3.1. Swarming Survey
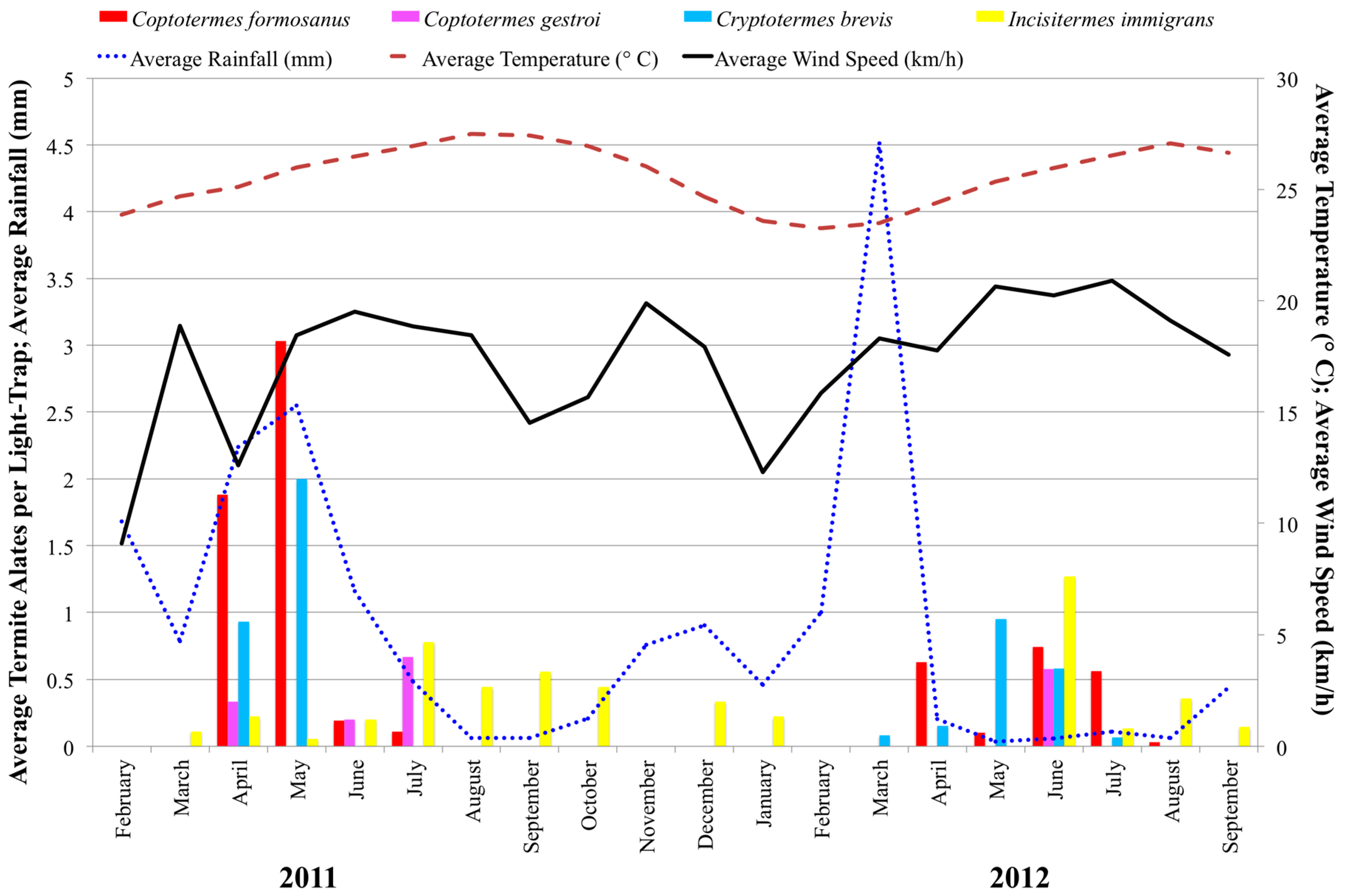
Termite alates were found every in month except February 2011, November 2011, and February 2012 (
Figure 2).
C. formosanus alates were captured between April and July 2011 and April and August 2012 from Aiea, Mililani, UH Mānoa, Waimanalo, and Washington Middle School (
Figure 2).
In months during which
C. formosanus alates were captured, a two-factor analysis of variance using combined location and month revealed capture rates differed by month (
F8,11 = 6.43,
p < 0.003), but not by location (
F4.15 = 1.16,
p < 0.367). Post-hoc Tukey’s HSD showed the peak month of capture to be May 2011 (
Table 4). Higa and Tamashiro [
31] found similar peaks in swarming during 1969 and 1970 with large numbers of
C. formosanus alates swarming from April to July.
The results of the multiple regression indicated that environmental factors predicted
C. formosanus alate capture (
R2 = 0.29,
F4,47 = 4.58,
p < 0.0025). Average wind speed (
β = −3.68,
p < 0.0006) and average rainfall (
β = 2.20,
p < 0.0325) significantly predicted
C. formosanus alate activity density, while average temperature and percent moon illumination did not (
Table 5a). Formosan subterranean termite swarming correlations with environmental factors are consistent with previous findings on Oahu that found wind velocity to be a key environmental factor regulating swarming [
32] (
Table 5b). Leong et al. [
32] found termite swarming to start when winds were below 3.7 km/h. However, Leong et al. [
32] used direct measurements, while the design of the present study used average wind speed per week, as termite alates were collected and combined by week. Despite these differences in methodology, wind velocity remains a predictive environmental factor for
C. formosanus swarming. When differences in wind speed are accounted for, average rainfall becomes a significant predictor of
C. formosanus alate capture.
C. formosanus termites’ high susceptibility to desiccation [
14] may explain the positive relationship of
C. formosanus capture with rainfall. Bess [
13] noted
C. formosanus swarmed on “humid days, often immediately followed by a shower”. Further, if rainfall is too high, it may also be detrimental to
C. formosanus [
31], which may contribute to the lower number of termite alates in 2012 compared to 2011.
Other factors than those tested in a multiple regression may influence the observed variable, possibly in a manner that correlates the observed variable to its past and future values, known as autocorrelation. Autocorrelation may inflate
t-values by decreasing the standard errors between time periods and overestimate the explanatory power of the multiple regressions’ factor(s). The Durbin-Watson statistic assessing serial autocorrelation was 1.67 (
p = 0.0697). A post-hoc comparison of the same regression of environmental factors to
C. formosanus alate activity density by trap monitor (citizen scientists vs. the termite lab) revealed differences in the Durbin–Watson statistic: D–W = 2.96 (
p = 0.9371) for citizen scientist-monitored traps, indicating no significant serial autocorrelation, and D–W = 1.01 (
p < 0.0004) for termite lab-monitored traps, indicating significant serial autocorrelation, which violates an assumption of the multiple regression analysis, which could be due to the study design. Light-traps for lab-monitored locations were hung above known
C. formosanus colonies, increasing the likelihood of consistent, and possibly auto-correlated, alate activity density. Another possible explanation for autocorrelation is the emergence pattern of
C. formosanus alates, in which alates emerge slowly over a period of three weeks or longer [
32]. Citizen scientist-monitored traps had a statistically insignificant Durbin-Watson statistic for autocorrelation; combination with lab data, therefore, decreased the multiple regression model’s likelihood for autocorrelation.
C. formosanus alate activity density in the summers of 2011 and 2012 were not similar. Average wind speed during swarming was 9.44 km/h (s.d. = 3.95) in 2011 and 16.81 km/h (s.d. = 4.89) in 2012, which may contribute to lower numbers of
C. formosanus alates observed in 2012. Other environmental variables, such as temperature, or intrinsic factors that influence
C. formosanus flight may also account for swarming variability. For example, in New Orleans, termite swarming showed a pause between swarming months, possibly due to developmental times [
48].
The height of light-traps may also influence the number of termite alates caught in light-traps. Lab-monitored traps were from 1.5 to 3 m high, with most about 2 m high. However, this variable was not recorded; further, it is not known at what height students hung traps, though it may be assumed they were within 1 to 3 m. The “PUB” light-trap from UH Mānoa was hung from a tree with a string-pulley that raised the trap to about 3 m. This trap also had the highest number of alates in a single week (n = 33). However, there were no significant differences between C. formosanus activity densities at different locations.
Data from the current study may provide a baseline for studies measuring termite suppression and changes in termite distributions on Oahu. However, studies comparing future swarming data over time should account for wind speed and rainfall, as these were significant predictors of C. formosanus alate capture.
Asian subterranean termite,
C. gestroi, alates were observed in April, June, and July 2011 (
n = 14) and in June of 2012 (
n = 8).
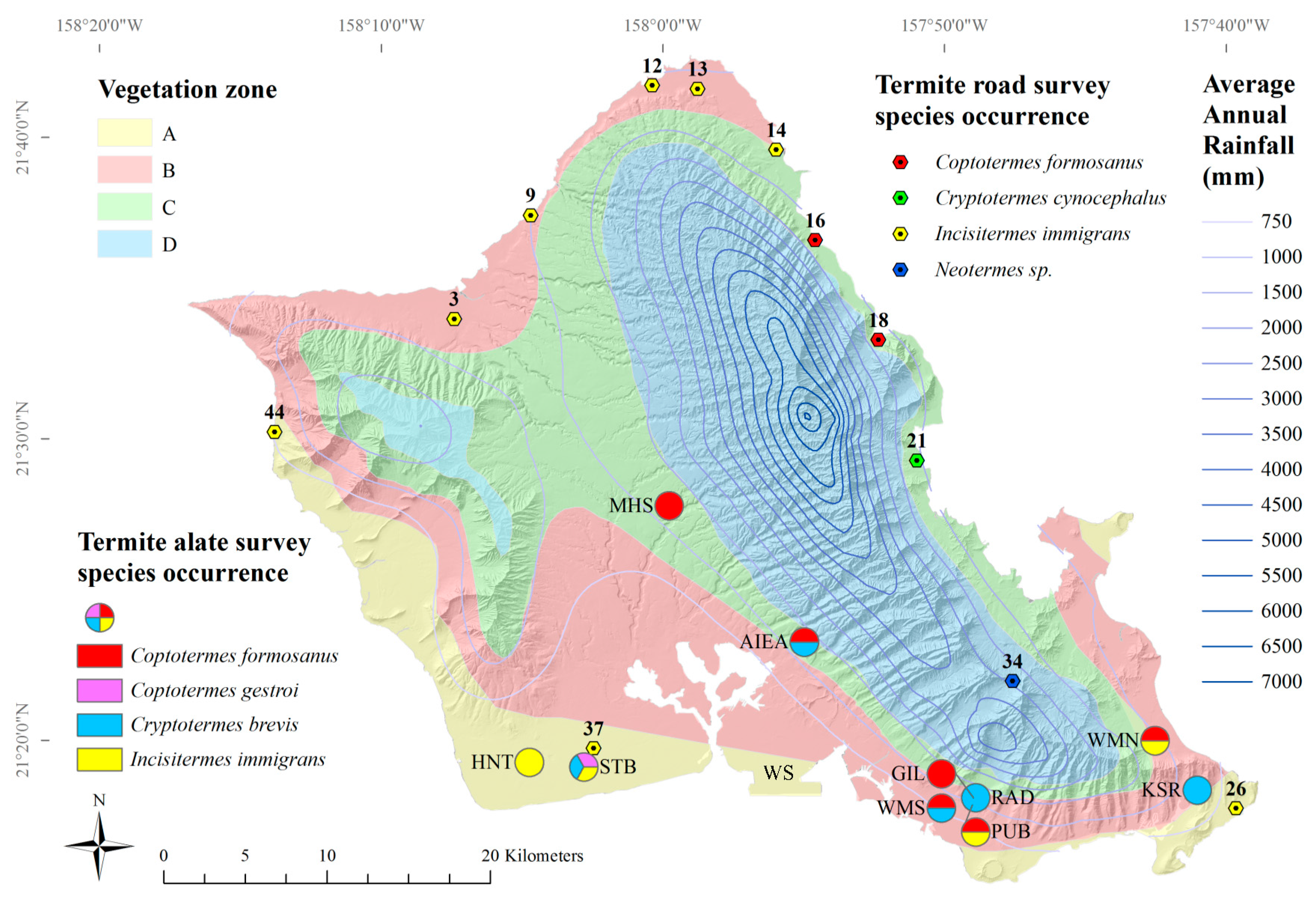
C. gestroi was found only in Kalaeloa (
Figure 3). West-Indian drywood termite,
Cr. brevis, alates were captured in April to June 2011 and April to July 2012.
Cr. brevis alates were found in Aiea, Kalaeloa, UH Mānoa, and Kaiser High School (
Figure 3).
Cr. brevis alates were captured primarily by citizen scientists, so the full range of swarming may not have been observed in 2011. Lowland tree termite,
I. immigrans, alates were observed every month except February and November in 2011 and from June through September in 2012.
I. immigrans alates were found in Kalaeloa, Makakilo, UH Mānoa, and Waimanalo (
Figure 3).
Indo-Malaysian drywood termite, Cr. cynocephalus, western drywood termite, I. minor, and Pacific dampwood termite, Z. angusticollis, alates were not observed in light-traps. A single desiccated Neotermes sp. was found in a Mililani High School student’s light-trap, and is assumed to be the forest tree termite, N. connexus, as it is the only species of this genus known from Hawaii, although its species could not be identified.
In addition to decreasing the autocorrelation in the multiple regression, citizen scientists expanded the geographic range of the study and increased the sample size. Participating teachers found the project a useful addition to the curriculum. Some students were able to use their data for further questions for science or senior projects. Light-trapping termites contributed to scientific literacy, as students gathered data, read scientific literature, and were taught to differentiate drywood and subterranean termite alates.
Not all species of termites that occur in Hawaii were found in light-traps. Future studies may include: continued alate trapping in current areas, alate trapping in new areas, and trapping near known locations of
Cr. cynocephalus,
N. connexus, and
Z. angusticollis to better define these species’ swarming phenologies on Oahu. The Western drywood termite,
I. minor, may not be attracted to light-traps, as they swarm during the day. The geographic range of the study was also limited, with no sites on the north or west portions of Oahu, and these areas may be targeted for future alate surveys. For
Cr. brevis, light-trapping may even be a control strategy, as it can decrease the number of dispersing alates [
49].
3.2. Hand-Collection Survey
Four species of termites were found from 44 roadside sites on Oahu, Hawaii (
Figure 4). The lowland tree termite,
I. immigrans, was most frequently encountered (
n = 8), followed by the Formosan subterranean termite,
C. formosanus (
n = 2) (
Table 6). The Indo-Malaysian drywood termite,
Cr. cynocephalus, was found at one site (
Table 6). A termite likely from the genus
Neotermes was found at one site (
Table 6), and only
Neotermes connexus is known from this genus in Hawaii.
C. gestroi,
Cr. brevis,
I. minor, and
Z. angusticollis were not found at any road survey site. Abiotic factors by species occurrences are given in
Table 7.
The lowland tree termite,
I. immigrans, was the most frequently encountered termite, consistent with the findings of Scheffrahn et al. [
20].
I. immigrans termites were found at around the same areas along the North Shore, Waianae Coast and down to Malaekahana State Park along the Windward Coast, although they were absent from several areas previously identified along the Windward coast [
20].
I. immigrans termites were found near alate light-traps that captured
I. immigrans,
C. gestroi, and
Cr. brevis (
Figure 3).
I. immigrans alates were found in four out of ten light-trap locations (
Figure 3).
I. immigrans was not significantly affected by average elevation or average annual rainfall (GLM,
F = 4.61,
p < 0.10).
The two road survey occurrences of
C. formosanus were not found near alate light-trap locations (
Figure 3).
C. formosanus alates were found in six out of ten light-trap locations (
Figure 3). Formosan subterranean termite,
C. formosanus, workers and soldiers were found crawling freely (not in mud tubes) along the base of a tree in Hauula and in a tree stump in Ahupuaa O Kahana State Park. Scheffrahn et al. [
20] found
C. formosanus colonies near the same area, but further south, along Kaneohe Bay, as well as Waahila Ridge, Heeia, Kahaluu, and Punaluu. The previous study [
20] also found
N. connexus termites where the current study found
C. formosanus. This may be because of both species’ relatively high moisture requirement.
C. formosanus distributions may further be delimited with increased light-trapping on the north and west sides of Oahu. It is not clear whether
C. formosanus is actually decreasing in range or if the current survey methodology resulted in fewer findings. However, years of termite education, prevention measures (e.g., Basaltic Termite Barrier, Termi-mesh, termite-resistant wood, etc.), bait-systems and other chemical controls may have contributed to smaller populations of
C. formosanus.
Cr. brevis alates were found at half of all light-trap sites (
Figure 3); however,
Cr. brevis termites were not found at any road survey location. This is not unexpected, since this species very rarely occurs in natural settings, although known to be well-established in structures throughout the island.
Cr. brevis was extremely common in alate light-traps (
Figure 3). Alate trapping is, therefore, recommended to study distributions of
Cr. brevis.
The Indo-Malaysian drywood termite,
Cr. cynocephalus, was found in fallen logs near a stream between the two sites at which it was first found in 1996: Waiahole Valley Road and Kualoa Regional Park [
20]. Several nymphs were found, along with alates, indicating a mature colony.
Cr. cynocephalus termites were not found at any surrounding survey sites, which may mean its dispersal is still somewhat limited.
The Western drywood termite, I. minor, was not found at any sites, possibly because it has yet to expand its range, or because it remains in structures that were not examined. I. minor swarms during the day, and may not be attracted to light-traps. Incidental reports may be the best method at present for locating I. minor infestations. Pacific dampwood termite, Z. angusticollis, was not found in this survey. Currently, its known distribution remains limited to Kula, Maui. Survey sites in this study were mostly at low elevations near coastlines. Tree branches with evidence of termites were found at multiple locations, but live termites were not always found, especially with regard to subterranean termites. Subterranean termites are cryptic, and light-trap surveys may be more effective in mapping their distribution than hand-collection techniques. Crowd sourcing, or gathering data from the public on occurrences of termites may also be an option, and pest control reports may also be useful.
At some locations, soldiers and imagoes (alate and dealate reproductives) were not found. At the Pali lookout site (map code 34), two pairs of dealate reproductive termites were found, but positive identification was difficult because of the lack of wings and absence of any soldiers. These dealates were darker, had a smaller pronotum width, a proportionately wider head to pronotum ratio, and larger eyes than voucher N. connexus alates. Further surveys in the area may reveal soldiers and more imago samples for positive identification.