The Myrmecofauna (Hymenoptera: Formicidae) of Hungary: Survey of Ant Species with an Annotated Synonymic Inventory
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Subfamily: Proceratiinae
3.1.1. Tribe: Proceratiini
Genus: Proceratium
3.2. Subfamily: Ponerinae
3.2.1. Tribe: Ponerini
Genus: Cryptopone
Genus: Hypoponera
Genus: Ponera
3.3. Subfamily: Myrmicinae
3.3.1. Tribe: Attini
Genus: Strumigenys
3.3.2. Tribe: Crematogastrini
Genus: Cardiocondyla
Genus: Crematogaster
Genus: Formicoxenus
Genus: Harpagoxenus
Genus: Leptothorax
Genus: Myrmecina
Genus: Strongylognathus
Genus: Temnothorax
Genus: Tetramorium
3.3.3. Tribe: Myrmicini
Genus: Manica
Genus: Myrmica
3.3.4. Tribe: Solenopsidini
Genus: Monomorium
Genus: Solenopsis
3.3.5. Tribe: Stenammini
Genus: Aphaenogaster
Genus: Messor
Genus: Stenamma
3.4. Subfamily: Dolichoderinae
3.4.1. Tribe: Bothriomyrmecini
Genus: Bothriomyrmex
3.4.2. Tribe: Dolichoderini
Genus: Dolichoderus
3.4.3. Tribe: Tapinomini
Genus: Liometopum
Genus: Tapinoma
3.5. Subfamily: Formicinae
3.5.1. Tribe: Camponotini
Genus: Camponotus
Genus: Colobopsis
3.5.2. Tribe: Formicini
Genus: Cataglyphis
Genus: Formica
Genus: Polyergus
3.5.3. Tribe: Lasiini
Genus: Lasius
Genus: Prenolepis
3.5.4. Tribe: Plagiolepidini
Genus: Plagiolepis
3.6. Species of Doubtful Occurrence
3.6.1. Temnothorax rabaudi (Bondroit, 1918)
3.6.2. Tetramorium impurum (Förster, 1850)
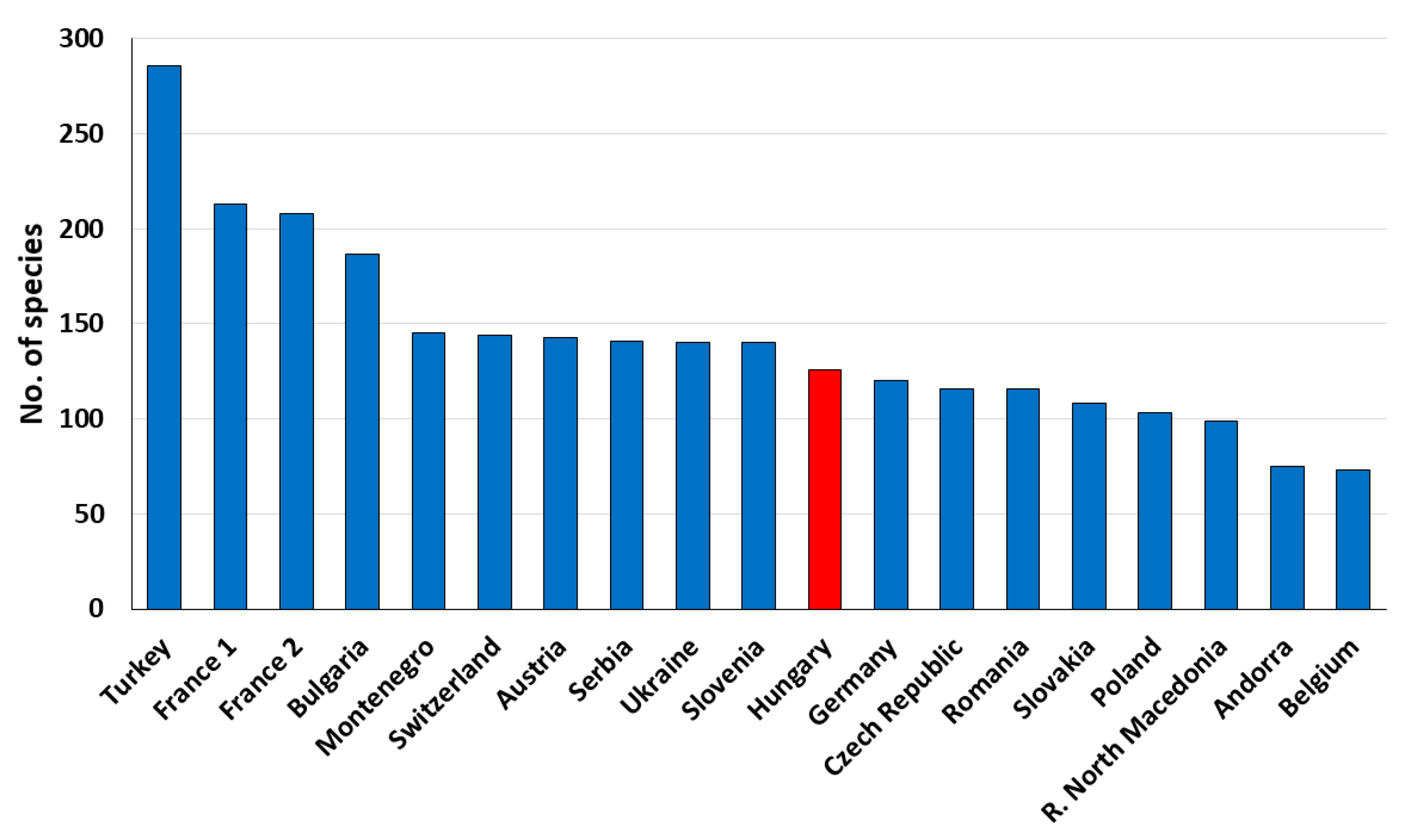
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Causey, D.; Janzen, D.H.; Peterson, A.T.; Vieglais, D.; Krishtalka, L.; Beach, J.H.; Wiley, E.O. Museum collections and taxonomy. Science 2004, 305, 1106. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, Q.D.; Raven, P.H.; Wilson, E.O. Taxonomy: Impediment or Expedient. Science 2004, 303, 285. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, M.R.; Bockmann, F.A.; Amorim, D.S.; de Vivo, M.; de Toledo-Piza, M.; Menezes, N.A.; de Figueiredo, J.L.; Castro, R.M.C.; Gill, A.C.; McEachran, J.D.; et al. Revisiting the taxonomic impediment. Science 2005, 307, 353. [Google Scholar] [CrossRef] [PubMed]
- Hallmann, C.A.; Sorg, M.; Jongejans, E.; Siepel, H.; Hofland, N.; Schwan, H.; Stenmans, W.; Müller, A.; Sumser, H.; Hörren, T.; et al. More than 75 percent decline over 27 years in total flying insect biomass in protected area. PLoS ONE 2017, 12, e0185809. [Google Scholar] [CrossRef]
- Rhodes, C.J. Are insect species imperilled? Critical factors and prevailing evidence for a potential global loss of the entomofauna: A current commentary. Sci. Prog. 2017, 102, 181–196. [Google Scholar] [CrossRef]
- Suprayitno, N.; Narakusumo, R.P.; von Rintelen, T.; Hendrich, L.; Balke, M. Taxonomy and Biogeography without frontiers–WhatsApp, Facebook and smartphone digital photography let citizen scientists in more remote localities step out of the dark. Biodivers. Data J. 2017, 5. [Google Scholar] [CrossRef]
- Devictor, V.; Mouillot, D.; Meynard, C.; Jiguet, F.; Thuiller, W.; Mouquet, N. Spatial mismatch and congruence between taxonomic, phylogenetic and functional diversity: The need for integrative conservation strategies in a changing world. Ecol. Lett. 2010, 13, 1030–1040. [Google Scholar] [CrossRef]
- McKinley, D.; Miller-Rushing, A.J.; Ballard, H.L.; Bonney, R.; Brown, H.; Cook-Patton, S.C.; Evans, D.M.; French, R.A.; Parrish, J.K.; Phillips, T.B.; et al. Citizen science can improve conservation science, natural resource management, and environmental protection. Biol. Conserv. 2017, 208, 15–28. [Google Scholar] [CrossRef]
- Freitag, H.; Pangantihon, C.V.; Njunjić, I. Three new species of Grouvellinus Champion.; 1923 from Maliau Basin, Sabah, Borneo, discovered by citizen scientists during the first Taxon Expedition (Insecta, Coleoptera, Elmidae). ZooKeys 2018, 754, 1–24. [Google Scholar] [CrossRef]
- Moulin, N. When Citizen Science highlights alien invasive species in France: The case of Indochina mantis, Hierodula patellifera (Insecta, Mantodea, Mantidae). Biodivers. Data J. 2020, 8. [Google Scholar] [CrossRef]
- Tanduo, V.; Golemaj, A.; Crocetta, F. Citizen-science detects the arrival and establishment of Branchiomma luctuosum (Grube, 1870) (Annelida: Polychaeta: Sabellidae) in Albania. Biodivers. Data J. 2020, 8. [Google Scholar] [CrossRef]
- Vinarski, M.V. Roots of the taxonomic impediment: Is the “integrativeness” a remedy? Integr. Zool. 2020, 15, 2–15. [Google Scholar] [CrossRef] [PubMed]
- Markó, B.; Sipos, B.; Csősz, S.; Kiss, K.; Boros, I.; Gallé, L. A comprehensive list of the ants of Romania (Hymenoptera: Formicidae). Myrmecol. Nachr. 2006, 9, 65–76. [Google Scholar]
- Radchenko, A.G. Zonal and zoogeographic characteristic of the ant fauna (Hymenoptera, Formicidae) of Ukraine. Vestn. Zool. 2011, 45, 30–39. [Google Scholar] [CrossRef][Green Version]
- Czechowski, W.; Radchenko, A.; Czechowska, W.; Vepsäläinen, K. The Ants of Poland with Reference to the Myrmecofauna of Europe-Fauna Poloniae Vol. 4 New Series; Museum and Institute of Zoology of the Polish Academy of Sciences and Natura optima dux Foundation: Warszawa, Poland, 2012. [Google Scholar]
- Czekes, Z.; Radchenko, A.G.; Csősz, S.; Szász-Len, A.-M.; Tăușan, I.; Benedek, K.; Markó, B. The genus Myrmica Latreille, 1804 (Hymenoptera: Formicidae) in Romania: Distribution of species and key for their identification. Entomol. Romanica 2012, 17, 29–50. [Google Scholar]
- Kiran, K.; Karaman, C. First annotated checklist of the ant fauna of Turkey (Hymenoptera: Formicidae). Zootaxa 2012, 3548, 1–38. [Google Scholar] [CrossRef]
- Bernadou, A.; Fourcassié, V.; Espadaler, X. A preliminary checklist of the ants (Hymenoptera, Formicidae) of Andorra. ZooKeys 2013, 277, 13–23. [Google Scholar] [CrossRef]
- Blatrix, R.; Galkowski, C.; Lebas, C.; Wegnez, P. Fourmis de France, de Belgique et du Luxembourg [The Ants of France, Belgium, and Luxembourg]; Delachaux & Niestlé: Paris, France, 2013; ISBN 978-2-603-01899-6. [Google Scholar]
- Monnin, T.; Espadaler, X.; Lenoir, A.; Peeters, C. Guide des Fourmis de France; Belin: Paris, France, 2013; ISBN 978-2-7011-6471-7. [Google Scholar]
- Bračko, G.; Gomboc, M.; Lupše, B.; Marić, R.; Pristovšek, U. New faunistic data on ants (Hymenoptera: Formicidae) of the southern part of Montenegro. Nat. Slov. 2014, 16, 41–51. [Google Scholar]
- Wagner, H.C. Die Ameisen Kärntens: Verbreitung, Biologie, Ökologie und Gefährdung [The Ants of Carinthia: Distribution, Biology, Ecology and Threats]; Naturwissentschaftlicher Verein für Kärnten: Klagenfurt, Austria, 2014; ISBN 9783853280669. [Google Scholar]
- Seifert, B. The Ants of Central and North. Europe; Lutra Verlags- und Vertriebsgesellschaft: Tauer, Germany, 2018; ISBN 9783936412079. [Google Scholar]
- Csősz, S.; Markó, B.; Gallé, L. The myrmecofauna (Hymenoptera: Formicidae) of Hungary: An updated checklist. North West. J. Zool. 2011, 7, 55–62. [Google Scholar]
- Mayr, G. Ungarn’s Ameisen. Program. der Städtischen Oberrealschule Pesth 1857, 3, 5–26. [Google Scholar]
- Mocsáry, A. Ordo Hymenoptera. In Fauna Regni Hungariae; Mocsáry, A., Ed.; Királyi Magyar Természettudományi Társulat: Budapest, Hungary, 1897; pp. 7–116. [Google Scholar]
- Somfai, E. Hangya alkatúak, Formicoidea. [Ants, Formicoidea.]. In Magyarország Állatvilága (Fauna Hungariae); Székessy, V., Ed.; Akadémiai Kiadó: Budapest, Hungary, 1959; Volume 13, pp. 1–79. [Google Scholar]
- Gallé, L.; Csősz, S.; Tartally, A.; Kovács, É. A check-list of Hungarian ants (Hymenoptera: Formicidae). Folia Entomol. Hung. 1998, 59, 213–220. [Google Scholar]
- Lőrinczi, G. Lasius nitidigaster Seifert, 1996–a new ant species (Hymenoptera: Formicidae) for the Hungarian fauna. Nat. Som. 2011, 19, 223–226. [Google Scholar]
- Seifert, B.; Galkowski, C. The Westpalaearctic Lasius paralienus complex (Hymenoptera: Formicidae) contains three species. Zootaxa 2016, 4132, 44–58. [Google Scholar] [CrossRef] [PubMed]
- Wagner, H.C.; Arthofer, W.; Seifert, B.; Muster, C.; Steiner, F.M.; Schlick-Steiner, B.C. Light at the end of the tunnel: Integrative taxonomy delimits cryptic species in the Tetramorium caespitum complex (Hymenoptera: Formicidae). Myrmecol. News 2017, 25, 95–129. [Google Scholar] [CrossRef]
- Steiner, F.M.; Csősz, S.; Markó, B.; Gamisch, A.; Rinnhofer, L.; Folterbauer, C.; Hammerle, S.; Stauffer, C.; Arthofer, W.; Schlick-Steiner, B.C. Turning one into five: Integrative taxonomy uncovers complex evolution of cryptic species in the harvester ant Messor “structor”. Mol. Phylogenet. Evol. 2018, 127, 387–404. [Google Scholar] [CrossRef]
- Wagner, H.C.; Seifert, B.; Aurenhammer, S.; Komposch, C. Temnothorax turcicus (Santschi, 1934)–eine arborikole Ameise (Hymenoptera: Formicidae) neu für Österreich. Ber. Nat. Med. Ver. Innsbr. 2011, 97, 59–71. [Google Scholar]
- Bolton, B. AntCat: An Online Catalog of Ants of the World. California Academy of Sciences. Available online: http://antcat.org/ (accessed on 21 May 2020).
- Csősz, S. A key to the Ponerinae species of the Carpathian Basin (Hymenoptera: Formicidae). Ann. Hist. Nat. Mus. Nat. Hung. 2003, 95, 147–160. [Google Scholar]
- Csősz, S.; Seifert, B. Ponera testacea Emery, 1895 stat n.–a sister species of P. coarctata (Latreille, 1802) (Hymenoptera, Formicidae). Acta Zool. Acad. Sci. Hung. 2003, 49, 201–214. [Google Scholar]
- Mayr, G.L. Die Europäischen Formiciden: Nach der Analytischen Methode Bearbeitet/von Gustav L. Mayr.; C. Gerold’s Sohn: Wien, Austria, 1861. [Google Scholar]
- Csősz, S.; Tartally, A. Adatok a Körös-Maros Nemzeti Park hangyafaunájához [Data to the ant fauna of the Körös-Maros National Park]. Crisicum 1998, 1, 180–194. [Google Scholar]
- Csősz, S. Taxonomic and distributional notes on two new and a rare Leptothorax Mayr, 1855 species for the Hungarian ant fauna (Hymenoptera, Formicidae). Ann. Hist. Nat. Mus. Nat. Hung. 2001, 93, 99–106. [Google Scholar]
- Móczár, L. Bátorliget hártyásszárnyú faunája, Hymenoptera [Hymenoptera fauna of Bátorliget]. In Bátorliget Élővilága [Flora and Fauna of Bátorliget]; Székessy, Ed.; Akadémiai Kiadó: Budapest, Hungary, 1953; pp. 286–316. [Google Scholar]
- Gallé, L.; Szőnyi, G. The checklist of ants (Hymenoptera, Formicoida) of a sandy grassland in Kiskunság National Park (Hungary). Acta Biol. Szeged. 1988, 34, 167–168. [Google Scholar]
- Mayr, G. Formicina austriaca. Beschreibungder bischer im östreichischen Kaiserstaate aufgefundenen Ameisen nebst Hinzufüngung jener in Deutschland, in der Schweiz und in Italien vorkommenden Ameisen. Verh. Des. Zool. Bot. Ver. Wien. 1855, 5, 273–478. [Google Scholar]
- Radchenko, A.G. The ant fauna of Őrség, western Hungary (Hymenoptera: Formicidae). In Natural History of Őrség Landscape Conervation Area III; Vig, K., Ed.; Savaria–a Vas Megyei Múzeumok Értesítője: Szombathely, Hungary, 1997; Volume 24, pp. 81–91. [Google Scholar]
- Röszler, P. Beiträge zur Kenntnis der Ameisenfauna von Siebenbürgen und Ungarn. Verhandlungen und Mitteilungen des Siebenbürgischen Vereins für Naturwissenschaften zu Hermannstadt 1935, 83–84, 72–83. [Google Scholar]
- Röszler, P. Myrmecologisches aus dem Jahre 1938. Zool. Anz. 1951, 146, 88–96. [Google Scholar]
- Csősz, S.; Markó, B. Redescription of Tetramorium hungaricum Röszler, 1935, a related species of T. caespitum (Linnaeus, 1758) (Hymenoptera: Formicidae). Myrmecol. Nachr. 2004, 6, 49–59. [Google Scholar]
- Schlick-Steiner, B.C.; Steiner, F.M.; Moder, K.; Seifert, B.; Sanetra, M.; Dyreson, E.; Stauffer, C.; Christian, E. A multidisciplinary approach reveals cryptic diversity in Western Palearctic Tetramorium ants (Hymenoptera: Formicidae). Mol. Phylogenet. Evol. 2006, 40, 259–273. [Google Scholar] [CrossRef]
- Csősz, S.; Wagner, H.C.; Bozsó, M.; Seifert, B.; Arthofer, W.; Schlick-Steiner, B.C.; Steiner, F.M.; Pénzes, Z. Tetramorium indocile Santschi, 1927 stat. rev. is the proposed scientific name for Tetramorium sp. C sensu based on combined molecular and morphological evidence (Hymenoptera: Formicidae). Zool. Anz. 2014, 253, 469–481. [Google Scholar] [CrossRef]
- Csősz, S.; Radchenko, A.G.; Schulz, A. Taxonomic revision of the Palaearctic Tetramorium chefketi species complex (Hymenoptera: Formicidae). Zootaxa 2007, 1405, 1–38. [Google Scholar] [CrossRef]
- Seifert, B.; Schlick-Steiner, B.C.; Steiner, F.M. Myrmica constricta Karavajev, 1934-a cryptic sister species of Myrmica hellenica Finzi, 1926 (Hymenoptera: Formicidae). Soil Org. 2009, 81, 53–76. [Google Scholar]
- Gallé, L.; Markó, B.; Kiss, K.; Kovács, É.; Dürgő, H.; Kőváry, K.; Csősz, S. Ant fauna of Tisza river basin (Hymenoptera: Formicidae). Tiscia Monogr. Ser. 2005, 7, 149–197. [Google Scholar]
- Gallé, L. Adatok a Bakony hegység hangyafaunájának ismeretéhez (Hymenoptera, Formicidae) [Contributions to the knowledge of the myrmecofauna (Hymenoptera, Formicidae) of the Bakony mountain]. Veszprém M. Múzeumok Közleményei 1979, 14, 239–244. [Google Scholar]
- Tartally, A.; Csősz, S. Adatok a Maculinea boglárkalepkék (Lepidoptera: Lycaenidae) kárpát-medencei hangyagazdáiról [Data on the ant hosts of the Maculinea butterflies (Lepidoptera: Lycaenidae) of Hungary]. Természetvédelmi Közlemények 2004, 11, 309–317. [Google Scholar]
- Csősz, S.; Markó, B.; Kiss, K.; Tartally, A.; Gallé, L. The ant fauna of the Fertő-Hanság National Park (Hymenoptera: Formicoidea). In The Fauna of the Fertő-Hanság National Park; Mahunka, S., Ed.; Hungarian Natural History Museum: Budapest, Hungary, 2002; pp. 617–629. [Google Scholar]
- Gallé, L. Ecological and zoocoenological conditions of the Formicoidea fauna at Tiszakürt. Tiscia 1967, 3, 67–72. [Google Scholar]
- Gallé, L. The ant fauna of the Kiskunság National Park. In The Fauna of Kiskunság National Park; Mahunka, S., Ed.; Akadémiai Kiadó: Budapest, Hungary, 1986; pp. 427–434. [Google Scholar]
- Gallé, L. Factors stabilizing the ant populations (Hymenoptera: Formicidae) in the grass associations of the Tisza Basin. Tiscia 1975, 10, 61–66. [Google Scholar]
- Gallé, L. Formicidae populations of the ecosystems in the environs of Tiszafüred. Tiscia 1972, 7, 59–68. [Google Scholar]
- Gallé, L. Habitat and niche analysis of grassland ants. Entomol. Gen. 1986, 11, 197–211. [Google Scholar] [CrossRef]
- Csősz, S. Hangya-faunisztikai adatok a Körös-Maros Nemzeti Parkból: A mályvádi erdők [Ant-faunistic investigations in the Körös-Maros National Park: The Mályvád-Forests]. Crisicum 2000, 3, 183–187. [Google Scholar]
- Markó, B.; Csősz, S. Die europäischen ameisenarten (Hymenoptera: Formicidae) des Herrmannstädter (Sibiu, Rumänien) Naturkundemuseums I.: Unterfamilien Ponerinae, Myrmicinae und Dolichoderinae. Ann. Hist. Nat. Mus. Nat. Hung. 2002, 94, 109–121. [Google Scholar]
- Gallé, L.; Kanizsai, O.; Maák, I.; Lőrinczi, G. Close nesting association of two ant species in artificial shelters: Results from a long-term experiment. Acta Zool. Acad. Sci. Hung. 2014, 60, 359–370. [Google Scholar]
- Seifert, B. Ameisen, Beobachten, Bestimmen; Naturbuch Verlag: Augsburg, Germany, 1996; ISBN 978-3894401702. [Google Scholar]
- Gallé, L. The Formicoid fauna of the Hortobágy. In The Fauna of the Hortobágy National Park; Mahunka, S., Ed.; Akadémiai Kiadó: Budapest, Hungary, 1981; pp. 307–311. [Google Scholar]
- Gallé, L. Ecological and zoocoenological investigation of the Formicoidea fauna of the flood area of the River Tisza. Tiscia 1966, 2, 113–118. [Google Scholar]
- Ionescu-Hirsch, A.; Markó, B.; Csősz, S. Camponotus tergestinus Müller, 1921 (Hymenoptera: Formicidae): First record of a rare species for Romania and Hungary. Entomol. Romanica 2009, 14, 19–22. [Google Scholar]
- Gallé, L. Study of ant populations in various grassland ecosystems. Acta Biol. Szeged. 1972, 18, 159–164. [Google Scholar]
- Tartally, A.; Csősz, S. Blaskovics-pusztai gyepek értékelése a rajtuk talált hangyaközösségek (Hymenoptera: Formicidae) alapján [The evaluation of the Blaskovics-puszta’s grasslands and their ant (Hymenoptera: Formicidae) communities]. Crisicum 1999, 2, 133–140. [Google Scholar]
- Csősz, S.; Markó, B. European Ant Species (Hymenoptera: Formicidae) in the ant collection of the natural history museum of Sibiu (Hermannstadt/Nagyszeben), Romania II. Subfamily Formicinae. Ann. Hist. Nat. Mus. Nat. Hung. 2005, 97, 225–240. [Google Scholar]
- Mocsáry, A. Ordo Hymenoptera. In Fauna Regni Hungariae. Arthropoda.; Paszlavsky, J., Ed.; Királyi Magyar Természettudományi Társulat: Budapest, Hungary, 1918. [Google Scholar]
- Van Loon, A.J.; Boomsma, A.; Andrásfalvy, A. A new polygynous Lasius species from Central Europe. Insectes Soc. 1990, 37, 348–362. [Google Scholar] [CrossRef]
- Barrett, K.E. Ants from Hungary and Bulgaria. Entomologist 1970, 103, 139–140. [Google Scholar]
- Csősz, S.; Salata, S.; Borowiec, L. Three Turano-European species of the Temnothorax interruptus group (Hymenoptera: Formicidae) demonstrated by quantitative morphology. Myrmecol. News 2018, 26, 101–119. [Google Scholar] [CrossRef]
- Varga, Z. Geographical patterns of biological diversity in the Palaearctic Region and the Carpathian Basin. Acta Zool. Acad. Sci. Hung. 1995, 41, 71–92. [Google Scholar]
- Borhidi, A. Magyarország Növénytársulásai [Plant. Communities of Hungary]; Akadémiai Kiadó: Budapest, Hungary, 2003; ISBN 963-05-7983-9. [Google Scholar]
- Varga, Z. Biogeográfia: Az élet földrajza [Biogeography: Geography of life]. In Biogeográfia: Az élet földrajza PARS Könyvek (12); Peregovits, L., Ed.; PARS Kft: Nagykovácsi, Hungary, 2019; ISBN 978-963-88339-9-0. [Google Scholar]
- Seifert, B. A taxonomic revision of the Palaearctic members of the subgenus Lasius s. str (Hymenoptera, Formicidae). Soil Org. 2020, 92, 15–86. [Google Scholar]
- Borowiec, L.; Lapeva-Gjonova, A.; Salata, S. Three species of Aphaenogaster Mayr, 1853 (Hymenoptera: Formicidae) new to Bulgarian fauna. Acta Zool. Bulg. 2019, 71, 613–616. [Google Scholar]
- Petrov, I.Z. Mravi Srbije i Crne Gore [The ants of Serbia and Montenegro]; SANU [Serbian Academy of Sciences and Arts]: Beograd, Serbia, 2006; ISBN 978-8670254084. [Google Scholar]
- Bračko, G. Checklist of the ants of Slovenia (Hymenoptera: Formicidae). Nat. Slov. 2007, 9, 15–24. [Google Scholar]
- Tăușan, I.; Lapeva-Gjonova, A. Camponotus samius Forel, 1889 (Hymenoptera: Formicidae)-at the north edge of its European distribution. North. West. J. Zool. 2017, 13, 352–354. [Google Scholar]
- Werner, P.; Bezděčka, P.; Bezděčková, K.; Pech, P. An updated checklist of the ants (Hymenoptera: Formicidae) of the Czech Republic. Acta Rerum Nat. 2018, 22, 5–12. [Google Scholar]
- Werner, P.; Wiezik, M. Vespoidea: Formicidae (mravencoviti). Acta Entomol. Mus. Nationnalis Pragae 2007, 11, 133–164. [Google Scholar]
- Bračko, G.; Wagner, H.C.; Schulz, A.; Gioahin, E.; Matičič, J.; Tratnik, A. New investigation and a revised checklist of the ants (Hymenoptera: Formicidae) of the Republic of Macedonia. North. West. J. Zool. 2014, 10, 10–24. [Google Scholar]
- Dekoninck, W.; Vankerkhoven, F. Checklist of the Belgian ant-fauna (Formicidae, Hymenoptera). Bull. Inst. R. Sci. Natur. Belg. Entomol. 2001, 71, 263–266. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Csősz, S.; Báthori, F.; Gallé, L.; Lőrinczi, G.; Maák, I.; Tartally, A.; Kovács, É.; Somogyi, A.Á.; Markó, B. The Myrmecofauna (Hymenoptera: Formicidae) of Hungary: Survey of Ant Species with an Annotated Synonymic Inventory. Insects 2021, 12, 78. https://doi.org/10.3390/insects12010078
Csősz S, Báthori F, Gallé L, Lőrinczi G, Maák I, Tartally A, Kovács É, Somogyi AÁ, Markó B. The Myrmecofauna (Hymenoptera: Formicidae) of Hungary: Survey of Ant Species with an Annotated Synonymic Inventory. Insects. 2021; 12(1):78. https://doi.org/10.3390/insects12010078
Chicago/Turabian StyleCsősz, Sándor, Ferenc Báthori, László Gallé, Gábor Lőrinczi, István Maák, András Tartally, Éva Kovács, Anna Ágnes Somogyi, and Bálint Markó. 2021. "The Myrmecofauna (Hymenoptera: Formicidae) of Hungary: Survey of Ant Species with an Annotated Synonymic Inventory" Insects 12, no. 1: 78. https://doi.org/10.3390/insects12010078
APA StyleCsősz, S., Báthori, F., Gallé, L., Lőrinczi, G., Maák, I., Tartally, A., Kovács, É., Somogyi, A. Á., & Markó, B. (2021). The Myrmecofauna (Hymenoptera: Formicidae) of Hungary: Survey of Ant Species with an Annotated Synonymic Inventory. Insects, 12(1), 78. https://doi.org/10.3390/insects12010078







