Beyond Human Nutrition of Edible Insects: Health Benefits and Safety Aspects
Abstract
Simple Summary
Abstract
1. Introduction
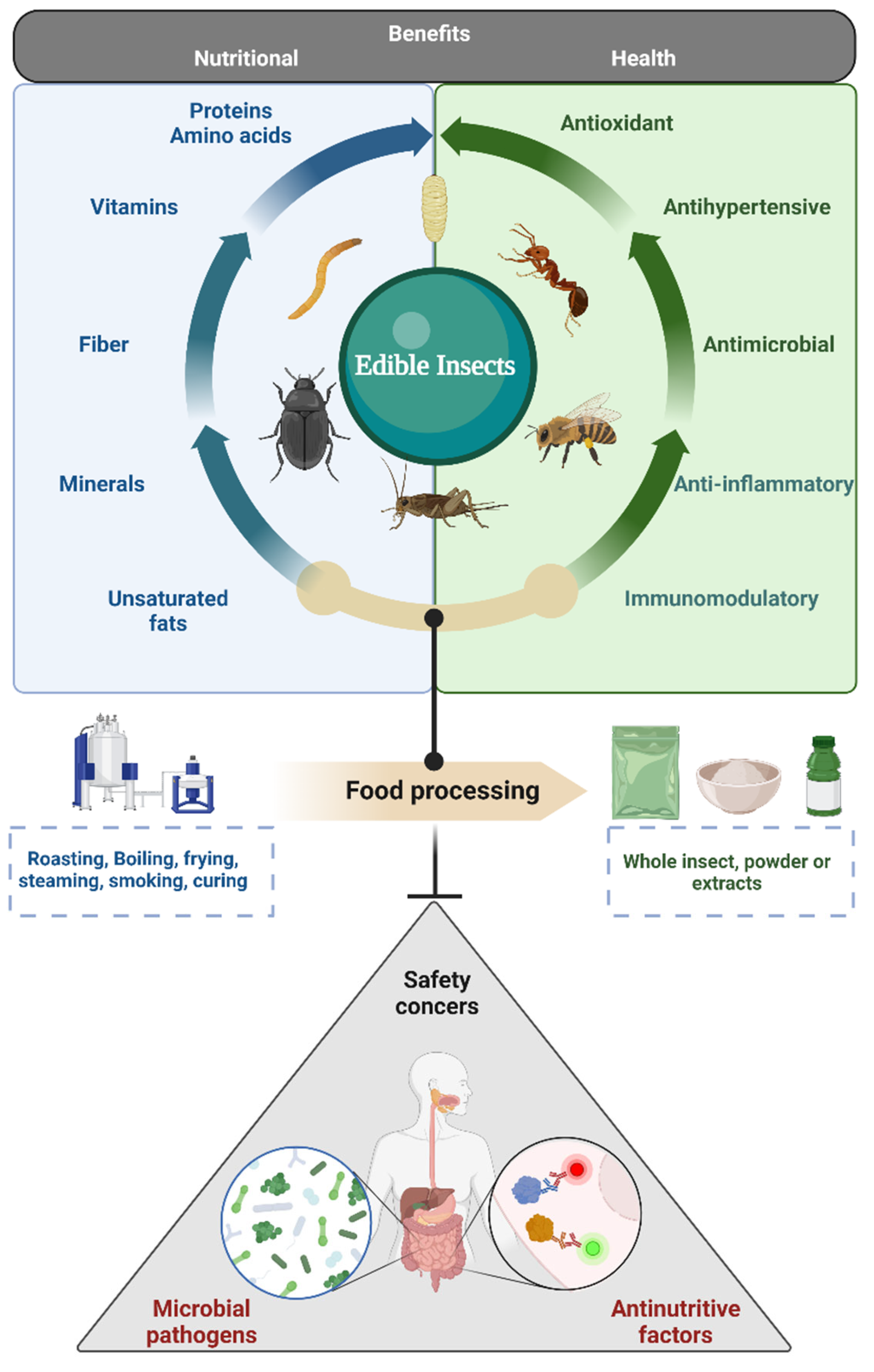
2. Health Benefits of Edible Insects
3. Safety Aspects of Edible Insects
3.1. Microbial Safety of Edible Insects
| Cooking or Processing Method | Insect Species | References |
|---|---|---|
| Boiling, blanching, steaming, sautéed | Alphitobius diaperinus (beetle) Ruspolia differens (grasshopper) Tenebrio molitor (yellow mealworm) Acheta domesticus (house cricket) | [53,54,55,56,57,58,59] |
| Drying (sun, lyophilization, fluidized-bed, microwave-assisted) | Ruspolia nitidula (grasshopper) Rhynchophorus phoenicis (palm weevil) Ruspolia differens (longhorn grasshopper) Tenebrio molitor (yellow mealworm) Polyrhachis vicina Roger (Black ant) Nauphoeta cinerea (speckled cockroach) | [53,55,56,60,61,62,63,64,65,66,67] |
| Enzyme technology; extrusion; sonication, fermentation, ultrasonication, pasteurization, microwave-assisted extractions | Acheta domesticus (house cricket) Schistocerca gregaria (desert locust) Spodoptera littoralis (leaf worm), Gryllodes sigillatus (tropical banded cricket) Tenebrio molitor (yellow mealworm) | [28,30,68,69,70,71,72,73,74] |
3.2. Anti-Nutritive Factors of Edible Insects
3.3. Other Safety Aspects of Edible Insects
3.3.1. Pesticides
3.3.2. Heavy Metals
3.3.3. Mycotoxins
3.3.4. Allergens
4. Conclusions and Future Directions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Imathiu, S. Benefits and food safety concerns associated with consumption of edible insects. NFS J. 2020, 18, 1–11. [Google Scholar] [CrossRef]
- Kim, T.-K.; Yong, H.I.; Kim, Y.-B.; Kim, H.-W.; Choi, Y.-S. Edible insects as a protein source: A review of public perception, processing technology, and research trends. Food Sci. Anim. Resour. 2019, 39, 521–540. [Google Scholar] [CrossRef] [PubMed]
- Orkusz, A. Edible insects versus meat—Nutritional comparison: Knowledge of their composition is the key to good health. Nutrients 2021, 13, 1207. [Google Scholar] [CrossRef] [PubMed]
- Zielińska, E.; Karaś, M.; Jakubczyk, A.; Zieliński, D.; Baraniak, B. Edible insects as source of proteins. In Bioactive Molecules in Food; Mérillon, J.-M., Ramawat, K.G., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 389–441. [Google Scholar]
- Liceaga, A.M.; Aguilar-Toalá, J.E.; Vallejo-Cordoba, B.; González-Córdova, A.F.; Hernández-Mendoza, A. Insects as an alternative protein source. Annu. Rev. Food Sci. Technol. 2022, 13, 19–34. [Google Scholar] [CrossRef] [PubMed]
- Searchinger, T.; Waite, R.; Hanson, C.; Ranganathan, J.; Dumas, P. Creating a Sustainable Food Future—A Menu of Solutions to Feed Nearly 10 Billion People by 205; World Resources Institute: Washington, DC, USA, 2019. [Google Scholar]
- Hall, F.; Reddivari, L.; Liceaga, A.M. Identification and characterization of edible cricket peptides on hypertensive and glycemic in vitro inhibition and their anti-inflammatory activity on RAW 264.7 macrophage cells. Nutrients 2020, 12, 3588. [Google Scholar] [CrossRef]
- Zielińska, E.; Baraniak, B.; Karaś, M. Identification of antioxidant and anti-inflammatory peptides obtained by simulated gastrointestinal digestion of three edible insects species (Gryllodes sigillatus, Tenebrio molitor, Schistocerca gragaria). Int. J. Food Sci. Technol. 2018, 53, 2542–2551. [Google Scholar] [CrossRef]
- Malm, M.; Liceaga, A.M. Physicochemical properties of chitosan from two commonly reared edible cricket species, and its application as a hypolipidemic and antimicrobial agent. Polysaccharides 2021, 2, 339–353. [Google Scholar] [CrossRef]
- Mohan, K.; Ganesan, A.R.; Muralisankar, T.; Jayakumar, R.; Sathishkumar, P.; Uthayakumar, V.; Chandirasekar, R.; Revathi, N. Recent insights into the extraction, characterization, and bioactivities of chitin and chitosan from insects. Trends Food Sci. Technol. 2020, 105, 17–42. [Google Scholar] [CrossRef]
- Nino, M.C.; Reddivari, L.; Osorio, C.; Kaplan, I.; Liceaga, A.M. Insects as a source of phenolic compounds and potential health benefits. J. Insects Food Feed 2021, 7, 1077–1087. [Google Scholar] [CrossRef]
- Baigts-Allende, D.; Doost, A.S.; Ramírez-Rodrigues, M.; Dewettinck, K.; Van der Meeren, P.; de Meulenaer, B.; Tzompa-Sosa, D. Insect protein concentrates from Mexican edible insects: Structural and functional characterization. LWT—Food Sci. Technol. 2021, 152, 112267. [Google Scholar] [CrossRef]
- Jantzen da Silva Lucas, A.; Menegon de Oliveira, L.; da Rocha, M.; Prentice, C. Edible insects: An alternative of nutritional, functional and bioactive compounds. Food Chem. 2020, 311, 126022. [Google Scholar] [CrossRef] [PubMed]
- Kouřimská, L.; Adámková, A. Nutritional and sensory quality of edible insects. NFS J. 2016, 4, 22–26. [Google Scholar] [CrossRef]
- Ekop, E.A.; Udoh, A.I.; Akpan, P.E. Proximate and anti-nutrient composition of four edible insects in Akwa Ibom State, Nigeria. World J. Appl. Sci. Technol. 2010, 2, 224–231. [Google Scholar]
- Melgar-Lalanne, G.; Hernández-Álvarez, A.-J.; Salinas-Castro, A. Edible insects processing: Traditional and innovative technologies. Compr. Rev. Food Sci. Food Saf. 2019, 18, 1166–1191. [Google Scholar] [CrossRef] [PubMed]
- Di Mattia, C.; Battista, N.; Sacchetti, G.; Serafini, M. Antioxidant activities in vitro of water and liposoluble extracts obtained by different species of edible insects and invertebrates. Front. Nutr. 2019, 6, 106. [Google Scholar] [CrossRef] [PubMed]
- Hall, F.; Johnson, P.E.; Liceaga, A. Effect of enzymatic hydrolysis on bioactive properties and allergenicity of cricket (Gryllodes sigillatus) protein. Food Chem. 2018, 262, 39–47. [Google Scholar] [CrossRef]
- Cito, A.; Botta, M.; Francardi, V.; Dreassi, E. Insects as source of angiotensin converting enzyme inhibitory peptides. J. Insects Food Feed 2017, 3, 231–240. [Google Scholar] [CrossRef]
- Tang, J.-J.; Fang, P.; Xia, H.-L.; Tu, Z.-C.; Hou, B.-Y.; Yan, Y.-M.; Di, L.; Zhang, L.; Cheng, Y.-X. Constituents from the edible Chinese black ants (Polyrhachis dives) showing protective effect on rat mesangial cells and anti-inflammatory activity. Food Res. Int. 2015, 67, 163–168. [Google Scholar] [CrossRef]
- Flores, D.R.; Casados, L.E.; Velasco, S.F.; Ramírez, A.C.; Velázquez, G. Comparative study of composition, antioxidant and antimicrobial activity of two adult edible insects from Tenebrionidae family. BMC Chem. 2020, 14, 55. [Google Scholar] [CrossRef]
- Chen, Y.; Zhao, J.; Zhang, W.; Zhao, T.; Zhang, Q.; Mao, G.; Feng, W.; Li, Q.; Yang, L. Purification of novel polypeptides from bee pupae and their immunomodulatory activity in vivo and in vitro. J. Insects Food Feed 2022, 8, 1117–1132. [Google Scholar] [CrossRef]
- Tang, Q.F.; Dai, Y. Immunomodulatory effects of supercritical fluid CO2 extracts from freeze-dried powder of Tenebrio molitor larvae (yellow mealworm). Food Sci. Technol. 2016, 36, 493–498. [Google Scholar] [CrossRef]
- Tukshipa, S.D.; Chakravorty, J.; Prveen. In vivo antioxidant and immunomodulatory effect of the aqueous extract of Mimela sp. on cyclophosphamide induced im-munocompromised mice. Int. J. Pharmacogn. Life Sci. 2022, 3, 13–19. [Google Scholar]
- Seo, M.; Goo, T.W.; Chung, M.Y.; Baek, M.; Hwang, J.S.; Kim, M.A.; Yun, E.Y. Tenebrio molitor larvae inhibit adipogenesis through AMPK and MAPKs signaling in 3T3-L1 adipocytes and obesity in high-fat diet-induced obese mice. Int. J. Mol. Sci. 2017, 18, 518. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.; Kim, K.-Y.; Kim, A.Y.; Kim, N.-S.; Kweon, H.; Ji, S.-D.; Koh, Y.H. Increased healthspan and resistance to Parkinson’s disease in Drosophila by boiled and freeze-dried mature silk worm larval powder. J. Asia-Pac. Entomol. 2016, 19, 551–561. [Google Scholar] [CrossRef]
- Mudd, N.; Martin-Gonzalez, F.S.; Ferruzzi, M.; Liceaga, A.M. In vivo antioxidant effect of edible cricket (Gryllodes sigillatus) peptides using a Caenorhabditis elegans model. Food Hydrocoll. Health 2022, 2, 100083. [Google Scholar] [CrossRef]
- Navarro del Hierro, J.; Gutiérrez-Docio, A.; Otero, P.; Reglero, G.; Martin, D. Characterization, antioxidant activity, and inhibitory effect on pancreatic lipase of extracts from the edible insects Acheta domesticus and Tenebrio molitor. Food Chem. 2020, 309, 125742. [Google Scholar] [CrossRef] [PubMed]
- Nino, M.C.; Reddivari, L.; Ferruzzi, M.G.; Liceaga, A.M. Targeted phenolic characterization and antioxidant bioactivity of extracts from edible Acheta domesticus. Foods 2021, 10, 2295. [Google Scholar] [CrossRef] [PubMed]
- Mendoza-Salazar, A.; Santiago-López, L.; Torres-Llanez, M.J.; Hernández-Mendoza, A.; Vallejo-Cordoba, B.; Liceaga, A.M.; González-Córdova, A.F. In vitro antioxidant and antihypertensive activity of edible insects flours (mealworm and grasshopper) fermented with Lactococcus lactis strains. Fermentation 2021, 7, 153. [Google Scholar] [CrossRef]
- Rumbos, C.I.; Karapanagiotidis, I.T.; Mente, E.; Psofakis, P.; Athanassiou, C.G. Evaluation of various commodities for the development of the yellow mealworm, Tenebrio molitor. Sci. Rep. 2020, 10, 11224. [Google Scholar] [CrossRef]
- Bergmans, R.S.; Nikodemova, M.; Stull, V.J.; Rapp, A.; Malecki, K.M.C. Comparison of cricket diet with peanut-based and milk-based diets in the recovery from protein malnutrition in mice and the impact on growth, metabolism and immune function. PLoS ONE 2020, 15, e0234559. [Google Scholar] [CrossRef]
- Baiano, A. Edible insects: An overview on nutritional characteristics, safety, farming, production technologies, regulatory framework, and socio-economic and ethical implications. Trends Food Sci. Technol. 2020, 100, 35–50. [Google Scholar] [CrossRef]
- Dillon, R.J.; Dillon, V.M. The gut bacteria of insects: Nonpathogenic interactions. Annu. Rev. Entomol. 2003, 49, 71–92. [Google Scholar] [CrossRef] [PubMed]
- Douglas, A.E. Multiorganismal insects: Diversity and function of resident microorganisms. Annu. Rev. Entomol. 2015, 60, 17–34. [Google Scholar] [CrossRef] [PubMed]
- Schlüter, O.; Rumpold, B.; Holzhauser, T.; Roth, A.; Vogel, R.F.; Quasigroch, W.; Vogel, S.; Heinz, V.; Jäger, H.; Bandick, N.; et al. Safety aspects of the production of foods and food ingredients from insects. Mol. Nutr. Food Res. 2017, 61, 1600520. [Google Scholar] [CrossRef] [PubMed]
- FAO Edible insects: Future prospects for food and feed security. Int. For. Rev. 2013, 16, 112.
- EFSA Risk profile related to production and consumption of insects as food and feed. EFSA J. 2015, 13, 4257. [CrossRef]
- Stoops, J.; Crauwels, S.; Waud, M.; Claes, J.; Lievens, B.; Van Campenhout, L. Microbial community assessment of mealworm larvae (Tenebrio molitor) and grasshoppers (Locusta migratoria migratorioides) sold for human consumption. Food Microbiol. 2016, 53, 122–127. [Google Scholar] [CrossRef]
- Nyenje, M.E.; Odjadjare, C.E.; Tanih, N.F.; Green, E.; Ndip, R.N. Foodborne pathogens recovered from ready-to-eat foods from roadside cafeterias and retail outlets in Alice, Eastern Cape Province, South Africa: Public health implications. Int. J. Environ. Res. Public Health 2012, 9, 2608–2619. [Google Scholar] [CrossRef] [PubMed]
- Klunder, H.C.; Wolkers-Rooijackers, J.; Korpela, J.M.; Nout, M.J.R. Microbiological aspects of processing and storage of edible insects. Food Control 2012, 26, 628–631. [Google Scholar] [CrossRef]
- Ali, A.; Mohamadou, B.A.; Saidou, C.; Aoudou, Y.; Tchiegang, C. Physio-chemical properties and safety of grasshoppers, important contributors to food security in far north region of Cameroon. Res. J. Anim. Sci. 2010, 4, 108–111. [Google Scholar]
- Chen, X.; Feng, Y.; Chen, Z. Common edible insects and their utilization in China. Entomol. Res. 2009, 39, 299–303. [Google Scholar] [CrossRef]
- Grabowski, N.T.; Klein, G. Microbiology of processed edible insect products–Results of a preliminary survey. Int. J. Food Microbiol. 2017, 243, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Mujuru, F.M.; Kwiri, R.; Nyambi, C.; Winini, C.; Moyo, D.N. Microbiological quality of Gonimbrasia belina processed under different traditional practices in Gwanda, Zimbabwe. Int. J. Curr. Microbiol. Appl. Sci. 2014, 3, 1085–1094. [Google Scholar]
- Garofalo, C.; Osimani, A.; Milanović, V.; Taccari, M.; Cardinali, F.; Aquilanti, L.; Riolo, P.; Ruschioni, S.; Isidoro, N.; Clementi, F. The microbiota of marketed processed edible insects as revealed by high-throughput sequencing. Food Microbiol. 2017, 62, 15–22. [Google Scholar] [CrossRef]
- Bonaccorsi, G.; Simoncini, E.; Gestri, D.; Talini, M.; Tesi, S.; Rastelli, F.; Nardone, G.; Lorini, C. The microbial safety of edible insects: A matter of processing. Ann. Ist. Super. Sanita 2019, 55, 203–204. [Google Scholar]
- Liceaga, A.M. Processing insects for use in the food and feed industry. Curr. Opin. Insect Sci. 2021, 48, 32–36. [Google Scholar] [CrossRef]
- Efsa Panel on Nutrition, N.F.; Food, A.; Turck, D.; Bohn, T.; Castenmiller, J.; De Henauw, S.; Hirsch-Ernst, K.I.; Maciuk, A.; Mangelsdorf, I.; McArdle, H.J.; et al. Safety of frozen and dried formulations from whole house crickets (Acheta domesticus) as a Novel food pursuant to Regulation (EU) 2015/2283. EFSA J. 2021, 19, e06779. [Google Scholar] [CrossRef]
- Fellows, P.J. 10-Heat processing. In Food Processing Technology, 3rd ed.; Fellows, P.J., Ed.; Woodhead Publishing: Cambridge, UK, 2009; pp. 339–366. [Google Scholar]
- Sruthy, G.N.; Sandhya, K.R.; Kumkum, C.R.; Mythri, R.; Sharma, M. Thermal processing technologies for food. In Current Developments in Biotechnology and Bioengineering; Tarafdar, A., Pandey, A., Sirohi, R., Soccol, C., Dussap, C.-G., Eds.; Elsevier: Oxford, UK, 2022; Chapter 10; pp. 263–300. [Google Scholar]
- Liceaga, A.M. Edible insects, a valuable protein source from ancient to modern times. Adv. Food Nutr. Res. 2022, 101, 129. [Google Scholar]
- Wynants, E.; Crauwels, S.; Verreth, C.; Gianotten, N.; Lievens, B.; Claes, J.; Van Campenhout, L. Microbial dynamics during production of lesser mealworms (Alphitobius diaperinus) for human consumption at industrial scale. Food Microbiol. 2018, 70, 181–191. [Google Scholar] [CrossRef]
- Grabowski, N.T.; Klein, G. Microbiology of cooked and dried edible Mediterranean field crickets (Gryllus bimaculatus) and superworms (Zophobas atratus) submitted to four different heating treatments. Food Sci. Technol. Int. 2017, 23, 17–23. [Google Scholar] [CrossRef]
- Fombong, F.T.; Van Der Borght, M.; Vanden Broeck, J. Influence of freeze-drying and oven-drying post blanching on the nutrient composition of the edible insect Ruspolia differens. Insects 2017, 8, 102. [Google Scholar] [CrossRef] [PubMed]
- Purschke, B.; Brüggen, H.; Scheibelberger, R.; Jäger, H. Effect of pre-treatment and drying method on physico-chemical properties and dry fractionation behaviour of mealworm larvae (Tenebrio molitor L.). Eur. Food Res. Technol. 2018, 244, 269–280. [Google Scholar] [CrossRef]
- Kamau, E.; Mutungi, C.; Kinyuru, J.; Imathiu, S.; Tanga, C.; Affognon, H.; Ekesi, S.; Nakimbugwe, D.; Fiaboe, K. Moisture adsorption properties and shelf-life estimation of dried and pulverised edible house cricket Acheta domesticus (L.) and black soldier fly larvae Hermetia illucens (L.). Food Res. Int. 2018, 106, 420–427. [Google Scholar] [CrossRef] [PubMed]
- Ssepuuya, G.; Aringo, R.; Mukisa, I.; Nakimbugwe, D. Effect of processing, packaging and storage-temperature based hurdles on the shelf stability of sautéed ready-to-eat Ruspolia nitidula. J. Insects Food Feed 2016, 2, 245–253. [Google Scholar] [CrossRef]
- Nyangena, D.N.; Mutungi, C.; Imathiu, S.; Kinyuru, J.; Affognon, H.; Ekesi, S.; Nakimbugwe, D.; Fiaboe, K.K. Effects of traditional processing techniques on the nutritional and microbiological quality of four edible insect species used for food and feed in East Africa. Foods 2020, 9, 574. [Google Scholar] [CrossRef]
- Tiencheu, B.; Womeni, H.M.; Linder, M.; Mbiapo, F.T.; Villeneuve, P.; Fanni, J.; Parmentier, M. Changes of lipids in insect (Rhynchophorus phoenicis) during cooking and storage. Eur. J. Lipid Sci. Technol. 2013, 115, 186–195. [Google Scholar] [CrossRef]
- Alves, A.V.; Sanjinez-Argandoña, E.J.; Linzmeier, A.M.; Cardoso, C.A.L.; Macedo, M.L.R. Food value of mealworm grown on Acrocomia aculeata pulp flour. PLoS ONE 2016, 11, e0151275. [Google Scholar] [CrossRef]
- Bußler, S.; Rumpold, B.A.; Fröhling, A.; Jander, E.; Rawel, H.M.; Schlüter, O.K. Cold atmospheric pressure plasma processing of insect flour from Tenebrio molitor: Impact on microbial load and quality attributes in comparison to dry heat treatment. Innov. Food Sci. Emerg. Technol. 2016, 36, 277–286. [Google Scholar] [CrossRef]
- Vandeweyer, D.; Lenaerts, S.; Callens, A.; Van Campenhout, L. Effect of blanching followed by refrigerated storage or industrial microwave drying on the microbial load of yellow mealworm larvae (Tenebrio molitor). Food Control 2017, 71, 311–314. [Google Scholar] [CrossRef]
- Kröncke, N.; Böschen, V.; Woyzichovski, J.; Demtröder, S.; Benning, R. Comparison of suitable drying processes for mealworms (Tenebrio molitor). Innov. Food Sci. Emerg. Technol. 2018, 50, 20–25. [Google Scholar] [CrossRef]
- de Oliveira, L.M.; da Silva Lucas, A.J.; Cadaval, C.L.; Mellado, M.S. Bread enriched with flour from cinereous cockroach (Nauphoeta cinerea). Innov. Food Sci. Emerg. Technol. 2017, 44, 30–35. [Google Scholar] [CrossRef]
- Lenaerts, S.; Van Der Borght, M.; Callens, A.; Van Campenhout, L. Suitability of microwave drying for mealworms (Tenebrio molitor) as alternative to freeze drying: Impact on nutritional quality and colour. Food Chem. 2018, 254, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Álvarez, A.-J.; Mondor, M.; Piña-Domínguez, I.-A.; Sánchez-Velázquez, O.-A.; Melgar Lalanne, G. Drying technologies for edible insects and their derived ingredients. Dry. Technol. 2021, 39, 1991–2009. [Google Scholar] [CrossRef]
- Hall, F.G.; Jones, O.G.; O’Haire, M.E.; Liceaga, A.M. Functional properties of tropical banded cricket (Gryllodes sigillatus) protein hydrolysates. Food Chem. 2017, 224, 414–422. [Google Scholar] [CrossRef]
- Zhao, X.; Vázquez-Gutiérrez, J.L.; Johansson, D.P.; Landberg, R.; Langton, M. Yellow mealworm protein for food purposes-extraction and functional properties. PLoS ONE 2016, 11, e0147791. [Google Scholar] [CrossRef]
- Mishyna, M.; Martinez, J.-J.I.; Chen, J.; Benjamin, O. Extraction, characterization and functional properties of soluble proteins from edible grasshopper (Schistocerca gregaria) and honey bee (Apis mellifera). Food Res. Int. 2019, 116, 697–706. [Google Scholar] [CrossRef] [PubMed]
- Zielińska, E.; Karaś, M.; Baraniak, B. Comparison of functional properties of edible insects and protein preparations thereof. LWT-Food Sci. Technol. 2018, 91, 168–174. [Google Scholar] [CrossRef]
- Carcea, M. Quality and nutritional/textural properties of durum wheat pasta enriched with cricket powder. Foods 2020, 9, 1298. [Google Scholar] [CrossRef]
- Otero, P.; Gutierrez-Docio, A.; Del Hierro, J.N.; Reglero, G.; Martin, D. Extracts from the edible insects Acheta domesticus and Tenebrio molitor with improved fatty acid profile due to ultrasound assisted or pressurized liquid extraction. Food Chem. 2020, 314, 126200. [Google Scholar] [CrossRef]
- Adámková, A.; Mlček, J.; Kouřimská, L.; Borkovcová, M.; Bušina, T.; Adámek, M.; Bednářová, M.; Krajsa, J. Nutritional potential of selected insect species reared on the island of Sumatra. Int. J. Environ. Res. Public Health 2017, 14, 521. [Google Scholar] [CrossRef]
- Stull, V.J.; Finer, E.; Bergmans, R.S.; Febvre, H.P.; Longhurst, C.; Manter, D.K.; Patz, J.A.; Weir, T.L. Impact of edible cricket consumption on gut microbiota in healthy adults, a double-blind, randomized crossover trial. Sci. Rep. 2018, 8, 10762. [Google Scholar] [CrossRef] [PubMed]
- Selenius, O.; Korpela, J.; Salminen, S.; Gallego, C.G. Effect of chitin and chitooligosaccharide on in vitro growth of Lactobacillus rhamnosus GG and Escherichia coli TG. Appl. Food Biotechnol. 2018, 5, 163–172. [Google Scholar]
- Fernandes, J.C.; Tavaria, F.K.; Soares, J.C.; Ramos, Ó.S.; João Monteiro, M.; Pintado, M.E.; Xavier Malcata, F. Antimicrobial effects of chitosans and chitooligosaccharides, upon Staphylococcus aureus and Escherichia coli, in food model systems. Food Microbiol. 2008, 25, 922–928. [Google Scholar] [CrossRef] [PubMed]
- Gemede, H.F.; Ratta, N. Antinutritional factors in plant foods: Potential health benefits and adverse effects. Int. J. Nutr. Food Sci. 2014, 3, 284–289. [Google Scholar] [CrossRef]
- Musundire, R.; Zvidzai, C.J.; Chidewe, C.; Samende, B.K.; Manditsera, F.A. Nutrient and anti-nutrient composition of Henicus whellani (Orthoptera: Stenopelmatidae), an edible ground cricket, in south-eastern Zimbabwe. Int. J. Trop. Insect Sci. 2014, 34, 223–231. [Google Scholar] [CrossRef]
- Omotoso, O.T. Nutritional quality, functional properties and anti-nutrient compositions of the larva of Cirina forda (Westwood) (Lepidoptera: Saturniidae). J. Zhejiang Univ. Sci. B 2006, 7, 51–55. [Google Scholar] [CrossRef]
- Nishimune, T.; Watanabe, Y.; Okazaki, H.; Akai, H. Thiamin is decomposed due to Anaphe spp. entomophagy in seasonal ataxia patients in Nigeria. J. Nutr. 2000, 130, 1625–1628. [Google Scholar] [CrossRef]
- Bush, R.K. Indoor allergens, environmental avoidance, and allergic respiratory disease. Allergy Asthma Proc. 2008, 29, 575–579. [Google Scholar] [CrossRef]
- Murefu, T.R.; Macheka, L.; Musundire, R.; Manditsera, F.A. Safety of wild harvested and reared edible insects: A review. Food Control 2019, 101, 209–224. [Google Scholar] [CrossRef]
- De Paepe, E.; Wauters, J.; Van Der Borght, M.; Claes, J.; Huysman, S.; Croubels, S.; Vanhaecke, L. Ultra-high-performance liquid chromatography coupled to quadrupole orbitrap high-resolution mass spectrometry for multi-residue screening of pesticides, (veterinary) drugs and mycotoxins in edible insects. Food Chem. 2019, 293, 187–196. [Google Scholar] [CrossRef]
- Poma, G.; Cuykx, M.; Amato, E.; Calaprice, C.; Focant, J.F.; Covaci, A. Evaluation of hazardous chemicals in edible insects and insect-based food intended for human consumption. Food Chem. Toxicol. 2017, 100, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Calatayud-Vernich, P.; Calatayud, F.; Simó, E.; Picó, Y. Pesticide residues in honey bees, pollen and beeswax: Assessing beehive exposure. Environ. Pollut. 2018, 241, 106–114. [Google Scholar] [CrossRef] [PubMed]
- Houbraken, M.; Spranghers, T.; De Clercq, P.; Cooreman-Algoed, M.; Couchement, T.; De Clercq, G.; Verbeke, S.; Spanoghe, P. Pesticide contamination of Tenebrio molitor (Coleoptera: Tenebrionidae) for human consumption. Food Chem. 2016, 201, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Bayo, F. Indirect effect of pesticides on insects and other arthropods. Toxics 2021, 9, 177. [Google Scholar] [CrossRef]
- Brühl, C.A.; Bakanov, N.; Köthe, S.; Eichler, L.; Sorg, M.; Hörren, T.; Mühlethaler, R.; Meinel, G.; Lehmann, G.U.C. Direct pesticide exposure of insects in nature conservation areas in Germany. Sci. Rep. 2021, 11, 24144. [Google Scholar] [CrossRef]
- Rani, L.; Thapa, K.; Kanojia, N.; Sharma, N.; Singh, S.; Grewal, A.S.; Srivastav, A.L.; Kaushal, J. An extensive review on the consequences of chemical pesticides on human health and environment. J. Clean. Prod. 2021, 283, 124657. [Google Scholar] [CrossRef]
- Saeed, T.; Dagga, F.; Saraf, M. Analysis of residual pesticides present in edible locusts captured in Kuwait. Arab. Gulf J. Sci. Res. 1993, 11, 1–5. [Google Scholar]
- Sabarwal, A.; Kumar, K.; Singh, R.P. Hazardous effects of chemical pesticides on human health–Cancer and other associated disorders. Environ. Toxicol. Pharmacol. 2018, 63, 103–114. [Google Scholar] [CrossRef]
- Lushchak, V.I.; Matviishyn, T.M.; Husak, V.V.; Storey, J.M.; Storey, K.B. Pesticide toxicity: A mechanistic approach. EXCLI J. 2018, 17, 1101–1136. [Google Scholar]
- Greenfield, R.; Akala, N.; van der Bank, F.H. Heavy metal concentrations in two populations of mopane worms (Imbrasia belina) in the Kruger National Park pose a potential human health risk. Bull. Environ. Contam. Toxicol. 2014, 93, 316–321. [Google Scholar] [CrossRef]
- Zhang, Z.-S.; Lu, X.-G.; Wang, Q.-C.; Zheng, D.-M. Mercury, cadmium and lead biogeochemistry in the soil–plant–insect system in Huludao City. Bull. Environ. Contam. Toxicol. 2009, 83, 255–259. [Google Scholar] [CrossRef] [PubMed]
- Banjo, A.; Lawal, O.; Fasunwon, B.; Alimi, G. Alkali and heavy metal contaminants of some selected edible arthropods in South Western Nigeria. Am.-Eurasian J. Toxicol. Sci. 2010, 2, 25–29. [Google Scholar]
- Hyun, S.-H.; Kwon, K.H.; Park, K.-H.; Jeong, H.C.; Kwon, O.; Tindwa, H.; Han, Y.S. Evaluation of nutritional status of an edible grasshopper, Oxya chinensis formosana. Entomol. Res. 2012, 42, 284–290. [Google Scholar] [CrossRef]
- Alengebawy, A.; Abdelkhalek, S.T.; Qureshi, S.R.; Wang, M.-Q. Heavy metals and pesticides toxicity in agricultural soil and plants: Ecological risks and human health implications. Toxics 2021, 9, 42. [Google Scholar] [CrossRef] [PubMed]
- Mirza Alizadeh, A.; Golzan, S.A.; Mahdavi, A.; Dakhili, S.; Torki, Z.; Hosseini, H. Recent advances on the efficacy of essential oils on mycotoxin secretion and their mode of action. Crit. Rev. Food Sci. Nutr. 2022, 62, 4726–4751. [Google Scholar] [CrossRef]
- Omotayo, O.P.; Omotayo, A.O.; Mwanza, M.; Babalola, O.O. Prevalence of mycotoxins and their consequences on human health. Toxicol. Res. 2019, 35, 1–7. [Google Scholar] [CrossRef]
- Pradanas-González, F.; Álvarez-Rivera, G.; Benito-Peña, E.; Navarro-Villoslada, F.; Cifuentes, A.; Herrero, M.; Moreno-Bondi, M.C. Mycotoxin extraction from edible insects with natural deep eutectic solvents: A green alternative to conventional methods. J. Chromatogr. A 2021, 1648, 462180. [Google Scholar] [CrossRef]
- Musundire, R.; Osuga, I.M.; Cheseto, X.; Irungu, J.; Torto, B. Aflatoxin contamination detected in nutrient and anti-oxidant rich edible stink bug stored in recycled grain containers. PLoS ONE 2016, 11, e0145914. [Google Scholar] [CrossRef]
- de Gier, S.; Verhoeckx, K. Insect (food) allergy and allergens. Mol. Immunol. 2018, 100, 82–106. [Google Scholar] [CrossRef]
- Feng, Y.; Chen, X.M.; Zhao, M.; He, Z.; Sun, L.; Wang, C.Y.; Ding, W.F. Edible insects in China: Utilization and prospects. Insect Sci. 2018, 25, 184–198. [Google Scholar] [CrossRef]
- Ribeiro, J.C.; Cunha, L.M.; Sousa-Pinto, B.; Fonseca, J. Allergic risks of consuming edible insects: A systematic review. Mol. Nutr. Food Res. 2018, 62, 1700030. [Google Scholar] [CrossRef] [PubMed]
- Pali-Schöll, I.; Meinlschmidt, P.; Larenas-Linnemann, D.; Purschke, B.; Hofstetter, G.; Rodríguez-Monroy, F.A.; Einhorn, L.; Mothes-Luksch, N.; Jensen-Jarolim, E.; Jäger, H. Edible insects: Cross-recognition of IgE from crustacean-and house dust mite allergic patients, and reduction of allergenicity by food processing. World Allergy Organ. J. 2019, 12, 100006. [Google Scholar] [CrossRef] [PubMed]
- Ayuso, R.; Reese, G.; Leong-Kee, S.; Plante, M.; Lehrer, S.B. Molecular basis of arthropod cross-reactivity: IgE-binding cross-reactive epitopes of shrimp, house dust mite and cockroach tropomyosins. Int. Arch. Allergy Immunol. 2002, 129, 38–48. [Google Scholar]
- Wong, L.; Huang, C.H.; Lee, B.W. Shellfish and house dust mite allergies: Is the link tropomyosin? Allergy Asthma Immunol. Res. 2016, 8, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Hall, F.; Liceaga, A. Isolation and proteomic characterization of tropomyosin extracted from edible insect protein. Food Chem. Mol. Sci. 2021, 3, 100049. [Google Scholar] [CrossRef]
- Abdelmoteleb, M.; Palmer, L.K.; Pavlovikj, N.; Marsh, J.T.; Johnson, P.E.; Goodman, R.E. Bioinformatics and proteomics evaluations to consider IgE binding assays for potential cross-reactivity between house-cricket (Acheta domesticus) used in food, crustaceans and cockroaches. J. Allergy Clin. Immunol. 2018, 141, AB263. [Google Scholar] [CrossRef]
- Lukiwati, D.R. Teak caterpillars and other edible insects in Java. In Forest Insects as Food: Humans Bite Back; FAO: Bangkok, Thailand, 2010; p. 99. [Google Scholar]
- Liu, X.; Tian, J.; Chen, D. An allergic shock case that resulted from consuming silkworm pupae. Chin. Recuper. Med. 2001, 10, 80. [Google Scholar]
- Vetter, R. A case of ingestant allergy from eating a grasshopper. Food Insects Newsl. 1995, 8. [Google Scholar]
- Srivastava, S.; Babu, N.; Pandey, H. Traditional insect bioprospecting-As human food and medicine. Indian J. Tradit. Knowl. 2009, 8, 485–494. [Google Scholar]
- Volpicella, M.; Leoni, C.; Dileo, M.C.; Ceci, L.R. Progress in the analysis of food allergens through molecular biology approaches. Cells 2019, 8, 1073. [Google Scholar]
- Leoni, C.; Volpicella, M.; Dileo, M.C.; Gattulli, B.A.; Ceci, L.R. Chitinases as food allergens. Molecules 2019, 24, 2087. [Google Scholar] [CrossRef] [PubMed]
- MacEvilly, C. Bugs in the system. Nutr. Bull. 2000, 25, 267–268. [Google Scholar]
- Zhao, R.-J.; Huo, C.-Y.; Qian, Y.; Ren, D.-F.; Lu, J. Ultra-high-pressure processing improves proteolysis and release of bioactive peptides with activation activities on alcohol metabolic enzymes in vitro from mushroom food protein. Food Chem. 2017, 231, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Rao, H.; Zhang, K.; Tao, S.; Xue, W.T. Effects of different thermal processing methods on the structure and allergenicity of peanut allergen Ara h 1. Food Sci. Nutr. 2018, 6, 1706–1714. [Google Scholar] [CrossRef] [PubMed]
- Mills, E.C.; Mackie, A.R. The impact of processing on allergenicity of food. Curr. Opin. Allergy Clin. Immunol. 2008, 8, 249–253. [Google Scholar] [CrossRef] [PubMed]
- Broekman, H.; Knulst, A.; den Hartog Jager, S.; Monteleone, F.; Gaspari, M.; De Jong, G.; Houben, G.; Verhoeckx, K. Effect of thermal processing on mealworm allergenicity. Mol. Nutr. Food Res. 2015, 59, 1855–1864. [Google Scholar] [CrossRef]
- Phiriyangkul, P.; Srinroch, C.; Srisomsap, C.; Chokchaichamnankit, D.; Punyarit, P. Effect of food thermal processing on allergenicity proteins in Bombay locust (Patanga succincta). Int. J. Food Eng. 2015, 1, 23–28. [Google Scholar]
- Van Broekhoven, S.; Bastiaan-Net, S.; de Jong, N.W.; Wichers, H.J. Influence of processing and in vitro digestion on the allergic cross-reactivity of three mealworm species. Food Chem. 2016, 196, 1075–1083. [Google Scholar]
- Izquierdo, F.J.; Peñas, E.; Baeza, M.L.; Gomez, R. Effects of combined microwave and enzymatic treatments on the hydrolysis and immunoreactivity of dairy whey proteins. Int. Dairy J. 2008, 18, 918–922. [Google Scholar] [CrossRef]
- Abd El-Salam, M.H.; El-Shibiny, S. Reduction of milk protein antigenicity by enzymatic hydrolysis and fermentation. A review. Food Rev. Int. 2019, 37, 276–295. [Google Scholar]
| Insect Species | Sample Used or Bioactive Compound Identified | Type of Study | Bioactivities | Reference |
|---|---|---|---|---|
| Mealworms (Tenebrio molitor), buffalo worms (Alphitobius diaperinus), Palm worm larvae (Rhynchophorus ferrugineus), Evening cicada (Tanna japonensis), Black ants (Lasius niger), African caterpillars (Imbrasia oyemensis), Silkworm (Bombyx mori), Grasshoppers (Calliptamus italicus), Crickets (Acheta domesticus), Mini crickets (Acheta domesticus), Giant water bugs (Lethocerus indicus), Scolopendra gigantea (Scolopendra) | Water and liposoluble extracts | In vitro | Antioxidant | [17] |
| Crickets (Gryllodes sigillatus) | Bioactive peptides | In vitro | Antioxidant, antihypertensive, antidiabetic, antiglycemic, anti-inflammatory | [7,18] |
| Weaver ants (Polyrhachis dives) | Thirteen non-peptide nitrogen compounds (most were identified as alkaloids) | In vitro | Anti-inflammatory, immunosuppressive, renoprotective | [20] |
| House cricket (Acheta domesticus) and tropical banded cricket (Gryllodes sigillatus) | Chitin and chitosan | In vitro | Hypolipidemic, antimicrobial | [9] |
| Mealworms (Tenebrio molitor) and Chinese beetle (Ulomoides dermestoides) | Extract with main components of saponins, carbohydrates, and proteins | In vitro | Antioxidant and antimicrobial | [21] |
| Bee pupae | Polypeptide components | In vitro and in vivo | Immunomodulatory | [22] |
| Mealworms (Tenebrio molitor) | Supercritical fluid CO2 extract | In vivo | Immunomodulatory | [23] |
| Green beetle (Mimela sp.) | Aqueous extract | In vivo | Antioxidant and immunomodulatory | [24] |
| Mealworms (Tenebrio molitor) | Ethanol extract | In vitro and in vivo | Anti-adipogenic and antiobesity | [25] |
| Silkworm (Bombyx mori) | Powder | In vivo | Anti-Parkinson activity | [26] |
| Crickets (Gryllodes sigillatus) | Bioactive peptides | In vivo | Antioxidant | [27] |
| House cricket (Acheta domesticus) and mealworms (Tenebrio molitor) | Polyphenolic ethanol and ethanol: water extracts | In vitro | Antioxidant and antiobesity | [28] |
| House cricket (Acheta domesticus) | Polyphenolic methanolic extracts | In vitro | Antioxidant | [29] |
| Mealworm (Tenebrio molitor) and grasshopper (Sphenarium purpurascens) | Flour fermented with Lactococcus lactis strains | In vitro | Antioxidant and antihypertensive | [30] |
| Silkworm (Bombyx mori) | Oil | In vivo | Antioxidant and anti-dyslipidemia | [26] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aguilar-Toalá, J.E.; Cruz-Monterrosa, R.G.; Liceaga, A.M. Beyond Human Nutrition of Edible Insects: Health Benefits and Safety Aspects. Insects 2022, 13, 1007. https://doi.org/10.3390/insects13111007
Aguilar-Toalá JE, Cruz-Monterrosa RG, Liceaga AM. Beyond Human Nutrition of Edible Insects: Health Benefits and Safety Aspects. Insects. 2022; 13(11):1007. https://doi.org/10.3390/insects13111007
Chicago/Turabian StyleAguilar-Toalá, José E., Rosy G. Cruz-Monterrosa, and Andrea M. Liceaga. 2022. "Beyond Human Nutrition of Edible Insects: Health Benefits and Safety Aspects" Insects 13, no. 11: 1007. https://doi.org/10.3390/insects13111007
APA StyleAguilar-Toalá, J. E., Cruz-Monterrosa, R. G., & Liceaga, A. M. (2022). Beyond Human Nutrition of Edible Insects: Health Benefits and Safety Aspects. Insects, 13(11), 1007. https://doi.org/10.3390/insects13111007









