Aedes Mosquito Distribution along a Transect from Rural to Urban Settings in Yaoundé, Cameroon
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
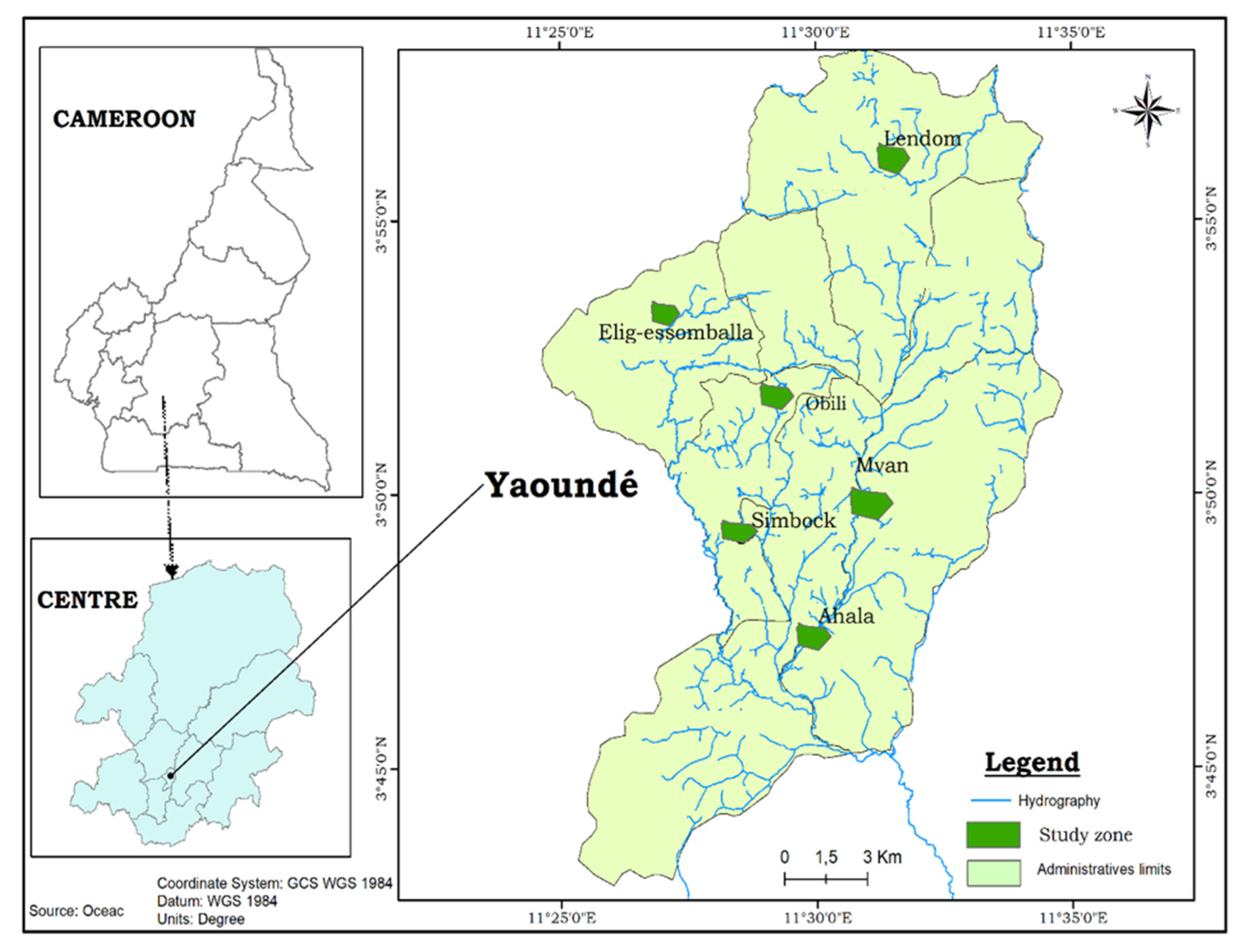
2.1. Description of Study Sites
2.2. Study Design
2.3. Data Analysis
3. Results
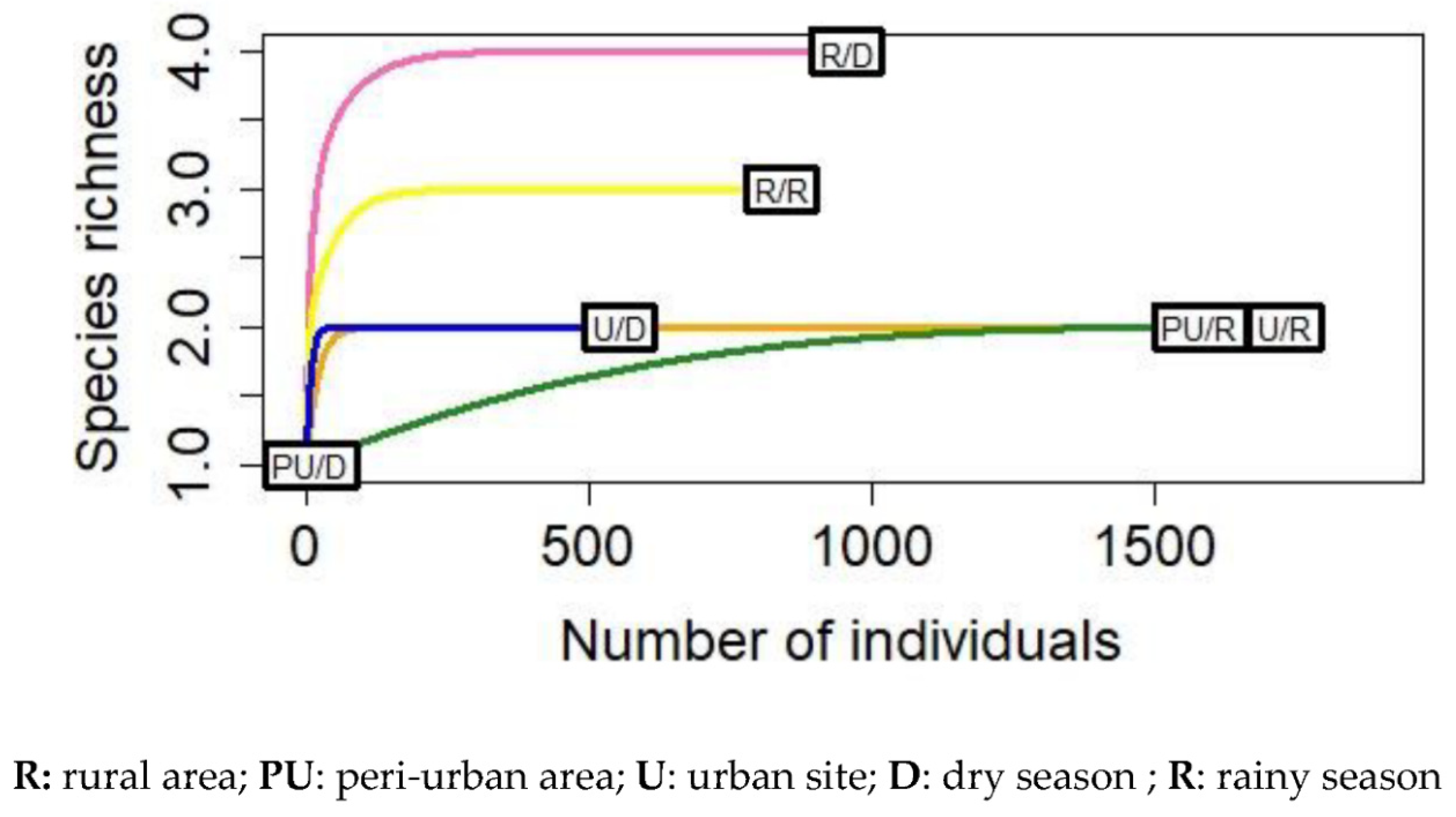
3.1. Mosquito Distribution across Ecological Settings
3.2. Distribution of Aedes Species According to Seasons and Ecological Settings
3.3. Types of Aedes Breeding Habitats
3.4. Co-Occurence of Aedes Species in Breeding Sites
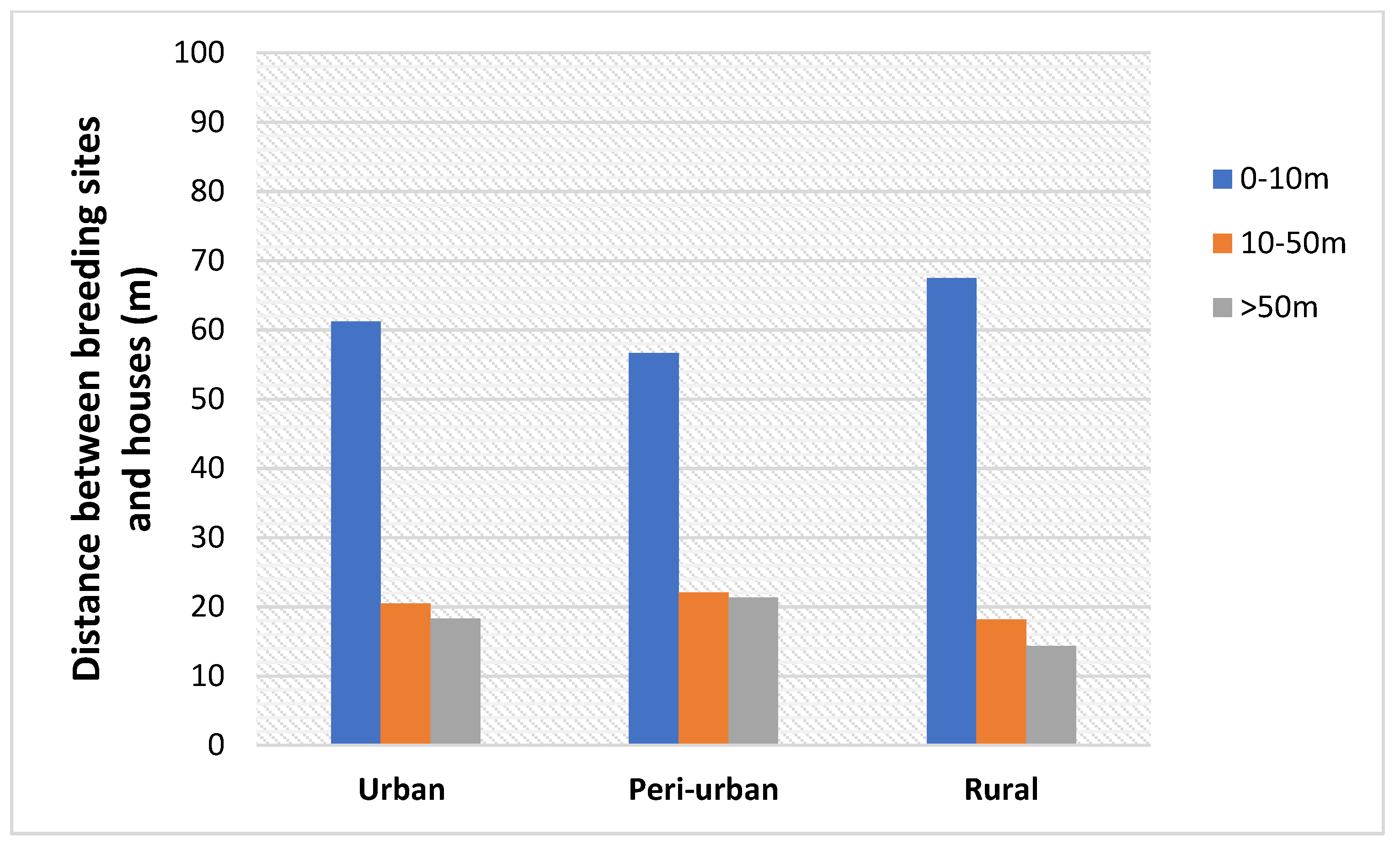
3.5. Distance of the Breeding Sites to the Nearest House
3.6. Estimation of Entomological Indices (Stegomyia Indices)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bhatt, S.; Gething, P.W.; Brady, O.J.; Messina, J.P.; Farlow, A.W.; Moyes, C.L.; Drake, J.M.; Brownstein, J.S.; Hoen, A.G.; Sankoh, O.; et al. The Global Distribution and Burden of Dengue. Nature 2013, 496, 504–507. [Google Scholar] [CrossRef]
- Weaver, S.C.; Reisen, W.K. Present and Future Arboviral Threats. Antiviral Res. 2010, 85, 328–345. [Google Scholar] [CrossRef] [Green Version]
- Ayolabi, C.I.; Olusola, B.A.; Ibemgbo, S.A.; Okonkwo, G.O. Detection of Dengue Viruses among Febrile Patients in Lagos, Nigeria and Phylogenetics of Circulating Dengue Serotypes in Africa. Infect. Genet. Evol. J. Mol. Epidemiol. Evol. Genet. Infect. Dis. 2019, 75, 103947. [Google Scholar] [CrossRef] [PubMed]
- Lutomiah, J.; Barrera, R.; Makio, A.; Mutisya, J.; Koka, H.; Owaka, S.; Koskei, E.; Nyunja, A.; Eyase, F.; Coldren, R.; et al. Dengue Outbreak in Mombasa City, Kenya, 2013–2014: Entomologic Investigations. PLoS Negl. Trop. Dis. 2016, 10, e0004981. [Google Scholar] [CrossRef] [PubMed]
- Otu, A.; Udoh, U.; Ita, O.; Hicks, J.; Ukpeh, I.; Walley, J. Prevalence of Zika and Malaria in Patients with Fever in Secondary Healthcare Facilities in South-Eastern Nigeria. Trop Doct. 2020, 50, 22–30. [Google Scholar] [CrossRef]
- Proesmans, S.; Katshongo, F.; Milambu, J.; Fungula, B.; Mavoko, H.M.; Ahuka-Mundeke, S.; da Luz, R.I.; Esbroeck, M.V.; Ariën, K.K.; Cnops, L.; et al. Dengue and Chikungunya among Outpatients with Acute Undifferentiated Fever in Kinshasa, Democratic Republic of Congo: A Cross-Sectional Study. PLoS Negl. Trop. Dis. 2019, 13, e0007047. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kading, R.C.; Brault, A.C.; Beckham, J.D. Global Perspectives on Arbovirus Outbreaks: A 2020 Snapshot. Trop. Med. Infect. Dis. 2020, 5, 142. [Google Scholar] [CrossRef] [PubMed]
- Arias-Goeta, C.; Moutailler, S.; Mousson, L.; Zouache, K.; Thiberge, J.-M.; Caro, V.; Rougeon, F.; Failloux, A.-B. Chikungunya Virus Adaptation to a Mosquito Vector Correlates with Only Few Point Mutations in the Viral Envelope Glycoprotein. Infect. Genet. Evol. 2014, 24, 116–126. [Google Scholar] [CrossRef]
- Novella, I.S.; Presloid, J.B.; Smith, S.D.; Wilke, C.O. Specific and Nonspecific Host Adaptation during Arboviral Experimental Evolution. Microb. Physiol. 2011, 21, 71–81. [Google Scholar] [CrossRef] [Green Version]
- Ramasamy, R. Adaptation of Freshwater Mosquito Vectors to Salinity Increases Arboviral Disease Transmission Risk in the Context of Anthropogenic Environmental Changes. In Global Virology I—Identifying and Investigating Viral Diseases; Shapshak, P., Sinnott, J.T., Somboonwit, C., Kuhn, J.H., Eds.; Springer: New York, NY, USA, 2015; pp. 45–54. ISBN 978-1-4939-2410-3. [Google Scholar]
- Gould, E.; Pettersson, J.; Higgs, S.; Charrel, R.; de Lamballerie, X. Emerging Arboviruses: Why Today? One Health 2017, 4, 1–13. [Google Scholar] [CrossRef]
- McMichael, A.J.; Woodruff, R.E.; Hales, S. Climate Change and Human Health: Present and Future Risks. Lancet 2006, 367, 859–869. [Google Scholar] [CrossRef]
- Wilder-Smith, A.; Ooi, E.-E.; Horstick, O.; Wills, B. Dengue. Lancet 2019, 393, 350–363. [Google Scholar] [CrossRef]
- WHO. WHO Global Vectors Control Response, 2017–2030; WHO: Geneva, Switzerland, 2017. [Google Scholar]
- Boyer, S.; Calvez, E.; Chouin-Carneiro, T.; Diallo, D.; Failloux, A.-B. An Overview of Mosquito Vectors of Zika Virus. Microbes Infect. 2018, 20, 646–660. [Google Scholar] [CrossRef] [PubMed]
- Diallo, D.; Sall, A.A.; Diagne, C.T.; Faye, O.; Faye, O.; Ba, Y.; Hanley, K.A.; Buenemann, M.; Weaver, S.C.; Diallo, M. Zika Virus Emergence in Mosquitoes in Southeastern Senegal, 2011. PLoS ONE 2014, 9, e109442. [Google Scholar] [CrossRef] [Green Version]
- Fontenille, D.; Toto, J.C. Aedes (Stegomyia) Albopictus (Skuse), a Potential New Dengue Vector in Southern Cameroon. Emerg. Infect. Dis. 2001, 7, 1066. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rageau, J.; Adam, J.-P. Culicinæ du cameroun. Ann. Parasitol. Hum. Comparée 1952, 27, 610–635. [Google Scholar] [CrossRef]
- Demanou, M.; Pouillot, R.; Grandadam, M.; Boisier, P.; Kamgang, B.; Hervé, J.P.; Rogier, C.; Rousset, D.; Paupy, C. Evidence of Dengue Virus Transmission and Factors Associated with the Presence of Anti-Dengue Virus Antibodies in Humans in Three Major Towns in Cameroon. PLoS Negl. Trop. Dis. 2014, 8, e2950. [Google Scholar] [CrossRef] [PubMed]
- Gake, B.; Vernet, M.A.; Leparc-Goffart, I.; Drexler, J.F.; Gould, E.A.; Gallian, P.; de Lamballerie, X.; Gake, B.; Vernet, M.A.; Leparc-Goffart, I.; et al. Low Seroprevalence of Zika Virus in Cameroonian Blood Donors. Braz. J. Infect. Dis. 2017, 21, 481–483. [Google Scholar] [CrossRef] [PubMed]
- Nemg Simo, F.B.; Sado Yousseu, F.B.; Evouna Mbarga, A.; Bigna, J.J.; Melong, A.; Ntoude, A.; Kamgang, B.; Bouyne, R.; Moundipa Fewou, P.; Demanou, M. Investigation of an Outbreak of Dengue Virus Serotype 1 in a Rural Area of Kribi, South Cameroon: A Cross-Sectional Study. Intervirology 2018, 61, 265–271. [Google Scholar] [CrossRef]
- Tchuandom, S.B.; Tchouangueu, T.F.; Antonio-Nkondjio, C.; Lissom, A.; Djang, J.O.N.; Atabonkeng, E.P.; Kechia, A.; Nchinda, G.; Kuiate, J.-R. Seroprevalence of Dengue Virus among Children Presenting with Febrile Illness in Some Public Health Facilities in Cameroon. Pan Afr. Med. J. 2018, 31. [Google Scholar] [CrossRef]
- Tchuandom, S.B.; Tchadji, J.C.; Tchouangueu, T.F.; Biloa, M.Z.; Atabonkeng, E.P.; Fumba, M.I.M.; Massom, E.S.; Nchinda, G.; Kuiate, J.-R. A Cross-Sectional Study of Acute Dengue Infection in Paediatric Clinics in Cameroon. BMC Public Health 2019, 19, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Yousseu, F.B.S.; Nemg, F.B.S.; Ngouanet, S.A.; Mekanda, F.M.O.; Demanou, M. Detection and Serotyping of Dengue Viruses in Febrile Patients Consulting at the New-Bell District Hospital in Douala, Cameroon. PLoS ONE 2018, 13, e0204143. [Google Scholar] [CrossRef]
- Kamgang, B.; Vazeille, M.; Tedjou, A.; Yougang, A.P.; Wilson-Bahun, T.A.; Mousson, L.; Wondji, C.S.; Failloux, A.-B. Different Populations of Aedes Aegypti and Aedes Albopictus (Diptera: Culicidae) from Central Africa Are Susceptible to Zika Virus Infection. PLoS Negl. Trop. Dis. 2020, 14, e0008163. [Google Scholar] [CrossRef]
- Kamgang, B.; Vazeille, M.; Tedjou, A.N.; Wilson-Bahun, T.A.; Yougang, A.P.; Mousson, L.; Wondji, C.S.; Failloux, A.-B. Risk of Dengue in Central Africa: Vector Competence Studies with Aedes Aegypti and Aedes Albopictus (Diptera: Culicidae) Populations and Dengue 2 Virus. PLoS Negl. Trop. Dis. 2019, 13. [Google Scholar] [CrossRef] [Green Version]
- Kamgang, B.; Vazeille, M.; Yougang, A.P.; Tedjou, A.N.; Wilson-Bahun, T.A.; Mousson, L.; Wondji, C.S.; Failloux, A.-B. Potential of Aedes Albopictus and Aedes Aegypti (Diptera: Culicidae) to Transmit Yellow Fever Virus in Urban Areas in Central Africa. Emerg. Microbes Infect. 2019, 8, 1636–1641. [Google Scholar] [CrossRef] [Green Version]
- Roslan, M.; Ngui, R.; Vythilingam, I.; Wan Sulaiman, W. Evaluation of Sticky Traps for Adult Aedes Mosquitoes in Malaysia: A Potential Monitoring and Surveillance Tool for the Efficacy of Control Strategies. J. Vector Ecol. 2017, 42, 298–307. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schoener, E.; Zittra, C.; Weiss, S.; Walder, G.; Barogh, B.S.; Weiler, S.; Fuehrer, H.-P. Monitoring of Alien Mosquitoes of the Genus Aedes (Diptera: Culicidae) in Austria. Parasitol. Res. 2019, 118, 1633–1638. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Y.; Kamara, F.; Zhou, G.; Puthiyakunnon, S.; Li, C.; Liu, Y.; Zhou, Y.; Yao, L.; Yan, G.; Chen, X.-G. Urbanization Increases Aedes Albopictus Larval Habitats and Accelerates Mosquito Development and Survivorship. PLoS Negl. Trop. Dis. 2014, 8, e3301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mayi, M.P.A.; Bamou, R.; Djiappi-Tchamen, B.; Fontaine, A.; Jeffries, C.L.; Walker, T.; Antonio-Nkondjio, C.; Cornel, A.J.; Tchuinkam, T. Habitat and Seasonality Affect Mosquito Community Composition in the West Region of Cameroon. Insects 2020, 11, 312. [Google Scholar] [CrossRef]
- PAHO. Dengue and Dengue Hemorrhagic Fever in the Americas: Guidelines for Prevention and Control; PAHO: Washington, DC, USA, 1994. [Google Scholar]
- Cameroun Fiche Pays PopulationData.Net 2018. Available online: https://www.populationdata.net/pays/cameroun/ (accessed on 16 May 2021).
- Low, G.K.-K.; Ogston, S.A.; Yong, M.-H.; Gan, S.-C.; Chee, H.-Y. Global Dengue Death before and after the New World Health Organization 2009 Case Classification: A Systematic Review and Meta-Regression Analysis. Acta Trop. 2018, 182, 237–245. [Google Scholar] [CrossRef]
- Sanchez, L.; Vanlerberghe, V.; Alfonso, L.; del Carmen Marquetti, M.; Guzman, M.G.; Bisset, J.; van der Stuyft, P. Aedes Aegypti Larval Indices and Risk for Dengue Epidemics. Emerg. Infect. Dis. 2006, 12, 800–806. [Google Scholar] [CrossRef] [Green Version]
- WHO. WHO Technical Guide for a System of Yellow Fever Surveillance; WHO: Geneva, Switzerland, 1971. [Google Scholar]
- Tedjou, A.N.; Kamgang, B.; Yougang, A.P.; Njiokou, F.; Wondji, C.S. Update on the Geographical Distribution and Prevalence of Aedes Aegypti and Aedes Albopictus (Diptera: Culicidae), Two Major Arbovirus Vectors in Cameroon. PLoS Negl. Trop. Dis. 2019, 13, e0007137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brottes, H.; Rickenbach, A.; Brès, P.; Williams, M.C.; Salaun, J.J.; Ferrara, L. Le virus Okola (YM 50/64) nouveau prototype d’arbovirus isolé au Cameroun à partir de moustiques. Ann. Inst. Pasteur 1969, 116, 543–551. [Google Scholar]
- Rickenbach, A.; Gonidec, L. L’incidence Des Arbovirus Isolés Des Moustiques Dans Une Région Forestière Du Sur Cameroun, La Région de Yaoundé. Bulletin de la Société de Pathologie Exotique 1976, 69, 372–381. [Google Scholar]
- Salaun, J.J.; Rickenbach, A.; Brès, P.; Brottes, H.; Germain, M.; Eouzan, J.-P.; Ferrara, L. Le virus Nkolbisson (YM 31/65) nouveau prototype d’arbovirus isolé au Cameroun. Ann. Inst. Pasteur 1969, 116, 254–260. [Google Scholar]
- Kamgang, B.; Yougang, A.P.; Tchoupo, M.; Riveron, J.M.; Wondji, C. Temporal Distribution and Insecticide Resistance Profile of Two Major Arbovirus Vectors Aedes Aegypti and Aedes Albopictus in Yaoundé, the Capital City of Cameroon. Parasit. Vectors 2017, 10, 469. [Google Scholar] [CrossRef]
- Hondt, O.E.N.; Ntonga, P.A.; Hiol, J.V.N.; Edou, D.N.; Tonga, C.; Foko, D.; Kekeunou, S. Adaptation Compétitive d’Aedes Albopictus Skuse, 1894 En Présence d’Aedes Aegypti Linné, 1862 Dans Quelques Gîtes Larvaires Temporaires de La Ville de Douala (Cameroun) Dans Un Contexte de Résistance Aux Pyréthrinoïdes. Bulletin de la Société de pathologie exotique 2020, 113, 79–87. [Google Scholar] [CrossRef]
- Coffinet, T.; Mourou, J.R.; Pradines, B.; Toto, J.C.; Jarjaval, F.; Amalvict, R.; Kombila, M.; Carnevale, P.; Pagès, F. First Record of Aedes Albopictus InGabon. J. Am. Mosq. Control Assoc. 2007, 23, 471–472. [Google Scholar] [CrossRef]
- Wat’senga Tezzo, F.; Fasine, S.; Manzambi Zola, E.; del Marquetti, M.C.; Binene Mbuka, G.; Ilombe, G.; Mundeke Takasongo, R.; Smitz, N.; Bisset, J.A.; Van Bortel, W.; et al. High Aedes Spp. Larval Indices in Kinshasa, Democratic Republic of Congo. Parasit. Vectors 2021, 14, 92. [Google Scholar] [CrossRef]
- Wilson-Bahun, T.A.; Kamgang, B.; Lenga, A.; Wondji, C.S. Larval Ecology and Infestation Indices of Two Major Arbovirus Vectors, Aedes Aegypti and Aedes Albopictus (Diptera: Culicidae), in Brazzaville, the Capital City of the Republic of the Congo. Parasit. Vectors 2020, 13, 492. [Google Scholar] [CrossRef]
- Chareonviriyaphap, T.; Bangs, M.J.; Suwonkerd, W.; Kongmee, M.; Corbel, V.; Ngoen-Klan, R. Review of Insecticide Resistance and Behavioral Avoidance of Vectors of Human Diseases in Thailand. Parasit. Vectors 2013, 6, 280. [Google Scholar] [CrossRef] [Green Version]
- Lounibos, L.P.; Suárez, S.; Menéndez, Z.; Nishimura, N.; Escher, R.L.; O’Connell, S.M.; Rey, J.R. Does Temperature Affect the Outcome of Larval Competition between Aedes Aegypti and Aedes Albopictus? J. Vector Ecol. J. Soc. Vector Ecol. 2002, 27, 86–95. [Google Scholar]
- Braks, M.A.H.; Honório, N.A.; Lounibos, L.P.; Lourenço-De-Oliveira, R.; Juliano, S.A. Interspecific Competition Between Two Invasive Species of Container Mosquitoes, Aedes Aegypti and Aedes Albopictus (Diptera: Culicidae), in Brazil. Ann. Entomol. Soc. Am. 2004, 97, 130–139. [Google Scholar] [CrossRef]
- Daugherty, M.P.; Alto, B.W.; Juliano, S.A. Invertebrate Carcasses as a Resource for Competing Aedes Albopictus and Aedes Aegypti (Diptera: Culicidae). J. Med. Entomol. 2000, 37, 364–372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Juliano, S.A. Species Introduction and Replacement among Mosquitoes: Interspecific Resource Competition or Apparent Competition? Ecology 1998, 79, 255–268. [Google Scholar] [CrossRef]
- Juliano, S.A.; O’Meara, G.F.; Morrill, J.R.; Cutwa, M.M. Desiccation and Thermal Tolerance of Eggs and the Coexistence of Competing Mosquitoes. Oecologia 2002, 130, 458–469. [Google Scholar] [CrossRef] [Green Version]
- Juliano, S.A.; Lounibos, L.P.; O’Meara, G.F. A Field Test for Competitive Effects of Aedes Albopictus on A. Aegypti in South Florida: Differences between Sites of Coexistence and Exclusion? Oecologia 2004, 139, 583–593. [Google Scholar] [CrossRef] [Green Version]
- Hornby, J.A.; Moore, D.E.; Miller, T.W. Aedes Albopictus Distribution, Abundance, and Colonization in Lee County, Florida, and Its Effect on Aedes Aegypti. J. Am. Mosq. Control Assoc. 1994, 10, 397–402. [Google Scholar]
- Simard, F.; Nchoutpouen, E.; Toto, J.C.; Fontenille, D. Geographic Distribution and Breeding Site Preference of Aedes Albopictus and Aedes Aegypti (Diptera: Culicidae) in Cameroon, Central Africa. J. Med. Entomol. 2005, 42, 726–731. [Google Scholar] [CrossRef]
- Tedjou, A.N.; Kamgang, B.; Yougang, A.P.; Wilson-Bahun, T.A.; Njiokou, F.; Wondji, C.S. Patterns of Ecological Adaptation of Aedes Aegypti and Aedes Albopictus and Stegomyia Indices Highlight the Potential Risk of Arbovirus Transmission in Yaoundé, the Capital City of Cameroon. Pathogens 2020, 9, 491. [Google Scholar] [CrossRef]
- Carvalho, R.G.; Lourenço-de-Oliveira, R.; Braga, I.A. Updating the Geographical Distribution and Frequency of Aedes Albopictus in Brazil with Remarks Regarding Its Range in the Americas. Mem. Inst. Oswaldo Cruz 2014, 109, 787–796. [Google Scholar] [CrossRef] [Green Version]
- Pancetti, F.G.M.; Honório, N.A.; Urbinatti, P.R.; Lima-Camara, T.N. Twenty-Eight Years of Aedes Albopictus in Brazil: A Rationale to Maintain Active Entomological and Epidemiological Surveillance. Rev. Soc. Bras. Med. Trop. 2015, 48, 87–89. [Google Scholar] [CrossRef] [PubMed]
- Braks, M.A.H.; Honório, N.A.; Lourenço-De-Oliveira, R.; Juliano, S.A.; Lounibos, L.P. Convergent Habitat Segregation of Aedes Aegypti and Aedes Albopictus (Diptera: Culicidae) in Southeastern Brazil and Florida. J. Med. Entomol. 2003, 40, 785–794. [Google Scholar] [CrossRef] [Green Version]
- Honório, N.A.; Castro, M.G.; de Barros, F.S.M.; Magalhães, M.D.A.F.M.; Sabroza, P.C. The Spatial Distribution of Aedes Aegypti and Aedes Albopictus in a Transition Zone, Rio de Janeiro, Brazil. Cad. Saúde Pública Rio de Janeiro 2009, 25, 1203–1214. [Google Scholar] [CrossRef] [PubMed]
- de Lima-Camara, T.N.; Honório, N.A.; Lourenço-de-Oliveira, R. Frequency and spatial distribution of Aedes aegypti and Aedes albopictus (Diptera, Culicidae) in Rio de Janeiro, Brazil. Cad. Saúde Pública 2006, 22, 2079–2084. [Google Scholar] [CrossRef]
- O’Meara, G.; Evans, L.; Gettman, A.; Cuda, J. Spread of Aedes Albopictus and Decline of Ae. Aegypti (Diptera: Culicidae) in Florida. J. Med. Entomol. 1995, 32, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Cox, J.; Grillet, M.E.; Ramos, O.M.; Amador, M.; Barrera, R. Habitat Segregation of Dengue Vectors along an Urban Environmental Gradient. Am. J. Trop. Med. Hyg. 2007, 76, 820–826. [Google Scholar] [CrossRef] [PubMed]
- Bagny, L.; Delatte, H.; Elissa, N.; Quilici, S.; Fontenille, D. Aedes (Diptera: Culicidae) Vectors of Arboviruses in Mayotte (Indian Ocean): Distribution Area and Larval Habitats. J. Med. Entomol. 2009, 46, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Barrera, R. Competition and Resistance to Starvation in Larvae of Container-Inhabiting Aedes Mosquitoes. Ecol. Entomol. 1996, 21, 117–127. [Google Scholar] [CrossRef]
- Alto, B.W.; Lounibos, L.P.; Higgs, S.; Juliano, S.A. Larval Compettition Differentially Affect Arbovirus Infection in Aedes Mosquitoes. Ecology 2005, 86, 3279–3288. [Google Scholar] [CrossRef] [Green Version]
- Murrell, E.G.; Juliano, S.A. Detritus Type Alters the Outcome of Interspecific Competition between Aedes Aegypti and Aedes Albopictus (Diptera: Culicidae). J. Med. Entomol. 2008, 45, 375–383. [Google Scholar] [CrossRef] [Green Version]
- Lounibos, L.P.; O’Meara, G.F.; Juliano, S.A.; Nishimura, N.; Escher, R.L.; Reiskind, M.H.; Cutwa, M.; Greene, K. Differential Survivorship of Invasive Mosquito Species in South Florida Cemeteries: Do Site-Specific Microclimates Explain Patterns of Coexistence and Exclusion? Ann. Entomol. Soc. Am. 2010, 103, 757–770. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Camara, D.C.P.; Codeço, C.T.; Juliano, S.A.; Lounibos, L.P.; Riback, T.I.S.; Pereira, G.R.; Honorio, N.A. Seasonal Differences in Density But Similar Competitive Impact of Aedes Albopictus (Skuse) on Aedes Aegypti (L.) in Rio de Janeiro, Brazil. PLoS ONE 2016, 11, e0157120. [Google Scholar] [CrossRef] [PubMed]

| Species | Ecological Zones | Total | |||||
|---|---|---|---|---|---|---|---|
| Urban | Peri-Urban | Rural | |||||
| Obili | Mvan | Simbock | Ahala | Lendom | Elig-Essomballa | ||
| Ae. albopictus | 459 | 1755 | 1252 | 260 | 295 | 239 | 4260 (67.27%) |
| Ae. aegypti | 3 | 68 | 71 | 7 | 588 | 577 | 1314 (20.75%) |
| Ae. simpsoni | 0 | 0 | 0 | 0 | 65 | 20 | 85 (1.34) |
| Cx. duttoni | 0 | 21 | 10 | 14 | 103 | 113 | 261 (4.12%) |
| Cx. antennatus | 0 | 0 | 0 | 0 | 26 | 0 | 26 (0.41%) |
| Cx. quinquefasciatus | 0 | 34 | 57 | 0 | 14 | 86 | 191 (3.01%) |
| Lutzia tigripes | 0 | 2 | 1 | 0 | 8 | 9 | 20 (0.31%) |
| Cx. (Culiciomyia) group. | 0 | 0 | 0 | 10 | 91 | 16 | 117 (1.84%) |
| Anopheles funestus | 0 | 0 | 0 | 0 | 0 | 9 | 9 (0.14%) |
| Eretmapodites spp. | 0 | 0 | 0 | 0 | 0 | 15 | 15 (0.23%) |
| Toxorhynchites sp. | 0 | 0 | 0 | 0 | 1 | 0 | 1 (0.01%) |
| Culex spp. | 0 | 0 | 12 | 0 | 2 | 6 | 20 (0.31%) |
| Aedes spp. | 0 | 0 | 0 | 0 | 13 | 0 | 13 (0.20%) |
| Total | 462 | 1880 | 1403 | 291 | 1206 | 1090 | 6332 |
| LR Chisq | Df | Pr (>Chisq) | |
|---|---|---|---|
| Mosquito Species | 22.18 | 3 | p < 0.0001 |
| Site (Urban/periurban/rural) | 939.33 | 2 | p < 0.0001 |
| Season (dry/rainy) | 108.13 | 1 | p < 0.0001 |
| Mosquito species vs. Site | 484.4 | 6 | p < 0.0001 |
| Mosquito species vs. Season | 0.81 | 3 | 0.8478 |
| Site vs. Season | 180.78 | 2 | p < 0.0001 |
| Mosquito species vs. Site vs. Season | 198.02 | 6 | p < 0.0001 |
| Breeding Habitats | Urban | Peri-Urban | Rural | Total | ||||
|---|---|---|---|---|---|---|---|---|
| Obili | Mvan | Simbock | Ahala | Lendom | Elig-Essomballa | |||
| Tires | N(a) | 20 (9) | 33(7) | 83(42) | 19(5) | 5(2) | 8(4) | 168(69) |
| Ae. albopictus | 358 | 1350 | 1159 | 260 | 1 | 10 | 3138 | |
| Ae. aegypti | 3 | 68 | 62 | 7 | 15 | 47 | 217 | |
| Plastic containers | N(a) | 14(1) | 7(2) | 19(4) | 2(0) | 48(31) | 47(30) | 137(68) |
| Ae. albopictus | 101 | 67 | 72 | 0 | 210 | 131 | 581 | |
| Ae. aegypti | 0 | 0 | 8 | 0 | 353 | 344 | 705 | |
| Ae. simpsoni | 0 | 0 | 0 | 0 | 65 | 2 | 67 | |
| Metallic containers | N(a) | 1(0) | 1(0) | 4(1) | 0 | 10(9) | 13(11) | 29(21) |
| Ae. albopictus | 0 | 0 | 21 | 0 | 30 | 27 | 78 | |
| Ae. aegypti | 0 | 0 | 1 | 0 | 109 | 94 | 204 | |
| Ae. simpsoni | 0 | 0 | 0 | 0 | 0 | 9 | 9 | |
| Metallic tanks | N(a) | 0 | 1(0) | 0 | 0 | 6(6) | 19(8) | 26(14) |
| Ae. albopictus | 0 | 0 | 0 | 0 | 10 | 34 | 44 | |
| Ae. aegypti | 0 | 0 | 0 | 0 | 65 | 13 | 78 | |
| Ae. simpsoni | 0 | 0 | 0 | 0 | 0 | 5 | 5 | |
| Plastic tanks | N(a) | 0 | 3(2) | 0 | 2(0) | 8(3) | 4(4) | 17(9) |
| Ae. albopictus | 0 | 338 | 0 | 0 | 39 | 18 | 395 | |
| Ae. aegypti | 0 | 1 | 0 | 0 | 15 | 9 | 25 | |
| Breeze block | N(a) | 0 | 3(0) | 0 | 2(0) | 2(1) | 3(2) | 10(3) |
| Ae. albopictus | 0 | 0 | 0 | 0 | 3 | 12 | 15 | |
| Ae. aegypti | 0 | 0 | 0 | 0 | 3 | 16 | 5 | |
| Ae. simpsoni | 0 | 0 | 0 | 0 | 0 | 4 | 4 | |
| Tree hole | N(a) | 0 | 1(0) | 0 | 0 | 0 | 0 | 1(0) |
| Ae. albopictus | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Ae. aegypti | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Plant leaves | N(a) | 0 | 0 | 0 | 0 | 0 | 1(1) | 1(1) |
| Ae. albopictus | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Ae. aegypti | 0 | 0 | 0 | 0 | 0 | 22 | 22 | |
| Others | N(a) | 0 | 3(0) | 6(0) | 0 | 5(4) | 1(1) | 15(5) |
| Ae. albopictus | 0 | 0 | 0 | 0 | 2 | 7 | 9 | |
| Ae. aegypti | 0 | 0 | 0 | 0 | 28 | 32 | 60 | |
| Species | Urban | Peri-Urban | Rural |
|---|---|---|---|
| Ae. albopictus | 22.09% (19/86) | 22.05% (30/136) | 3.86% (7/181) |
| Ae. aegypti | 0 | 0 | 14.91% (27/181) |
| Ae. albopictus + Ae. aegypti | 3.48% (3/86) | 10.29% (14/136) | 12.15% (22/181) |
| Ae. albopictus + Ae. simpsoni | 0 | 0 | 0.55% (1/181) |
| Ae. albopictus + Ae. aegypti + Ae. simpsoni | 0 | 0 | 4.41% (8/181) |
| Ae. albopictus + Ae. aegypti + Ae. simpsoni + Cx. culiciomayia group | 0 | 0 | 1.10% (2/181) |
| Ae. albopictus + Ae. aegypti + Ae. simpsoni + Cx. Duttoni | 0 | 0 | 0.55% (1/181) |
| Ae. albopictus + Ae. aegypti + Ae. simpsoni + Cx. Culiciomayia group + Cx. antennatus | 0 | 0 | 0.55% (1/181) |
| Ae. albopictus + Cx. quinquefasciatus | 2.94% (4/136) | 0.73% (1/136) | 0.55% (1/181) |
| Ae. albopictus + Cx. duttoni | 2.20% (3/136) | 0.73% (1/136) | 0.55% (1/181) |
| Ae. albopictus + Lutzia tigripes | 0 | 0.73% (1/136) | 0 |
| Ae. albopictus + Cx. Culiciomayia group | 0 | 0.73% (1/136) | 0 |
| Ae. albopictus + Cx. Culiciomayia group + Cx. Duttoni | 0 | 0.73% (1/136) | 0 |
| Ae. albopictus + Cx. Culiciomayia group + Cx. quinquefasciatus | 0 | 0 | 0.55% (1/181) |
| Ae. albopictus + Cx. quinquefasciatus + Lutzia tigripes | 0.73% (1/136) | 0 | 0 |
| Ae. aegypti + Cx. Culiciomayia group | 0 | 0 | 1.65% (3/181) |
| Ae. aegypti + Cx. Culiciomayia group + Cx. quinquefasciatus + Lutzia tigripes | 0 | 0 | 0.55% (1/181) |
| Ae. aegypti + Cx. duttoni + Lutzia tigripes | 0 | 0 | 1.10% (2/181) |
| Ae.aegypti + Ae. simpsoni + Lutzia tigripes | 0 | 0 | 0.55% (1/181) |
| Ae.aegypti + Cx. quinquefasciatus + Eretmapodites sp | 0 | 0 | 0.55% (1/181) |
| Ae. albopictus + Ae. aegypti + Cx. quinquefasciatus | 0.73% (1/136) | (4/136) | 1.65% (3/181) |
| Ae. albopictus + Ae. aegypti + Cx. duttoni | 0 | 0.73% (1/136) | 2.76% (5/181) |
| Ae. albopictus + Ae. aegypti + Cx. antennatus | 0 | 0 | 0.55% (1/181) |
| Ae. albopictus + Ae. aegypti + Lutzia tigripes | 0 | 0 | 0.55% (1/181) |
| Ae. albopictus + Ae. aegypti + Cx. Culiciomayia group | 0 | 0.73% (1/136) | 1.10% (2/181) |
| Ae. albopictus + Ae. aegypti + Cx. duttoni + Cx. quinquefasciatus | 0 | 0.73% (1/136) | 1.65% (3/181) |
| Ae. albopictus + Ae. aegypti + Cx. Culiciomayia group + Cx. duttoni | 0 | 0 | 0.55% (1/181) |
| Ae. albopictus + Ae. aegypti + Cx. Culiciomayia group + Cx. antennatus | 0 | 0 | 0.55% (1/181) |
| Ae. albopictus + Ae. aegypti + Cx. duttoni + Lutzia tigripes | 0 | 0 | 0.55% (1/181) |
| Ae. aegypti + Ae. albopictus + Eretmapodites | 0 | 0 | 1.10% (2/181) |
| Ae. albopictus + Ae. aegypti + Cx. Culiciomayia group + Cx. quinquefasciatus + Lutzia tigripes + An. funestus | 0 | 0 | 0.55% (1/181) |
| Ae. albopictus + Ae. aegypti + Ae. simpsoni + Cx. Culiciomayia group + Cx. quinquefasciatus | 0 | 0 | 0.55% (1/181) |
| Stegomyia Indices | |||
|---|---|---|---|
| Sites | Breteau Index (95% CI) | Container Index (95% CI) | House Index (95% CI) |
| Urban | 49.38% (41.07–57.69) | 70.02% (67.64–72.41) | 40.72% (40.38–41.07) |
| Peri-urban | 40.27% (25–55.55) | 42.71% (39.13–46.29) | 41.94% (25–58.88) |
| Rural | 42.31% (35.44–49.18) | 34.62% (29.78–39.47) | 44.91% (44.26–45.56) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Djiappi-Tchamen, B.; Nana-Ndjangwo, M.S.; Tchuinkam, T.; Makoudjou, I.; Nchoutpouen, E.; Kopya, E.; Talipouo, A.; Bamou, R.; Mayi, M.P.A.; Awono-Ambene, P.; et al. Aedes Mosquito Distribution along a Transect from Rural to Urban Settings in Yaoundé, Cameroon. Insects 2021, 12, 819. https://doi.org/10.3390/insects12090819
Djiappi-Tchamen B, Nana-Ndjangwo MS, Tchuinkam T, Makoudjou I, Nchoutpouen E, Kopya E, Talipouo A, Bamou R, Mayi MPA, Awono-Ambene P, et al. Aedes Mosquito Distribution along a Transect from Rural to Urban Settings in Yaoundé, Cameroon. Insects. 2021; 12(9):819. https://doi.org/10.3390/insects12090819
Chicago/Turabian StyleDjiappi-Tchamen, Borel, Mariette Stella Nana-Ndjangwo, Timoléon Tchuinkam, Idene Makoudjou, Elysée Nchoutpouen, Edmond Kopya, Abdou Talipouo, Roland Bamou, Marie Paul Audrey Mayi, Parfait Awono-Ambene, and et al. 2021. "Aedes Mosquito Distribution along a Transect from Rural to Urban Settings in Yaoundé, Cameroon" Insects 12, no. 9: 819. https://doi.org/10.3390/insects12090819
APA StyleDjiappi-Tchamen, B., Nana-Ndjangwo, M. S., Tchuinkam, T., Makoudjou, I., Nchoutpouen, E., Kopya, E., Talipouo, A., Bamou, R., Mayi, M. P. A., Awono-Ambene, P., Wondji, C., & Antonio-Nkondjio, C. (2021). Aedes Mosquito Distribution along a Transect from Rural to Urban Settings in Yaoundé, Cameroon. Insects, 12(9), 819. https://doi.org/10.3390/insects12090819








