The Impact of Diet Protein and Carbohydrate on Select Life-History Traits of The Black Soldier Fly Hermetia illucens (L.) (Diptera: Stratiomyidae)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Insect Colony
2.2. Artificial Diet Experiments
2.3. Data Analysis
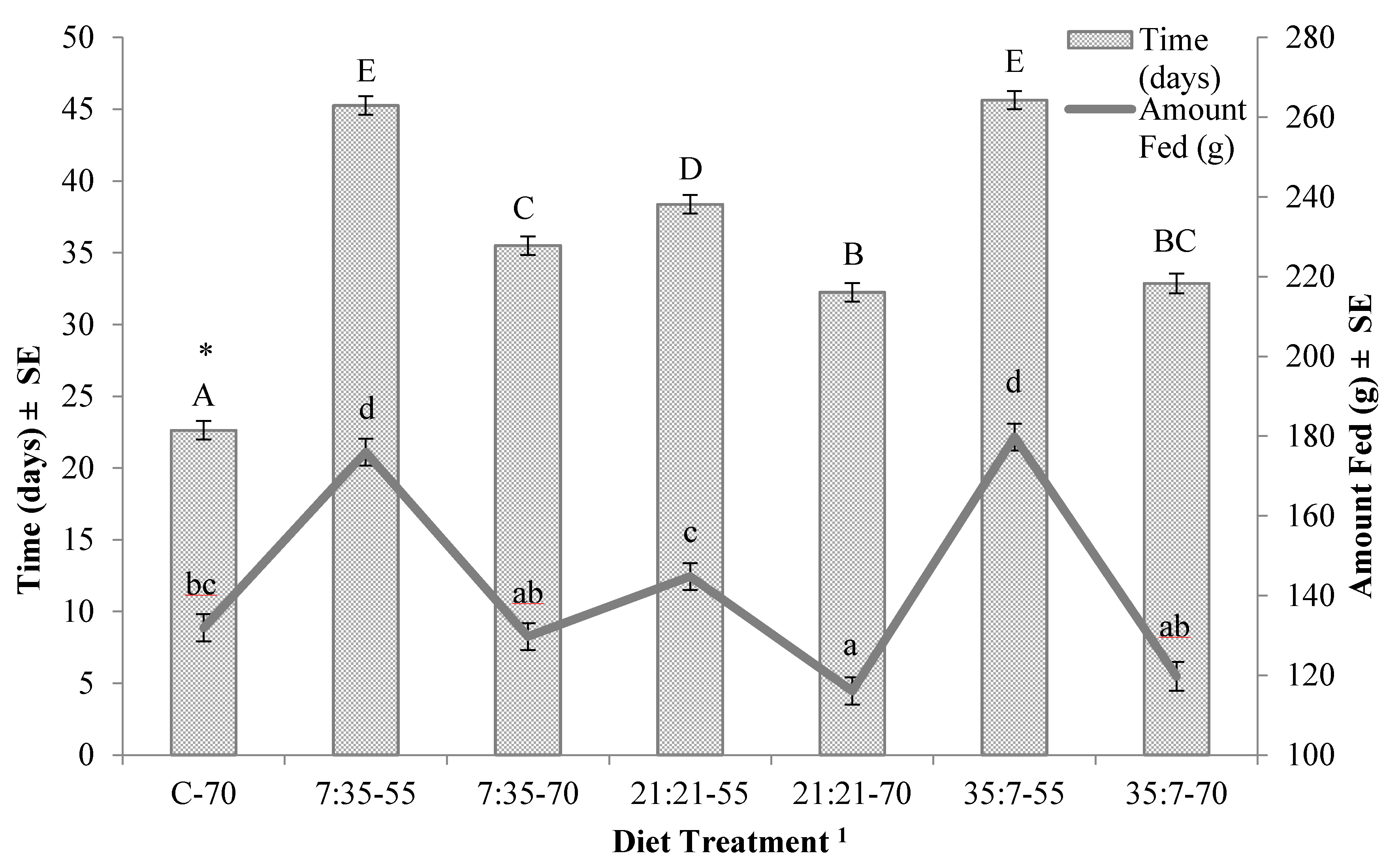
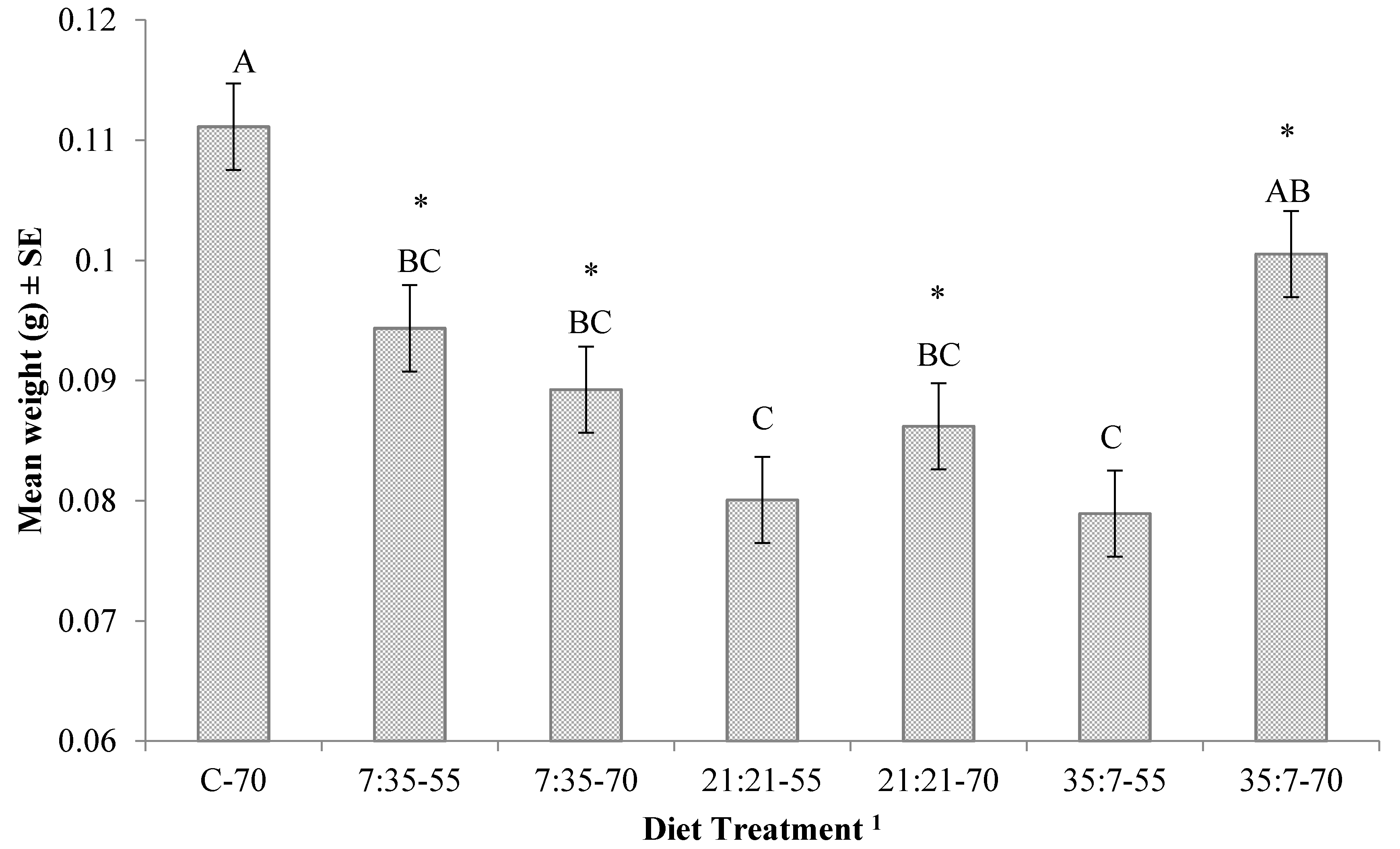
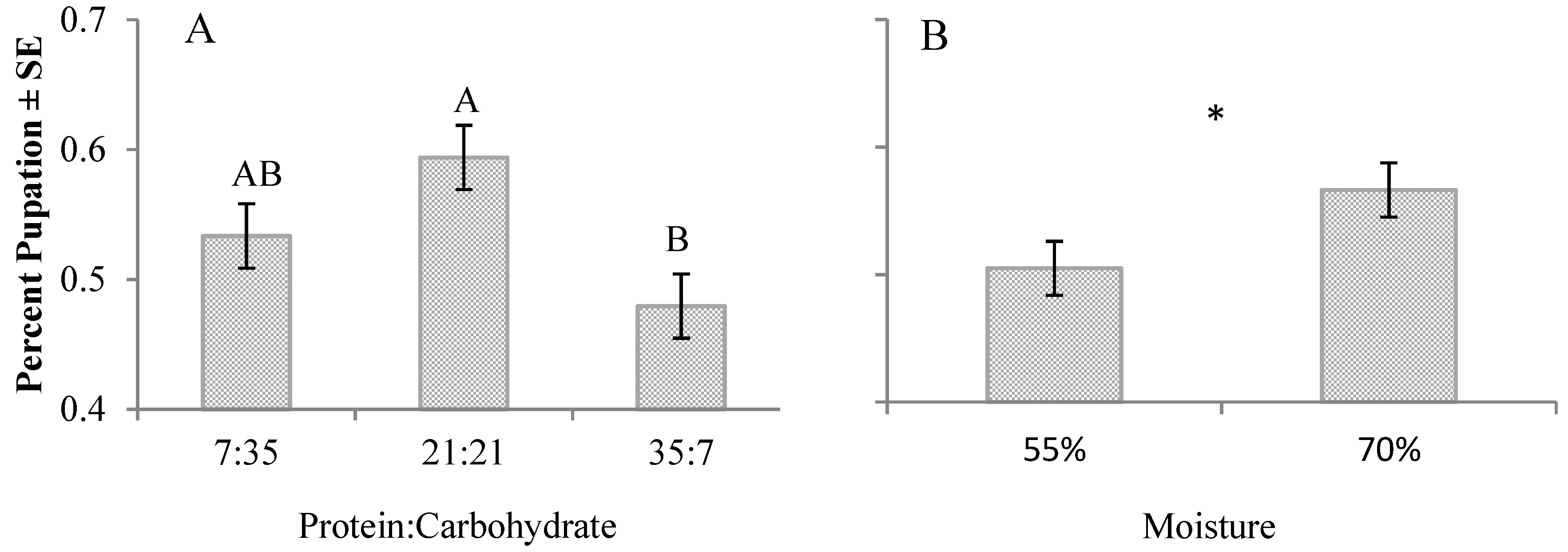
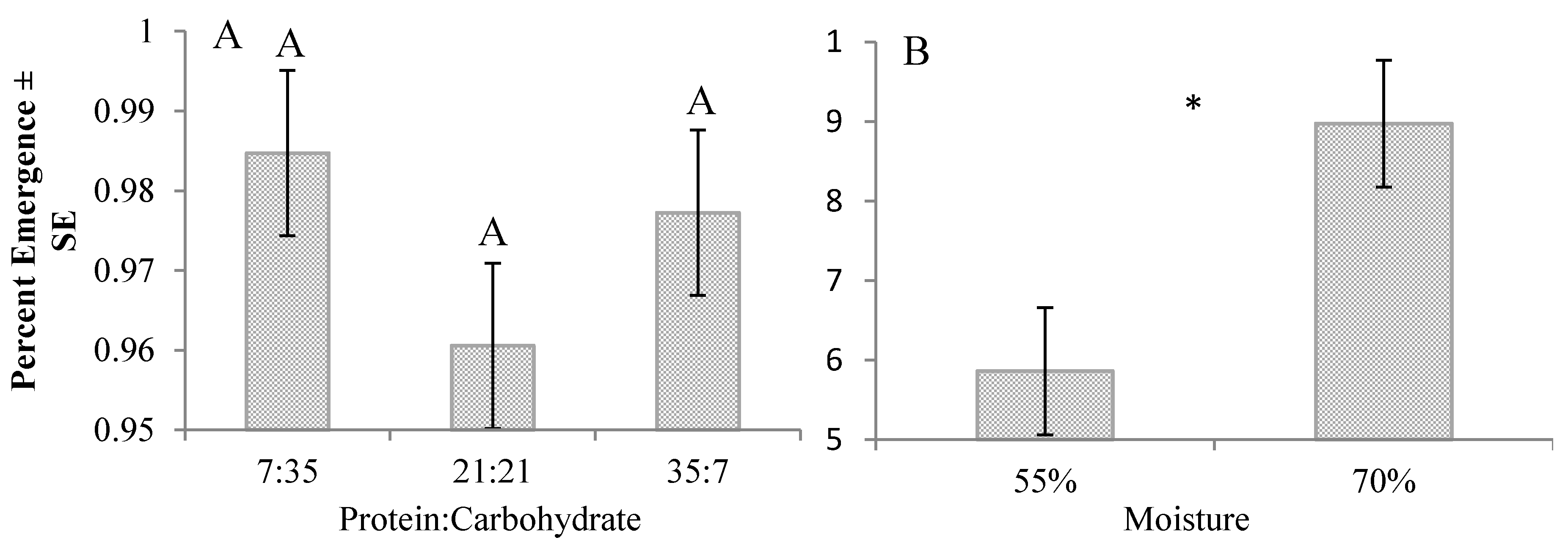
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hoornweg, D.; Bhada-Tata, P.; Kennedy, C. Waste production must peak this century. Nature 2013, 502, 615–617. [Google Scholar] [CrossRef] [PubMed]
- Food Wastage Footprint. Impacts on Natural Resources. Summary Report; Food and Agriculture Organization of the United Nations: Rome, Italy, 2013; p. 63.
- Stone, A.; Sabrosky, C.W.; Wirth, W.W.; Foote, R.H.; Coulson, J. A catalog of the diptera of America north of Mexico; U.S. Government Printing Office: Washington, DC, USA, 1965.
- Callan, E.M. Hermetia illucens (Dipt., Stratiomyidae), A cosmopolitan american species long established in Australia and New-Zealand. Entomol. Mon. Mag. 1974, 109, 232–234. [Google Scholar]
- Stephens, C.S. Hermetia illucens (Diptera: Stratiomyidae) as a banana pest in Panama. Trop. Agric. 1975, 52, 173–178. [Google Scholar]
- Tomberlin, J.K.; Sheppard, D.C.; Joyce, J.A. Black soldier fly (Diptera: Stratiomyidae) colonization of pig carrion in south Georgia. J. Forensic. Sci. 2005, 50, 152–153. [Google Scholar] [CrossRef] [PubMed]
- Dunn., L.H. Hermetia illucens breeding in a human cadaver (Dipt.). Entomol. News 1916, 27, 59–60. [Google Scholar]
- Pujol-Luz, J.R.; Francez, P.; Ururahy-Rodrigues, A.; Constantino, R. The black soldier-fly, Hermetia illucens (Diptera, Stratiomyidae), used to estimate the postmortem interval in a case in Amapa State, Brazil. J. Forensic. Sci. 2008, 53, 476–478. [Google Scholar] [CrossRef] [PubMed]
- St-Hilaire, S.; Cranfill, K.; McGuire, M.A.; Mosley, E.E.; Tomberlin, J.K.; Newton, L.; Sealey, W.; Sheppard, C.; Irving, S. Fish offal recycling by the black soldier fly produces a foodstuff high in omega-3 fatty acids. J. World Aquacult. Soc. 2007, 38, 309–313. [Google Scholar] [CrossRef]
- Diener, S.; Solano, N.M.S.; Gutierrez, F.R.; Zurbrugg, C.; Tockner, K. Biological treatment of municipal organic waste using black soldier fly larvae. Waste Biomass Valorization 2011, 2, 357–363. [Google Scholar] [CrossRef]
- Nguyen, T.T.X.; Tomberlin, J.K.; Vanlaerhoven, S. Ability of black soldier fly (Diptera: Stratiomyidae) larvae to recycle food waste. Environ. Entomol. 2015, 44, 406–410. [Google Scholar] [CrossRef] [PubMed]
- Oonincx, D.G.A.B.; van Broekhoven, S.; van Huis, A.; van Loon, J.J. Feed conversion, survival and development, and composition of four insect species on diets composed of food by-products. PLoS ONE 2015, 10, e0144601. [Google Scholar] [CrossRef] [PubMed]
- Banks, I.J.; Gibson, W.T.; Cameron, M.M. Growth rates of black soldier fly larvae fed on fresh human faeces and their implication for improving sanitation. Trop. Med. Int. Health 2014, 19, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Myers, H.M.; Tomberlin, J.K.; Lambert, B.D.; Kattes, D. Development of black soldier fly (Diptera: Stratiomyidae) larvae fed dairy manure. Environ. Entomol. 2008, 37, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Oonincx, D.G.A.B.; van Huis, A.; van Loon, J.J.A. Nutrient utilisation by black soldier flies fed with chicken, pig, or cow manure. J. Insects Food Feed 2015, 1, 131–139. [Google Scholar] [CrossRef]
- Fatchurochim, S.; Geden, C.J.; Axtell, R.C. Filth fly (Diptera) oviposition and larval development in poultry manure of various moisture levels. J. Entomol. Sci. 1989, 24, 224–231. [Google Scholar]
- Sheppard, D.C.; Tomberlin, J.K.; Joyce, J.A.; Kiser, B.; Sumner, S.M. Rearing methods for the black soldier fly (Diptera: Stratiomyidae). J. Med. Entomol. 2002, 39, 695–698. [Google Scholar] [CrossRef] [PubMed]
- Newton, G.L.; Sheppard, D.C.; Watson, D.W.; Burtle, G.J.; Dove, C.R.; Tomberlin, J.K.; Thelen, E.E. The Black Soldier Fly, Hermetia Illucens, As a Manure Management/resource Recovery Tool. In Proceedings of the Symposium, State of teh Science, Animal Manure and Waste Management, San Antonio, TX, USA, 5–7 January 2005. [Google Scholar]
- Sheppard, D.C.; Newton, G.L.; Thompson, S.A.; Savage, S. A value added manure management system using the black soldier fly. Bioresource Technol. 1994, 50, 275–279. [Google Scholar] [CrossRef]
- Bondari, K.; Sheppard, D.C. Soldier fly, Hermetia illucens L., as feed for channel catfish, Ictalurus punctatus (Rafinesque), and blue tilapia, Oreochromis aureus (Steindachner). Aquacult. Fish. Manag. 1987, 18, 209–220. [Google Scholar] [CrossRef]
- St-Hilaire, S.; Sheppard, C.; Tomberlin, J.K.; Irving, S.; McGuire, M.A.; Mosley, E.E.; Hardy, R.W.; Sealey, W. Fly prepupae as a feedstuff for rainbow trout, Oncorhynchus mykiss. J. World Aquacult. Soc. 2007, 38, 59–67. [Google Scholar] [CrossRef]
- Magalhaes, R.; Sanchez-Lopez, A.; Leal, R.S.; Martinez-Llorens, S.; Oliva-Teles, A.; Peres, H. Black soldier fly (Hermetia illucens) pre-pupae meal as a fish meal replacement in diets for European seabass (Dicentrarchus labrax). Aquaculture 2017, 476, 79–85. [Google Scholar] [CrossRef]
- Newton, G.L.; Booram, C.V.; Barker, R.W.; Hale, O.M. Dried Hermetia illucens larvae meal as a supplement for swine. J. Anim. Sci. 1977, 44, 395–400. [Google Scholar] [CrossRef]
- Hale, O.M. Dried hermetia illucens larvae (stratiomyidae) as a feed additive for poultry. J. Georgia Entomol. Soc. 1973, 8, 16–20. [Google Scholar]
- Schiavone, A.; Cullere, M.; De Marco, M.; Meneguz, M.; Biasato, I.; Bergagna, S.; Dezzutto, D.; Gai, F.; Dabbou, S.; Gasco, L.; Dalle Zotte, A. Partial or total replacement of soybean oil by black soldier fly larvae (Hermetia illucens L.) fat in broiler diets: effect on growth performances, feed-choice, blood traits, carcass characteristics and meat quality. Italian J. Anim. Sci. 2017, 16, 93–100. [Google Scholar] [CrossRef]
- Makkar, H.P.S.; Tran, G.; Heuze, V.; Ankers, P. State-of-the-art on use of insects as animal feed. Anim. Feed Sci. Tech. 2014, 197, 1–33. [Google Scholar] [CrossRef]
- 2016 AAFCO Annual Meeting Agenda and Committee Reports; Association of American Feed Control Officials: Pittsburgh, PA, USA, 2016; p. 112.
- Li, Q.; Zheng, L.; Qiu, N.; Cai, H.; Tomberlin, J.K.; Yu, Z. Bioconversion of dairy manure by black soldier fly (diptera: Stratiomyidae) for biodiesel and sugar production. Waste Manag. 2011, 31, 1316–1320. [Google Scholar] [CrossRef] [PubMed]
- Surendra, K.C.; Olivier, R.; Tomberlin, J.K.; Jha, R.; Khanal, S.K. Bioconversion of organic wastes into biodiesel and animal feed via insect farming. Renew. Energy 2016, 98, 197–202. [Google Scholar] [CrossRef]
- Park, B.K.; Kim, M.M. Applications of chitin and its derivatives in biological medicine. Int. J. Mol. Sci. 2010, 11, 5152–5164. [Google Scholar] [CrossRef] [PubMed]
- Hamed, I.; Ozogul, F.; Regenstein, J.M. Industrial applications of crustacean by-products (chitin, chitosan, and chitooligosaccharides): A review. Trends Food. Sci. Tech. 2016, 48, 40–50. [Google Scholar] [CrossRef]
- Deans, C.A.; Sword, G.A.; Behmer, S.T. Revisiting macronutrient regulation in the polyphagous herbivor Helicoverpa zea (Lepidoptera: Noctuidae): New insights via nutritional geometry. J. Insect Physiol. 2015, 81, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Joern, A.; Provin, T.; Behmer, S.T. Not just the usuaul suspects: Insect herbivore populations and communities are associated with multiple plant nutrients. Ecology 2012, 93, 1002–1015. [Google Scholar] [CrossRef] [PubMed]
- Raubenheimer, D.; Simpson, S.J. Nutrient balancing in grasshoppers: Behavioural and physiological correlates of dietary breadth. J. Exp. Biol. 2003, 206, 1669–1681. [Google Scholar] [CrossRef] [PubMed]
- Simpson, S.J.; Abisgold, J.D. Compensation by locusts for changes in dietary nutrients—behavioral mechanisms. Physiol. Entomol. 1985, 10, 443–452. [Google Scholar] [CrossRef]
- Hogsette, J.A. New diets for production of house flies and stable flies (Diptera: Muscidae) in the laboratory. J. Econ. Entomol. 1992, 85, 2291–2294. [Google Scholar] [CrossRef] [PubMed]
- Behmer, S.T.; Raubenheimer, D.; Simpson, S.J. Frequency-dependent food selection in locusts: A geometric analysis of the role of nutrient balancing. Anim. Behav. 2001, 61, 995–1005. [Google Scholar] [CrossRef]
- Tomberlin, J.K.; Sheppard, D.C.; Joyce, J.A. Selected life-history traits of black soldier flies (Diptera: Stratiomyidae) reared on three artificial diets. Ann. Entomol. Soc. Am. 2002, 95, 379–386. [Google Scholar] [CrossRef]
- Roeder, K.A.; Behmer, S.T. Lifetime consequences of food protein-carbohydrate content for an insect herbivore. Funct. Ecol. 2014, 28, 1135–1143. [Google Scholar] [CrossRef]
- Nash, W.J.; Chapman, T. Effect of dietary components on larval life history characteristics in the medfly (Ceratitis capitata: Diptera, Tephritidae). PLoS ONE 2014, 9, e86029. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.T.X.; Tomberlin, J.K.; Vanlaerhoven, S. Influence of resources on Hermetia illucens (Diptera: Stratiomyidae) larval development. J. Med. Entomol. 2013, 50, 898–906. [Google Scholar] [CrossRef] [PubMed]
- Tomberlin, J.K.; Adler, P.H.; Myers, H.M. Development of the black soldier fly (Diptera: Stratiomyidae) in relation to temperature. Environ. Entomol. 2009, 38, 930–934. [Google Scholar] [CrossRef] [PubMed]
- Runagall-McNaull, A.; Bonduriansky, R.; Crean, A.J. Dietary protein and lifespan across the metamorphic boundary: Protein-restricted larvae develop into short-lived adults. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Sentinella, A.T.; Crean, A.J.; Bonduriansky, R. Dietary protein mediates a trade-off between larval survival and the development of male secondary sexual traits. Funct. Ecol. 2013, 27, 1134–1144. [Google Scholar] [CrossRef]
- Li, C.; Xu, B.; Wang, Y.; Yang, Z.; Yang, W. Protein content in larval diet affects adult longevity and antioxidant gene expresson in honey bee workers. Entomol. Exp. Appl. 2014, 151, 19–26. [Google Scholar] [CrossRef]
- Tocher, D.R. Fatty acid requirements in ontogeny of marine and freshwater fish. Aquac. Res. 2010, 41, 717–732. [Google Scholar] [CrossRef]
- Tomberlin, J.K.; van Huis, A.; Benbow, M.E.; Jordan, H.; Astuti, D.A.; Azzollini, D.; Banks, I.; Bava, V.; Borgmeister, C.; Cammack, J.A.; et al. Protecting the environment through insect farming as a means to produce protein for use as livestock, poultry, and aquaculture feed. J. Insects Food Feed 2015, 1, 307–309. [Google Scholar] [CrossRef]
- Raubenheimer, D.; Simpson, S.J.; Mayntz, D. Nutrition, ecology, and nutritional ecology: Toward an integrated framework. Funct. Ecol. 2009, 23, 4–16. [Google Scholar] [CrossRef]
- Simpson, S.J.; Raubenheimer, D. The nature of nutrition: A unifying framework. Aust. J. Zool. 2011, 59, 350–368. [Google Scholar] [CrossRef]
- Ur Rehman, K.; Rehman, A.; Cai, M.; Zheng, L.; Xiao, X.; Somroo, A.A.; Wanh, H.; Li, W.; Yu, Z.; Zhang, J. Conversion of mixtures od dairy manure and soybean curd residue by black soldier fly larvae (Hermetia illucens L.). J. Clean. Prod. 2017, 154, 366–373. [Google Scholar] [CrossRef]
| Quantity/100 g | Poultry Feed | Swine Liver | Swine Manure | Restaurant Waste | Fruits and Vegetables | Fish Rendering | |
|---|---|---|---|---|---|---|---|
| Waste Stream | Calories | 310.48 | 442.68 | 295.23 | 484.32 | 375 | 502.76 |
| Protein (g) | 18.02 | 76.71 | 22.66 | 20.41 | 20.07 | 50 | |
| Fat (g) | 2.52 | 12.84 | 1.4 | 19.58 | 1.55 | 36.18 | |
| Carbohydrate (g) | 53.62 | 4.74 | 47.61 | 56.79 | 68.95 | 0.55 | |
| Protein:carbohydrate | 1:2.97 | 16.18:1 | 1:2.1 | 1:2.78 | 1:3.44 | 90.9:1 | |
| Resulting BSF * Prepupae | Calories | 130 | 214 | − | − | 105 | 233 |
| Protein (g) | 14.7 | 21 | − | 21.2 | 12.9 | 19.4 | |
| Fat (g) | 4.02 | 8.39 | − | − | 2.22 | 11.6 | |
| Carbohydrate (g) | 8.75 | 13.7 | − | − | 8.38 | 12.7 |
| Study | Diet | Mean Duration (d = Day) of Larval Development ± SE | Percent Survival 2 ± SE |
|---|---|---|---|
| Myers et al. [14] | Cow (Dairy) Manure | 30.4 ± 0.1 | 77.3 ± 4.55 |
| Nguyen et al. [41] 1 | Poultry Feed | 23.0 ± 0.6 | 80.8 ± NA |
| Pork Liver | 22.5 ± 0.7 | 57.2 ± NA | |
| Swine Manure | 34.0 ± 1.4 | 74.3 ± NA | |
| Kitchen Waste | 23.8 ± 0.4 | 46.7 ± NA | |
| Fruits and Vegetables | 28.7 ± 0.8 | 76.7 ± NA | |
| Fish Offal | 26.5 ± 0.9 | 47.2 ± NA | |
| Oonincx et al. [12] 6 | HPHF | 21 ± 1.4 | 86 ± 18.0 |
| HPLF | 33 ± 5.4 | 77 ± 19.8 | |
| LPHF | 37 ± 10.6 | 72 ± 12.9 | |
| LPLF | 37 ± 5.8 | 74 ± 23.5 | |
| Control | 21 ± 1.1 | 75 ± 31.0 | |
| Current Study 3 | Control (T1) 4 | 23.5 ± 0.4 | 87.0 ± 3.30 |
| (T2) | 21.8 ± 0.4 | ||
| 7:35–55 | 45.3 ± 0.7 | 48.9 ± 3.30 | |
| 7:35–70 | 35.5 ± 0.7 | 57.8 ± 3.30 | |
| 21:21–55 | 38.4 ± 0.7 | 56.7 ± 3.30 | |
| 21:21–70 (T1) 5 | 32.3 ± 0.7 | 68.2 ± 1.96 | |
| (T2) | 56.0 ± 1.96 | ||
| 35:7–55 | 45.6 ± 0.7 | 46.0 ± 3.30 | |
| 35:7–70 | 32.9 ± 0.7 | 52.9 ± 3.53 |
| Study 1 | Diet | Duration 2 (Days) of Larval Development ± SE | Percent Difference between Published Studies and Current Study 3 Data | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Control | 7:35–55 | 7:35–70 | 21:21–55 | 21:21–70 | 35:7–55 | 35:7–70 | |||
| Myers et al. [14] | Cow (Dairy) Manure | 30.4 ± 0.1 | −29% | +39% | +15% | +23% | +6% | +40% | +8% |
| Nguyen et al. [41] | Poultry Feed | 23.0 ± 0.6 | −2% | +65% | +43% | +50% | +34% | +66% | +35% |
| Pork Liver | 22.5 ± 0.7 | +0.6% | +67% | +45% | +52% | +36% | +68% | +37% | |
| Swine Manure | 34.0 ± 1.4 | −40% | +28% | +4% | +12% | −5% | +29% | −3% | |
| Kitchen Waste | 23.8 ± 0.4 | −5% | +62% | +39% | +47% | +30% | +63% | +32% | |
| Fruits and Vegetables | 28.7 ± 0.8 | −24% | +45% | +21% | +29% | +12% | +46% | +14% | |
| Fish Offal | 26.5 ± 0.9 | −16% | +52% | +29% | +37% | +20% | +53% | +21% | |
| Oonincx et al. [12] 4 | HPHF | 21 ± 1.4 | +7% | +73% | +51% | +59% | +43% | +74% | +44% |
| HPLF | 33 ± 5.4 | −37% | +31% | +7% | +15% | −2% | +32% | −0.4% | |
| LPHF | 37 ± 10.6 | −48% | +20% | −4% | +4% | −13% | +21% | −12% | |
| LPLF | 37 ± 5.8 | −48% | +20% | −4% | +4% | −13% | +21% | −12% | |
| Control | 21 ± 1.1 | +7% | +73% | +51% | +59% | +43% | +74% | +44% | |
| Mean difference | −19% | +48% | +25% | +32% | +16% | +49% | +17% | ||
| Study 1 | Diet | Percent Survival ± SE | Percent Difference between Published Studies and Current Study 2 Data | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Control | 7:35–55 | 7:35–70 | 21:21–55 | 21:21–70 | 35:7–55 | 35:7–70 | |||
| Myers et al. [14] | Cow (Dairy) Manure | 77.3 ± 4.55 | +12% | −45% | −29% | −31% | −22% | −51% | −37% |
| Nguyen et al. [41] | Poultry Feed | 80.8 ± NA | +7% | −49% | −33% | −35% | −26% | −55% | −42% |
| Pork Liver | 57.2 ± NA | +41% | −16% | +1% | −1% | +8% | −22% | −8% | |
| Swine Manure | 74.3 ± NA | +16% | −41% | −25% | −27% | −18% | −47% | −34% | |
| Kitchen Waste | 46.7 ± NA | +60% | +5% | +21% | +19% | +28% | −2% | +12% | |
| Fruits and Vegetables | 76.7 ± NA | +12% | −44% | −28% | −30% | −21% | −50% | −37% | |
| Fish Offal | 47.2 ± NA | +60% | +4% | +20% | +18% | +27% | −3% | +11% | |
| Oonincx et al. [12] 3 | HPHF | 86 ± 18.0 | +1% | −55% | −39% | −41% | −32% | −61% | −48% |
| HPLF | 77 ± 19.8 | +12% | −45% | −28% | −30% | −21% | −51% | −37% | |
| LPHF | 72 ± 12.9 | +19% | −38% | −22% | −24% | −15% | −44% | −31% | |
| LPLF | 74 ± 23.5 | +16% | −41% | −25% | −26% | −17% | −47% | −33% | |
| Control | 75 ± 31.0 | +15% | −42% | −25% | −28% | −19% | −48% | −35% | |
| Mean difference | +17% | −16% | −6% | −7% | −2% | −19% | −11% | ||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cammack, J.A.; Tomberlin, J.K. The Impact of Diet Protein and Carbohydrate on Select Life-History Traits of The Black Soldier Fly Hermetia illucens (L.) (Diptera: Stratiomyidae). Insects 2017, 8, 56. https://doi.org/10.3390/insects8020056
Cammack JA, Tomberlin JK. The Impact of Diet Protein and Carbohydrate on Select Life-History Traits of The Black Soldier Fly Hermetia illucens (L.) (Diptera: Stratiomyidae). Insects. 2017; 8(2):56. https://doi.org/10.3390/insects8020056
Chicago/Turabian StyleCammack, Jonathan A., and Jeffery K. Tomberlin. 2017. "The Impact of Diet Protein and Carbohydrate on Select Life-History Traits of The Black Soldier Fly Hermetia illucens (L.) (Diptera: Stratiomyidae)" Insects 8, no. 2: 56. https://doi.org/10.3390/insects8020056
APA StyleCammack, J. A., & Tomberlin, J. K. (2017). The Impact of Diet Protein and Carbohydrate on Select Life-History Traits of The Black Soldier Fly Hermetia illucens (L.) (Diptera: Stratiomyidae). Insects, 8(2), 56. https://doi.org/10.3390/insects8020056





