2. Materials and Methods
The studied specimen of fossil ant (
Figure 1,
Figure 2 and
Figure 3G,H;
Supplementary Materials) originated from the Baltic amber, Priabonian age (Late Eocene, 37.8–33.9 Ma) [
8], the Prussian Formation, on the Sambia Peninsula near Kaliningrad, Kaliningrad region, Russia. The holotype of the new species is kept in the collection of the Kaliningrad Amber Museum, Kaliningrad, Russia, under the collection number KAM BX 34/22.1. The amber was hand processed for study (cut and polished) by one of the authors (DD) at the St. Petersburg State University (PalaeoEntLab).
The studies were performed on the equipment of the Research Park of St. Petersburg State University (“Centre for X-ray Diffraction Studies”, project No. 103-23769; “Resource Centre for Microscopy and Microanalysis”, project No. 112-23465 and “Computing Centre”, project No. 110-27449).
Photography and morphological analysis of samples were performed using a Leica M205C motorized stereomicroscope. SEM micrographs were obtained on Desktop Scanning Electron Microscope Hitachi TM3000 (Hitachi Corp., Tokyo, Japan). Subsequent image processing was carried out using the Helicon Focus Pro 8, Kritta 5.0.2 and Inkscape 1.2 software.
Micro-computed tomography allowed us to make accurate measurements of the specimen and to study all of the traditional diagnostic features of the new species. For a clearer graphical representation of the information, we made a reconstruction of a new species in the form of a digitally painted and straightened 3D model. Arrays of microtomographic sections were obtained using a desktop high-resolution X-ray microtomograph SkyScan 1172. Visualization, volume rendering, and segmentation of tomographic sections were performed in 3DSlicer 5.1 and Drishti 3.0 software. The holotype was scanned with the following parameters: voltage 40 kV, current 250 µA, without a filter, with a pixel size of 2.46 microns and a resolution of 2848 × 2692 pixels per slice with a continuous 360° rotation, and a shutter speed of 1300 ms per frame (2268 X-ray projections).
The inclusion was inverted and subsequently isolated from the surrounding background using the software segmentation editor. Part of poorly preserved morphological structures were previously segmented in the segmentation editor by manually labeling each slice.
The measurements indicated below were performed by volume rendering of the sample in 3DSlicer which made all morphological structures available for study (as opposed to studying samples directly in amber with the help of a microscope) and made it possible to measure with precision of 0.01 mm. The results of segmentation (in 3DSlicer) in the file format PLY were imported into the free and open Blender 3.1 software for “straightening” (changing the position of limbs and other parts of the body), painting, and animation of the ant reconstruction. The sample was “straightened” using the Auto-Rig PRO tool. All the individual parts of the reconstruction were transformed into polygonal grids using the Smart UV project tool and painted manually (the color of the models is not the natural color of the specimen or the assumed lifetime color).
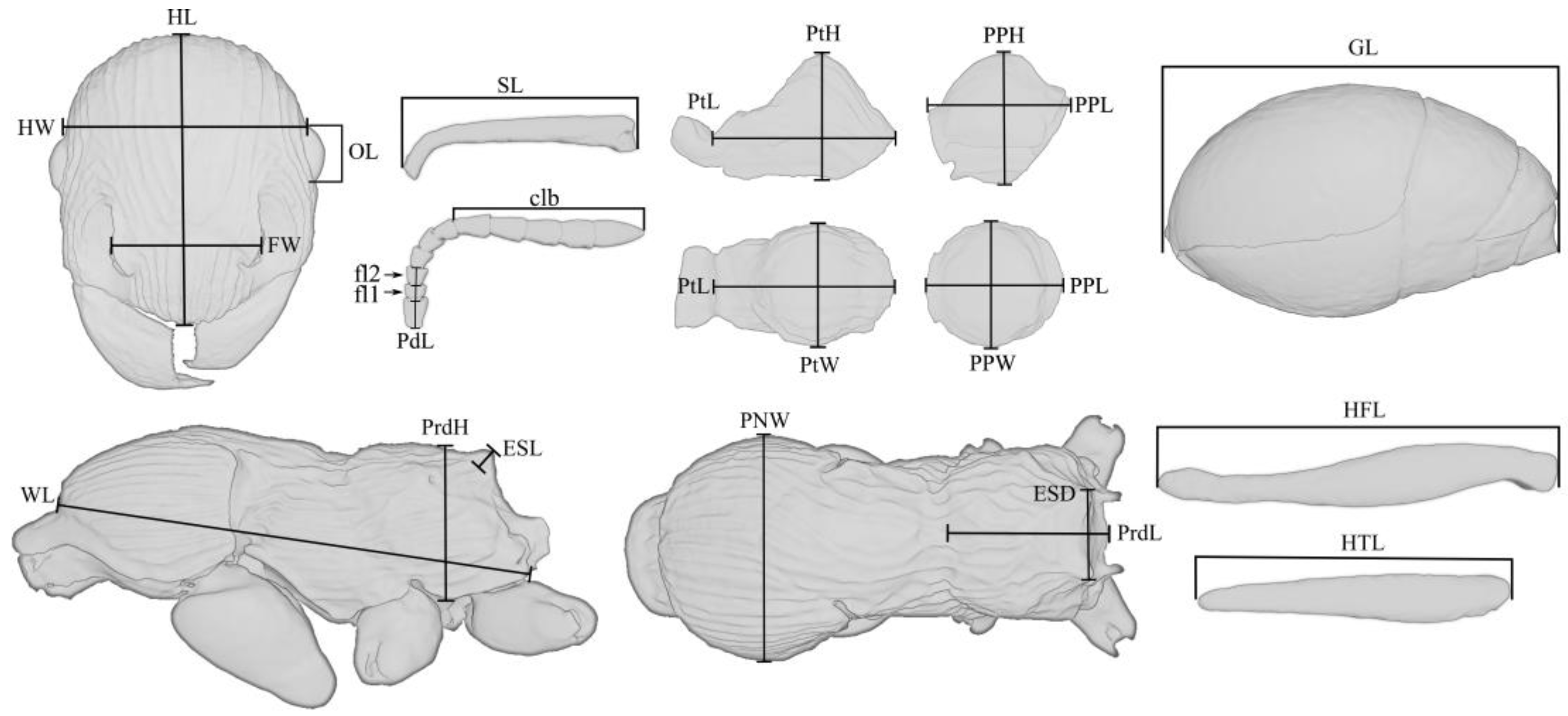
The dimensional values of morphological structures are given in millimeters. The following designations are used in the text:
HW—head width, measured along the upper line of the eyes;
HL—head length, maximum length of the head, measured from the posterior margin of the head to the anterior-most extremity of the clypeus;
SL—the maximum straight-line length of the scape measured from antennal bulb to the apex;
FW—the maximum width measured between the frontal lobes;
PdL—pedicellum length;
FI1—the length of the first flagellomere;
FI2—the length of the second flagellomere;
OL—the maximum eye length measured by maximum diameter;
MdL—the length of the mandible, measured from the mandibular apex to the anterior clypeus margin, or to the transverse line connecting the anterior-most points in those taxa where the margin is concave medially;
WL—Weber’s length, the diagonal length of the mesosoma in profile from the point at which the pronotum meets the cervical shield to the posterior basal angle of the metapleuron;
PNW—the maximum width of the pronotum in dorsal view;
ESL—maximum length of propodeal tubercles (or spines) in profile, measured along the tubercle/spine from its tip to the deepest point of the propodeal constriction at its base;
ESD—distance between the tips of propodeal tubercles/spines in dorsal view;
PtL—the length of the petiolar node in profile, measured as the distance from the place of attachment to the propodeum to the place of attachment to the postpetiole;
PtH—the height of the petiolar node in profile, measured as the perpendicular distance from the ventral edge to the highest point of the petiolar node;
PtW—the maximum width of the petiolar node in dorsal view;
PPL—the length of the postpetiole in profile, measured as the distance from the place of attachment to the petiolar node to the place of attachment to the gaster;
PPH—the height of the postpetiole in profile, measured as the perpendicular distance from the ventral edge to the highest point of the postpetiole;
PPW—the maximum width of the postpetiole in dorsal view;
HFL—the maximum length of hind femur, measured in anterior view;
HTL—the maximum length of hind tibia, measured in anterior view;
PrdL—the maximum length of the propodeum in dorsal view;
PrdH—the height of the propodeum in profile, measured as the perpendicular distance from the ventral edge to the highest point of the propodeum;
GL—the length of the gaster, measured as the distance from the place of attachment of the postpetiole to the top of the gaster in ventral view;
TL—the total length of the ant (=HL + MdL + WL + PtL + PPL + GL).
Indices:
CI (cephalic index) = HL/HW
SI1 (scape length index) = SL/HL
SI2 (scape width index) = SL/HW
FLI (frontal lobes index) = FW/HW
OI1 (eye length index) = OL/HL
OI2 (eye width index) = OL/HW
PI1 (petiole height index) = PtL/PtH
PI2 (petiole width index) = PtL/PtW
PPI1 (postpetiole height index) = PPL/PPH
PPI2 (postpetiole width index) = PPH/PPW
PPI3 (postpetiole-petiole index) = PPW/PtW
ESLI (propodeal spine length index) = ESL/HW
ESDI (propodeal spine distance index) = ESD/ESL
MI (mesosomal index) = WL/PNW
PRI (propodeal index) = PrdL/PrdH
4. Discussion
Over the past 65 million years, geological events and changing climatic conditions have profoundly affected the distribution of plants and animals [
9]. Biogeographic data and paleontological findings may provide some clues about the diversification of the tribe Myrmicini and the origin of its modern genera. Based on the estimates of Ward et al. [
7], the Myrmicini tribe forms a monophyletic group that is sister to all other in the Myrmicinae subfamily. Myrmicini is considered morphologically the most primitive in the subfamily Myrmicinae, based on the plesiomorphic states of many features such as 6−segmented maxillary and 4−segmented labial palps, 11–12−segmented antennae in females, and 12–13−segmented antennae in males, usually a well-developed pectinate spur on the middle and hind tibiae, the mesosomal structure with clearly defined sutures (what is noted in the genus
Manica, for example see
Figure 2 and
Figure 3;
Supplementary Materials), etc., [
6]. Moreover, in comparison with
Myrmica, the genus
Manica has a more “primitive” biology [
10,
11,
12]. Given the plesiomorphic features, the modern subordinate position of
Manica species in modern montane ant societies and absence of paleontological finds (prior to this work), there is limited reason to think that
Manica were once widespread ants. This may be an advantage due to the lower prevalence of dominant species of ants in such an environment. Representatives of the genus
Manica, as in the case of its related genus
Myrmica and some other representatives of the subfamily Myrmicinae, are among the ants subordinate in the hierarchy of dominance and, therefore, less able to protect resources [
13]. Considering the modern lifestyle of
Manica i.e., confined to the mountains, with small colonies, and with workers rarely coming to the surface [
2], the finding of this genus in amber was rather unlikely.
Presently
Myrmica species are distributed mainly in the Holarctic, but also in the northern parts of the Indomalayan and Neotropical regions.
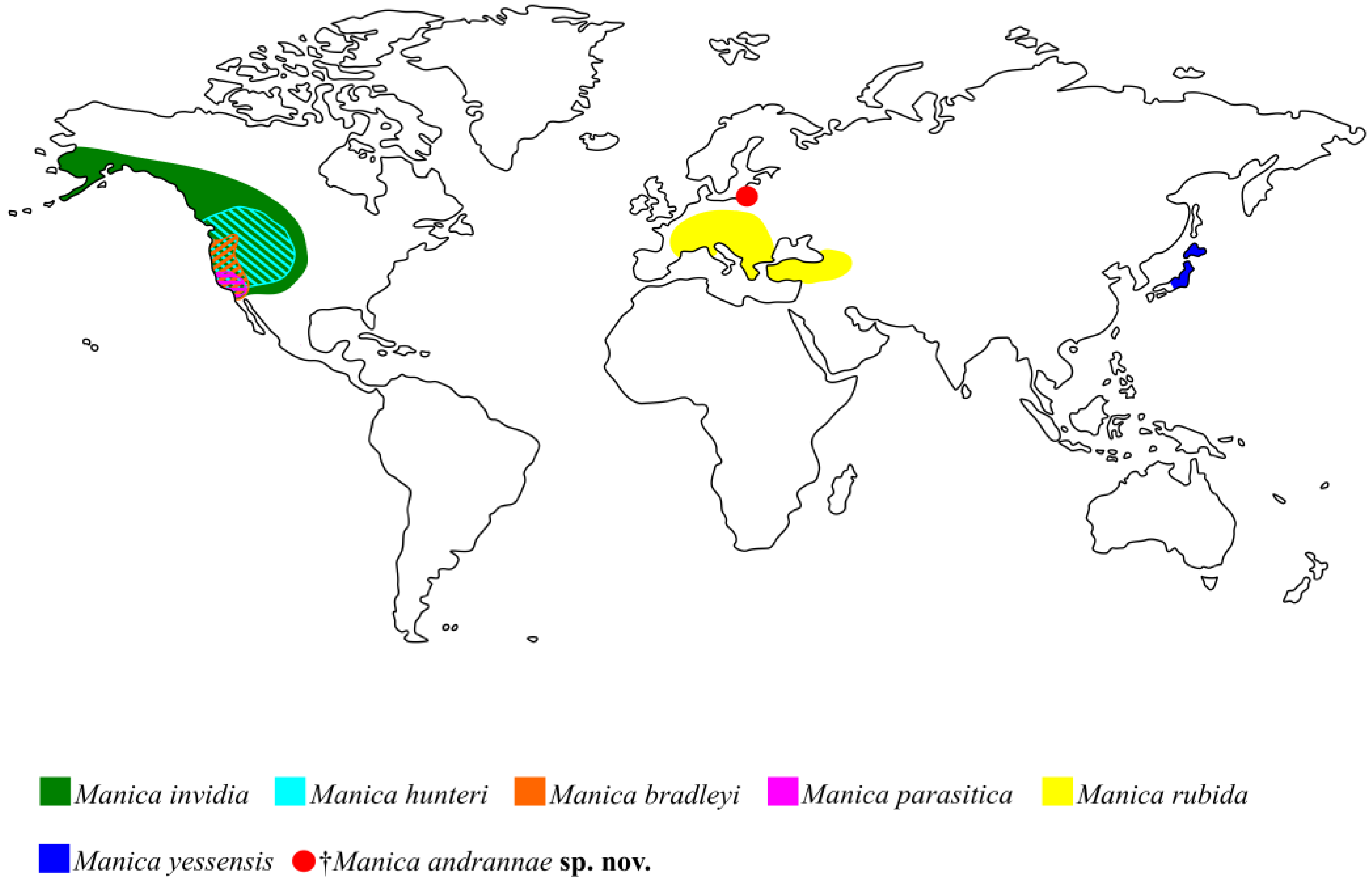
Manica has an exclusively Holarctic distribution (
Figure 4). There is not much paleontological evidence for the geographical origin of the genus
Myrmica. In comparison with numerous other recent genera, the samples of
Myrmica fossils are quite rare and occur exclusively in late Eocene European amber [
14]. Even considering that Eocene European amber is the most studied fossil ant fauna in the world [
15,
16,
17], only five species of
Myrmica [
14,
18,
19], two species of
Plesiomyrmex Dlussky and Radchenko, 2009 and
Protomyrmica Dlussky and Radchenko, 2009 [
20], and one species of
Manica described here are known for the Myrmicini tribe. Their lifestyle hardly favored getting them into the resin, but by the late Eocene they, apparently, were already quite diverse.
Recent
Myrmica species in Central and Southeast Asia (Himalayas, southern China, Burma, Thailand, Vietnam, and Taiwan) demonstrate the most diverse morphology and also contain the largest number of species, most of which are endemic and have many plesiomorphic states. For example,
Myrmica mirabilis Elmes and Radchenko, 1998, has primitive features that can bring it closer to
Manica: general shape and large body size, promesonotal suture weak in dorsal view, non-obvious 4-jointed apical club (it happens to
Manica too) [
21]. Multidentate mandibles with more than 10 denticles of
Myrmica hecate Weber, 1947 is also a feature characteristic of the genus
Manica. In more southern areas, species of
Myrmica live exclusively in mountain forests and alpine meadows at altitudes of more than 1200–2000 m, and sometimes even higher than 4800 m [
6,
14]. For example, the west Palearctic ant paleofauna was very similar in composition (at generic level) and structure to the ones observed now only in the mountain area of the Indomalayan region [
22]. That is, they are being displaced, such as
Manica, by more competitive species. It seems logical to assume that the genus
Myrmica first appeared in the World, and not in the New World, where none of the species is similar to the species found in Late Eocene amber, unlike the species of the Old World. Paleontological data from amber show that the species of the
ritae group and other ancient forms of
Myrmica were certainly present in Europe in the Late Eocene (ca. 37.8–33.9 Mya).
Manica, in turn, has 4 out of 6 species that live in North America (
Figure 4) [
1]. Based on the morphology of
Manica and taking into account our new finding, we can suggest the place of origin of this genus. Wheeler and Wheeler [
2] state that
Manica originated from the New World based on the presence of a larger number of species in the Nearctic and what is found there, according to the authors, the putative ancestral species—
M. invidia. They considered
M. invidia as the supposed ancestor, based on the fact that this species is the most “plastic” and has the largest range. However, the authors assumed the origin of
M. rubida in the Old World, not yet knowing about the existence of
M. yessensis in Japan. Radchenko and Elmes [
23] also believed that place of origin of
Manica is most likely the New World.
Three of the six extant species, namely the Nearctic
M. hunteri and the Palaearctic
M. rubida and
M. yessensis have a ventral postpetiolar process, similarly to the new fossil
†M. andrannae sp. n..
M. hunteri also has the anterior clypeal margin medially unnotched, such as the fossil species. Based on these morphological features, it can be assumed that the ancestral state observed in
M. hunteri,
M. invidia, and
M. rubida are very similar, but
M. invidia differs from these species in the absence of a ventral process of postpetiole and the presence of deep medially notched clypeus. †
M. andrannae sp. n. is most similar to
M. yessensis in general body rugosity, as well as
M. yessensis has a sharp edge of the propodeum, as if there are small teeth. It seems unlikely that the ancestor of
Manica spread from Europe to Asia and vice versa, even through the existing shores or islands of Tethys Ocean. Since, apparently, the Western Palearctic fauna developed in a different way to a greater extent isolated from the Eastern Palearctic fauna [
9]. This suggests that
Manica could have formed in the New World, after which the ancestor of
M. yessensis got to the eastern Palearctic through Beringia, and the ancestor of
M. rubida to Europe through Thulean Bridge.
Based on the discussion above, we assume that the center of origin of
Manica was in the New World. If this scenario is accepted, then the common ancestor of the modern genera Myrmicini must have existed before the complete separation of North America and Eurasia. From the Mid Cretaceous (ca. 100 Mya) throughout the Tertiary, northeastern Eurasia was almost permanently connected to the Nearctic via Beringia [
24,
25,
26]. Additionally, the Atlantic Thulean and De Geer routes connected Europe and eastern North America in the late Cretaceous and Early Tertiary [
26,
27]. Most important for the exchange of flora and fauna was the Thulean Bridge, which connected Europe to Greenland via the British Isles and persisted until the early Eocene (about 50 Ma) [
9,
23,
25]. On this basis, it could be assumed that the ancestor of Myrmicini had already evolved by the time of the destruction of the Thulean Bridge (50 Mya), which corresponds to the estimated time of origin of tribe based on molecular and paleontological data [
7].
Interesting in the context of this work is †
Myrmica paradoxa Radchenko et al., 2007 [
14] from Bitterfeld amber, which looks superficially similar to †
Manica andrannae sp. n. in some features: it has short propodeal tubercles that are not found in other
Myrmica, and the promesonotal suture is faint but visible on the dorsum. However, according to the set of features in the description, and which can be seen from the only and poor photo, it is unlikely to be a
Manica (3 jointed club, uncharacteristic head sculpture for
Manica). Micro-computed tomography scanning is required to check all available morphological features in more detail of †
Myrmica paradoxa.
Our finding does not contradict, but rather confirms the idea of Radchenko and Elmes [
23], that
Manica and
Myrmica most likely originated from a common ancestor at about the same time about 50 million years ago during the surge of diversification of ants and before North America was completely separated from Eurasia. The common ancestor of these genera apparently had spines on the propodeum, a full complement of mesosomal sutures and the promesonotal suture impressed in dorsal view. However, if we take into account the modern diversity of these genera and the paleontological finds, it seems that these genera diverged much earlier than in the Eocene.
Manica could have formed in the New World and then ended up in the Palearctic, and
Myrmica could have formed in the Old World.