The Morphological Diversity of Antlion Larvae and Their Closest Relatives over 100 Million Years
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Material
2.2. Documentation Methods
2.3. Digital Drawings
2.4. Shape Analysis
2.5. Statistical Analysis
2.6. Note on Terminology
3. Results
3.1. Short Descriptions of New Specimens
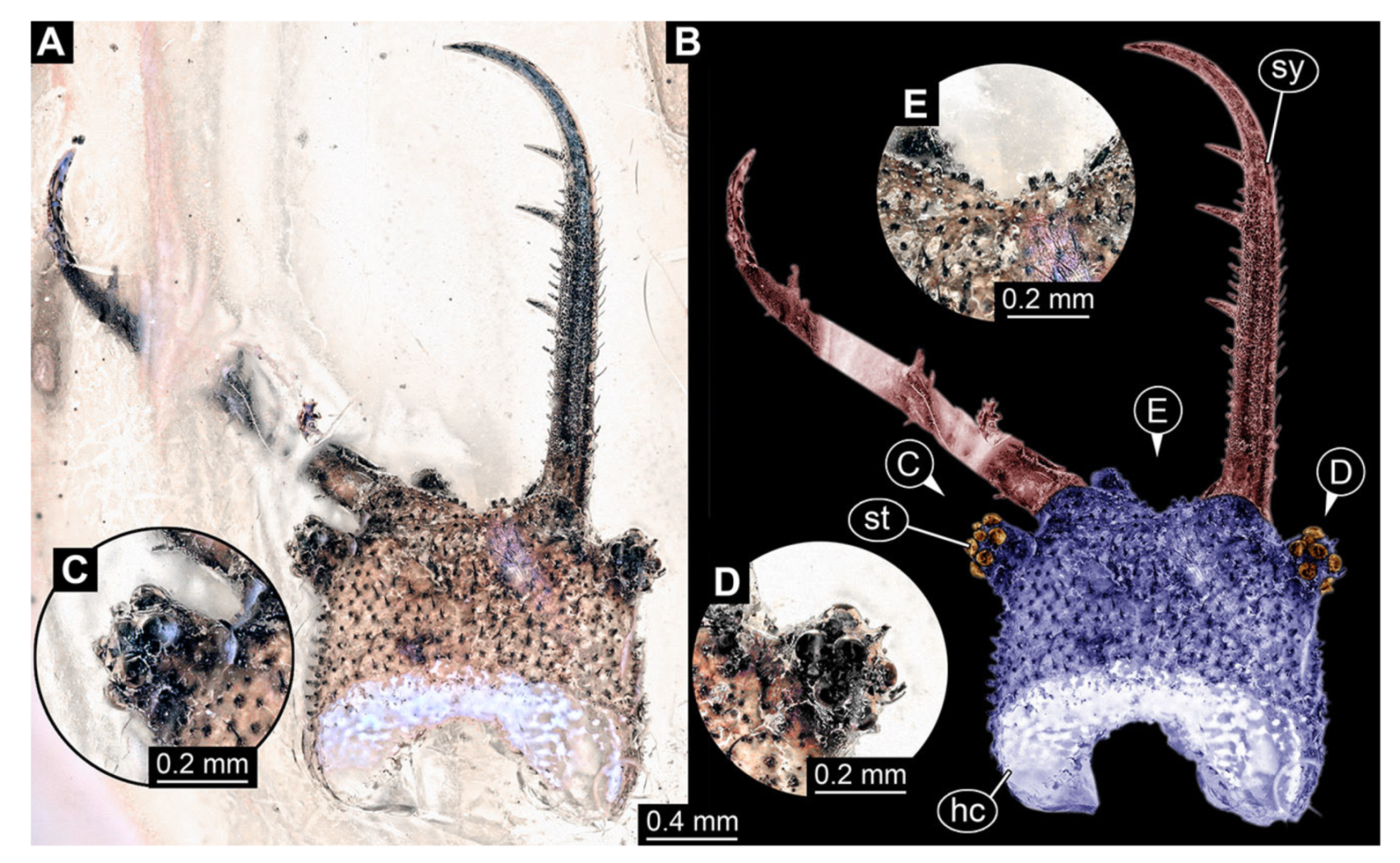
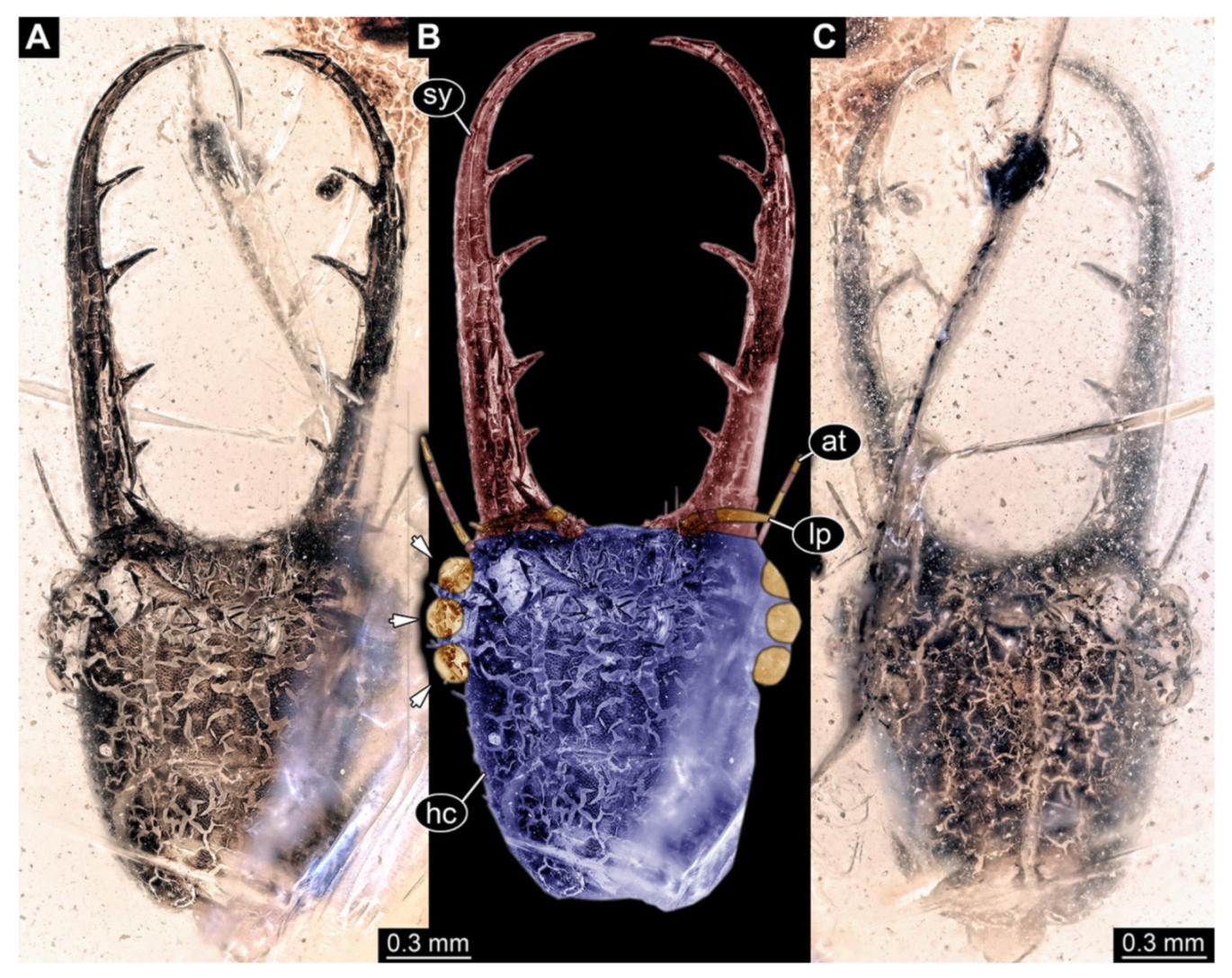
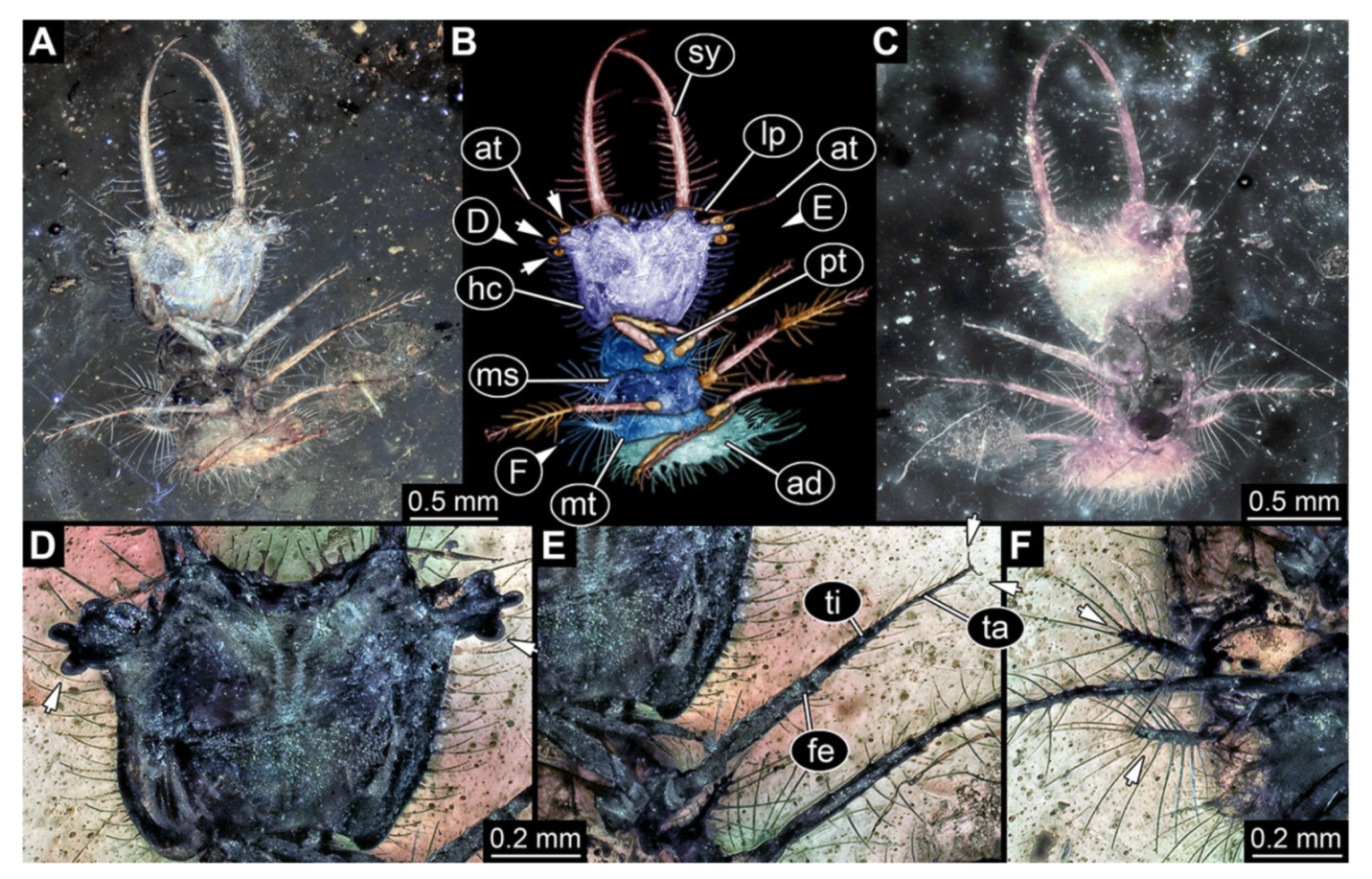
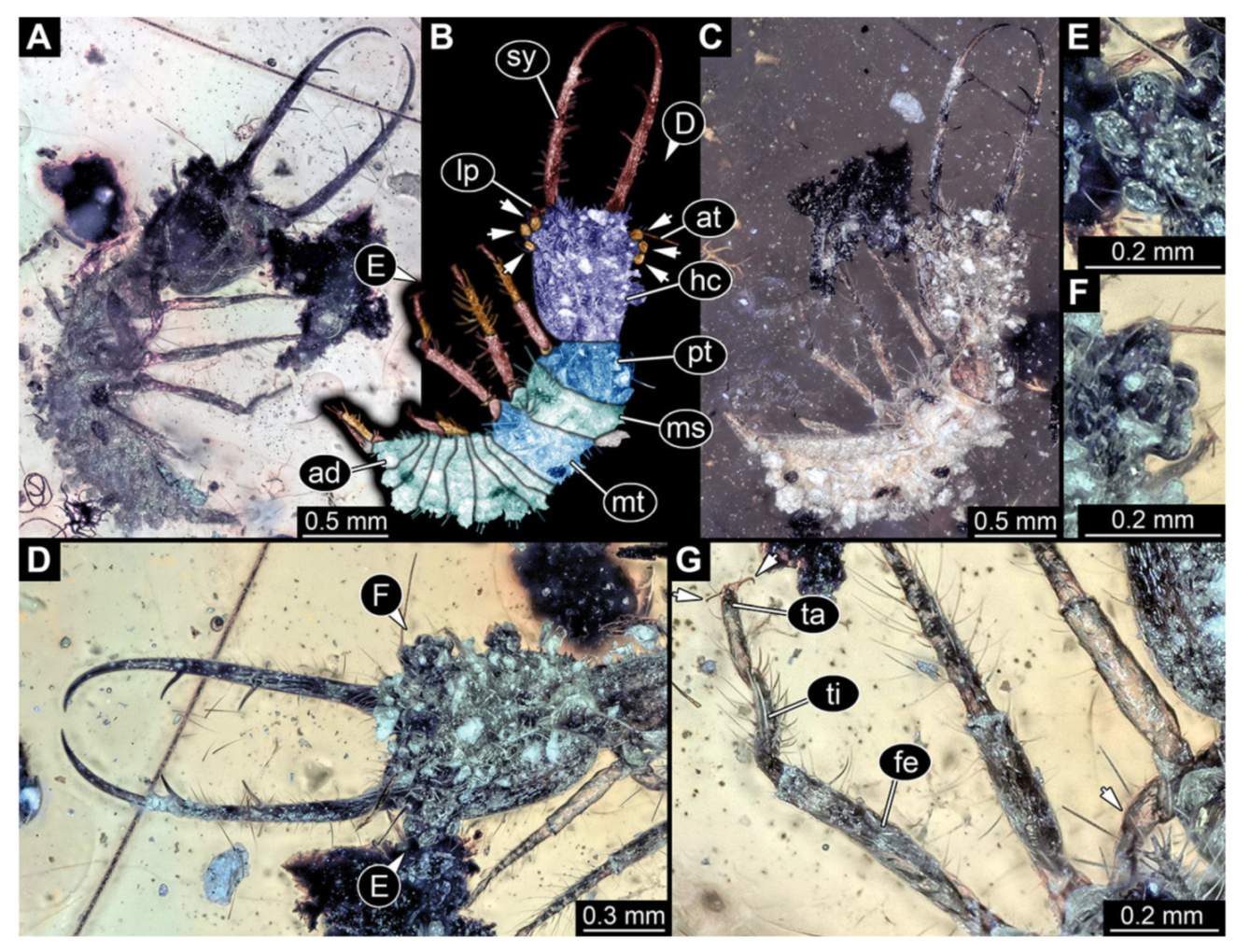
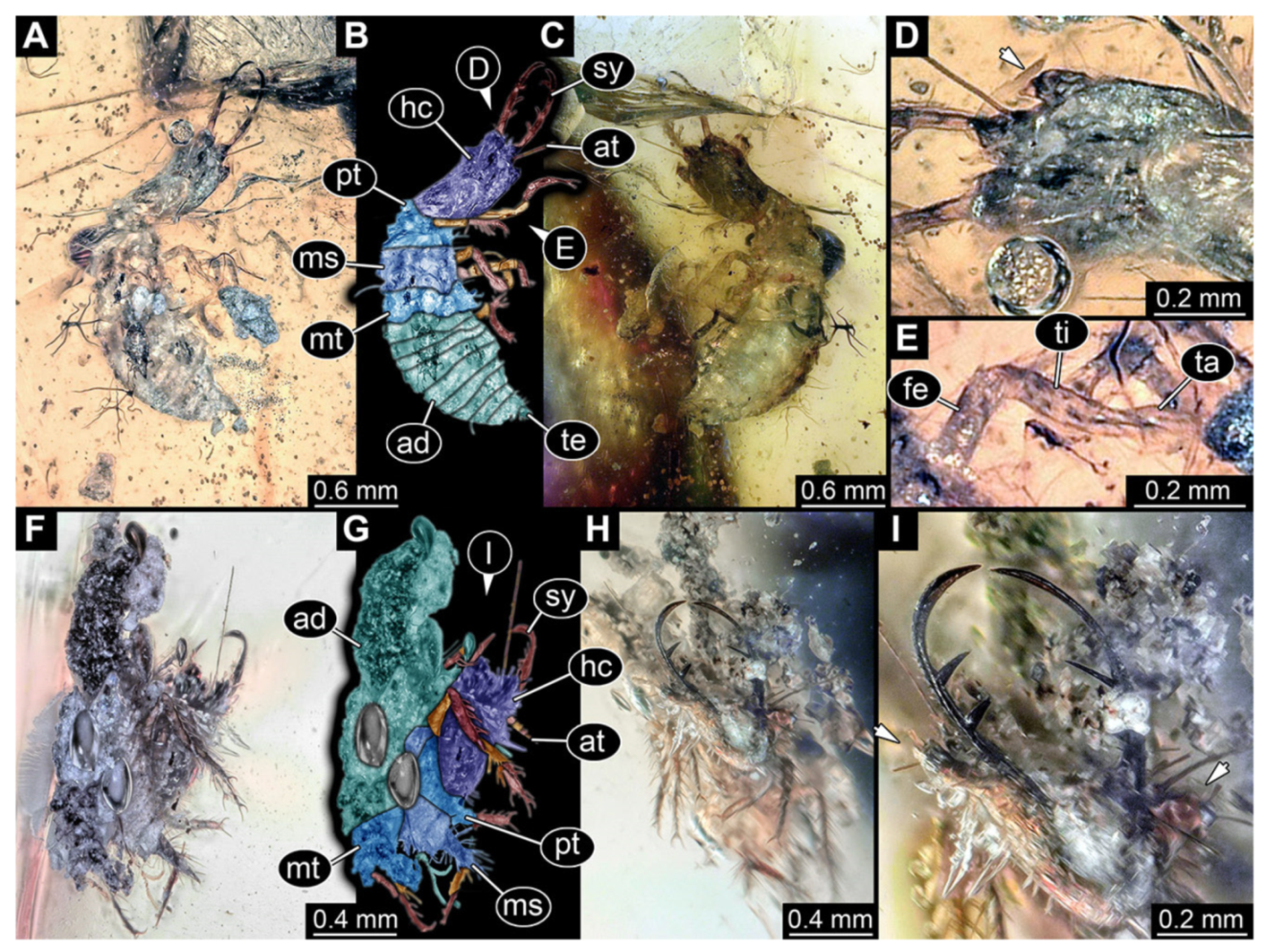
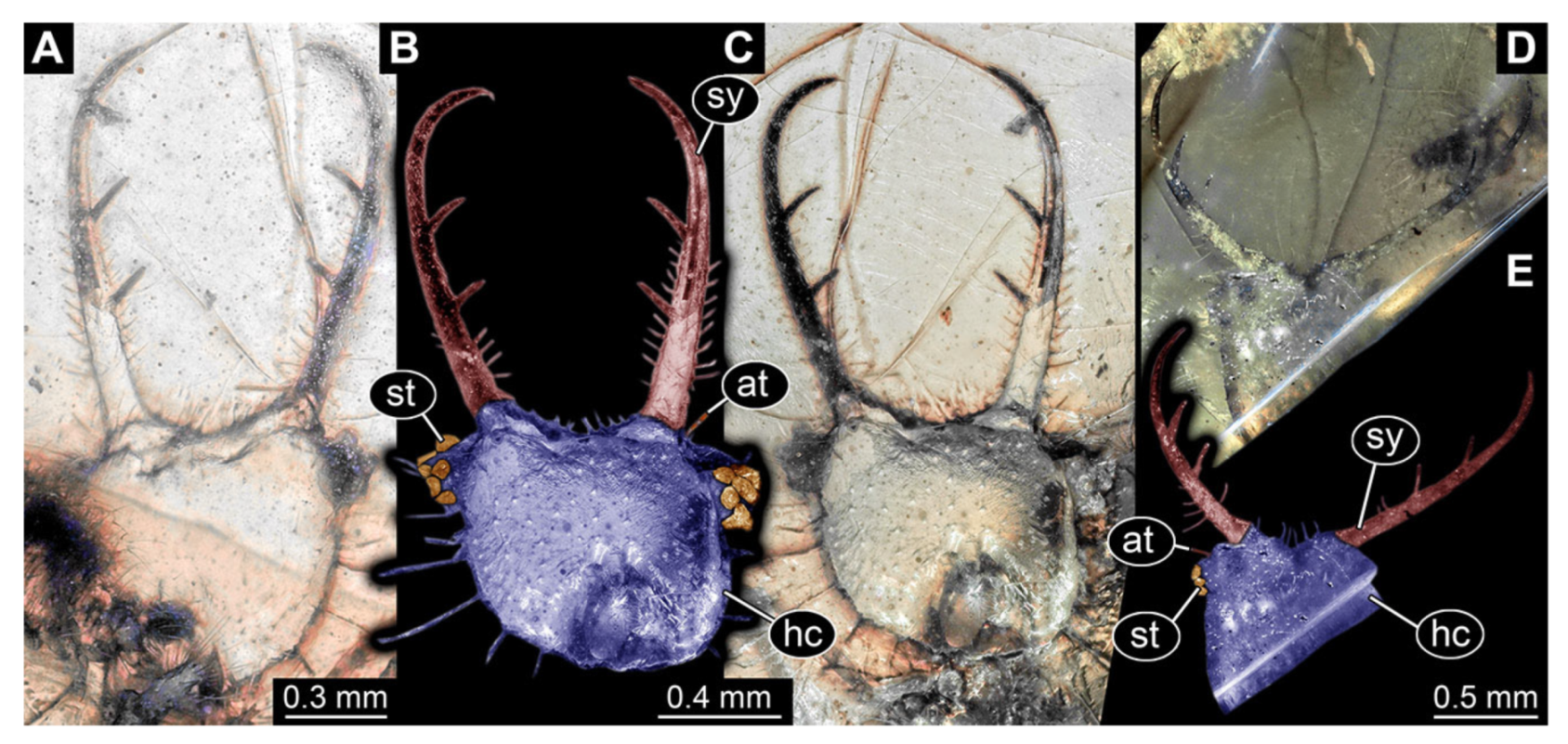
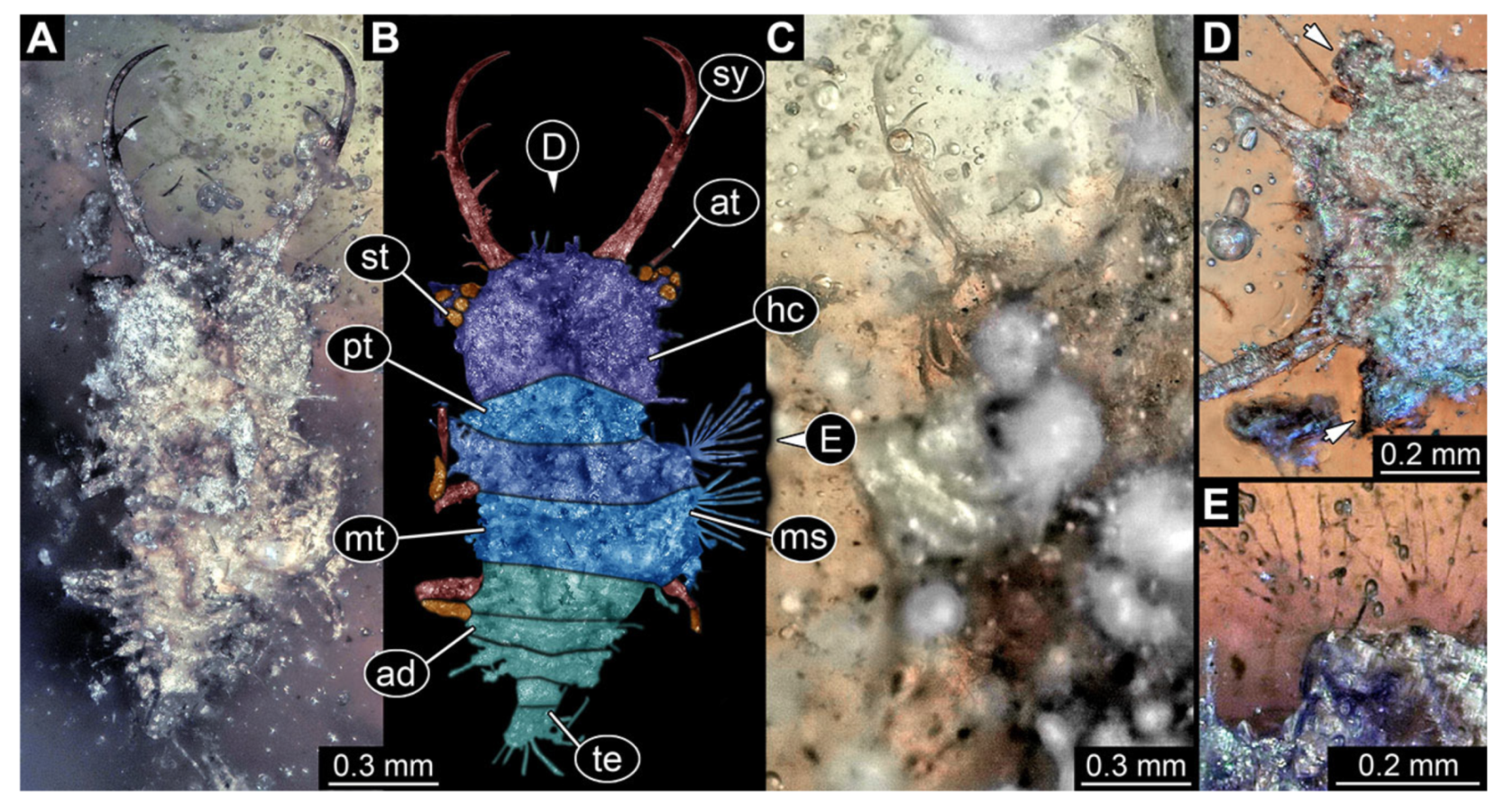
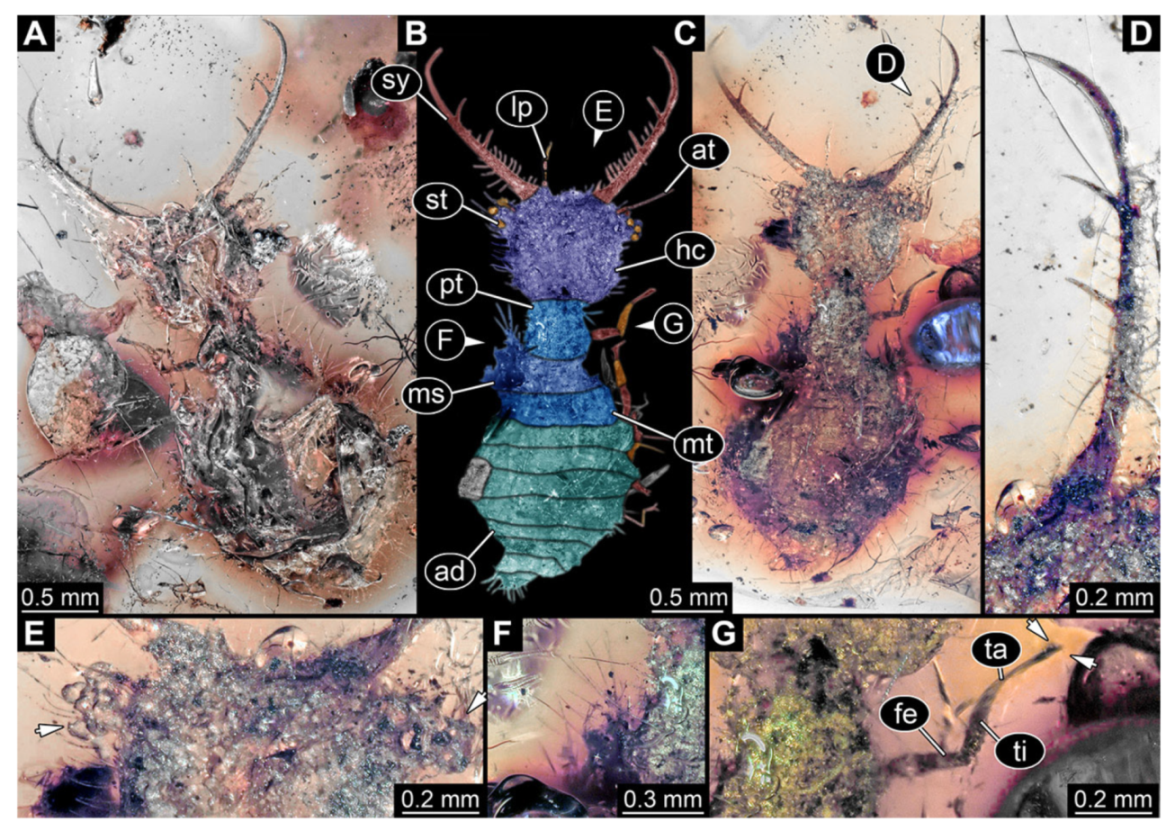
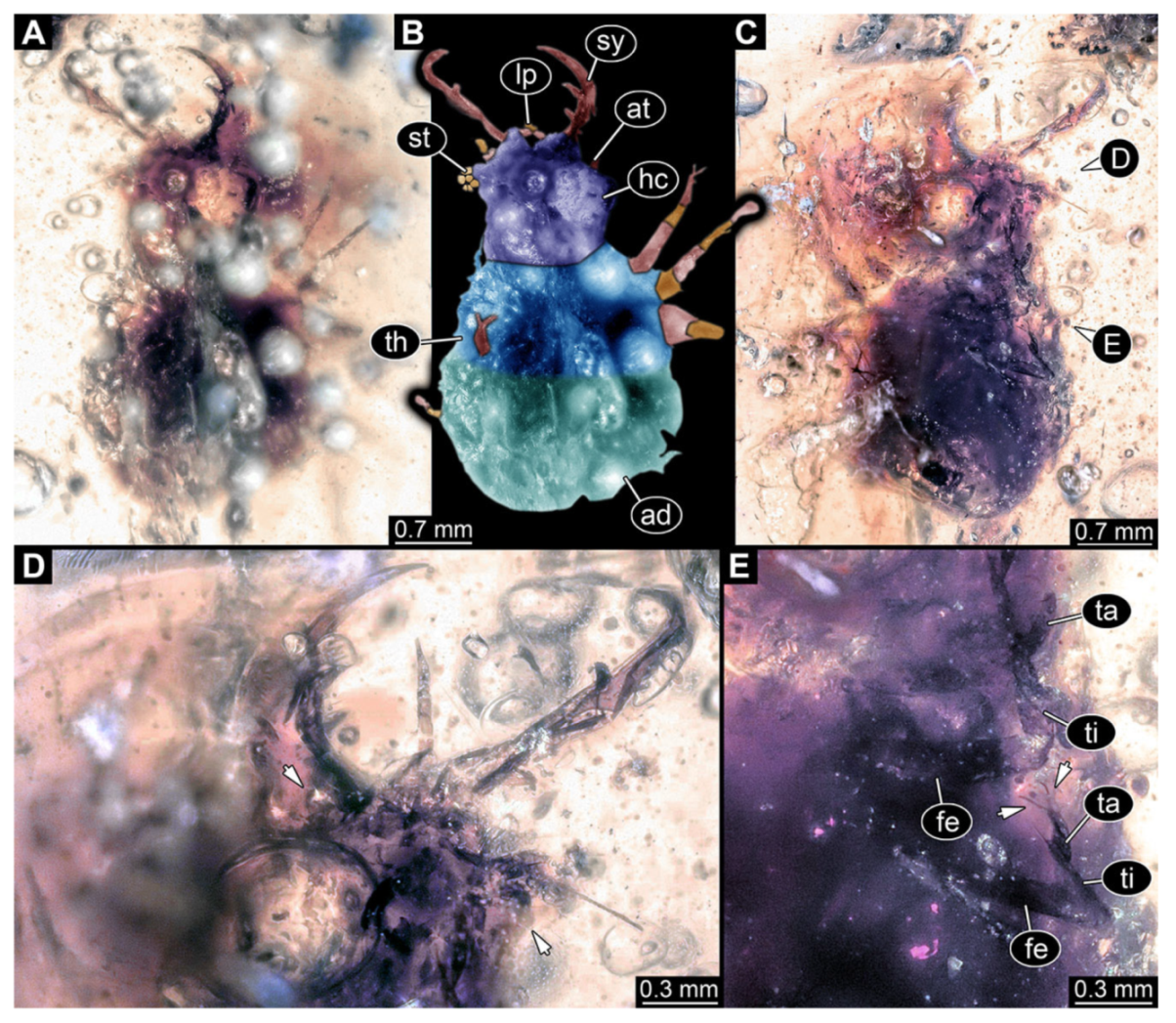
- Specimen 3102 (CCGG 2525), preserved in Eocene Baltic amber. Only the head preserved. Specimen well accessible in dorsal view, with the left stylet partially obscured (Figure 1A,B). Head capsule roughly square-shaped in dorsal view; stylets longer than head capsule (Figure 1A,B). Stemmata on prominent protrusions on each side of head capsule (Figure 1C,D). Each stylet has three major teeth and numerous smaller teeth or robust setae (Figure 1A,B). Anterior rim of head medially with a pair of larger tubercles on each side, close to the midline (Figure 1E). No antenna or palp apparent. Preserved part of specimen is 3.40 mm.
- 2.
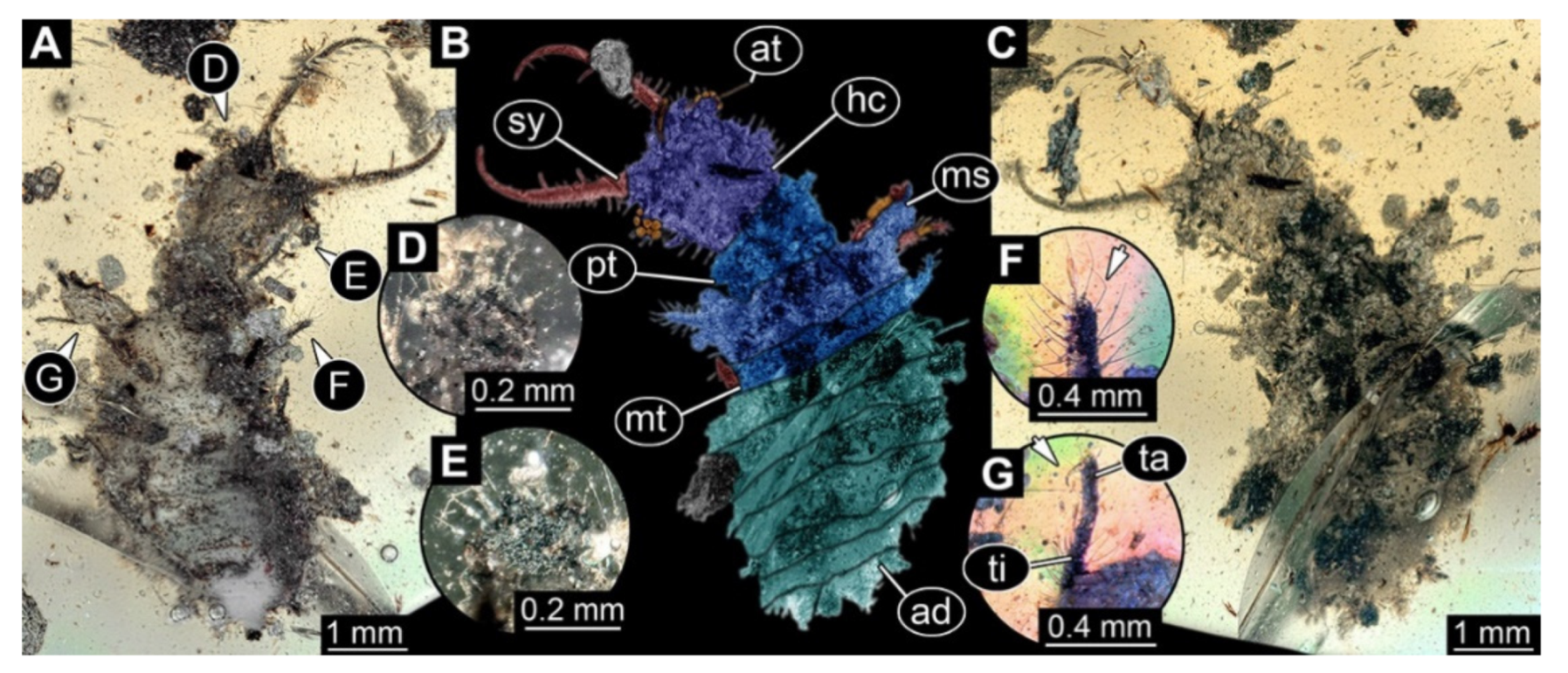
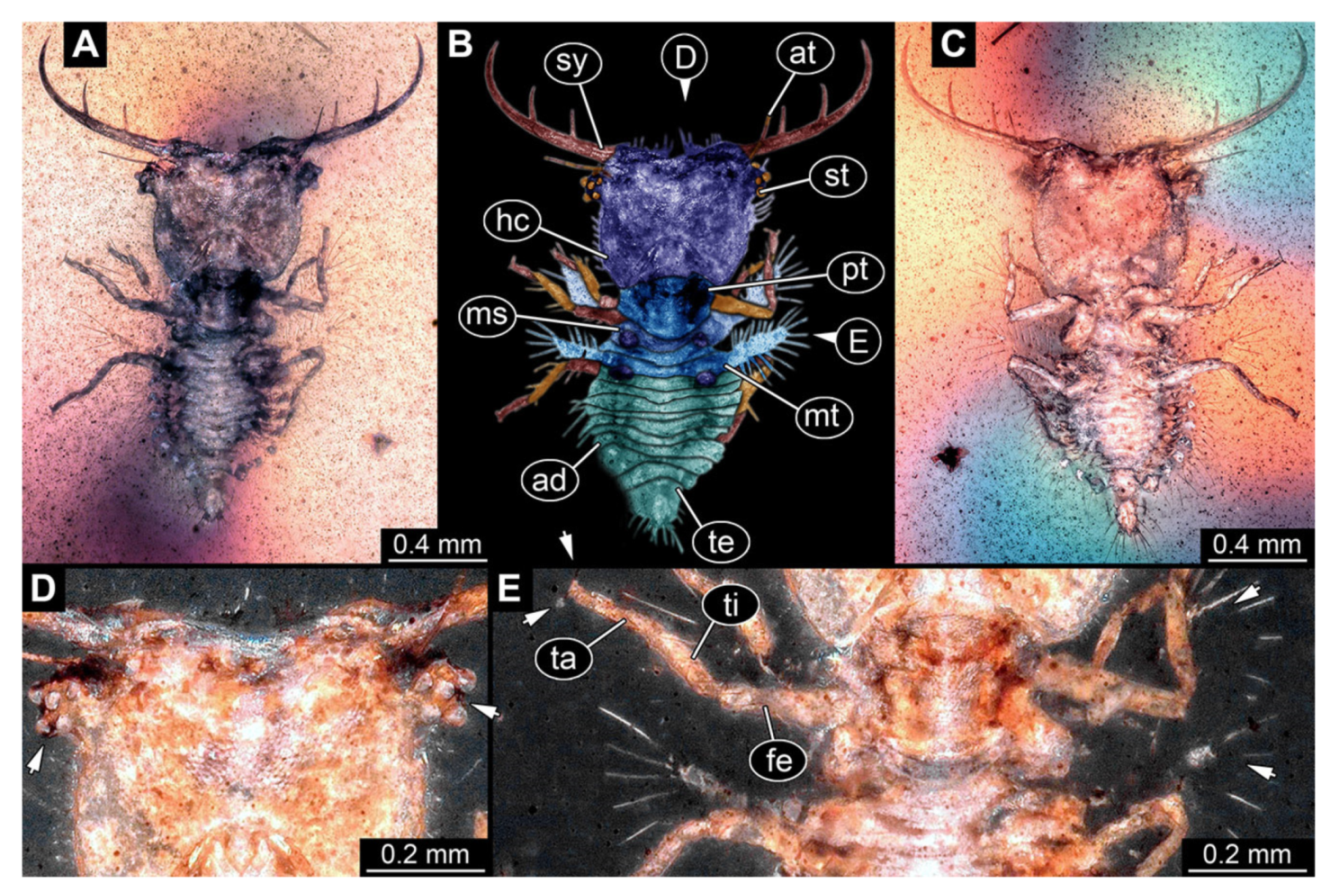
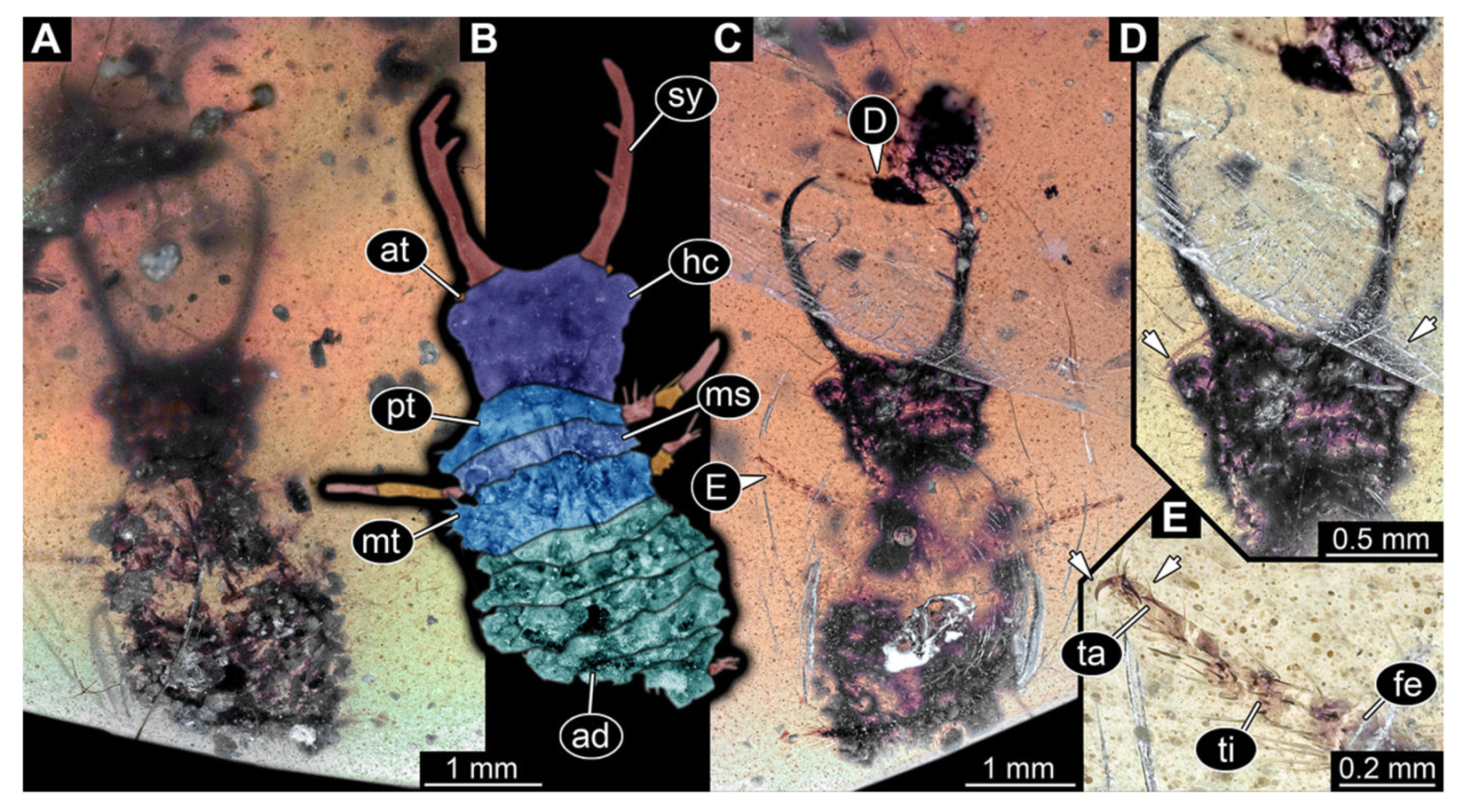
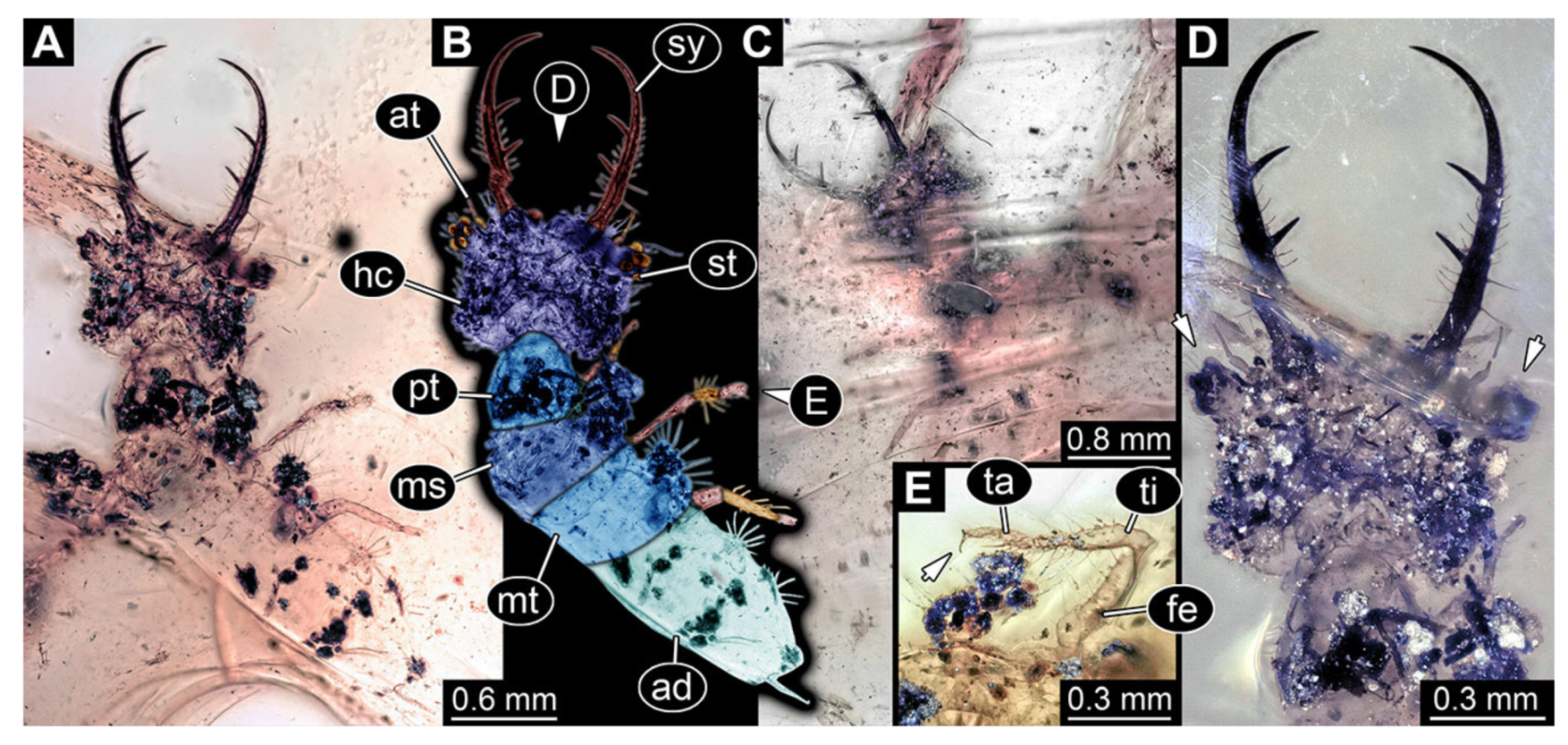
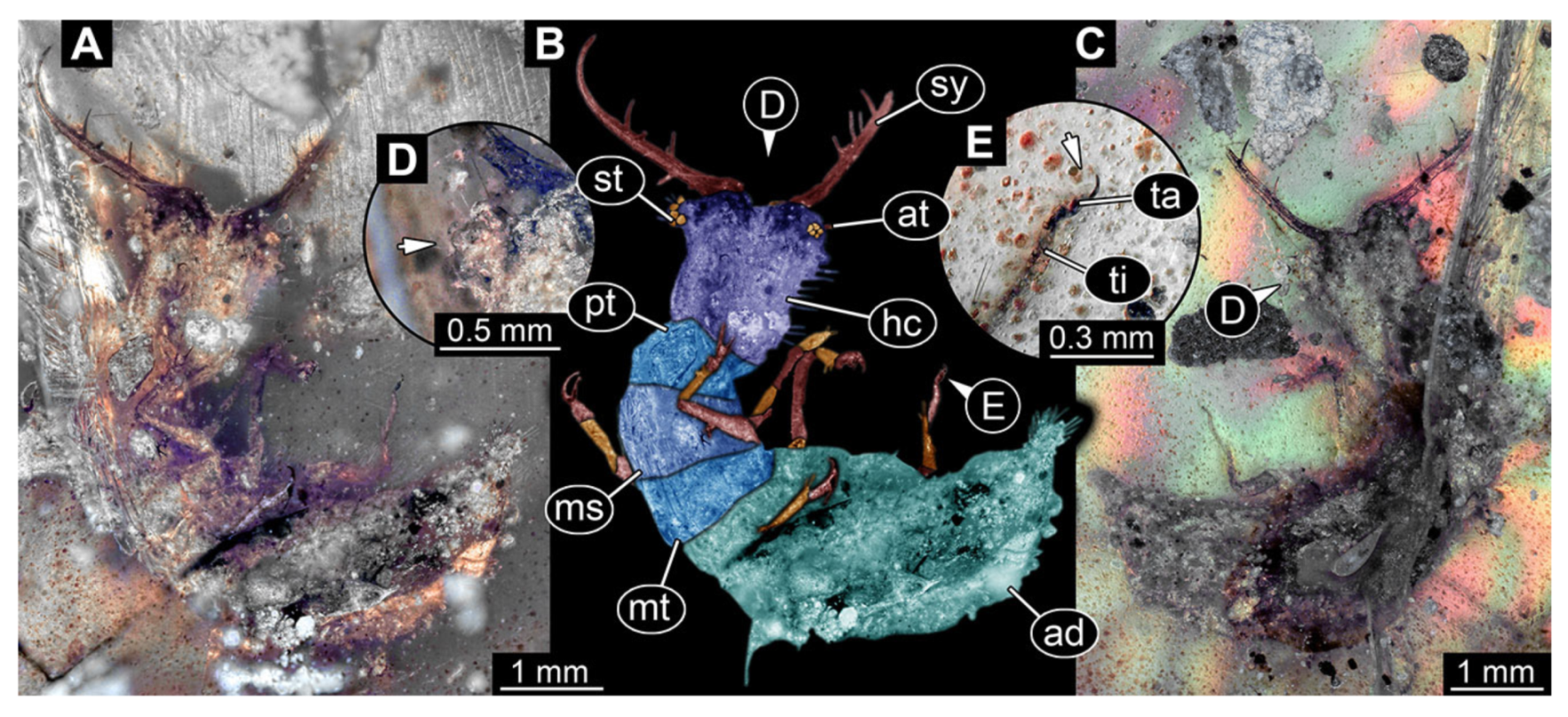
- Specimen 3213 (IGR.ARC-236.3), preserved in Cretaceous Charentese amber, France. Specimen was already reported in Wang et al. [47] (Figure 3E p. 4), but re-figured here to add some details. Only the head is preserved. Specimen well accessible in ventral (Figure 2A,B) and dorsal view (Figure 2C). Head capsule roughly rectangular in dorsal view, longer than wide; stylets longer than head capsule (Figure 2A–C). Stemmata on slight elevations on each side of head capsule (Figure 2A–C). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet bearing four major teeth, but no smaller teeth or robust setae. Short robust labial palps, with three elements each, antero-lateral, close to stylets. Preserved part of specimen is 3.14 mm.
- 3.
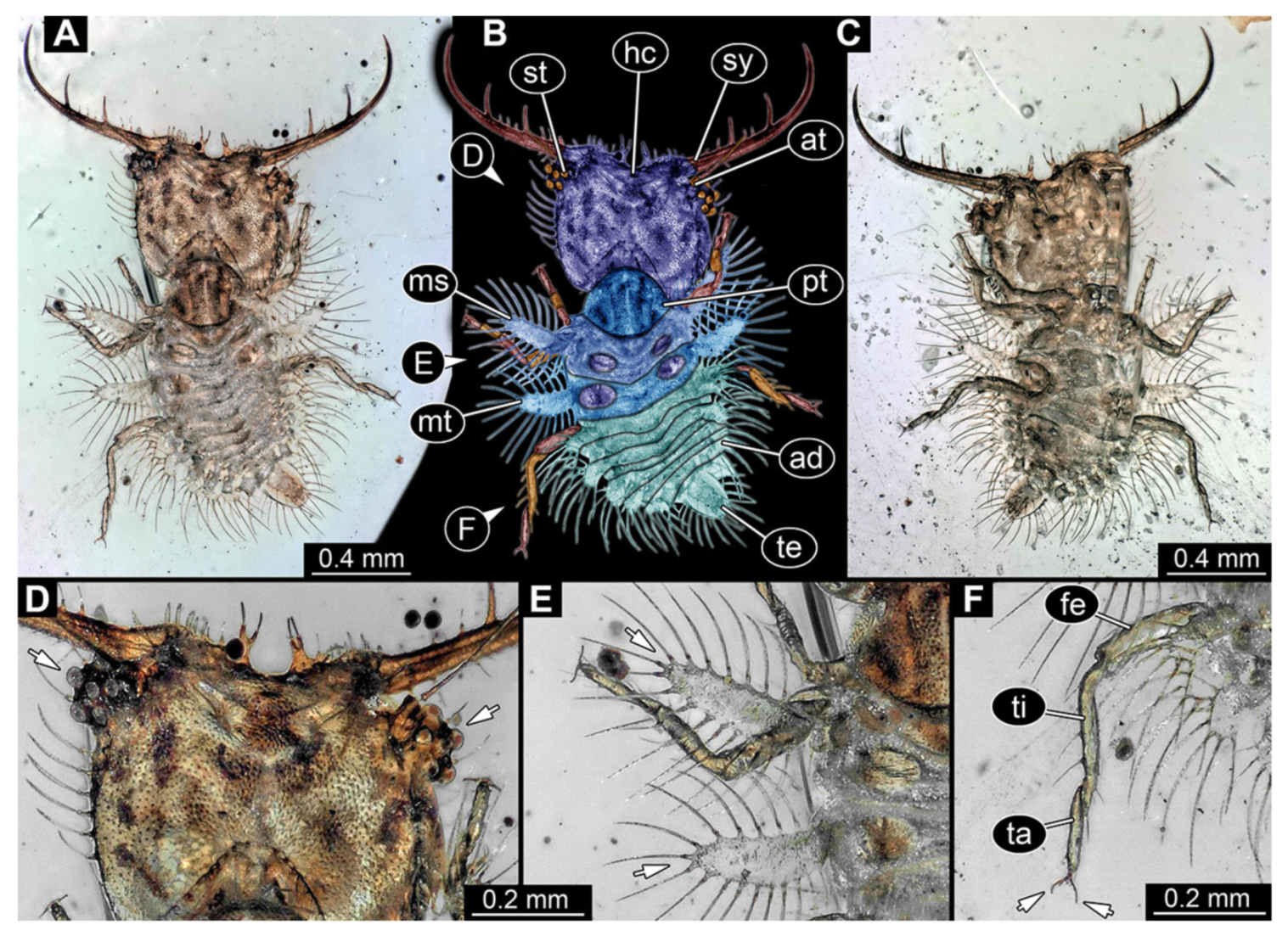
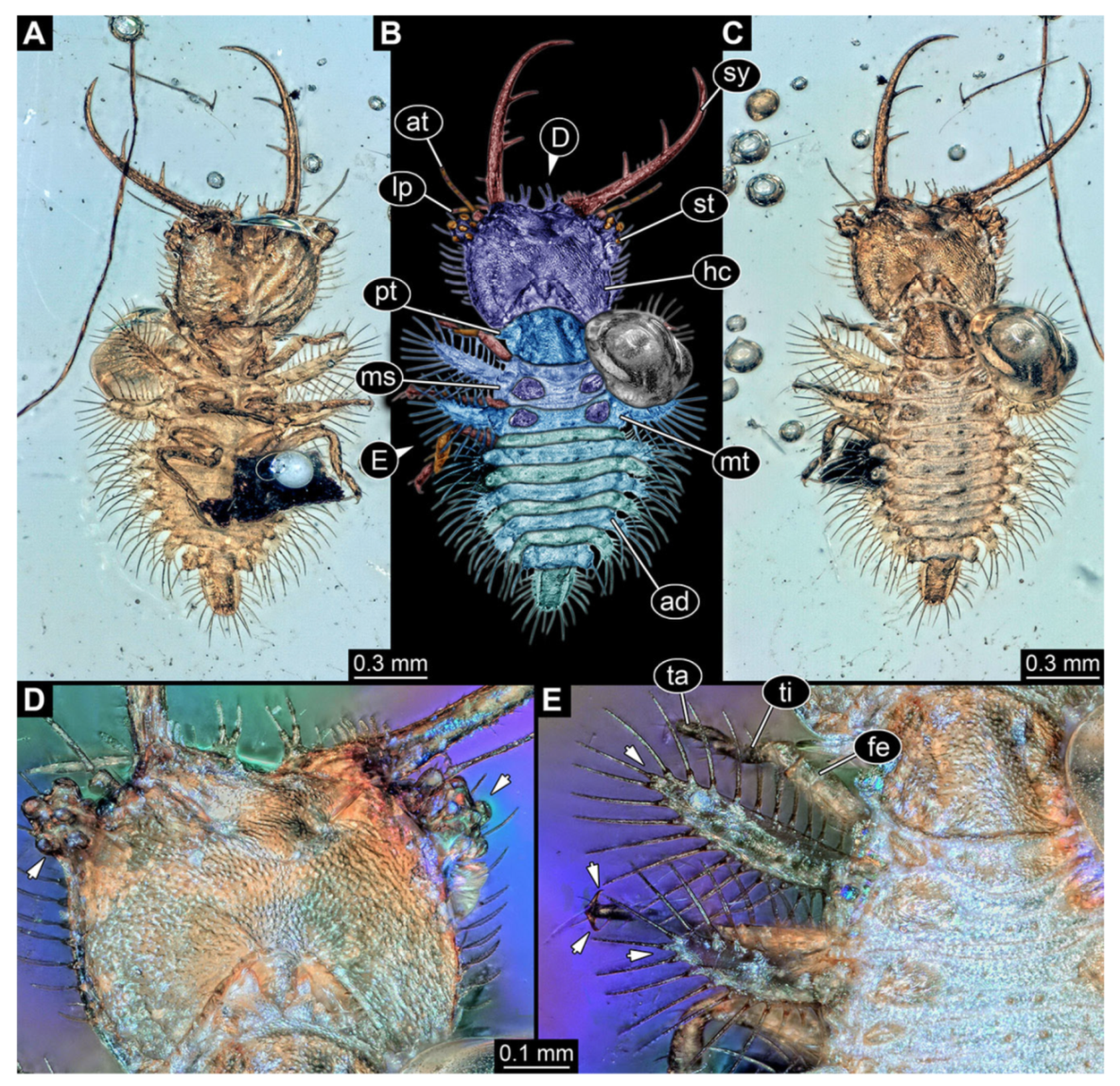
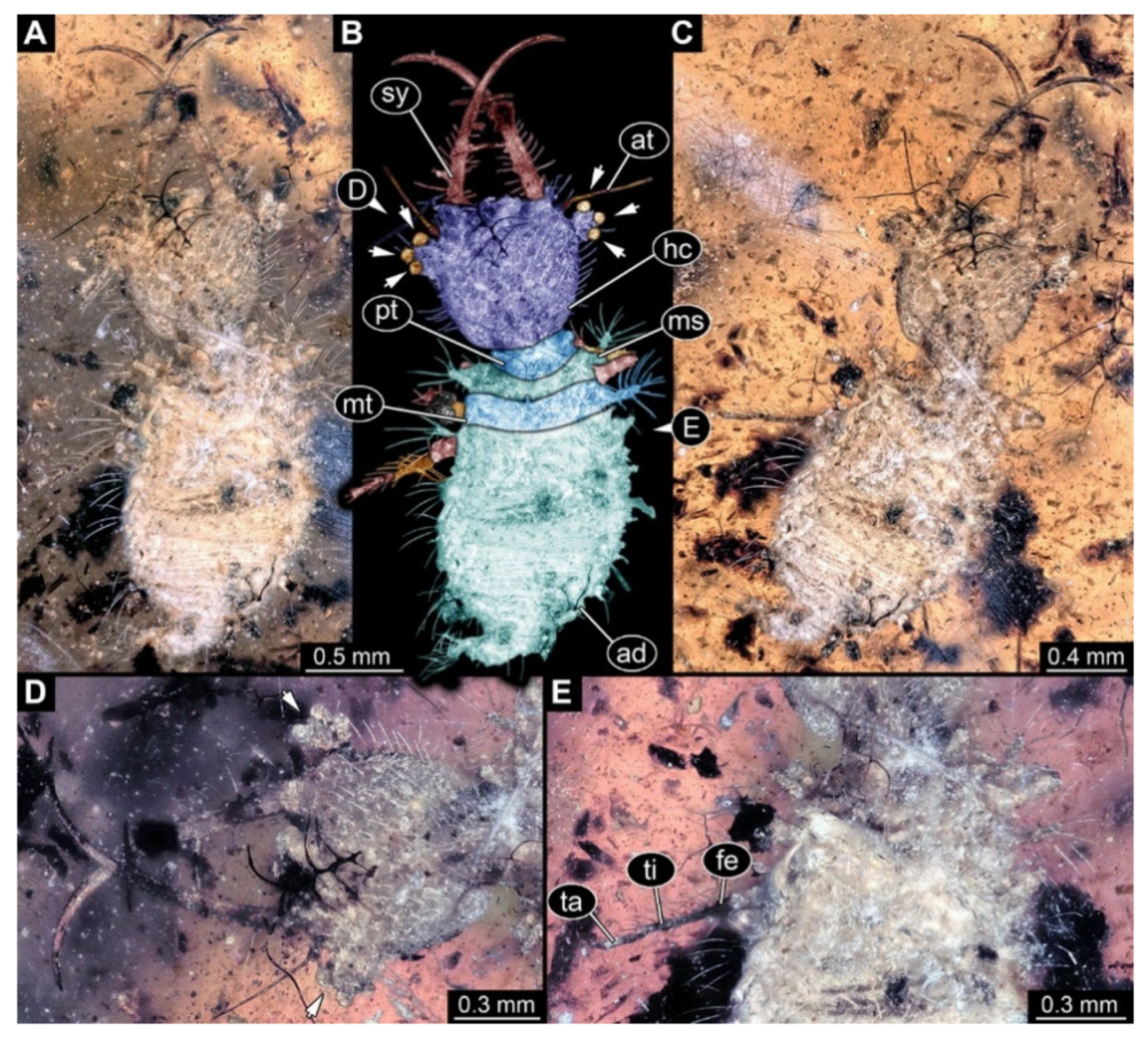
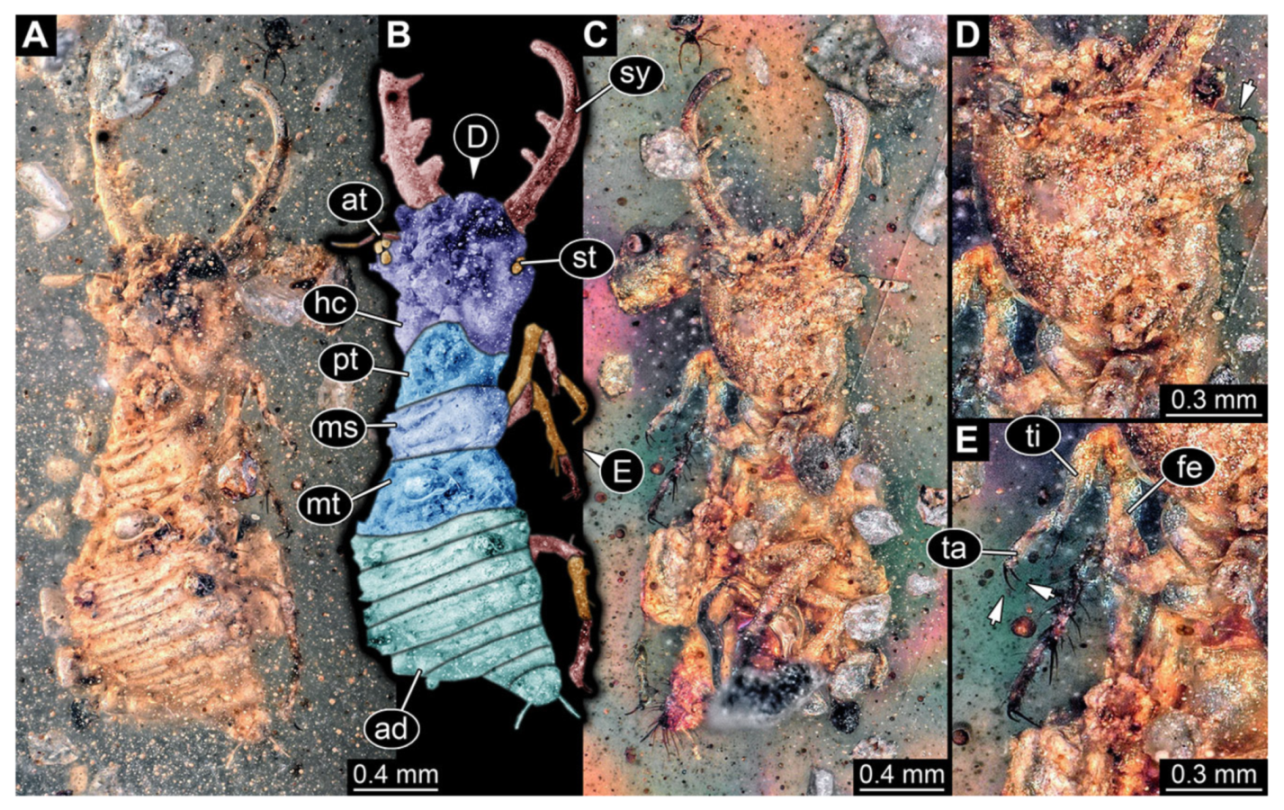
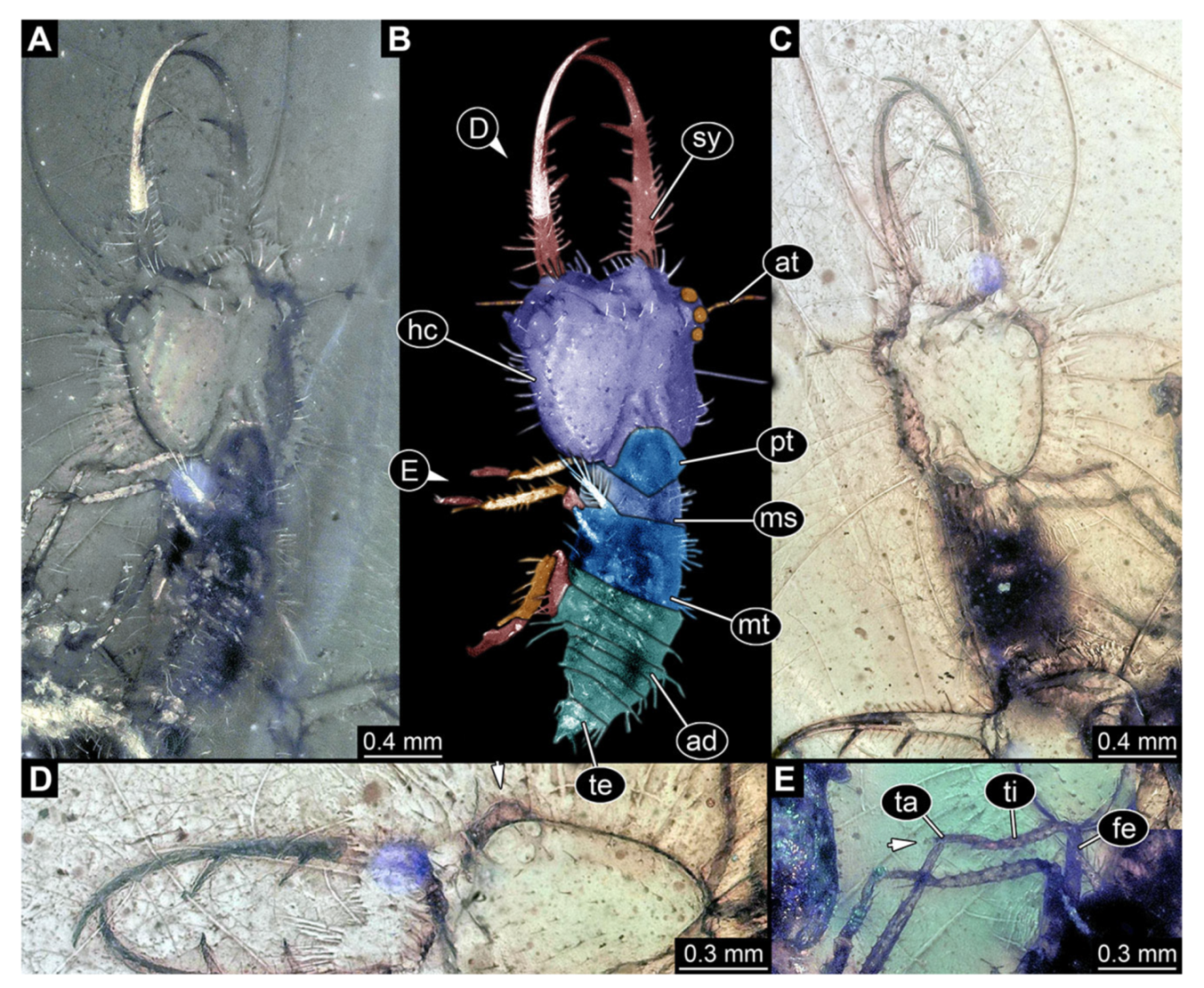
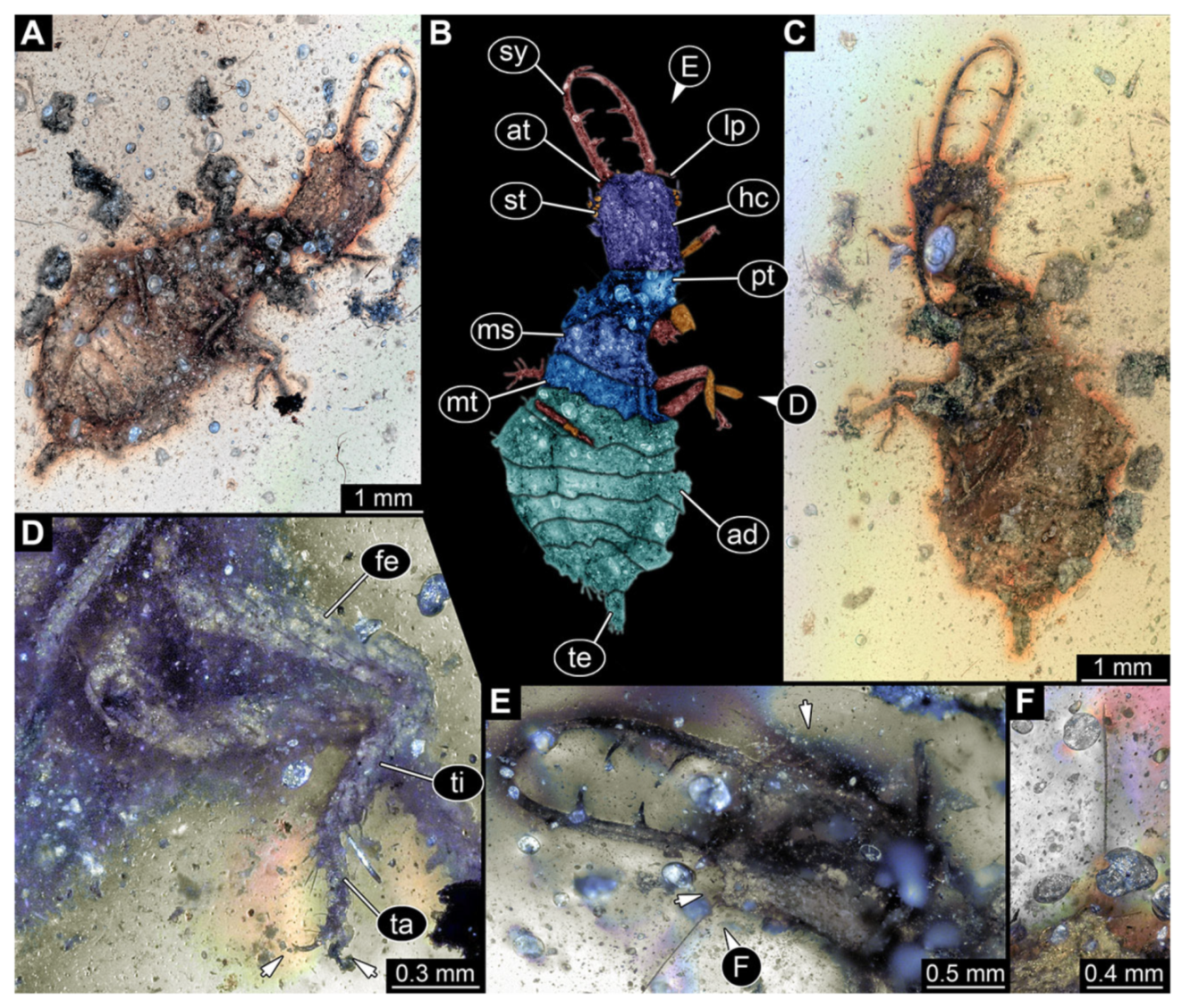
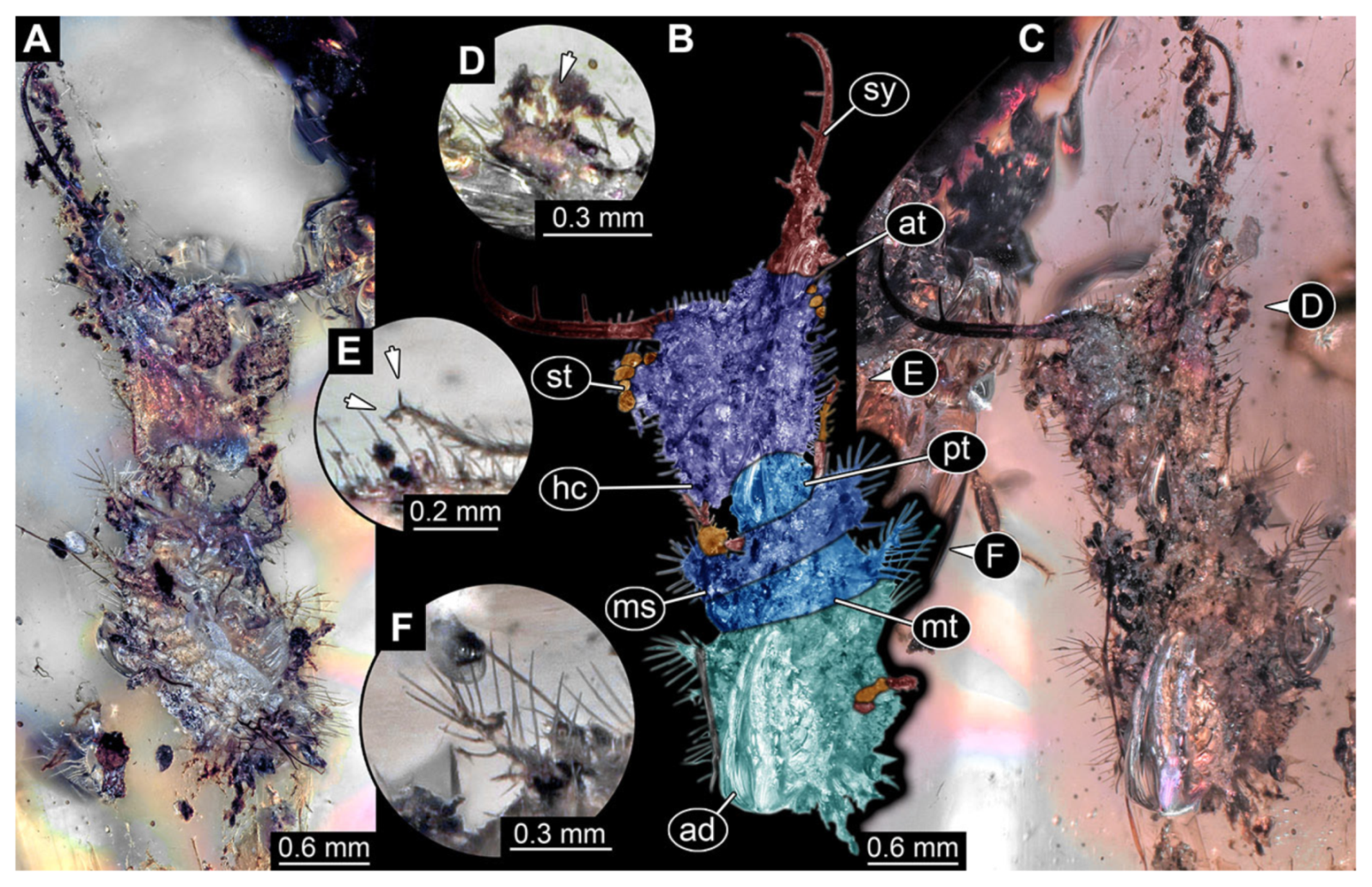
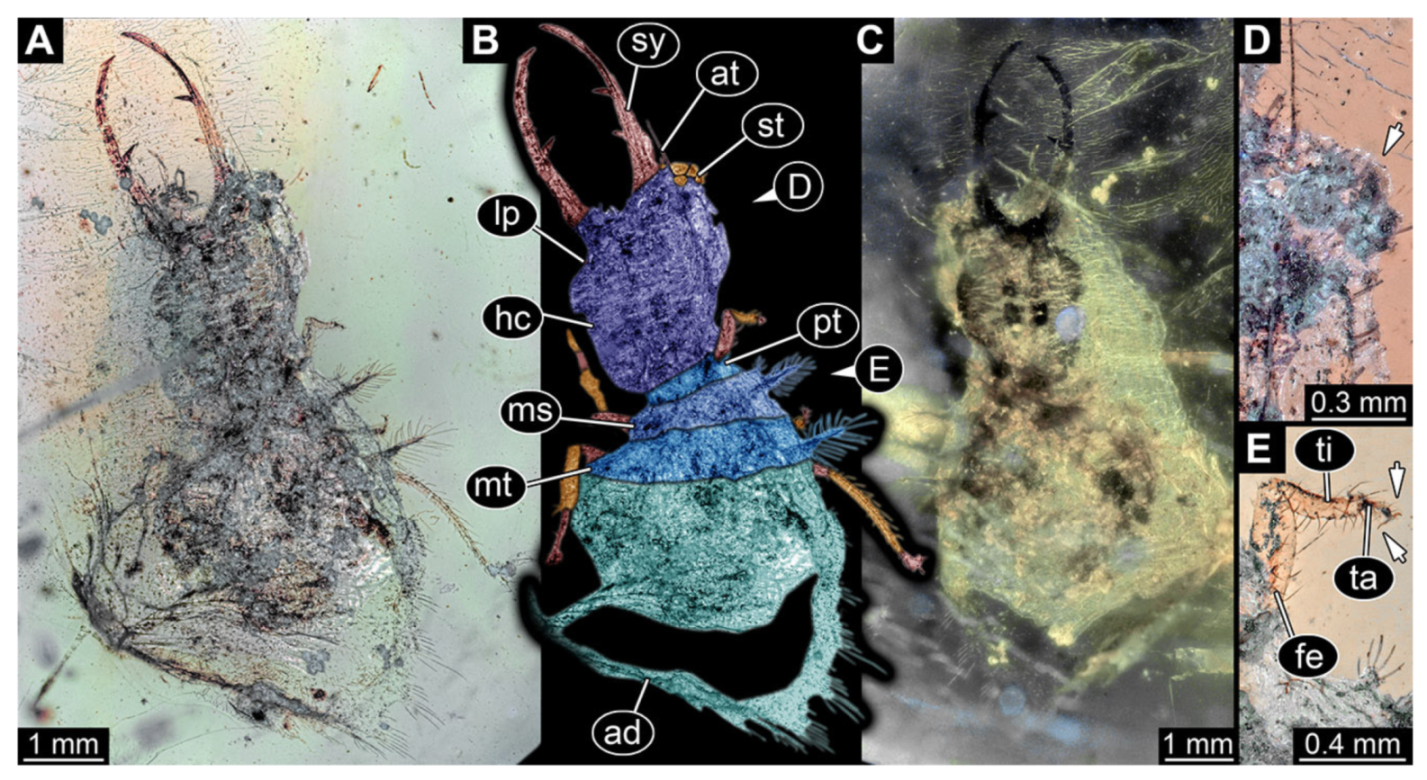
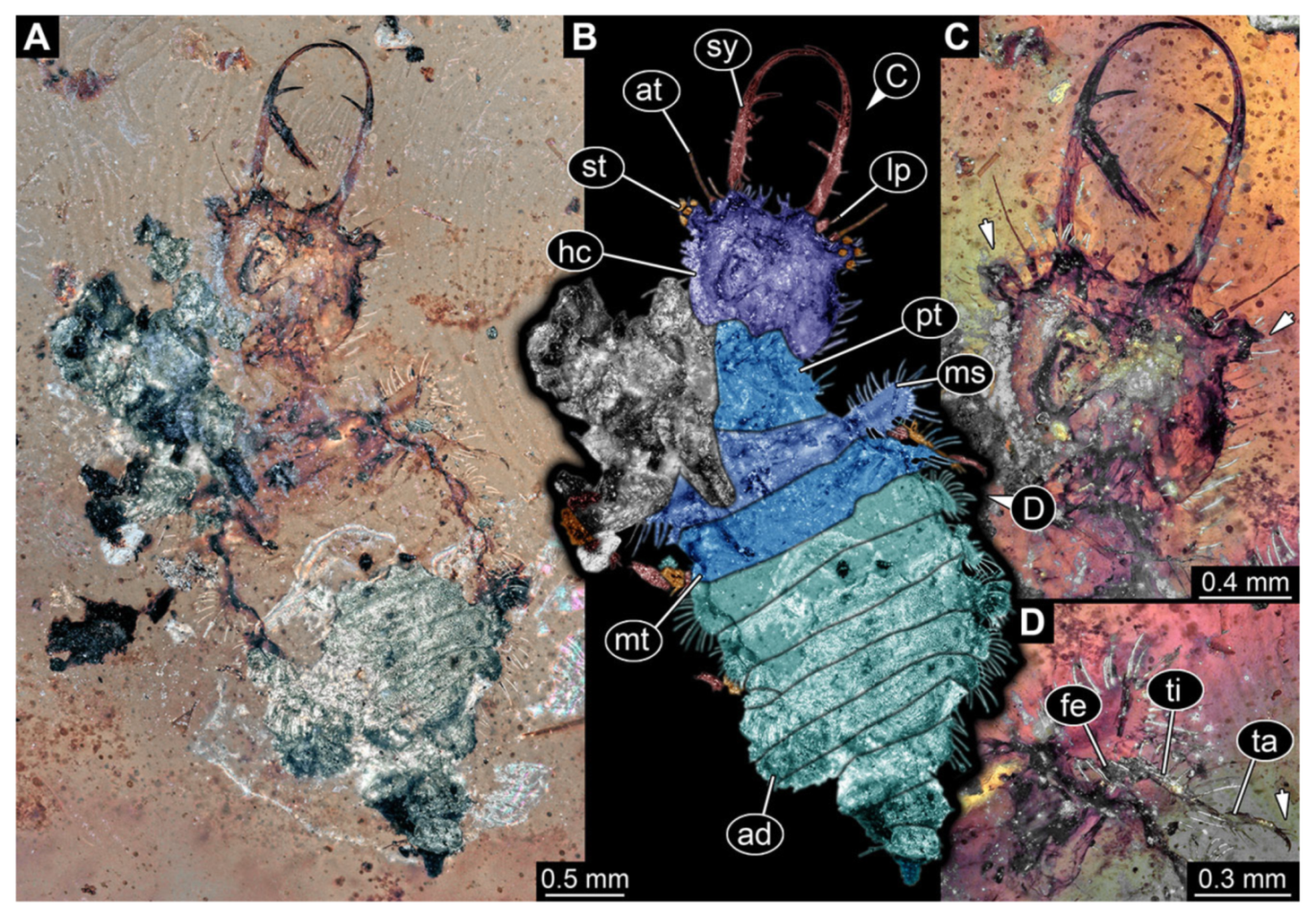
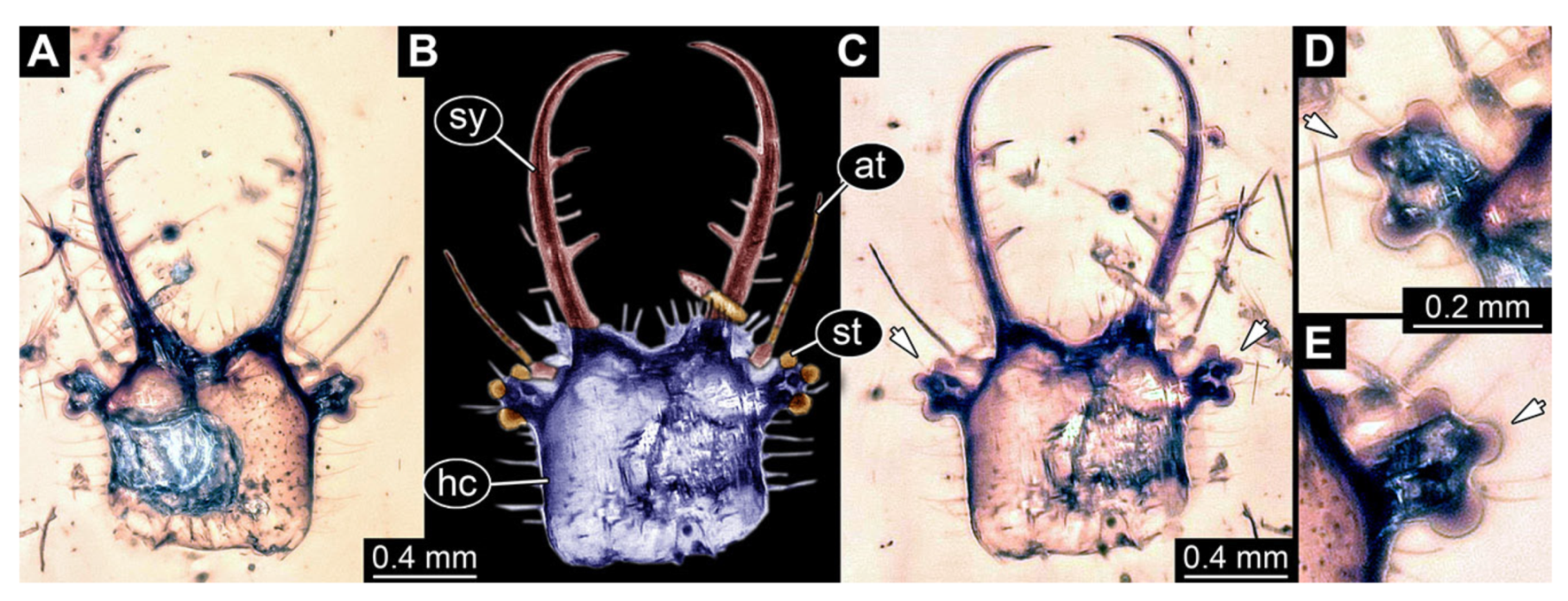
- Specimen 3214 (BUB 1430), preserved in Cretaceous Kachin amber, Myanmar. Specimen very complete, well accessible in dorsal (Figure 3A,B) and ventral view (Figure 3C). Head capsule roughly square-shaped in dorsal view, only slightly wider than long; stylets longer than head capsule (Figure 3A–C). Anterior rim with deeply forked spine, one on each side of midline (Figure 3D). Stemmata on prominent protrusions on each side of head capsule (Figure 3D). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 3A–C). No palp apparent. Pronotum cap-like. Mesothorax and metathorax with very prominent dorso-lateral protrusions bearing numerous setae (Figure 3E). Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated, also seen in leg three (Figure 3F). Distal tarsus with pair of claws, no empodium apparent. Anterior abdomen segments also with protrusions, but smaller ones. Trunk end is elongate, longer than wide in dorsal view, with numerous setae. Total length of specimen is 2.15 mm.
- 4.
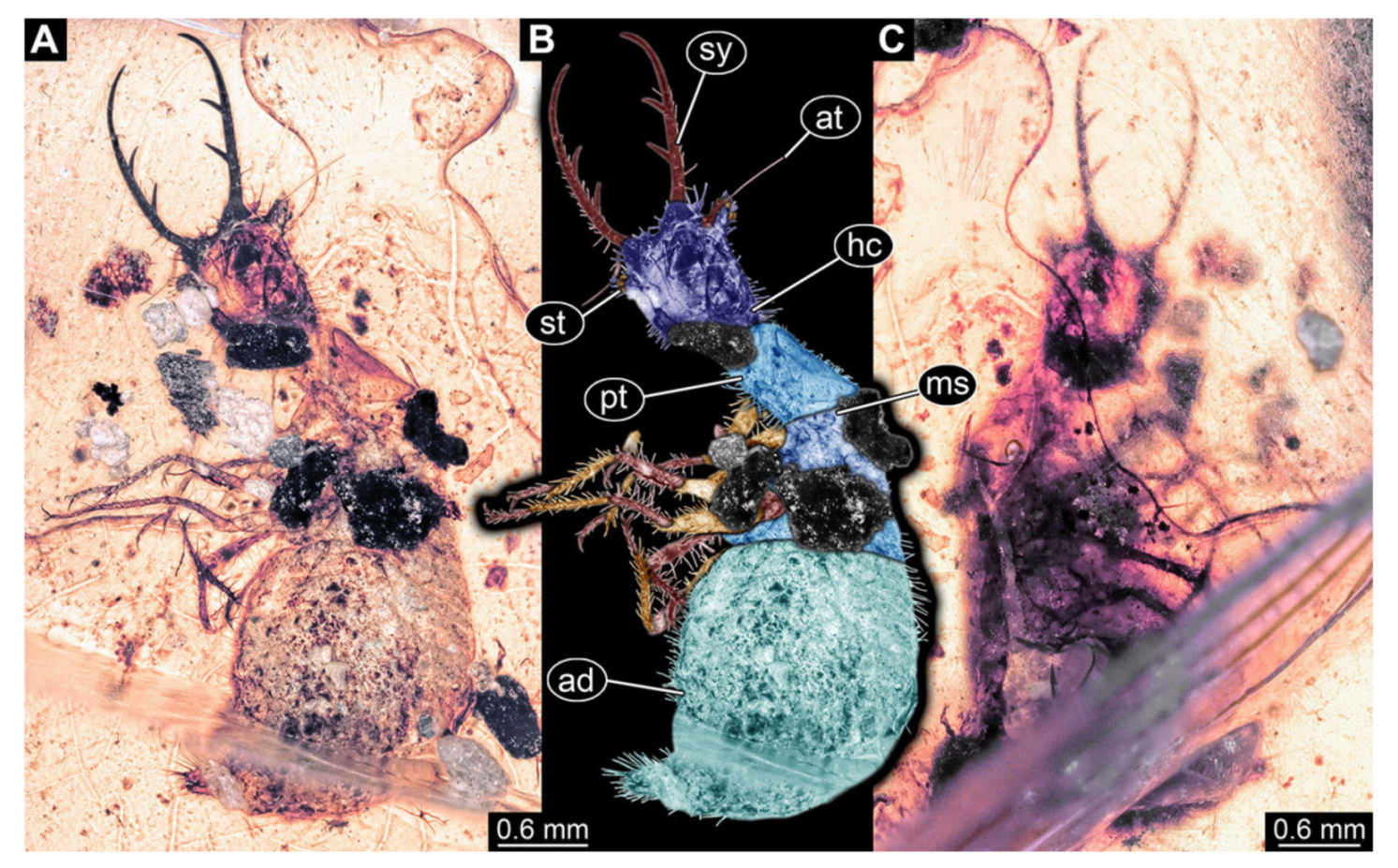
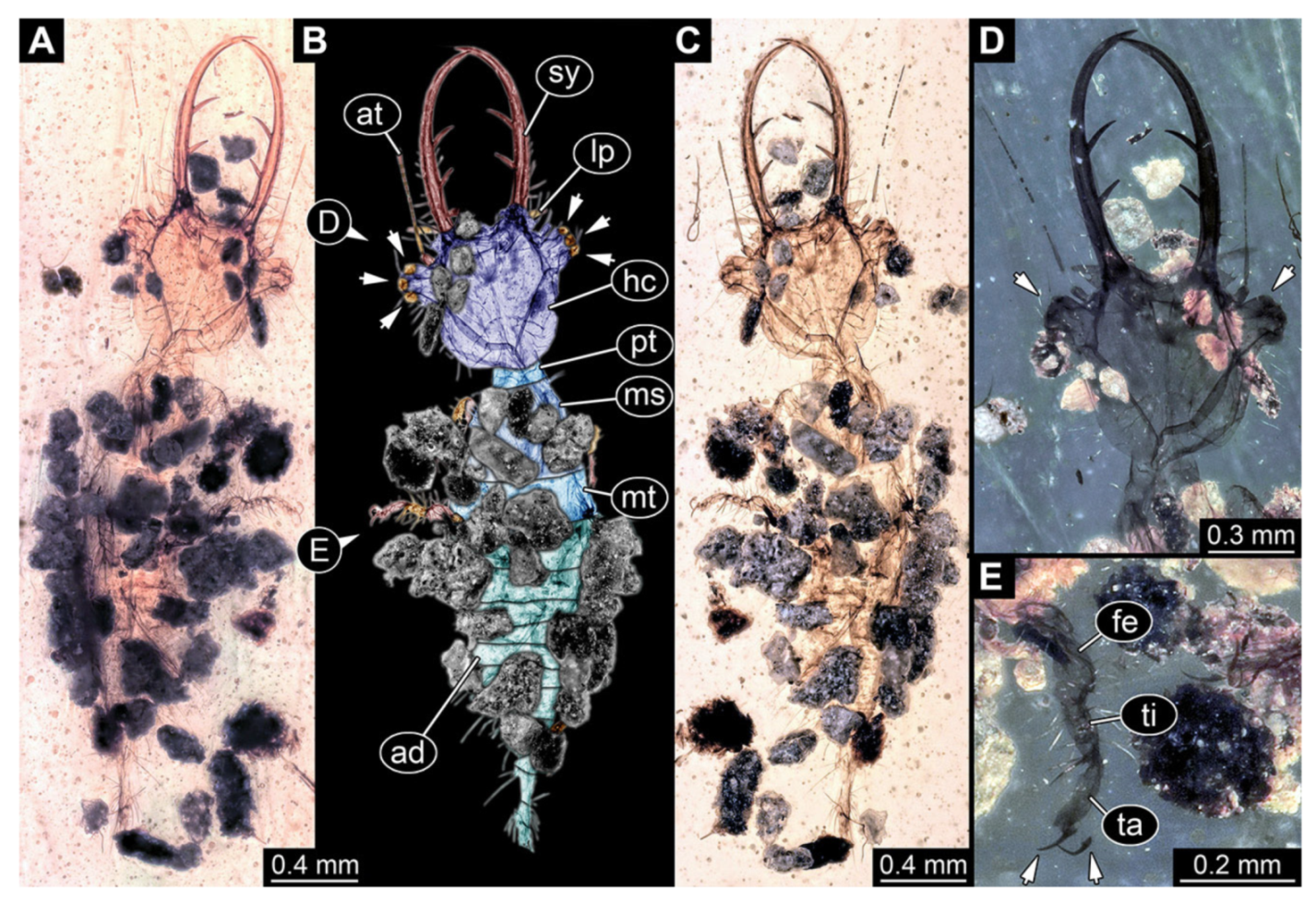
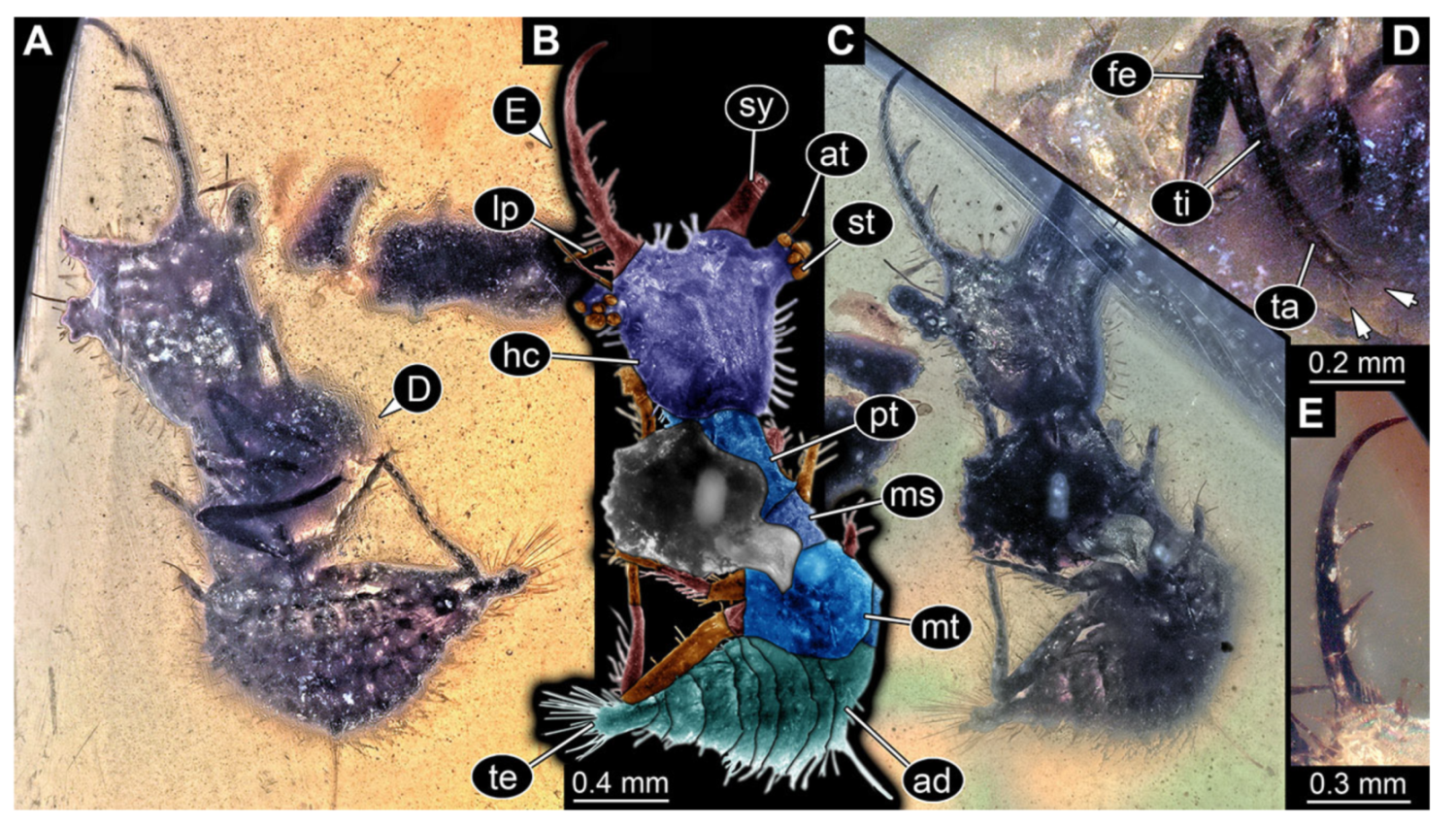
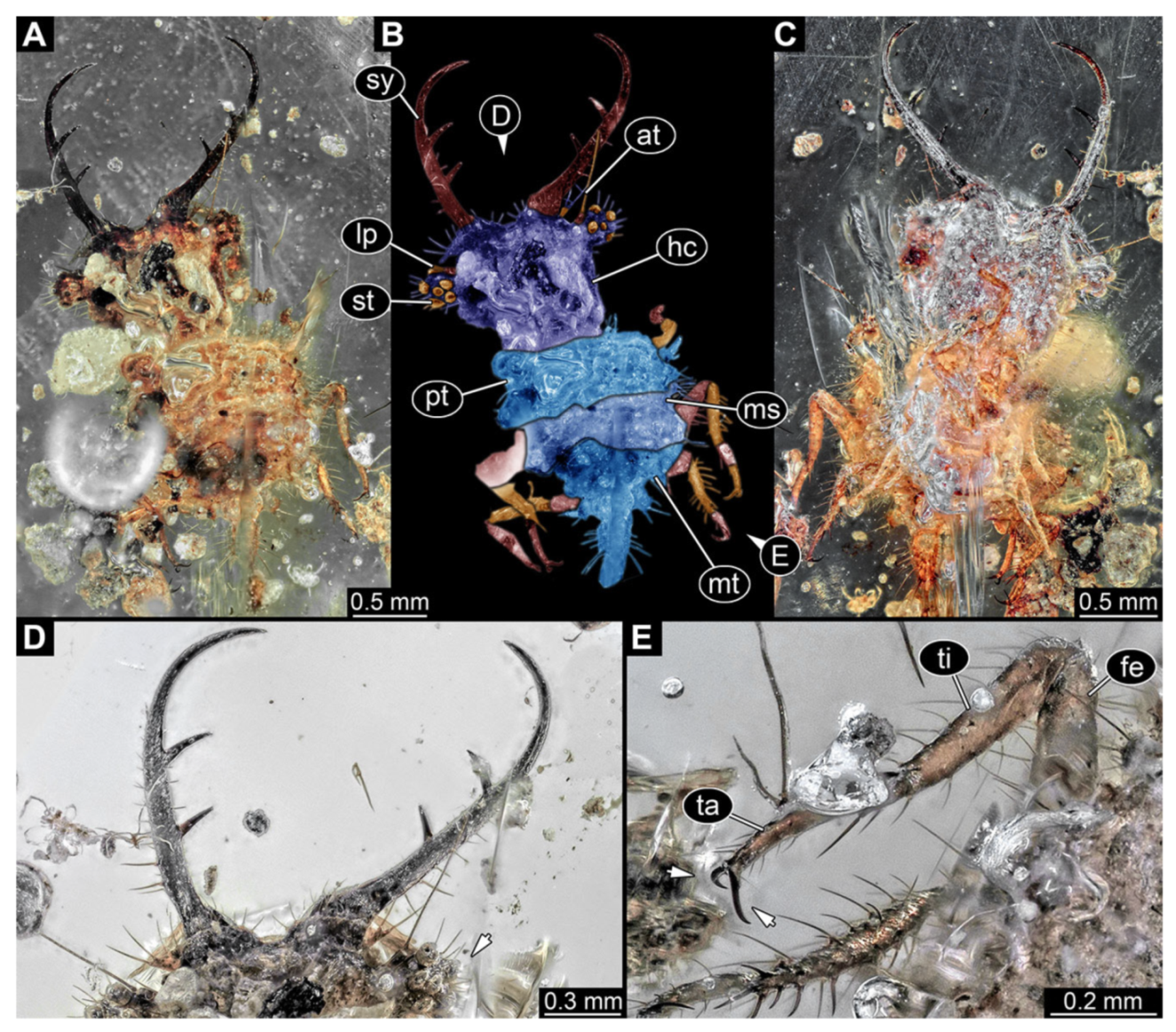
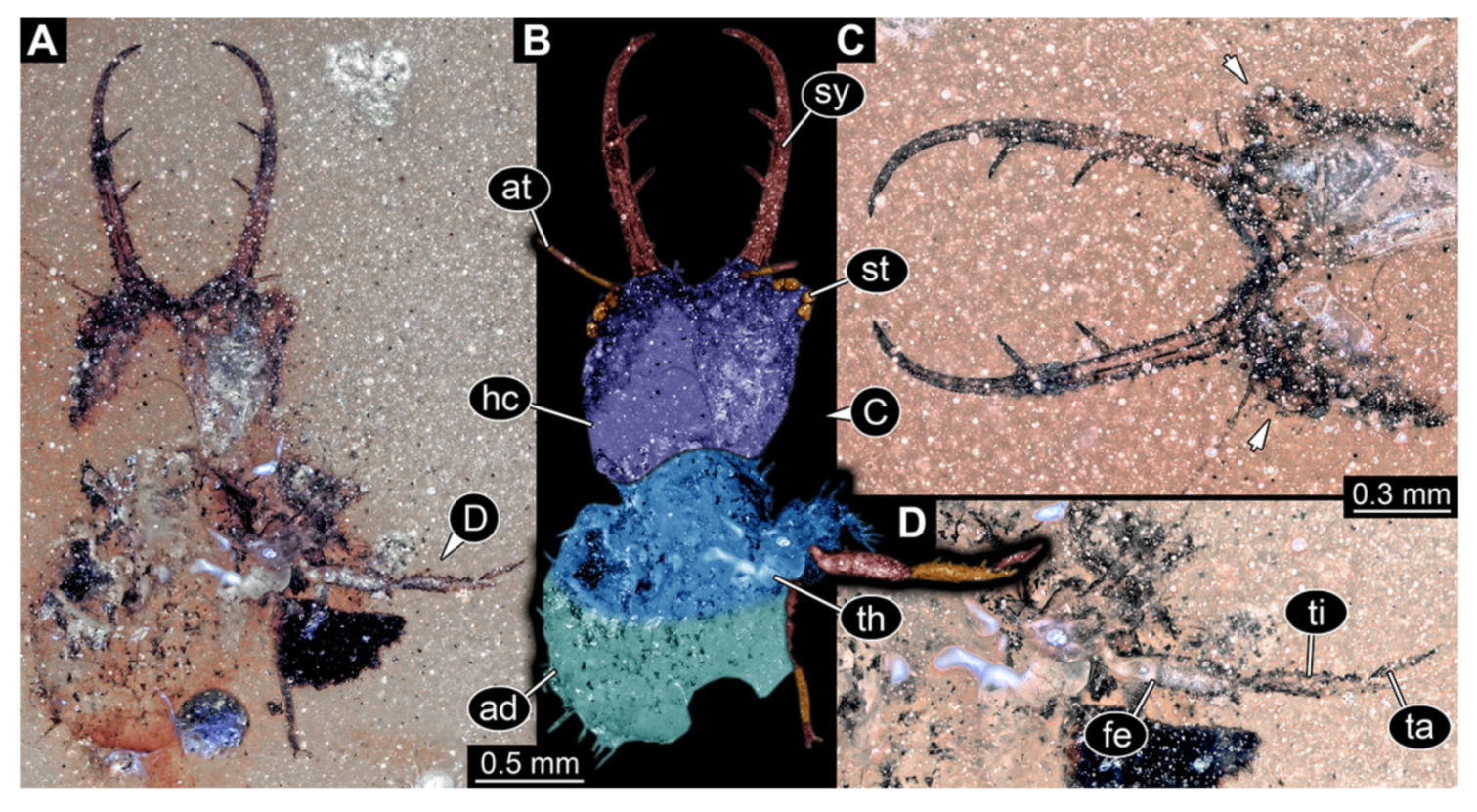
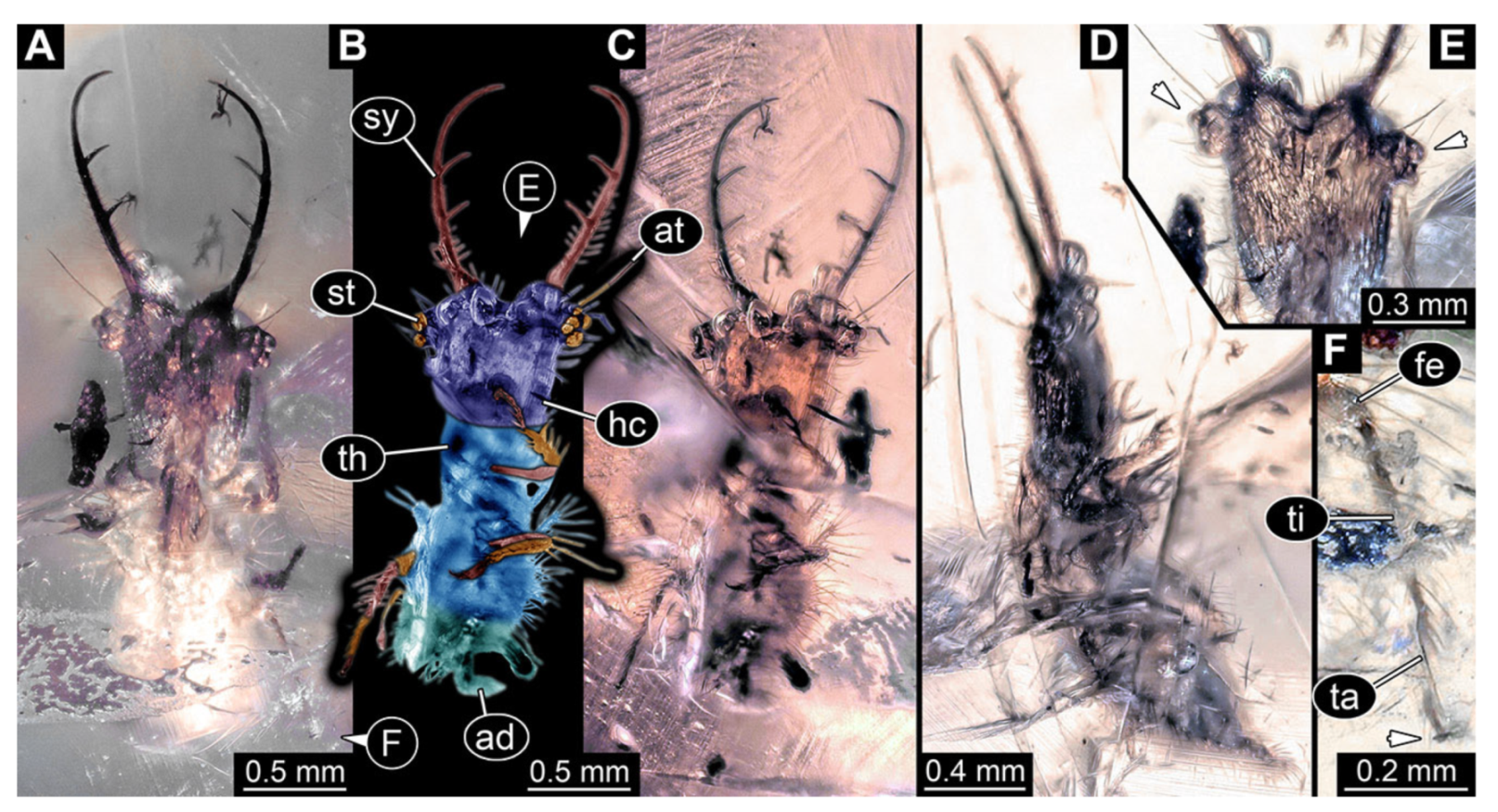
- Specimen 3215 (BUB 2537), preserved in Cretaceous Kachin amber, Myanmar. Specimen is well accessible in ventral view (Figure 4A), dorsally partially concealed by air bubble (Figure 4B,C). Head capsule roughly square-shaped in dorsal view, only slightly wider than long; stylets longer than head capsule (Figure 4A–C). Anterior rim with deeply forked spine, one on each side of midline (Figure 4D). Stemmata on prominent protrusions on each side of head capsule (Figure 4D). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 4A–C). Labial palp is short, robust. Pronotum cap-like. Mesothorax and metathorax with very prominent dorso-lateral protrusions bearing numerous setae (Figure 4E). Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 4E). Distal tarsus with a pair of claws, no empodium apparent. Anterior abdomen segments also with protrusions, but smaller ones. Trunk end is elongated, longer than wide in dorsal view, with numerous setae. Total length of specimen is 2.42 mm.
- 5.
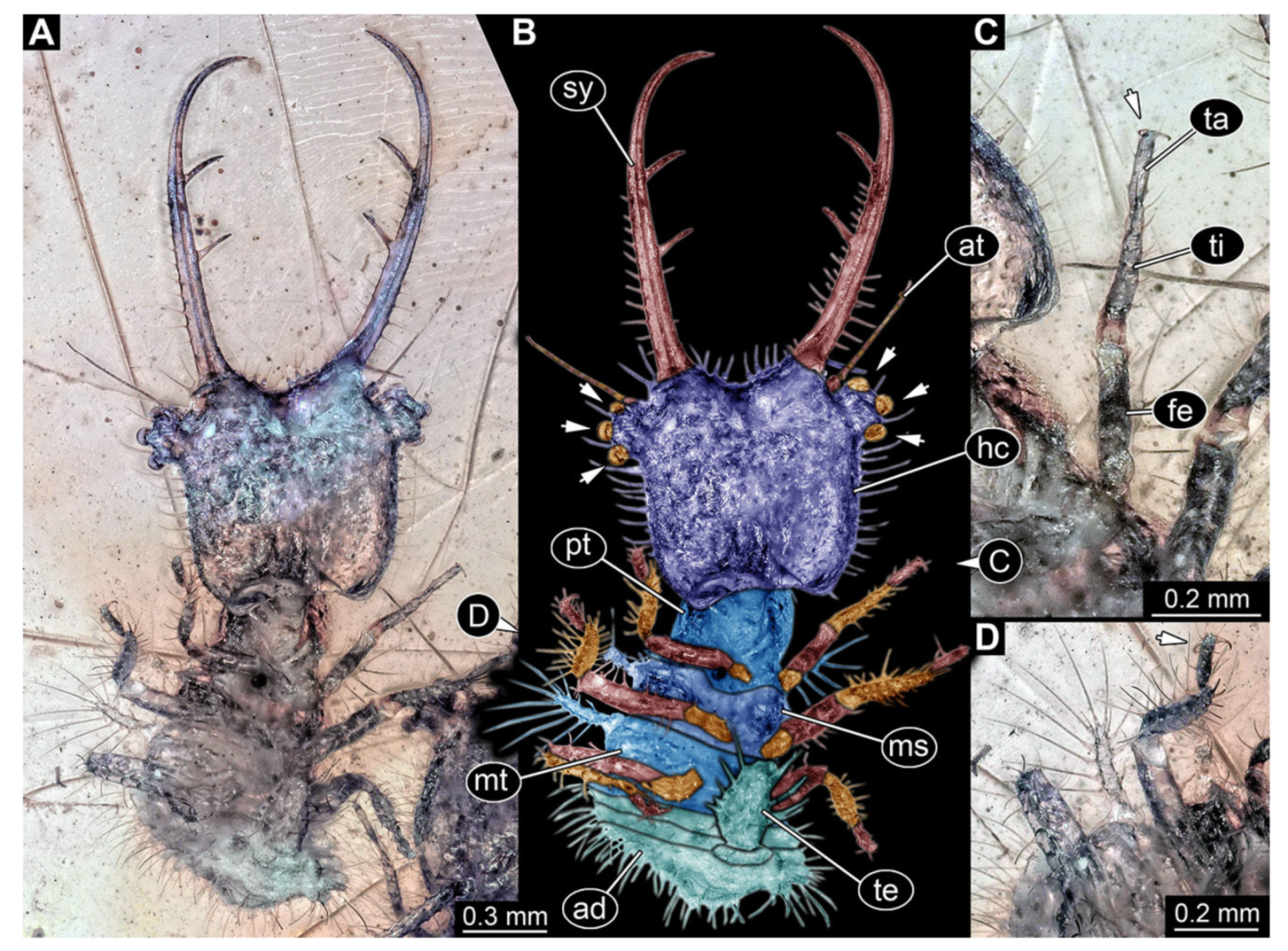
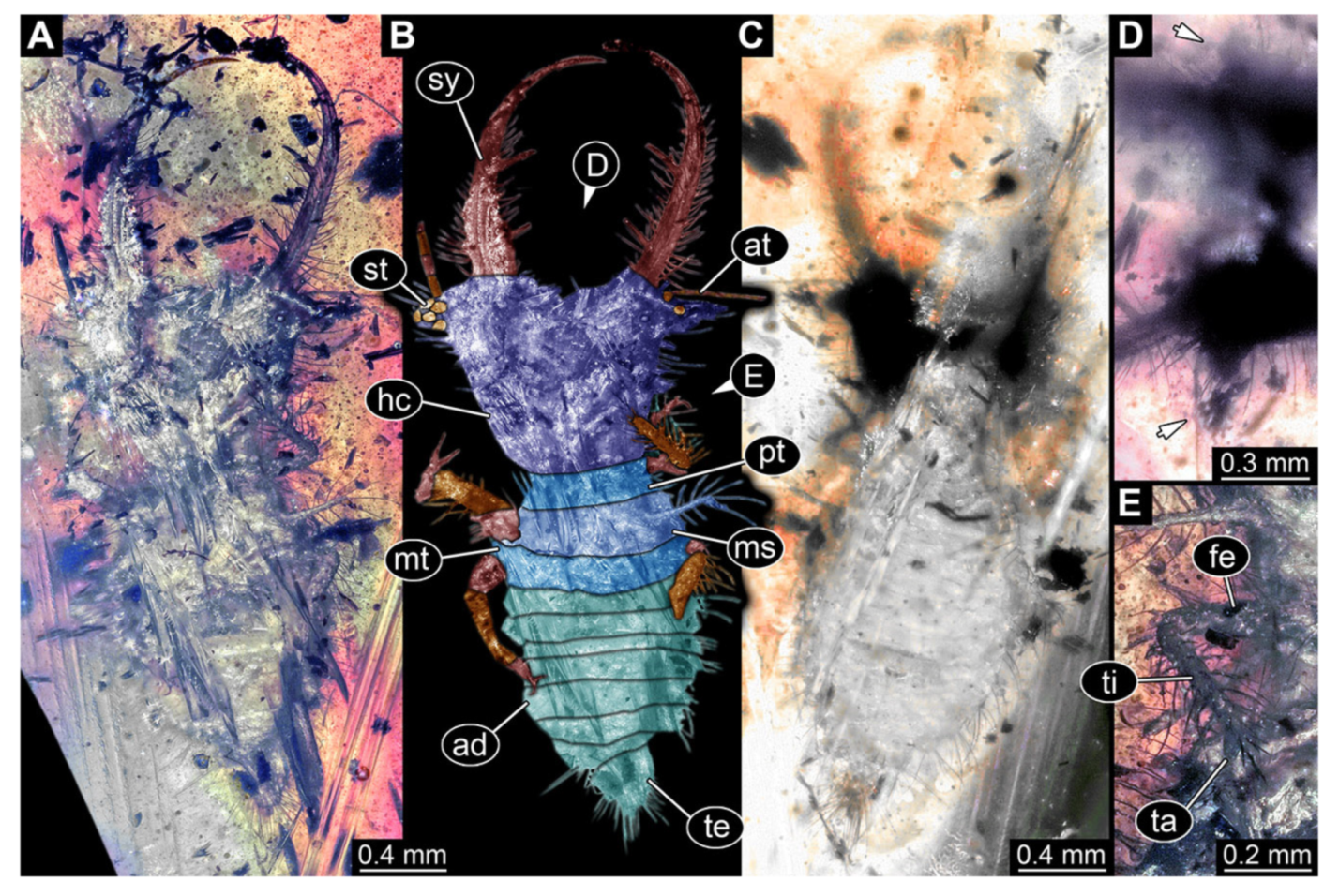
- Specimen 3216 (BUB 3343), preserved in Cretaceous Kachin amber, Myanmar. Anterior region of specimen is well accessible in ventral (Figure 5A,B) and dorsal view (Figure 5C); posterior part of body folded forward, not well accessible. Head capsule roughly square-shaped in dorsal view, only slightly wider than long; stylets longer than head capsule (Figure 5A–C). Anterior rim with simple setae (Figure 5D). Stemmata on prominent protrusions on each side of head capsule (Figure 5D). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 5A–C). Labial palp is short, robust. Pronotum cap-like. Mesothorax and metathorax with very prominent dorso-lateral protrusions bearing numerous setae (Figure 5F). Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 5E). Distal tarsus with a pair of claws, no empodium apparent. Anterior abdomen segments also with protrusions, but smaller ones. Trunk end is elongated, longer than wide in dorsal view, with numerous setae. Total length of specimen is 2.60 mm.
- 6.
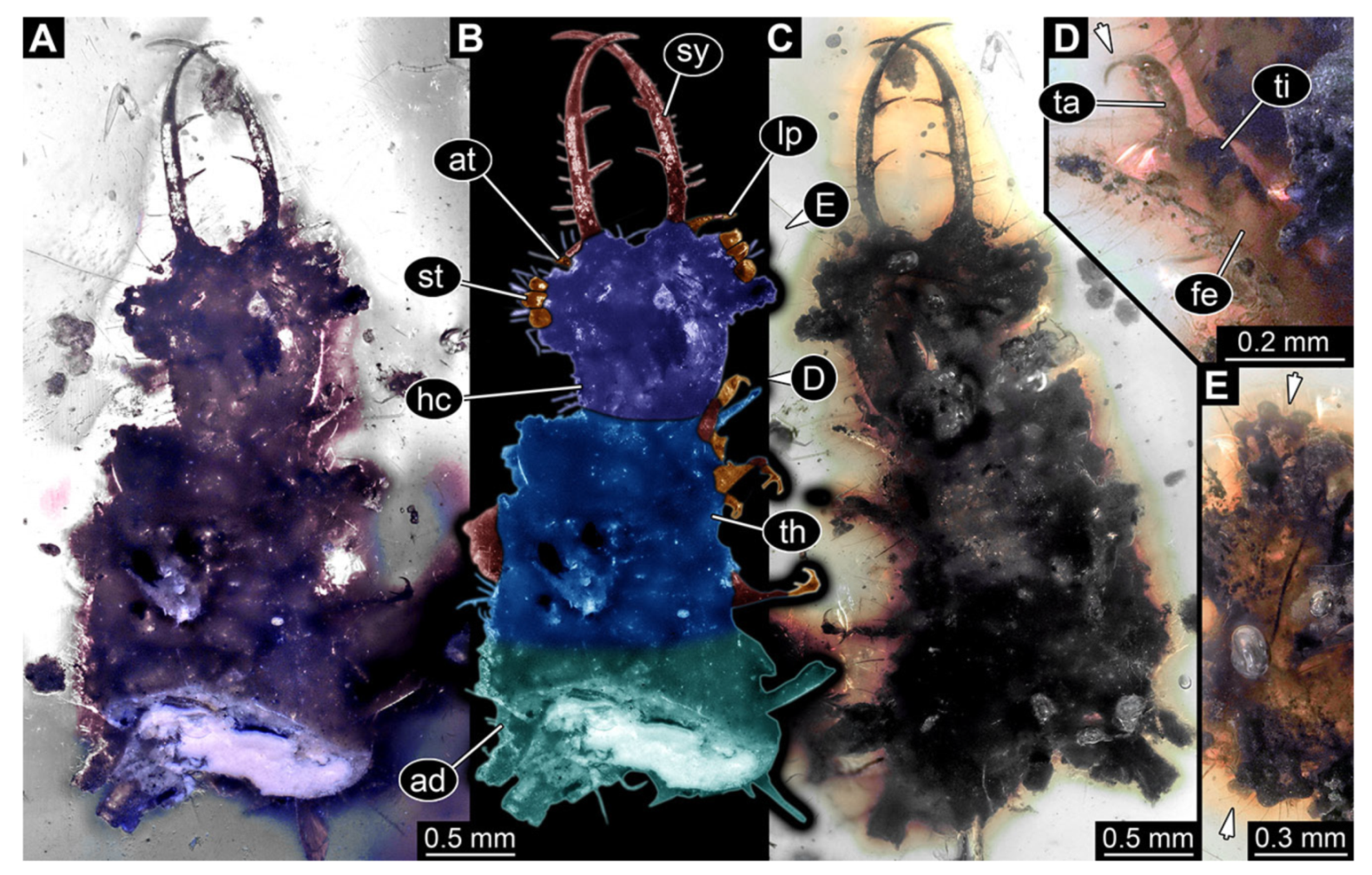
- Specimen 3217 (BUB 3348), preserved in Cretaceous Kachin amber, Myanmar. Specimen is well accessible in ventral (Figure 6A) and dorsal view (Figure 6B,C), yet partly concealed by debris, apparently attached to body forming camouflaging cloak. Head capsule roughly square-shaped in dorsal view; stylets longer than head capsule (Figure 6A–C). Stemmata on prominent protrusions on each side of head capsule (Figure 6D,E). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 6A–C). No palp apparent. Pronotum cap-like. Mesothorax and metathorax with prominent dorso-lateral protrusions bearing numerous setae (Figure 6F). Thorax segments ventrally with locomotory appendages. Legs distally with two prominent elements (tibia, tarsus), with the tibia and tarsus well separated (Figure 6G). Distal tarsus with a pair of claws, no empodium apparent. Details of abdomen not well accessible due to debris. Total length of specimen is 9.00 mm.
- 7.
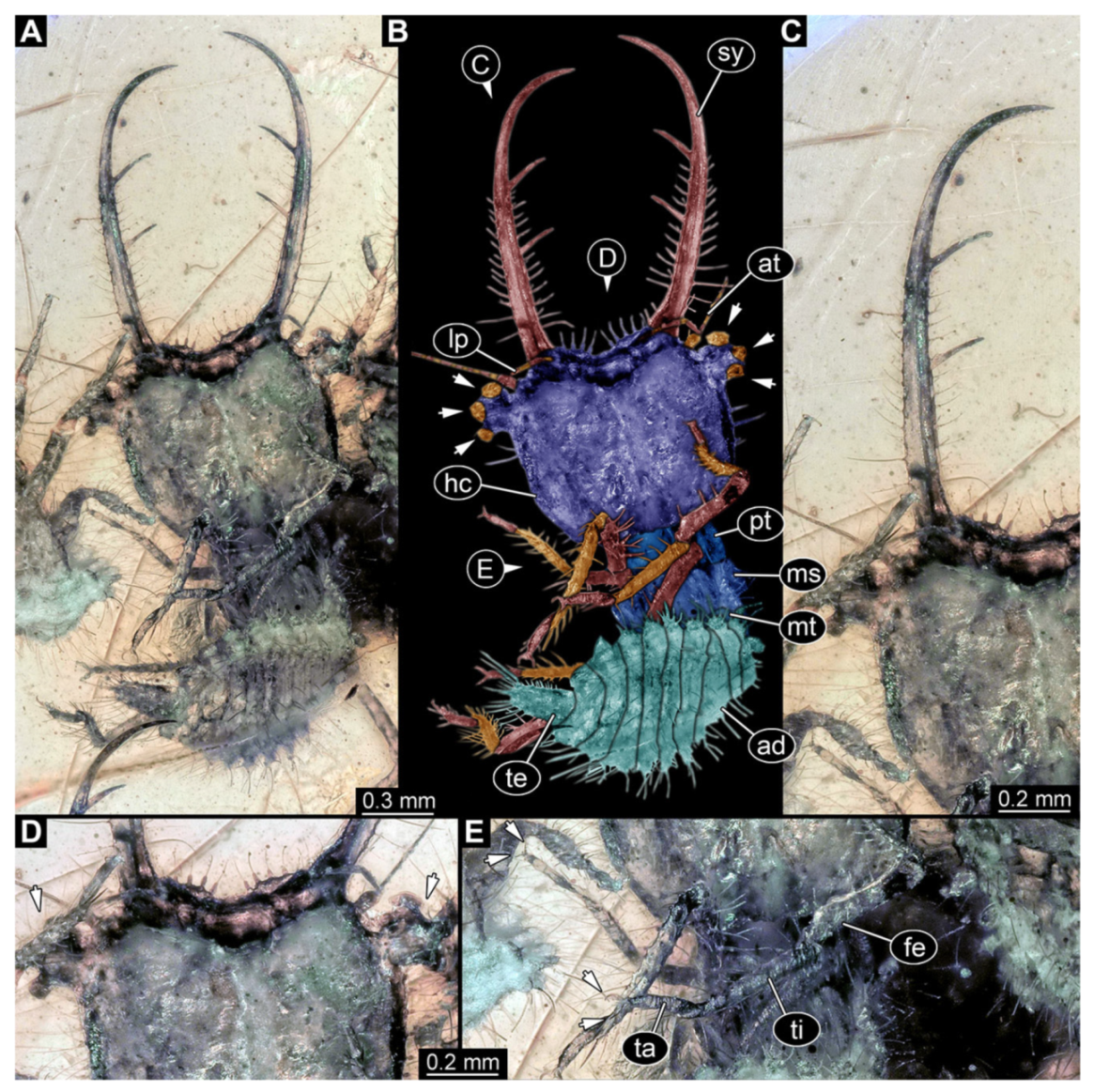
- Specimen 3218 (BUB 3378), preserved in Cretaceous Kachin amber, Myanmar. Specimen is well accessible in dorsal view (Figure 7A–C). Head capsule roughly trapezium-shaped in dorsal view, anterior wider than posterior; stylets longer than head capsule (Figure 7D). Stemmata on prominent protrusions on each side of head capsule (Figure 7D). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 7D). No palp apparent. Pronotum cap-like. Mesothorax and metathorax with prominent dorso-lateral protrusions bearing numerous setae (Figure 7B). Thorax segments ventrally with locomotory appendages. Legs distally with two prominent elements (tibia, tarsus), with the tibia and tarsus well separated (Figure 7E). Anterior abdomen segments also with protrusions, but smaller ones. Trunk end is elongated, longer than wide in dorsal view, with numerous setae. Total length of specimen is 3.26 mm.
- 8.
- Specimen 3219 (BUB 3380), preserved in Cretaceous Kachin amber, Myanmar. Specimen is accessible in oblique dorsal (Figure 8A,B) and oblique ventral view (Figure 8C), but partly concealed by debris. Head capsule roughly trapezium-shaped in dorsal view, anterior slightly wider than posterior; stylets longer than head capsule (Figure 8). Stemmata on prominent protrusions on each side of head capsule (Figure 8). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 8). No palp apparent. Pronotum cap-like. Mesothorax and metathorax largely concealed (Figure 8A,B). Thorax segments ventrally with locomotory appendages. Legs distally with two prominent elements (tibia, tarsus), with the tibia and tarsus well separated (Figure 8B). Anterior abdomen segments without apparent protrusions. Trunk end with numerous setae. Total length of specimen is 6.00 mm.
- 9.
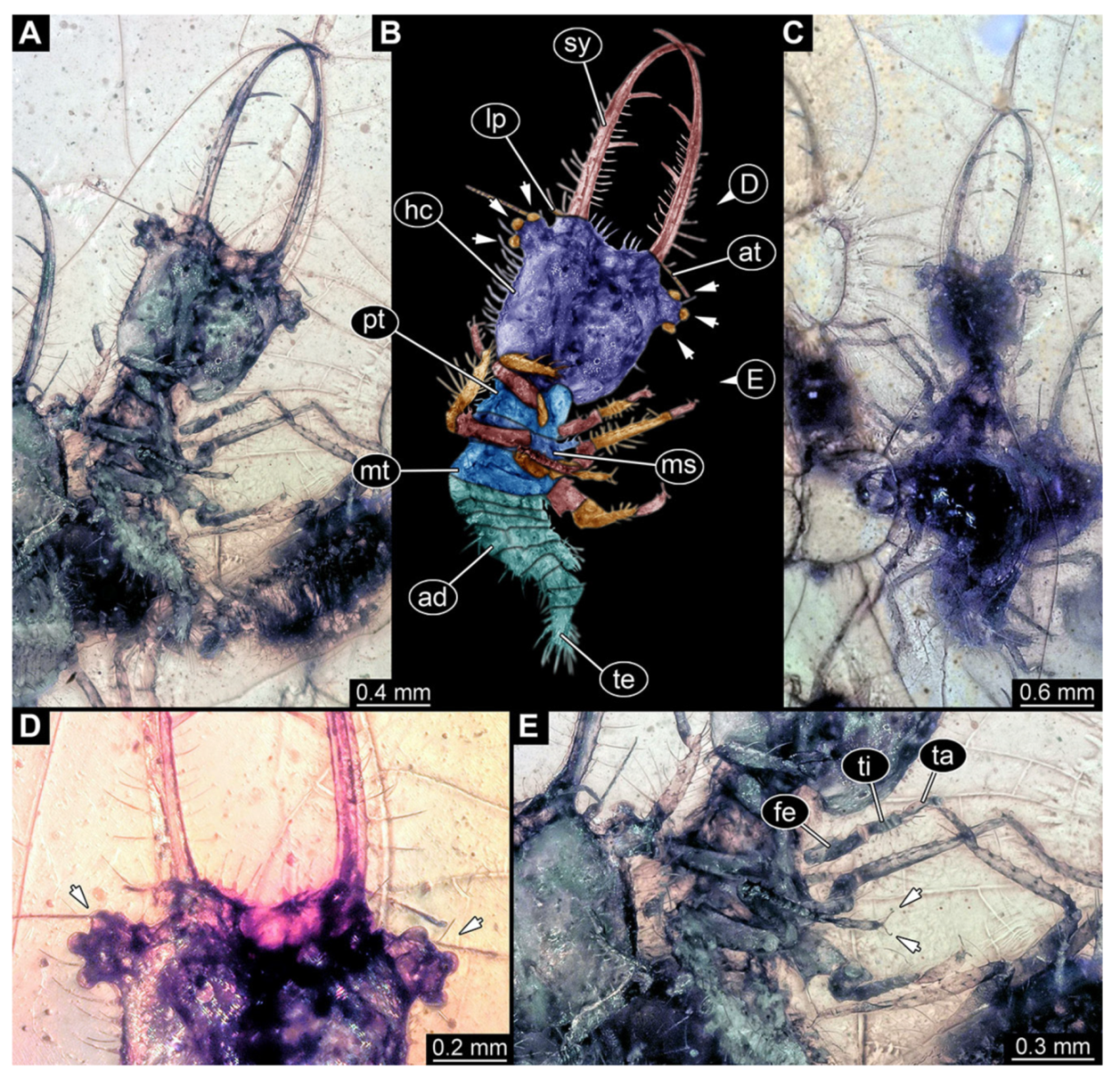
- Specimen 3221 (BUB unnumbered), preserved in Cretaceous Kachin amber, Myanmar. Specimen is well accessible in oblique ventral (Figure 9A) and oblique dorsal view (Figure 9B,C). Head capsule roughly trapezium-shaped in dorsal view, anterior wider than posterior; stylets longer than head capsule (Figure 9D). Stemmata on prominent protrusions on each side of head capsule (Figure 9E,F). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 9D). No palp apparent. Pronotum cap-like. Mesothorax and metathorax bearing numerous setae (Figure 9A–C). Thorax segments ventrally with locomotory appendages. Legs distally with two prominent elements (tibia, tarsus), with the tibia and tarsus well separated (Figure 9G). Anterior abdomen segments also with numerous setae. Trunk end is elongated, longer than wide in dorsal view, with numerous setae. Total length of specimen is 3.6 mm.
- 10.
- Specimen 3222 (BUB 3062), preserved in Cretaceous Kachin amber, Myanmar. Specimen is well accessible in dorsal (Figure 10A,B) and ventral view (Figure 10C). Head capsule roughly square-shaped in dorsal view, only slightly wider than long; stylets longer than head capsule (Figure 10A–C). Anterior rim with deeply forked spine, one on each side of midline (Figure 10A,B). Stemmata on prominent protrusions on each side of head capsule (Figure 10D). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 10A–C). No palps apparent. Pronotum cap-like. Mesothorax and metathorax with very prominent dorso-lateral protrusions bearing numerous setae (Figure 10A–C). Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 10E). Distal tarsus with a pair of claws, no empodium apparent. Anterior abdomen segments also with protrusions, but smaller ones. Trunk end is elongated, longer than wide in dorsal view, with numerous setae. Total length of specimen is 2.00 mm.
- 11.
- Specimen 3223 (BUB 3063), preserved in Cretaceous Kachin amber, Myanmar. Many details of the specimen are not well accessible in dorsal (Figure 11A,B) and ventral view (Figure 11C), due to a reflective film covering the specimen. Head capsule roughly square-shaped in dorsal view, only slightly longer than wide; stylets longer than head capsule (Figure 11D). Stemmata on slight elevations on each side of head capsule (Figure 11D). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 11A–C). No palp apparent. Pronotum cap-like. No apparent protrusions on trunk segments. Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 11E). Distally tarsus with pair of claws, no empodium apparent. Trunk end is elongated, longer than wide in dorsal view, with numerous setae. Total length of specimen is 4.97 mm.
- 12.
- Specimen 3224 (BUB 3724), preserved in Cretaceous Kachin amber, Myanmar. Specimen is complete and is well accessible in ventral (Figure 12A) and dorsal view (Figure 12B,C), yet partly concealed by debris, apparently attached to the body forming camouflaging cloak. Head capsule roughly square-shaped in dorsal view, only slightly wider than long (Figure 12D); stylets longer than head capsule (Figure 12D). Stemmata on prominent protrusions on each side of head capsule (Figure 12D). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 12A–D). Labial palp is short, robust. Pronotum cap-like. No apparent protrusions on trunk segments. Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 12E). Distal tarsus with a pair of claws, no empodium apparent. Trunk end is elongated, longer than wide in dorsal view, with numerous setae. Total length of specimen is 3.68 mm.
- 13.
- Specimen 3225 (CJW F 3199), preserved in Cretaceous Kachin amber, Myanmar. Specimen is accessible in oblique dorsal (Figure 13A,B) and oblique ventral view (Figure 13C). Head capsule roughly square-shaped in dorsal view, only slightly longer than wide; stylets about the same length as head capsule (Figure 13A–C). Stemmata on prominent protrusions on each side of head capsule, but obscured on one side (Figure 13D). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 13A,B). No palps apparent. Pronotum cap-like. Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 13E). Total length of specimen is 2.68 mm.
- 14.
- Specimen 3226 (CJW F 3421), preserved in Cretaceous Kachin amber. Specimen is not well accessible, strongly folded, body visible in oblique lateral view (Figure 13F,G), head also in ventral view (Figure 13H,I). Body largely concealed by debris, apparently attached to the body forming a camouflaging cloak. Head capsule roughly square-shaped in dorsal view, only slightly longer than wide; stylets longer than head capsule (Figure 13H,I). Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 13F,G). Total length of specimen is 2.72 mm.
- 15.
- Specimen 3227 (PED 0083a), preserved in Cretaceous Kachin amber, Myanmar. Specimen is well accessible in ventral view (Figure 14A,B), trunk partly folded. Head capsule roughly square-shaped in ventral view; stylets longer than head capsule (Figure 14A,B). Stemmata on prominent protrusions on each side of head capsule (Figure 14A,B). Anterior rim with simple setae. Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 14A,B). No palps apparent. Mesothorax and metathorax with prominent dorso-lateral protrusions bearing numerous setae (Figure 14A,B). Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 14C,D). Distal tarsus with a pair of claws, no empodium apparent (Figure 14C,D). Anterior abdomen segments also with protrusions, but smaller ones. Trunk end is elongated, longer than wide in dorsal view (folded forwards, therefore seen from dorsally), with numerous setae. Total length of specimen is 3.01 mm.
- 16.
- Specimen 3228 (PED 0083b), preserved in Cretaceous Kachin amber, Myanmar, same piece as previous specimen. Specimen is well accessible in mostly ventral view (Figure 15A,B), but partly twisted and folded, hence the posterior trunk is seen in the dorsal view. Head capsule roughly square-shaped in dorsal view, only slightly wider than long; stylets longer than head capsule (Figure 15A,B). Anterior rim with simple setae. Stemmata on prominent protrusions on each side of head capsule (Figure 15D). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 15C). Labial palp is short, robust. Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 15E). Distal tarsus with a pair of claws, no empodium apparent. Anterior abdomen segments with protrusions. Trunk end is elongate, longer than wide in dorsal view, with numerous setae. Total length of specimen is 3.60 mm.
- 17.
- Specimen 3229 (PED 0083c), preserved in Cretaceous Kachin amber, Myanmar, same piece as two previous specimens. Specimen is well accessible in slightly oblique ventral view (Figure 16A,B), not well accessible in dorsal view, partially concealed by other individuals (Figure 16C). Head capsule roughly square-shaped in dorsal view, only slightly wider than long; stylets longer than head capsule (Figure 16A,B). Anterior rim with simple setae. Stemmata on prominent protrusions on each side of head capsule (Figure 16D). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 16A,B). Labial palp is short, robust. Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 16E). Distal tarsus with a pair of claws, no empodium apparent. Anterior abdomen segments with protrusions. Trunk end is elongated, longer than wide in dorsal view, with numerous setae. Total length of specimen is 3.48 mm.
- 18.
- Specimen 3230 (PED 0083d), preserved in Cretaceous Kachin amber, Myanmar, same piece as three previous specimens. Specimen is well accessible in slightly oblique dorsal view (Figure 17A,B), and is less well accessible in ventral view (Figure 17C), partly obscured by other individuals. Head capsule roughly square-shaped in dorsal view; stylets longer than head capsule (Figure 17A,B). Anterior rim with simple setae. Stemmata on elevations on each side of head capsule (Figure 17C). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 17D). Labial palp is short, robust. Pronotum is cap-like. Mesothorax and metathorax with very prominent dorso-lateral protrusions bearing numerous setae (Figure 17A,B). Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 17E). Distal tarsus with a pair of claws, no empodium apparent. Anterior abdomen segments also with protrusions, but smaller ones. Trunk end is elongated, longer than wide in dorsal view, with numerous setae. Total length of specimen is 3.62 mm.
- 19.
- Specimen 3231 (PED 0083e), preserved in Cretaceous Kachin amber, Myanmar, same piece as four previous specimens. Largely concealed by other individuals, mostly the head is accessible in the dorsal (Figure 18A,B) and ventral view (Figure 18C). Head capsule roughly square-shaped in dorsal view, slightly wider than long; stylets longer than head capsule (Figure 18A–C). Stemmata on slight elevations on each side of head capsule (Figure 18A–C). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet bears two major teeth and few smaller teeth or robust setae (Figure 18A–C). Preserved part of specimen is 1.96 mm.
- 20.
- An additional partial specimen (PED 0083f) is preserved within the same amber piece as the five previous specimens. Yet, the specimen (Figure 18D,E) is too incomplete to further consider it here.
- 21.
- Specimen 3232 (PED 0087), preserved in Cretaceous Kachin amber, Myanmar. Specimen is well accessible in ventral (Figure 19A) and dorsal view (Figure 19B,C). Head capsule roughly square-shaped in dorsal view; stylets longer than head capsule (Figure 19A–C). Stemmata on prominent protrusions on each side of head capsule (Figure 19A–C). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 19E). Labial palp is short, robust. Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 19D). Distal tarsus with a pair of claws, no empodium apparent. Anterior abdomen segments with slight protrusions. Trunk end is elongated, longer than wide in dorsal view, with numerous setae. Total length of specimen is 3.76 mm.
- 22.
- Specimen 3233 (PED 0249), preserved in Cretaceous Kachin amber, Myanmar. Head of specimen is well accessible in dorsal (Figure 20A,B) and ventral view (Figure 20C). Trunk partly concealed by debris, apparently attached to the body forming a camouflaging cloak. Posterior end of trunk seems not to be preserved inside the amber piece. Head capsule roughly square-shaped in dorsal view; stylets longer than head capsule (Figure 20A–C). Stemmata on protrusions on each side of head capsule (Figure 20E). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 20A–C). Labial palp is short, robust. Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 20D). Distal tarsus with a pair of claws, no empodium apparent. Abdomen segments not accessible. Preserved part of specimen is 4.47 mm.
- 23.
- Specimen 3234 (PED 0272), preserved in Cretaceous Kachin amber, Myanmar. Specimen is well accessible in dorsal view (Figure 21A,B), ventrally largely concealed by dirt particles (Figure 21C). Head capsule roughly square-shaped in dorsal view, only slightly wider than long; stylets longer than head capsule (Figure 21A,B). Anterior rim with deeply forked spine, one on each side of midline (Figure 21D). Stemmata on prominent protrusions on each side of head capsule (Figure 21D). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 21A–C). No palps apparent. Pronotum cap-like. Mesothorax and metathorax with very prominent dorso-lateral protrusions bearing numerous setae (Figure 21E). Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated. Anterior abdomen segments also with protrusions, but smaller ones. Trunk end is elongated, longer than wide in dorsal view, with numerous setae. Total length of specimen is 2.23 mm.
- 24.
- Specimen 3235 (PED 0282) is preserved in Cretaceous Kachin amber, Myanmar. Specimen is well accessible in dorsal (Figure 22A,B) and ventral view (Figure 22C), with the further posterior part of trunk slightly concealed by debris, apparently attached to the body forming a camouflaging cloak. Head capsule roughly square-shaped in dorsal view, slightly wider than long; stylets longer than head capsule (Figure 22A–C). Stemmata on protrusions on each side of head capsule (Figure 22D). Antennae and palps not apparent, only very proximal part of antennae visible. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 22D). Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 22E). Distal tarsus with a pair of claws, no empodium apparent. Anterior abdomen segments also with protrusions, but smaller ones. Total length of specimen is 5.54 mm.
- 25.
- Specimen 3236 (PED 0318), preserved in Cretaceous Kachin amber, Myanmar. Specimen is accessible in ventral (Figure 23A,B) and dorsal view (Figure 23C). Head capsule roughly square-shaped in dorsal view, slightly longer than wide; stylets slightly longer than head capsule (Figure 23E). Stemmata on elevations on each side of head capsule (Figure 23E). Antennae small, very slender, far lateral, slightly anterior to stemmata (Figure 23F). Each stylet has two major teeth and few smaller teeth or robust setae (Figure 23E). No palps apparent. Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 23D). Distal tarsus with a pair of claws, no empodium apparent. Anterior abdomen segments also with protrusions, but smaller ones. Trunk end is elongated, longer than wide in dorsal view, with numerous setae. Total length of specimen is 7.35 mm.
- 26.
- Specimen 3237 (PED 0319), preserved in Cretaceous Kachin amber, Myanmar. Specimen is well accessible in dorsal view (Figure 24A,B), ventrally partly concealed by irregularities in the amber (Figure 24C). Head capsule roughly square-shaped in dorsal view, only slightly wider than long; stylets longer than head capsule (Figure 24A–C). Anterior rim with simple setae. Stemmata on prominent protrusions on each side of head capsule (Figure 24D). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 24D). Labial palp short, robust. Metathorax with very prominent dorso-lateral protrusions bearing numerous setae (Figure 24A,B). Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 24E). Distal tarsus with a pair of claws, no empodium apparent. Abdomen not preserved. Total length of specimen is 3.78 mm.
- 27.
- Specimen 3238 (PED 0320), preserved in Cretaceous Kachin amber, Myanmar. Specimen is partly concealed by irregularities of the amber in ventral view (Figure 25A), dorsally better accessible (Figure 25B,C). Head capsule roughly square-shaped in dorsal view, only slightly wider than long; stylets longer than head capsule (Figure 25A–C). Stemmata on prominent protrusions on each side of head capsule (Figure 25E). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 25D). Pronotum cap-like. Mesothorax and metathorax with very prominent dorso-lateral protrusions bearing numerous setae (Figure 25B,C,F). Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 25G). Distally tarsus with pair of claws, no empodium apparent. Trunk end is elongated rounded, with numerous setae. Total length of specimen is 4.00 mm.
- 28.
- Specimen 3239 (PED 0378) is preserved in Cretaceous Kachin amber, Myanmar. Specimen is accessible in dorsal view (Figure 26A,B), ventrally largely concealed (Figure 26C), trunk only preserved as outline. Head capsule roughly square-shaped in dorsal view, only slightly wider than long; stylets longer than head capsule (Figure 26D). Stemmata on prominent protrusions on each side of head capsule (Figure 26D). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 26D). No palps apparent. Thorax is largely concealed by debris, apparently attached to the body forming a camouflaging cloak. Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with he tibia and tarsus well separated (Figure 26E). Distal tarsus with a pair of claws, no empodium apparent. Further details of trunk not available. Total length of specimen is 4.30 mm.
- 29.
- Specimen 3240 (PED 0520), preserved in Cretaceous Kachin amber, Myanmar. Specimen is well accessible in ventral (Figure 27A) and dorsal view (Figure 27B,C). Head capsule roughly square-shaped in dorsal view, only slightly wider than long; stylets longer than head capsule (Figure 27A–C). Anterior rim with simple setae. Stemmata on prominent protrusions on each side of head capsule (Figure 27D). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 27A–C). No palps apparent. Pronotum cap-like. Mesothorax and metathorax with very prominent dorso-lateral protrusions bearing numerous setae (Figure 27F). Thorax segments ventrally with locomotory appendages. Distal tarsus with pair of claws, no empodium apparent (Figure 27E). Anterior abdomen segments also with protrusions, but smaller ones. Trunk end not apparent due to air bubble. Total length of specimen is 5.20 mm.
- 30.
- Specimen 3241 (PED 0563), preserved in Cretaceous Kachin amber, Myanmar. Specimen is accessible in dorsal view (Figure 28A,B); many details not accessible due to numerous small dirt particles; posterior end of trunk not preserved. Head capsule roughly square-shaped in dorsal view; stylets longer than head capsule (Figure 28A,B). Stemmata on elevations on each side of head capsule (Figure 28C). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 28C). No palps apparent. Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 28D). Preserved part of specimen is 3.33 mm.
- 31.
- Specimen 3242 (PED 0575), preserved in Cretaceous Kachin amber, Myanmar. Specimen is not well accessible, neither in the dorsal (Figure 29A,B) nor ventral view (Figure 29C). Head capsule roughly square-shaped in dorsal view; stylets about as long as head capsule (Figure 29A–C). Stemmata on elevations on each side of head capsule (Figure 29D). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth (Figure 29A–C). No palps apparent. Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 29E). Distal tarsus with a pair of claws, no empodium apparent. Details of posterior trunk not accessible. Preserved part of specimen is 4.2 mm.
- 32.
- Specimen 3243 (PED 0583), preserved in Cretaceous Kachin amber, Myanmar. Specimen is accessible in ventral (Figure 30A,B) and dorsal view (Figure 30C), yet dirt and other impurities conceal many details. Head capsule roughly square-shaped in dorsal view, only slightly longer than wide; stylets longer than head capsule (Figure 30A–C). Stemmata on prominent protrusions on each side of head capsule (Figure 30D). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 30A–C). No palps apparent. Thorax segments ventrally with locomotory appendages. Legs distally with two prominent elements (tibia, tarsus), which are well separated (Figure 30E). Distal tarsus with a pair of claws, no empodium apparent. No details of abdomen segments accessible. Trunk end is elongated, longer than wide in ventral view, with numerous setae. Total length of specimen is 8.20 mm.
- 33.
- Specimen 3244 (PED 0684), preserved in Cretaceous Kachin amber, Myanmar. Specimen is accessible in dorsal view (Figure 31A,B), largely concealed in ventral view (Figure 31C), posterior end incomplete, damaged. Head capsule roughly square-shaped in dorsal view, only slightly longer than wide; stylets about as long as head capsule (Figure 31A–C). Stemmata on elevations on each side of head capsule (Figure 31D). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth (Figure 31A–C). No palps apparent. Mesothorax and metathorax with prominent dorso-lateral protrusions bearing numerous setae (Figure 31A,B). Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 31E). Distal tarsus with a pair of claws, no empodium apparent. Details of abdomen segments not well accessible. Preserved part of specimen is 8.06 mm.
- 34.
- Specimen 3245 (PED 0944), preserved in Cretaceous Kachin amber, Myanmar. Specimen is well accessible in the dorsal (Figure 32A,B) and ventral view (Figure 32C). Head capsule roughly square-shaped in dorsal view, but wider than long; stylets longer than head capsule (Figure 32D). Anterior rim with deeply forked spine, one on each side of midline (Figure 32D). Stemmata on prominent protrusions on each side of head capsule (Figure 32D). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 32D). Labial palp short, robust. Pronotum cap-like. Mesothorax and metathorax with very prominent dorso-lateral protrusions bearing numerous setae (Figure 32A,B). Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 32E–G), also in leg 3 (Figure 32E). Distal tarsus with a pair of claws, no empodium apparent. Anterior abdomen segments also with protrusions, but smaller ones. Trunk end is elongated, longer than wide in dorsal view, with numerous setae. Total length of specimen is 2.07 mm.
- 35.
- Specimen 3246 (PED 0975), preserved in Cretaceous Kachin amber, Myanmar. Specimen is accessible in dorsal (Figure 33A), ventral (Figure 33B,C) and lateral view (Figure 33D). Head capsule roughly square-shaped in dorsal view, but slightly wider than long; stylets longer than head capsule (Figure 33A–C). Stemmata on prominent protrusions on each side of head capsule (Figure 33E). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 33A,B). No palps apparent. Mesothorax and metathorax with very prominent dorso-lateral protrusions bearing numerous setae (Figure 33A,B). Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 33F). Distal tarsus with a pair of claws, no empodium apparent. Abdomen segments not well accessible. Total length of specimen is 2.65 mm.
- 36.
- Specimen 3247 (PED 1047), preserved in Cretaceous Kachin amber, Myanmar. Specimen is accessible in dorsal view (Figure 34A,B). Head capsule roughly square-shaped in dorsal view, only slightly wider than long; stylets longer than head capsule (Figure 34C). Anterior rim with deeply forked spine, one on each side of midline (Figure 34C). Stemmata on prominent protrusions on each side of head capsule (Figure 34C). Antennae are small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 34C). Labial palp short, robust. Pronotum cap-like. Mesothorax and metathorax with very prominent dorso-lateral protrusions bearing numerous setae (Figure 34A,B). Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 34D). Anterior abdomen segments also with protrusions, but smaller ones. Trunk end is elongated, longer than wide in dorsal view, with numerous setae. Total length of specimen is 5.35 mm.
- 37.
- Specimen 3248 (PED 1206), preserved in Cretaceous Kachin amber, Myanmar. Only the head is preserved. Specimen is well accessible in dorsal and ventral view (Figure 35A–C). Head capsule roughly square-shaped in dorsal view; stylets longer than head capsule (Figure 35A–C). Stemmata on prominent protrusions on each side of head capsule (Figure 35D,E). Each stylet has two major teeth and numerous smaller teeth or robust setae (Figure 35A–C). Anterior rim medially with deeply forked spine, one on each side of midline (Figure 35A–C). Antennae are small, very slender, far lateral, slightly anterior to stemmata. No palps apparent. Preserved part of specimen is 2.04 mm.
- 38.
- Specimen 3249 (Weiterschan BuB 23), preserved in Cretaceous Kachin amber, Myanmar. Specimen is well accessible in ventral view (Figure 36A,B), largely concealed in dorsal view (Figure 36C). Head capsule roughly square-shaped in dorsal view, slightly wider than long; stylets longer than head capsule (Figure 36A–C). Stemmata on prominent protrusions on each side of head capsule (Figure 36D). Antennae small, very slender, far lateral, slightly anterior to stemmata. Each stylet has two major teeth and few smaller teeth or robust setae (Figure 36A–C). No palps apparent. Mesothorax and metathorax with very prominent dorso-lateral protrusions bearing numerous setae (Figure 36A,B). Thorax segments ventrally with locomotory appendages. Legs distally with three prominent elements (femur, tibia, tarsus), with the tibia and tarsus well separated (Figure 36E). Anterior abdomen segments also with protrusions, but smaller ones. Trunk end about as long as wide in dorsal view, with numerous setae. Total length of specimen is 3.68 mm.
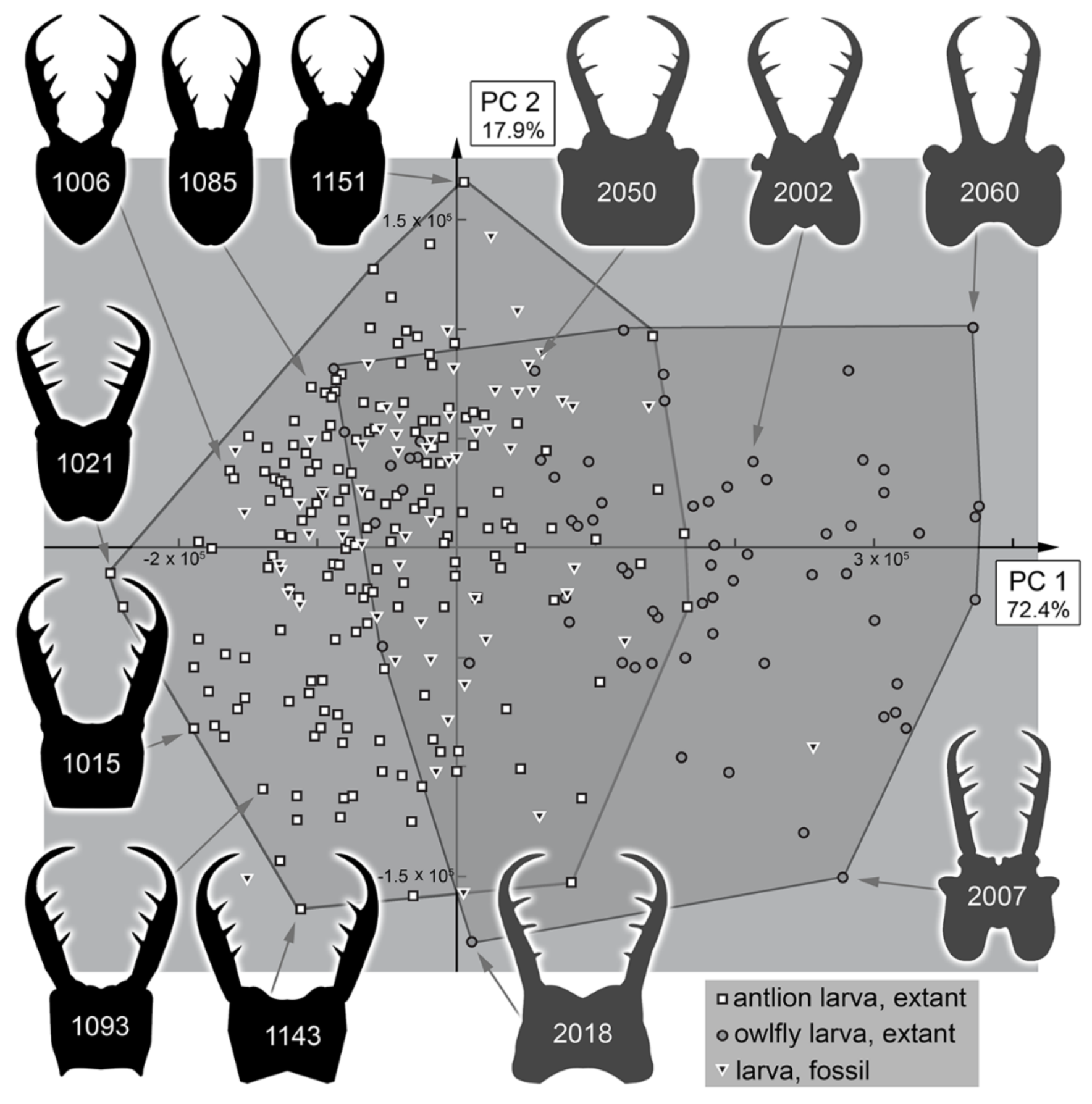
3.2. Shape Analysis
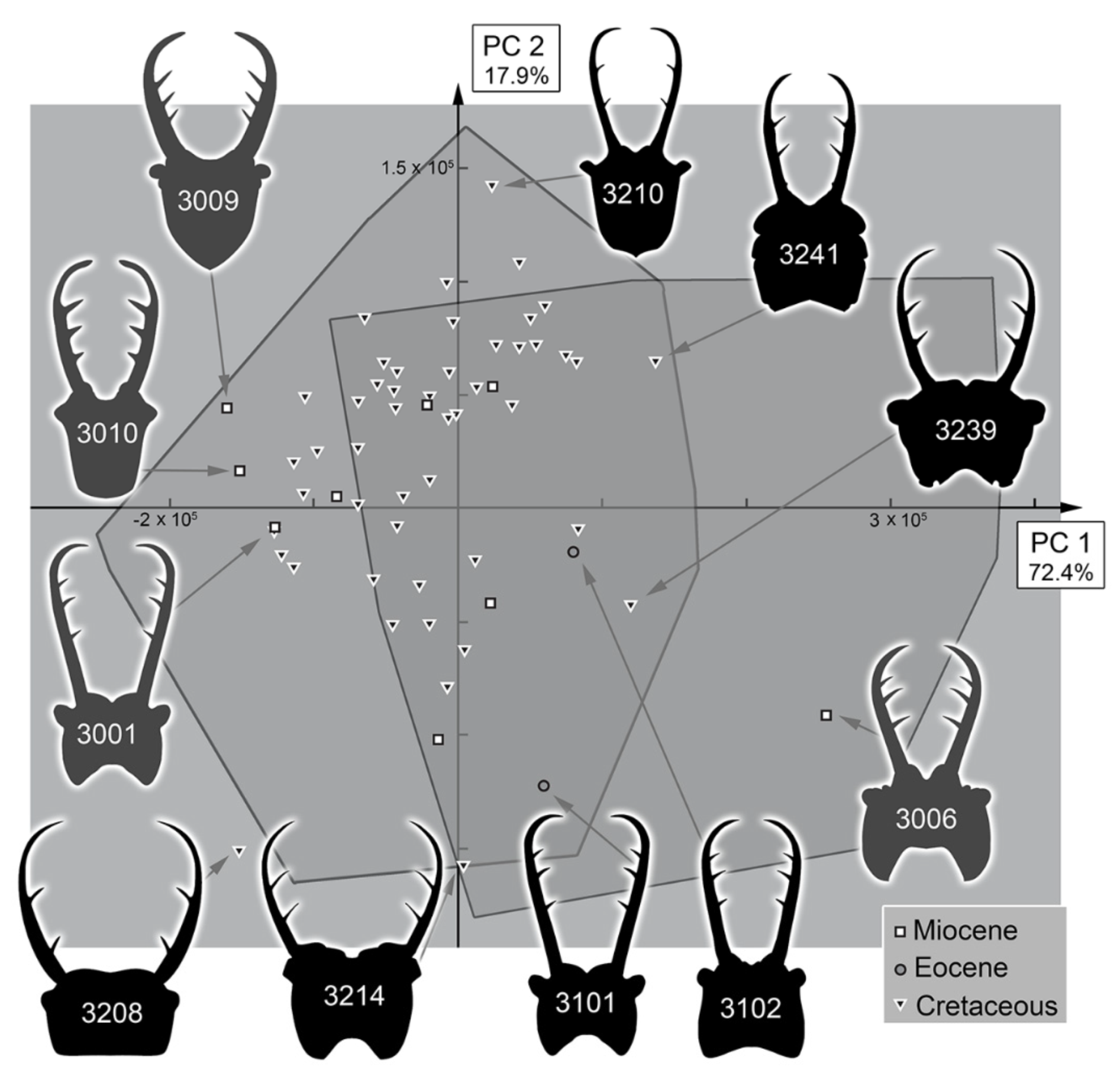
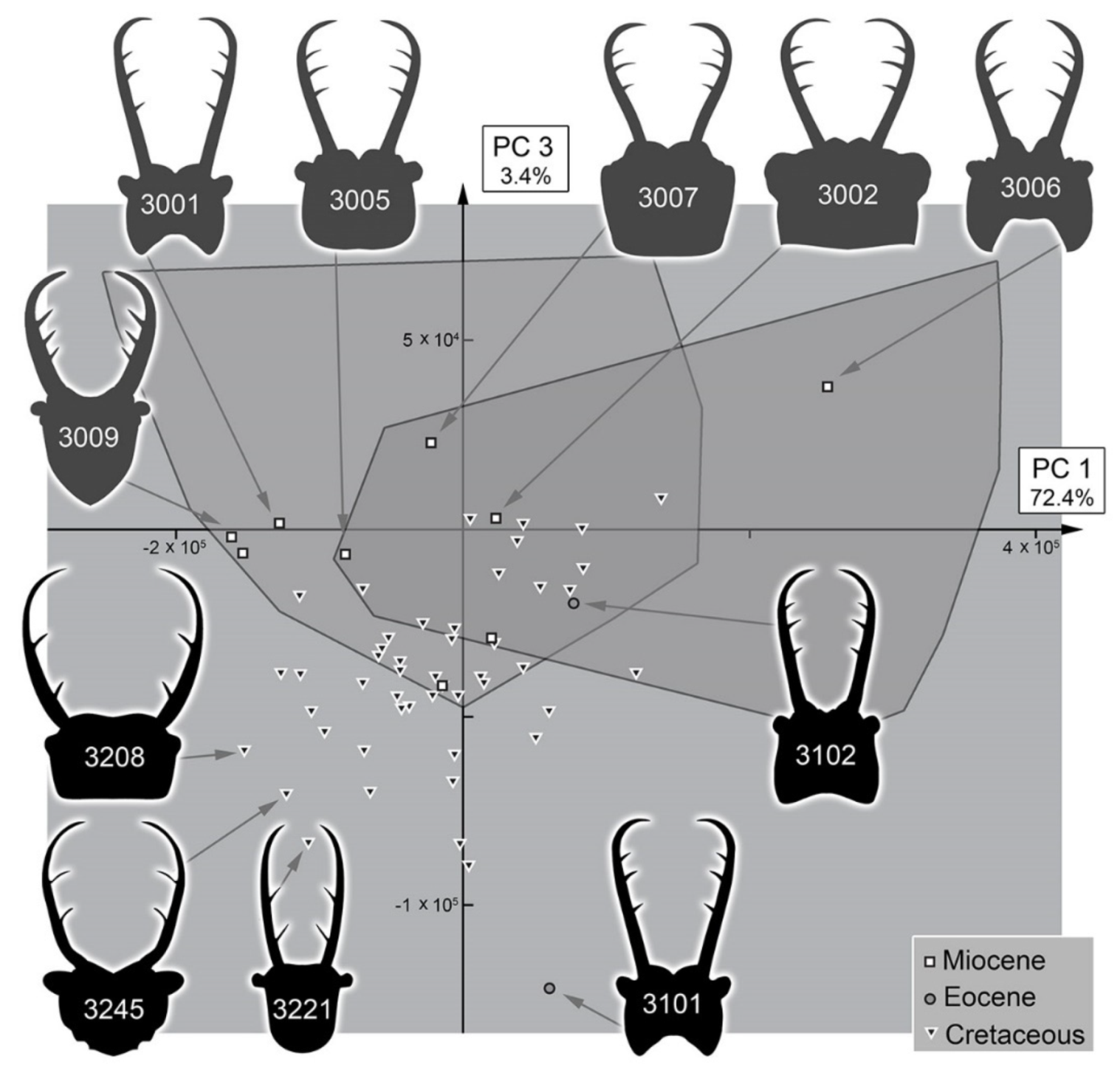
3.3. Statistical Differences in the Morphospace of Different Time Groups
4. Discussion
4.1. Identity of the Specimens Reported Here
4.2. Sub-Sample Sizes
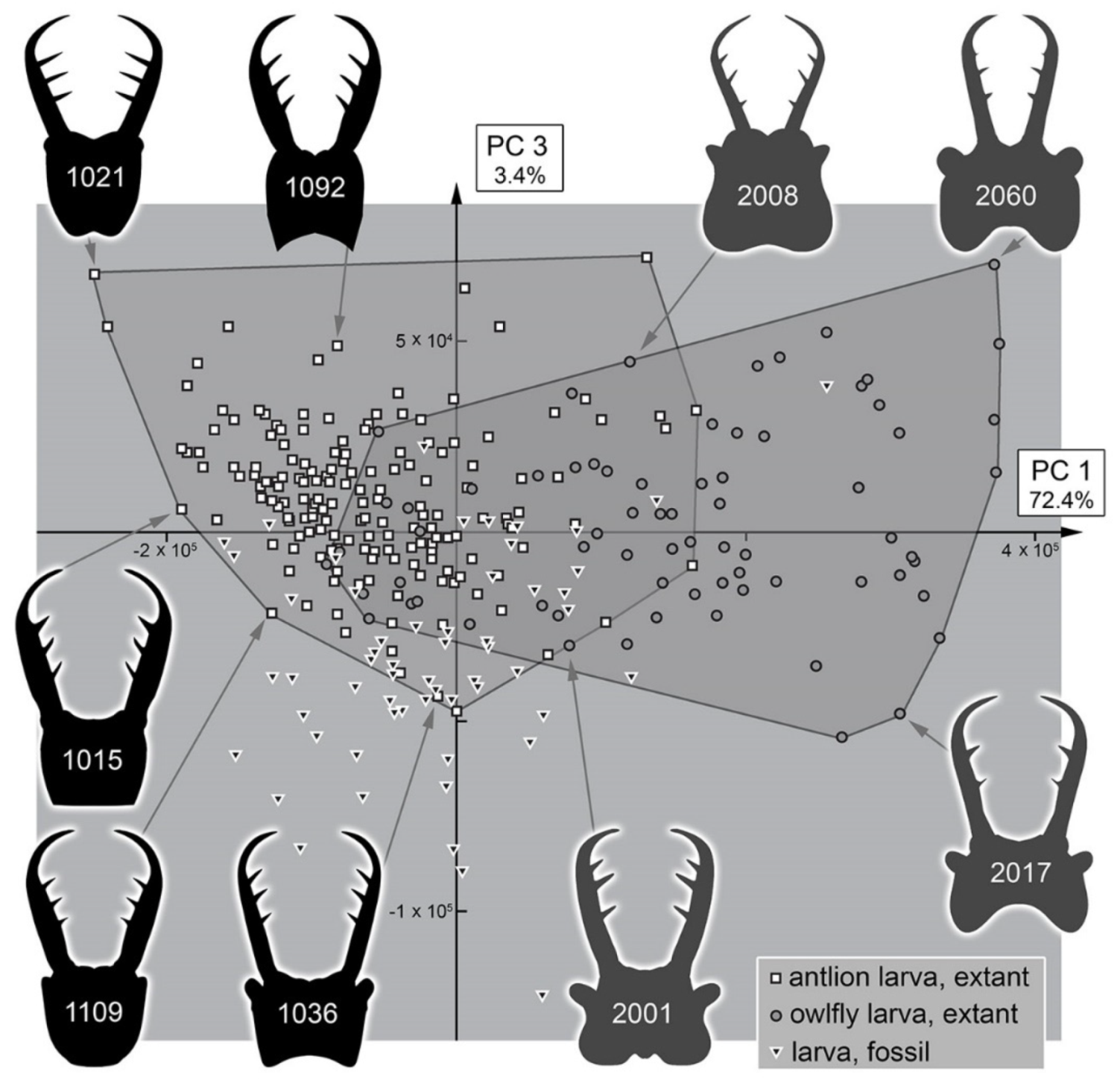
4.3. Morphospace Occupation of the Extant Larvae
4.4. Morphospace Occupation of the Fossil Larvae
4.5. Diversity Changes through Time in Owllion Larvae
4.6. Diversity Changes through Time in Comparison to Other Myrmeleontiformian Lacewings
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WWF. Living Planet Report 2020-Bending the Curve of Biodiversity Loss; Almond, R.E.A., Grooten, M., Peterson, T., Eds.; World Wildlife Fund: Gland, Switzerland, 2020; 162p. [Google Scholar]
- Sánchez-Bayo, F.; Wyckhuys, K.A. Worldwide decline of the entomofauna: A review of its drivers. Biol. Conserv. 2019, 232, 8–27. [Google Scholar] [CrossRef]
- Hallmann, C.A.; Sorg, M.; Jongejans, E.; Siepel, H.; Hofland, N.; Schwan, H.; Stenmans, W.; Müller, A.; Sumser, H.; Hörren, T.; et al. More than 75 percent decline over 27 years in total flying insect biomass in protected areas. PLoS ONE 2017, 12, e0185809. [Google Scholar] [CrossRef]
- Hallmann, C.A.; Ssymank, A.; Sorg, M.; de Kroon, H.; Jongejans, E. Insect biomass decline scaled to species diversity: General patterns derived from a hoverfly community. Proc. Natl. Acad. Sci. USA 2021, 118, e2002554117. [Google Scholar] [CrossRef]
- van der Sluijs, J.P. Insect decline, an emerging global environmental risk. Curr. Opin. Environ. Sustain. 2020, 46, 39–42. [Google Scholar] [CrossRef]
- Wagner, D.L. Insect declines in the Anthropocene. Annu. Rev. Entomol. 2020, 65, 457–480. [Google Scholar] [CrossRef]
- Wagner, D.L.; Grames, E.M.; Forister, M.L.; Berenbaum, M.R.; Stopak, D. Insect decline in the Anthropocene: Death by a thousand cuts. Proc. Natl. Acad. Sci. USA 2021, 118, e2023989118. [Google Scholar] [CrossRef]
- Blüthgen, N.; Staab, M.; Achury, R.; Weisser, W.W. Unravelling insect declines: Can space replace time? Biol. Lett. 2022, 18, 20210666. [Google Scholar] [CrossRef] [PubMed]
- Klein, A.M.; Vaissiere, B.E.; Cane, J.H.; Steffan-Dewenter, I.; Cunningham, S.A.; Kremen, C.; Tscharntke, T. Importance of pollinators in changing landscapes for world crops. Proc. R. Soc. B 2007, 274, 303–313. [Google Scholar] [CrossRef]
- Aizen, M.A.; Garibaldi, L.A.; Cunningham, S.A.; Klein, A.M. How much does agriculture depend on pollinators? Lessons from long-term trends in crop production. Ann. Bot. 2009, 103, 1579–1588. [Google Scholar] [CrossRef] [PubMed]
- Minelli, A.; Brena, C.; Deflorian, G.; Maruzzo, D.; Fusco, G. From embryo to adult—beyond the conventional periodization of arthropod development. Dev. Genes Evol. 2006, 216, 373–383. [Google Scholar] [CrossRef] [PubMed]
- Bybee, S.M.; Hansen, Q.; Buesse, S.; Cahill Wightman, H.M.; Branham, M.A. For consistency’s sake: The precise use of larva, nymph and naiad within Insecta. Syst. Entomol. 2015, 40, 667–670. [Google Scholar] [CrossRef]
- Sahlén, G.; Suhling, F.; Martens, A.; Gorb, S.N.; Fincke, O.M. For consistency’s sake? A reply to Bybee et al. Syst. Entomol. 2016, 41, 307–308. [Google Scholar] [CrossRef]
- Büsse, S.; Bybee, S.M. Larva, nymph and Naiad–A response to the replies to Bybee et al. (2015) and the results of a survey within the entomological community. Syst. Entomol. 2017, 42, 11–14. [Google Scholar] [CrossRef]
- Haug, J.T. Why the term “larva” is ambiguous, or what makes a larva. Acta Zool. 2020, 101, 167–188. [Google Scholar] [CrossRef]
- Schreiber, S.; Rudolf, V.H. Crossing habitat boundaries: Coupling dynamics of ecosystems through complex life cycles. Ecol. Lett. 2008, 11, 576–587. [Google Scholar] [CrossRef]
- Romero, G.Q.; Srivastava, D.S. Food-web composition affects cross-ecosystem interactions and subsidies. J. Anim. Ecol. 2010, 79, 1122–1131. [Google Scholar] [CrossRef]
- Kristensen, N.P. Phylogeny of endopterygote insects, the most successful lineage of living organisms. Eur. J. Entomol. 1999, 96, 237–254. [Google Scholar]
- McMahon, D.P.; Hayward, A. Why grow up? A perspective on insect strategies to avoid metamorphosis. Ecol. Entomol. 2016, 41, 505–515. [Google Scholar] [CrossRef]
- New, T.R. Neuroptera. In The Insects of Australia: A Textbook for Students and Research Workers, 2nd ed.; Commonwealth Scientific and Industrial Research Organisation, Division of Entomology, Ed.; Melbourne University Press: Victoria, Australia, 1991; Volume 2, pp. 525–542. [Google Scholar]
- Aspöck, U.; Aspöck, H. Kamelhälse, Schlammfliegen, Ameisenlöwen. Wer sind sie? (Insecta: Neuropterida: Raphidioptera, Megaloptera, Neuroptera). Stapfia 1999, 60, 1–34. [Google Scholar]
- Aspöck, U.; Aspöck, H. Verbliebene Vielfalt vergangener Blüte. Zur Evolution, Phylogenie und Biodiversität der Neuropterida (Insecta: Endopterygota). Denisia 2007, 20, 451–516. [Google Scholar]
- Winterton, S.L.; Lemmon, A.R.; Gillung, J.P.; Garzón, I.J.; Badano, D.; Bakkes, D.K.; Breitkreuz, L.C.V.; Engel, M.S.; Moriarty Lemmon, E.; Liu, X.-Y.; et al. Evolution of lacewings and allied orders using anchored phylogenomics (Neuroptera, Megaloptera, Raphidioptera). Syst. Entomol. 2018, 43, 330–354. [Google Scholar] [CrossRef]
- Machado, R.J.; Gillung, J.P.; Winterton, S.L.; Garzón-Orduña, I.J.; Lemmon, A.R.; Lemmon, E.M.; Oswald, J.D. Owlflies are derived antlions: Anchored phylogenomics supports a new phylogeny and classification of Myrmeleontidae (Neuroptera). Syst. Entomol. 2019, 44, 418–450. [Google Scholar] [CrossRef]
- Winterton, S.L.; Hardy, N.B.; Wiegmann, B.M. On wings of lace: Phylogeny and Bayesian divergence time estimates of Neuropterida (Insecta) based on morphological and molecular data. Syst. Entomol. 2010, 35, 349–378. [Google Scholar] [CrossRef]
- Badano, D.; Fratini, M.; Maugeri, L.; Palermo, F.; Pieroni, N.; Cedola, A.; Haug, J.T.; Weiterschan, T.; Velten, J.; Mei, M.; et al. X-ray microtomography and phylogenomics provide insights into the morphology and evolution of an enigmatic Mesozoic insect larva. Syst. Entomol. 2021, 46, 672–684. [Google Scholar] [CrossRef]
- Henry, C.S. Eggs and rapagula of Ululodes and Ascaloptynx (Neuroptera: Ascalaphidae): A comparative study. Psyche 1972, 79, 054050. [Google Scholar] [CrossRef]
- Nardi, J.B. Life in the Soil. A Guide for Naturalists and Gardeners; The University of Chicago Press: Chicago, IL, USA; London, UK, 2007; 293p. [Google Scholar]
- Devetak, D.; Klokočovnik, V.; Lipovsek, S.; Bock, E.; Leitinger, G. Larval morphology of the antlion Myrmecaelurus trigrammus (Pallas, 1771) (Neuroptera, Myrmeleontidae), with notes on larval biology. Zootaxa 2013, 3641, 491–500. [Google Scholar] [CrossRef] [PubMed]
- Badano, D.; Pantaleoni, R.A. The larvae of European Ascalaphidae (Neuroptera). Zootaxa 2014, 3796, 287–319. [Google Scholar] [CrossRef]
- Gupta, A.; Badano, D. Larval morphology and life history of Ascalaphus dicax Walker, 1853 (Neuroptera: Myrmeleontidae, Ascalaphinae). Fragm. Entomol. 2021, 53, 1–8. [Google Scholar] [CrossRef]
- MacLeod, E.G. A Comparative Morphological Study of the Head Capsule and Cervix of Larval Neuroptera (Insecta). Doctoral Dissertation, Harvard University, Cambridge, MA, USA, 1964. [Google Scholar]
- Engel, M.S.; Grimaldi, D.A. The neuropterid fauna of Dominican and Mexican amber (Neuropterida, Megaloptera, Neuroptera). Am. Mus. Novit. 2007, 3587, 1–58. [Google Scholar] [CrossRef]
- Zimmermann, D.; Randolf, S.; Aspöck, U. From chewing to sucking via phylogeny—from sucking to chewing via ontogeny: Mouthparts of Neuroptera. In Insect Mouthparts—Form, Function, Development and Performance, Zoological Monographs; Krenn, H., Ed.; Springer: Cham, Switzerland, 2019; Volume 5, pp. 361–385. [Google Scholar] [CrossRef]
- Nicoli Aldini, R. Observations on the larval morphology of the antlion Myrmeleon bore (Tjeder, 1941) (Neuroptera Myrmeleontidae) and its life cycle in the Po Valley (northern Italy). Ann. Mus. Civ. Stor. Nat. Ferrara 2007, 8, 59–66. [Google Scholar]
- Pantaleoni, R.A.; Cesaroni, C.; Nicoli Aldini, R. Myrmeleon mariaemathildae n. sp.: A new Mediterranean pit-building antlion (Neuropterida Myrmeleontidae). Bull. Insectology 2010, 63, 91–98. [Google Scholar]
- Franks, N.R.; Worley, A.; Falkenberg, M.; Sendova-Franks, A.B.; Christensen, K. Digging the optimum pit: Antlions, spirals and spontaneous stratification. Proc. R. Soc. B 2009, 286, 20190365. [Google Scholar] [CrossRef] [PubMed]
- Büsse, S.; Büscher, T.H.; Heepe, L.; Gorb, S.N.; Stutz, H.H. Sand-throwing behaviour in pit-building antlion larvae: Insights from finite-element modelling. J. R. Soc. Interface 2021, 18, 20210539. [Google Scholar] [CrossRef] [PubMed]
- McEwen, P.K.; New, T.R.; Whittington, A.E. Lacewings in the Crop Environment; Cambridge University Press: Cambridge, UK, 2001; 546p. [Google Scholar]
- Zheng, Y.C.; Liu, X.Y. First description of immature stages of the antlion Bullanga florida (Navás, 1913) (Neuroptera, Myrmeleontidae, Dendroleontini). Zootaxa 2020, 4858, 394–404. [Google Scholar] [CrossRef]
- Zheng, Y.C.; Hayashi, F.; Price, B.W.; Liu, X.Y. Unveiling the evolutionary history of a puzzling antlion genus Gatzara Navás (Neuroptera, Myrmeleontidae, Dendroleontinae) based on systematic revision, molecular phylogenetics, and biogeographic inference. Insect Syst. Div. 2020, 6, 4. [Google Scholar] [CrossRef]
- Belušič, G.; Pirih, P.; Zupančič, G.; Stušek, P.; Drašlar, K. The visual ecology of the owlfly (Libelloides macaronius). In Proceedings of the 10th International Symposium on Neuropterology, Piran, Slovenia, 22–25 June 2008; Devetak, D., Lipovšek, S., Arnett, A.E., Eds.; University of Maribor: Maribor, Slovenia, 2010; pp. 89–96. [Google Scholar]
- Devetak, D.; Klokočovnik, V. The feeding biology of adult lacewings (Neuroptera): A review. Trends Entomol. 2016, 12, 29–42. [Google Scholar]
- Labandeira, C.C. A compendium of fossil insect families. Milwaukee Public Mus. Contrib. Biol. Geol. 1994, 88, 11–16. [Google Scholar]
- Oswald, J.D.; Machado, R.J. Biodiversity of the Neuropterida (Insecta: Neuroptera, Megaloptera, and Raphidioptera). In Insect Biodiversity: Science and Society, 1st ed.; Foottit, R.G., Adler, P.H., Eds.; Wiley: Hoboken, NY, USA, 2018; Volume 2, pp. 627–672. [Google Scholar] [CrossRef]
- Labandeira, C.C.; Yang, Q.; Santiago-Blay, J.A.; Hotton, C.L.; Monteiro, A.; Wang, Y.-J.; Goreva, Y.; Shih, C.K.; Siljeström, S.; Rose, T.R.; et al. The evolutionary convergence of mid-Mesozoic lacewings and Cenozoic butterflies. Proc. R. Soc. B Biol. Sci. 2016, 283, 20152893. [Google Scholar] [CrossRef]
- Wang, B.; Xia, F.; Engel, M.S.; Perrichot, V.; Shi, G.; Zhang, H.; Chen, J.; Jarzembowski, E.A.; Wappler, T.; Rust, J. Debris-carrying camouflage among diverse lineages of Cretaceous insects. Sci. Adv. 2016, 2, e1501918. [Google Scholar] [CrossRef]
- Zhang, W.W. Frozen dimensions. In The Fossil Insects and Other Invertebrates in Amber; Chongqing University Press: Chongqin, China, 2017; 692p. [Google Scholar]
- Liu, Q.; Lu, X.; Zhang, Q.; Chen, J.; Zheng, X.; Zhang, W.; Liu, X.; Wang, B. High niche diversity in Mesozoic pollinating lacewings. Nat. Commun. 2018, 9, 3793. [Google Scholar] [CrossRef]
- Badano, D.; Engel, M.S.; Basso, A.; Wang, B.; Cerretti, P. Diverse Cretaceous larvae reveal the evolutionary and behavioural history of antlions and lacewings. Nat. Commun. 2018, 9, 3257. [Google Scholar] [CrossRef] [PubMed]
- Makarkin, V.N. Re-description of Grammapsychops lebedevi Martynova, 1954 (Neuroptera: Psychopsidae) with notes on the Late Cretaceous psychopsoids. Zootaxa 2018, 4524, 581–594. [Google Scholar] [CrossRef] [PubMed]
- Herrera-Flórez, A.F.; Braig, F.; Haug, C.; Neumann, C.; Wunderlich, J.; Hörnig, M.K.; Haug, J.T. Identifying the oldest larva of a myrmeleontiformian lacewing–A morphometric approach. Acta Palaeontol. Pol. 2020, 65, 235–250. [Google Scholar] [CrossRef]
- Khramov, A.V.; Bashkuev, A.S.; Lukashevich, E.D. The fossil record of long-proboscid nectarivorous insects. Entomol. Rev. 2020, 100, 881–968. [Google Scholar] [CrossRef]
- Lu, X.; Wang, B.; Zhang, W.; Ohl, M.; Engel, M.S.; Liu, X. Cretaceous diversity and disparity in a lacewing lineage of predators (Neuroptera: Mantispidae). Proc. R. Soc. B Biol. Sci. 2020, 287, 20200629. [Google Scholar] [CrossRef]
- Nakamine, H.; Yamamoto, S.; Takahashi, Y. Hidden diversity of small predators: New thorny lacewings from mid-Cretaceous am- ber from northern Myanmar (Neuroptera: Rhachiberothidae: Paraberothinae). Geol. Mag. 2020, 157, 1149–1175. [Google Scholar] [CrossRef]
- Shi, C.; Yang, Q.; Shih, C.; Labandeira, C.C.; Pang, H.; Ren, D. Cretaceous mantid lacewings with specialized raptorial forelegs illuminate modification of prey capture (Insecta: Neuroptera). Zool. J. Linn. Soc. 2020, 190, 1054–1070. [Google Scholar] [CrossRef]
- MacLeod, E.G. The Neuroptera of the Myanmar Amber. I. Ascalaphidae, Nymphidae, and Psychopsidae. Psyche 1970, 77, 147–180. [Google Scholar] [CrossRef]
- Poinar, G.O., Jr.; Poinar, R. The Amber Forest: A Reconstruction of a Vanished World; Princeton University Press: Princeton, NJ, USA, 1999; xviii+239p. [Google Scholar]
- Scheven, J. Bernstein-Einschlüsse: Eine untergegangene Welt bezeugt die Schöpfung. Erinnerungen an die Welt vor der Sintflut; Kuratorium Lebendige Vorwelt: Hofheim a.T., Germany, 2004; 160p. [Google Scholar]
- Grimaldi, D.; Engel, M.S. Evolution of the Insects; Cambridge University Press: Cambridge, UK, 2005; 755p. [Google Scholar]
- Engel, M.S.; Grimaldi, D.A. Diverse Neuropterida in Cretaceous amber, with particular reference to the paleofauna of Myanmar (Insecta). Nova Suppl. Entomol. 2008, 20, 1–86. [Google Scholar]
- Haug, C.; Haug, G.T.; Baranov, V.A.; Solórzano-Kraemer, M.M.; Haug, J.T. An owlfly larva preserved in Mexican amber and the Miocene record of lacewing larvae. Bol. Soc. Geol. Mex. 2021, 73, A271220. [Google Scholar] [CrossRef]
- Pérez-de la Fuente, R.; Delclòs, X.; Peñalver, E.; Speranza, M.; Wierzchos, J.; Ascaso, C.; Engel, M.S. Early evolution and ecology of camouflage in insects. Proc. Natl. Acad. Sci. USA 2012, 109, 21414–21419. [Google Scholar] [CrossRef] [PubMed]
- Pérez-de la Fuente, R.; Delclos, X.; Penalver, E.; Engel, M.S. A defensive behavior and plant-insect interaction in Early Cretaceous amber–the case of the immature lacewing Hallucinochrysa diogenesi. Arthropod Struct. Dev. 2016, 45, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Pérez-de la Fuente, R.; Peñalver, E.; Azar, D.; Engel, M.S. A soil-carrying lacewing larva in Early Cretaceous Lebanese amber. Sci. Rep. 2018, 8, 16663. [Google Scholar] [CrossRef] [PubMed]
- Pérez-de la Fuente, R.; Engel, M.S.; Azar, D.; Peñalver, E. The hatching mechanism of 130-million-year-old insects: An association of neonates, egg shells and egg bursters in Lebanese amber. Palaeontology 2019, 62, 547–559. [Google Scholar] [CrossRef]
- Pérez-de la Fuente, R.; Engel, M.S.; Delclòs, X.; Peñalver, E. Straight-jawed lacewing larvae (Neuroptera) from Lower Cretaceous Spanish amber, with an account on the known amber diversity of neuropterid immatures. Cretac. Res. 2020, 106, 104200. [Google Scholar] [CrossRef]
- Xia, F.; Yang, G.; Zhang, Q.; Shi, G.; Wang, B. Amber: Life through Time and Space; Science Press: Beijing, China, 2015; 197p. [Google Scholar]
- Liu, X.; Zhang, W.; Winterton, S.L.; Breitkreuz, L.C.; Engel, M.S. Early morphological specialization for insect-spider associations in Mesozoic lacewings. Curr. Biol. 2016, 26, 1590–1594. [Google Scholar] [CrossRef]
- Liu, X.; Shi, G.; Xia, F.; Lu, X.; Wang, B.; Engel, M.S. Liverwort mimesis in a Cretaceous lacewing larva. Curr. Biol. 2018, 28, 1475–1481. [Google Scholar] [CrossRef]
- Liu, H.; Luo, C.; Jarzembowski, E.A.; Xiao, C. Acanthochrysa langae gen. et sp. nov., a new lacewing larva (Neuroptera: Chrysopoidea) from mid-Cretaceous Kachin amber. Cretac. Res. 2022, 133, 105146. [Google Scholar] [CrossRef]
- Haug, C.; Herrera-Flórez, A.F.; Müller, P.; Haug, J.T. Cretaceous chimera–an unusual 100-million-year old neuropteran larva from the “experimental phase” of insect evolution. Palaeodiversity 2019, 12, 1–11. [Google Scholar] [CrossRef]
- Haug, J.T.; Müller, P.; Haug, C. A 100-million-year old predator: A fossil neuropteran larva with unusually elongated mouthparts. Zool. Lett. 2019, 5, 29. [Google Scholar] [CrossRef]
- Haug, J.T.; Müller, P.; Haug, C. A 100-million-year old slim insectan predator with massive venom-injecting stylets-A new type of neuropteran larva from Burmese amber. Bull. Geosci. 2019, 94, 431–440. [Google Scholar] [CrossRef]
- Haug, G.T.; Haug, C.; Pazinato, P.G.; Braig, F.; Perrichot, V.; Gröhn, C.; Müller, P.; Haug, J.T. The decline of silky lacewings and morphological diversity of long-nosed antlion larvae through time. Palaeontol. Electron. 2020, 23, a39. [Google Scholar] [CrossRef]
- Haug, J.T.; Baranov, V.; Schädel, M.; Müller, P.; Gröhn, C.; Haug, C. Challenges for understanding lacewings: How to deal with the incomplete data from extant and fossil larvae of Nevrorthidae? (Neuroptera). Fragm. Entomol. 2020, 52, 137–167. [Google Scholar] [CrossRef]
- Haug, J.T.; Pazinato, P.G.; Haug, G.T.; Haug, C. Yet another unusual new type of lacewing larva preserved in 100-million-year old amber from Myanmar. Riv. Ital. Paleontol. Stratigr. 2020, 126, 821–832. [Google Scholar] [CrossRef]
- Haug, G.T.; Baranov, V.; Wizen, G.; Pazinato, P.G.; Müller, P.; Haug, C.; Haug, J.T. The morphological diversity of long-necked lacewing larvae (Neuroptera: Myrmeleontiformia). Bull. Geosci. 2021, 96, 431–457. [Google Scholar] [CrossRef]
- Haug, G.T.; Haug, C.; Haug, J.T. The morphological diversity of spoon-winged lacewing larvae and the first possible fossils from 99 million-year-old Myanmar amber. Palaeodiversity 2021, 14, 133–152. [Google Scholar] [CrossRef]
- Haug, J.T.; Baranov, V.; Müller, P.; Haug, C. New extreme morphologies as exemplified by 100 million-year-old lacewing larvae. Sci. Rep. 2021, 11, 20432. [Google Scholar] [CrossRef]
- Haug, J.T.; Haug, G.T.; Zippel, A.; van der Wal, S.; Müller, P.; Gröhn, C.; Wunderlich, J.; Hoffeins, C.; Hoffeins, H.-W.; Haug, C. Changes in the morphological diversity of larvae of lance lacewings, mantis lacewings and their closer relatives over 100 million years. Insects 2021, 12, 860. [Google Scholar] [CrossRef]
- Haug, G.T.; Haug, C.; van der Wal, S.; Müller, P.; Haug, J.T. Split-footed lacewings declined over time: Indications from the morphological diversity of their antlion-like larvae. PalZ 2022, 96, 29–50. [Google Scholar] [CrossRef]
- Haug, J.T.; Linhart, S.; Haug, G.T.; Gröhn, C.; Hoffeins, C.; Hoffeins, H.-W.; Müller, P.; Weiterschan, T.; Wunderlich, J.; Haug, C. The diversity of aphidlion-like larvae over the last 130 million years. Insects 2022, 13, 336. [Google Scholar] [CrossRef]
- Haug, J.T.; Kiesmüller, C.; Haug, G.T.; Haug, C.; Hörnig, M.K. A fossil aphidlion preserved together with its prey in 40 million-year-old Baltic amber. Palaeobio. Palaeoenv. 2020, 1–9. [Google Scholar] [CrossRef]
- Zippel, A.; Kiesmüller, C.; Haug, G.T.; Müller, P.; Weiterschan, T.; Haug, C.; Hörnig, M.K.; Haug, J.T. Long-headed predators in Cretaceous amber—fossil findings of an unusual type of lacewing larva. Palaeoentomology 2021, 4, 475–498. [Google Scholar] [CrossRef]
- Hörnig, M.K.; Haug, C.; Müller, P.; Haug, J.T. Not quite social–possible cases of gregarious behaviour of immatures of various lineages of Insecta preserved in 100-million-year-old amber. Bull. Geosci. 2022, 97, 69–87. [Google Scholar] [CrossRef]
- Luo, C.; Liu, H.; Jarzembowski, E.A. High morphological disparity of neuropteran larvae during the Cretaceous revealed by a new large species. Geol. Mag. 2022, 159, 954–962. [Google Scholar] [CrossRef]
- Haug, J.T.; Haug, C. Beetle larvae with unusually large terminal ends and a fossil that beats them all (Scraptiidae, Coleoptera). PeerJ 2019, 7, e7871. [Google Scholar] [CrossRef]
- Kerp, H.; Bomfleur, B. Photography of plant fossils—New techniques, old tricks. Rev. Palaeobot. Palynol. 2011, 166, 117–151. [Google Scholar] [CrossRef]
- Iwata, H.; Ukai, Y. SHAPE: A computer program package for quantitative evaluation of biological shapes based on elliptic Fourier descriptors. J. Hered. 2002, 93, 384–385. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.R-project.org/ (accessed on 23 June 2022).
- Guillerme, T. dispRity: A modular R package for measuring disparity. Methods Ecol. Evol. 2018, 9, 1755–1763. [Google Scholar] [CrossRef]
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016; 260p. [Google Scholar]
- Wickham, H. Reshaping Data with the reshape Package. J. Stat. Softw. 2007, 21, 1–20. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; et al. Vegan: Community Ecology Package. R Package Version 2.5-7. 2020. Available online: https://CRAN.R-project.org/package=vegan (accessed on 23 June 2022).
- Neuwirth, E. RColorBrewer: ColorBrewer Palettes. R Package Version 1.1-2. 2014. Available online: https://CRAN.R-project.org/package=RColorBrewer (accessed on 23 June 2022).
- Guillerme, T.; Puttick, M.N.; Marcy, A.E.; Weisbecker, V. Shifting spaces: Which disparity or dissimilarity measurement best summarize occupancy in multidimensional spaces? Ecol. Evol. 2020, 10, 7261–7275. [Google Scholar] [CrossRef]
- Anderson, M.J. A new method for non-parametric multivariate analysis of variance. Aust. Ecol. 2001, 26, 32–46. [Google Scholar] [CrossRef]
- Ereshefsky, M. Linnaean ranks: Vestiges of a bygone era. Philos. Sci. 2002, 69, S305–S315. [Google Scholar] [CrossRef]
- Laurin, M. The subjective nature of Linnaean categories and its impact in evolutionary biology and biodiversity studies. Contrib. Zool. 2010, 79, 131–146. [Google Scholar] [CrossRef]
- Lambertz, M.; Perry, S.F. Chordate phylogeny and the meaning of categorial ranks in modern evolutionary biology. Proc. R. Soc. B Biol. Sci. 2015, 282, 20142327. [Google Scholar] [CrossRef] [PubMed]
- Modesto, S.; Anderson, J. The phylogenetic definition of Reptilia. Syst. Biol. 2004, 53, 815–821. [Google Scholar] [CrossRef] [PubMed]
- Ábrahám, L.; Papp, Z. Preliminary report on the larva of Myrmecaelurus zigan Aspöck, Aspöck et Hölzel, 1980 (Planipennia: Myrmeleontidae). Folia Historico-Naturalia Musei Matraensis 1990, 15, 37–42. [Google Scholar]
- Acevedo, F.; Monserrat, V.J.; Badano, D. Comparative description of larvae of the European species of Distoleon Banks: D. annulatus (Klug, 1834) and D. tetragrammicus (Fabricius, 1798) (Neuroptera, Myrmeleontidae). Zootaxa 2013, 3721, 488–494. [Google Scholar] [CrossRef]
- Aspöck, H.; Aspöck, U. Synopsis der Systematik, Ökologie und Biogeographie der Neuropteren Mitteleuropas im Spiegel der Neuropteren-Fauna von Linz und Oberösterreich, sowie Bestimmungs-Schlüssel für die mitteleuropäischen Neuropteren und Beschreibung von Coniopteryx lentiae nov. spec. Naturkundliches Jahrbuch der Stadt Linz 1964, 1964, 127–282. [Google Scholar]
- Badano, D. The Larvae of European Myrmeleontidae and Ascalaphidae (Neuroptera). Ph.D. Dissertation, Università degli Studi di Sassari, Sassari, Italy, 2012. [Google Scholar]
- Brauer, F.M. Beiträge zur Kenntniss des inneren Baues und der Verwandlung der Neuropteren. Verh. Zool. -Bot. Ver. Wien 1854, 4, 463–472. [Google Scholar]
- Beutel, R.G.; Friedrich, F.; Aspöck, U. The larval head of Nevrorthidae and the phylogeny of Neuroptera (Insecta). Zool. J. Linn. Soc. 2010, 158, 533–562. [Google Scholar] [CrossRef]
- BugGuide Contributors BugGuide. 2022. Available online: https://bugguide.net (accessed on 8 May 2022).
- Gepp, J. Erforschungsstand der Neuropteren. Larven der Erde (mit einem Schlüssel zur Larvaldiagnose der Familien, einer Übersicht von 340 beschriebenen Larven und 600 Literaturzitaten). In Progress in Worlds Neuropterology. In Proceedings of the First International Symposium on Neuropterology, Graz, Austria, 21–23 August 1984; pp. 183–239. [Google Scholar]
- Henry, C.S. Some aspects of the external morphology of larval owlflies (Neuroptera: Ascalaphidae), with particular reference to Ululodes and Ascalopterynx. Psyche 1976, 83, 1–31. [Google Scholar] [CrossRef]
- Kamiya, A.; Ando, H. External Morphogenesis of the Embryo of Ascalaphus ramburi (Neuroptera, Ascalaphidae); Ando, H., Miya, K., Eds.; Recent Advances in Insect Embryology in Japan; ISEBU Co., Ltd.: Tsukuba, Japan, 1985; pp. 203–213. [Google Scholar]
- Matsuno, S. A non-destructive method for observation of body-surface fine structure of ethanol-preserved insect larvae. Jpn. J. Environ. Entomol. Zool. 2017, 28, 1–4. [Google Scholar]
- McClendon, J.F. The life history of Ulula hyalina Latreille. Am. Nat. 1902, 36, 421–429. [Google Scholar] [CrossRef]
- Miller, R.B.; Stange, L.A. A revision of the genus Eremoleon Banks (Neuroptera: Myrmeleontidae: Nemoleontini). Insecta Mundi 2016, 0495, 1–111. [Google Scholar]
- Penny, N.D. Neuroptera of the Amazon Basin. Part 3 Ascalaphidae. Acta Amaz. 1981, 11, 605–651. [Google Scholar] [CrossRef][Green Version]
- Peterson, A. Larvae of Insects. An Introduction to Nearctic Species. Part II. Coleoptera, Diptera, Neuroptera, Siphonaptera, Mecoptera, trichoptera. Larvae of Insects. An Introduction to Nearctic Species; Edward Brothers: Columbus, OH, USA, 1957; p. 416. [Google Scholar]
- Principi, M.M. Contributi allo studio dei Neurotteri italiani. II. Myrmeleon inconspicuus Ramb. ed Euroleon nostras Fourcroy. Bollettino dell’Istituto di Entomologia della R. Università degli Studi di Bologna 1943, 14, 131–192. [Google Scholar]
- Riek, E.F. Neuroptera (Lacewings); Waterhouse, D.F., Ed.; The Insects of Australia; Melbourne University Press: Melbourne, Australia, 1970; pp. 472–494. [Google Scholar]
- Satar, A.; Suludere, Z.; Canbulat, S.A.V.A.Ş.; Oezbay, C. Rearing the larval stages of Distoleon tetragrammicus (Fabricius, 1798) (Neuroptera, Myrmeleontidae) from egg to adult, with notes on their behaviour. Zootaxa 2006, 1371, 57–64. [Google Scholar] [CrossRef]
- Satar, A.; Tusun, S.; Bozdoğan, H. Third instars larvae of Gepus gibbosus Hölzel, 1968 (Neuroptera: Myrmeleontindae). Zootaxa 2014, 3793, 281–285. [Google Scholar] [CrossRef]
- Satar, A.; Tusun, S.; Aykut, M. Morphology and surface structure of third instar larvae of Solter ledereri Navás, 1912 (Neuroptera: Myrmeleontidae) from Turkey. Entomol. News 2014, 124, 67–72. [Google Scholar] [CrossRef]
- Planipennia, S.H.; Schultze, P. (Eds.) Biologie der Tiere Deutschlands. Lfg. 33, Teil 35; Borntraeger: Berlin, Germany, 1931; pp. 67–304. [Google Scholar]
- Townsend, L.H. Lacewings and their allies. Sci. Mon. 1939, 48, 350–357. [Google Scholar]
- Withycombe, C.L. XV. Some aspects of the biology and morphology of the Neuroptera. With special reference to the immature stages and their possible phylogenetic significance. Trans. R. Entomol. Soc. Lond. 1925, 72, 303–411. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haug, C.; Posada Zuluaga, V.; Zippel, A.; Braig, F.; Müller, P.; Gröhn, C.; Weiterschan, T.; Wunderlich, J.; Haug, G.T.; Haug, J.T. The Morphological Diversity of Antlion Larvae and Their Closest Relatives over 100 Million Years. Insects 2022, 13, 587. https://doi.org/10.3390/insects13070587
Haug C, Posada Zuluaga V, Zippel A, Braig F, Müller P, Gröhn C, Weiterschan T, Wunderlich J, Haug GT, Haug JT. The Morphological Diversity of Antlion Larvae and Their Closest Relatives over 100 Million Years. Insects. 2022; 13(7):587. https://doi.org/10.3390/insects13070587
Chicago/Turabian StyleHaug, Carolin, Victor Posada Zuluaga, Ana Zippel, Florian Braig, Patrick Müller, Carsten Gröhn, Thomas Weiterschan, Jörg Wunderlich, Gideon T. Haug, and Joachim T. Haug. 2022. "The Morphological Diversity of Antlion Larvae and Their Closest Relatives over 100 Million Years" Insects 13, no. 7: 587. https://doi.org/10.3390/insects13070587
APA StyleHaug, C., Posada Zuluaga, V., Zippel, A., Braig, F., Müller, P., Gröhn, C., Weiterschan, T., Wunderlich, J., Haug, G. T., & Haug, J. T. (2022). The Morphological Diversity of Antlion Larvae and Their Closest Relatives over 100 Million Years. Insects, 13(7), 587. https://doi.org/10.3390/insects13070587






