Simple Summary
The European pepper moth is an important pest of many crops; however, some countries, such as Brazil, do not have insecticides registered for combating this pest. In particular, the egg stage of the biological cycle of this moth is the most difficult life stage to control. In this sense, biological control agents, including egg parasitoids and entomopathogenic fungi, can be an alternative to pesticides. In this study, laboratory tests were conducted to evaluate the susceptibility of two species of egg parasitoids (Trichogrammatidae family) to entomopathogenic fungal strains (including two commercial bioinsecticides). These fungal strains were applied on eggs of the European pepper moth before and after parasitism by the parasitoids. Overall, the entomopathogens reduced the parasitism rate, adult emergence, and longevity of adult parasitoids by less than 30%. The results obtained constitute the first step in designing effective pest control strategies. Future research should investigate the sub-lethal effects of the fungal strains on the parasitoids in the field.
Abstract
The European pepper moth, Duponchelia fovealis (Lepidoptera: Crambidae), is a key pest in strawberry production. Entomopathogenic fungi (EF) and parasitoids of the Trichogrammatidae family are effective biological control agents of this pest with the potential to be used jointly for improved efficacy. This study aims to evaluate the susceptibility of Trichogramma atopovirilia and Trichogramma pretiosum to two Beauveria bassiana strains (B2 and B3) and two commercial bioinsecticides (Bovemax® and Methamax®) by applying them to D. fovealis eggs in pre- and post-parasitism periods. Pre-parasitism application of B2 and B3 did not affect the percentage of D. fovealis eggs parasitized by either Trichogramma species, except in the case of T. atopovirilia when eggs were sprayed with B3 at 1.5 × 105 conidia mL−1 (16.7% less than the control). In contrast, eggs sprayed with 1.5 × 108 conidia mL−1 of the commercial bioinsecticides were not parasitized by any Trichogramma species. Overall, the EF tested reduced the parasitism rate, adult emergence, and longevity of Trichogramma adults by less than 30% in all cases. The adverse effects of the B. bassiana strains and commercial products on the biological traits of both Trichogramma species were minimal, meaning that these agents can be used jointly in D. fovealis control strategies.
1. Introduction
In Brazil, strawberry (Fragaria x ananassa) cultivation is of particular socioeconomic importance because of its prevalence in small rural properties, providing farmers with additional income [1]. Damage due to disease and pests over the growing season can severely limit yields. The European pepper moth, Duponchelia fovealis (Lepidoptera: Crambidae), which is native to the Mediterranean region, has become a major pest in strawberry fields in Brazil [2,3]. Its activity in strawberry cultivation has been previously noted in Portugal, Italy, France, and Turkey [4,5,6]. Duponchelia fovealis larvae feed on leaves and flowers, resulting in physical damage that can create entry points for pathogenic microorganisms and increase the risk of crop losses [3]. No authorized chemical pesticides are currently available in Brazil to control this exotic pest [7].
In this context, biological agents are important tools for controlling D. fovealis populations in strawberry plantations. Hymenoptera parasitoids represent one of the most important functional groups in insect pest regulation and are used in many crop systems worldwide [8]. Species of the Trichogrammatidae family are parasitoids that develop in the eggs of a wide range of insect species, making them one of the leading biological control agents employed worldwide. Indeed, they are involved in pest control programs for over 200 insect species [8]. In particular, members of the Trichogrammatidae family have considerable potential as cost-effective control agents against Lepidoptera pests due to their ability to be easily mass-produced [8,9,10,11,12]. However, the use of non-selective pesticides in agricultural production systems limits the successful implementation of biological control programs that use Trichogrammatidae species due to the sensitivity of these organisms [11]. Moreover, pesticide application can negatively affect beneficial arthropods, cause toxicity to humans, and may contaminate the final crop products, thus becoming especially relevant for those that are consumed in fresh, such as fruits and vegetables, including strawberries [13]. Effectively integrating different biological control agents could, therefore, minimize the use of aggressive chemicals.
Entomopathogenic fungi (EF) are a potential component of integrated pest management (IPM) programs for strawberry cultivation [14]. While some studies have reported laboratory infections of non-target organisms [15,16,17], EF are less effective under field conditions [18], and generally do not harm ecosystem service providers such as pollinators, earthworms, predatory arthropods, and parasitoids, when at low concentrations. However, given the potential of EF to have negative side effects on biocontrol agents [19], preliminary studies are necessary to evaluate the compatibility of EF with other beneficial organisms. This highlights the need for research on the selectivity and effectiveness of commercial microbial insecticides against D. fovealis since this may uncover opportunities to implement sustainable strawberry pest management strategies. In 2010, during the collection of D. fovealis individuals in Paraná state, Brazil, researchers observed that larvae and pupae of this species were infected by Beauveria bassiana ((Balsamo-Crivelli) Vuillemin) (Hypocreales: Cordycipitaceae) [3]. Subsequent spray applications of two B. bassiana strains that were isolated from Coleoptera species present in strawberry crops caused high mortality among D. fovealis larvae (>60%) [20]. Recent research on B. bassiana infections of D. fovealis has shown that although the fungus effectively controlled larvae and pupae, it did not affect the eggs [21]. Indeed, eggs are considered to be the most difficult insect life stage to kill because of their protective structure, which provides a barrier against penetration by insecticides and pathogens [22,23,24].
In the field, D. fovealis deposits its eggs on the abaxial side of strawberry leaves [2], reducing the effectiveness of chemical applications. This appears to suggest that the combined use of EF and egg parasitoids could increase pest mortality. However, there is a paucity of information available about the use of egg parasitoids for controlling D. fovealis. Trials involving Trichogramma species on D. fovealis eggs indicated that Trichogramma galloi Zucchi and Trichogramma pretiosum Riley (Hymenoptera: Trichogrammatidae) are promising at controlling the pest’s eggs [9]. Studies have also determined the ideal number of such biological agents to be released in strawberry fields [25]. Other species from the Trichogrammatidae family have been reported as effective biological agents against D. fovealis, including Trichogramma exiguum Pinto and Platner, Trichogramma atopovirilia Oatman and Platner [10], Trichogramma cacoeciae Marchal, and Trichogramma brassicae Bezdenko [26].
Combining biological control agents like egg parasitoids and EF may result in more effective pest control [16,19,24]. For instance, the selectivity of B. bassiana and Metarhizium anisopliae (Metschnikoff) Sorokin to T. pretiosum was assessed previously, and the results suggested that the simultaneous use of both EF with this parasitoid is possible [27]. Even though M. anisopliae decreased emergence and increased mortality among the parasitoids, there were no significant changes in the number of parasitized eggs and sex ratio when M. anisopliae was applied pre-parasitism [27]. However, the risk of using B. bassiana has been shown to vary depending on the fungal strain, insect host, and parasitoid being evaluated [22]. The interaction between T. pretiosum and the two strains of B. bassiana used against Ephestia kuniella Zeller (Lepidoptera: Pyralidae) was considered innocuous because the parasitism rate was reduced by less than 30% when these fungal strains were applied either pre- or post-parasitism [22]. Given these results, further studies are needed to determine how to combine different biological control agents without impairing their effectiveness while developing strategies that may improve their efficacy [16,17].
This study aims to assess the susceptibility of T. pretiosum and T. atopovirilia to two B. bassiana strains and two commercial bioinsecticides applied to eggs of D. fovealis, either pre- or post-parasitism, in non-choice tests. In addition, a free-choice bioassay was conducted. The resulting information will be useful for designing future biological control strategies in IPM programs against D. fovealis.
2. Materials and Methods
2.1. Duponchelia fovealis Rearing
Specimens of D. fovealis were obtained from a laboratory colony (Universidade Federal do Paraná, Curitiba, Paraná, Brazil) established from locally collected insects and reared at 25 ± 2 °C, 70 ± 10% RH, and 14 L:10 D photoperiod [28]. Adults were kept in plastic cages (15 × 15 × 12.5 cm) and fed on a nutritional solution consisting of 0.5 g nipagin, 0.5 g sorbic acid, 30.0 g sugar, 10 mL honey, 170 mL beer, and 500 mL distilled water. The walls of these cages were wrapped with a paper towel for egg deposition. Strips of paper towels containing eggs were fixed in sterile, vented, plastic containers (7 × 4 × 4.5 cm). Larvae were fed on an artificial diet consisting of seeds of Phaseolus vulgaris L. “carioca” cultivar (65 g), wheat germ (50 g), textured soy protein (25 g), casein (25 g), beer yeast (31 g), bacteriological grade agar (20 g), ascorbic acid (3 g), sorbic acid (1.5 g), methylparaben-nipagin (2.5 g), tetracycline (0.14 g), 30% formaldehyde (3 mL), Vanderzant vitamin mixture (8 mL), and distilled water (1000 mL), to which V8® vegetable juice (50 mL) was added as a phagostimulant [28]. Pupae were transferred to sterile Petri dishes (15 × 2 cm) with moist filter paper until adult emergence.
2.2. Trichogramma Rearing
To obtain a sufficient population of T. pretiosum and T. atopovirilia, these parasitoids were reared on Mythimma sequax Franclemont (Lepidoptera: Noctuidae) eggs. Newly hatched M. sequax larvae were kept in plastic containers (5 × 4 cm) and fed on an artificial diet until they reached the 4th instar [29]. They were then individually placed in glass tubes (2 × 8 cm), where they were kept until they reached the pupae stage. Adults were kept in cages (45 × 30 × 22 cm) and fed on honey (10%) and water. Strips of paper were provided for oviposition, and the eggs were used to maintain the Trichogramma populations. Rearing was conducted under controlled conditions (20 ± 1 °C, 70 ± 10% RH, and 12 L:12 D photoperiod). Populations of T. pretiosum and T. atopovirilia were kept in glass tubes (1 × 10 cm) under the same controlled conditions as those described for M. sequax. At 48 h intervals, approximately 100 M. sequax eggs were offered for parasitism. Adult parasitoids were fed diets of pure honey.
2.3. Entomopathogenic Fungal Strains and Commercial Bioinsecticides
Beauveria bassiana B2 (Genbank: KU751847) and B3 (Genbank: KU751848) strains were selected because they have been shown to cause high mortality rates in D. fovealis (>60%) in previous studies [20]. Monospore cultures of each isolate were inoculated into Petri dishes containing Niger Seed Agar (NSA) and incubated for 14 days (28 ± 2 °C) [30]. The mycelia and conidia were subsequently scraped from the Petri dishes and transferred to vials (20 mL) containing 0.85% saline solution (NaCl containing 0.01% Tween 80). The vials were agitated on a vortex mixer, and the resulting suspensions were filtered to remove mycelial fragments. The concentrations of the resulting suspensions were determined by hemocytometer counts and were adjusted to 1.5 × 105 and 1.5 × 108 conidia mL−1. The viability of the isolates and bioinsecticides was verified by spraying 100 μL of a suspension containing 104 conidia mL−1 onto Petri dishes containing Sabouraud Agar. The dishes were maintained at 25 °C and 100% RH for 16 h. Viability was determined by viewing 200 conidia under a microscope (400× magnification). Conidia that had produced a germ tube were considered viable. This procedure was used for all isolates, and viability was >90% in every case.
In addition, the following two commercial bioinsecticides available as emulsifiable concentrates (EC) were used as positive controls in the bioassays: Bovemax®EC (Beauveria bassiana Strain CG 716, containing 1.5 × 109 viable conidia per mL of commercial product) and Methamax®EC (Metarhizium anisopliae Strain IBCB 348, containing 2.5 × 109 viable spores per mL of commercial product). These bioinsecticides were purchased from Novozymes BioAg (Brazil) and were diluted to obtain test concentrations of 1.5 × 105 and 1.5 × 108 conidia mL−1. The highest of these two concentrations corresponds to the recommended dose for field applications. Throughout this paper, Bovemax®EC and Methamax®EC will be referred to as Bb CG716 and Ma IBCB348, respectively.
2.4. Trichogramma atopovirilia and Trichogramma pretiosum Parasitism in Free-Choice Test
Paper strips with 20 eggs (24 h) of D. fovealis (from the population reared as described in Section 2.1) were sprayed with 0.5 mL of a suspension containing either 1.5 × 105 or 1.5 × 108 conidia mL−1 of each B. bassiana strain, commercial bioinsecticide, or the control (sterilized distilled water + 0.01% Tween 80). Spraying was performed using an airbrush (Pneumatic Sagyma® SW775, São Paulo, SP, Brazil) coupled to a pump at constant pressure (1.2 kgf cm−2). After being sprayed, paper strips were left to dry in a flow chamber for 1 h. A single paper strip sprayed with a fungal treatment was then placed into a sterilized glass tube (2.5 × 8.0 cm), along with a non-sprayed control and a T. atopovirilia or T. pretiosum female (24 h old) from the population reared as described in Section 2.2. The tubes were closed and kept under controlled conditions (25 ± 2 °C, 70 ± 10% RH, and 12 L:12 D photoperiod).
After 24 h, the parasitoids were collected and isolated in glass tubes. After their death, they were transferred into sterilized Eppendorf tubes containing moistened cotton and kept at 28 ± 2 °C for 14 days to verify the occurrence of fungal extrusion. The paper strips with eggs were placed into separate glass tubes (5 × 1 cm) and kept under the same controlled conditions. The percentage of parasitism by each Trichogramma species was assessed using a free-choice test based on the numbers of parasitized eggs on the control and treated paper strips. Specifically, this test compared each B. bassiana strain (B2 and B3) or bioinsecticide (Bb CG716 and Ma IBCB348) with its corresponding control. Fifteen replicates per treatment combination were performed. Each replicate consisted of a tube with two paper strips (one treated and the other non-treated) with 20 D. fovealis eggs each and a female parasitoid.
2.5. Spraying of Entomopathogenic Fungi Pre- and Post-Parasitism (No-Choice Tests)
The pre-parasitism bioassay was performed using 0.5 mL suspensions containing 1.5 × 108 conidia mL−1 of each B. bassiana strain or commercial bioinsecticide, or the control (sterilized distilled water + 0.01% Tween 80) sprayed on unparasitized eggs of D. fovealis (ca. 24 h old, from the population reared as described in Section 2.1) fixed on paper strips. Spraying was conducted with the same equipment as in the bioassay described in Section 2.4. After being sprayed, paper strips were left to dry in a flow chamber for 1 h. Then, 15 paper strips (containing 20 D. fovealis eggs each) for each fungal strain, commercial bioinsecticide, and control groups were placed into separate sterilized glass tubes (2.5 × 8.0 cm) with a T. atopovirilia or a T. pretiosum female (24 h old) from the population reared as described in Section 2.2. The tubes were closed and kept for 24 h under controlled conditions (25 ± 2 °C, 70 ± 10% RH, and 12 L:12 D photoperiod). The parasitoids were transferred into glass tubes and, after their death, they were isolated in sterilized Eppendorf tubes and kept at 28 ± 2 °C for 14 days to verify the occurrence of fungal extrusion. Mortality due to the fungus was confirmed by observing the insect under a stereomicroscope at 20× or 40× magnification [31].
In the post-parasitism bioassay, a female of T. atopovirilia or T. pretiosum (24 h old) was confined with a paper strip containing 20 D. fovealis eggs in a glass vial for 24 h. After this period, the parasitoids were removed, and the paper strips were sprayed in the same manner as in the former bioassay and kept for 1 h in a flow chamber. Then, the paper strips were isolated in glass tubes and kept under the same controlled conditions as previously described. The parasitoids used in this bioassay were not isolated to assess their cause of death because they were not in contact with the fungi.
In both bioassays, the following biological parameters were assessed daily: (1) number of parasitized D. fovealis eggs; (2) percentage of emergence (%E) calculated as %E = (TE/TO) × 100, where TE is the total number of emerged adults and TO is the total number of parasitized eggs; (3) longevity of emerged adults; (4) egg-to-adult development period; (5) offspring sex ratio (R), computed as R = TF/TE, where TF is the total number of emerged females. The percentage reductions (PR) in emergence due to parasitized eggs, adult longevity, and percentage of parasitism relative to the control were calculated using the following equation: PR (%)= [1 − (Q/q) × 100], where PR is the percentage reduction in the relevant biological parameter, Q is the average value of the parameter for the EF treatment, and q represents the mean value of the parameter obtained in the control.
The selectivity/toxicity of B. bassiana isolates and commercial bioinsecticides to egg parasitism by T. atopovirilia and T. pretiosum was classified according to the IOBC (International Organization for Biological Control) criteria for laboratory tests, where: 1 = innocuous (<30%), 2 = slightly harmful (30–79%), 3 = moderately harmful (80–99%), and 4 = harmful (>99%) [32]. The experimental design was completely randomized, with four treatments and one control per parasitoid. Fifteen replicates were used per combination, and each replicate consisted of a tube with D. fovealis eggs (a paper strip with 20 eggs) and a parasitoid.
2.6. Statistical Analysis
Data were checked for normality and homoscedasticity using the Shapiro-Wilk and Bartlett tests, respectively [33,34]. When required, data were arcsine transformed. Data from the free-choice test were analyzed using the Wilcoxon non-parametric test. In the pre- and post-parasitism bioassays, data were submitted to an analysis of variance (ANOVA), with period of parasitism, Trichogramma species, and entomopathogen treatment, as well as their interactions, as fixed factors. Means were compared with the Tukey honest significant difference (HSD) test [35]. Statistical analyses were conducted in R v.3.6.2 [36].
3. Results
3.1. Trichogramma atopovirilia and Trichogramma pretiosum Parasitism in Free-Choice Test
In general, the B2 and B3 strains did not affect the percentage of D. fovealis eggs parasitized by either Trichogramma species (Figure 1 and Figure 2).
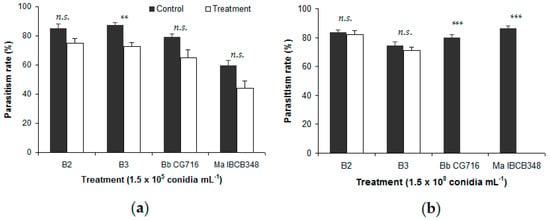
Figure 1.
Percentage of Duponchelia fovealis eggs parasitized by Trichogramma atopovirilia in free-choice tests, comparing eggs sprayed with Beauveria bassiana strains (B2 and B3) and commercial bioinsecticides (Bb CG716 and Ma IBCB348) vs. control solutions: (a) 1.5 × 105 conidia mL−1; (b) 1.5 × 108 conidia mL−1. Error bars indicate standard errors (n = 15). Significant differences between a given treatment (entomopathogen) and its corresponding control (water), according to the Wilcoxon test, are shown over the bars as follows: n.s. = not significant; ** = p-value < 0.01; *** = p-value < 0.001.
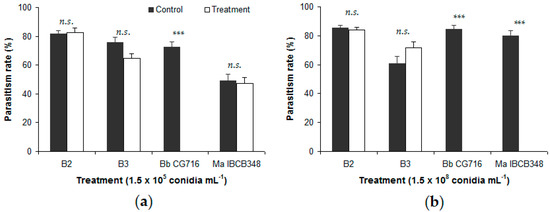
Figure 2.
Percentage of Duponchelia fovealis eggs parasitized by Trichogramma pretiosum in free-choice tests, comparing eggs sprayed with Beauveria bassiana strains (B2 and B3) and commercial bioinsecticides (Bb CG716 and Ma IBCB348) vs. control solutions: (a) 1.5 × 105 conidia mL−1; (b) 1.5 × 108 conidia mL−1. Error bars indicate standard errors (n = 15). Significant differences between a given treatment (entomopathogen) and its corresponding control (water), according to the Wilcoxon test, are shown over the bars as follows: n.s. = not significant; *** = p-value < 0.001.
No significant differences were detected between either B. bassiana strain and the respective control in the percentage of eggs parasitized, except for B3 at 1.5 × 105 conidia mL−1 in the presence of T. atopovirilia (Figure 1a). In contrast, commercial bioinsecticides affected the parasitism process negatively. Eggs sprayed with 1.5 × 108 conidia mL−1 of Bb CG716 or Ma IBCB348 were not parasitized by either Trichogramma species, whereas their controls showed parasitism rates of between 79.9% and 86.2% of eggs (Figure 1b and Figure 2b). Eggs sprayed with Bb CG716 at the lowest concentration (1.5 × 105 conidia mL−1) were not parasitized by T. pretiosum (Figure 2a), while parasitism by T. atopovirilia remained unaffected (Figure 1a). Spraying with Ma IBCB348 at 1.5 × 105 conidia mL−1 did not affect the parasitism behavior of either Trichogramma species (Figure 1a and Figure 2a).
Infection rates among parasitoids kept under controlled conditions for 14 days were uniformly low (<30%) following exposure to both B. bassiana strains and commercial bioinsecticides at concentrations of 1.5 × 105 conidia mL−1 (F3119 = 1.22; p-value = 0.305) or 1.5 × 108 conidia mL−1 (F3119 = 0.97; p-value = 0.408). Moreover, no differential tolerance to EF products was observed between T. atopovirilia and T. pretiosum at both 1.5 × 105 conidia mL−1 (F1119 = 0.33; p-value = 0.565) and 1.5 × 108 conidia mL−1 (F1119 = 0.07; p-value = 0.795). Mortality in the control groups was not fungus-related.
3.2. Spraying of Entomopathogenic Fungi Pre- and Post-Parasitism (No-Choice Tests)
The biological parameters of the Trichogramma species (parasitism rate, emergence rate, offspring sex ratio, and duration of the egg-to-adult period) were significantly affected by the period in which parasitoids were exposed to the EF (pre- or post-parasitism), the EF treatments themselves and the interactions among them, as shown by a full ANOVA model (p-values ranging from < 0.001 to 0.039). Moreover, sex ratio (p-value < 0.001) and duration of the egg-to-adult period (p-value = 0.013) differed between Trichogramma species. Consequently, data were analyzed separately for each period of parasitism and Trichogramma species.
EF application during the pre-parasitism period did not result in any change in the biological parameters studied relative to the control treatment (Table 1). In the case of T. pretiosum, the only significant difference was observed in the emergence rate between the B2 and B3 treatments (Table 1). In addition, the percentage of emerged adults was lower in T. atopovirilia than in T. pretiosum when they were treated with B2, though the opposite was observed following application of Bb CG716 (Table 1). The length of the egg-to-adult period was not affected by any treatment in either of the Trichogramma species (Figure 3a). The longevity of T. atopovirilia was reduced by the application of Bb CG716 and Ma IBCB348, while the B. bassiana strains had no effect relative to the control (Figure 4). The treatments did not affect the longevity of T. pretiosum (Figure 4).

Table 1.
Biological parameters (% eggs parasitized, % emerged adults, and offspring sex ratio) of Trichogramma atopovirilia and Trichogramma pretiosum individuals emerged from Duponchelia fovealis eggs sprayed with Beauveria bassiana strains (B2 and B3) and commercial bioinsecticides (Bb CG716 and Ma IBCB348) pre-parasitism. Data represent averages ± standard errors (n = 15).
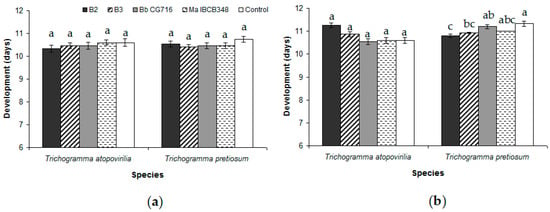
Figure 3.
Average length of the egg-to-adult period of development of Trichogramma atopovirilia and Trichogramma pretiosum individuals emerged from Duponchelia fovealis eggs sprayed with Beauveria bassiana strains (B2 and B3) and commercial bioinsecticides (Bb CG716 and Ma IBCB348) pre- (a) and post-parasitism (b). Error bars are standard errors (n = 15). Small letters over the bars indicate significant differences among treatments according to the Tukey HSD test (p-value < 0.05).
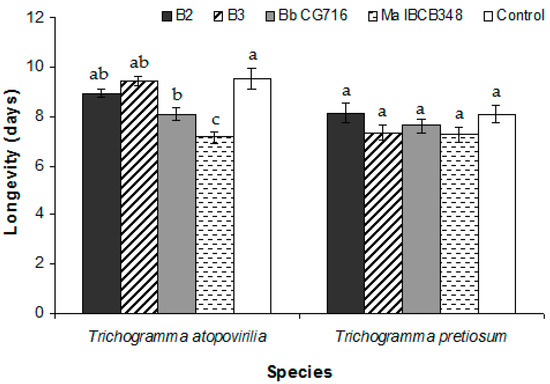
Figure 4.
Average longevity of Trichogramma atopovirilia and Trichogramma pretiosum individuals emerged from Duponchelia fovealis eggs sprayed with Beauveria bassiana strains (B2 and B3) and commercial bioinsecticides (Bb CG716 and Ma IBCB348) pre-parasitism. Error bars are standard errors (n = 15). Small letters over the bars indicate significant differences among treatments according to the Tukey HSD test (p-value < 0.05).
The percentage of eggs parasitized in the post-parasitism bioassay was not affected by the spraying of EF for either Trichogramma species (Table 2). The percentage of emerged T. atopovirilia adults following B2 application was lower than the control (Table 2). The offspring sex ratio in T. atopovirilia was not affected by the entomopathogens; on the other hand, for T. pretiosum, this biological trait was reduced by all the entomopathogen treatments considered in this study, especially B2 (Table 2). The only parameter that was significantly different between the Trichogramma species was the offspring sex ratio, which was greater for T. pretiosum under the control, Bb CG716 and Ma IBCB348 treatments, and greater for T. atopovirilia under the B2 treatment (Table 2). The length of the egg-to-adult period was not affected by EF in T. atopovirilia, while T. pretiosum individuals had a shorter development under the B2 and B3 treatments than under the control treatment (Figure 3b). The B. bassiana strains and bioinsecticides considered in this study caused reductions of under 30% in the percentage of eggs parasitized, emergence of adults, and longevity in both the pre- and post-parasitism bioassays for the two Trichogramma species. Therefore, all EF treatments were classified as innocuous (Class 1) according to the IOBC criteria.

Table 2.
Biological parameters (% eggs parasitized, % emerged adults and offspring sex ratio) of Trichogramma atopovirilia and Trichogramma pretiosum individuals emerged from Duponchelia fovealis eggs sprayed with Beauveria bassiana strains (B2 and B3) and commercial bioinsecticides (Bb CG716 and Ma IBCB348) post-parasitism. Data represent averages ± standard errors (n = 15).
4. Discussion
In the free-choice bioassay, parasitism rate was completely absent in the treatments with commercial bioinsecticides at 1.5 × 108 conidia mL−1; however, at a lower concentration (1.5 × 105 conidia mL−1), Bb CG716 did not affect the parasitism rate of T. atopovirilia, although it completely inhibited parasitism of T. pretiosum. In contrast, the B. bassiana strains tested (B2 and B3) did not affect the parasitism rates of either Trichogramma species. In a similar study, significant reductions in the parasitism rate of T. pretiosum were observed in a free-choice test when B. bassiana strains Unioeste 47 and Unioeste 57 were applied at concentrations of 1.5 × 109 conidia mL−1 on the eggs of Ephestia kuehniella Zeller (Lepidoptera: Pyralidae) [22]. These contrasting observations may be due to differences in the conidia concentrations applied, the B. bassiana strains used, or the target organism [22]. It is possible that T. pretiosum females can recognize the presence of a harmful or repellent substance deposited on the host eggs before parasitizing them [22,37]. Females may be able to recognize whether the host is infected by a fungus by walking back and forth over the eggs, touching them with the gustatory sensilla of their antennae [22,37,38]. Meanwhile, commercial products may contain carrying substances in their formulations, which could partially explain the stronger effect of these bioinsecticides on the Trichogramma species studied.
The pre- and post-parasitism (no-choice) EF application tests showed that all of the products were innocuous according to the IOBC selectivity/toxicity classification since parasitism was reduced by <30%. These results are consistent with previous observations on T. pretiosum [22]. Similarly, prior studies did not observe significant reductions in parasitism by T. pretiosum and T. atopovirilia when B. bassiana and M. anisopliae strains were applied on host eggs in no-choice tests [23,27,39]. The pattern observed in the current study of parasitoids being repelled by EF in the free-choice tests but accepting the infected eggs in the no-choice tests may indicate that the host eggs are suitable for parasitism by T. atopovirilia and T. pretiosum. In fact, Trichogramma females probe potential host eggs prior to parasitizing them by inserting the ovipositor and verifying the nutritional quality of the egg [40].
The egg-to-adult development period was not affected in either of the Trichogramma species by any entomopathogen treatment in the no-choice tests performed prior to parasitism in this study. In contrast, a minimal effect was observed in T. pretiosum development following post-parasitism with EF application. This is consistent with previous studies of T. pretiosum in which host eggs were sprayed with strains of B. bassiana and M. anisopliae [22,27]. Alterations of the length of the Trichogramma parasitoids’ egg-to-adult period may be related to changes in the host caused by the fungus [38]. However, the near absence of alterations in the egg-to-adult period reinforces prior test findings that D. fovealis eggs were not affected by EF [21].
The percentage of emergence in the no-choice tests performed in this study was not significantly affected by most entomopathogen treatments, while any significant reductions were under 30% relative to the control. Of all the pre-parasitism treatments, only the B. bassiana strains B2 and B3 caused reductions in this biological parameter. Meanwhile, only T. atopovirilia had a significantly lower percentage of emerged adults following post-parasitism application of B2 (75.8%) compared with the control (93.5%). These results are consistent with previous research showing that the emergence of T. pretiosum adults did not differ following pre- and post-parasitism spraying with B. bassiana strains (no-choice test) [22]. In contrast, significant reductions of T. pretiosum emergence have been reported as a result of the application of M. anisopliae (Unioeste 22) to eggs of E. kuehniella in no-choice tests, though this reduction was only observed following pre-parasitism application and accounted for less than 30% [27]. Furthermore, these authors did not observe significant reductions in this parameter when eggs were sprayed with B. bassiana strain Unioeste 1 [27]. Moreover, a study found that B. bassiana (Boveril WP PL63) and M. anisopliae (Metarril WP E9) sprayed onto eggs of Spodoptera frugiperda (Smith) (Lepidoptera: Noctuidae) prior to parasitism did not affect the emergence rate of T. atopovirilia [39].
The offspring sex ratio differed between the two Trichogramma species considered in this study. In both cases, this ratio was affected by EF application during the post-parasitism period. These results differ from previous observations of T. pretiosum in which offspring sex ratio was not altered by the host eggs being sprayed with B. bassiana and M. anisopliae strains either pre- or post- parasitism [22,27]. Alterations in sex ratio could occur due to interference with the behavior of Trichogramma adults or an effect on the fertilization of ova [41]. However, although significant changes were observed in the offspring sex ratio for both Trichogramma species, this ratio was greater than 0.6 in all treatments, which is considered satisfactory since sex ratio values of over 0.5 are suitable for mass rearing and do not hinder the success of biological control programs [42].
Concerning the longevity of emerged adults, only the commercial biopesticides significantly affected this trait in T. atopovirilia, in which longevity was reduced following pre-parasitism application of these compounds. In contrast, previous research did not report changes in longevity due to pre-parasitism application of B. bassiana and M. anisopliae strains (no-choice tests) [39]. The longevity of T. pretiosum was not affected by any of the treatments, which is consistent with previous reports involving T. pretiosum and B. bassiana and M. anisopliae strains [22,27].
The percentage of fungus-related mortality of adult parasitoids in the current study was low and consistent across the strains applied, which is crucial for the viability of combining parasitoids and EF within IPM strategies. The mortality patterns may be explained by the way in which EF were applied in the current study (no direct contact between EF and parasitoids) and the period of application. Previous research has shown that confirmed fungus-related mortality depends on the virulence of fungal strains, EF species, Trichogramma species, and the application period (pre- or post-parasitism) [22,27,39]. Furthermore, the interactions between the EF and Trichogramma species were minimal for all biological parameters considered in this study, especially when compared with chemical insecticides [11,43,44,45].
The effectiveness of the commercial biopesticides and EF strains tested in the present study in controlling D. fovealis has already been demonstrated [20]. Meanwhile, this study suggests that the residual effects of these biocontrol agents on T. atopovirilia and T. pretiosum were minimal. Moreover, these EF strains have shown low adverse impacts on two natural enemies of D. fovealis that are commonly present in Brazilian strawberry fields, Podisus nigrispinus (Dallas) (Hemiptera: Pentatomidae) and Harmonia axyridis (Pallas) (Coleoptera: Coccinellidae) [16]. Thus, the combined use of these biological control agents may be suitable for IPM programs. The use of EF with a parasitoid may improve the effectiveness of biological control strategies since these organisms act on the same pest species at different stages of its life cycle [23,39].
Overall, the results of this study suggest that the safety of EF is specific and depends on the fungal strain and conidia concentration, as well as the particular host and parasitoid under evaluation. Free and no-choice bioassays are, therefore, essential to confirm the selectivity and to better understand the effect that a given EF strain might have on a certain species of the Trichogramma genus. Because the results of free and no-choice bioassays can vary greatly, performing a single test would lead to biases or partial conclusions on the selectivity of an EF strain for a given Trichogramma species [22,23]. Moreover, combining the two approaches allows for a better understanding of the situations in which the EF would cause lower impacts on the performance and biological parameters of these parasitoids, which can inform the design of IPM strategies.
5. Conclusions
This study demonstrates that the adverse effects observed on the biological parameters of T. pretiosum and T. atopovirilia were minimal, suggesting that the B. bassiana strains and the commercial products tested could be harmless to these parasitoids; however, further research is needed to confirm this. Effectiveness against a particular pest and the innocuity against non-target organisms are key characteristics of good IPM practices. Thus, the results obtained constitute a first step in designing effective D. fovealis control strategies involving both EF and parasitoids. Future research should investigate the sub-lethal effects of these fungal strains on such parasitoids, besides involving field studies.
Author Contributions
Conceptualization, A.S.P., C.G.P., M.A.C.Z., and I.C.P.; formal analysis, E.S.A., A.S.P., and J.M.M.-A.; investigation, A.S.P. and C.G.P.; resources, M.A.C.Z. and I.C.P.; data curation, E.S.A. and J.M.M.-A.; writing—original draft preparation, E.S.A. and J.M.M.-A.; writing—review and editing, all authors; supervision, M.A.C.Z. and I.C.P.; project administration, M.A.C.Z. and I.C.P.; funding acquisition, J.M.M.-A., M.A.C.Z. and I.C.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by grants from the Coordination for the Improvement of Higher-Level Personnel (CAPES), the National Council for Scientific and Technological Development (CNPq), and the Araucária Foundation for the Support of Scientific and Technological Development (FA). The APC was funded through discount vouchers obtained by José M. Mirás-Avalos for reviewing several MDPI journals.
Acknowledgments
The authors are grateful to Tamara Akemi Takahashi and Luis Amilton Foerster, Laboratório de Controle Integrado de Insetos, Universidade Federal do Paraná, for providing the Trichogramma species. The authors express their deepest gratitude to Karin Hoch Fehlauer Ale and her team at EscritaLAB for correcting the English language of this manuscript. We are also grateful to Matthew Franklyn Sturgis for reading the manuscript as a native English speaker and providing us with suggestions.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study, in the collection, analysis, or interpretation of data, in the writing of the manuscript, or in the decision to publish the results.
References
- Ponce Ados, R.; Bastiani, M.I.D.; Minim, V.P.; Vanetti, M.C.D. Características físico-químicas e microbiológicas de morango minimamente processado. Ciência Tecnol. Aliment. 2009, 30, 113–118. [Google Scholar] [CrossRef]
- Zawadneak, M.A.C.; Vidal, H.R.; Santos, B. Lagarta-da-coroa, Duponchelia fovealis (Lepidoptera: Crambidae). In Pragas Introduzidas: Insetos e Acaros; Vilela, E., Zucchi, R.A., Eds.; FEALQ: Piracicaba, Brazil, 2015. [Google Scholar]
- Zawadneak, M.A.C.; Gonçalves, R.B.; Pimentel, I.C.; Schuber, J.M.; Santos, B.; Poltronieri, A.S.; Solis, M.A. First record of Duponchelia fovealis (Lepidoptera: Crambidae) in South America. Idesia 2016, 34, 91–95. [Google Scholar] [CrossRef]
- Bonsignore, C.P.; Vacante, V. Duponchelia fovealis (Zeller). A new emergency for strawberry? Protez. Colt. 2010, 3, 40–43. [Google Scholar]
- Franco, M.C.; Baptista, M.C. Duponchelia fovealis Zeller—Nova praga em Portugal. Frutas Legumes Flores 2010, 110, 34–35. [Google Scholar]
- Efil, L.; Özgür, O.; Efil, F. A new pest, Duponchelia fovealis Zeller, on strawberries in Turkey: Damage, distribution and parasitoid. J. Entomol. Zool. Stud. 2014, 2, 328–334. [Google Scholar]
- BRASIL, Ministério da Agricultura, Pecuária e Abastecimento. Agrofit—Sistema de Agrotóxicos Fitossanitários. Available online: http://agrofit.agricultura.gov.br/agrofit_cons/principal_agrofit_cons (accessed on 21 February 2020).
- Jalali, S.K. Natural occurrence, host range and distribution of trichogrammatid egg parasitoids. In Biological Control of Insect Pests Using Egg Parasitoids; Sithanantham, S., Ballal, C., Jalali, S., Bakthavatsalam, N., Eds.; Springer: New Delhi, India, 2013; pp. 67–76. [Google Scholar]
- Paes, J.P.P.; Lima, V.L.S.; Pratissoli, D.; de Carvalho, J.R.; de Freitas Bueno, R.C.O. Selection of parasitoids of the genus Trichogramma (Hymenoptera: Trichogrammatidae) and parasitism at different eggs ages of Duponchelia fovealis (Lepidoptera: Crambidae). Acta Sci. Biol. 2018, 40, e42216. [Google Scholar] [CrossRef]
- Rodrigues, R.; Jaras, L.I.; Poltronieri, A.S.; Pimentel, I.C.; Zawadneak, M.A.C. Selectivity of growth regulators and botanical insecticides in the parasitism of three Trichogramma species on eggs of Duponchelia fovealis Zeller (Lepidoptera: Crambidae). EntomoBrasilis 2017, 10, 26–32. [Google Scholar] [CrossRef]
- Feltrin-Campos, E.; Ringenberg, R.; Carvalho, G.A.; Glaeser, D.F.; de Oliveira, H.N. Selectivity of insecticides against adult Trichogramma pretiosum Riley (Hymenoptera: Trichogrammatidae) on cassava. J. Agric. Sci. 2019, 11, 546–552. [Google Scholar] [CrossRef]
- Schäfer, L.; Herz, A. Suitability of European Trichogramma species as biocontrol agents against the tomato leaf miner Tuta absoluta. Insects 2020, 11, 357. [Google Scholar] [CrossRef]
- Fenik, J.; Tankiewicz, M.; Biziuk, M. Properties and determination of pesticides in fruits and vegetables. Trac. Trend Anal. Chem. 2011, 30, 814–826. [Google Scholar] [CrossRef]
- Srinivasan, R.; Sevgan, S.; Ekesi, S.; Tamò, M. Biopesticide based sustainable pest management for safer production of vegetable legumes and brassicas in Asia and Africa. Pest. Manag. Sci. 2019, 75, 2446–2454. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, G. Review on safety of the entomopathogenic fungus Metarhizium anisopliae. Biocontrol. Sci. Technol. 2007, 17, 879–920. [Google Scholar] [CrossRef]
- Araujo, E.S.; Benatto, A.; Rizzato, F.B.; Poltronieri, A.S.; Poitevin, C.G.; Zawadneak, M.A.C.; Pimentel, I.C. Combining biocontrol agents with different mechanisms of action to control Duponchelia fovealis, an invasive pest in South America. Crop. Prot. 2020, 134, 105184. [Google Scholar] [CrossRef]
- Miranda-Fuentes, P.; Quesada-Moraga, E.; Aldebis, H.K.; Yousef-Naef, M. Compatibility between the endoparasitoid Hyposoter didymator and the entomopathogenic fungi Metarhizium brunneum: A laboratory simulation for the simultaneous use to control Spodoptera littoralis. Pest. Manag. Sci. 2020, 76, 1060–1070. [Google Scholar] [CrossRef]
- Jaronski, S.T.; Goettel, M.S.; Lomer, C.J. Regulatory requirements for ecotoxicological assessments of microbial insecticides—How relevant are they? In Environmental Impacts of Microbial Insecticides. Progress in Biological Control; Hokkanen, H.M.T., Hajek, A.E., Eds.; Springer: Dordrecht, The Netherlands, 2003; Volume 1, pp. 237–260. [Google Scholar]
- Jazzar, C.; Hammad, E.A.F. Efficacy of multiple biocontrol agents against the sweet potato whitefly Bemissia tabaci (Gennadius) (Homoptera: Aleyrodidae) on tomato. J. Appl. Entomol. 2004, 128, 188–194. [Google Scholar] [CrossRef]
- Poitevin, C.G.; Porsani, M.V.; Poltronieri, A.S.; Zawadneak, M.A.C.; Pimentel, I.C. Fungi isolated from insects in strawberry crops act as potential biological control agents of Duponchelia fovealis (Lepidoptera: Crambidae). Appl. Entomol. Zool. 2018, 53, 323–331. [Google Scholar] [CrossRef]
- Baja, F.; Poitevin, C.G.; Araujo, E.S.; Mirás-Avalos, J.M.; Zawadneak, M.A.C.; Pimentel, I.C. Infection of Beauveria bassiana and Cordyceps javanica on different immature stages of Duponchelia fovealis Zeller (Lepidoptera: Crambidae). Crop. Prot. 2020, 138, 105347. [Google Scholar] [CrossRef]
- Potrich, M.; Alves, L.F.A.; Lozano, E.; Roman, J.C.; Pietrowski, V.; Neves, P.M.O.J. Interactions between Beauveria bassiana and Trichogramma pretiosum under laboratory conditions. Entomol. Exper. Appl. 2015, 154, 213–221. [Google Scholar] [CrossRef]
- Potrich, M.; Alves, L.F.A.; Lozano, E.R.; Bonini, A.K.; Neves, P.M.O.J. Potential side effects of the entomopathogenic fungus Metarhizium anisopliae on the egg parasitoid Trichogramma pretiosum (Hymenoptera: Trichogrammatidae) under controlled conditions. J. Econ. Entomol. 2017, 110, 2318–2324. [Google Scholar] [CrossRef]
- Campbell, B.E.; Pereira, R.M.; Koehler, P.G. Complications with controlling insect eggs. In Insecticides Resistance; Trdan, S., Ed.; IntechOpen: Rijeka, Croatia, 2016; pp. 83–96. [Google Scholar]
- Pirovani, V.D.; Pratissoli, D.; Tibúrcio, M.O.; de Carvalho, J.R.; Damascena, A.P.; Faria, L.V. Trichogramma galloi and Trichogramma pretiosum for the management of Duponchelia fovealis (Lepidoptera: Crambidae) in strawberry plants. Pesq. Agr. Bras. 2017, 52, 690–693. [Google Scholar] [CrossRef]
- Zimmermann, O. Use of Trichogramma wasps in Germany: Present status of research and commercial application of egg parasitoids against lepidopterous pests for crop. Gesunde Pflanz. 2004, 56, 157–166. [Google Scholar] [CrossRef]
- Potrich, M.; Alves, L.F.A.; Haas, J.; Da Silva, E.R.L.; Daros, A.; Pietrowski, V.; Neves, P.M.O.J. Seletividade de Beauveria bassiana e Metarhizium anisopliae a Trichogramma pretiosum Riley (Hymenoptera: Trichogrammatidae). Neotrop. Entomol. 2009, 38, 822–826. [Google Scholar] [CrossRef] [PubMed]
- Zawadneak, M.A.C.; Gonçalves, R.B.; Poltronieri, A.S.; Santos, B.; Bischoff, M.A.; Borba, M.A.; Pimentel, I.C. Biological parameters of Duponchelia fovealis (Lepidoptera: Crambidae) reared in the laboratory on two diets. Eur. J. Entomol. 2017, 114, 291–294. [Google Scholar] [CrossRef]
- Marchioro, C.A.; Foerster, L.A. Performance of the wheat armyworm Pseudaletia sequax Franclemont, on natural and artificial diets. Neotrop. Entomol. 2012, 41, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Ito, F.A.D.; Pimentel, I.C.; Poitevin, C.G. Niger seed agar as an inductor of sporulation of filamentous fungi with potential in biological control of agricultural diseases and pests. Int. J. Microbiol. Res. 2019, 11, 1509–1513. [Google Scholar]
- Alves, S.B.; Almeida, J.E.M.; Moino Júnior, A.; Alves, L.F.A. Controle microbiano de insetos. In Técnicas de Laboratorio; Alves, S.B., Ed.; FEALQ: Piracicaba, Brazil, 1998; pp. 637–712. [Google Scholar]
- Hassan, A.S. Métodos padronizados para testes de seletividade, com ênfase em Trichogramma. In Trichogramma e Controle Biológico Aplicado; Parra, J.R.P., Zucchi, R.A., Eds.; FEALQ: Piracicaba, Brazil, 1997; pp. 207–233. [Google Scholar]
- Shapiro, S.S.; Wilk, M.B. An analysis of variance test for normality. Biometrika 1965, 52, 591–611. [Google Scholar] [CrossRef]
- Bartlett, M.S. Properties of sufficiency and statistical tests. Proc. R. Stat. Soc. A 1937, 160, 268–282. [Google Scholar]
- Haddad, M.L.; Vendramin, J.D. Comparação de porcentagens observadas com casos extremos de 0 e 100%. Soc. Entomol. Bras. 2000, 29, 835–837. [Google Scholar] [CrossRef][Green Version]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: https://www.R-project.org/ (accessed on 21 January 2020).
- Brivio, M.F.; Mastore, M. When appearance misleads: The role of the entomopathogen surface in the relationship with its host. Insects 2020, 11, 387. [Google Scholar] [CrossRef]
- Vinson, S.B. Comportamento de seleção hospedeira de parasitoides de ovos, com ênfase na família Trichogrammatidae. In Trichogramma e Controle Biológico Aplicado; Parra, J.R.P., Zucchi, R.A., Eds.; FEALQ: Piracicaba, Brazil, 1997; pp. 67–119. [Google Scholar]
- Polanczyk, R.A.; Pratissoli, D.; Dalvi, L.P.; Grecco, E.D.; Franco, C.R. Efeito de Beauveria bassiana (Bals.) Vuillemin e Metarhizium anisopliae (Metsch.) Sorokin nos parâmetros biológicos de Trichogramma atopovirilia Oatman & Platner, 1983 (Hymenoptera: Trichogrammatidade). Cienc. Agrotec. 2010, 34, 1412–1416. [Google Scholar]
- Cônsoli, F.L.; Vinson, S.B. Parasitóides (Hymenoptera). Bioecologia e nutrição de insetos. In Base Para o Manejo Integrado de Pragas; Panizzi, A.R., Parra, J.R.P., Eds.; Embrapa Informacão Tecnológica: Brasília, Brazil, 2009; pp. 837–873. [Google Scholar]
- Desneux, N.; Decourtye, A.; Delpuech, J.M. The sublethal effects of pesticides on beneficial arthropods. Annu. Rev. Entomol. 2007, 52, 81–106. [Google Scholar] [CrossRef] [PubMed]
- Navarro, M.A. Trichogramma spp Producción, uso y Manejo en Colombia; Impretec: Guadalajara de Buga, Colombia, 1998; p. 176. [Google Scholar]
- Goulart, R.M.; Volpe, H.X.L.; Vacari, A.M.; Thuler, R.T.; De Bortoli, S.A. Insecticide selectivity to two species of Trichogramma in three different hosts, as determined by IOBC/WPRS methodology. Pest. Manag. Sci. 2012, 68, 240–244. [Google Scholar] [CrossRef]
- Dos Santos, V.P.; Pratissoli, D.; Paes, J.P.P.; Fragoso, D.F.M.; de Carvalho, J.R. Desempenho de Trichogramma pretiosum Riley, 1879 (Hymenoptera: Trichogrammatidae), submetido a inseticidas e fungicidas em dois hospedeiros. Rev. Ceres 2016, 63, 653–660. [Google Scholar] [CrossRef]
- Gallego, J.R.; Guerrero-Manzano, J.; Fernández-Maldonado, F.J.; Cabello, T. Susceptibility of the egg parasitoid Trichogramma achaeae (Hymenoptera: Trichogrammatidae) to selected insecticides used in tomato greenhouses. Span. J. Agric. Res. 2019, 17, e1009. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).