Trait Variation in Moths Mirrors Small-Scaled Ecological Gradients in A Tropical Forest Landscape
Simple Summary
Abstract
1. Introduction
2. Material and Methods
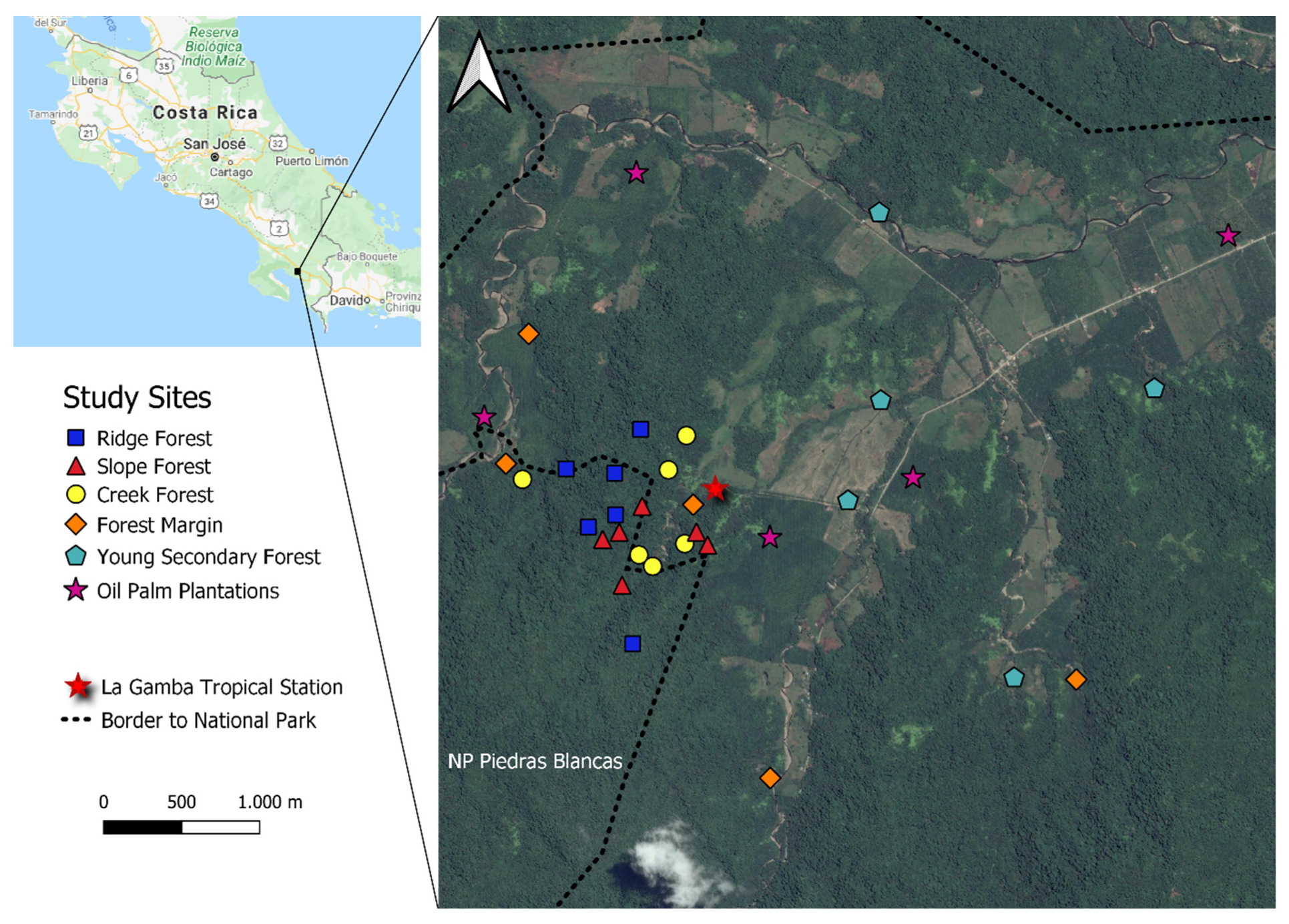
2.1. Study Sites
2.2. Moth Sampling and Processing
2.3. Data Collection and Statistical Analysis
3. Results
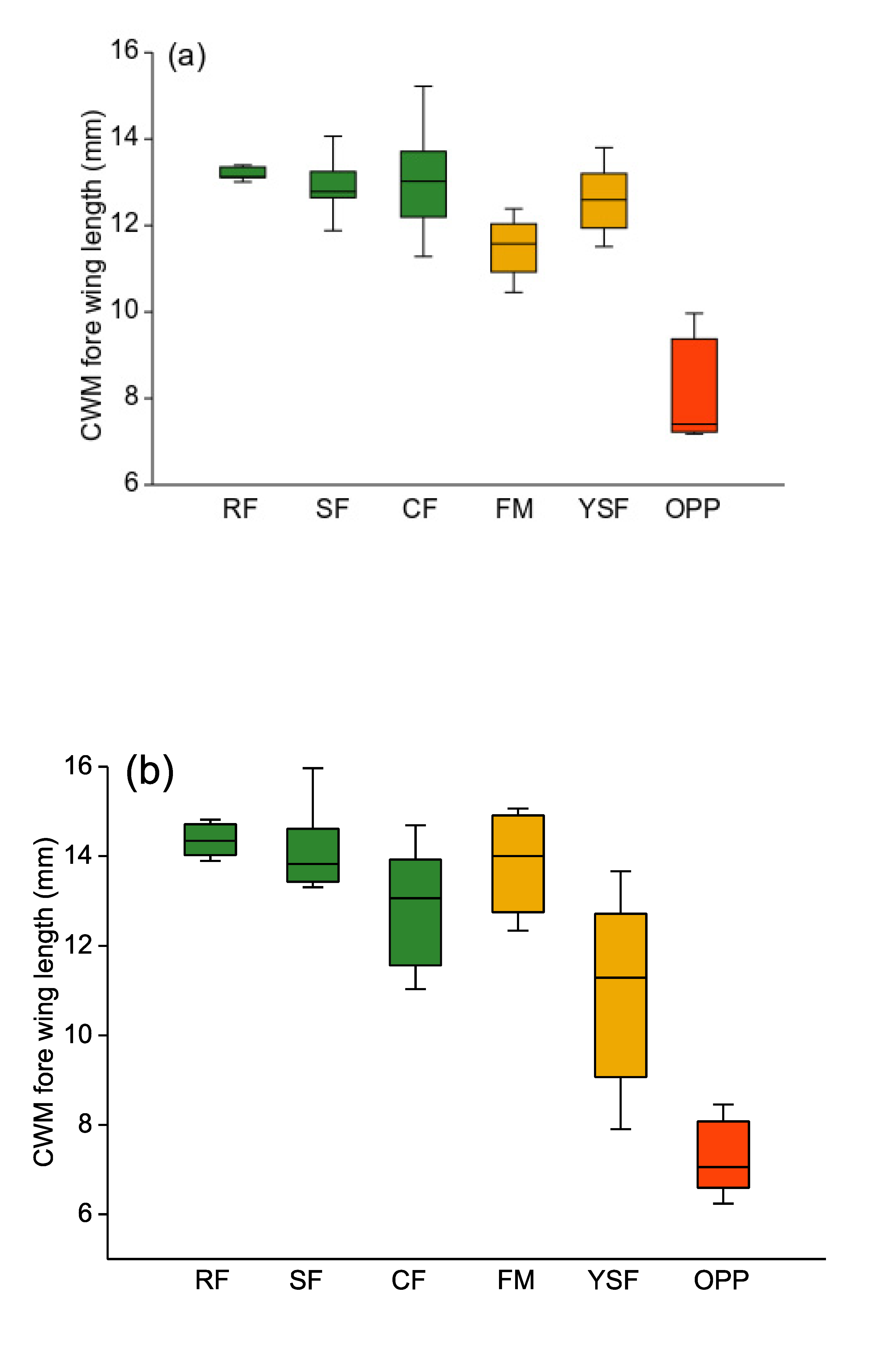
3.1. Body Size
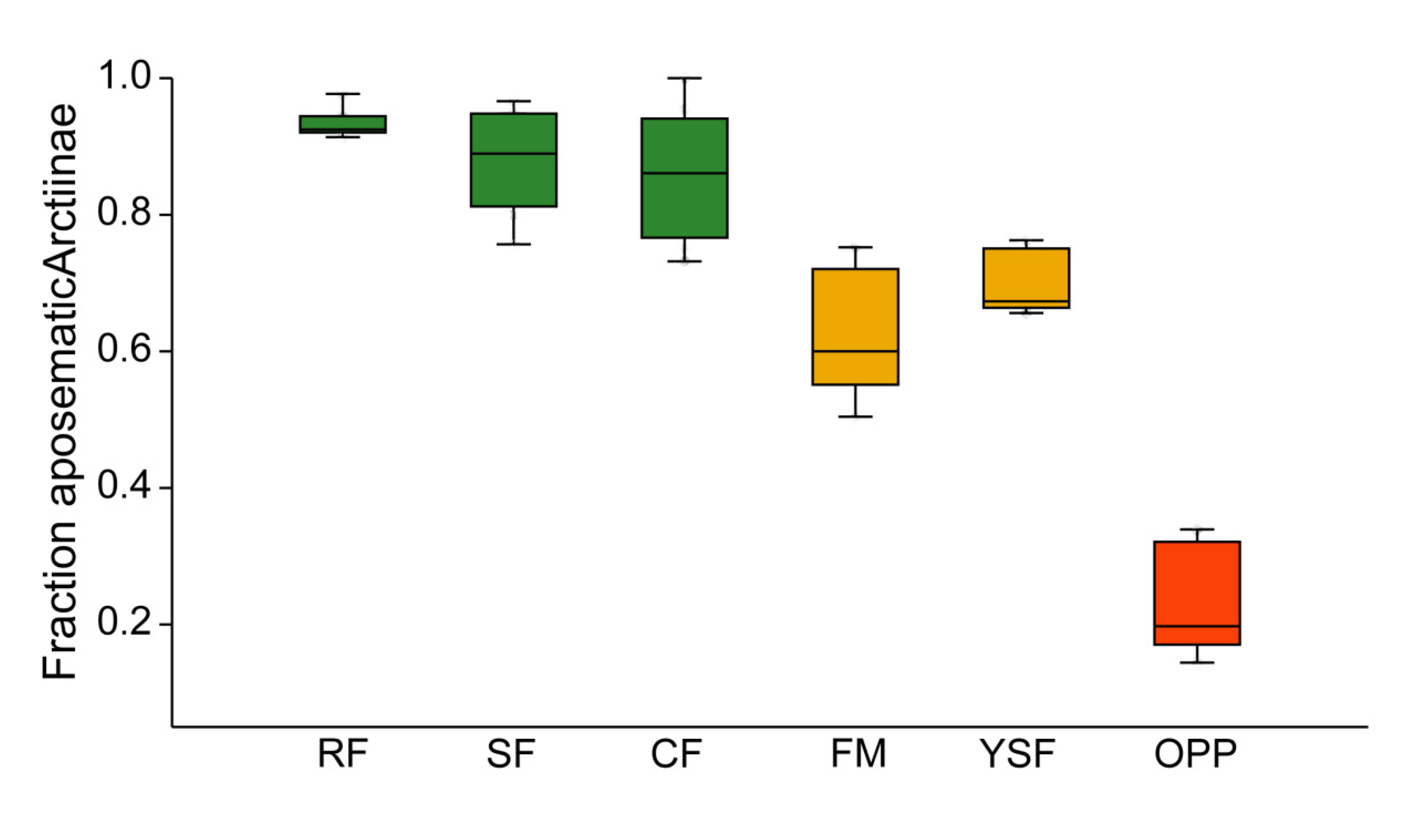
3.2. Incidence of Aposematism in Arctiinae
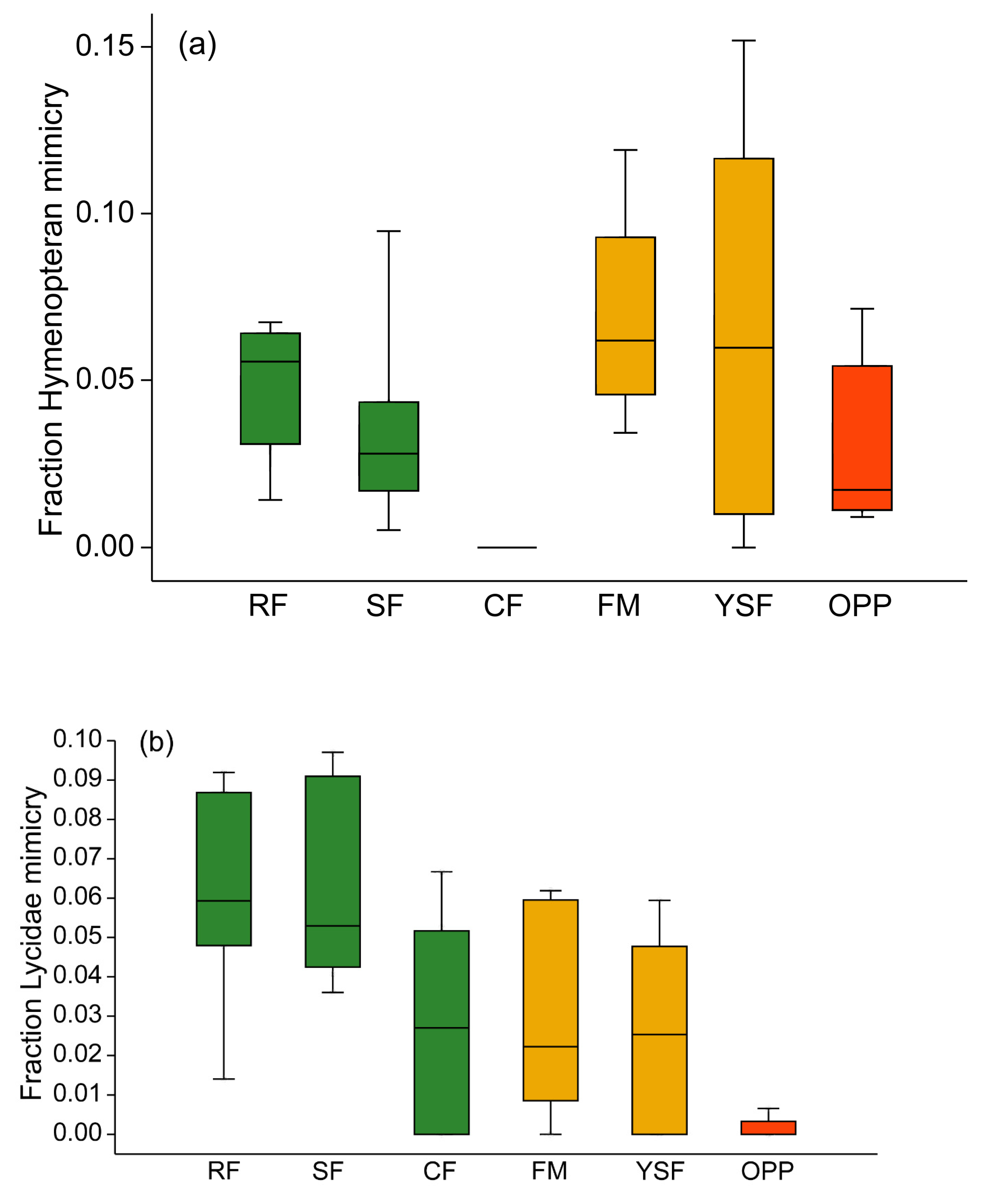
3.3. Incidence of Mimicry Among Arctiinae
4. Discussion
4.1. Body Size
4.2. Aposematism
4.3. Hymenopteran and Lycid Mimicry Rings
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kraft, N.J.; Adler, P.B.; Godoy, O.; James, E.C.; Fuller, S.; Levine, J.M. Community assembly, coexistence and the environmental filtering metaphor. Funct. Ecol. 2015, 29, 592–599. [Google Scholar] [CrossRef]
- Axmacher, J.C.; Holtmann, G.; Scheuermann, L.; Brehm, G.; Müller-Hohenstein, K.; Fiedler, K. Diversity of geometrid moths (Lepidoptera: Geometridae) along an Afrotropical elevational rainforest transect. Divers. Distrib. 2004, 10, 293–302. [Google Scholar] [CrossRef]
- Hilt, N.; Brehm, G.; Fiedler, K. Diversity and ensemble composition of geometrid moths along a successional gradient in the Ecuadorian Andes. J. Trop. Ecol. 2006, 22, 155–166. [Google Scholar] [CrossRef]
- Ashton, L.A.; Kitching, R.L.; Maunsell, S.; Bito, D.; Putland, D. Macrolepidopteran assemblages along an altitudinal gradient in subtropical rainforest—Exploring indicators of climate change. Mem. Queensl. Mus. 2011, 55, 375–389. Available online: https://www.qm.qld.gov.au/~/media/Documents/QM/About+Us/Publications/Memoirs+-+Nature/N55-2/n-55-2-ashton.pdf (accessed on 7 September 2020).
- Alonso-Rodríguez, A.M.; Finegan, B.; Fiedler, K. Neotropical moth assemblages degrade due to oil palm expansion. Biodiv. Conserv. 2017, 26, 2295–2326. [Google Scholar] [CrossRef]
- Rabl, D.; Gottsberger, B.; Brehm, G.; Hofhansl, F.; Fiedler, K. Moth assemblages in Costa Rica rain forest mirror small-scale topographic heterogeneity. Biotropica 2020, 52, 288–301. [Google Scholar] [CrossRef]
- Boggs, C.L. Understanding insect life histories and senescence through a resource allocation lens. Funct. Ecol. 2009, 23, 27–37. [Google Scholar] [CrossRef]
- Chown, S.L.; Gaston, K.J. Body size variation in insects: A macroecological perspective. Biol. Rev. 2010, 85, 139–169. [Google Scholar] [CrossRef]
- Brose, U.; Jonsson, T.; Berlow, E.L.; Warren, P.; Banašek-Richter, C.; Bersier, L.F.; Blanchard, J.L.; Brey, T.; Carpenter, S.R.; Cattin Blandenier, M.F.; et al. Consumer-resource body-size relationships in natural food webs. Ecology 2006, 87, 2411–2417. [Google Scholar] [CrossRef]
- Bersier, L.F.; Kehrli, P. The signature of phylogenetic constraints on food-web structure. Ecol. Complex. 2008, 5, 132–139. [Google Scholar] [CrossRef]
- Ruxton, G.D.; Allen, W.L.; Sherratt, T.N.; Speed, M.P. Avoiding Attack: The Evolutionary Ecology of Crypsis, Aposematism, and Mimicry; Oxford University Press: Oxford, UK, 2019. [Google Scholar]
- Gilbert, L.E.; Christen, C.A.; Altrichter, M.; Longino, J.T.; Sherman, P.M.; Plowes, R.; Swartz, M.B.; Winemiller, K.O.; Weghorst, J.A.; Vega, A.; et al. The southern pacific lowland evergreen moist forest of the Osa region. In Costa Rican Ecosystems; Kappelle, M., Ed.; University of Chicago Press: Chicago, IL, USA, 2016; pp. 360–411. [Google Scholar] [CrossRef]
- Huber, W.; Weissenhofer, A. The La Gamba research station in Costa Rica—History, nature, and research. Acta ZooBot Austria 2019, 156, 3–11. [Google Scholar]
- Weissenhofer, A.; Huber, W.; Mayer, V.; Pamperl, S.; Weber, A. Natural and Cultural History of the Golfo Dulce Region, Costa Rica; Biologiezentrum des Oberösterreichischen Landesmuseums: Linz, Austria, 2008. [Google Scholar]
- Höbinger, T.; Schindler, S.; Seaman, B.S.; Wrbka, T.; Weissenhofer, A. Impact of oil palm plantations on the structure of the agroforestry mosaic of La Gamba, southern Costa Rica: Potential implications for biodiversity. Agrofor. Syst. 2012, 85, 367–381. [Google Scholar] [CrossRef]
- Fournier, M.L.; Castillo, L.E.; Ramírez, F.; Moraga, G.; Ruepert, C. Evaluación preliminar de la influencia del área agrícola sobre la calidad del agua en el Golfo Dulce, Costa Rica. Rev. Cienc. Ambient. 2019, 53, 92–112. [Google Scholar] [CrossRef]
- Gallmetzer, N.; Schulze, C.H. Impact of oil palm agriculture on understory amphibians and reptiles: A Mesoamerican perspective. Glob. Ecol. Conserv. 2015, 4, 95–109. [Google Scholar] [CrossRef]
- Freudmann, A.; Mollik, P.; Tschapka, M.; Schulze, C.H. Impacts of oil palm agriculture on phyllostomid bat assemblages. Biodivers. Conserv. 2015, 24, 3583–3599. [Google Scholar] [CrossRef]
- Falk, M.; Schulze, C.H.; Fiedler, K. Ground-dwelling ant assemblages severely degrade in oil-palm plantations—A case study from the Golfo Dulce region, SW Costa Rica. Acta ZooBot Austria 2019, 156, 115–133. [Google Scholar]
- Morera-Beita, A.; Sánchez, D.; Wanek, W.; Hofhansl, F.; Huber, W.; Chacón-Madrigal, E.; Montero-Muñoz, J.L.; Silla, F. Beta diversity and oligarchic dominance in the tropical forests of southern Costa Rica. Biotropica 2019, 51, 117–128. [Google Scholar] [CrossRef]
- Hofhansl, F.; Chacón-Madrigal, E.; Fuchslueger, L.; Jenking, D.; Morera, A.; Plutzar, C.; Silla, F.; Andersen, K.M.; Buchs, D.; Dullinger, S.; et al. Climatic and edaphic controls over tropical forest diversity and vegetation carbon storage. Sci. Rep. 2020, 10, 5066. [Google Scholar] [CrossRef]
- Brehm, G.; Axmacher, J.C. A comparison of manual and automatic moth sampling methods (Lepidoptera: Arctiidae, Geometridae) in a rain forest in Costa Rica. Environ. Ent. 2006, 35, 757–764. [Google Scholar] [CrossRef]
- Truxa, C.; Fiedler, K. Attraction to light—From how far do moths (Lepidoptera) return to weak artificial sources of light? Eur. J. Ent. 2012, 109, 77–84. [Google Scholar] [CrossRef]
- McGeachie, W.J. The effects of moonlight illuminance, temperature and wind speed on light-trap catches of moths. Bull. Ent. Res. 1989, 79, 185–192. [Google Scholar] [CrossRef]
- Jaimes Niño, L.M.; Mörtter, R.; Brehm, G. Diversity and trait patterns of moths at the edge of an Amazonian rainforest. J. Insect Conserva. 2019, 23, 751–763. [Google Scholar] [CrossRef]
- Brehm, G.; Zeuss, D.; Colwell, R.K. Moth body size increases with elevation along a complete tropical elevational gradient for two hyperdiverse clades. Ecography 2019, 42, 632–642. [Google Scholar] [CrossRef]
- Eisner, T.; Schroeder, F.C.; Snyder, N.; Grant, J.B.; Aneshansley, D.J.; Utterback, D.; Meinwald, J.; Eisner, M. Defensive chemistry of lycid beetles and of mimetic cerambycid beetles that feed on them. Chemoecology 2008, 18, 109–119. [Google Scholar] [CrossRef]
- Do Nascimento, E.A. Estudos do mimetismo em Lycidae (Insecta: Coleoptera). Ph. D. Thesis, Universidade de São Paulo, São Paulo, Brasil, 2009. [Google Scholar]
- Lyytinen, A.; Alatalo, R.V.; Lindström, L.; Mappes, J. Are European white butterflies aposematic? Evol. Ecol. 1999, 13, 709–719. [Google Scholar] [CrossRef]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 1–9. Available online: https://folk.uio.no/ohammer/past/ (accessed on 7 September 2020).
- Wong, M.K.; Guénard, B.; Lewis, O.T. Trait-based ecology of terrestrial arthropods. Biol. Rev. 2019, 94, 999–1022. [Google Scholar] [CrossRef]
- Schulze, C.H.; Linsenmair, K.E.; Fiedler, K. Understorey versus canopy: Patterns of vertical stratification and diversity among Lepidoptera in a Bornean rain forest. Plant Ecol. 2001, 153, 133–152. [Google Scholar] [CrossRef]
- Brehm, G.; Fiedler, K. Bergmann’s rule does not apply to geometrid moths along an elevational gradient in an Andean montane rain forest. Glob. Ecol. Biogeogr. 2004, 13, 7–14. [Google Scholar] [CrossRef]
- De Andrade, R.B.; Barlow, J.; Louzada, J.; Vaz-de-Mello, F.Z.; Silveira, J.M.; Cochrane, M.A. Tropical forest fires and biodiversity: Dung beetle community and biomass responses in a northern Brazilian Amazon forest. J. Insect Conserv. 2014, 18, 1097–1104. [Google Scholar] [CrossRef]
- Strutzenberger, P.; Brehm, G.; Gottsberger, B.; Bodner, F.; Seifert, C.L.; Fiedler, K. Diversification rates, host plant shifts and an updated molecular phylogeny of Andean Eois moths (Lepidoptera: Geometridae). PLoS ONE 2017, 12, e0188430. [Google Scholar] [CrossRef]
- Dillon, M.E.; Frazier, M.R. Thermodynamics constrains allometric scaling of optimal development time in insects. PLoS ONE 2013, 8, e84308. [Google Scholar] [CrossRef]
- Darras, K.F.; Corre, M.D.; Formaglio, G.; Tjoa, A.; Potapov, A.; Brambach, F.; Kibrom, T.S.; Grass, I.; Rubiano, A.A.; Buchori, D.; et al. Reducing fertilizer and avoiding herbicides in oil palm plantations ‒ ecological and economic valuations. Front. For. Glob. Change 2019, 2, 65. [Google Scholar] [CrossRef]
- Lees, A.C.; Moura, N.G.; de Almeida, A.S.; Vieira, I.C. Poor prospects for avian biodiversity in Amazonian oil palm. PLoS ONE 2015, 10, e0122432. [Google Scholar] [CrossRef]
- Koh, L.P. Birds defend oil palms from herbivorous insects. Ecol. Appl. 2008, 18, 821–825. [Google Scholar] [CrossRef]
- Caras, T.; Korine, C. Effect of vegetation density on the use of trails by bats in a secondary tropical rain forest. J. Trop. Ecol. 2009, 25, 97–101. [Google Scholar] [CrossRef]
- Beck, J.; McCain, C.M. Just bird food? On the value of invertebrate macroecology. Front. Biogeogr. 2020, 12, e47684. [Google Scholar] [CrossRef]
- Conner, W.E.; Weller, S.J. A quest for alkaloids: The curious relationship between tiger moths and plants containing pyrrolizidine alkaloids. In Advances in Insect Chemical Ecology; Cardé, R.T., Millar, J.G., Eds.; Cambridge University Press: Cambridge, UK, 2004; pp. 248–282. [Google Scholar]
- Bowers, M.D. Chemical defenses in woolly bears: Sequestration and efficacy against predators and parasitoids. In Tiger Moths and Woolly Bears: Behavior, Ecology, and Evolution of the Arctiidae; Conner, W.E., Ed.; Oxford University Press: New York, NY, USA, 2009; pp. 83–102. [Google Scholar]
- Zaspel, J.M.; Weller, S.J.; Wardwell, C.T.; Zahiri, R.; Wahlberg, N. Phylogeny and evolution of pharmacophagy in tiger moths (Lepidoptera: Erebidae: Arctiinae). PLoS ONE 2014, 9, e101975. [Google Scholar] [CrossRef]
- Martins, C.H.; Cunha, B.P.; Solferini, V.N.; Trigo, J.R. Feeding on host plants with different concentrations and structures of pyrrolizidine alkaloids impacts the chemical-defense effectiveness of a specialist herbivore. PLoS ONE 2015, 10, e0141480. [Google Scholar] [CrossRef]
- Reudler, J.H.; Lindstedt, C.; Pakkanen, H.; Lehtinen, I.; Mappes, J. Costs and benefits of plant allelochemicals in herbivore diet in a multi enemy world. Oecologia 2015, 179, 1147–1158. [Google Scholar] [CrossRef]
- Hegna, R.H.; Galarza, J.A.; Mappes, J. Global phylogeography and geographical variation in warning coloration of the wood tiger moth (Parasemia plantaginis). J. Biogeogr. 2015, 42, 1469–1481. [Google Scholar] [CrossRef]
- Théry, M.; Gomez, D. Insect colours and visual appearance in the eyes of their predators. Adv. Insect Physiol. 2010, 38, 267–353. [Google Scholar] [CrossRef]
- Mappes, J.; Marples, N.; Endler, J.A. The complex business of survival by aposematism. Trends Ecol. Evol. 2005, 20, 598–603. [Google Scholar] [CrossRef]
- Lehmann, K.D.; Goldman, B.W.; Dworkin, I.; Bryson, D.M.; Wagner, A.P. From cues to signals: Evolution of interspecific communication via aposematism and mimicry in a predator-prey system. PLoS ONE 2014, 9, e91783. [Google Scholar] [CrossRef]
- Brehm, G. Contrasting patterns of vertical stratification in two moth families in a Costa Rican lowland rain forest. Basic Appl. Ecol. 2007, 8, 44–54. [Google Scholar] [CrossRef]
- Simmons, R.B. Adaptive coloration and mimicry. In Tiger Moths and Woolly Bears: Behavior, Ecology, and Evolution of the Arctiidae; Conner, W.E., Ed.; Oxford University Press: New York, NY, USA, 2009; pp. 115–126. [Google Scholar]
- Simmons, R.B.; Weller, S.J. What kind of signals do mimetic tiger moths send? A phylogenetic test of wasp mimicry systems (Lepidoptera: Arctiidae: Euchromiini). Proc. R. Soc. Lond. B Biol. Sci. 2002, 269, 983–990. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rabl, D.; Alonso-Rodríguez, A.M.; Brehm, G.; Fiedler, K. Trait Variation in Moths Mirrors Small-Scaled Ecological Gradients in A Tropical Forest Landscape. Insects 2020, 11, 612. https://doi.org/10.3390/insects11090612
Rabl D, Alonso-Rodríguez AM, Brehm G, Fiedler K. Trait Variation in Moths Mirrors Small-Scaled Ecological Gradients in A Tropical Forest Landscape. Insects. 2020; 11(9):612. https://doi.org/10.3390/insects11090612
Chicago/Turabian StyleRabl, Dominik, Aura M. Alonso-Rodríguez, Gunnar Brehm, and Konrad Fiedler. 2020. "Trait Variation in Moths Mirrors Small-Scaled Ecological Gradients in A Tropical Forest Landscape" Insects 11, no. 9: 612. https://doi.org/10.3390/insects11090612
APA StyleRabl, D., Alonso-Rodríguez, A. M., Brehm, G., & Fiedler, K. (2020). Trait Variation in Moths Mirrors Small-Scaled Ecological Gradients in A Tropical Forest Landscape. Insects, 11(9), 612. https://doi.org/10.3390/insects11090612




