Simple Summary
It is of great significance to develop and utilize plant-derived compounds for the sustainable control of grasshopper. This study assessed the adverse effects of rutin and quercetin on grasshopper, as well as the insect’s physiological response to these two plant-derived compounds. Rutin and quercetin all exhibited toxic effects on grasshopper, with quercetin showing a stronger toxicity, which indicated that they—especially quercetin—have the potential to be developed as biopesticides to control the grasshopper.
Abstract
Insect-resistant substances from plants are important natural resources that human beings can potentially develop and use to control pests. In this study, we explored the adverse effects of rutin and quercetin on grasshopper (Calliptamus abbreviatus), as well as the insect’s physiological response to these substances in laboratory and field experiments. These two plant compounds exhibited toxic effects on C. abbreviatus, with quercetin showing a stronger toxicity, indicated by a lower survival, slower development, and higher induced gene expression and activities of UDP-glucuronosyltransferase, cytochrome P450s, superoxide dismutase, peroxidase and catalase, compared to rutin. These compounds, especially quercetin, have the potential to be developed as biopesticides to control grasshoppers.
1. Introduction
Calliptamus abbreviatus (Orthoptera: Acrididae) is a dominant grasshopper species in grasslands in northern China. Its high occurrence negatively affects agricultural and animal husbandry production [1,2,3]. At present, the use of chemical pesticides is the main method for the control of C. abbreviatus. Although chemical pesticides play an important role in plant protection, their irrational use often leads to problems of pesticide residues, pest resistance and resurgence, which in turn adversely affects the ecosystem [4,5,6]. In recent decades, research efforts have focused on the identification of bio-pesticides that are low-toxicity, biodegradable and low-residue [7,8]. Insect-resistant substances from plants, such as flavonoids, terpenoids, alkaloids, and sterols, exhibit the above characteristics and are important natural resources to be developed as biological insecticides [9,10]. Therefore, it is of great significance to develop and utilize plant-derived compounds for the sustainable control of C. abbreviatus.
To resist insect feeding or other stress, plants produce a large number of compounds, serving as “chemical weapons”, to resist damage [11,12]. Some of these compounds can adversely affect the feeding behavior, growth, development or reproduction of insects, or even directly poison them [13]. For example, plant derived substances such as tannin, nicotine and matrine can directly hinder the normal growth and development of pests such as Helicoverpa armigera, Papilio polyxenes, Bemisia tabaci, Spodoptera eridania and Pectinophora gossypiella [14,15,16]. In addition, other compounds can be induced for the indirect control of pests by attracting predatory or parasitic natural enemies, such as that exhibited by some terpenoids on Pyrausta nubilalis and Spodoptera exigua [17,18]. The mechanism underlying the toxicity of plant secondary compounds on insects includes the inhibition of the activities of acetylcholinesterase, acetylcholine receptor, and enzyme activities; or destroying the aminobutyric acid gated chlorine channels, blocking Na+/K+ exchange and Ca2+ channels, and interfering with the series of reactions of respiration and tyramine and the balance of ecdysone or juvenile hormone [19]. Compounds such as azadirachtin can induce reactive oxygen species (ROS) in insects, resulting in oxidative damage, which inhibits the normal development of insects [20,21,22].
Herbivorous insects have evolved mechanisms to detoxify these toxic plant compounds to reduce their harm. These include the synthesis and secretion of cytochrome P450s, UDP-glucuronosyltransferase, glutathione sulfotransferase, carboxylesterase and other detoxification enzymes in guts and fat bodies [3,16,23,24,25]. Moreover, insects can also secrete peroxidase, catalase, superoxide dismutase and other protective enzymes to alleviate oxidative damage [26,27]. The changes in ROS content, gene expression or the activities of detoxification enzymes and protective enzymes in insects can indicate an adaptation response to the toxicities of these plant compounds.
Rutin and quercetin are flavonoids which have been identified in a wide plant species. They can inhibit the normal growth and development of pests such as Spodoptera litura, Lymantria dispar, Ostrinia nubilalis, Helicoverpa zea, Coptotermes formosanus, Pectinophora gossypiella and Acheta domesticus, resulting in increased pest mortality, decreased development rate and fecundity [28,29,30,31]. Their adverse effects on pests suggest that they are an important resource, which can be potentially developed and used to control pests. In this study, we assessed the adverse effects of rutin and quercetin on C. abbreviatus, as well as the insect’s physiological response to these two plant-derived compounds. The aim of this research was to provide a theoretical basis for the further development and utilization of rutin or quercetin to control grasshopper.
2. Materials and Methods
2.1. Field Grasshopper Collection
Third instar nymphs of C. abbreviatus were collected from an alfalfa field by a sweep net (40 cm diameter) in Yishui, Shandong Province, northeastern China (35.839° N, 118.296° E) in June 2021 and 2022. The vegetation in this alfalfa field was dominated by Medicago sativa L. Collected nymphs were maintained temporarily in a cage, and then used in the indoor trial in 2021 and field cage study in 2022.
2.2. Indoor Feeding Trial
A total of 1600 healthy third instar nymphs of C. abbreviatus were selected and starved for 24 h to conduct the indoor feeding trial in June 2021. They were randomly assigned to 80 mesh cages (size: 40 cm × 30 cm × 20 cm), with each cage containing 20 nymphs. A series of rutin and quercetin concentrations (0%, 0.001%, 0.01%, 0.1% and 1.0%) were prepared using dimethyl sulfoxide (DMSO). For each cage, 10 mL of the prepared solution was evenly applied to 50 g of fresh M. sativa using a 50 mL hand sprayer, allowed to dry, and then provided to C. abbreviatus. Each treatment included 8 replicates (cages). Grasshopper survival was inspected daily until all surviving individuals became adults. For each treatment, 8 adult individuals were randomly collected to weigh the body mass (mg) and immediately frozen in liquid nitrogen. These frozen samples were stored at −80 °C and used to measure gene expressions, rutin and quercetin contents, ROS level and enzyme activity (see below). Grasshopper survival rate (%) and development time (days) were calculated using the described method by Li et al. [32]. The survival rate (%) of grasshoppers was calculated by the number of surviving adulthoods/numbers of initial third instar individuals (n = 20). Development time (days) of C. abbreviatus from third instar to adult was calculated by the formula: , where i is the number of days from third instar to adult; Ni is the number of grasshopper individuals with the development time corresponding to that value of “i”; Nt is the number of grasshopper C. abbreviates that successfully survived to adulthood.
2.3. Gene Expression
The relative gene expressions of β-glucosidase, UDP-glucuronosyltransferase, cytochrome P450 6k1, superoxide dismutase, peroxidase and catalase were measured using a real time PCR. The sequences of these genes were acquired from our previous published transcriptome profiles of C. abbreviatus (SRA database: SRR10289990). Gene-specific primers were designed using the Primer 3 software (version 4.1.0, http://bioinfo.ut.ee/primer3-0.4.0/, accessed on 20 January 2024) and have been provided in Supplementary Table S1. The same method of real time PCR was used, as described in our published paper [3]. In brief, the total RNA from each grasshopper sample was extracted using TRIzol reagent (Invitrogen, San Diego, CA, USA). After RNA extraction, the AMV reverse transcriptase (Invitrogen, San Diego, CA, USA) was used to synthesize cDNA. Experiments were performed using SYBR® green PCR mix (Hilden, Germany) according to the manufacturer’s instructions. Relative gene expressions were calculated using the 2−ΔΔCT method, with β-actin as the reference gene. Expression values were adjusted by setting the expression of controls to 1 for each gene. The PCR runs for each gene of each treatment included eight biological replicates with three technical replicates.
2.4. Analysis of Rutin and Quercetin Content in Grasshopper
The rutin and quercetin content in each grasshopper sample for each treatment were measured by high-performance liquid chromatography (HPLC), using the same method described in a previously published paper [33]. In brief, the flavonoids in each grasshopper sample were extracted by soaking in 70% ethanol. After ultrasonic extraction for 35 min, the solution was put in a water bath at 60 °C to evaporate the ethanol. Anhydrous methanol was added until it reached a volume of 50.00 mL. After centrifugation, supernatant was collected and filtrated through a 0.45 μm filter membrane. Ultrasonic degassing was carried out to obtain the flavonoids extract. Each reference substance was dissolved by anhydrous methanol and filtrated by a 0.45 μm filter membrane. Finally, rutin and quercetin were detected by chromatographic experiments conducted using the Agilent 1260 (Santa Clara, CA, USA) HPLC system. The retention times were used for a qualitative analysis, and peak areas were used for a quantitative analysis.
2.5. ELISA Analysis of ROS Level and Enzyme Activities
ROS level, and the enzyme activities of β-glucosidase, UDP-glucuronosyltransferase, cytochrome P450s, superoxide dismutase, peroxidase and catalase in the grasshopper samples were measured following the method described by Li et al. [33]. In brief, grasshopper samples were homogenized in 1 mL phosphate buffered saline (PBS), and the resulting suspension subjected to ultrasonication to further break the cell membranes. After that, we centrifuged the homogenates for 15 min at 5000 rpm, collected the supernatants and stored at −20 °C until required for further analysis. The ELISA procedures were conducted according to the manufacturer’s instructions.
2.6. Field Cage Trial
A field cage study was conducted on an alfalfa area to evaluate the field survival of C. abbreviatus exposed to rutin and quercetin in June 2022. A total of 50 mesh cages (size: 1 m × 1 m × 1 m) were installed in this alfalfa area. All other plant species were removed to ensure that only M. sativa remained in these field cages. M. sativa plants were mainly at the vegetative stage, with heights ranging from ~53.2 to ~62.7 cm and dry biomass ranging from 132.4 to 167.8 g/m2. M. sativa in each cage was sufficient for grasshopper feeding until the end of the field experiment. Before adding C. abbreviatus, all potential natural enemies in these field cages were removed. A total of 1000 third instar nymphs of C. abbreviatus were collected and assigned randomly to 50 cages. Each cage included 20 healthy individuals. The prepared solutions of rutin and quercetin of different concentrations (0%, 0.001%, 0.01%, 0.1% and 1.0%) were applied evenly to grass M. sativa in field cages using a hand sprayer. In each cage, 100 mL of solution was sprayed on M. sativa. This field trial included ten treatments with five replications per treatment. The number of surviving individuals was determined on days 7 and 14 to calculate the survival rate (%) by the number of surviving individuals/the number of initial third instar individuals (n = 20).
2.7. Data Analyses
A one-way ANOVA was used to compare the difference in survival rate, body mass, development time, gene expression, ROS level, rutin and quercetin content, and enzyme activities in grasshoppers treated with the different concentrations of rutin or quercetin. A Student’s t-test was used to compare the differences in the grasshopper variables between rutin and quercetin treatments within the same concentration. All analyses were carried out using SAS version 9.0 (SAS Institute, Inc., Cary, NC, USA).
3. Results
3.1. Grasshopper Survival, Body Mass and Developmental Time
The survival of C. abbreviatus negatively correlated with increased rutin and quercetin concentration, respectively (Figure 1A, grasshoppers exposed to rutin: R2 = 0.9816, p < 0.01; grasshoppers exposed to quercetin: R2 = 0.9794, p < 0.01). The survival rate of C. abbreviatus treated with 0–1% rutin significantly (F = 19.25, df = 4, 35, p < 0.05) decreased from 92.3% to 40.5%, with that of grasshoppers treated with 0–1% quercetin significantly (F = 20.68, df = 4, 35, p < 0.05) decreasing from 90.8% to 38.9%. In addition, grasshoppers treated with quercetin had a lower survival rate than rutin-treated grasshoppers at a concentration of 0.001–0.1%.
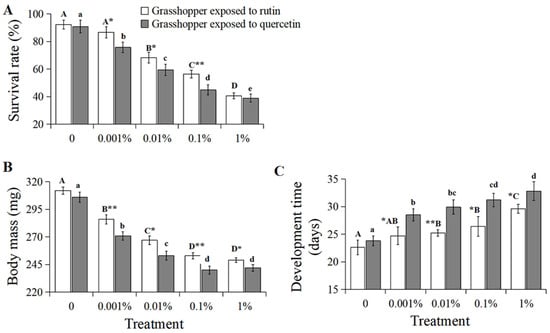
Figure 1.
The survival (A), body mass (B) and developmental time (C) of Calliptamus abbreviatus exposed to rutin and quercetin. Bars with different uppercase letters indicate significant differences within the treatment with rutin, based on Turkey’s HSD analysis at p < 0.05. Bars with different lowercase letters indicate significant differences within the treatment with quercetin, based on Turkey’ s HSD analysis at p < 0.05. A Student’s t-test was used to compare the differences in grasshoppers treated with rutin and quercetin of the same concentration. * p < 0.05, ** p < 0.01.
The body mass of C. abbreviatus also inversely correlated with increased rutin and quercetin concentration (Figure 1B, grasshoppers exposed to rutin: R2 = 0.9359, p < 0.05; grasshoppers exposed to quercetin: R2 = 0.9082, p < 0.05). The body mass of grasshoppers treated with 0–1% rutin significantly (F = 18.32, df = 4, 35, p < 0.05) decreased from 312 mg to 249 mg, with that of grasshoppers treated with 0–1% quercetin decreasing from 306 mg to 242 mg. And grasshoppers treated with quercetin had a lower body mass than rutin-treated grasshoppers at a concentration of 0.001–1%.
The developmental time of C. abbreviatus positively correlated with increased rutin and quercetin concentration (Figure 1C, grasshoppers exposed to rutin: R2 = 0.9280, p < 0.05; grasshoppers exposed to quercetin: R2 = 0.9099, p < 0.05). Developmental time significantly (F = 15.63, df = 4, 35, p < 0.05) increased from 22.6 days to 29.6 days for increased rutin concentration from 0% to 1%, with that of grasshoppers treated with 0–1% quercetin increasing from 23.8 days to 32.8 days (F = 19.31, df = 4, 35, p < 0.05). And grasshoppers treated with quercetin had a slower developmental time than rutin-treated grasshoppers at a concentration of 0.001–1%.
3.2. Gene Expressions in Grasshopper
The expression of the grasshopper β-glucosidase gene positively correlated with increased rutin concentration (Figure 2A, grasshopper exposed to rutin: R2 = 0.9816, p < 0.01). The relative gene expression in 0–1% rutin-treated grasshoppers increased significantly to 26.63 (F = 18.27, df = 4, 35, p < 0.05). It was also higher in rutin-treated than in quercetin-treated grasshoppers at a concentration of 0.001–1%.
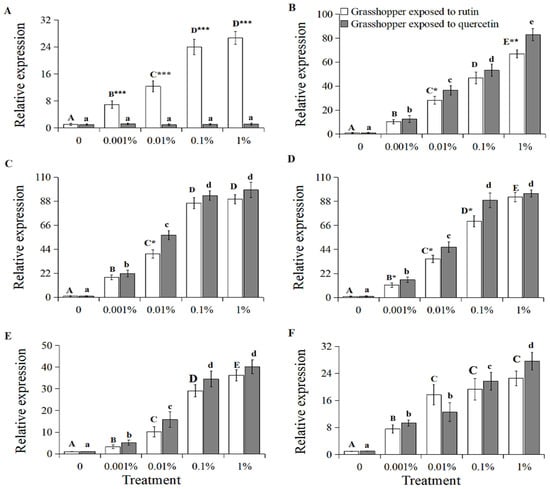
Figure 2.
The relative gene expression of β-glucosidase (A), UDP-glucuronosyltransferase (B), cytochrome P450 6k1 (C), superoxide dismutase (D), peroxidase (E), and catalase (F) in Calliptamus abbreviatus exposed to rutin and quercetin. Bars with different uppercase letters indicate significant differences within rutin treatment, based on Turkey’s HSD analysis at p < 0.05. Bars with different lowercase letters indicate significant differences within quercetin treatment, based on Turkey’ s HSD analysis at p < 0.05. A Student’s t-test was used to compare the differences between grasshoppers treated with rutin and quercetin at the same concentration. * p < 0.05, ** p < 0.01, *** p < 0.001.
The gene expressions of the grasshopper UDP-glucuronosyltransferase, cytochrome P450 6k1, superoxide dismutase, peroxidase and catalase also positively correlated with increased rutin and quercetin concentration (Figure 2B–F, R2 > 0.92, p < 0.05). The relative expressions of these genes in 0–1% rutin-treated grasshoppers increased significantly to 66.82, 89.8, 91.92, 36.25 and 22.53 (F = 8.56–26.57, df = 4, 35, p < 0.05), respectively. Likewise, the relative expressions of these genes in the 0–1% quercetin-treated grasshoppers increased significantly to 82.97, 98.3, 95.27, 40.18 and 27.61(F = 9.32–21.13, df = 4, 35, p < 0.05), respectively. Among them, the gene expressions of UDP-glucuronosyltransferase, cytochrome P450 6k1 and superoxide dismutase in grasshoppers treated with quercetin were higher than those in rutin-treated grasshoppers at a concentration of 0.001–1%.
3.3. Grasshopper Rutin and Quercetin Content
The rutin content in grasshoppers treated with rutin positively correlated with increased rutin concentration (Figure 3A, R2 = 0.9814, p < 0.01). The rutin content in grasshoppers treated with 0–1% rutin significantly (F = 17.93, df = 4, 35, p < 0.05) increased from 0.012 mg/g to 0.696 mg/g. And grasshoppers treated with rutin had a higher rutin content than quercetin-treated grasshoppers at a concentration of 0.001–1%.
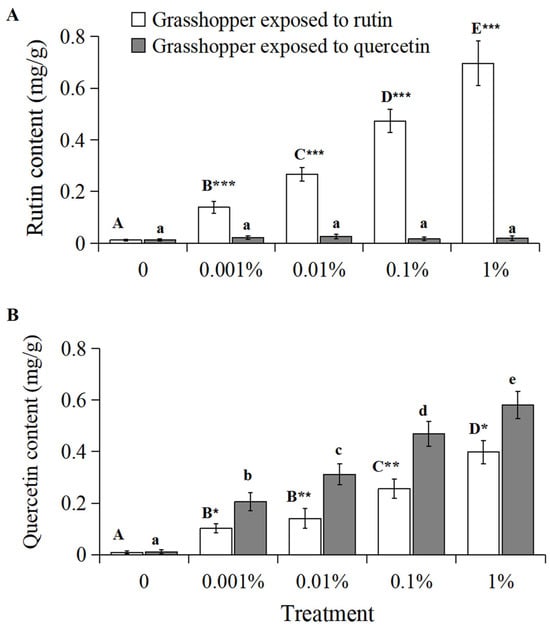
Figure 3.
The rutin (A) and quercetin content (B) in Calliptamus abbreviatus exposed to rutin and quercetin. Bars with different uppercase letters indicate significant differences within rutin treatment, based on Turkey’s HSD analysis at p < 0.05. Bars with different lowercase letters indicate significant differences within quercetin treatment, based on Turkey’ s HSD analysis at p < 0.05. A Student’s t-test was used to compare the differences between grasshoppers treated with rutin and quercetin at the same concentration. * p < 0.05, ** p < 0.01, *** p < 0.001.
The quercetin content in grasshoppers treated with rutin and quercetin positively correlated with increased rutin and quercetin concentration, respectively (Figure 3B, grasshoppers exposed to rutin: R2 = 0.9633, p < 0.01; grasshoppers exposed to quercetin: R2 = 0.9906, p < 0.05). The quercetin content in grasshoppers treated with 0–1% rutin significantly (F = 13.18, df = 4, 35, p < 0.05) increased from 0.009 mg/g to 0.398 mg/g. The quercetin content in grasshoppers treated with 0–1% quercetin significantly (F = 14.45, df = 4, 35, p < 0.05) increased from 0.011 mg/g to 0.581 mg/g. Grasshoppers treated with quercetin had a higher quercetin content than rutin-treated grasshoppers at a concentration of 0.001–1%.
3.4. Grasshopper ROS Level
The ROS level in the grasshoppers had a significantly positive relationship with rutin and quercetin concentration (Figure 4, grasshoppers exposed to rutin: R2 = 0.9969, p < 0.01; grasshoppers exposed to quercetin: R2 = 0.9677, p < 0.01). The ROS level in grasshoppers treated with 0–1% rutin significantly increased (F = 16.56, df = 4, 35, p < 0.05) from ~216 pg/g to ~498 pg/g. The ROS level in grasshoppers treated with 0–1% quercetin significantly increased (F = 19.29, df = 4, 35, p < 0.05) from ~198 pg/g to ~566 pg/g. And the ROS level in grasshoppers treated with quercetin was higher than that in rutin-treated grasshoppers at a concentration of 0.01–1%.
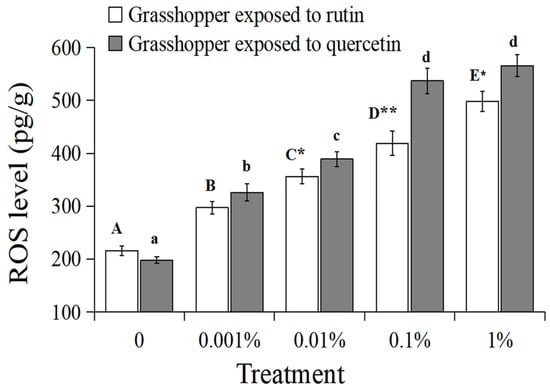
Figure 4.
The ROS level in Calliptamus abbreviatus exposed to rutin and quercetin. Bars with different uppercase letters indicate significant differences within rutin treatment, based on Turkey’s HSD analysis at p < 0.05. Bars with different lowercase letters indicate significant differences within quercetin treatment, based on Turkey’ s HSD analysis at p < 0.05. A Student’s t-test was used to compare the differences between grasshoppers treated with rutin and quercetin at the same concentration. * p < 0.05, ** p < 0.01.
3.5. Grasshopper Enzyme Activity
β-glucosidase activity positively correlated with increased rutin concentration (Figure 5A, R2 = 0.9505, p < 0.01). β-glucosidase activity in 0–1% rutin-treated grasshoppers increased significantly from 123 U/g to 495 U/g (F = 18.92, df = 4, 35, p < 0.05). And grasshoppers treated with rutin had higher β-glucosidase activity than in quercetin-treated grasshoppers at a concentration of 0.001–1%.
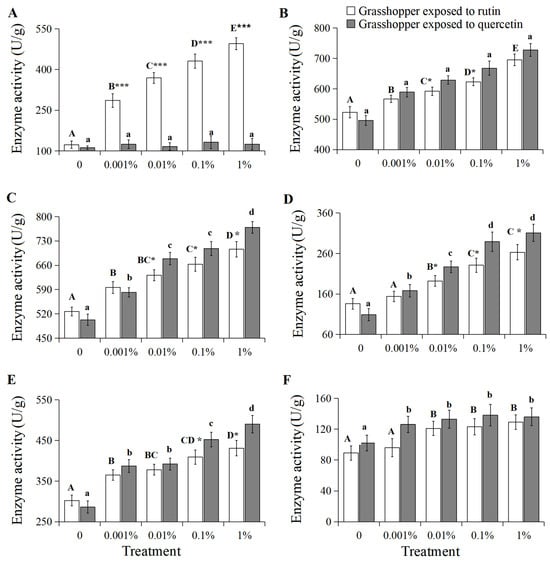
Figure 5.
β-glucosidase (A), UDP-glucuronosyltransferase (B), cytochrome P450s (C), superoxide dismutase (D), peroxidase (E) and catalase (F) activities in Calliptamus abbreviatus exposed to rutin and quercetin. Bars with different uppercase letters indicate significant differences within rutin treatment, based on Turkey’s HSD analysis at p < 0.05. Bars with different lowercase letters indicate significant differences within quercetin treatment, based on Turkey’ s HSD analysis at p < 0.05. Student’s t-tests were used to compare the differences between grasshoppers treated with rutin and quercetin at the same concentration. * p < 0.05, *** p < 0.001.
UDP-glucuronosyltransferase, cytochrome P450 6k1, superoxide dismutase, peroxidase and catalase activities also positively correlated with increased rutin and quercetin concentrations (Figure 5B–E, R2 > 0.9, p < 0.05). Their activities in 0–1% rutin-treated grasshoppers increased significantly from 523 U/g, 527 U/g, 136 U/g, 302 U/g and 89 U/g to 695 U/g, 706 U/g, 263 U/g, 431 U/g and 129 U/g, respectively (F = 7.86–23.69, df = 4, 35, p < 0.05). They also increased significantly from 496 U/g, 503 U/g, 109 U/g, 286 U/g and 102 U/g to 728 U/g, 769 U/g, 311 U/g, 490 U/g and 136 U/g, respectively (F = 9.21–19.63, df = 4, 35, p < 0.05) in 0–1% quercetin-treated grasshoppers. The enzyme activities in grasshoppers treated with quercetin were higher than those in rutin-treated grasshoppers at a concentration of 0.001–1%, with the exception of catalase.
3.6. Grasshopper Survival Rate in Field Cage
The survival rate of grasshoppers at 7 d negatively correlated with an increased concentration of rutin and quercetin (R2 > 0.9601, p < 0.05, Figure 6A). The survival rate of 0–1% rutin-treated grasshoppers at 7 d decreased significantly from 93.6% to 47.5% (F = 17.13, df = 24, p < 0.05). Likewise, that of quercetin-treated grasshoppers decreased significantly from 91.8% to 39.6% (F = 9.82, df = 24, p < 0.05). And grasshoppers treated with quercetin had a lower survival rate at 7 d than that of rutin-treated grasshoppers at a concentration of 0.001–1%.
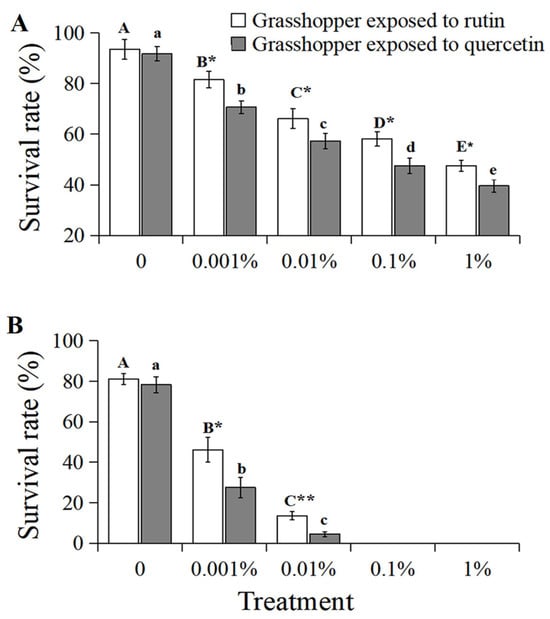
Figure 6.
The survival rate of Calliptamus abbreviatus exposed to rutin and quercetin at 7 d (A) and 14 d (B) in field cages. Bars with different uppercase letters indicate significant differences within rutin treatment, based on Turkey’s HSD analysis at p < 0.05. Bars with different lowercase letters indicate significant differences within quercetin treatment, based on Turkey’ s HSD analysis at p < 0.05. A Student’s t-test was used to compare the differences between grasshoppers treated with rutin and quercetin at the same concentration. * p < 0.05, ** p < 0.01.
The survival rate of 0–0.01% rutin-treated grasshoppers at 14 d decreased significantly from 81.2% to 13.6% (F = 15.01, df = 24, p < 0.05, Figure 6B). Also, that of quercetin-treated grasshoppers decreased significantly from 78.3% to 4.5% (F = 12.16, df = 24, p < 0.05, Figure 6B). Grasshoppers treated with quercetin had a lower survival rate at 14 d than that of rutin-treated grasshoppers at concentrations of 0.001% and 0.01%. No survival was recorded at 14 d for the 0.1% and 1% concentration treatments of these two chemicals.
4. Discussion
In recent years, with the aggravation of grasshoppers in China, the discovery and utilization of plant-derived compounds for the sustainable control of these insects have attracted much attention. In this study, we found that rutin and quercetin significantly reduced the survival rate and inhibited the development of Calliptamus abbreviatus, indicating that they were toxic to C. abbreviatus and have the potential to be developed as biopesticides. In addition, we found that quercetin showed stronger toxicity than rutin to grasshoppers, as indicated by a lower survival rate and slower development than those of rutin.
Generally, herbivorous insects produce large amounts of detoxification enzymes, such as UDP-glucuronosyltransferase and cytochrome P450s, in response to toxic plant-derived compounds. This is to convert or degrade them to reduce tissue damage and enhance their survival under toxicity stress [23,25,26,27,34,35]. For example, cytochrome P450s and UDP-glucuronosyltransferase activities in Spodoptera litura, Helicoverpa zea and Manduca sexta treated with toxic plant-derived compounds were significantly higher than those in the control group [36,37,38,39,40]. In this study, we also found that the gene expressions and activities of these two detoxification enzymes increased significantly after treatment with rutin and quercetin, indicating that they played an important role in the resistance of C. abbreviatus to the toxic compounds. We also found that C. abbreviatus showed higher detoxification enzyme activities to convert or degrade toxic quercetin than for rutin. The detoxification of harmful plant compounds is a high-energy process [41,42,43,44,45,46]. Therefore, the detoxification of quercetin in grasshoppers may have been energetically costly, which resulted in lower survival and development than in those treated with rutin. However, the molecular mechanism underlying the detoxification of rutin and quercetin in C. abbreviatus requires further research.
Exposure to toxic compounds usually leads to an increase in ROS concentration in organisms, triggering oxidative damage and programmed apoptosis [47,48]. Therefore, ROS concentration can reflect the degree of environmental stress [7,21,22]. In this study, ROS levels in grasshoppers significantly increased after treatment with toxic rutin and quercetin, with higher levels recorded under quercetin treatment. This also supports the observation that quercetin showed stronger toxicity than rutin in C. abbreviatus. ROS levels in insects are mainly regulated by superoxide dismutase, peroxidase and catalase [24,25]. They are induced to synergically reduce ROS levels, to maintain the normal growth and development of organisms [21,23,49]. These protective enzymes also play an important role in the resistance to oxidative stress caused by toxic plant compounds. In this study, expressions of the genes and superoxide dismutase, peroxidase and catalase activities in grasshoppers all increased after treatment with rutin and quercetin; however, those in quercetin-treated grasshoppers were higher than those in rutin-treated grasshoppers.
Gene expression and related enzyme function were the underpinning mechanisms of herbivorous insects resisting toxic compounds [16]. Those changed genes were the basis of genetic adaptation and allowed the rapid induction of arrays of broader or more robustly active digestive, antioxidant or detoxifying enzymes in herbivorous insects after the consumption of toxic compounds [23]. These rapid biochemical responses to toxic compounds are vital for insect survival and growth. In this study, we also found that those gene expressions were in accordance with their related enzyme activities. C. abbreviatus treated by rutin or quercetin had high gene expression and related enzyme activity associated with digestion, oxidation resistance and detoxification, which implied that they play an important role in the resistance of C. abbreviatus to toxic rutin and quercetin in order to survive.
Rutin (C27H30O16) is an important flavonoid glycoside widely distributed in plants and is composed of quercetin and glucose ligands. It is the glycoside form of quercetin [29,30,31]. β-glucosidase, a hydrolase found in the membrane of animal midgut epithelial cells, plays a role in the metabolism of carbohydrates taken by animals, especially in the transmembrane metabolism of flavonoid glycosides. Mature β-glucosidase can hydrolyze flavonoid glycosides into free aglygen and glucose ligands [17,50]. In this study, we also found that C. abbreviatus showed increased gene expression and β-glucosidase activity when exposed to rutin, which corresponded with a higher quercetin content in grasshoppers. β-glucosidase maybe involved in the hydrolyzation of rutin to quercetin in C. abbreviatus. However, this requires further study.
Rutin and quercetin reduced the survival and inhibited the development of C. abbreviatus. However, the higher toxicity of quercetin suggests its potential to be developed as a biopesticide for grasshopper control. In this study, we preliminarily evaluated the effects of rutin and quercetin on the physiological response of C. abbreviatus. We propose that further research should be conducted to understand the complex relationship between C. abbreviatus and these two plant-derived compounds, to accelerate their application in pest control. For example, more studies on the feeding behavior of grasshoppers should be conducted before the application of control measures based on quercetin and rutin in the field. Studies on the consumption of quercetin and rutin-treated plants by grasshoppers should also be conducted. The fact that the body mass of the grasshoppers dropped together with an increase in quercetin and rutin concentrations suggested that these botanicals may have an inhibited ability to feed. The next step should be to provide more toxicology tests to determine the lethal medium concentration (LC50), lethal medium dose (LD50) lethal medium time (LT50), and effective methods for applying rutin or quercetin to control C. abbreviatus.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/insects15020095/s1, Table S1: Designed sequences of gene primers for real time PCR.
Author Contributions
Conceptualization, X.H. and L.Z.; methodology, X.H.; software, X.H.; validation, X.H., L.Z. and Y.W.; formal analysis, X.H.; investigation, X.H.; resources, X.H.; data curation, Y.W.; writing—original draft preparation, X.H. and L.Z.; writing—review and editing, X.H. and Y.W; visualization, X.H.; supervision, L.Z.; project administration, X.H.; funding acquisition, X.H. All authors have read and agreed to the published version of the manuscript.
Funding
This work was funded by the National Natural Science Foundation of China (NSFC) (32102199).
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Acknowledgments
We thank DMB editing for its linguistic assistance during the preparation of this manuscript, and the anonymous reviewers for their valuable suggestions to improve an earlier draft.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Wei, S.H.; Huang, W.G.; Zhang, R.; Gao, L.Y.; Yu, Z.; Zhu, M.M. Biological and ecological characteristics of Calliptamus abbreviatus Ikonnikov (Orthoptera: Catantopidae). Chin. J. Appl. Entomol. 2015, 52, 998–1005. [Google Scholar]
- Zhao, F.J.; Wang, Z.H.; Wang, H.P.; Wu, H.H.; Liu, H.W.; Wang, G.J.; Zhang, Z.H. The efects of hyper spectral change on grassland biomass after damage by Clliptamus abbreviates populations of diferent densities. Acta Prataculturae Sin. 2015, 24, 195–203. [Google Scholar]
- Huang, X.B.; Hidayat, U.; Wang, Y.Y. Growth performance and transcriptomic response of Calliptamus abbreviatus Ikonn (Orthoptera: Acrididae) to suitable and unsuitable host plant species. Arthropod-Plant Interact. 2020, 14, 605–612. [Google Scholar] [CrossRef]
- Atreya, K.; Sitaula, B.K.; Overgaard, H.; Bajracharya, R.M.; Sharma, S. Knowledge, attitude and practices of pesticide use and acetylcholinesterase depression among farm workers in Nepal. Int. J. Environ. Health Res. 2012, 22, 401–415. [Google Scholar] [CrossRef] [PubMed]
- Cevizci, S.; Babaoglu, U.T.; Bakar, C. Evaluating pesticide use and safety practices among farmworkers in gallipoli peninsula, Turkey. Southeast Asian J. Trop. Med. Public. Health 2015, 46, 143–154. [Google Scholar] [PubMed]
- Jallow, M.F.; Awadh, D.G.; Albaho, M.S.; Devi, V.Y.; Thomas, B.M. Pesticide knowledge and safety practices among farm workers in Kuwait: Results of a survey. Int. J. Environ. Res. Public Health 2017, 14, 340. [Google Scholar] [CrossRef] [PubMed]
- Senthil-Nathan, S. Physiological and biochemical effect of neem and other Meliaceae plants secondary metabolites against Lepidopteran insects. Front. Physiol. 2013, 4, 359. [Google Scholar] [CrossRef]
- De Oliveira, J.L.; Campos, E.V.R.; Bakshi, M.; Abhilash, P.C.; Fraceto, L.F. Application of nanotechnology for the encapsulation of botanical insecticides for sustainable agriculture: Prospects and promises. Biotechnol. Adv. 2014, 32, 1550–1561. [Google Scholar] [CrossRef]
- Campos, E.V.R.; Proença, P.L.F.; Oliveira, J.L.; Bakshi, M.; Abhilash, P.C.; Fraceto, L.F. Use of botanical insecticides for sustainable agriculture: Future perspectives. Ecol. Indic. 2018, 105, 483–495. [Google Scholar]
- Monsreal-Ceballos, R.J.; Ruiz-Sánchez, E.; Ballina-Gómez, H.S.; Reyes-Ramírez, A.; González-Moreno, A. Effects of botanical insecticides on hymenopteran parasitoids: A meta-analysis approach. Neotrop. Entomol. 2018, 47, 681–688. [Google Scholar] [CrossRef]
- Mouden, S.; Klinkhamer, P.G.L.; Choi, Y.H.; Leiss, K.A. Towards eco-friendly crop protection: Natural deep eutectic solvents and defensive secondary metabolites. Phytochem. Rev. 2017, 16, 935–951. [Google Scholar] [CrossRef]
- Michael, W. Plant secondary metabolites modulate insect behavior-steps toward addiction? Front. Physiol. 2018, 9, 364. [Google Scholar]
- Rosa, E.; Woestmann, L.; Biere, A.; Saastamoinen, M. A plant pathogen modulates the effects of secondary metabolites on the performance and immune function of an insect herbivore. Oikos 2018, 127, 1539–1549. [Google Scholar] [CrossRef]
- Wang, Y.L.; Guan, Z.G.; Jia, X.S.; Wu, S.Y.; Wei, H.G. Study progress of matrine application in farming pest control. J. Shanxi Agric. Sci. 2012, 36, 27–32. [Google Scholar]
- Zanardi, O.Z.; Ribeiro, L.; Ansante, T.F.; Santos, M.S.; Bordini, G.P.; Yamamoto, P.T.; Vendramim, J.D. Bioactivity of a matrinebased biopesticide against four pest species of agricultural importance. Crop Prot. 2015, 67, 160–167. [Google Scholar] [CrossRef]
- Roy, A.; Walker, W.B.; Vogel, H.; Chattington, S.; Larsson, M.C.; Anderson, P.; Heckel, D.G.; Schlyter, F. Diet dependent metabolic responses in three generalist insect herbivores Spodoptera spp. Insect Biochem. Mol. 2016, 71, 91–105. [Google Scholar] [CrossRef]
- Poreddy, S.; Mitra, S.; Schöttner, M.; Chandran, J.; Schneider, B.; Baldwin, I.T.; Kumar, P.; Pandit, S.S. Detoxification of hostplant’s chemical defence rather than its anti-predator co-option drives β-glucosidase-mediated lepidopteran counter adaptation. Nat. Commun. 2015, 6, 8525. [Google Scholar] [CrossRef]
- Züst, T.; Agrawal, A.A. Trade-offs between plant growth and defense against insect herbivory: An emerging mechanistic synthesis. Annu. Rev. Plant Biol. 2017, 68, 513–534. [Google Scholar] [CrossRef] [PubMed]
- Deng, H.F.; Sang, X.Q.; Zhou, L.J. Insecticidal mechanisms of plant secondary metabolites. World Pestic. 2011, 33, 17–21. [Google Scholar]
- Aucoin, R.R.; Phiogène, B.J.R.; Arnason, J.T. Antioxidant enzymes as biochemical defenses against phototoxin-induced oxidative stress in three species of herbivorous Lepidoptera. Arch. Insect Biochem. Physiol. 1991, 16, 139–152. [Google Scholar] [CrossRef]
- Huang, J.; Lü, C.; Hu, M.; Zhong, G. The mitochondria-mediate apoptosis of lepidopteran cells induced by azadirachtin. PLoS ONE 2013, 8, e58499. [Google Scholar] [CrossRef]
- Richards, L.A.; Glassmire, A.E.; Ochsenrider, K.M.; Smilanich, A.M.; Dyer, L.A. Phytochemical diversity and synergistic effects on herbivores. Phytochem. Rev. 2016, 15, 1153–1166. [Google Scholar] [CrossRef]
- Després, L.; David, J.P.; Gallet, C. The evolutionary ecology of insect resistance to plant chemicals. Trends Ecol. Evol. 2007, 22, 298–307. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.K.; Mukhopadhyay, A. Growth, nutritional indices and digestive enzymes of Hyposidra infixaria Walker (Lepidoptera: Geometridae) on artificial and natural (tea) diets. J. Asia-Pac. Entomol. 2016, 19, 167–172. [Google Scholar] [CrossRef]
- Birnbaum, S.S.L.; Rinker, D.C.; Gerardo, N.M.; Abbot, P. Transcriptional profile and differential fitness in a specialist milkweed insect across host plants varying in toxicity. Mol. Ecol. 2017, 26, 6742–6761. [Google Scholar] [CrossRef] [PubMed]
- Schuler, M.A.; Berenbaum, M.R. Structure and function of cytochrome P450S in insect adaptation to natural and synthetic toxins: Insights gained from molecular modeling. J. Chem. Ecol. 2013, 39, 1232–1245. [Google Scholar] [CrossRef]
- Wang, R.L.; Liu, S.W.; Baerson, S.R.; Qin, Z.; Ma, Z.H.; Su, Y.J.; Zhang, J.E. Identification and functional analysis of a novel cytochrome P450 gene CYP9A105 associated with pyrethroid detoxification in Spodoptera exigua Hübner. Int. J. Mol. Sci. 2018, 19, 737. [Google Scholar] [CrossRef]
- Abou-Zaid, M.M.; Beninger, C.W.; Arnason, J.T. The effect of one flavone, two catechins and four flavonols on mortality and growth of the European corn borer (Ostrinia nubilalis Hubner). Biochem. Syst. Ecol. 1993, 21, 415–420. [Google Scholar] [CrossRef]
- Silva, T.R.F.B.; Almeida, A.C.D.S.; Moura, T.D.L.; Silvaet, A.R.D.; Freitas, S.D.S.; Jesus, F.G.D. Effect of the flavonoid rutin on the biology of Spodoptera frugiperda (Lepidoptera: Noctuidae). Acta Sci. Agron. 2016, 38, 165–170. [Google Scholar] [CrossRef]
- Chen, C.; Han, P.; Yan, W.; Wang, S.; Shi, X.; Zhou, X.; Desneux, N.; Gao, X. Uptake of quercetin reduces larval sensitivity to lambda-cyhalothrin in Helicoverpa armigera. J. Pest Sci. 2017, 91, 919–926. [Google Scholar] [CrossRef]
- Bentivenha1, J.P.F.; Canassa, V.F.; Baldin, E.L.L.; Borguini, M.G.; Lima, G.P.P.; Lourenção, A.L. Role of the rutin and genistein flavonoids in soybean resistance to Piezodorus guildinii (Hemiptera: Pentatomidae). Arthropod-Plant Interact. 2018, 12, 311–320. [Google Scholar] [CrossRef]
- Li, S.; Huang, X.; McNeill, M.R.; Liu, W.; Tu, X.; Ma, J.; Lv, S.; Zhang, Z. Dietary stress from plant secondary metabolites contributes to grasshopper (Oedaleus asiaticus) migration or plague by regulating insect insulin-like signaling pathway. Front. Physiol. 2019, 10, 531. [Google Scholar] [CrossRef]
- Chen, H.; Liu, W. Determination of rutin and quercetin content in Yantai Bupleurum by HPLC. Med. Plant 2014, 5, 57–64. [Google Scholar]
- Herde, M.; Howe, G.A. Host plant-specific remodeling of midgut physiology in the generalist insect herbivore Trichoplusia ni. Insect Biochem. Mol. Biol. 2014, 50, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Erb, M.; Robert, C.A. Sequestration of plant secondary metabolites by insect herbivores: Molecular mechanisms and ecological consequences. Curr. Opin. Insect Sci. 2016, 14, 8–11. [Google Scholar] [CrossRef] [PubMed]
- Luque, T.; Okano, K.; Reilly, D.R.O. Characterization of a novel silkworm (Bombyx mori) phenol UDP-glucosyltransferase. Eur. J. Biochem. 2002, 269, 819–825. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.J.; Gao, X.W.; Lei, M.Q.; Zheng, B.Z. Effects of tannic acid on glutathione S-transferases in Helicoverpa armigera (Hübner). Acta Entomol. Sin. 2003, 46, 684–690. [Google Scholar]
- Li, X.C.; Baudry, J.; Berenbaum, M.R.; Schuler, M.A. Structural and functional divergence of insect CYP6B proteins: From specialist to generalist cytochrome P450. Proc. Natl. Acad. Sci. USA 2004, 101, 2939–2944. [Google Scholar] [CrossRef]
- Celorio-Mancera, M.L.; Ahn, S.J.; Vogel, H.; Heckel, D.G. Transcriptional responses underlying the hormetic and detrimental effects of the plant secondary metabolite gossypol on the generalist herbivore Helicoverpa armigera. BMC Genom. 2011, 12, 575. [Google Scholar] [CrossRef]
- Wang, R.L.; Sun, Y.L.; Liang, X.T.; Song, Y.Y.; Su, Y.J.; Zhu, K.Y.; Zeng, R.S. Effects of six plant secondary metabolites on activities of detoxification enzymes in Spodoptera litura. Acta Ecol. Sin. 2012, 32, 5191–5198. [Google Scholar] [CrossRef]
- Whitman, D.W.; Ananthrakrishnan, T.N. Phenotypic Plasticity of Insects: Mechanisms and Consequences; Science Publishers: Enfield, NH, USA, 2009. [Google Scholar]
- Castañeda, L.E.; Figueroa, C.C.; Nespolo, R.F. Do insect pests perform better on highly defended plants? Costs and benefits of induced detoxification defences in the aphid Sitobion avenae. J. Evol. Biol. 2010, 23, 2474–2483. [Google Scholar] [CrossRef]
- Rivero, A.; Magaud, A.; Nicot, A.; Vezilier, J. Energetic cost of insecticide resistance in Culex pipiens mosquitoes. J. Med. Entomol. 2011, 48, 694–700. [Google Scholar] [CrossRef] [PubMed]
- Gordon, J.R.; Potter, M.F.; Haynes, K.F. Insecticide resistance in the bed bug comes with a cost. Sci. Rep. 2015, 5, 10807. [Google Scholar] [CrossRef] [PubMed]
- Schwenke, R.A.; Lazzaro, B.P.; Wolfner, M.F. Reproductionimmunity trade-offs in insects. Annu. Rev. Entomol. 2016, 61, 239–256. [Google Scholar] [CrossRef] [PubMed]
- Rane, R.V.; Walsh, T.K.; Pearce, S.L.; Jermiin, L.S.; Gordon, K.H.; Richards, S.; Oakeshott, J.G. Are feeding preferences and insecticide resistance associated with the size of detoxifying enzyme families in insect herbivores? Curr. Opin. Insect Sci. 2016, 13, 70–76. [Google Scholar] [CrossRef]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef]
- Matsumura, T.; Matsumoto, H.; Hayakawa, Y. Heat stress hardening of oriental armyworms is induced by a transient elevation of reactive oxygen species during sublethal stress. Arch. Insect Biochem. Physiol. 2017, 96, e21421. [Google Scholar] [CrossRef]
- Krishnan, N.; Kodrík, D. Antioxidant enzymes in Spodoptera littoralis (Boisduval): Are they enhanced to protect gut tissues during oxidative stress? J. Insect Physiol. 2006, 52, 11–20. [Google Scholar] [CrossRef]
- Kohl, K.D.; Samuni-Blank, M.; Lymberakis, P.; Kurnath, P.; Izhaki, I.; Arad, Z.; Karasov, W.H.; Dearing, M.D. Effects of fruit toxins on intestinal and microbial β-glucosidase activities of seed-predating and seed-dispersing rodents (Acomys spp.). Physiol. Biochem. Zool. 2016, 89, 198–205. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).