Beneficial Insect Attraction to Milkweeds (Asclepias speciosa, Asclepias fascicularis) in Washington State, USA
Abstract
:1. Introduction
2. Materials and Methods
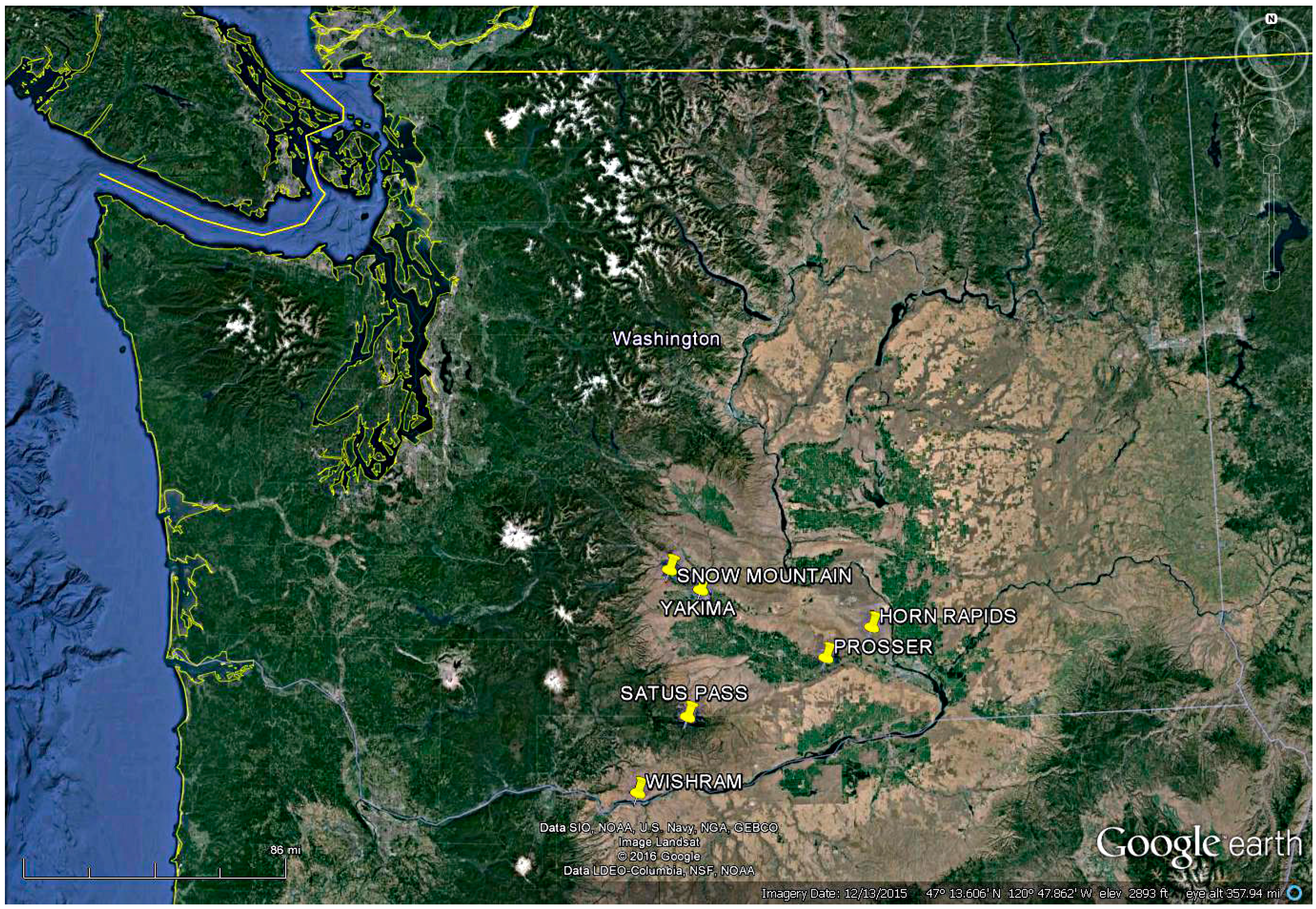
2.1. Sites
2.2. Traps
2.3. Trap Processing
2.4. Data Analysis
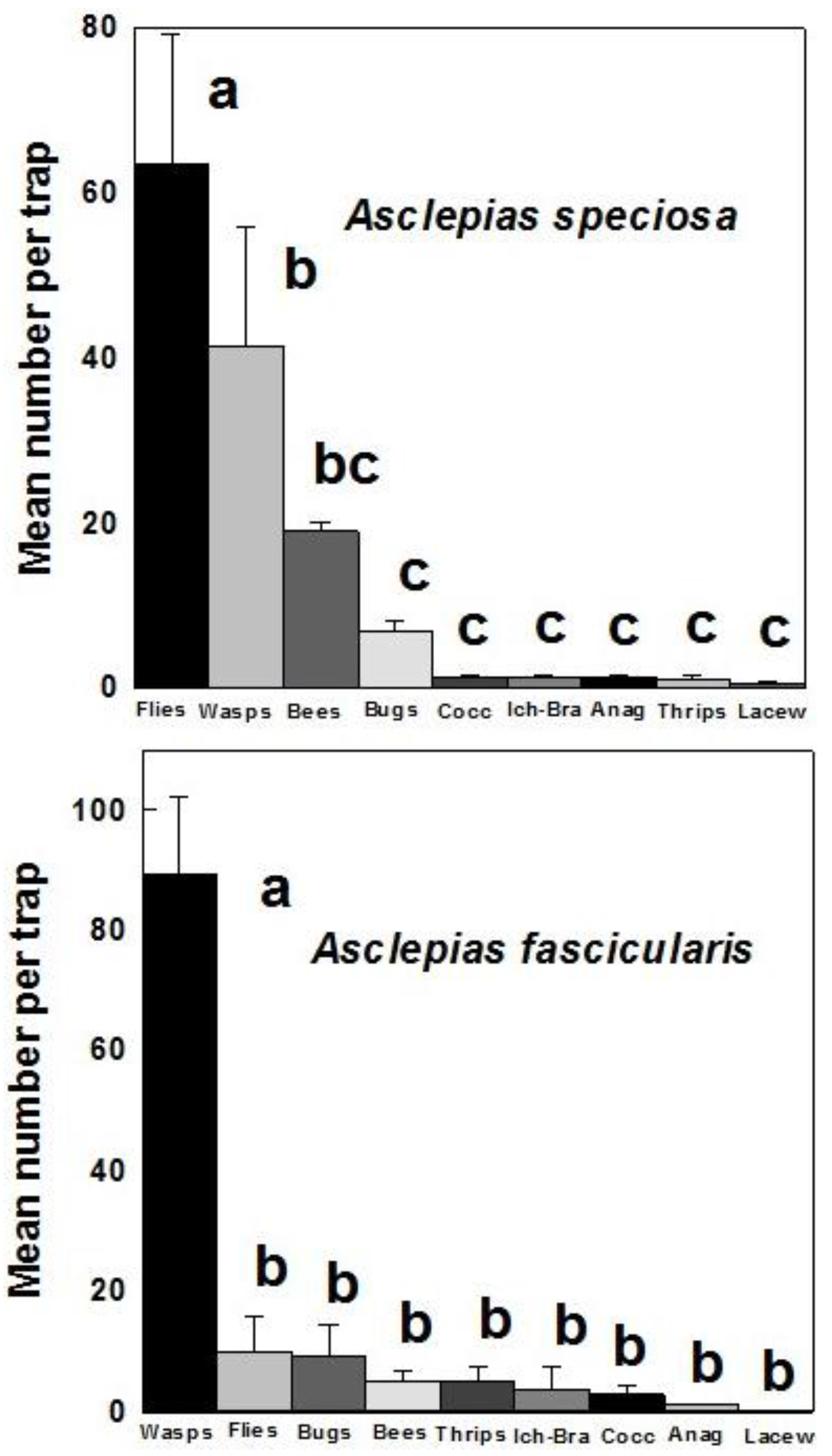
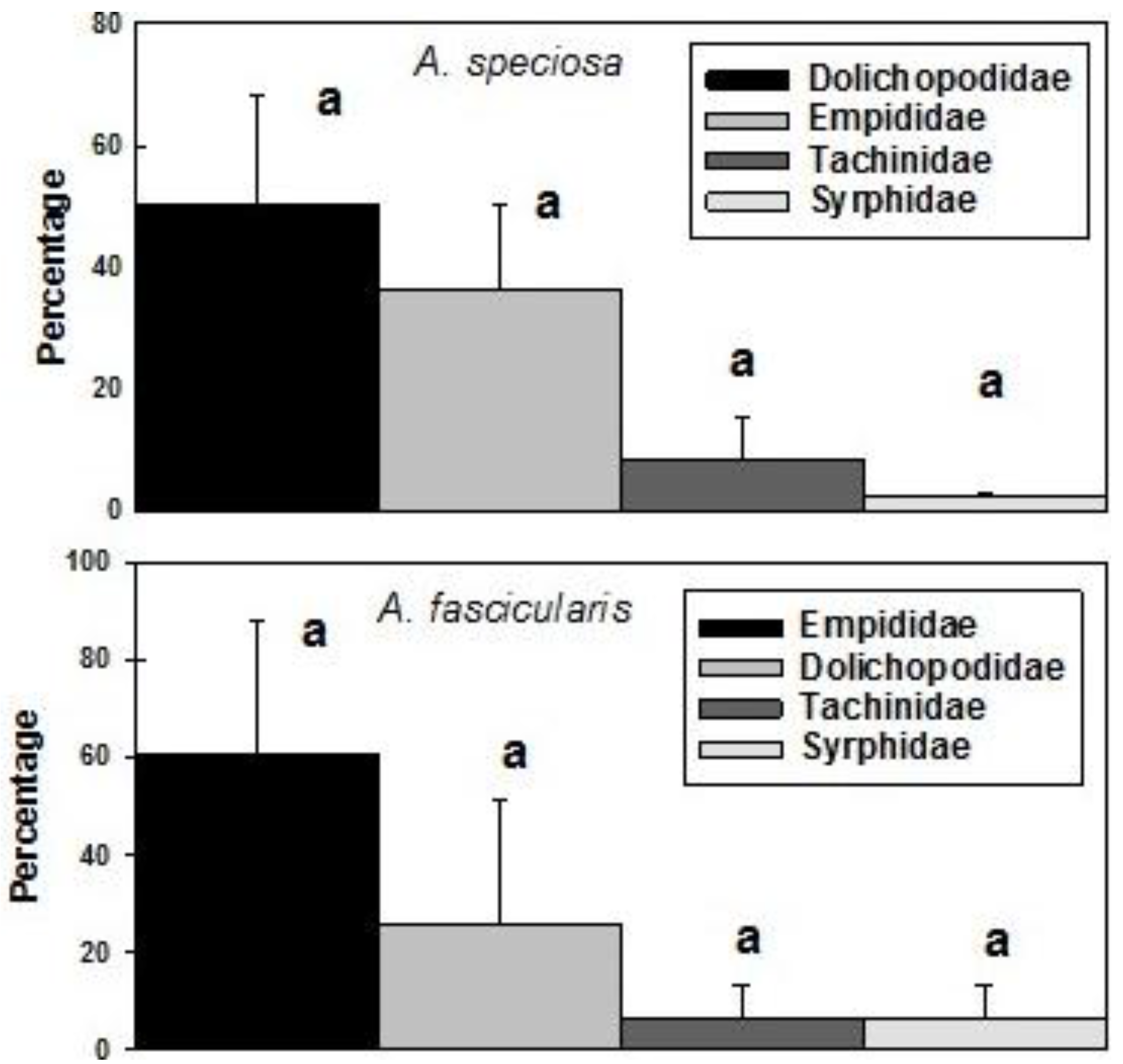
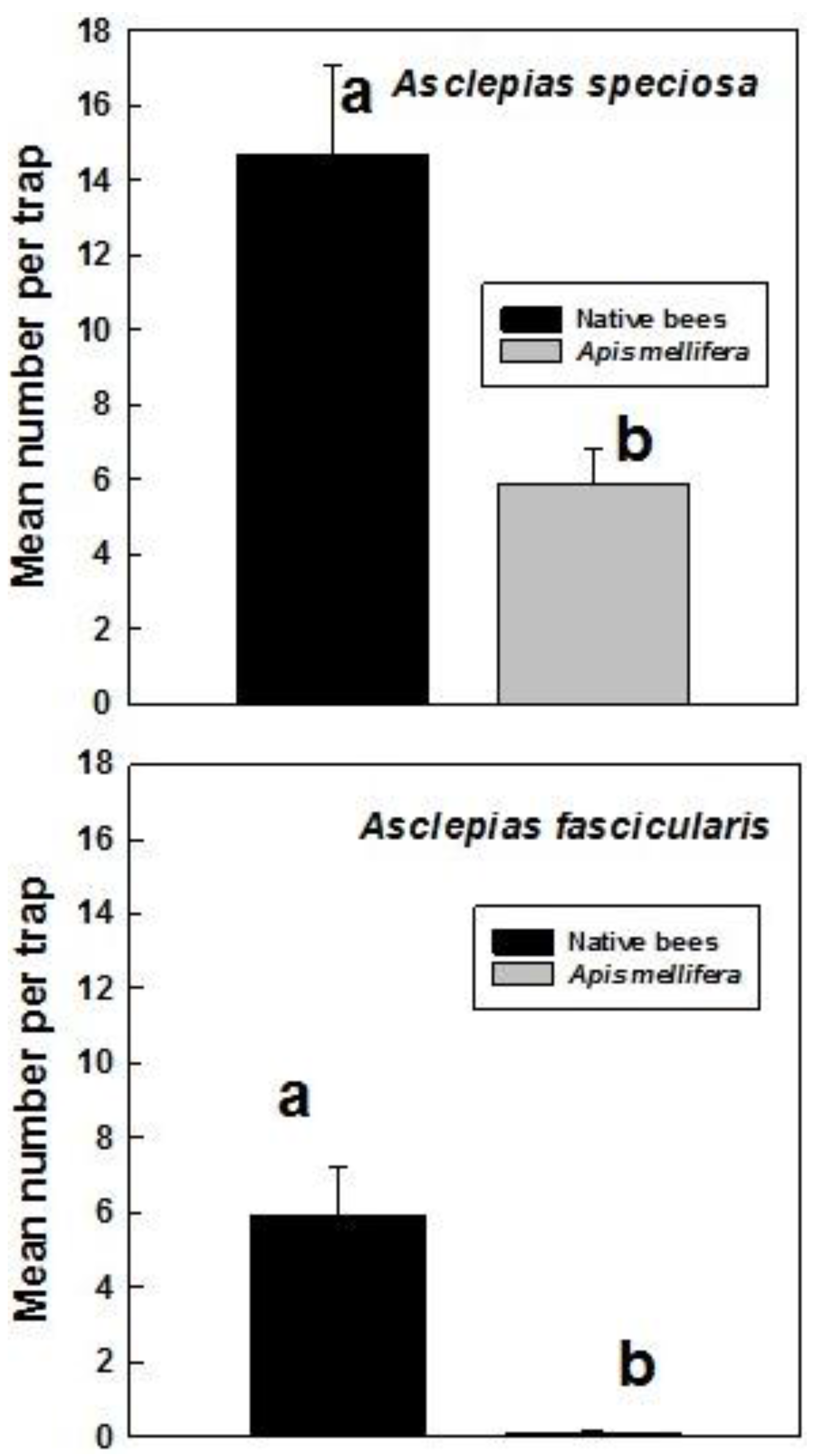
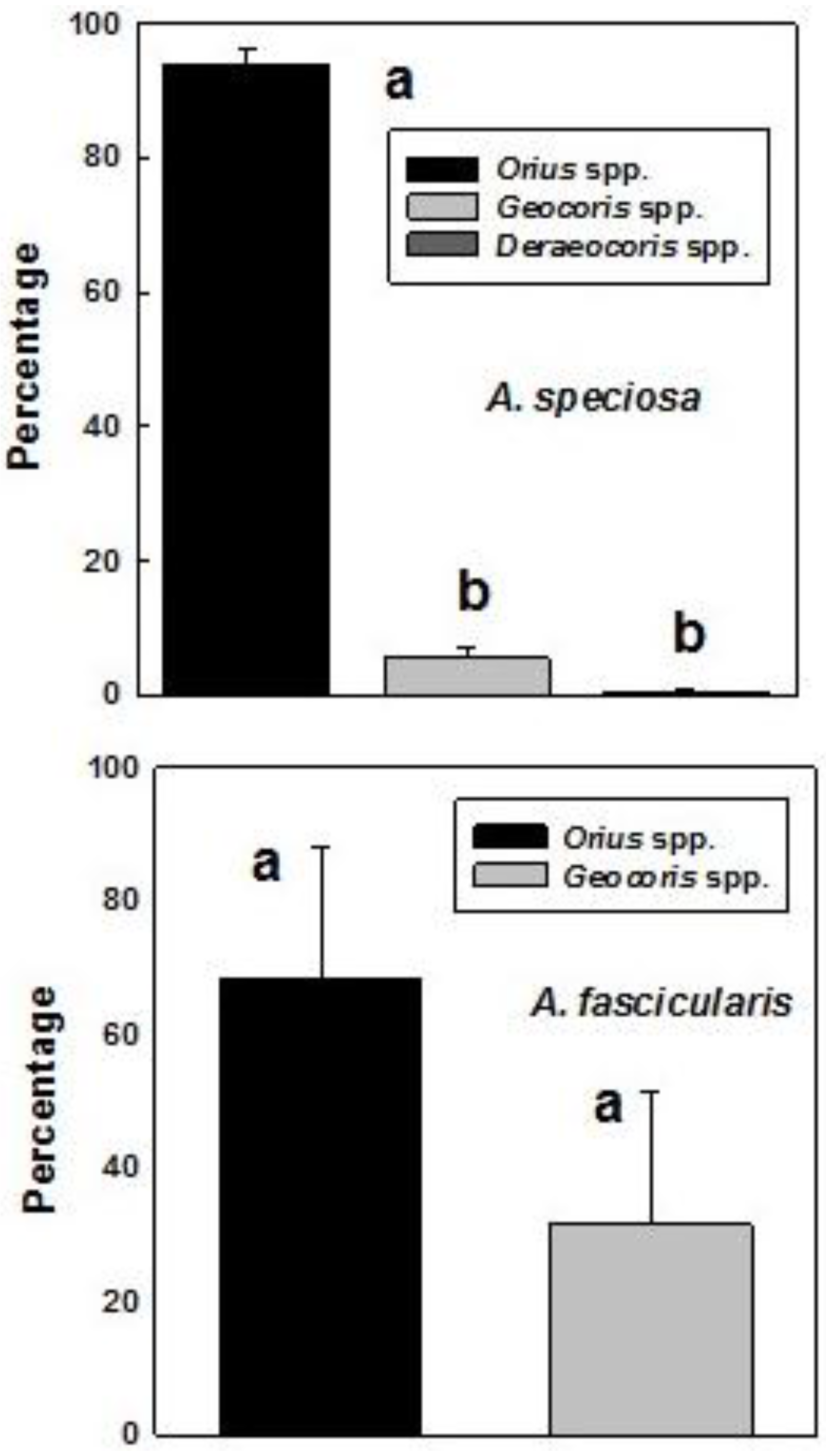
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schellhorn, N.A.; Bianchi, F.J.J.A.; Hsu, C.L. Movement of entomophagous arthropods in agricultural landscapes: Links to pest suppression. Annu. Rev. Entomol. 2014, 59, 559–581. [Google Scholar] [CrossRef] [PubMed]
- Fiedler, A.K.; Landis, D.A.; Wratten, S.D. Maximizing ecosystem services from conservation biological control: The role of habitat management. Biol. Control 2008, 45, 254–271. [Google Scholar] [CrossRef]
- Isaacs, R.J.; Tuell, A.; Fiedler, A.; Gardiner, M.; Landis, D.A. Maximizing arthropod-mediated ecosystem services in agricultural landscapes: The role of native plants. Front. Ecol. Environ. 2009, 7, 196–203. [Google Scholar] [CrossRef]
- Fiedler, A.; Landis, D.A. Attractiveness of Michigan native plants to arthropods natural enemies and herbivores. Environ. Entomol. 2007, 36, 751–765. [Google Scholar] [CrossRef] [PubMed]
- Bennett, A.B.; Gratton, C. Floral diversity increases beneficial arthropod richness and decreases variability in arthropod community composition. Ecol. Appl. 2013, 23, 86–95. [Google Scholar] [CrossRef]
- Frank, S.D.; Shrewsbury, P.M.; Esiekpe, O. Spatial and temporal variation in natural enemy assemblages on Maryland native plant species. Environ. Entomol. 2008, 37, 478–486. [Google Scholar] [CrossRef] [PubMed]
- Ballard, M.; Hough-Golstein, J.; Tallamy, D. Arthropod communities on nonnative early successional plants. Environ. Entomol. 2013, 42, 851–859. [Google Scholar] [CrossRef] [PubMed]
- Pisani Gareau, T.L.; Letourneau, D.K.; Shennan, C. Relative densities of natural enemy and pest insects within California hedgerows. Environ. Entomol. 2013, 42, 688–702. [Google Scholar] [CrossRef] [PubMed]
- Tschumi, M.; Albrecht, M.; Entling, M.H.; Jacot, M. High effectiveness of tailored flower strips in reducing pests and crop plant damage. Proc. Biol. Sci. 2015. [Google Scholar] [CrossRef] [PubMed]
- Gaigher, R.; Pryke, J.S.; Samways, M.J. High parasitoid density in remnant natural vegetation, but limited spillover into the agricultural matrix in South African vineyard ecosystems. Biol. Conserv. 2015, 186, 69–74. [Google Scholar] [CrossRef]
- James, D.G.; Seymour, L.; Lauby, G.; Buckley, K. Beneficial insects attracted to native flowering buckwheats (Eriogonum Michx) in central Washington. Environ. Entomol. 2014, 43, 942–948. [Google Scholar] [CrossRef] [PubMed]
- James, D.G.; Lauby, G.; Seymour, L.; Buckley, K. Beneficial insects associated with stinging nettle, Urtica dioica Linnaeus, in central Washington State. Pan-Pacific Entomol. 2015, 91, 82–90. [Google Scholar] [CrossRef]
- Luna, T.; Dumroese, R.K. Monarchs (Danaus plexippus) and milkweeds (Asclepias species). Native Plants 2013, 14, 5–15. [Google Scholar] [CrossRef]
- Ackery, P.R.; Vane-Wright, R.I. Milkweed Butterflies: Their Cladistics and Biology; Cornell University Press: Ithaca, NY, USA, 1984. [Google Scholar]
- Hartzler, R.G. Reduction in common milkweed (Asclepias syriaca) occurrence in Iowa cropland from 1999–2009. Crop Prot. 2010, 29, 363–366. [Google Scholar] [CrossRef]
- Pleasants, J.M.; Oberhauser, K.S. Milkweed loss in agricultural fields because of herbicide use: Effect on the monarch butterfly population. Insect Conserv. Divers. 2013, 6, 135–144. [Google Scholar] [CrossRef]
- MonarchWatch. Bring back the Monarchs. Available online: http://monarchwatch.org/bring-back-the-monarchs/milkweed (accessed on 29 March 2016).
- New Steps to Protect Pollinators, Critical Contributors to Our Nation’s Economy. Available online: https://www.whitehouse.gov/blog/2014/06/20/new-steps-protect-pollinators-critical-contributors-our-nation-s-economy (accessed on 29 March 2016).
- Project Milkweed. Available online: http://www.xerces.org/milkweed/ (accessed on 29 March 2016).
- Robertson, C. Flowers and Insects; Science Press: Lancaster, PA, USA, 1928. [Google Scholar]
- Willson, M.F.; Bertin, R.I. Flower visitors, nectar production and inflorescence size of Asclepias syriaca. Can. J. Bot. 1979, 57, 1380–1388. [Google Scholar] [CrossRef]
- Kephart, S.R. The partitioning of pollinators among three species of Asclepias. Ecology 1983, 64, 120–133. [Google Scholar] [CrossRef]
- Wyatt, R.; Broyles, S.B. Ecology and evolution of reproduction in milkweeds. Ann. Rev. Ecol. Syst. 1994, 25, 423–441. [Google Scholar] [CrossRef]
- Fishbein, M.; Venable, D.L. Diversity and temporal change in the effective pollinators of Asclepias tuberosa. Ecology 1996, 77, 1061–1073. [Google Scholar] [CrossRef]
- Tillman, P.G.; Carpenter, J.E. Milkweed (Gentianales: Apocynaceae); A farmscape resource for increasing parasitism of stink bugs (Hemiptera: Pentatomidae) and provding nectar to insect pollinators and monarch butterflies. Environ. Entomol. 2014, 43, 370–376. [Google Scholar] [CrossRef] [PubMed]
- James, D.G. Beneficial Arthropods. In Field Guide for Integrated Pest Management in Pacific Northwest Vineyards; Moyers, M.M., O’Neal, S., Eds.; Washington State University: Pullman, WA, USA, 2013; pp. 49–64. [Google Scholar]
- James, D.G. Pest Management in Washington State Vineyards. In Farming with Native Beneficial Insects; Mader, E.L., Hopwood, J., Morandin, L., Vaughan, M., Black, S.H., Eds.; The Xerces Society, Storey Publishing: North Adams, MA, USA, 2014; pp. 26–77. [Google Scholar]
- James, D.G.; Seymour, L.; Lauby, G.; Buckley, K. Beauty with benefits: Butterfly conservation in Washington State, USA, wine grape vineyards. J. Insect Conserv. 2015, 19, 341–348. [Google Scholar] [CrossRef]
- Brower, L.P.; Taylor, O.R.; Williams, E.H.; Slayback, D.A.; Zubieta, R.R.; Ramirez, M.I. Decline of monarch butterflies overwintering in Mexico: Is the migratory phenomenon at risk? Insect Conserv. Divers. 2012, 5, 95–100. [Google Scholar] [CrossRef]
- Make Way for Monarchs. Available online: http://makewayformonarchs.org/i/ (accessed on 29 March 2016).
- Elliot, N.C.; Kieckhefer, R.W.; Michels, G.J.; Giles, K.L. Predator abundance in alfalfa fields in relation to aphids, within-field vegetation and landscape matrix. Environ. Entomol. 2002, 31, 253–260. [Google Scholar] [CrossRef]
- Cumming, J.M.; Cooper, B.E. Techniques for obtaining adult-associated immature stages of predacious tachydromiine flies (Diptera: Empidoidea) with implications for rearing and biocontrol. Entomol. News 1993, 104, 93–101. [Google Scholar]
- Coulibaly, B. Dolichopodidae (Diptera) in the biological control of certain insects harmful to forest ecosystems. Insect Sci. Appl. 1993, 14, 85–87. [Google Scholar]
- Oberhauser, K.; Anderson, M.; Anderson, S.; Caldwell, W.; De Anda, A.; Hunter, M.; Kaiser, M.C.; Solensky, M.J. Lacewings Wasps and flies-Oh my: Insect Natural Enemies Take a Bite Out of Monarchs. In Monarchs in a Changing World; Oberhauser, K., Nail, K.R., Altizer, S., Eds.; Cornell University Press: New York, NY, USA, 2015; pp. 71–82. [Google Scholar]
- Tuell, J.K.; Fiedler, A.K.; Landis, D.; Isaacs, R. Visitation by wild and managed bees (Hymenoptera: Apoidea) to eastern US natives plants for use in conservation programs. Environ. Entomol. 2008, 37, 707–718. [Google Scholar] [CrossRef]
- Miliczky, E.; Horton, D.R. Occurrence of the western flower thrips, Frankliniella occidentalis, and potential predators on host plants in near-orchard habitats of Washington and Oregon (Thysanoptera: Thripidae). J. Entomol. Soc. Br. Columbia 2011, 108, 11–28. [Google Scholar]
- Greenleaf, S.S.; Kremen, C. Wild bees enhance honey bees’ pollination of hybrid sunflower. Proc. Natl. Acad. Sci. USA 2006, 103, 13890–13895. [Google Scholar] [CrossRef] [PubMed]
- James, D.G. Population biology of monarch butterflies, Danaus plexippus L. (Lepidoptera: Nymphalidae) at a milkweed-rich summer breeding site in central Washington. J. Lep. Soc. 2016. accepted. [Google Scholar]
| Beneficial Insect Categories | Species, Genera or Family Included |
|---|---|
| Neuroptera (Lacewings) | Chrysoperla plorabunda (Fitch) Chrysopa nigricornis Burmeister Chrysopa coloradensis Banks Chrysopa oculata Say |
| Coccinellidae (Ladybeetles) | Harmonia axyridis (Pallas) Coccinella septempunctata L. Coccinella transversogutatta Mulsant Hippodamia convergens (Guerin-Meneville) Stethorus picipes Casey Stethorus punctillum (Weise) |
| Heteroptera (Predatory bugs) | Deraeocoris brevis (Uhler) Geocoris pallens Stal Orius spp. |
| Aeolothripidae (Predatory thrips) | Franklinothrips spp. Aeolothrips spp. |
| Diptera (Predatory and parasitic flies) | Empididae Syrphidae Dolichopodidae Tachinidae |
| Icheumonidae and Braconidae (Ichneumonid and braconid wasps) | |
| Mymaridae (Fairy flies) | Anagrus spp. |
| Other parasitic wasps | Pteromalidae, Eulophidae, Trichogrammatidae, Scelionidae |
| Apoidea (Bees) | Apis mellifera L., Andrenidae, Halictidae, Megachilidae, Apidae, Colletidae |
| Year | A. speciosa | A. fascicularis | ||
|---|---|---|---|---|
| No. of Traps | Trapping Period | No. of Traps | Trapping Period | |
| 2010 | 12 | 28 June–19 July | 0 | – |
| 2011 | 30 | 19 July–25 August | 0 | – |
| 2012 | 51 | 16 May–23 July | 6 | 26 June–10 July |
| 2013 | 28 | 11 June–24 July | 6 | 27 June–10 July |
| 2014 | 0 | – | 3 | 24 June–8 July |
| All years | 121 | 16 May–25 August | 15 | 24 June–10 July |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
James, D.G.; Seymour, L.; Lauby, G.; Buckley, K. Beneficial Insect Attraction to Milkweeds (Asclepias speciosa, Asclepias fascicularis) in Washington State, USA. Insects 2016, 7, 30. https://doi.org/10.3390/insects7030030
James DG, Seymour L, Lauby G, Buckley K. Beneficial Insect Attraction to Milkweeds (Asclepias speciosa, Asclepias fascicularis) in Washington State, USA. Insects. 2016; 7(3):30. https://doi.org/10.3390/insects7030030
Chicago/Turabian StyleJames, David G., Lorraine Seymour, Gerry Lauby, and Katie Buckley. 2016. "Beneficial Insect Attraction to Milkweeds (Asclepias speciosa, Asclepias fascicularis) in Washington State, USA" Insects 7, no. 3: 30. https://doi.org/10.3390/insects7030030
APA StyleJames, D. G., Seymour, L., Lauby, G., & Buckley, K. (2016). Beneficial Insect Attraction to Milkweeds (Asclepias speciosa, Asclepias fascicularis) in Washington State, USA. Insects, 7(3), 30. https://doi.org/10.3390/insects7030030







