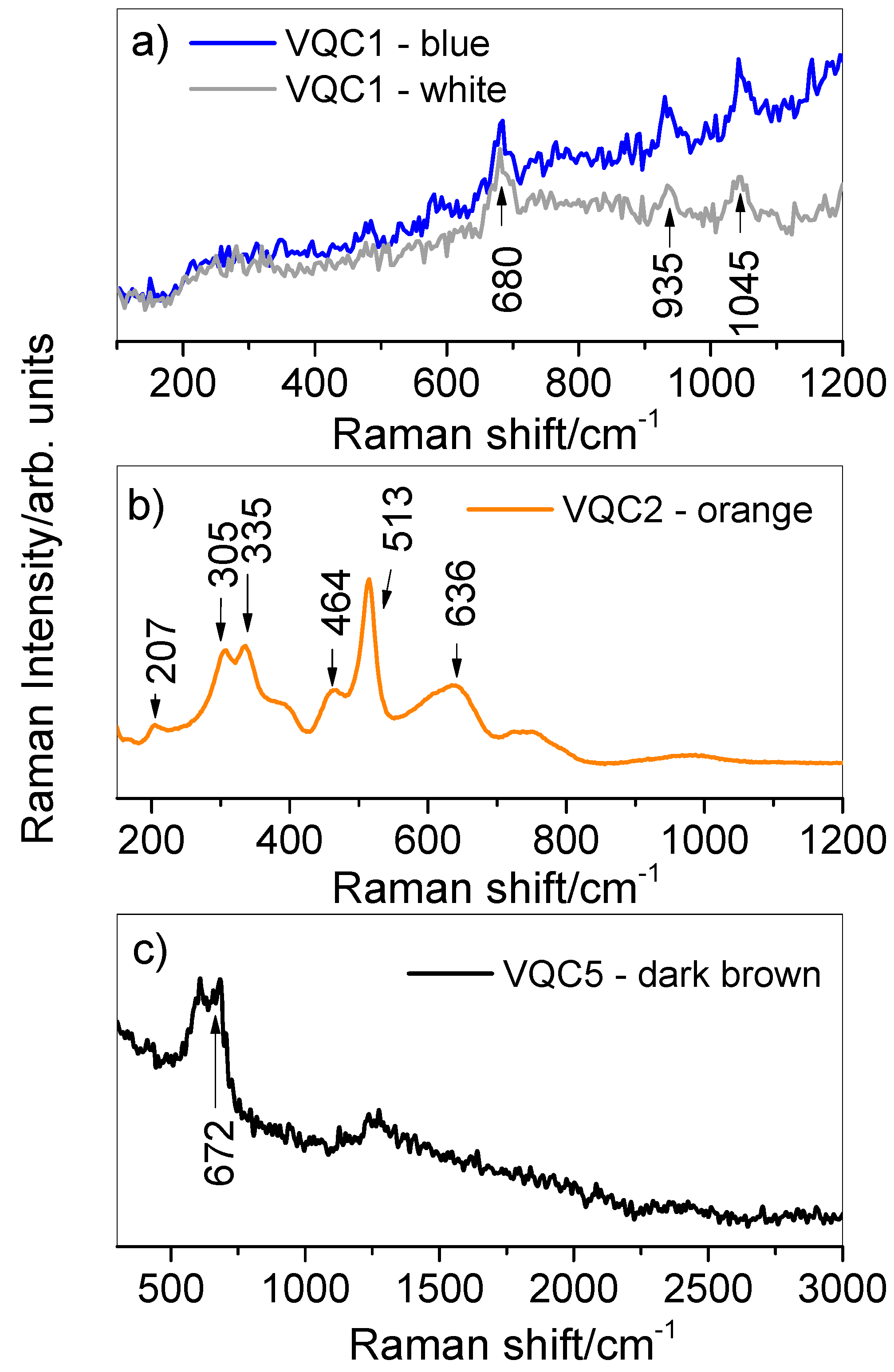
3.2. X-Ray Fluorescence (XRF) and Micro-Raman Spectroscopy
3.3. EMPA-EDS
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gajić-Kvaščev, M.; Bikić, V.; Wright, V.J.; Evans, I.R.; Damjanović-Vasilić, L. Archaeometric study of 17th/18th century painted pottery from the Belgrade Fortress. J. Cult. Herit. 2018, 32, 9–21. [Google Scholar] [CrossRef]
- Ferreira, L.F.V.; Machado, I.F.; Casimiro, T.M.; Pereira, M.F.C.; Santos, L.F. Portuguese blue-on-blue 16th–17th century pottery. Archaeometry 2018, 60, 695–712. [Google Scholar] [CrossRef]
- Ricci, A. La villa dei Quintili sulla via Appia Antica: Riuso e spoliazione. Seminari di Archeologia Cristiana 1991, 67, 467–469. [Google Scholar]
- D’Agostino, S.; Bellomo, M. Excavation, restoration and conservation of archaeological sites. The Villa dei Quintili on the Appia Antica in Rome. WIT Trans. Built Environ. 1999, 6, 451–460. [Google Scholar]
- Frontoni, R. Forma Urbis-Itinerari Nascosti di Roma Antica; ANNO: Rome, Italy, 2000. [Google Scholar]
- Paris, R. Appia Antica. In Villa dei Quintili; Tomei, M.A., Ed.; Memorie dal Sottosuolo: Verona, Italy, 2006. [Google Scholar]
- Frontoni, R.; Galli, G. Calce e calcara nella Villa dei Quintili. Arkos Scienza e Restauro 2010, 25, 66–73. [Google Scholar]
- Belfiore, C.M.; Ricca, M.; la Russa, M.F.; Ruffolo, S.A.; Galli, G.; Barca, D.; Malagodi, M.; Vallefuoco, M.; Sprovieri, M.; Pezzino, A. Provenance study of building and statuary marbles from the Roman archaeological site of “Villa dei Quintili” Rome, Italy. Ital. J. Geosci. 2016, 135, 236–249. [Google Scholar] [CrossRef]
- Bruun, C. Fistule acquarie e proprietary terrier nel suburbium. In Suburbium: Il Suburbio di Roma dalla Crisi del Sistema delle Ville a Gregorio Magno; Pergola, P., Valenziani, R.S., Volpe, R., Eds.; École Française de Rome: Rome, Italy, 2003; pp. 485–501. [Google Scholar]
- Paris, R. Via Appia. La Villa dei Quintili. Collana Soprintendenza Archeologica di Roma; Mondadori Electa: Milano, Italy, 2000. [Google Scholar]
- Frontoni, R.; Galli, G.; Paris, R.; Rotondi, A. Forma Urbis—Itinerari Nascosti di Roma Antica, Anno XVII; ANNO: Roma, Italy, 2012. [Google Scholar]
- Panagopoulou, A.; Lampakis, D.; Christophilos, D.; Beltsios, K.; Ganetsos, T. Technological examination of Iznik ceramics by SEM-EDX, Raman, XRD, PLM: A case study. Sci. Cult. 2018, 4, 27–33. [Google Scholar]
- Rahim, N.S.A. Analytical study and conservation of archaeological Terra Sigιllata ware from Roman Period, Tripoli, Libya. Sci. Cult. 2016, 2, 19–27. [Google Scholar]
- Fındık, N.Ö.; Akyol, A.A.; Sarı, N. Archaeometric analyses of Hasankeyf unglazed ceramics. Mediterr. Archaeol. Archaeom. 2014, 14, 261–271. [Google Scholar]
- Garcia-Iñañez, J.; Garrigós, J.B.I.; Fernández, M.M.I.; Esparraguera, J.M.G.I.; Cerdà, J.A.C.I. Archaeometric characterization of Middle Age and Renaissance tin lead glazed pottery from Barcelona. In Archaeometric and Archaeological Approaches to Ceramics; BAR International Series, 1691; Yona Waksman, S., Ed.; Archaeopress: Oxford, UK, 2007; p. 175. [Google Scholar]
- Bell, I.M.; Clark, R.J.H.; Gibbs, P.J. Raman spectroscopic library of natural and synthetic pigments pre-≈ 1850 AD. Spectrochim. Acta A 1997, 53, 2159–2179. [Google Scholar] [CrossRef]
- Buzgar, N.; Apopei, A.I.; Buzatu, A. Database of Raman Spectroscopy. 2009. Available online: rdrs.uaic.ro (accessed on 16 April 2019).
- Lafuente, B.; Downs, R.T.; Yang, H.; Stone, N. The power of databases: The RRUFF project. In Highlights in Mineralogical Crystallography; Armbruster, T., Danisi, R.M., Eds.; W. De Gruyter: Berlin, Germany, 2015; pp. 1–30. [Google Scholar]
- Whitbread, I.K. Greek Transport Amphorae. A Petrological and Archaeological Study; The British School at Athens: Athens, Greece, 1995. [Google Scholar]
- Aloise, P.; Ricca, M.; la Russa, M.F.; Ruffolo, S.A.; Belfiore, C.M.; Padeletti, G.; Crisci, G.M. Diagnostic analysis of stone materials from underwater excavations: The case study of the Roman archaeological site of Baia Naples, Italy. Appl. Phys. A 2014, 1143, 655–662. [Google Scholar] [CrossRef]
- Barca, D.; la Russa, M.F.; Crisci, G.M. Technological and geochemical study of two red figured vases of unknown provenance by various analytical techniques. Appl. Phys. A 2010, 1003, 911–917. [Google Scholar] [CrossRef]
- Montero, M.A.; Jordán, M.M.; Almendro-Candel, M.B.; Sanfeliu, T.; Hernández-Crespo, M.S. The use of a calcium carbonate residue from the stone industry in manufacturing of ceramic tile bodies. Appl. Clay Sci. 2009, 43, 186–189. [Google Scholar] [CrossRef]
- Ricca, M.; Comite, V.; la Russa, M.F.; Barca, D. Diagnostic analysis of bricks from the underwater archaeological site of Baia Naples, Italy: Preliminary results. Rend. Online Soc. Geol. Ital. 2016, 38, 85–88. [Google Scholar] [CrossRef]
- Rovella, N.; Comite, V.; Ricca, M. The methodology of investigation on red-and black-figured pottery of unknown provenance. Int. J. Conserv. Sci. 2016, 72, 954–964. [Google Scholar]
- Maritan, L.; Nodari, L.; Mazzoli, C.; Milano, A.; Russo, U. Influence of firing condi-tions on ceramic products: Experimental study on clay rich in organic matter. Appl. Clay Sci. 2006, 31, 1–15. [Google Scholar] [CrossRef]
- Riccardi, M.P.; Messiga, B.; Duminuco, P. An approach to the dynamics of clayfiring. Appl. Clay Sci. 1999, 15, 393–409. [Google Scholar] [CrossRef]
- Kreimeyer, R. Some notes on the Firing Colour of Clay Bricks. Appl. Clay Sci. 1987, 2, 175–183. [Google Scholar] [CrossRef]
- Garrigós, J.B.I. Alteration and contamination of archaeological ceramics: The perturbation problem. J. Archaeol. Sci. 1999, 26, 29–313. [Google Scholar]
- Messina, M.; Arcifa, L.; Barone, G.; Finocchiaro, C.; Mazzoleni, P. Islamic Pottery Production in Eastern Sicily (10th–11th Centuries): Preliminary archaeometric data on local and imported products from Paternò (Sicily). Mediterr. Archaeol. Archaeom. 2018, 5, 207–223. [Google Scholar]
- Crupi, V.; Majolino, D.; Venuti, V.; Barone, G.; Mazzoleni, P.; Pezzino, A.; la Russa, M.F.; Ruffolo, S.A.; Bardelli, F. Non-destructive identification of green and yellow pigments: The case of some Sicilian Renaissance glazed pottery. Appl. Phys. A 2010, 100, 845–853. [Google Scholar] [CrossRef]
- Crupi, V.; Galli, G.; la Russa, M.F.; Longo, F.; Maisano, G.; Majolino, D.; Malagodi, M.; Pezzino, A.; Ricca, M.; Rossi, B.; et al. Multi-technique investigation of Roman decorated plasters from Villa dei Quintili Rome, Italy. Appl. Surf. Sci. 2015, 349, 924–930. [Google Scholar] [CrossRef]
- Crupi, V.; Fazio, B.; Fiocco, G.; Galli, G.; la Russa, M.F.; Licchelli, M.; Majolino, D.; Malagodi, M.; Ricca, M.; Ruffolo, S.A.; et al. Multi-analytical study of Roman frescoes from Villa dei Quintili Rome, Italy. J. Archaeol. Sci. Rep. 2018, 21, 422–432. [Google Scholar] [CrossRef]
- Scarpelli, R.; Clark, R.J.; de Francesco, A.M. Archaeometric study of black-coated pottery from Pompeii by different analytical techniques. Spectrochim. Acta A 2014, 120, 60–66. [Google Scholar] [CrossRef]
- Appolonia, L.; Vaudan, D.; Chatel, V.; Aceto, M.; Mirti, P. Combined use of FORS, XRF and Raman spectroscopy in the study of mural paintings in the Aosta Valley (Italy). Anal. Bioanal. Chem. 2009, 395, 2005–2013. [Google Scholar] [CrossRef]
- Casuccio, G.; Bunker, K.; Kennedy, S.; Sparrow, M.; Pacolay, B.; Ioannidis, P.; Foulke, R. Portable XRF and Raman analysis of a ‘Modigliani’ signature painting. Microsc. Microanal. 2012, 18, 958–959. [Google Scholar] [CrossRef]
- Tite, M.S.; Pradell, T.; Shortland, A. Discovery, production and use of tin-based opacifiers in glasses, enamels and glazes from the Late Iron Age onwards: A reassessment. Archaeometry 2008, 50, 67–84. [Google Scholar] [CrossRef]
- Tite, M.S. The production technology of Italian maiolica: A reassessment. J. Archaeol. Sci. 2009, 36, 2065–2080. [Google Scholar] [CrossRef]
- Colomban, P.; Zhang, Y.; Zhao, B. Non-invasive Raman analyses of Chinese huafalang and related porcelain wares. Searching for evidence for innovative pigment technologies. Ceram. Int. 2017, 43, 12079–12088. [Google Scholar] [CrossRef]
- Gratuze, B.; Soulier, I.; Barradon, J.N.; Foy, D. De l’origine du cobalt dans les verres. Rev. Archeom. 1992, 16, 97–108. [Google Scholar] [CrossRef]
- Gratuze, B.; Soulier, I.; Blet, M.; Vallauri, L. De l’origine du cobalt: Du verre à lacéramique. Rev. Archeom. 1996, 20, 77–94. [Google Scholar]
- Zucchiatti, A.; Bouquillon, A.; Katona, I.; D’Alessandro, A. The “Della Robbia blue”: A case study for the use of cobalt pigments in ceramics during the Italian renaissance. Archaeometry 2006, 48, 131–152. [Google Scholar] [CrossRef]
- Colomban, P.; Arberet, L.; Kirmizi, B. On-site analysis of 17th–18th centuries Limoges enamels. Arsenic and the technological relationship between enamelled Limoges and Chinese wares. Ceram. Int. 2017, 43, 10158–10165. [Google Scholar] [CrossRef]
- Dell’Aquila, C.; Laviano, R.; Vurro, F.J. Chemical and mineralogical investigations of majolicas 16th–19th centuries from Laterza, southern Italy. Geol. Soc. 2006, 257, 151–162. [Google Scholar] [CrossRef]
- Roldán, C.; Coll, J.; Ferrero, J. EDXRF analysis of blue pigments used in Valencian ceramics from the 14th century to modern times. J. Cult. Herit. 2006, 7, 134–138. [Google Scholar] [CrossRef]
- Schurr, M.R.; Donohue, P.H.; Simonetti, A.; Dawson, E.L. Multi-element and lead isotope characterization of early nineteenth century pottery sherds from Native American and Euro-American sites. J. Archaeol. Sci. Rep. 2018, 20, 39–399. [Google Scholar] [CrossRef]
- Šefců, R.; Chlumská, Š.; Hostašová, A. An investigation of the lead tin yellows type I and II and their use in Bohemian panel paintings from the Gothic period. Herit. Sci. 2015, 3, 1–15. [Google Scholar] [CrossRef]
- Sandalinas, C.; Ruiz-Moreno, S.; Lòpez-Gil, A.; Miralles, J. Experimental confirmation by Raman spectroscopy of a Pb–Sn–Sb triple oxide yellow pigment in sixteenth-century Italian pottery. J. Raman Spectrosc. 2006, 37, 1146–1153. [Google Scholar] [CrossRef]
- Bevilacqua, N.; Borgioli, L.; Gracia, I.A. I Pigmenti-nell’arte. Dalla Preistoria Alla Rivoluzione Industrilae; Matteini: Saonara, Italy, 2010. [Google Scholar]
- Shortland, A.J. The use and origin of antimonate colorants in early Egyptian glass. Archaeometry 2002, 44, 517–530. [Google Scholar] [CrossRef]
- Freestone, I.C.; Stapleton, C.P.; Rigby, V. The production of red glass and enamel in the Late Iron Age, Roman and Byzantine periods. In Through a Glass Brightly: Studies in Byzantine and Medieval Art and Archaeology; Entwistle, C., Buckton, D., Eds.; Oxbow Books: Oxford, UK, 2003; ISBN 978-1785702518. [Google Scholar]
- Lahlil, S.; Biron, I.; Galoisy, L.; Morin, G. Technological processes to produce antimonate opacified glass throughout history. Annales du 2009, 17, 571–578. [Google Scholar]
- Molina, G.; Odin, G.P.; Pradell, T.; Shortland, A.J.; Tite, M.S. Production technology and replication of lead antimonate yellow glass from New Kingdom Egypt and the Roman Empire. J. Archaeol. Sci. 2014, 41, 171–184. [Google Scholar] [CrossRef]
- Maltoni, S.; Silvestri, A. Investigating Production Technologies of Roman Glass Tesserae from Northeastern Italy. Minerals 2018, 86, 255. [Google Scholar] [CrossRef]
- Legodi, M.; de Waal, D. The preparation of magnetite, goethite, hematite and maghemite of pigment quality from mill scale iron waste. Dyes Pigments 2007, 74, 161–168. [Google Scholar] [CrossRef]
- Montagner, C.; Sanches, D.; Pedroso, J.; Melo, M.J.; Vilarigues, M. Ochres and earths: Matrix and chromophores characterization of 19th and 20th century artist materials. Spectrochim. Acta A 2013, 103, 409–416. [Google Scholar] [CrossRef]
- Hradil, D.; Grygar, T.; Hradilovà, J.; Bezdička, P. Clay and iron oxide pigments in the history of painting. Appl. Clay Sci. 2003, 22, 223–236. [Google Scholar] [CrossRef]
- Genestar, C.; Pons, C. Earth pigments in painting: Characterisation and differentiation by means of FTIR spectroscopy and SEM-EDS microanalysis. Anal. Bioanal. Chem. 2005, 3822, 269–274. [Google Scholar] [CrossRef]
- Matin, M.; Tite, M.; Watson, O. On the origins of tin-opacified ceramic glazes: New evidence from early Islamic Egypt, the Levant, Mesopotamia, Iran, and Central Asia. J. Archaeol. Sci. 2018, 97, 42–66. [Google Scholar] [CrossRef]
- Campanella, L.; Casoli, A.; Colombini, M.P.; Marini, R.B.; Matteini, M.; Migneco, L.M.; Montenero, A.; Nodari, L.; Piccioli, C.; Zappalà, M.P.; et al. Chimica per L’arte; Zanichelli: Bologna, Italy, 2007; ISBN 9788808068538. [Google Scholar]
- Eppler, R.A. Colorants for Ceramics; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2013. [Google Scholar]
- Paris, R.; Frontoni, R.; Galli, G.; Lalli, C. Dalla villa al casale: Attività produttive nella Villa dei Quintili. In Proceedings of the International Conference of Studies, Prato, Italy, 20–21 October 2014; pp. 195–210. [Google Scholar]
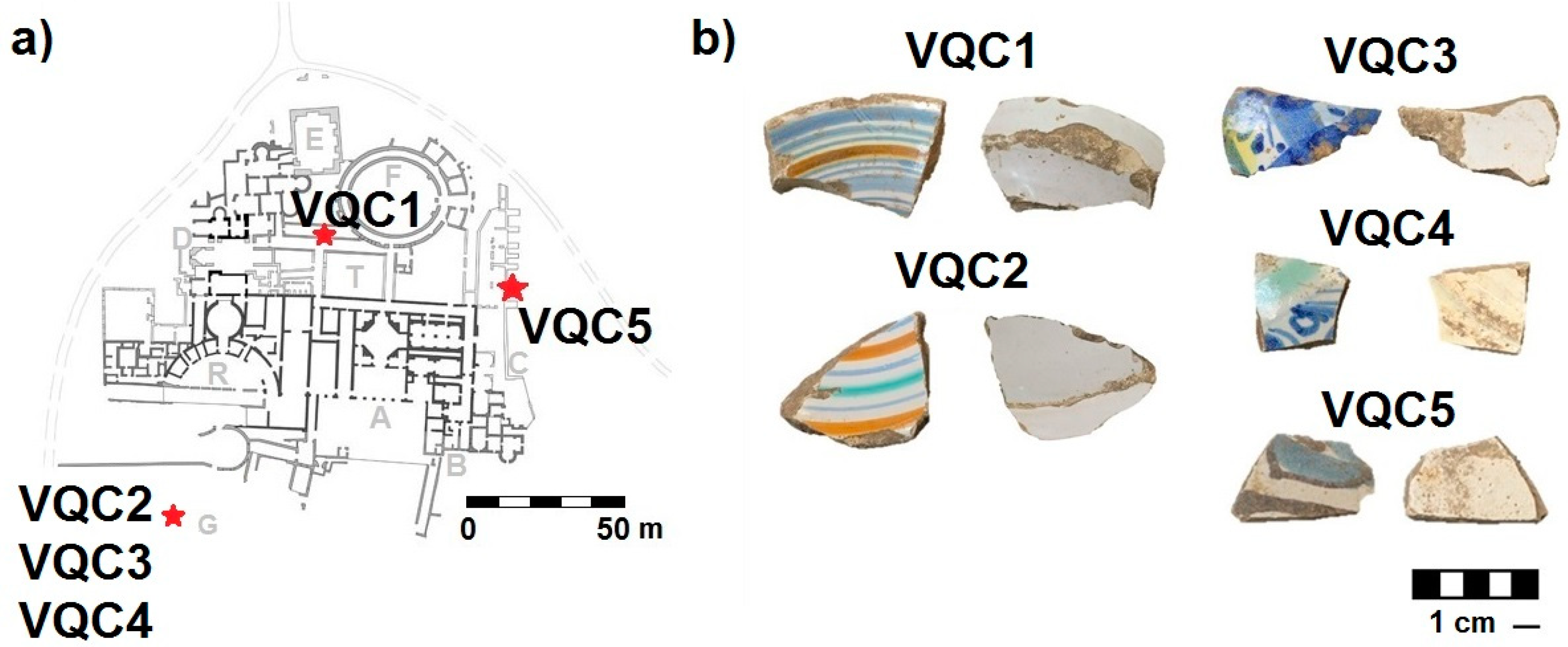

| Sample | Description | Recovering Area |
|---|---|---|
| VQC1 | Open shape (probably a plate) with polychrome bands decoration on a white background. Predominant colours: light blue, blue, orange | Corridor of the Ludus-Viridarium |
| VQC2 | Open shape (probably a plate) with polychrome bands decoration on a white background. Predominant colours: green, blue, orange | Faced with the Great cistern |
| VQC3 | Closed shape (probably a jug) with polychrome floral decoration on a white background. Predominant colours: light blue, blue, green, yellow | Faced with the Great cistern |
| VQC4 | Closed shape (probably a jug) with polychrome floral decoration on a white background. Predominant colours: blue, green | Faced with the Great cistern |
| VQC5 | Closed shape (probably a jug) with polychrome floral decoration on a white background. Predominant colours: light blue, dark-brown | Basis Villae |
| Minero-Petrographic Features by POM | ||||||||
| Sample | Groundmass | Other Inclusions | Size of Inclusions min-max (µm) | Sorting | Common Grain Shape | c/f/v Ratio (%) | ||
| VQC1 | Ho | Qtz, Fds, Fe-ox | 30–90 | WS | medium-low sphericity, sub-angular rounding | 45/40/15 | ||
| VQC2 | Ho | Qtz, Fds, Fe-ox | 30–300 | S to WS | medium-low sphericity, from sub-angular to rounded | 50/40/10 | ||
| VQC3 | Ho | Qtz, Fds, Fe-ox, Pl | 30–120 | WS | medium-high sphericity, from sub-angular to rounded | 45/40/15 | ||
| VQC4 | Ho | Qtz, Fds, Fe-ox, Pl | 30–200 | WS | medium-high sphericity, from sub-angular to rounded | 45/40/15 | ||
| VQC5 | Ho | Qtz, Fds, Fe-ox | 30–200 | S to WS | medium-low sphericity, from sub-angular to sub-rounded | 50/40/10 | ||
| Mineralogical Phases by XRD | ||||||||
| Sample | Qtz | Fds | Cal | Gh | Di | Pl | Gy | Clay Minerals Group |
| VQC1 | P | P | P | P | P | / | P | P |
| VQC2 | P | P | P | P | P | / | P | P |
| VQC3 | P | P | P | P | P | P | / | P |
| VQC4 | P | P | P | P | P | P | / | P |
| VQC5 | P | P | P | P | P | / | P | P |
| Sample | Analysed Area | Detected Chemical Elements by XRF |
|---|---|---|
| VQC1 | Light Blue | S, Pb, K, As, Ca, Cl, Sn (Fe, Ti, Co, Zn, Cd, Mn, Ba, Sr) |
| Orange | S, Pb, K, Ca, As, Cl, Sn (Fe, Co, Ti, Ni, Cd, Zn, Mn, Ba) | |
| White background | S, Pb, K, Sn, Cl, Ca (As, Fe, Ti, Cd, Mn, Ba) | |
| Ceramic body | Ca, Fe, K (Ti, Sr, Pb, Mn, Ba, Cr, Zn) | |
| VQC2 | Green | S, Pb, Ca, K, Cl, Cu, Sn (Fe, As, Ti, Sr, Mn, Ba) |
| Orange | S, Pb, K, Ca, Cl, Sn, Fe, Sb (As, Cd, Mn) | |
| White background | S, Pb, Ca, K, Fe, Cl (Sn, As, Ti, Sr, Mn, Ba, Cu, Cd, Cr) | |
| Ceramic body | Ca, Fe, K (Ti, Sr, Pb, Mn, Ba, Cr, Zn) | |
| VQC3 | Light Blue | S, Pb, K, Ca, Sn, As, Cl (Co, Fe, Ni, Cu, Ti, Zn, Mn, Cd, Ba, Sr) |
| Blue | Pb, S, K, Ca, As, Fe, Co, Sn, Cl (Ni, Ti, Cu, Zn, Mn, Sr, Cd, Ba) | |
| Yellow | Pb, S, K, Ca, Sn, Cl, Fe (As, Sb, Co, Ti, Zn, Cu, Ni, Sr, Mn, Ba) | |
| White background | S, Pb, Ca, K, Cl, Fe, As (Sn, Ti, Mn, Sr, Cu, Ba, Zn) | |
| Ceramic body | Ca, Fe, K (Ti, Sr, Pb, Mn, Ba, Cr, Zn) | |
| VQC4 | Green | S, Pb, k, Cl, Ca, Sn, As (Cu, Fe, Ti, Zn, Cd, Mn, Ba) |
| Blue | S, Pb, K, Cl, As, Sn, Ca (Co, Fe, Ni, Cu, Ti, Zn, Cd, Ba, Mn) | |
| White background | S, Pb, K, Ca, Cl, As (Fe, Sn, Ti, Cu, Sr, Mn, Zn, Ba) | |
| Ceramic body | Ca, Fe, K (Ti, Sr, Pb, Mn, Ba, Cr, Zn) | |
| VQC5 | Dark-brown | S, Pb, K, Mn, Ca, Cl, Sn, As (Fe, Cu, Ba, Cd, Sr) |
| Light Blue | S, Pb, K, Cl, Ca, Sn, As, Mn (Fe, Co, Ni, Cu, Cd, Ba) | |
| White background | S, Pb, K, Ca, Cl, Sn, As (Fe, Ti, Cd, Mn, Zn, Ba, Sr) | |
| Ceramic body | Ca, Fe, K (Ti, Sr, Pb, Mn, Ba, Cr, Zn) |
| Detected Chemical Elements by EMPA-EDS | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample | Analysed Area | CaO | Na2O | MgO | Al2O3 | SiO2 | P2O5 | K2O | FeO | As2O3 | SnO2 | PbO | CoO | Cr2O3 | CuO | MnO |
| VQC1 | Blue | 1.35 | 1.25 | 0.31 | 4.56 | 51.78 | 6.69 | 0.82 | 0.09 | 1.26 | 31.73 | 0.16 | ||||
| σ | 0.09 | 0.10 | 0.04 | 0.03 | 5.12 | 0.40 | 0.05 | 0.03 | 0.13 | 0.83 | 0.03 | |||||
| Light Blue | 1.45 | 1.14 | 0.26 | 4.15 | 52.15 | 0.35 | 5.62 | 0.45 | 0.10 | 1.20 | 33.07 | 0.06 | ||||
| σ | 0.20 | 0.15 | 0.03 | 0.05 | 4.88 | 0.05 | 0.72 | 0.03 | 0.02 | 0.15 | 1.23 | 0.03 | ||||
| Orange | 1.87 | 1.31 | 0.34 | 3.28 | 40.97 | 2.93 | 6.83 | 10.10 | 0.24 | 32.12 | ||||||
| σ | 0.25 | 0.88 | 0.05 | 0.10 | 3.35 | 0.50 | 0.88 | 1.22 | 0.02 | 1.48 | ||||||
| White background | 1.31 | 1.03 | 0.20 | 3.53 | 48.08 | 5.80 | 0.29 | 0.04 | 1.82 | 37.90 | ||||||
| σ | 0.08 | 0.12 | 0.07 | 0.17 | 2.45 | 0.30 | 0.04 | 0.01 | 0.40 | 0.97 | ||||||
| VQC2 | Blue | 1.00 | 0.99 | 0.12 | 2.39 | 47.06 | 4.20 | 0.81 | 0.15 | 0.86 | 42.21 | 0.21 | ||||
| σ | 0.05 | 0.10 | 0.04 | 0.30 | 4.48 | 0.60 | 0.05 | 0.03 | 0.12 | 1.34 | 0.05 | |||||
| Green | 1.01 | 1.08 | 0.18 | 3.26 | 44.40 | 4.19 | 0.15 | 2.02 | 40.00 | 3.70 | ||||||
| σ | 0.05 | 0.07 | 0.05 | 0.61 | 3.07 | 0.18 | 0.02 | 0.55 | 0.89 | 0.80 | ||||||
| Orange | 0.61 | 1.39 | 0.25 | 2.77 | 44.04 | 5.92 | 6.95 | 0.14 | 37.89 | 0.04 | ||||||
| σ | 0.10 | 0.56 | 0.05 | 0.30 | 3.48 | 0.67 | 0.07 | 0.02 | 1.78 | 0.01 | ||||||
| White background | 0.79 | 1.20 | 0.13 | 3.36 | 49.45 | 4.27 | 0.13 | 0.10 | 1.18 | 39.39 | ||||||
| σ | 0.06 | 0.10 | 0.06 | 0.22 | 3.15 | 0.80 | 0.03 | 0.01 | 0.40 | 1.12 | ||||||
| VQC3 | Blue | 1.43 | 1.53 | 0.24 | 3.73 | 56.24 | 4.13 | 1.40 | 30.49 | 0.81 | ||||||
| σ | 0.12 | 0.10 | 0.04 | 0.89 | 6.02 | 0.70 | 0.10 | 1.15 | 0.11 | |||||||
| Light Blue | 1.88 | 1.53 | 4.16 | 61.86 | 4.51 | 0.59 | 0.11 | 1.04 | 24.21 | 0.11 | ||||||
| σ | 0.15 | 0.21 | 0.14 | 4.94 | 0.63 | 0.03 | 0.02 | 0.15 | 0.98 | 0.02 | ||||||
| Yellow | 1.15 | 1.97 | 0.30 | 5.34 | 68.54 | 5.65 | 0.33 | 1.52 | 15.20 | |||||||
| σ | 0.01 | 0.16 | 0.07 | 0.93 | 5.58 | 0.86 | 0.05 | 0.25 | 1.68 | |||||||
| Green | 0.82 | 2.34 | 0.25 | 4.15 | 65.36 | 5.93 | 1.50 | 0.13 | 1.20 | 17.84 | 0.48 | |||||
| σ | 0.04 | 0.10 | 0.05 | 0.72 | 3.67 | 0.23 | 0.02 | 0.05 | 0.98 | 0.03 | ||||||
| White background | 3.02 | 2.07 | 0.44 | 3.97 | 64.94 | 5.80 | 0.49 | 1.69 | 17.58 | |||||||
| σ | 0.10 | 0.22 | 0.07 | 0.32 | 4.55 | 0.45 | 0.05 | 0.56 | 1.11 | |||||||
| VQC4 | Blue | 2.09 | 1.95 | 0.45 | 3.51 | 51.55 | 3.92 | 0.68 | 1.20 | 34.50 | 0.15 | |||||
| σ | 0.09 | 0.11 | 0.04 | 0.77 | 5.86 | 0.88 | 0.12 | 0.05 | 2.22 | 0.04 | ||||||
| Green | 2.53 | 1.98 | 0.55 | 3.56 | 51.26 | 3.70 | 0.64 | 0.78 | 33.92 | 1.08 | ||||||
| σ | 0.10 | 0.29 | 0.04 | 0.79 | 4.48 | 0.24 | 0.03 | 0.08 | 1.54 | 0.12 | ||||||
| White background | 1.98 | 2.27 | 0.53 | 4.29 | 54.45 | 4.13 | 0.69 | 0.79 | 30.87 | |||||||
| σ | 0.06 | 0.09 | 0.06 | 0.62 | 3.97 | 0.44 | 0.04 | 0.08 | 1.44 | |||||||
| VQC5 | Light Blue | 1.45 | 1.00 | 0.24 | 2.98 | 45.56 | 4.06 | 0.57 | 0.87 | 43.08 | 0.19 | |||||
| σ | 0.15 | 0.15 | 0.03 | 0.05 | 4.54 | 0.89 | 0.03 | 0.15 | 2.18 | 0.03 | ||||||
| Dark-brown | 1.56 | 1.23 | 0.41 | 3.15 | 45.23 | 4.00 | 0.65 | 0.72 | 42.32 | 0.73 | ||||||
| σ | 0.15 | 0.22 | 0.02 | 0.20 | 5.78 | 0.76 | 0.05 | 0.05 | 2.78 | 0.05 | ||||||
| White background | 2.49 | 0.86 | 0.34 | 2.92 | 40.30 | 3.40 | 0.69 | 3.26 | 45.74 | |||||||
| σ | 0.25 | 0.12 | 0.06 | 0.44 | 3.78 | 0.32 | 0.04 | 0.33 | 2.78 | |||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ricca, M.; Paladini, G.; Rovella, N.; Ruffolo, S.A.; Randazzo, L.; Crupi, V.; Fazio, B.; Majolino, D.; Venuti, V.; Galli, G.; et al. Archaeometric Characterisation of Decorated Pottery from the Archaeological Site of Villa dei Quintili (Rome, Italy): Preliminary Study. Geosciences 2019, 9, 172. https://doi.org/10.3390/geosciences9040172
Ricca M, Paladini G, Rovella N, Ruffolo SA, Randazzo L, Crupi V, Fazio B, Majolino D, Venuti V, Galli G, et al. Archaeometric Characterisation of Decorated Pottery from the Archaeological Site of Villa dei Quintili (Rome, Italy): Preliminary Study. Geosciences. 2019; 9(4):172. https://doi.org/10.3390/geosciences9040172
Chicago/Turabian StyleRicca, Michela, Giuseppe Paladini, Natalia Rovella, Silvestro Antonio Ruffolo, Luciana Randazzo, Vincenza Crupi, Barbara Fazio, Domenico Majolino, Valentina Venuti, Giuliana Galli, and et al. 2019. "Archaeometric Characterisation of Decorated Pottery from the Archaeological Site of Villa dei Quintili (Rome, Italy): Preliminary Study" Geosciences 9, no. 4: 172. https://doi.org/10.3390/geosciences9040172
APA StyleRicca, M., Paladini, G., Rovella, N., Ruffolo, S. A., Randazzo, L., Crupi, V., Fazio, B., Majolino, D., Venuti, V., Galli, G., & La Russa, M. F. (2019). Archaeometric Characterisation of Decorated Pottery from the Archaeological Site of Villa dei Quintili (Rome, Italy): Preliminary Study. Geosciences, 9(4), 172. https://doi.org/10.3390/geosciences9040172










