Effects of Climatic Change on the Potential Distribution of Lycoriella Species (Diptera: Sciaridae) of Economic Importance
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Occurrence Data
2.2. Environmental Variables
2.3. Model Calibration and Evaluation
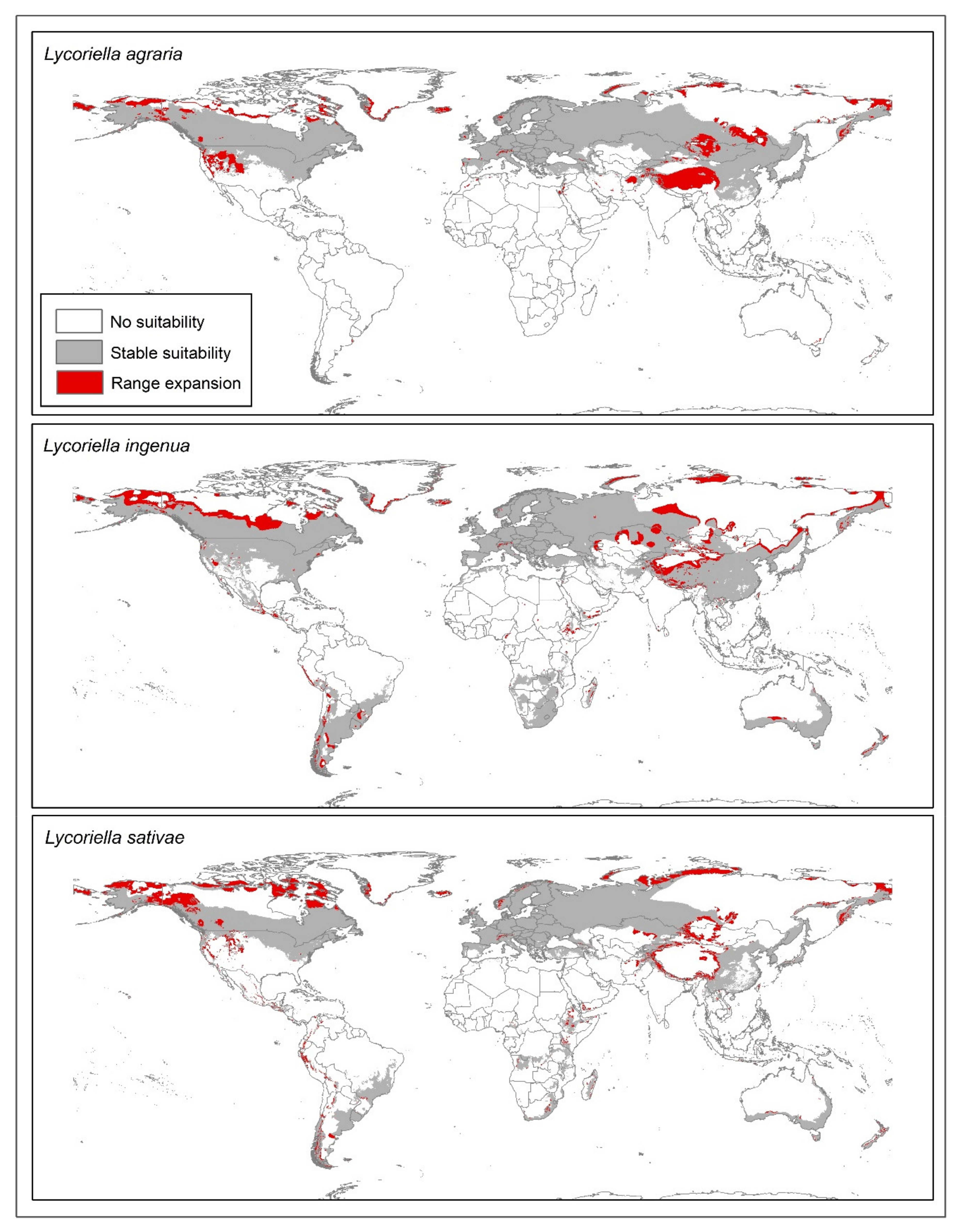
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vilkamaa, P. Checklist of the family Sciaridae (Diptera) of Finland. Zookeys 2014, 441, 151–164. [Google Scholar] [CrossRef] [Green Version]
- Han, Q.X.; Cheng, D.M.; Luo, J.; Zhou, C.Z.; Lin, Q.S.; Xiang, M.M. First report of Bradysia difformis (Diptera: Sciaridae) damage to Phalaenopsis orchid in China. J. Asia-Pac. Entomol. 2015, 18, 77–81. [Google Scholar] [CrossRef]
- Mohrig, W.; Heller, K.; Hippa, H.; Vilkamaa, P.; Menzel, F. Revision of black fungus gnats (Diptera: Sciaridae) of North America. Studia Dipterol. 2013, 19, 141–286. [Google Scholar]
- Shin, S.-G.; Lee, H.-S.; Lee, S. Dark winged fungus gnats (Diptera: Sciaridae) collected from shiitake mushroom in Korea. J. Asia-Pac. Entomol. 2012, 15, 174–181. [Google Scholar] [CrossRef]
- Ye, L.; Leng, R.; Huang, J.; Qu, C.; Wu, H. Review of three black fungus gnat species (Diptera: Sciaridae) from greenhouses in China: Three greenhouse sciarids from China. J. Asia-Pac. Entomol. 2017, 20, 179–184. [Google Scholar] [CrossRef]
- Shamshad, A. The development of integrated pest management for the control of mushroom sciarid flies, Lycoriella ingenua (Dufour) and Bradysia ocellaris (Comstock), in cultivated mushrooms. Pest Manag. Sci. 2010, 66, 1063–1074. [Google Scholar] [CrossRef]
- Grimm, D.; Wösten, H.A.B. Mushroom cultivation in the circular economy. Appl. Microbiol. Biotechnol. 2018, 102, 7795–7803. [Google Scholar] [CrossRef] [Green Version]
- Royse, D.J.; Baars, J.; Tan, Q. Current overview of mushroom production in the world. In Edible and Medicinal Mushrooms; Diego, C.Z., Pardo-Giménez, A., Eds.; John Wiley & Sons, Ltd.: Chichester, UK, 2017; pp. 5–13. [Google Scholar] [CrossRef]
- National Agricultural Statistics Service (USDA), Agricultural Statistics Board. Mushrooms. Washington, DC, USA, 2020. Available online: https://www.nass.usda.gov/Statistics_by_State/Wisconsin/Publications/Crops/2020/US-Mushrooms-08-20.pdf (accessed on 13 February 2021).
- Lewandowski, M.; Sznyk, A.; Bednarek, A. Biology and morphometry of Lycoriella ingenua (Diptera: Sciaridae). Biol. Lett. 2004, 41, 41–50. [Google Scholar]
- Yi, J.-H.; Park, I.-K.; Choi, K.-S.; Shin, S.-C.; Ahn, Y.-J. Toxicity of medicinal plant extracts to Lycoriella ingenua (Diptera: Sciaridae) and Coboldia fuscipes (Diptera: Scatopsidae). J. Asia-Pac. Entomol. 2008, 11, 221–223. [Google Scholar] [CrossRef]
- Rinker, D.L. Insect, mite, and nematode pests of commercial mushroom production. In Edible and Medicinal Mushrooms; Zied, C., Pardo-Giménez, A., Eds.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2017; pp. 221–237. [Google Scholar] [CrossRef]
- Cloyd, R.A.; Zaborski, E.R. Fungus gnats, Bradysia spp. (Diptera: Sciaridae), and other arthropods in commercial bagged soilless growing media and rooted plant plugs. J. Econ. Entomol. 2004, 97, 503–510. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hulme, P.E. Trade, transport and trouble: Managing invasive species pathways in an era of globalization. J. Appl. Ecol. 2009, 46, 10–18. [Google Scholar] [CrossRef]
- Peterson, A.; Soberón, J.; Pearson, R.G.; Anderson, R.; Martínez-Meyer, E.; Nakamura, M.; Araújo, M. Ecological Niches and Geographic Distributions; Princeton University Press: Princeton, NJ, USA, 2011. [Google Scholar]
- Compton, T.J.; Leathwick, J.R.; Inglis, G.J. Thermogeography predicts the potential global range of the invasive European green crab (Carcinus maenas). Divers. Distrib. 2010, 16, 243–255. [Google Scholar] [CrossRef]
- Gallien, L.; Münkemüller, T.; Albert, C.H.; Boulangeat, I.; Thuiller, W. Predicting potential distributions of invasive species: Where to go from here? Divers. Distrib. 2010, 16, 331–342. [Google Scholar] [CrossRef]
- Thuiller, W.; Richardson, D.M.; Pyšek, P.; Midgley, G.F.; Hughes, G.O.; Rouget, M. Niche-based modelling as a tool for predicting the risk of alien plant invasions at a global scale. Glob. Chang. Biol. 2005, 11, 2234–2250. [Google Scholar] [CrossRef]
- Papp, L.; Darvas, B. ‘Contributions to a Manual of Palaearctic Diptera.’ (Science Herald). Available online: https://agris.fao.org/agris-search/search.do?recordID=US201300058453 (accessed on 3 March 2021).
- GBIF. Download Lycoriella agraria Occurrences. 2020. Available online: https://doi.org/10.15468/dl.hrntwd (accessed on 15 November 2020).
- GBIF. Download Sciara multiseta Occurrences. 2020. Available online: https://doi.org/10.15468/dl.e6qf9b (accessed on 15 November 2020).
- GBIF. Download Lycoriella ingenua Occurrences. 2020. Available online: https://doi.org/10.15468/dl.7y5g8w (accessed on 15 November 2020).
- GBIF. Download Sciara pauciseta Occurrences. 2020. Available online: https://doi.org/10.15468/dl.uhk2m4 (accessed on 15 November 2020).
- GBIF. Download Lycoriella sativae Occurrences. 2020. Available online: https://doi.org/10.15468/dl.qdcafo (accessed on 15 November 2020).
- GBIF. Download Lycoriella auripila Occurrences. 2020. Available online: https://doi.org/10.15468/dl.yjtvtr (accessed on 15 November 2020).
- GBIF. Download Lycoriella castanescens Occurrences. 2020. Available online: https://doi.org/10.15468/dl.yobjuj (accessed on 15 November 2020).
- Aiello-Lammens, M.E.; Boria, R.A.; Radosavljevic, A.; Vilela, B.; Anderson, R.P. spThin: An R package for spatial thinning of species occurrence records for use in ecological niche models. Ecography 2015, 38, 541–545. [Google Scholar] [CrossRef]
- Hijmans, R.J.; Cameron, S.E.; Parra, J.L.; Jones, P.G.; Jarvis, A. Very high resolution interpolated climate surfaces for global land areas. Int. J. Climatol. 2005, 25, 1965–1978. [Google Scholar] [CrossRef]
- Escobar, L.E.; Lira-Noriega, A.; Medina-Vogel, G.; Peterson, A.T. Potential for spread of the white-nose fungus (Pseudogymnoascus destructans) in the Americas: Use of Maxent and NicheA to assure strict model transference. Geospat. Health 2014, 9, 221–229. [Google Scholar] [CrossRef]
- CCAFS. GCM Downscaled Data Portal—Spatial Downscaling Data. 2020. Available online: http://www.ccafs-climate.org/data_spatial_downscaling/ (accessed on 15 January 2021).
- Cobos, M.E.; Peterson, A.T.; Osorio-Olvera, L.; Jiménez-García, D. An exhaustive analysis of heuristic methods for variable selection in ecological niche modeling and species distribution modeling. Ecol. Inform. 2019, 53, 100983. [Google Scholar] [CrossRef]
- Phillips, S.J.; Dudík, M.; Schapire, R.E. A Maximum Entropy Approach to Species Distribution Modeling. In Proceedings of the Twenty-First International Conference on Machine Learning—ICML ’04, Banff, AB, Canada, 4–8 July 2004; ACM Press: New York, NY, USA, 2004; p. 83. [Google Scholar]
- Cobos, M.E.; Peterson, A.T.; Barve, N.; Osorio-Olvera, L. kuenm: An R package for detailed development of ecological niche models using Maxent. PeerJ 2019, 7, e6281. [Google Scholar] [CrossRef] [Green Version]
- Peterson, A.T.; Papeş, M.; Soberón, J. Rethinking receiver operating characteristic analysis applications in ecological niche modeling. Ecol. Model. 2008, 213, 63–72. [Google Scholar] [CrossRef]
- Warren, D.L.; Seifert, S.N. Ecological niche modeling in Maxent: The importance of model complexity and the performance of model selection criteria. Ecol. Appl. 2011, 21, 335–342. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anderson, R.P.; Raza, A. The effect of the extent of the study region on GIS models of species geographic distributions and estimates of niche evolution: Preliminary tests with montane rodents (genus Nephelomys) in Venezuela. J. Biogeogr. 2010, 37, 1378–1393. [Google Scholar] [CrossRef]
- Barve, N.; Barve, V.; Jiménez-Valverde, A.; Lira-Noriega, A.; Maher, S.P.; Peterson, A.T.; Soberón, J.; Villalobos, F. The crucial role of the accessible area in ecological niche modeling and species distribution modeling. Ecol. Model. 2011, 222, 1810–1819. [Google Scholar] [CrossRef]
- Owens, H.L.; Campbell, L.P.; Dornak, L.L.; Saupe, E.E.; Barve, N.; Soberón, J.; Ingenloff, K.; Lira-Noriega, A.; Hensz, C.M.; Myers, C.E.; et al. Constraints on interpretation of ecological niche models by limited environmental ranges on calibration areas. Ecol. Model. 2013, 263, 10–18. [Google Scholar] [CrossRef]
- Osorio-Olvera, L.; Lira-Noriega, A.; Soberón, J.; Peterson, A.T.; Falconi, M.; Contreras-Díaz, R.G.; Martínez-Meyer, E.; Barve, V.; Barve, N. ntbox: An R package with graphical user interface for modelling and evaluating multidimensional ecological niches. Methods Ecol. Evol. 2020, 11, 1199–1206. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2014: Synthesis Report. Intergovernmental Panel on Climate Change, (New York, NY, USA). 2014. Available online: https://www.ipcc.ch/pdf/assessment-3report/ar5/syr/AR5_SYR_FINAL_SPM.pdf (accessed on 15 February 2020).
- Sawangproh, W.; Ekroos, J.; Cronberg, N. The effect of ambient temperature on larvae of Scatopsciara cunicularius (Diptera: Sciaridae) feeding on the thallose liverwort Marchantia polymorpha. Eur. J. Entomol. 2016, 113, 259–264. [Google Scholar] [CrossRef] [Green Version]
- Chidziya, E.; Mutangadura, D.; Jere, J.; Siziba, L. A comparative evaluation of locally available substrates for rearing and studying biology of sciarid fly, Lycoriella mali. Acad. J. Biotechnol. 2013, 1, 57–61. [Google Scholar]
- Katumanyane, A.; Kanzi, A.M.; Malan, A.R. Sciarid pests (Diptera: Sciaridae) from undercover crop production in South Africa. S. Afr. J. Sci. 2020, 116, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Gaitán-Hernández, R. Uso del Sustrato Residual del Cultivo de Pleurotus spp. ‘Sánchez JE and Royse DJ. La Biología, el Cultivo y las Propiedades Nutricionales y Medicinales de las Setas Pleurotus spp.’; El Colegio de la Frontera Sur: Tapachula, México, 2017; pp. 261–282. [Google Scholar]
- Duarte, J.L.P.; da Fonseca Duarte, A.; da Cunha, U.S. A new threat for mushroom growers in South America: First record of Coboldia fuscipes (Meigen, 1830) (Diptera, Scatopsidae) massive damage to Pleurotus spp. Int. J. Trop. Insect Sci. 2020, 41, 887–890. [Google Scholar] [CrossRef]
- Menzel, F.; Smith, J.E.; Colauto, N.B. Bradysia difformis Frey and Bradysia ocellaris (Comstock): Two additional Neotropical species of black fungus gnats (Diptera: Sciaridae) of economic importance: A redescription and review. Ann. Entomol. Soc. Am. 2003, 96, 448–457. [Google Scholar] [CrossRef]
- Broadley, A.; Kauschke, E.; Mohrig, W. Black fungus gnats (Diptera: Sciaridae) found in association with cultivated plants and mushrooms in Australia, with notes on cosmopolitan pest species and biosecurity interceptions. Zootaxa 2018, 4415, 201–242. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jess, S.; Schweizer, H. Biological control of Lycoriella ingenua (Diptera: Sciaridae) in commercial mushroom (Agaricus bisporus) cultivation: A comparison between Hypoaspis miles and Steinernema feltiae. Pest Manag. Sci. 2009, 65, 1195–1200. [Google Scholar] [CrossRef] [PubMed]

| Lycoriella Species | Mean AUC Ratio | pROC p Value | Omission Rate at 5% | AICc | Delta AICc | Reg. Multiplier | Feature Classes |
|---|---|---|---|---|---|---|---|
| L. agraria 1170 models | 1.000 | 0 | 0.04 | 829.260 | 0.000 | 1 | lqpt |
| 1.049 | 0 | 0.04 | 830.493 | 1.232 | 1 | lqpt | |
| 1.000 | 0 | 0 | 830.664 | 1.401 | 3 | lqpth | |
| 1.000 | 0 | 0 | 830.667 | 1.407 | 3 | lqpth | |
| 1.000 | 0 | 0 | 830.667 | 1.407 | 3 | lqpth | |
| 1.000 | 0 | 0.04 | 831.205 | 1.945 | 1 | lqpt | |
| 1.000 | 00 | 0.04 | 831.208 | 1.948 | 1 | lqpt | |
| L. ingenua 15,561 models | 1.036 | 0 | 0.01 | 2425.36 | 0 | 3 | l |
| 1.035 | 0 | 0.03 | 2425.366 | 0.005 | 0.1 | l | |
| 1.036 | 0 | 0.03 | 2425.366 | 0.005 | 0.3 | l | |
| 1.036 | 0 | 0.03 | 2425.366 | 0.005 | 0.5 | l | |
| 1.035 | 0 | 0.03 | 2425.366 | 0.005 | 0.7 | l | |
| 1.035 | 0 | 0.03 | 2425.366 | 0.005 | 1 | l | |
| L. sativae 5400 models | 1.052 | 0 | 0.031 | 2766.137 | 0 | 3 | l |
| 1.047 | 0 | 0.046 | 2766.874 | 0.736 | 0.1 | l | |
| 1.044 | 0 | 0.046 | 2766.874 | 0.736 | 0.3 | l | |
| 1.046 | 0 | 0.046 | 2766.874 | 0.736 | 0.5 | l | |
| 1.045 | 0 | 0.031 | 2766.874 | 0.736 | 0.7 | l | |
| 1.043 | 0 | 0.015 | 2766.874 | 0.736 | 1 | l | |
| 1.000 | 0 | 0 | 2767.922 | 1.784 | 3 | pth |
| Species | Uncorrelated Variables | Variable Contribution (%) |
|---|---|---|
| L. agraria | Mean diurnal range | 4.60 |
| Mean temperature of warmest quarter | 48.67 | |
| Mean temperature of coldest quarter | 0.00 | |
| Precipitation of wettest quarter | 22.67 | |
| Precipitation of driest quarter | 24.05 | |
| L. ingenua | Temperature seasonality | 28.90 |
| Maximum temperature of warmest month | 0.00 | |
| Mean temperature of coldest quarter | 49.80 | |
| Precipitation of wettest quarter | 21.30 | |
| L. sativae | Mean diurnal range | 38.26 |
| Maximum temperature of warmest month | 29.44 | |
| Temperature annual range | 0.00 | |
| Mean temperature of coldest quarter | 7.89 | |
| Annual precipitation | 8.18 | |
| Precipitation of wettest quarter | 5.77 | |
| Precipitation of driest quarter | 10.41 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marques, R.; Duarte, J.L.P.; Duarte, A.d.F.; Krüger, R.F.; Silva da Cunha, U.; Osorio-Olvera, L.; Contreras-Díaz, R.G.; Jiménez-García, D. Effects of Climatic Change on the Potential Distribution of Lycoriella Species (Diptera: Sciaridae) of Economic Importance. Insects 2021, 12, 831. https://doi.org/10.3390/insects12090831
Marques R, Duarte JLP, Duarte AdF, Krüger RF, Silva da Cunha U, Osorio-Olvera L, Contreras-Díaz RG, Jiménez-García D. Effects of Climatic Change on the Potential Distribution of Lycoriella Species (Diptera: Sciaridae) of Economic Importance. Insects. 2021; 12(9):831. https://doi.org/10.3390/insects12090831
Chicago/Turabian StyleMarques, Roberta, Juliano Lessa Pinto Duarte, Adriane da Fonseca Duarte, Rodrigo Ferreira Krüger, Uemmerson Silva da Cunha, Luis Osorio-Olvera, Rusby G. Contreras-Díaz, and Daniel Jiménez-García. 2021. "Effects of Climatic Change on the Potential Distribution of Lycoriella Species (Diptera: Sciaridae) of Economic Importance" Insects 12, no. 9: 831. https://doi.org/10.3390/insects12090831
APA StyleMarques, R., Duarte, J. L. P., Duarte, A. d. F., Krüger, R. F., Silva da Cunha, U., Osorio-Olvera, L., Contreras-Díaz, R. G., & Jiménez-García, D. (2021). Effects of Climatic Change on the Potential Distribution of Lycoriella Species (Diptera: Sciaridae) of Economic Importance. Insects, 12(9), 831. https://doi.org/10.3390/insects12090831







