Agricultural-Grade Apple Cider Vinegar Is Remarkably Attractive to Drosophila suzukii (Diptera: Drosophiliadae) in Mexico
Abstract
1. Introduction
2. Materials and Methods
2.1. Insect Colony
2.2. Evaluation of Apple-Cider-Vinegar-Based Attractants
2.3. Laboratory Cage Attraction of D. suzukii to Vinegars and Other Attractants
2.4. Attraction to Agricultural-Grade ACV in Polytunnel Raspberry Crops
2.5. Statistical Analysis
3. Results
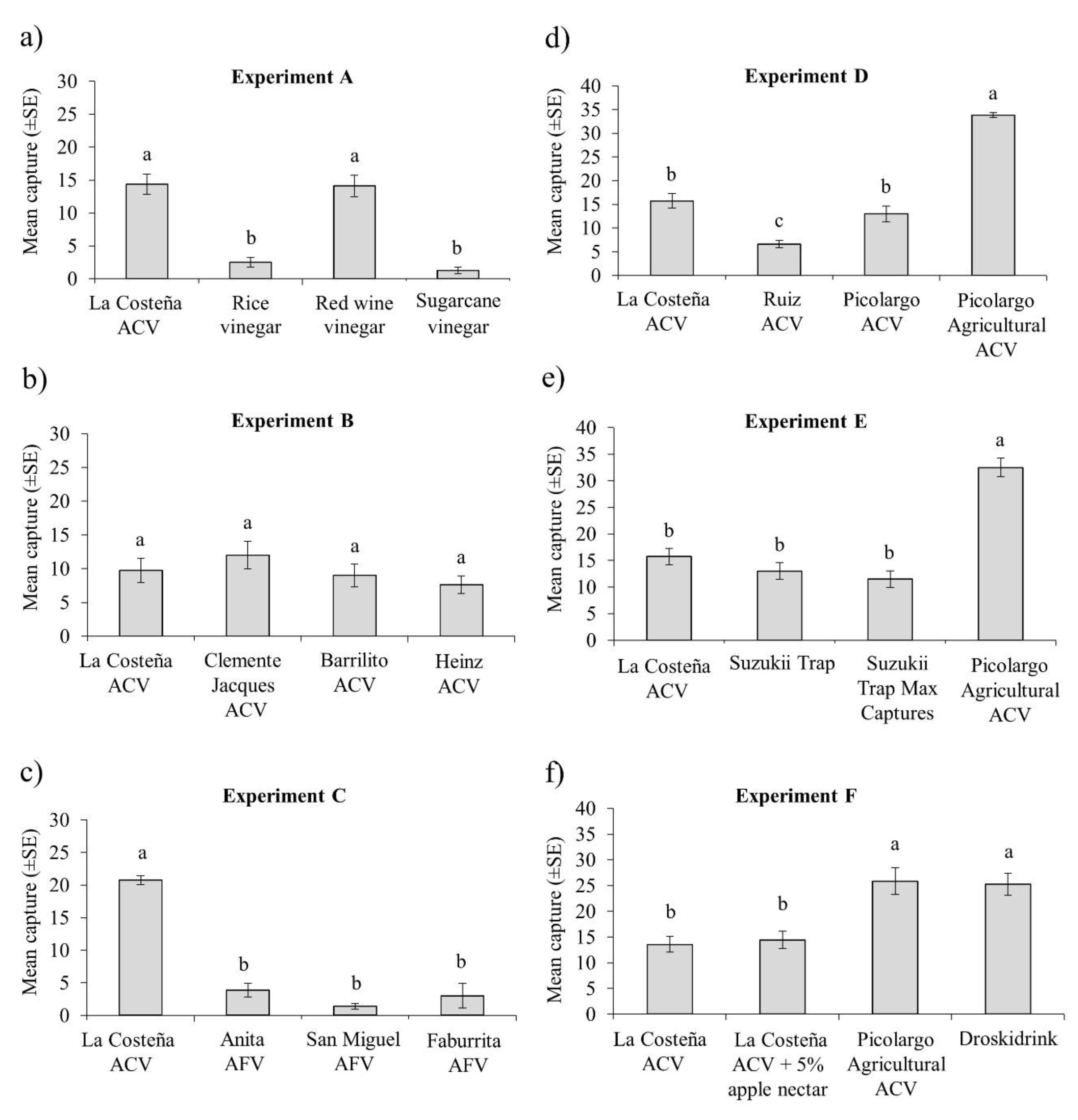
3.1. Evaluation of Apple Cider Vinegar-Based Attractants
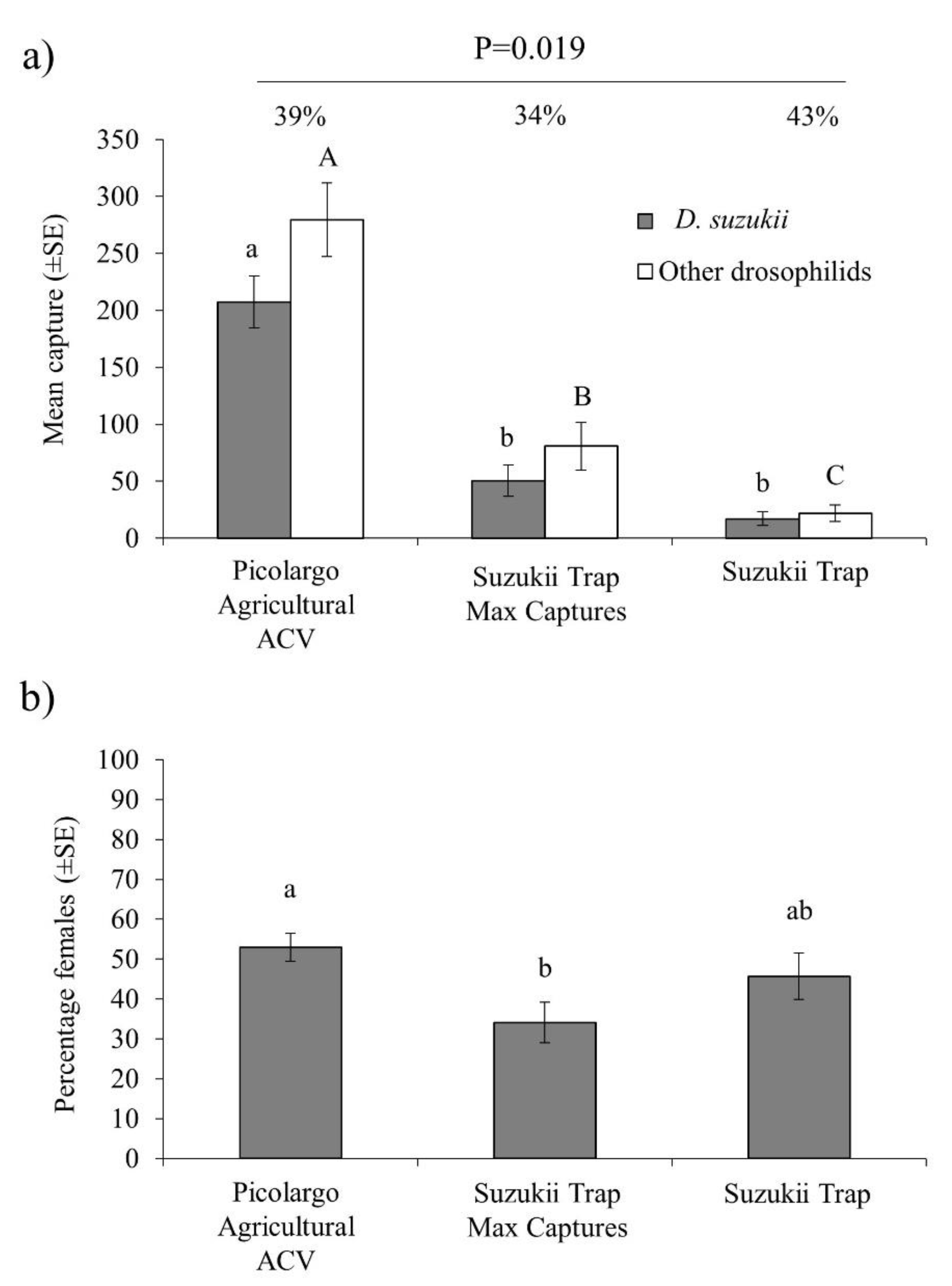
3.2. Laboratory Cage Attraction of D. suzukii to Vinegars and Other Attractants
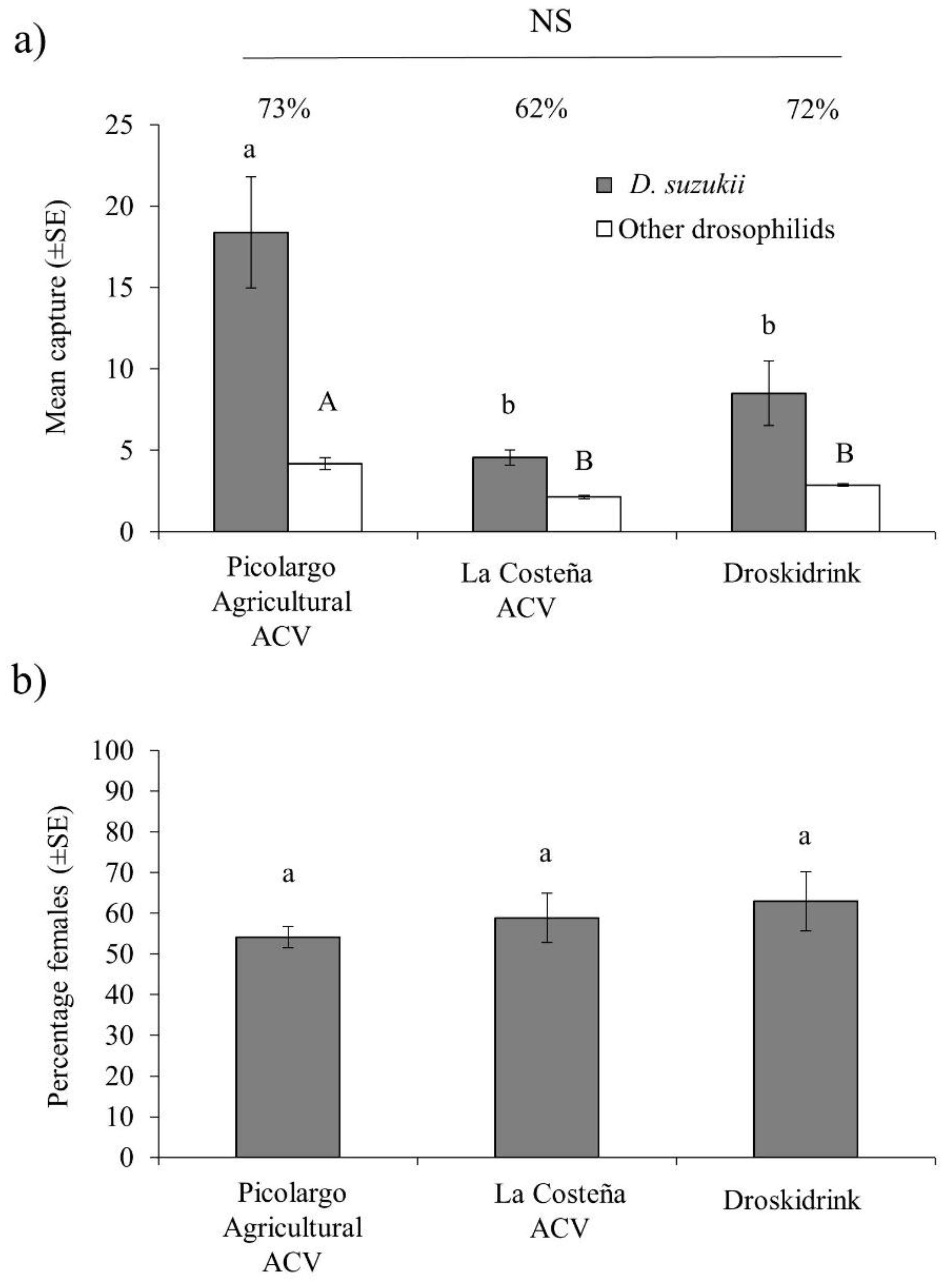
3.3. Attraction to Agricultural-Grade ACV in Polytunnel Raspberry Crops
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hauser, M. A historic account of the invasion of Drosophila suzukii (Matsumura) (Diptera: Drosophilidae) in the continental United States, with remarks on their identification. Pest Manag. Sci. 2011, 67, 1352–1357. [Google Scholar] [CrossRef]
- Asplen, M.K.; Anfora, G.; Biondi, A.; Choi, D.S.; Chu, D.; Daane, K.M.; Gibert, P.; Gutierrez, A.P.; Hoelmer, K.A.; Hutchison, W.D.; et al. Invasion biology of spotted wing drosophila (Drosophila suzukii): A global perspective and future priorities. J. Pest Sci. 2015, 88, 469–494. [Google Scholar] [CrossRef]
- SAGARPA. Secretaría de Agricultura Ganadería del Desarrollo Rural Pesca y Alimentación. Mosca del vinagre de alas manchadas (Drosophila suzukii Mastsumura). Dirección General de Sanidad Vegetal. In Sistema Nacional de Vigilancia Epidemiológica Fitosanitaria; Ficha Técnica 7; SAGARPA: Mexico City, Mexico, 2013; p. 22. Available online: https://prod.senasica.gob.mx/SIRVEF/ContenidoPublico/Fichas%20tecnicas/Ficha%20T%C3%A9cnica%20Mosca%20del%20vinagre%20de%20alas%20manchadas.pdf (accessed on 30 June 2020).
- Walsh, D.B.; Bolda, M.P.; Goodhue, R.E.; Dreves, A.J.; Lee, J.C.; Bruck, D.J.; Walton, V.M.S.; O’Neal, D.; Zalom, F.G. Drosophila suzukii (Diptera: Drosophilidae): Invasive pest of ripening soft fruit expanding its geographic range and damage potential. J. Integr. Pest Manag. 2011, 2, G1–G7. [Google Scholar] [CrossRef]
- Cini, A.; Ioriatti, C.; Anfora, G. A review of the invasion of Drosophila suzukii in Europe and a draft research agenda for integrated pest management. Bull. Insectol. 2012, 65, 149–160. [Google Scholar]
- Lee, J.C.; Dreves, A.; Cave, A.; Kawai, S.; Isaacs, R.; Miller, J.; Van Timmeren, S.; Bruck, D. Infestation of wild and ornamental non-crop fruits by Drosophila suzukii (Diptera: Drosophilidae). Ann. Entomol. Soc. Am. 2015, 108, 117–129. [Google Scholar] [CrossRef]
- Atallah, J.; Teixeira, L.; Salazar, R.; Zaragoza, G.; Kopp, A. The making of a pest: The evolution of a fruit-penetrating ovipositor in Drosophila suzukii and related species. Proc. R. Soc. B. 2014, 281, 20132840. [Google Scholar] [CrossRef]
- Abraham, J.; Zhang, A.; Angeli, S.; Abubeker, S.; Michel, C.; Feng, Y.; Rodriguez-Saona, C. Behavioral and antennal responses of Drosophila suzukii (Diptera: Drosophilidae) to volatiles from fruit extracts. Environ. Entomol. 2015, 44, 356–367. [Google Scholar] [CrossRef] [PubMed]
- CABI. Invasive Species Compendium: Detailed Coverage of Invasive Species Threatening Livelihoods and the Environment Worldwide. Drosophila suzukii (Spotted Wing Drosophila). CAB International: Wallingford, UK, 2020. Available online: https://www.cabi.org/isc/datasheet/109283 (accessed on 30 June 2020).
- Lee, J.C.; Burrack, H.J.; Barrantes, L.D.; Beers, E.H.; Dreves, A.J.; Hamby, K.A.; Haviland, D.R.; Isaacs, R.; Richardson, T.A.; Shearer, P.W.; et al. Evaluation of monitoring traps for Drosophila suzukii (Diptera: Drosophilidae) in North America. J. Econ. Entomol. 2012, 105, 1350–1357. [Google Scholar] [CrossRef]
- Basoalto, E.; Hilton, R.; Knight, A. Factors affecting the efficacy of a vinegar trap for Drosophila suzukii (Diptera; Drosophilidae). J. Appl. Entomol. 2013, 137, 561–570. [Google Scholar] [CrossRef]
- Lee, J.C.; Shearer, P.W.; Barrantes, L.D.; Beers, E.H.; Burrack, H.J.; Dalton, D.T.; Dreves, A.J.; Gut, L.J.; Hamby, K.A.; Haviland, D.L.; et al. Trap designs for monitoring Drosophila suzukii (Diptera: Drosophilidae). Environ. Entomol. 2013, 48, 2–8. [Google Scholar] [CrossRef]
- Burrack, H.J.; Asplen, M.; Bahder, L.; Collins, J.; Drummond, F.A.; Guédot, C.; Isaacs, R.; Johnson, D.; Blanton, A.; Lee, J.C.; et al. Multistate comparison of attractants for monitoring Drosophila suzukii (Diptera: Drosophilidae) in blueberries and caneberries. Environ. Entomol. 2015, 44, 704–712. [Google Scholar] [CrossRef] [PubMed]
- Ioriatti, C.; Walton, V.; Dalton, D.; Anfora, G.; Grassi, A.; Maistri, S.; Mazzoni, V. Drosophila suzukii (Diptera: Drosophilidae) and its potential impact to wine grapes during harvest in two cool climate wine grape production regions. J. Econ. Entomol. 2015, 108, 1148–1155. [Google Scholar] [CrossRef] [PubMed]
- Arnó, J.; Solà, M.; Riudavets, J.; Gabarra, R. Population dynamics, non-crop hosts, and fruit susceptibility of Drosophila suzukii in Northeast Spain. J. Pest Sci. 2016, 89, 713–723. [Google Scholar] [CrossRef]
- Briem, F.; Dominic, A.R.; Golla, B.; Hoffmann, C.; Englert, C.; Herz, A.; Vogt, H. Explorative data analysis of Drosophila suzukii trap catches from a seven-year monitoring program in Southwest Germany. Insects 2018, 9, 125. [Google Scholar] [CrossRef]
- Clymans, R.; Van Kerckvoorde, V.; Bangels, E.; Akkermans, W.; Alhmedi, A.; De Clercq, P.; Beliën, T.; Bylemans, D. Olfactory preference of Drosophila suzukii shifts between fruit and fermentations cues over the season: Effects of physiological status. Insects 2019, 10, 200. [Google Scholar] [CrossRef]
- Akasaka, N.; Higashikubo, H.; Ishii, Y.; Sakoda, H.; Fujiwara, S. Polyamines in brown rice vinegar function as potent attractants for the spotted wing drosophila. J. Biosci. Bioeng. 2017, 123, 78–83. [Google Scholar] [CrossRef]
- Willbrand, B.N.; Pfeiffer, D.G. Brown rice vinegar as an olfactory field attractant for Drosophila suzukii (Matsumura) and Zaprionus indianus Gupta (Diptera: Drosophilidae) in Cherimoya in Maui, Hawaii, with implications for attractant specificity between species and estimation of relative abundance. Insects 2019, 10, 80. [Google Scholar]
- Landolt, P.; Adams, T.; Rogg, H. Trapping spotted wing drosophila, Drosophila suzukii (Matsumura) (Diptera: Drosophilidae), with combinations of vinegar and wine, and acetic acid and ethanol. J. Appl. Entomol. 2012, 136, 148–154. [Google Scholar] [CrossRef]
- Iglesias, L.E.; Nyoike, T.W.; Liburd, O.E. Effect of trap design, bait type, and age on captures of Drosophila suzukii (Diptera: Drosophilidae) in berry crops. J. Econ. Entomol. 2014, 107, 1508–1518. [Google Scholar] [CrossRef]
- Frewin, A.J.; Renquema, J.; Fraser, H.; Hallett, H. Evaluation of attractants for monitoring Drosophila suzukii (Diptera: Drosophilidae). J. Econ. Entomol. 2017, 110, 1156–1163. [Google Scholar] [CrossRef]
- Landolt, P.; Adams, T.; Davis, S.; Rogg, H. Spotted wing drosophila, Drosophila suzukii (Diptera: Drosophilidae), trapped with combinations of wines and vinegars. Fla. Entomol. 2012, 95, 326–332. [Google Scholar] [CrossRef]
- Huang, J.; Gut, L.; Grieshop, M. Evaluation of food-based attractants for Drosophila suzukii (Diptera: Drosophilidae). Environ. Entomol. 2017, 46, 878–884. [Google Scholar] [CrossRef]
- Lasa, R.; Tadeo, E.; Toledo-Hernández, R.; Carmona, L.; Lima, I.; Williams, T. Improved capture of Drosophila suzukii by a trap baited with two attractants in the same device. PLoS ONE 2017, 12, e0188350. [Google Scholar] [CrossRef]
- Tonina, L.; Grassi, A.; Caruso, S.; Mori, N.; Gottardello, A.; Anfora, G.; Giomi, F.; Vaccari, G.; Ioriatti, C. Comparison of attractants for monitoring Drosophila suzukii in sweet cherry orchards in Italy. J. Appl. Entomol. 2018, 142, 18–25. [Google Scholar] [CrossRef]
- Cha, D.H.; Adams, T.; Rogg, H.; Landolt, P.J. Identification and field evaluation of fermentation volatiles from wine and vinegar that mediate attraction of spotted wing drosophila, Drosophila suzukii. J. Chem. Ecol. 2012, 38, 1419–1431. [Google Scholar] [CrossRef] [PubMed]
- Cha, D.H.; Adams, T.; Werle, C.T.; Sampson, B.J.; Adamczyk, J.J.; Rogg, H., Jr.; Landolt, P.J. A four-component synthetic attractant for Drosophila suzukii (Diptera: Drosophilidae) isolated from fermented bait headspace. Pest Manag. Sci. 2014, 70, 324–331. [Google Scholar] [CrossRef]
- Feng, Y.; Bruton, R.; Park, A.; Zhang, A. Identification of attractive blend for spotted wing drosophila, Drosophila suzukii, from apple juice. J. Pest Sci. 2018, 91, 1251–1267. [Google Scholar] [CrossRef]
- Renkema, J.M.; Wright, D.; Buitenhuis, R.; Hallett, R.H. Plant essential oils and potassium metabisulfite as repellents for Drosophila suzukii (Diptera: Drosophilidae). Sci. Rep. 2016, 6, 21432. [Google Scholar] [CrossRef]
- Kirkpatrick, D.M.; McGhee, P.S.; Gut, L.J.; Miller, J.R. Improving monitoring tools for spotted wing drosophila, Drosophila suzukii. Ent. Exp. Appl. 2017, 164, 87–93. [Google Scholar] [CrossRef]
- Cha, D.H.; Hesler, S.P.; Wallingford, A.K.; Zaman, F.; Jentsch, P.; Nyrop, J.; Loeb, G.M. Comparison of commercial lures and food baits for early detection of fruit infestation risk by Drosophila suzukii (Diptera: Drosophilidae). J. Econ. Entomol. 2018, 111, 645–652. [Google Scholar] [CrossRef]
- Jaffe, B.D.; Avanesyan, A.; Bal, H.K.; Feng, Y.; Grant, J.; Grieshop, M.J.; Lee, J.C.; Liburd, O.E.; Rhodes, E.; Rodriguez-Saona, C.; et al. Multistate comparison of attractants and the impact of fruit development stage on trapping Drosophila suzukii (Diptera: Drosophilidae) in raspberry and blueberry. Environ. Entomol. 2018, 47, 935–945. [Google Scholar] [CrossRef] [PubMed]
- SAGARPA. Secretaría de Agricultura Ganadería del Desarrollo Rural Pesca y Alimentación. Manual para el manejo fitosanitario de la mosca del vinagre de las alas manchadas (Drosophila suzukii Matsumura). Dirección General de Sanidad Vegetal. In Sistema Nacional de Vigilancia Epidemiológica Fitosanitaria; SAGARPA: Mexico City, Mexico, 2014; p. 18. [Google Scholar]
- Lasa, R.; Toledo-Hernández, R.A.; Rodríguez, D.; Williams, T. Raspberry as a source for the development of Drosophila suzukii attractants: Laboratory and commercial polytunnel trials. Insects 2019, 10, 137. [Google Scholar] [CrossRef] [PubMed]
- Hamby, K.A.; Becher, P.G. Current knowledge of interactions between Drosophila suzukii and microbes, and their potential utility for pest management. J. Pest Sci. 2016, 89, 621–630. [Google Scholar] [CrossRef]
- Lasa, R.; Navarro-de-la-Fuente, L.; Gschaedler-Mathis, A.C.; Kirchmayr, M.R.; Williams, T. Yeast species, strains, and growth media mediate attraction of Drosophila suzukii (Diptera: Drosophilidae). Insects 2019, 10, 228. [Google Scholar] [CrossRef] [PubMed]
- Grassi, A.; Anfora, G.; Maistri, S.; Gottardello, A.; Maddalena, G.; De Cristofaro, A.; Savini, G.; Ioriatti, C. Development and efficacy of Droskidrink, a food bait for trapping Drosophila suzukii. IOBC WPRS Bull. 2014, 109, 197–204. [Google Scholar]
- Khan, M.S.; Ullah, F.; Badshah, H.; Ahmad, B.; Shahjehan, I.A.; Clatayud, P.A. Bait attractants based on artificial fruit-essence for trapping and monitoring Drosophila suzukii (Diptera: Drosophilidae) females in Peshawar-Pakistan. Phytoparasitica 2019, 47, 179–184. [Google Scholar] [CrossRef]
- Dalton, D.; Walton, V.; Shearer, P.; Walsh, D.; Caprile, J.; Isaacs, R. Laboratory survival of Drosophila suzukii under simulated winter conditions of the Pacific Northwest and seasonal field trapping in five primary regions of small and stone fruit production in the United States. Pest Manag. Sci. 2011, 67, 1368–1374. [Google Scholar] [CrossRef]
- Jamovi Statistical Software, Version 1.0.7.0. Available online: https://www.jamovi.org (accessed on 14 April 2020).
- Hamby, K.A.; Bolda, M.P.; Sheehan, M.E.; Zalom, F.G. Seasonal monitoring for Drosophila suzukii (Diptera: Drosophilidae) in California commercial raspberries. Environ. Entomol. 2014, 43, 1008–1018. [Google Scholar] [CrossRef]
- Joshi, V.K.; Sharma, S. Cider vinegar: Microbiology, technology and quality. In Vinegars of the World; Solieri, L., Guidici, P., Eds.; Springer: Milan, Italy, 2009; pp. 197–207. [Google Scholar]
- Mazzetto, F.; Gonella, E.; Crotti, E.; Vacchini, V.; Syrpas, M.; Pontini, M.; Mangelinckx, S.; Daffonchio, D.; Alma, A. Olfactory attraction of Drosophila suzukii by symbiotic acetic acid bacteria. J. Pest Sci. 2016, 89, 783–792. [Google Scholar] [CrossRef]
| Experiment | Treatments | Mean Capture ± SE | Sig. | Statistical Values |
|---|---|---|---|---|
| Exp. 1 | ACV | 19.4 ± 2.0 | NS | t = 0.357, df = 7, p = 0.731 |
| Suzukii Trap | 18.1 ± 1.8 | |||
| Exp. 2 | ACV | 17.8 ± 3.3 | NS | t = 0.326, df = 7, p = 0.754 |
| ACV 80% | 15.9 ± 2.7 | |||
| ACV | 18.9 ± 2.1 | NS | t = 1.48, df = 7, p = 0.181 | |
| ACV 60% | 12.8 ± 2.4 | |||
| ACV | 26.3 ± 1.0 | *** | t = 5.57, df = 7, p < 0.001 | |
| ACV 40% | 10.3 ± 2.0 | |||
| Exp. 3 | ACV | 24.9 ± 2.5 | *** | t = 7.72, df = 7, p < 0.001 |
| ACV + 5% acetic acid | 9.0 ± 2.0 | |||
| ACV | 21.6 ± 1.8 | *** | t = 8.75, df = 7, p < 0.001 | |
| ACV + 2.5% propionic acid | 3.4 ± 0.5 | |||
| Exp. 4 | ACV | 15.6 ± 2.3 | NS | t = 0.683, df = 7, p = 0.516 |
| ACV + 8% Captor | 17.9 ± 1.7 | |||
| ACV | 17.3 ± 1.8 | NS | t = 0.710, df = 7, p = 0.501 | |
| ACV + 8% Winner | 14.8 ± 2.2 | |||
| ACV | 20.6 ± 2.3 | NS | t = 2.06, df = 7, p = 0.079 | |
| ACV + 8% Flyral | 13.6 ± 2.7 | |||
| ACV | 15.1 ± 1.2 | NS | t = 0.848, df = 7, p = 0.425 | |
| ACV + 8% CeraTrap | 13.5 ± 1.2 | |||
| Exp. 5 | ACV | 15.8 ± 1.8 | NS | t = 0.738, df = 7, p = 0.485 |
| ACV + 0.1% raspberry flavor | 15.9 ± 1.9 | |||
| ACV | 17.6 ± 2.7 | NS | t = 0.504, df = 7, p = 0.630 | |
| ACV + 0.1% blackberry flavor | 12.5 ± 1.6 | |||
| ACV | 13.8 ± 2.2 | NS | t = 0.895, df = 7, p = 0.400 | |
| ACV + 0.1% strawberry flavor | 17.3 ± 2.3 | |||
| ACV | 16.6 ± 1.2 | NS | t = 1.24, df = 7, p = 0.225 | |
| ACV + 0.1% apple flavor | 14.9 ± 1.6 | |||
| Exp. 6 | ACV | 19.1 ± 2.8 | NS | t = 1.01, df = 7, p = 0.344 |
| ACV + 5% grape nectar | 15.1 ± 1.5 | |||
| ACV | 13.5 ± 1.4 | NS | t = 0.654, df = 7, p = 0.606 | |
| ACV + 5% pineapple nectar | 12.3 ± 1.2 | |||
| ACV | 11.1 ± 1.0 | *** | t = 5.165, df = 7, p = 0.001 | |
| ACV + 5% apple nectar | 21.8 ± 1.4 | |||
| ACV | 14.9 ± 1.3 | NS | t = 1.50, df = 7, p = 0.178 | |
| ACV + 15% apple nectar | 20.1 ± 2.3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lasa, R.; Aguas-Lanzagorta, S.; Williams, T. Agricultural-Grade Apple Cider Vinegar Is Remarkably Attractive to Drosophila suzukii (Diptera: Drosophiliadae) in Mexico. Insects 2020, 11, 448. https://doi.org/10.3390/insects11070448
Lasa R, Aguas-Lanzagorta S, Williams T. Agricultural-Grade Apple Cider Vinegar Is Remarkably Attractive to Drosophila suzukii (Diptera: Drosophiliadae) in Mexico. Insects. 2020; 11(7):448. https://doi.org/10.3390/insects11070448
Chicago/Turabian StyleLasa, Rodrigo, Saide Aguas-Lanzagorta, and Trevor Williams. 2020. "Agricultural-Grade Apple Cider Vinegar Is Remarkably Attractive to Drosophila suzukii (Diptera: Drosophiliadae) in Mexico" Insects 11, no. 7: 448. https://doi.org/10.3390/insects11070448
APA StyleLasa, R., Aguas-Lanzagorta, S., & Williams, T. (2020). Agricultural-Grade Apple Cider Vinegar Is Remarkably Attractive to Drosophila suzukii (Diptera: Drosophiliadae) in Mexico. Insects, 11(7), 448. https://doi.org/10.3390/insects11070448





