Responses of Crop Pests and Natural Enemies to Wildflower Borders Depends on Functional Group
Abstract
1. Introduction
2. Methods
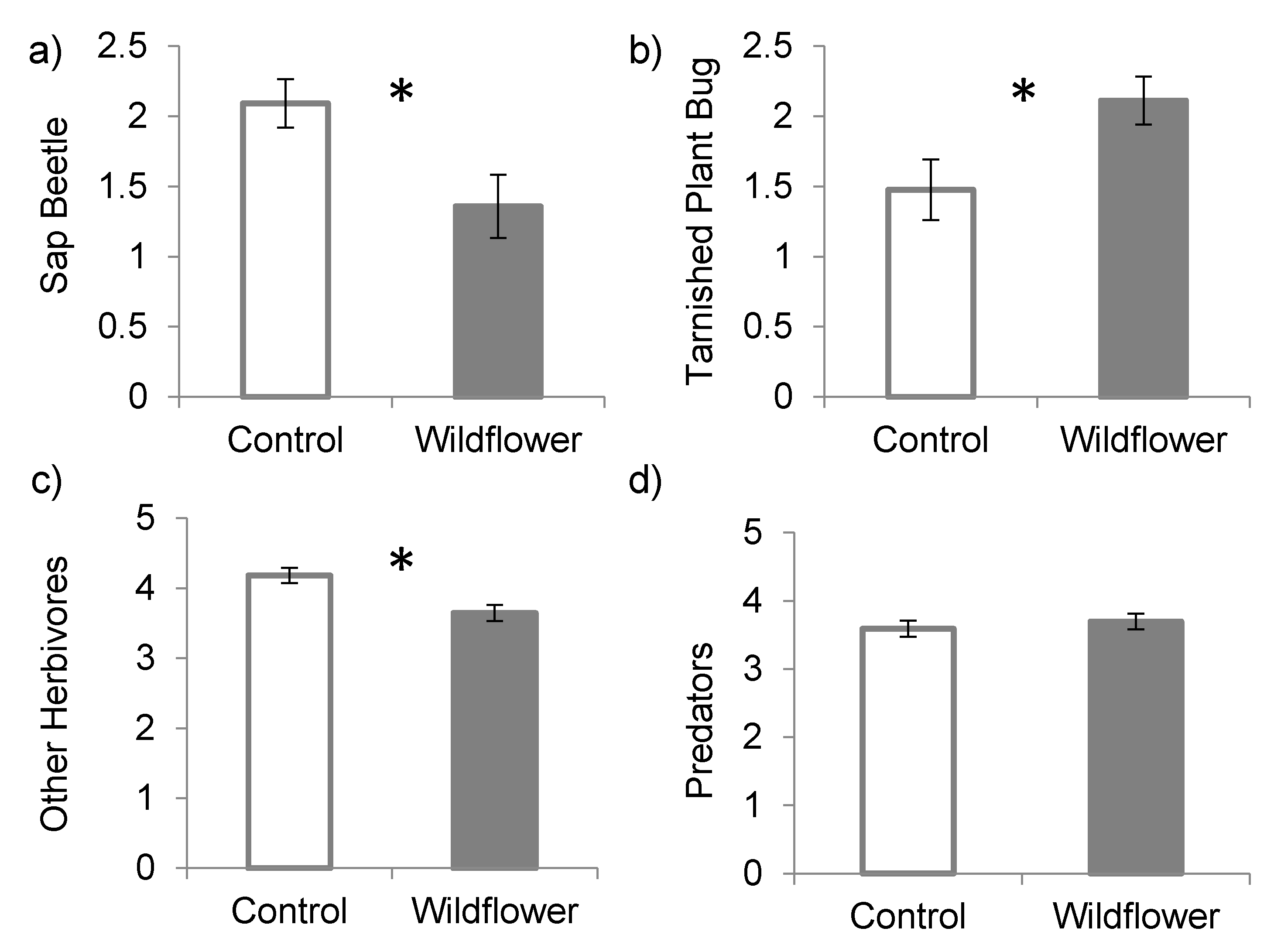
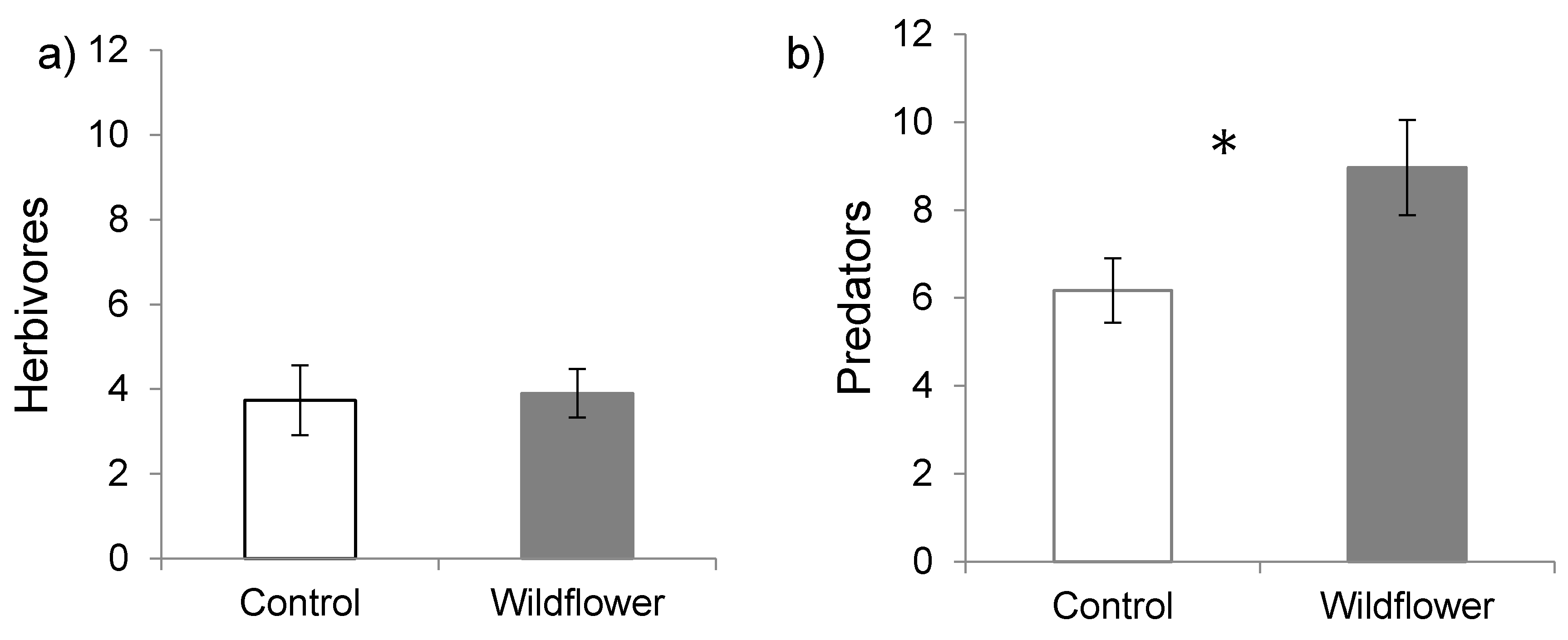
3. Results
4. Discussion
5. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Altieri, M. The ecological role of biodiversity in agroecosystem. Agric. Ecosyst. Environ. 1999, 74, 19–31. [Google Scholar] [CrossRef]
- Pywell, R.F.; Heard, M.S.; Woodcock, B.A.; Hinsley, S.; Ridding, L.; Nowakowski, M.; Bullock, J.M. Wildlife-friendly farming increases crop yield: Evidence for ecological intensification. Proc. R. Soc. B Biol. Sci. 2015, 282. [Google Scholar] [CrossRef] [PubMed]
- Holzschuh, A.; Dudenhöffer, J.H.; Tscharntke, T. Landscapes with wild bee habitats enhance pollination, fruit set and yield of sweet cherry. Biol. Conserv. 2012, 153, 101–107. [Google Scholar] [CrossRef]
- Thies, C.; Tscharntke, T. Landscape structure and biological control in agroecosystems. Science 1999, 285, 893–895. [Google Scholar] [CrossRef] [PubMed]
- Tscharntke, T.; Klein, A.M.; Kruess, A.; Steffan-Dewenter, I.; Thies, C. Landscape perspectives on agricultural intensification and biodiversity—Ecosystem service management. Ecol. Lett. 2005, 8, 857–874. [Google Scholar] [CrossRef]
- Newbold, T.; Hudson, L.N.; Hill, S.L.L.; Contu, S.; Lysenko, I.; Senior, R.A.; Börger, L.; Bennett, D.J.; Choimes, A.; Collen, B.; Day, J.; et al. Global effects of land use on local terrestrial biodiversity. Nature 2015, 520, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Tscharntke, T.; Clough, Y.; Wanger, T.C.; Jackson, L.; Motzke, I.; Perfecto, I.; Vandermeer, J.; Whitbread, A. Global food security, biodiversity conservation and the future of agricultural intensification. Biol. Conserv. 2012, 151, 53–59. [Google Scholar] [CrossRef]
- Kennedy, C.M.; Lonsdorf, E.; Neel, M.C.; Williams, N.M.; Ricketts, T.H.; Winfree, R.; Bommarco, R.; Brittain, C.; Burley, A.L.; Cariveau, D.; et al. A global quantitative synthesis of local and landscape effects on wild bee pollinators in agroecosystems. Ecol. Lett. 2013, 16, 584–599. [Google Scholar] [CrossRef] [PubMed]
- Rusch, A.; Chaplin-Kramer, R.; Gardiner, M.M.; Hawro, V.; Holland, J.; Landis, D.; Thies, C.; Tscharntke, T.; Weisser, W.W.; Winqvist, C.; et al. Agricultural landscape simplification reduces natural pest control: A quantitative synthesis. Agric. Ecosyst. Environ. 2016, 221, 198–204. [Google Scholar] [CrossRef]
- Chaplin-Kramer, R.; O’Rourke, M.E.; Blitzer, E.J.; Kremen, C. A meta-analysis of crop pest and natural enemy response to landscape complexity. Ecol. Lett. 2011, 14, 922–932. [Google Scholar] [CrossRef] [PubMed]
- Bommarco, R.; Kleijn, D.; Potts, S.G. Ecological intensification: Harnessing ecosystem services for food security. Trends Ecol. Evol. 2013, 28, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Fiedler, A.K.; Landis, D.A.; Wratten, S.D. Maximizing ecosystem services from conservation biological control: The role of habitat management. Biol. Control 2008, 45, 254–271. [Google Scholar] [CrossRef]
- Landis, D.A.; Wratten, S.D.; Gurr, G.M. Habitat management to conserve natural enemies of arthropod pests in agriculture. Annu. Rev. Entomol. 2000, 45, 175–201. [Google Scholar] [CrossRef] [PubMed]
- van Emden, H.F.; Emden, V.; Helmut, F. Conservation Biological Control: From Theory to Practice. In Proceedings of the International Symposium on Biological Control of Arthropods, Honolulu, HI, USA, 14–18 January 2002. [Google Scholar]
- Walton, N.J.; Isaacs, R. Influence of native flowering plant strips on natural enemies and herbivores in adjacent blueberry fields. Environ. Entomol. 2011, 40, 697–705. [Google Scholar] [CrossRef] [PubMed]
- Blaauw, B.R.; Isaacs, R. Wildflower plantings enhance the abundance of natural enemies and their services in adjacent blueberry fields. Biol. Control 2015, 91, 94–103. [Google Scholar] [CrossRef]
- Pfiffner, L.; Luka, H.; Schlatter, C.; Juen, A.; Traugott, M. Impact of wildflower strips on biological control of cabbage lepidopterans. Agric. Ecosyst. Environ. 2009, 129, 310–314. [Google Scholar] [CrossRef]
- Balmer, O.; Géneau, C.E.; Belz, E.; Weishaupt, B.; Förderer, G.; Moos, S.; Ditner, N.; Juric, I.; Luka, H. Wildflower companion plants increase pest parasitation and yield in cabbage fields: Experimental demonstration and call for caution. Biol. Control 2014, 76, 19–27. [Google Scholar] [CrossRef]
- Tschumi, M.; Albrecht, M.; Collatz, J.; Dubsky, V.; Entling, M.H.; Najar-Rodriguez, A.J.; Jacot, K. Tailored flower strips promote natural enemy biodiversity and pest control in potato crops. J. Appl. Ecol. 2016, 53, 1169–1176. [Google Scholar] [CrossRef]
- Tschumi, M.; Albrecht, M.; Entling, M.H.; Jacot, K. High effectiveness of tailored flower strips in reducing pests and crop plant damage. Proc. Biol. Sci. 2015. [Google Scholar] [CrossRef] [PubMed]
- Balzan, M.V.; Bocci, G.; Moonen, A.C. Augmenting flower trait diversity in wildflower strips to optimise the conservation of arthropod functional groups for multiple agroecosystem services. J. Insect Conserv. 2014, 18, 713–728. [Google Scholar] [CrossRef]
- Olson, D.M.; Wäckers, F.L. Management of field margins to maximize multiple ecological services. J. Appl. Ecol. 2007, 44, 13–21. [Google Scholar] [CrossRef]
- Berndt, L.A.; Wratten, S.D.; Scarratt, S.L. The influence of floral resource subsidies on parasitism rates of leafrollers (Lepidoptera: Tortricidae) in New Zealand vineyards. Biol. Control 2006, 37, 50–55. [Google Scholar] [CrossRef]
- Phillips, B.W.; Gardiner, M.M. Does local habitat management or large-scale landscape composition alter the biocontrol services provided to pumpkin agroecosystems? Biol. Control 2016, 92, 181–194. [Google Scholar] [CrossRef]
- Tscharntke, T.; Karp, D.S.; Chaplin-Kramer, R.; Batáry, P.; DeClerck, F.; Gratton, C.; Hunt, L.; Ives, A.; Jonsson, M.; Larsen, A.; et al. When natural habitat fails to enhance biological pest control—Five hypotheses. Biol. Conserv. 2016, 204, 449–458. [Google Scholar] [CrossRef]
- Jonsson, M.; Straub, C.S.; Didham, R.K.; Buckley, H.L.; Case, B.S.; Hale, R.J.; Gratton, C.; Wratten, S.D. Experimental evidence that the effectiveness of conservation biological control depends on landscape complexity. J. Appl. Ecol. 2015, 52, 1274–1282. [Google Scholar] [CrossRef]
- Wilson, H.; Miles, A.F.; Daane, K.M.; Altieri, M.A. Landscape diversity and crop vigor outweight influence of local diversification on biological control of a vineyard pest. Ecosphere 2017, 8. [Google Scholar] [CrossRef]
- Bommarco, R.; Miranda, F.; Bylund, H.; Björkman, C. Insecticides suppress natural enemies and increase pest damage in cabbage. J. Econ. Entomol. 2011, 104, 782–791. [Google Scholar] [CrossRef] [PubMed]
- Forehand, A.L.M.; Orr, D.B.; Linker, H.M. Insect communities associated with beneficial insect habitat plants in North Carolina. Environ. Entomol. 2006, 35, 1541–1549. [Google Scholar] [CrossRef]
- Feeny, P. Plant apparency and chemical defense. In Biochemical Interactions Between Plants and Insects; Wallace, J., Mansell, R., Eds.; Springer: London, UK, 1976; pp. 1–40. [Google Scholar]
- Castagneyrol, B.; Giffard, B.; Christelle, P. Plant apparency, an overlooked driver of associational resistance to insect herbivory. J. Ecol. 2013, 418–429. [Google Scholar] [CrossRef]
- Young, O. Host plants of the tarnished plant bug, Lygus lineolaris (Heteroptera: Miridae). Ann. Entomol. Soc. Am. 1986, 79, 747–762. [Google Scholar] [CrossRef]
- Schaefers, G.A. Yield effects of tarnished plant bug feeding on june-bearing strawberry varieties in New York State. J. Econ. Entomol. 1980, 73, 721–725. [Google Scholar] [CrossRef]
- Bostainian, N.J. The Tarnished Plant Bug and Strawberry Production; Agriculture and Agri-Food Canada: Ottawa, Cannada, 1994. [Google Scholar]
- Balzan, M.V.; Bocci, G.; Moonen, A.C. Utilisation of plant functional diversity in wildflower strips for the delivery of multiple agroecosystem services. Entomol. Exp. Appl. 2016, 158, 304–319. [Google Scholar] [CrossRef]
- Connell, W.A. Stelidota geminata (Say) infestations of strawberries (Coleoptera: Nitidulidae). Entomol. News 1980, 91, 55–56. [Google Scholar]
- Weber, R.G.; Connell, W.A. Stelidota geminata (Say): Studies of its biology (Coleoptera: Nitidulidae). Ann. Entomol. Soc. Am. 1975, 68, 649–653. [Google Scholar] [CrossRef]
- Loughner, R.L.; Loeb, G.M.; Demchak, K.; Schloemann, S. Evaluation of strawberry sap beetle (Coleoptera: Nitidulidae) use of habitats surrounding strawberry plantings as food resources and overwintering sites. Environ. Entomol. 2007, 36, 1059–1065. [Google Scholar] [CrossRef]
- Day, W.H. Evaluation of biological control of the tarnished plant bug (Hemiptera: Miridae) in alfalfa by the introduced parasite Peristenus digoneutis (Hymenoptera: Braconidae). Environ. Entomol. 1996, 25, 512–518. [Google Scholar] [CrossRef]
- Weiss, M.J.; Williams, R.N. Some host-parasite relationships of Microctonus nitidulidid and Stelidota geminata. Ann. Entomol. Soc. Am. 1980, 73, 323–326. [Google Scholar] [CrossRef]
- Tuell, J.K.; Fiedler, A.K.; Landis, D.; Isaacs, R.; Tuell, J.K.; Fiedler, A.K.; Landis, D. Visitation by Wild and Managed Bees (Hymenoptera : Apoidea) to Eastern U.S. Native Plants for Use in Conservation Programs. Environ. Entomol. 2008, 37, 707–718. [Google Scholar] [CrossRef]
- Isaacs, R.; Tuell, J.; Fiedler, A.; Gardiner, M.; Landis, D. Maximizing arthropod-mediated ecosystem services in agricultural landscapes: The role of native plants. Front. Ecol. Environ. 2009, 7, 196–203. [Google Scholar] [CrossRef]
- Baars, M.A. Catches in pitfall traps in relation to mean densities of carabid beetles. Oecologia 1979, 41, 25–46. [Google Scholar] [CrossRef] [PubMed]
- Spence, J.R.; Niemelä, J.K. Sampling Carabid Asseblages with pitfall traps: The madness and the method. Can. Entomol. 1994, 126, 881–894. [Google Scholar] [CrossRef]
- Lang, A.-G. The pitfalls of pitfalls: A comparison of pitfall trap catches and absolute density estimates of epigeal invertebrate predators in arable land. J. Pest Sci. 2000, 73, 99–106. [Google Scholar]
- Hunter, M.D.; Price, P.W. Playing Chutes and Ladders : Heterogeneity and the relative roles of bottom-up and top-down forces in natural communities. Ecology 1992, 73, 724–732. [Google Scholar]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McCabe, E.; Loeb, G.; Grab, H. Responses of Crop Pests and Natural Enemies to Wildflower Borders Depends on Functional Group. Insects 2017, 8, 73. https://doi.org/10.3390/insects8030073
McCabe E, Loeb G, Grab H. Responses of Crop Pests and Natural Enemies to Wildflower Borders Depends on Functional Group. Insects. 2017; 8(3):73. https://doi.org/10.3390/insects8030073
Chicago/Turabian StyleMcCabe, Ellie, Gregory Loeb, and Heather Grab. 2017. "Responses of Crop Pests and Natural Enemies to Wildflower Borders Depends on Functional Group" Insects 8, no. 3: 73. https://doi.org/10.3390/insects8030073
APA StyleMcCabe, E., Loeb, G., & Grab, H. (2017). Responses of Crop Pests and Natural Enemies to Wildflower Borders Depends on Functional Group. Insects, 8(3), 73. https://doi.org/10.3390/insects8030073





