3.2. Taxonomic Descriptions
Characters that are similar among species are not repeated after the description of the first species. The order of descriptions is from the most common and widespread to the less common species. Distribution records are given for Israel, but all species probably occur in Jordan and Egypt, and possibly in other countries in the region. Main morphological characters for the species described here are given in
Table 2.
3.2.1. Asiodiplosis largifica Dorchin, New Species
Host plants: Caroxylon vermiculatum (L.) Akhani & Roalson, C. incanescens (C.A.Mey.) Akhani & Roalson.
Gall and biology: This is one of the most-common and widespread
Asiodiplosis species in Israel. It develops in very-common and conspicuous bud galls on
C. vermiculatum (
Figure 2A–C)—sometimes hundreds on the same plant—and is much-less-common and conspicuous on
C. incancescens (
Figure 2D). Most galls develop in apical buds, but they are also common in lateral buds. Galls on
C. vermiculatum vary considerably in size and shape, with some much hairier and more compact than others. Large galls can be 2 cm long and 1–1.5 cm in diameter and are less hairy, whereas smaller galls are about 6 mm long and 4 mm wide and usually contain a mass of white, wooly hairs at their center. Smaller galls are often clumped together in groups of three to five individual units. Each gall is composed of a group of soft, tapered leaves that widen at their bases, accompanied by many long, white hairs, and contains a single central chamber at its base. The side of the leaves facing the center of the gall is bright green, whereas their other side is covered by short, white hairs. The large, almost-spherical larvae and pupae fill the larval chamber almost completely. Old, empty galls remain on the plants for several months. Galls occasionally contain an unidentified inquilinous moth larva that feeds at the center of the gall inside a loose silky cocoon. On
C. incanescens, the galls are less common and much smaller—about 5 mm in diameter, and composed of short, soft leaves that form a small rosette (
Figure 2D). Empty galls remain on the plant for several months.
This species completes several generations a year. Adults were reared from Caroxylon vermiculatum from February to August, with a peak in early spring (February–March), and from C. incanescens in June to early September. This is the only Israeli species that has more than one host plant. The perennial shrub C. vermiculatum is apparently the primary host, whereas the annual C. incanescens is used by the midges in summer, when it is at its peak growth phase. Overwintering is probably in C. vermiculatum buds as first-instar larvae.
Adult description: General color: Male head and thorax grey, abdomen brownish (
Figure 2E); female dull to bright orange (
Figure 2F).
Head: Eye facets round. Antennal flagellomeres 11–12 in both sexes; when 11, apical flagellomere composed of two fused units and is variably shaped. Male flagellomeres each composed of two nodes separated by short neck and ending with distal neck (
Figure 3A) except in apical flagellomere; necks successively longer along proximal half of antenna; same length along distal half. Each node with one circumfilar whorl subtended by row of strong setae and evenly setulose (
Figure 4A). Circumfilar loops about half length of node (
Figure 4A). Nodes of first flagellomere cylindrical rather than spherical and not distinctly separated as nodes of subsequent flagellomeres (
Figure 3B); strong setae on proximal node of first flagellomere not forming row as on subsequent flagellomeres. Apical flagellomere without distal neck, occasionally with short narrow projection, often composed of two merged units comprising three to four nodes not separated by necks (
Figure 3C). Female flagellomeres cylindrical and successively shorter along antennae (
Figure 3D); first five or six with distinct constriction in mid part and distinct neck, the remaining without constriction and necks. First flagellomere longer than second, sometimes partially fused with it (
Figure 3E). When 11 flagellomeres (the usual case), apical flagellomere 1.5–2 times longer than preceding, appears to consist of two merged units, sometimes with small apical projection (
Figure 3F). Each flagellomere with two whorls of simple, appressed circumfilla with longitudinal connections, two whorls of long setae, and otherwise evenly setulose (
Figure 4D). Frontoclypeal membrane on each side with 4–8 long setae in female, 5–14 in male. Palpus 1-segmented, 1–2 times as long as wide, spherical to slightly cylindrical or wider distally than proximally, occasionally tapered, completely setulose, bearing several long setae (
Figure 5A).
Thorax: Wing (
Figure 5C) transparent, sparsely and evenly covered by fine microtrichia. R
1 reaches C near wing mid-length, R
4+5 straight, slightly curved before reaching C beyond wing apex; M
1+2 present as fold; M
4 weak, forming a fork with CuA; CuA thick along straight proximal half, weak along distal curved half. C with long hair-like setae to slightly beyond meeting point with R
4+5; bases of R
1, R
4+5, and CuA with few long hair-like setae. Wing length 1.50–2.26 mm in female (
n = 36), 1.76–2.69 mm in male (
n = 42). Legs densely setose; claw untoothed, evenly curved, empodium extending beyond bend in claw (
Figure 5B); pulvilli about third length of claw.
Female abdomen (
Figure 6A): subspherical. Sclerites usually weakly pigmented. Tergites 1–7 with anterior pair of sensory setae and 1–2 posterior rows of long setae; tergite 8 with pair of sensory setae the only vestiture. Sternites 2–7 without discernible sensory setae; if pigmented, each with two separate patches of weak pigmentation and 1–2 posterior rows of long setae. Sternite eight not differentiated from surrounding tissue. Ovipositor 13.2–21.2 times as long as tergite 8 (
n = 31); segment 8 grooved by longitudinal lines of setulae; segment 9 covered by stacked ridges, with numerous setae about 0.3 times as long as segment height (
Figure 6B and
Figure 7A). Cerci minute, closely appressed together to form single unit with visible seam (
Figure 6B), with several setae much longer than setae on segment 9. Hypoproct about half as long as cerci.
Male abdomen: Sclerites usually weakly pigmented, often only in mid-section. Tergites 1–7 with anterior pair of distinct sensory setae, and 1–2 posterior rows of long setae; tergite 8 with anterior pair of sensory setae and few setae posteriorly. Sternites without discernible sensory setae anteriorly, with several long setae on most of surface, not forming clear posterior row. Terminalia (
Figure 8A and
Figure 9A,B): Gonocoxite robust, wide rectangular at base, gradually narrows distally, completely setulose, with many evenly distributed strong setae. Gonostylus cylindrical, almost same width throughout length, densely setulose, with numerous evenly distributed setae, ending with wide, brush-like tooth apparently composed of large group of fused setae. Gonostyli typically bent anteriorly to lie over gonocoxites. Cerci short and wide, densely setulose, separated only along distal half to form two triangular lobes much shorter than hypoproct, each with 5–6 long, straight apical setae on prominent bases (
Figure 9A). Hypoproct completely separated into two long cylindrical lobes, splayed along median margins, arched apically around aedeagus, with each lobe divided by longitudinal groove into wide dorsal section and narrower ventral section, with about 10 strong apical setae. Aedeagus wide at base and tapered towards rounded apex in dorsal view (
Figure 9A), cylindrical and dorsally curved in lateral view (
Figure 9B), with pair of sensillae on each side.
Larva (third instar) (
Figure 10A): Light orange; wide cylindrical, almost spherical. Integument with very delicate rugosity, seen only under SEM. Spiracles dark, situated on elevated projections. Antennae 1.5 times as long as wide; posterolateral apodemes about same length as head capsule (
Figure 10B). Spatula rudimentary, comprising small, vaguely defined pigmented patch with only hint of two anterior teeth and no shaft (
Figure 10C). Sternal papillae asetose; 3 lateral papillae on each side of spatula, one of which more distant from other two, all with barely visible setae. Other papillae not discernible.
Pupa (
Figure 11A,B): Light orange. Antennal bases not forming “horns,” with minute tips. Face smooth, without any papillae. Cephalic seta short. Prothoracic spiracle short and blunt, 1–1.5 as long as wide; trachea reaches apex. Abdominal segments evenly covered by tiny spicules.
Distribution: This species is common on its main host plant,
C. vermiculatum, from the southern Golan Heights (around the Sea of Galilee) in the north, to the northern Arava Valley in the south, and the Judaean and Negev deserts in the west. It is particularly common along the Jordan Valley and the Dead Sea. On its second host plant,
C. incanescens, it was found only in the central Jordan Valley, although the plant is very common in the Negev. The galls of this species may be the ones depicted in Houard [
48] from Algeria and Tunisia and attributed to an unidentified gall midge. This would mean that
A. largifica is widespread at least along the southern and eastern Mediterranean Basin.
Etymology: The species name is Latin for bountiful, with reference to the wide distribution and abundance of the galls at almost all times of year.
Holotype: ♂, Israel, Ma’agar Tirza, 32.0667, 35.5061, 23.ii.2021, N. Dorchin, ex bud gall on Caroxylon vermiculatum. On permanent microscope slide in Euparal, deposited in SMNHTAU.
Paratypes: Ex Caroxylon vermiculatum: 1♂, 1♀, Nahal Qumeran, Rt. 90, 31.7375, 35.4597, 19.iii.1995, N. Dorchin; 1♂, 1♀, Nahal Qumeran, 31.7375, 35.4597, 11.v.1995, N. Dorchin; 1♂, 2♀, Nahal Qumeran, 31.7375, 35.4597, 13.ii.1996; 2♂, 1♀, Avenat, 31.6797, 35.4406, 10.vii.1996, N. Dorchin; 1♂, Zomet Mezada, 5 km S, Rt. 90, 31.3131, 35.3833, 27.iv.2014, N. Dorchin; 2♂, Zomet Mezada, 31.3131, 35.3833, 16.ii.2015, N. Dorchin; 8♂, 13♀, HaMeshar, Nahal Terashim, 30.4553, 34.9356, 12.v.2020, N. Dorchin (1♂, 1♀ ZFMK); 4♂, 1♀, Wadi Malha Nature Reserve, 32.0217, 35.4678, 18.viii.2020, N. Dorchin; 4♂, Tomer, Rt. 90, 32.0214, 35.4469, 18.viii.2020, N. Dorchin; 1♂, 2♀, Ma’agar Tirza, 32.0667, 35.5061, 23.ii.2021, N. Dorchin.
Ex Caroxylon incanescens: 1♂, 2♀, Zomet Peza’el, 32.0481, 35.4640, 26.ix.1997, N. Dorchin; 11♂, 12♀, Wadi Malha Nature Reserve, 32.0217, 35.4678, 1.vi.2021, Y. Kenigsberg.
Other material examined: Ex Caroxylon vermiculatum 6 larvae, Enot Zuqim, 31.7156, 35.4514, 15.1.1997, N. Dorchin; 1♂, 2♀, Zomet Mezada, 31.3131, 35.3833, 16.ii.2015, N. Dorchin; 3♂, 8 exuviae, HaMeshar, Nahal Terashim, 30.4553, 34.9356, 12.v.2020, N. Dorchin; 4 larvae, Maagar Tirza, 32.0667, 35.5061, 20.ii.2021, N. Dorchin.
Ex Caroxylon incanescens: 1 larva, Wadi Malha Nature Reserve, 32.0217, 35.4678, 1.vi.2021, Y. Kenigsberg; 12 exuviae, Wadi Malha Nature Reserve, 32.0217, 35.4678, 1.vi.2021, Y. Kenigsberg.
Comments: To date, 24 species of
Asiodiplosis were described from
Caroxylon host plants (originally under
Salsola) but none of them from
C. vermiculatum or
C. incanescens.
Asiodiplosis largifica belongs to a large group of
Asiodiplosis species with short setae on segment 9 of the ovipositor, and has the longest ovipositor among the Israeli species (
Table 2). The larvae have a rudimentary spatula, contrary to the well-developed spatula or its complete absence in other Israeli species for which larvae are known. The male apical flagellomeres are often fused, and its two-lobed hypoproct is divided longitudinally—two attributes that are seen in many other species in this genus, but these species do not overlap geographically with
A. largifica and differ in gall morphology and life-history attributes. The two host plants of this species have an Irano-Turanian and Saharo-Arabian distribution, not reaching Central Asia, from which all other
Asiodiplosis species were described. Taken together with the high level of host specificity seen among Israeli species, we are confident that
A. largifica is not conspecific with any of the Central-Asian species.
3.2.2. Asiodiplosis paradoxa Dorchin, New Species
Host plants: Anabasis setifera Moq.
Gall and biology: This species induces striking and very common galls in stem joints (
Figure 12A–D). The gall constitutes an amorphic enlargement at the base of a joint, accompanied by dense tufts of white hair that seem to burst out of the stem, which makes for a conspicuous sight on the otherwise smooth, glabrous plant. Galls vary considerably in size, ranging from 0.5 to more than 2.5 cm in diameter, and are often grouped together on the same shoot. The size of a gall is not indicative of its age, as small galls may contain a small number of mature larvae or pupae. Each gall contains 1–6 larval chambers embedded in the mass of white hairs immediately above the point at which leaves are attached to the stem. The chambers are rigid capsules composed of several yellowish tapered scales, which appear in the gall when it contains second-instar larvae. First-instar larvae are found in the midst of the green, juicy tissue at the base of the gall without clear chambers. In February and early March, galls contained first-instar larvae, and adults were reared from March to October. Different galls on the same plant may contain larvae of different ages. Clearly this species completes several generations per year, with a peak in spring, and is less common during the summer and fall (July–November), when the plants bear fruits. The galls occasionally contain unidentified moth larvae that feed on the gall tissues as inquilines. Old galls remain on the plants for several months.
Adult description: General color: Female pinkish-orange, male grey to pale brown.
Head: Antennal flagellomeres 12 in male, 11–12 in female. Male apical flagellomere not fused with preceding, with clearly separated nodes, without apical projection. Circumfilar loops about as long as length of node. Female flagellomeres cylindrical, first 2–3 with slight constriction in mid part, subsequent without clear constriction (
Figure 4E), all but two apical flagellomeres with clear necks; apical flagellomere rounded apically, without projection. Frontoclypeal membrane on each side with 5–6 long setae. Palpus 1.5–2 times as long as wide, rounded apically.
Thorax: Wing length: 1.28–2.37 mm in female (n = 32), 1.45–2.35 mm in male (n = 41).
Female abdomen: Sclerites usually hardly pigmented. Tergites 1–7 with posterior row of long setae. Ovipositor 5.6–16.9 times as long as tergite 8 (
n = 27); setae on segment 9 almost as long as segment height (
Figure 7A).
Male abdomen: Tergites 1–7 with posterior row of setae. Sternites 2–6 with posterior row of setae and few long setae medially; sternites 7–8 with long setae on most of surface. Terminalia (
Figure 8B and
Figure 9C,D): Gonocoxite hardly narrowed distally. Cerci almost entirely fused, narrowed abruptly at apex to form two small, rounded lobes, each with three strong, straight setae (
Figure 9C). Lobes of hypoproct slightly arched around aedeagus, divided longitudinally into wide fusiform dorsal section and narrow, tapered ventral section ending with 3 strong setae on prominent bases (
Figure 9C,D). Aedeagus with two longitudinal grooves in dorsal view.
Larva: Integument virtually smooth, with very shallow, delicate verrucae. Antennae minute, as long as wide. Spatula absent. Sternal, lateral, and terminal papillae not discernible. Pleural and dorsal papillae asetose.
Pupa (
Figure 11C,D): Antennal bases form small, tapered horns, pointed ventrally. Head trapezoidal in frontal view.
Holotype: ♂, Israel, Nahal Zeruya, Rt. 90, 31.4386, 35.3831, 27.iv.2014, N. Dorchin, ex bud gall on Anabasis setifera. On permanent microscope slide in Euparal, deposited in SMNHTAU.
Paratypes: 1♂, 1♀, 2 exuviae, En Gedi nature reserve, 31.4549, 35.3942, 19.iii.1995, N. Dorchin; 1♂, 1♀, Nahal Qumeran, Rt. 90, 31.7375, 35.4597, 11.v.1995, N. Dorchin; 2♂, 1♀, Elifaz, 29.7883, 35.0180, 5.iv.1997, N. Dorchin; 1♂, 1♀, Enot Zuqim, 31.7156, 35.4514, 25.iii.2012, N. Dorchin; 3♂, Qalya, Rt. 90, 31.7463, 35.4735, 7.iv.2013, G. Danon; 4♂, 6♀, Enot Zuqim, 31.7156, 35.4514, 17.x.2013, N. Dorchin; 3♂, 2♀, Rt. 206, 3 km S Zomet Rotem, 30.9892, 35.0636, 12.xi.2013, N. Dorchin; 2♂, 1 exuviae, Neot HaKikkar, 30.9519, 35.3635, 27.iv.2014, N. Dorchin; 2♂, 3♀, 8 exuviae, Nahal Zeruya, Rt. 90, 31.4386, 35.3831, 27.iv.2014, N. Dorchin; 6♂, 5♀, 18 exuviae, Mezad Tamar, 31.0275, 35.2417, 27.iv.2014, N. Dorchin (1♂, 1♀ ZFMK); 2♂, 1♀, Arad, 10 km E, Rt. 31, 31.1909, 35.2815, 18.xi.2014, N. Dorchin; 1♂, Nahal Zeruya, Rt. 90, 31.4386, 35.3831, 16.iii.2015, N. Dorchin; 3♂, Nahal Qumeran, Rt. 90, 31.7375, 35.4597, 1.iii.2020, N. Dorchin; 1♂, 1♀, Mezad Tamar, 31.0275, 35.2417, 22.vii.2020, N. Dorchin.
Other material examined: 1♀, En Gedi nature reserve, 31.4549, 35.3942, 19.iii.1995, N. Dorchin; 1♀, Elifaz, 29.7883, 35.0180, 5.iv.1997, N. Dorchin; 1♂, 1♀, Enot Zuqim, 31.7156, 35.4514, 25.iii.2012, N. Dorchin; 2♂, 1♀, Enot Zuqim, 31.7156, 35.4514, 17.x.2013, N. Dorchin; 1♂, Nahal Yishay, 31.4841, 35.3969, 17.x.2013, N. Dorchin, G. Danon; 1♂, Rt. 206, 3 km S Zomet Rotem, 30.9892, 35.0636, 12.xi.2013, N. Dorchin; 4 larvae, Nahal Qumeran, Rt. 90, 31.7375, 35.4597, 13.iv.2021, N. Dorchin.
Distribution: Very common species throughout the distribution range of its host plant in Israel, from the northern coast of the Dead Sea to the southern Arava Valley in the south and into the Judaean and Negev deserts in the west.
Etymology: The species name means strange or unexpected, referring to the striking appearance of the white tufts of hair bursting from the distorted but otherwise smooth, glabrous stems of the host plant.
Comments: This is the only Israeli species whose pupae possess distinct antennal horns, and are also distinct in the trapezoidal shape of the head compared to the round pupal heads of other species, in particular, A. admirabilis and A. bimoda from other Anabasis species in Israel. The larvae do not have a spatula, contrary to those of most Israeli species for which larvae are known and in stark contrast to the well-defined spatula in A. bimoda. Adults generally resemble those of A. admirabilis, but the male antennal flagellomeres do not end with an apical projection, their circumfilar loops are longer, and the ventral, setae-bearing section of the hypoproct is slenderer. The Saharo-Arabian distribution of the host plant, Anabasis setifera, its easily recognizable habitus, and the unique appearance of the galls make it highly unlikely that Asiodiplosis paradoxa is conspecific with any of the 24 species recorded from Central Asia. Together with A. largifica, this is the most-common Asiodiplosis species in Israel in terms of gall abundance and adult activity, with adult emergence almost year-round.
3.2.3. Asiodiplosis admirabilis Dorchin, New Species
Host plants: Anabasis articulata (Forssk.) Moq.
Gall and biology: This species develops in conspicuous, artichoke-like galls in spring (April) (
Figure 13A,B) and in smaller galls in summer and fall (May–September) (
Figure 13C). The large spring galls are composed of fleshy scale-like projections accompanied by short, white hairs at the center of the gall and may exceed 2.5 cm in diameter. They are uncommon and difficult to find in most places, but at some sites specific plants harbored tens or even hundreds of galls year after year. Each gall contains 6–10 larval chambers composed of yellowish, tapered scales that are embedded in the white hairs. Old galls remain on the plant for over a year, becoming yellow and eventually grey (
Figure 13D). Summer galls are smaller and less conspicuous, usually 5–10 mm in diameter, developing at the bases of joints. They are evident as tufts of white, woolly hairs accompanied by a few green leaf-like scales (
Figure 13C) and tend to be found in small groups. Each such gall contains 3–6 larval chambers similar in structure to the larval chambers in spring galls. Adults were notoriously difficult to rear from this host plant compared to the two other
Asiodiplosis species on
Anabasis host-plants in Israel. Obviously, this species completes several generations per year in spring and summer. Overwintering probably takes place as first-instar larvae in buds.
Adult description:
Head: Antennal flagellomeres 12 in male, 10–11 in female. Necks of male flagellomeres about same length throughout antenna (not successively longer); circumfilar loops about half length of node; apical flagellomere not merged with preceding, sometimes with small elongate projection (
Figure 14A). Female flagellomeres, except for apical, all with short necks; length of necks about the same throughout antenna; occasionally, two adjacent flagellomeres partially fused; flagellomeres 2–5 with slight constriction, others without constriction; apical flagellomere composed of 2–3 fused units, sometimes with elongate apical projection (
Figure 14B). Frontoclypeal membrane mostly bare, with 1–2 closely adjacent setae on each side. Palpus 1–1.5 as long as wide.
Thorax: Wing length: 1.60–2.50 mm in female (n = 16), 1.62–2.65 mm in male (n = 13).
Female abdomen: Sclerites weakly but usually clearly pigmented; pigmentation on tergites often receding on ventral area except around posterior row of setae. Ovipositor 8.58–11.31 times as long as tergite 8 (n = 16); setae on segment 9 at most half as long as segment height.
Male abdomen: Sclerites usually clearly pigmented; pigmentation on tergites receding on ventral part, often absent along thin line just before posterior line of setae. Terminalia (
Figure 9E): Gonocoxite only slightly narrowed distally. Cerci almost completely fused, separated distally to form very short lobes, each with 3–4 long setae apically. Hypoproct almost completely separated into two robust, cylindrical lobes, each divided longitudinally into wide dorsal section and narrower ventral section ending with 3–4 long setae on prominent bases. Aedeagus with two longitudinal grooves in dorsal view.
Larva: Not studied.
Pupa (
Figure 11E,F): Antennal bases not forming horns, without any projections.
Holotype: ♂, Israel, Nahal Baraq, Rt. 90, 30.4242, 35.1491, 9.ix.2020, N. Dorchin, ex bud gall on Anabasis articulata. On permanent microscope slide in Euparal, deposited in SMNHTAU.
Paratypes: 6♀, 1♂, Mamshit, 31.0342, 35.0674, 9.vi.1997, N. Dorchin; 2♂, 2♀, Nahal Lavan, 30.9552, 34.3826, 7.v.1998, N. Dorchin; 1♀, Nahal Shezaf, near Hazeva, 30.7395, 35.2621, 19.iv.2014, N. Dorchin; 4♀, Mezad Tamar, 31.0275, 35.2417, 27.iv.2014, N. Dorchin; 1♂, 1♀, Nahal Ashosh, Rt. 90, 30.5114, 35.1801, 28.iv.2014, N. Dorchin; 3♂, Nahal Baraq, Rt. 90, 30.4242, 35.1491, 9.ix.2020, N. Dorchin; 3♂, 2♀, Nahal Shezaf, near Hazeva, 30.7395, 35.2621, 13.iv.2021, N. Dorchin.
Other material examined: 6♀, Mishor Rotem, Rt. 25, 31.0375, 35.1382, 9.vi.1997, N. Dorchin; 1♂, 3 exuviae, Mezad Tamar, 31.0275, 35.2417, 27.iv.2014, N. Dorchin; 3 exuviae, Nahal Baraq, Rt. 90, 30.4242, 35.1491, 9.ix.2020, N. Dorchin; 6 exuviae, Nahal Shezaf, near Hazeva, 30.7395, 35.2621, 13.iv.2021, N. Dorchin.
Distribution: A generally uncommon or sporadic species that is regularly observed in the most arid habitats in Israel, in the Judaean and Negev deserts, and along the Arava valley, particularly in dry riverbeds that experience flash floods once or several times per year. Galls have never been found along the Dead Sea, although the host plant is common in that region.
Etymology: The species name is Latin for admirable or remarkable, referring to the occurrence of the galls in the most extreme desert habitats in Israel, sometimes in great numbers.
Comments: Adults of this species resemble closely those of
A. paradoxa, but the apical flagellomeres in both sexes often end with a long projection that is absent in
A. paradoxa, and the circumfilar loops of the male flagellomeres are shorter. The male hypoproct is somewhat wider in
A. admirabilis than in
A. paradoxa, in particular, the ventral, setae-bearing section. Pupae of
A. admirabilis resemble those of
A. bimoda but clearly differ from those of
A. paradoxa in the lack of any projections on the antennal bases compared to the short, pointed antennal horns in
A. paradoxa. These two species overlap along much of their distribution range but develop on different host plants, and their galls differ profoundly. The scaly and hairy galls of
A. admirabilis resemble those of
A. bimoda and those of several
Asiodiplosis species from
Anabasis in Central Asia (e.g.,
A. anabasidicola Fedotova,
A. anabasidigemmae Fedotova, and
A. palpata Marikovskij [
13,
46]). Nevertheless, the Saharo-Aarabian distribution of
Anabasis articulata and differences in life-history attributes between
A. admirabilis and Central-Asian species make it unlikely that it is conspecific with any of them.
3.2.4. Asiodiplosis bimoda Dorchin, New Species
Host plants: Anabasis syriaca Iljin.
Gall and biology: This species induces two types of galls at different times of the year. Spring galls (March–April) are large and very conspicuous cone-like structures, up to 2 cm in diameter and composed of fleshy leaf-like projections accompanied by long, white hairs (
Figure 15A,B). In late March, such galls contain third-instar larvae, and adults emerge from them in April. In summer and fall, a much smaller type of gall develops on the same plants in the form of a small tuft of short, white hairs in slightly swollen stem joints, causing the joint to bend and distort (
Figure 15C,D). Adults emerge from these galls from June to October. Both types of galls contain several larval chambers—5–10 in spring galls and 1–6 in summer galls—that are embedded in the wooly hairs, composed of yellowish tapered scales at the base of the gall. Both types of galls can be found on the plants during summer, but the larger spring galls are already empty at that time. They remain on the plant for over a year, turning yellow and eventually grey as they dry up. Obviously, this species completes several generations per year in spring and summer. Overwintering probably takes place as first-instar larvae in dormant buds.
Adult description: Head: Antennal flagellomeres 12 in male, 11 in female. Necks of male flagellomeres about same length throughout antenna; circumfilar loops about half length of node; apical flagellomere not fused with preceding, occasionally with small apical projection. Female flagellomeres with short necks, all but apical three with median constriction; apical flagellomere without distal projection. Frontoclypeal membrane with 5–7 setae on each side. Palpus usually 1.0–1.5 times as long as wide, infrequently longer, up to 4.0 times as long as wide.
Thorax: Wing length: 1.75–2.68 mm in female (n = 41), 2.12–2.86 mm in male (n = 44).
Female abdomen: Ovipositor 9.00–15.13 as long as tergite 8 (n = 36); setae on segment 9 0.5–0.7 times as long as segment height.
Male abdomen: Sclerites usually clearly pigmented. Terminalia (
Figure 9F): Cerci separated to base, each with 3 long apical setae. Hypoproct divided longitudinally into wide-dorsal and thinner-ventral sections, thinner section with 3 long apical setae on prominent bases. Aedeagus with small apical cleft.
Larva (third instar): Varies greatly in size; some mature larvae collected at the same locality and date were tenth the size of others. Spatula (
Figure 16A) well-developed, with very wide shaft, narrowed anteriorly into single triangular tooth. Sternal papillae asetose; 3 three lateral papillae on each side, arranged in group of two setose and one distant asetose papilla. Other papillae not discernible.
Pupa (
Figure 17A,B): Antennal bases smooth, without any projections.
Holotype: ♂, Nahal Zin, Rt. 40, Zomet haRuhot, 5 km N, 30.7092, 34.7839, 16.vi.2014, N. Dorchin & I. Hayon, ex bud gall on Anabasis syriaca. On permanent microscope slide in Euparal, deposited in SMNHTAU.
Paratypes: 3♂, 3♀, Nahal Ye’elim, Rt. 31, 31.2388, 35.2357, 19.iv.1995, N. Dorchin; 2♂, 2♀, Ezuz, 30.8021, 34.4661, 17.iv.1998, A. Freidberg; 2♂, 2♀, Nevatim, 31.2156, 34.8799, 14.x.1998, N. Dorchin; 1♂, 1♀, Nahal Zin, Rt. 40, 30.7092, 34.7839, 27.vi.2012, N. Dorchin; 7♂, 6♀, Nahal Boqer, Rt. 40, 30.9096, 34.7779, 28.iii.2013, G. Danon; 8♂, 8♀, Nahal Ye’elim, Rt. 31, 31.2388, 35.2357, 17.iv.2013, N. Dorchin; 1♂, 1♀, Nahal Ye’elim, Rt. 31, 31.2388, 35.2357, 9.iv.2014, N. Dorchin; 5♂, 5♀, El Atrash, Rt. 31, nr. Hura, 31.2693, 34.9519, 11.vi.2014, Y. Sapir; 6♂, 2♀, Zomet haNegev, 31.0666, 34.8381, 21.iv.2014, G. Danon; 5♂, 6♀, Nahal Zin, Rt. 40, Zomet haRuhot, 5 km N, 30.7092, 34.7839, 16.vi.2014, N. Dorchin & I. Hayon; 1♂, 1♀, Nahal Zin, Rt. 40, 30.7092, 34.7839, 22.vii. 2020, N. Dorchin; 8 larvae, Nahal Ye’elim, Rt. 40, 31.2388, 35.2357, 25.iii.2021, N. Dorchin.
Other material examined: 2♂, 3♀, Nahal Boqer, Rt. 40, 30.9096, 34.7779, 28.iii.2013, G. Danon; 1♀, Nahal Ye’elim, Rt. 31, 31.2388, 35.2357, 17.iv.2013, N. Dorchin; 16 exuviae, Zomet HaNegev, 31.0666, 34.8381, 21.iv.2014, G. Danon.
Distribution: This species is found in high elevations in the Negev desert, wherever its host plant, Anabasis syriaca, occurs.
Etymology: The species name refers to the two types of galls it induced at different times of the year.
Comments: Adults, pupae, and galls of this species resemble those of
A. admirabilis from
Anabasis articulata, but the two species do not overlap in Israel (
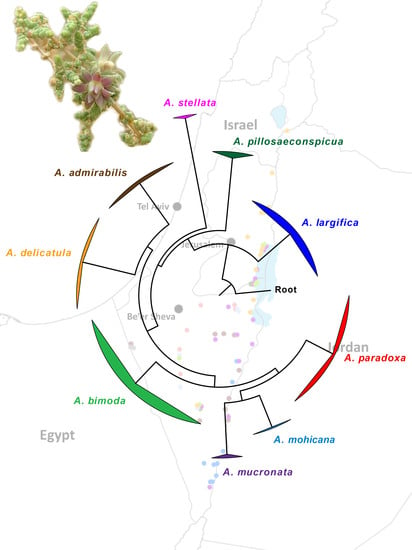
Figure 1) and are clearly distinct from each other genetically (
Figure 18). See comments under
A. admirabilis and
A. paradoxa for more differences between them and other species from
Anabasis from Central Asia.
3.2.5. Asiodiplosis delicatula Dorchin, New Species
Host plants: Haloxylon persicum Bunge.
Gall and biology: This species develops in rosette-like bud galls, about 3 mm in diameter (
Figure 19B,C). Very often several galls are grouped in 5–10 units to form spherical aggregations, 6–8 mm in diameter (
Figure 19A,D). The gall is composed of elongate, tapered leaf-like scales that are bent outwards except for 2–3 scales at the center of the gall, which are closely appressed to form a rigid cone with a cup-like bottom.
The leaf-like scales are green to brownish or magenta, and the galls stand out on the background of the plant’s leafless stems despite their small size. A single plant can harbor hundreds of galls mostly on young green shoots but also on older, woody shoots. Empty galls dry up, turn yellow, and stay on the plant for several weeks. The large, almost-spherical larvae fill up the larval chamber, as do their hymenopteran parasitoids. This species has only one generation per year, from January to February, when galls are very common at almost all sites where the host plant occurs in Israel. Galls found on plants in later months were empty.
Adult description:
Head: Antennal flagellomeres 12 in male, 11 in female. Male flagellomeres with proximal node larger than distal node, rhomboidal rather than spherical (
Figure 14C); distal necks successively longer; circumfilar loops as long as node (
Figure 4B); apical flagellomere often with short-to-medium-sized projection (
Figure 14D,E). Female flagellomeres successively shorter, except apical flagellomere longer than preceding; proximal 6–7 flagellomeres with slight constriction and short necks; distal flagellomeres without clear constriction and necks. Frontoclypeal membrane with 3–8 long setae on each side. Palpus usually spherical, as long as wide, occasionally 1.5–2.0 times as long as wide.
Thorax: Wing length: 2.54–2.94 mm in female (n = 23), 2.76–3.04 mm in male (n = 20).
Female abdomen: Sclerites usually clearly pigmented, pigmentation receding on antero-ventral part. Ovipositor 6.46–16.21 times as long as tergite 8 (
n = 21); segment 9 with dense cover of very long hair-like setae, denser and longer along proximal half; setae much longer than ovipositor width (
Figure 7C).
Male abdomen: Sclerites as in females. Terminalia (
Figure 20A,B): Cerci separated apically to form two short, rounded lobes, each with 4 strong setae apically. Hypoproct separated almost to base into two lobes, each divided longitudinally into massive, rectangular dorsal lobe and thinner ventral lobe with three tapered apical projections bearing long setae.
Larva (third instar): Integument virtually smooth. Spatula (
Figure 16B) conspicuous and strongly pigmented, with short and wide rectangular shaft and single, tapered tooth. Sternal papillae asetose, lateral papillae minute, setiform, usually arranged in two pairs of two, but occasionally one papilla in more-distal group lost or three papillae grouped together in proximal group. Terminal papillae, 3–4 on each side, asetose, barely visible.
Pupa: Antennal bases smooth, without any projections.
Holotype: ♂, Israel, Lotan, 29.9811, 35.0857, 10.ii.2021, N. Dorchin, ex bud gall on Haloxylon persicum. On permanent microscope slide in Euparal, deposited in SMNHTAU.
Paratypes: 2♂, 3♀, 2 larvae, Yotvata, 29.8841, 35.0771, 9.i.1996, N. Dorchin; 4♂, 3♀, Yotvata salt marsh, 29.8359, 35.0459, 12.ii.2013, N. Dorchin; 7♂, 8♀, En Yahav, 30.6450, 35.2085, 12.ii.2013, N. Dorchin (1♂, 1♀ ZFMK); 2♂, 3♀, 5 larvae, Lotan, 29.9811, 35.0857,10.ii.2021, N. Dorchin; 3♂, 3♀, Holot Kasuy, 29.9839, 34.9785, 11.ii.2021, N. Dorchin, M. Spodek.
Other material examined: 1♂, 2 exuviae, Yotvata, 29.8841, 35.0771, 9.i.1996, N. Dorchin; 3♀, 3 exuviae, En Yahav, 30.6450, 35.2085, 12.ii.2013, N. Dorchin.
Distribution: Common along the Arava Valley.
Etymology: The species is named after its small, soft-leaved galls, which are more delicate than galls of other Asiodiplosis species in Israel.
Comments: This species is the only representative in Israel of a group within
Asiodiplosis in which segment 9 of the ovipositor is covered by very long hair-like setae (e.g.,
A. botryoidea (Fedotova),
A. densipila (Marikovskij),
A. festinans (Marikovskij), and
A. rhaphidophytonis (Fedotova) [
6,
8,
49,
50]. It is also readily distinguishable from other Israeli species in the rectangular lobes of the male hypoproct and the conspicuous projection of the apical antennal flagellomere in both sexes. The “hairy” ovipositor and rectangular male hypoproct are often, but not always, correlated, and are common among the 18
Asiodiplosis species from
Haloxylon, all from Central Asia. Only two of these species—
A. floripara (Mamaev) and
A. saxauli (Kaplin)—were recorded from
Haloxylon persicum, with the remaining from
H. ammodendron (C.A.Mey.) Bunge ex Fenzl (=
H. aphyllum). Mamaev [
16] did not include any Figures in the description of
A. floripara from Uzbekistan, but the description of the galls and the male hypoproct do not fit those of
A. delicatula.
Asiodiplosis saxauli, described by Kaplin [
50] from Turkmenistan, has male flagellomeres that are almost devoid of necks and ovipositor with sparse, short setae. These attributes, together with the description of the galls, leave no doubt that
A. saxauli is distinct from
A. delicatula.
Asiodiplosis vernalis Marikovskij, described from
H. ammodendron in Kazakhstan and Turkmenistan [
6], is rather similar morphologically to
A. delicatula, but its galls (depicted by Mamaev [
51]) are scaly aggregations rather than soft-leaved rosettes as those of
A. delicatula.
3.2.6. Asiodiplosis stellata Dorchin, New Species
Host plants: Caroxylon tetrandrum (Forssk.) Akhani and Roalson.
Gall and biology: This species induces elegant galls reminiscent of waterlily flowers, 5–15 mm in diameter. The size of a gall does not reflect its age, as small galls contained mature larvae or pupae. The gall is composed of several fleshy, triangular leaves that are much larger and that differ considerably in shape from the normal, scaly leaves of the host plant (
Figure 21A,B), and is accompanied by very few hairs. Despite this, the galls are rather cryptic due to their infrequent occurrence and the fact that most of them are green. Some galls that are pinkish to dark magenta are easier to locate. Old galls turn yellow and remain on the plants for several months. Each gall contains a single very large larva that fills the round and rigid central chamber completely. Viable galls were found only from mid-January to March, suggesting that this species has a single generation per year.
Adult description: A strikingly large and dark gall midge. General color of both sexes tan-brown (including wings), female abdomen dark red.
Head: Antennal flagellomeres12 in male, 11–12 in female. necks of male flagellomeres successively longer (
Figure 22A); apical flagellomere without neck between nodes; circumfilar loops as long as node. Female flagellomeres (
Figure 22B) successively shorter, often without constriction; proximal flagellomeres cylindrical, distal flagellomeres almost spherical. If 11, apical flagellomere longer than preceding, composed of two merged units; necks same length to flagellomere 6 or 7, shorter thereafter. Frontoclypeal membrane on each side with 15–20 setae in male, 5–12 setae in female. Palpus 1–2 times as long as wide.
Thorax: Wing (
Figure 22C): tan-brown; length: 1.58–2.10 mm in female (
n = 16), 2.62–3.56 mm in male (
n = 6).
Female abdomen (
Figure 22D): tan-brown; sclerites usually strongly pigmented, tergite pigmentation receding on antero-ventral part; posterior row of setae extending into 2–3 rows on ventral part of tergites 1–7. Sternites with posterior row of setae and numerous setae elsewhere, posterior sternite more setose than anterior ones. Pleura densely covered by minute dark spicules except for bare spherical areas, creating white mottled pattern. Ovipositor 9.3–13.0 times as long as tergite 8; setae on segment 9 about 0.3 times as long as ovipositor height.
Male abdomen (
Figure 22E): Sclerites usually strongly pigmented. Tergite pigmentation receding on antero-ventral part, tergites with 3–4 lines of setae occupying posterior quarter to half of tergite length. White mottled pattern on pleura weaker than in female. Terminalia (
Figure 20C): Cerci shrot, almost completely fused, separated by very shallow notch apically, each with 4 long setae. Hypoproct separated into two cylindrical lobes, not arched towards aedeagus, weakly grooved into wider-dorsal and slightly narrower-ventral sections, each lobe with several strong setae pointed apically. Aedeagus very wide, truncate apically.
Larva (third instar): Bright orange. Head short and wide, posterolateral apodemes as long as head capsule, antennae tapered, about twice as long as wide. Spatula (
Figure 16C) well-developed, with single blunt tooth and long shaft, widest just proximal to tooth. Sternal papillae asetose; lateral papillae in one group of 3 on each side, with tiny setae; occasionally, only two lateral papillae in group, or two grouped and one set further away; arrangement may vary between two sides of same individual.
Pupa (
Figure 23A): Antennal bases with minute tapered projection.
Holotype: ♂, Israel, Nahal Zeruya, 31.4386, 35.3831, Rt. 90, 3.iii.2015, G. Danon, ex bud gall on Caroxylon tetrandrum. On permanent microscope slide in Euparal. Deposited in SMNHTAU.
Paratypes: 5♀, 5 larvae, En Gedi, Rt. 90, 31.4549, 35.3942, 25.1.1996, N. Dorchin; 2♀, 2♂, En Gedi, Rt. 90, 31.4549, 35.3942, 13.ii.1996, N. Dorchin; 1♀, 3 larvae, En Gedi, Rt. 90, 31.4549, 35.3942, 16.i.1997, N. Dorchin; 1♂, Nahal Zeruya, Rt. 90, 31.4386, 35.3831,3.iii.2015, G. Danon; 1♀, HaMeshar, 30.4553, 34.9356, 20.ii.2020, N. Dorchin; 1 larva, Nahal Zeruya, Rt. 90, 31.4386, 35.3831, 16.iv.2020, N. Dorchin, O. Segal; 1 larva, Nahal Zeruya, Rt. 90, 31.4386, 35.3831, 2.iii.2021, Y. Kenigsberg, O. Fischer; 5♀, 2♂, Nahal Zeruya, Rt. 90, 31.4386, 35.3831, 14.ii.2021, O. Fischer, Y. Kenigsberg, R. Raz.
Other material examined: 2♀, En Gedi, Rt. 90, 31.4549, 35.3942, 25.i.1996, N. Dorchin; 3 exuviae, Nahal Zeruya, Rt. 90, 31.4386, 35.3831, 14.iii.2021, O. Fischer, Y. Kenigsberg, R. Raz.
Distribution: Rare species that was regularly found in small numbers at only one locality in the Dead Sea area and on one occasion in the Negev Desert.
Etymology: This species is named after its galls, which are reminiscent of small stars.
Comments: This species stands out among other Israeli
Asiodiplosis species for its large size and conspicuously dark color. It is also unique for the barrel-shaped female flagellomeres that lack median constrictions in some individuals and the tergite setation, consisting of 3–4 lines of posterior setae as opposed to 1–2 lines in other species. It develops on the same host plant with
A. pillosaeconspicua, but its less-common galls (
Figure 21A,B) are easily distinguishable from the hairy galls of
A. pillosaeconspicua (
Figure 21C,D). In the laboratory, the heavy adults were not inclined to fly and were found stumbling around on the bottom of the rearing cage.
3.2.7. Asiodiplosis pillosaeconspicua Dorchin, New Species
Host plants: Caroxylon tetrandrum
Gall and biology: This species develops in compact, hairy galls, 1.0–1.5 cm long and 1.0 cm wide, composed of flat, elongate, green leaves that differ markedly from the normal scaly leaves of the plant, and a mass of woolly white hairs between and around the leaves (
Figure 21C,D). Galls are very common and are usually found in groups along the same shoots. Each gall contains 1–5 larvae in chambers composed of yellowish, triangular scales at the base of the gall. Adults were reared from mid-February to late June, but some galls dissected in March and April contained first-instar larvae. Therefore, this species appears to complete several generations in spring and early summer, whereas, in other times of the year, it is probably present as inactive first-instar larvae in the buds. Old galls remain on the plant for at least several weeks. These galls are much more common than those of
A. stellata on the same host plant, and differ from them clearly in structure and pilosity (compare
Figure 21A and
Figure 21C).
Adult description:
Head: Antennal flagellomeres 12 in male, 11–12 in female. Male flagellomeres with successively longer necks, proximal node sphereical, distal node oval. First two flagellomeres fused, first 2–4 often without necks between two nodes; apical flagellomere oval, without division into two nodes; circumfilar loops about half node length (
Figure 4C). Female flagellomeres 1–8 or 1–10 with short necks; when 11, apical flagellomere about twice as long as preceding, without apical projection. Palpus 1–1.5 times as long as wide. Frontoclypeal membrane with 2–4 setae on each side in female, 3–6 in male.
Thorax: Wing length: 1.26–2.15 mm in female (n = 11), 1.70–2.45 mm in male (n = 24).
Female abdomen: Sclerites usually clearly pigmented. Tergites 1–7 with one posterior row of setae, reduced to 2–4 setae on lower part of distal tergites. pleura with mottled pattern created by bare spherical areas on background of minute dark spicules. Ovipositor 6.2–13.6 times as long as tergite 8 (n = 9); setae on segment 9 about half length of ovipositor height.
Male abdomen: Sclerites usually clearly pigmented. Tergite setation and pleura as in female. Terminalia (
Figure 8D): Virtually similar to those of
A. largifica. Cerci almost completely fused, separated by shallow notch apically, each with 3 long apical setae. Hypoproct separated to base into two cylindrical lobes, weakly grooved into wider dorsal section and slightly narrower ventral section; each lobe with 6–7 long setae apically. Aedeagus wide, rounded to truncate apically.
Larva (third instar): Spatula (
Figure 16D) with clearly pigmented, nearly rectangular proximal lobe and weakly pigmented shaft. Lateral papillae in group of 2–3 on each side, with tiny setae.
Pupa (
Figure 17E,F): Antennal bases with minute tapered projection.
Holotype: ♂, Israel, HaMeshar, Rt. 40, 30.4553, 34.9356, 27.iii.2020, N. Dorchin, ex bud gall on Caroxylon tetrandrum. On permanent microscope slide in Euparal, deposited in SMNHTAU.
Paratypes: 1♀, Qalya, 31.7463, 35.4735, 15.i.1997, N. Dorchin; 1♂, En Avedat, 30.8337, 34.7722, 18.ii.2015, N. Dorchin; 1♀, 4♂, HaMeshar, 30.4553, 34.9356, 20.ii.2020, N. Dorchin; 3♀, 9♂, HaMeshar, Rt. 40, 30.4553, 34.9356, 27.iii.2020, N. Dorchin.
Other material examined: 2 larvae, Qalya, 31.7463, 35.4735, 15.i.1997, N. Dorchin; 2 exuviae, Nahal Zeruya, Rt. 90, 31.4386, 35.3831, 27.iv.2014, N. Dorchin; 1♂, En Avedat, 30.8337, 34.7722, 18.ii.2015, N. Dorchin; 6♀, 8♂, HaMeshar, 30.4553, 34.9356, 20.ii.2020, N. Dorchin; 1 larva, Nahal Qumeran, 31.7375, 35.4597, 2.iii.2021, N. Dorchin.
Distribution: Common along the Dead Sea and the Negev and Judaean deserts.
Etymology: This species is named after its hairy, gregarious galls, which are common and easily detected.
Comments: This species is rather similar morphologically to A. stellata, which develops on the same host plant, but is notably smaller and not as dark, and the galls of the two species are readily distinguishable. The two species share the white mottled pattern on the abdominal pleura, although it is much more pronounced in A. stellata, as well as the structure of male terminalia and the pupal morphology. Differences among them include the shorter circumfilar loops on the male flagellomeres in A. pillosaeconspicua, the almost-spherical rather than cylindrical distal female flagellomeres in A. stellata, the more-setose tergites in A. stellata, and the quadrate rather than tapered anterior lobe of the spatula in A. pillosaeconspicua.
3.2.8. Asiodiplosis mucronata Dorchin, New Species
Host plants: Cornulaca monacantha Delile.
Gall and biology: This species develops in scaly, spiny artichoke-shaped galls (
Figure 24A,B) that are locally very common. Fully-grown galls are 5–7 mm in diameter, often in groups of 2–4 galls together. They are composed of scores of small, tapered scale-like leaves, the internal ones thinner and softer, the external ones wider, more rigid, and concave, with a formidable apical thorn. The scales are mixed with long, white hairs. Each gall contains a single central larval chamber composed of closely appressed scales. Although the galls are found by the thousands, it is extremely difficult to rear the gall midges from them because the eclosion period is very short and galls that are collected too early do not yield adults. There is only one generation per year, with adult emergence in the first half of May. Galls that were sampled in March were fully developed but contained tiny first-instars; galls sampled in April did not yield adults, and galls collected after mid-May were already empty.
Adultdescription: Head: Antennal flagellomeres 12 in male, 11–12 in female. Male flagellomeres with very short necks between nodes and short and wide distal necks, slightly longer successively along antenna; two apical flagellomeres fused; apical flagellomere without separation to two nodes, sometimes with small apical projection; circumfilar loops about half length of node. Female flagellomeres about same length or slightly successively shorter along antenna. except longer first and last flagellomeres; when 11, last flagellomere composed of two merged units, sometimes with small apical projection; all flagellomeres with median constriction and wide necks, neck about same length throughout antenna. Palpus 1.5–3.0 times as long as wide, often widest distally. Frontoclypeal membrane with single long seta on each side.
Thorax: Wing length: 2.57–3.02 mm in female (n = 10), 2.51–3.02 mm in male (n = 10).
Female abdomen: Posterior row of setae on tergites expanded into 2–3 rows along ventral half of tergite. Ovipositor as long as tergite 8 (n = 7). Setae on segment 9 0.5–1 times as long as segment height.
Male abdomen: Terminalia (
Figure 20D): Gonostylus with slight shallow depression along proximal margin close to base. Cerci completely fused, sometimes with slight, shallow depression apically and 2–3 long setae on each side. Hypoproct divided into two lobes almost to base, parallel-sided laterally, narrowed abruptly to rounded notch medially, slightly arched distally; each lobe divided longitudinally into narrow ventral section ending with 3–4 long setae and somewhat wider dorsal section. Aedeagus wide and truncate apically.
Larva (third instar): Spatula absent. Discernible papillae include asetose sternals and pleurals, and two slightly protruding, asetose terminal papillae.
Pupa (
Figure 23B,C): Antennal bases smooth, without any projections.
Holotype: ♂, Israel, Sede Halamish, 30.9219, 34.4053, 3.v.2020, A. Dorchin, ex bud gall on Cornulaca monacantha. On a permanent microscope slide in Euparal, deposited in SMNHTAU.
Paratypes: 1♀, 4♂, Nizzana 5 km N., Rt. 10, 30.9294, 34.3789, 12.iv.2002, N. Dorchin; 3♀, 2♂, Nahal Lavan, 30.9552, 34.3826, 7.v.1998, N. Dorchin; 2♀, Sede Halamish, 30.9219, 34.4053, 3.v.2020, A. Dorchin.
Other material examined: 2♂, Nizzana 5 km N., Rt. 10, 12.iv.2002, N. Dorchin; 2 larvae, Sede Halamish, 30.9219, 34.4053, 27.iii.2020, N. Dorchin; 4♀, 1♂, Sede Halamish, 30.9219, 34.4053, 3.v.2020, A. Dorchin.
Distribution: The galls of this species are found in great numbers in the single small area in the western Negev Desert where the host plant occurs in Israel. The galls were depicted in Houard [
48], where they were reported from Tunisia and attributed to an unidentified gall midge. No doubt this species occurs also in Egypt.
Etymology: The species name refers to its spiny artichoke-shaped galls.
Comments: This species is distinct among the Israeli species for the narrow, almost-parallel-sided lobes of the male hypoproct and the completely fused male cerci. It is also notable that the frontoclypeal membrane bears only two long setae compared to numerous setae on each side in other Israeli species.
3.2.9. Asiodiplosis mohicana Dorchin, New Species
Host plants: Agathophora allopecuroides (Delile) Fenzl ex Bunge.
Gall and biology: The large galls of this species are 1–1.5 cm in diameter and are often found in small groups. They are composed of fleshy cylindrical leaves that bear a long apical bristle typical to normal leaves of the host plant, and are mixed with long, white hairs that grow densely in the gall (
Figure 24C,D). Some of these leaves are wider at their bases and are divided into several tapered lobes apically. Each gall contains 1–3 larval chambers at its base, composed of a somewhat rigid capsule surrounded by a group of thin, tapered scales. The galls are uncommon and found sporadically, and adults are rather difficult to rear. Except for one adult reared in mid-October, all adults were reared in February–April, which is the main activity season for this species.
Adult description: Head: Antennal flagellomeres 11–12 in both sexes. Male flagellomeres with short necks between nodes, occasionally without necks at all, and short distal necks about same length along antenna; circumfilar loops about half length of node. Female flagellomeres about same length along antenna, with slight constriction or without constriction medially, short necks to flagellomere 9 or 10, without neck thereafter; when 11 flagellomeres, apical flagellomere usually twice as long as preceding, with short apical projection. Frontoclypeal membrane with 5–6 setae on each side in male, 2–4 setae on each side in female. Palpus 1.5–2.5 times as long as wide.
Thorax: Wing length: 2.14–3.00 mm in female (n = 10), 2.28–2.90 mm in male (n = 6).
Female abdomen: Ovipositor 8.4–13.9 times as long as tergite 8 (n = 10). Setae on segment 9 0.5–1 as long as segment height.
Male abdomen: Terminalia: Cerci separated along distal half, with 3 apical setae on each side. Hypoproct lobes divided longitudinally into wide dorsal section and narrower ventral section with 3 long setae apically. Aedeagus tapered apically.
Larva: Not studied.
Pupa (
Figure 23D,E): Antennal bases without any projections. Dorsal side of head case conspicuously inflated to form elongate, cylindrical lobe extending posteriorly.
Holotype: ♀, Israel, upper Nahal Ye’elim, 31.2389, 35.2358, 19.iv.1995, N. Dorchin, ex bud gall on Agathophora allopecuroides. On permanent microscope slide in Euparal, deposited in SMNHTAU.
Paratypes: 3♂, upper Nahal Ye’elim, 31.2389, 35.2358, 19.iv.1995, N. Dorchin; 1♂, Midreshet Ben-Gurion, upper Nahal Qarqash, 30.8538, 34.7694, 13.x.1997, N. Dorchin; 5♀, 1♂, 2 exuviae, HaMeshar, 30.4553, 34.9356, 27.ii.2020, N. Dorchin.
Other material examined: 1♂, upper Nahal Ye’elim, 31.2389, 35.2358, 19.iv.1995, N. Dorchin; 5♀, HaMeshar, 30.4553, 34.9356, 27.ii.2020, N. Dorchin.
Distribution: Uncommon species, found sporadically at several sites in the Negev desert.
Etymology: This species is named after the long, peculiar bulge on the pupal head, which is reminiscent of a Mohawk hairstyle.
Comments: The adults of this species resemble those of A. largifica but differ from them in the female antennal flagellomeres, which virtually lack medial constriction and in the male cerci, which are fused almost entirely. The pupae stand out among all other Israeli species for the conspicuous posterior extension of the head case, the nature of characters can be seen clearly in both pupae and exuviae.
3.2.10. Halodiplosis fugax Dorchin, New Species
Host plants: Anabasis articulata.
Gall and biology: This species was reared only once from galls of
Asiodiplosis admirabilis collected in early June. The immature stages went unnoticed when dissecting the galls, but a single larva was found actively crawling around on an empty gall. This larva was markedly different from larvae of the gall inducer, and was able to spring around using its well-developed spatula, as do larvae of many cecidomyiid species that leave their galls to pupate in the soil [
3]. This observation suggests that larvae of this species develop as inquilines in
A. admirabilis galls and leave them to pupate in the soil, as do other
Halodiplosis species [
46]. No other details about the life history of this species are known.
Adult description:
Head: Eye facets round. Antennal flagellomeres 12 in both sexes. Male flagellomere 1 effeminate, without neck between nodes, with short apical neck; flagellomere 2 with clearer separation between nodes; flagellomeres 3–11 (
Figure 25A) composed of oval proximal node, wider than long, and cylindrical distal node, longer than wide; proximal nodes with one circumfilar whorl subtended by whorl of strong setae; distal nodes with two circumfilar whorls, separated by whorl of strong setae; circumfilar whorls about same length as proximal node; flagellomere necks about same length along antenna; flagellomere 12 tapered apically (
Figure 25C). Female flagellomeres (
Figure 25B) cylindrical, with clear medial constriction, first 5 consecutively shorter, remaining about same length, all with very short necks; each with proximal whorl of strong setae (grouped rather than lined on flagellomere 1) subtending closely appressed circumfilar whorl and distal whorl of strong setae; flagellomere 12 tapered apically (
Figure 25D). Frontoclypeal membrane with 3–4 long setae on each side. Palpus (
Figure 26A) 3-segmented, segment 1 about 1.5 times as long as wide, segments 2 and 3 about twice as long as wide, all setulose and bearing several long setae.
Thorax: Wing venation and setation as described above for
A. largifica. Wing length: 1.75 mm in female (
n = 1), 1.35 mm in male (
n = 1). Claw untoothed, evenly curved, empodium and pulvilli rudimentary (
Figure 26B).
Female abdomen (
Figure 26C): Tergites 1–7 with anterior pair of sensory setae and posterior row of long setae; tergite 8 with pair of sensory setae the only vestiture. Sternites 2–7 pigmented along proximal half and posterior row of setae; sternite 8 with small pigmented patch and two setae. Ovipositor 11.8 times as long as tergite 8 (
n = 1); segment 9 with numerous setae, less than half length of segment height; cerci closely appressed, with several setae 2–3 times as long as setae on segment 9.
Male abdomen: Tergites 1–7 with anterior pair of sensory setae and posterior row of long setae; tergite 8 with small pigmented patch, pair of sensory setae, and single seta posteriorly. Sternites pigmented on proximal half, without anterior pair of sensory setae, with posterior row of setae and several setae medially; sternite 8 not differentiated from surrounding membrane. Terminalia (
Figure 27A): Gonocoxite rectangular at base, narrows gradually toward apex, with numerous evenly distributed long setae. Gonostylus cylindrical, only slightly narrowed toward apex, evenly setose and setulose; apical tooth wide, brush-like, appears composed of closely packed setae. Cerci densely setulose, separated along apical half to form two short, rounded lobes, each with 4–5 long apical setae. Hypoproct completely separated into two cylindrical lobes, narrowed toward rounded apex, completely setulose, without strong setae apically. Aedeagus wide at base, narrowed toward rounded apex.
Larva (third instar): Elongate and bright orange. Integument covered by prominent triangular spicules. Spatula long-shafted, with trapezoidal, shallowly indented anterior lobe (
Figure 27B). Sternal papillae asetose; 4 lateral papillae on each side of spatula, arranged in group of three papillae, two of which setiform, and one asetose papilla farther away. Ventral papillae minute, asetose, dorsal and pleural papillae with conspicuously long setae. Terminal segment (
Figure 27C) with pair of massive, coniform pigmented papillae and two pairs of much-smaller setiform papillae.
Pupa: Unknown.
Holotype: ♂, Israel, Mamshit, 31.0342, 35.0674, 9.vi.1997, N. Dorchin, ex bud gall on Anabasis articulata. On permanent microscope slide in Euparal, deposited in SMNHTAU.
Paratypes: 1♀, 1 larva, Mamshit, 31.0342, 35.0674, 9.vi.1997, N. Dorchin.
Distribution: Reared only once from Mamshit in the Negev desert.
Etymology: “Fugax” is Latin for elusive or shy, with reference to the rarity of this species and its elusive life history.
Comments: Three of the 13 described species retained here in
Halodiplosis develop as inquilines in galls on
Anabasis host plants in Kazakhstan or Turkmenistan. These are
H. anabasidis from galls of an unknown species on
A. aphylla L.,
H. constricta (Mamaeva) from galls of
Asiodplosis iliensis Marikovskij on
A. aphylla, and
H. filipievi (Fedotova) in galls of
Asiodiplosis anabasidis (Fedotova) on
A. salsa (Ledeb.) Benth. ex Volkens [
47]. These species are generally similar to
H. fugax morphologically, but it is unlikely that they are conspecific given the geographic distance and the different host plants. Morphological differences among these species include the uniquely pigmented female cerci of
H. constricta that appear to be sclerotized, and the male hypoproct of
H. anabasidis and
H. filipjevi that is not deeply divided into two lobes as that of
H. fugax. The male of
H. salsolae, reared from galls on
Salsola tetragona in Tunisia, was described as having nearly glabrous gonostyli [
13], in contrast to the completely setulose gonostyli of
H. fugax. The remaining species of
Halodiplosis, all from Kazakhstan, do not develop in distinct galls and have other morphological and/or life-history attributes that distinguish them from
H. fugax [
46,
47].
3.2.11. Molecular Analysis
The results of the phylogenomic analysis based on the concatenated 16S and COI datasets are shown in
Figure 18. The ML and BI analyses produced identical tree topologies; thus,
Figure 18 presents the ML tree with both posterior probabilities and bootstrap-support values for each node. Our results clearly demonstrate the monophyly of the nine Israeli species of
Asiodiplosis, with maximal support (i.e., PP = 1 and BS = 100%). Support values for deeper nodes were gradually more ambiguous as expected, but the ancestral position of
A. largifica relative to all other species was strongly supported.
Intraspecific divergence within the nine
Asiodiplosis species was low and never exceeded 1% in both mean K2P and uncorrected divergence (
Table 3). Interspecific divergence, on the other hand, was an order of magnitude higher, and ranged between 10% and 20% in K2P values and 9% to 18% in the uncorrected values.