Cold Storage Effects on Fitness of the Whitefly Parasitoids Encarsia sophia and Eretmocerus hayati
Abstract
1. Introduction
2. Materials and Methods
2.1. Insects
2.2. Temperature Treatments
2.3. Parasitoid Fitness after Storage
2.3.1. Emergence Rate and Time
2.3.2. Longevity
2.3.3. Adult Size
2.3.4. Parasitism
2.4. Statistical Analysis
3. Results
3.1. Emergence Rate
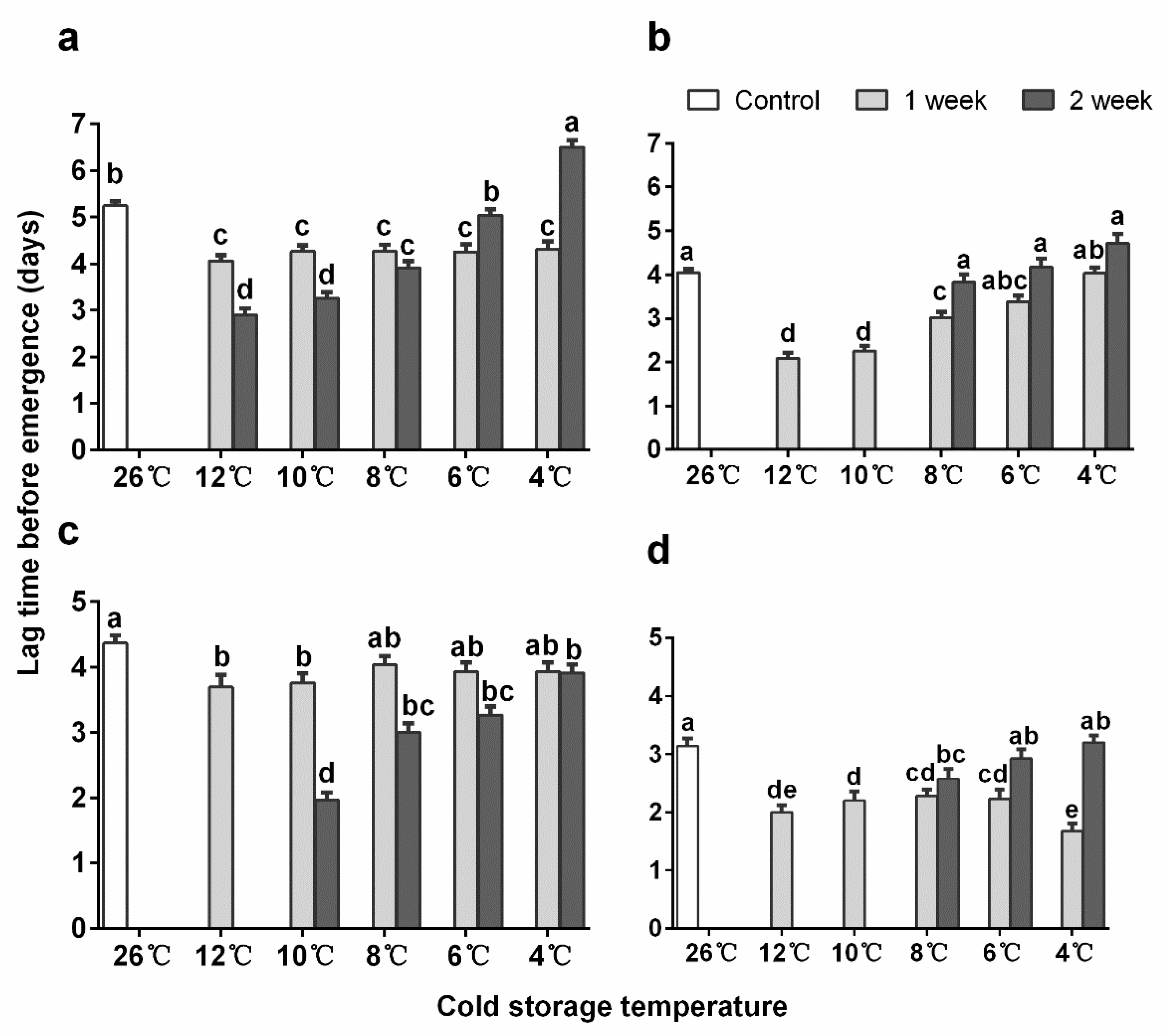
3.2. Emergence Time
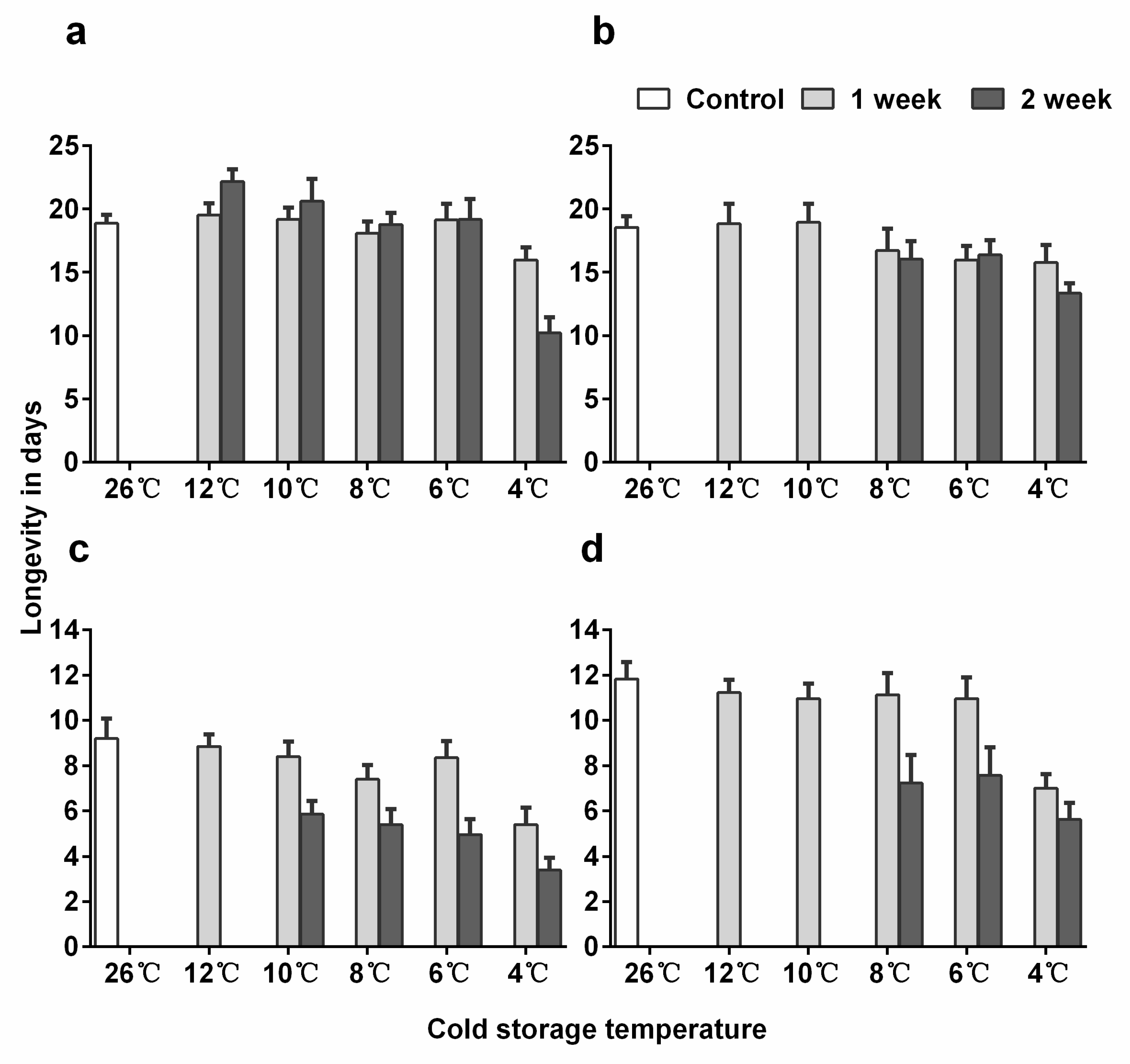
3.3. Longevity
3.4. Adult Body Size
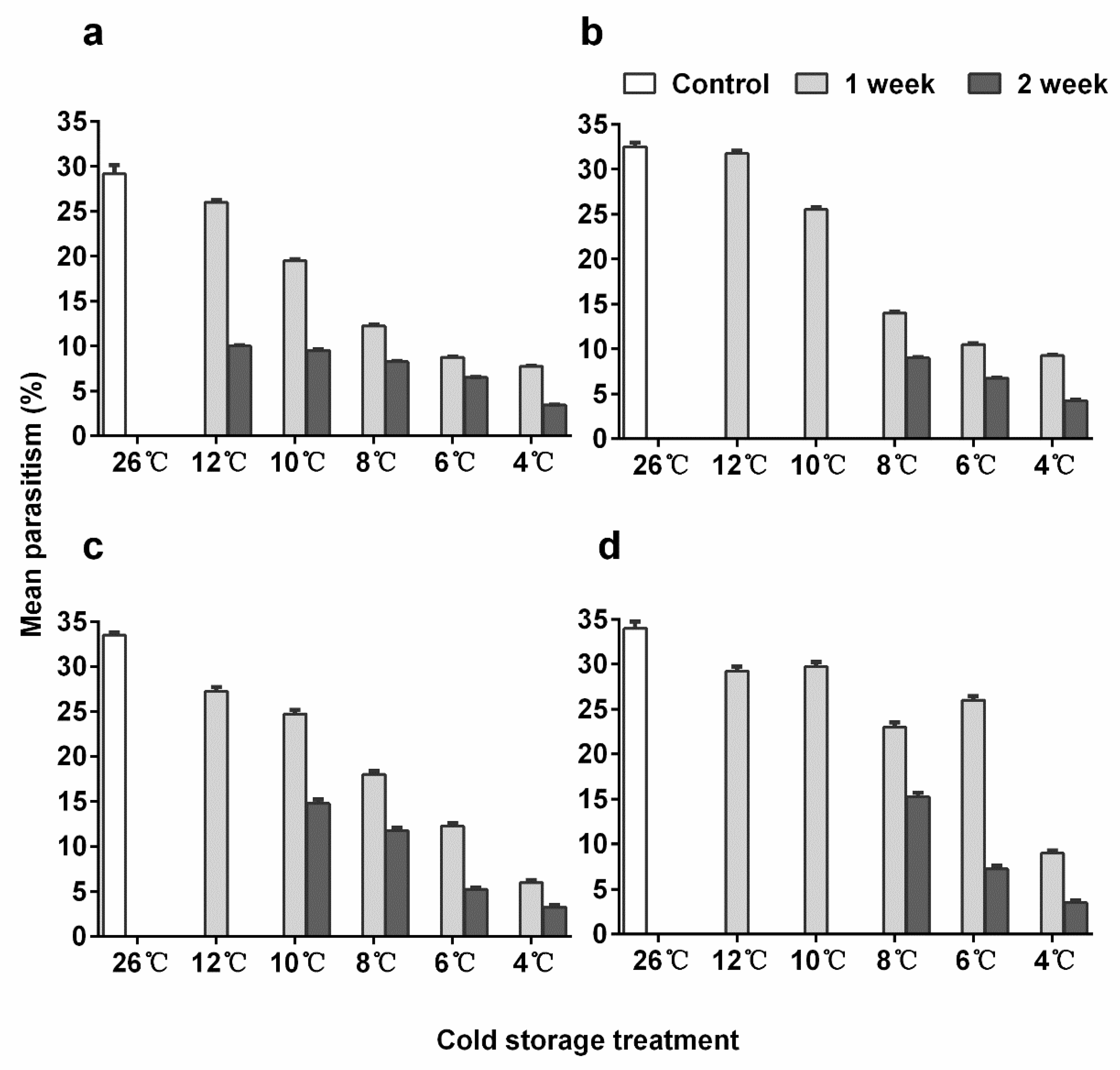
3.5. Parasitization Ability
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Colinet, H.; Boivin, G. Insect parasitoids cold storage: A comprehensive review of factors of variability and consequences. Biol. Control 2011, 58, 83–95. [Google Scholar] [CrossRef]
- Gardner, J.; Hoffmann, M.P.; Pitcher, S.A.; Nyrop, J.P. Recurrent warming to improve cold storage of Trichogrammatids (Hymenoptera: Trichogrammatidae). Biocontrol Sci. Technol. 2012, 22, 261–270. [Google Scholar] [CrossRef]
- Spinola-Filho, P.R.C.; Leite, G.L.D.; Soares, M.A.; Alvarenga, A.C.; Paulo, P.D.; Tuffi-Santos, L.D.; Zanuncio, J.C. Effects of duration of cold storage of host eggs on perecent parasitism and adult emergence of each ten Trichogrammatidae (Hymenoptera) species. Fla. Entomol. 2014, 97, 14–21. [Google Scholar] [CrossRef]
- Tezze, A.A.; Botto, E.N. Effect of cold storage on the quality of Trichogramma nerudai (Hymenoptera: Trichogrammatidae). Biol. Control 2004, 30, 11–16. [Google Scholar] [CrossRef]
- Chen, W.L.; Leopold, R.A.; Boetel, M.A. Cold storage of adult Gonatocerus ashmeadi (Hymenoptera: Mymaridae) and effects on maternal and progeny fitness. J. Econ. Entomol. 2008, 101, 1760–1770. [Google Scholar] [CrossRef]
- Kostal, V.; Korbelova, J.; Stetina, T.; Poupardin, R.; Colinet, H.; Zahradnickova, H.; Opekarova, I.; Moos, M.; Simek, P. Physiological basis for low-temperature survival and storage of quiescent larvae of the fruit fly Drosophila melanogaster. Sci. Rep. 2016, 6, 32346. [Google Scholar] [CrossRef] [PubMed]
- van Lenteren, J.; Tommasini, M. Mass production, storage, shipment and quality control of natural enemies. In Mass Production, Storage, Shipment and Quality Control of Natural Enemies, Integrated Pest and Disease Management in Greenhouse Crops; Albajes, R., Gullino, M.L., van Lenteren, J.C., Elad, Y., Eds.; Springer: Dordrecht, The Netherlands, 2012; pp. 276–294. [Google Scholar]
- Yan, Z.; Yue, J.J.; Bai, C.; Peng, Z.Q.; Zhang, C.H. Effects of cold storage on the biological characteristics of Microplitis prodeniae (Hymenoptera: Braconidae). Bull. Entomol. Res. 2017, 107, 506–512. [Google Scholar] [CrossRef]
- Colinet, H.; Renault, D.; Hance, T.; Vernon, P. The impact of fluctuating thermal regimes on the survival of a cold-exposed parasitic wasp, Aphidius colemani. Physiol. Entomol. 2006, 31, 234–240. [Google Scholar] [CrossRef]
- Colinet, H.; Boivin, G.; Hance, T. Manipulation of parasitoid size using the temperature-size rule: Fitness consequences. Oecologia 2007, 152, 425–433. [Google Scholar] [CrossRef]
- Colinet, H.; Sinclair, B.J.; Vernon, P.; Renault, D. Insects in fluctuating thermal environments. Annu. Rev. Entomol. 2015, 60, 123–140. [Google Scholar] [CrossRef]
- Yocum, G.D.; Rinehart, J.P.; Kemp, W.P. Duration and frequency of a high temperature pulse affect survival of emergence-ready Megachile rotundata (Hymenoptera: Megachilidae) during low-temperature incubation. J. Econ. Entomol. 2012, 105, 14–19. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Leopold, R.A.; Rojas, R.R.; Atkinson, P. Post pupariation cold storage of three species of flies: Increasing chilling tolerance by acclimation and recurrent recovery periods. Cryobiology 1998, 36, 213–224. [Google Scholar] [CrossRef] [PubMed]
- Renault, D.; Nedved, O.; Hervant, F.; Vernon, P. The importance of fluctuating thermal regimes for repairing chill injuries in the tropical beetle Alphitobius diaperinus (Coleoptera: Tenebrionidae) during exposure to low temperature. Physiol. Entomol. 2004, 29, 139–145. [Google Scholar] [CrossRef]
- Ismail, M.; Vanbaaren, J.; Hance, T.; Pierre, J.S.; Vernon, P. Stress intensity and fitness in the parasitoid Aphidius ervi (Hymenoptera: Braconidae): Temperature below the development threshold combined with a fluctuating thermal regime is a must. Ecol. Entomol. 2013, 38, 355–363. [Google Scholar] [CrossRef]
- Hance, T.; van Baaren, J.; Vernon, P.; Boivin, G. Impact of extreme temperatures on parasitoids in a climate change perspective. Annu. Rev. Entomol. 2007, 52, 107–126. [Google Scholar] [CrossRef] [PubMed]
- Lee, R.E. A primer on insect cold-tolerance. In Low Temperature Biology of Insects; Denlinger, D.L., Lee, R.E., Eds.; Cambridge University Press: Cambridge, UK, 2010; pp. 3–35. [Google Scholar]
- Ismail, M.; van Baaren, J.; Briand, V.; Pierre, J.S.; Vernon, P.; Hance, P. Fitness consequences of low temperature storage of Aphidius ervi. BioControl 2014, 59, 139–148. [Google Scholar] [CrossRef][Green Version]
- van Baaren, J.; Outreman, Y.; Boivin, G. Effect of low temperature exposure on host oviposition behaviour and patch exploitation strategy in an egg parasitoid. Anim. Behav. 2005, 70, 153–163. [Google Scholar] [CrossRef]
- van Baaren, J.; Boivin, G.; Outreman, Y. Deleterious effects of low temperature exposure on learning expression in a parasitoid. J. Comp. Psychol. 2006, 19, 368–385. [Google Scholar]
- Moiroux, J.; Brodeur, J.; Boivin, G. Sex ratio variations with temperature in an egg parasitoid: Behavioural adjustment and physiological constraint. Anim. Behav. 2014, 91, 61–66. [Google Scholar] [CrossRef]
- Kidane, D.; Yang, N.W.; Wan, F.H. Effect of cold storage on the biological fitness of Encarsia sophia (Hymenoptera: Aphelinidae), a parasitoid of Bemisia tabaci (Hemiptera: Aleyrodidae). Eur. J. Entomol. 2015, 112, 460–469. [Google Scholar] [CrossRef]
- Jalali, S.K.; Singh, S.P. Biology and feeding potential of Curinus coeruleus (Mulsant) and Chrysoperla carnea (Stephens) on subabul psyllid, Heteropsylla cubana Crawford. J. Insect. Sci. 1992, 5, 89–90. [Google Scholar]
- Levie, A.; Vernon, P.; Hance, T. Consequences of acclimation on survival and reproductive capacities of cold-stored Aphidius rhopalosiphi (Hymenoptera: Aphidiinae). J. Econ. Entomol. 2005, 98, 704–708. [Google Scholar] [CrossRef] [PubMed]
- Kostal, V.; Renault, D.; Mehrabianova, A.; Bastl, J. Insect cold tolerance and repair of chill-injury at fluctuating thermal regimes: Role of ion homeostasis. Comp. Biochem. Physiol. A 2007, 147, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Daane, K.M.; Wang, X.G.; Johnson, M.W.; Cooper, M.L. Low temperature storage effects on two olive fruit fly parasitoids. BioControl 2013, 58, 175–185. [Google Scholar] [CrossRef]
- Bowler, K.; Terblanche, J.S. Insect thermal tolerance: What is the role of ontogeny, ageing and senescence? Biol. Rev. 2008, 83, 339–355. [Google Scholar] [CrossRef]
- Lu, B.; Tang, Z.; Glenn, A.; Bellis, G.A.; Mark, G.; Wright, M.G.; Jin, Q.; Peng, Z.; Wen, H.; Wan, J.; et al. Biological and physiological response of a tropical parasitoid, Tetrastichus brontispae (Ferriere) following exposure to low temperature. BioControl 2016, 61, 649–659. [Google Scholar] [CrossRef]
- Mahi, H.; Rasekh, A.; Michaud, J.P.; Shishehbor, P. Biology of Lysiphlebus fabarum following cold storage of larvae and pupae. Entomol. Exp. Appl. 2015, 153, 10–19. [Google Scholar] [CrossRef]
- Foerster, L.A.; Doetzer, A.K.; Castro, L.C.F. Emergence, longevity and fecundity of Trissolcus basalis and Telenomus podisi after cold storage in the pupal stage. Pesqui. Agropecu. Bras. 2004, 39, 9. [Google Scholar] [CrossRef]
- Luczynski, A.; Nyrop, J.P.; Shi, A. Influence of cold storage on pupal development and mortality during storage and on post-storage performance of Encarsia formosa and Eretmocerus eremicus (Hymenoptera: Aphelinidae). Biol. Control 2007, 40, 107–117. [Google Scholar] [CrossRef]
- Zang, L.S.; Liu, T.X. Host feeding of three whitefly parasitoid species on Bemisia tabaci B biotype, with implication for whitefly biological control. Entomol. Exp. Appl. 2008, 127, 55–63. [Google Scholar] [CrossRef]
- De Barro, P.J.; Coombs, M.T. Post-release evaluation of Eretmocerus hayati Zolnerowich and Rose in Australia. Bull. Entomol. Res. 2009, 99, 193–206. [Google Scholar] [CrossRef]
- Yang, N.W.; Wan, F.H. Host suitability of different instars of Bemisia tabaci biotype B for the parasitoid Eretmocerus hayati. Biol. Control 2011, 59, 313–317. [Google Scholar] [CrossRef]
- Wan, F.H.; Yang, N.W. Invasion and Management of Agricultural Alien Insects in China. Annu. Rev. Entomol. 2016, 61, 77–98. [Google Scholar] [CrossRef]
- Hoelmer, K.A.; Roltsch, W.J. Evaluation of exotic parasitoids and predators in field cages in California. In Classical Biological Control of Bemisia tabaci in the United States: A Review of Interagency Research and Implementation; Gould, J., Hoelmer, K., Goolsby, J., Eds.; Springer: Dordrecht, The Netherlands, 2008; pp. 129–145. [Google Scholar]
- Yang, N.W.; Ji, L.L.; Lovei, G.L.; Wan, F.H. Shifting preference between oviposition vs. host-feeding under changing host densities in two aphelinid parasitoids. PLoS ONE 2012, 7, e41189. [Google Scholar] [CrossRef]
- Yang, N.W.; Zang, L.S.; Wang, S.; Guo, J.Y.; Xu, H.X.; Zhang, F.; Wan, F.H. Biological pest management by predators and parasitoids in the greenhouse vegetables in China. Biol. Control 2014, 68, 92–102. [Google Scholar] [CrossRef]
- Xu, H.Y.; Yang, N.W.; Wan, F.H. Field cage evaluation of interspecific interaction of two aphelinid parasitoids and biocontrol effect on Bemisia tabaci (Hemiptera: Aleyrodidae) Middle East-Asia Minor 1. Entomol. Sci. 2014, 18, 237–244. [Google Scholar] [CrossRef]
- Zhang, X.M.; Ferrante, M.; Wan, F.H.; Yang, N.W.; Lövei, G.L. The parasitoid Eretmocerus hayati is compatible with barrier cropping to decrease whitefly (Bemisia tabaci MED) densities on cotton in China. Insects 2020, 11, 57. [Google Scholar] [CrossRef]
- Akaike, H. Information theory and an extension of the maximum likelihood principle. In Selected Papers of Hirotugu Akaike; Parzen, E., Tanabe, K., Kitagawa, G., Eds.; Springer Series in Statistics; Springer: New York, NY, USA, 1998; pp. 199–213. [Google Scholar]
- Zuur, A.F.; Elena, N.I.; Chris, S.E. A protocol for data exploration to avoid common statistical problems. Methods Ecol. Evol. 2010, 3–14. [Google Scholar] [CrossRef]
- Lenth, R.V. Least-square means: The R package lsmeans. J. Stat. Software 2016, 69, 1–13. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: https://www.R-project.org/ (accessed on 10 June 2020).
- R Studio Team. RStudio: Integrated Development for R; RStudio, Inc.: Boston, MA, USA, 2018; Available online: http://www.rstudio.com/ (accessed on 17 June 2020).
- Abdullah, K.; Ellington, J.J.; Yasmin, S. Storage studies of Anaphes iole Girault (Hymenoptera: Mymaridae) at different developmental stages, temperatures and light-dark regimes. Agric. Conspec. Sci. 2009, 74, 319–326. [Google Scholar]
- Kidane, Z.D. The Effect of Banker Plant and Cold Storage in Biological Control of Bemisia tabaci Middle East-Asia Minor 1 (MEAM1) (Hemiptera: Aleyrodidae) Using Two Aphelinidae Parasitoids. Ph.D. Thesis, Chinese Academy of Agricultural Sciences, Beijing, China, 2015; p. 7.
- Bayram, A.; Ozcan, H.; Kornosor, S. Effect of cold storage on the performance of Telenomus busseolae Gahan (Hymenoptera: Scelionidae), an egg parasitoid of Sesamia nonagrioides (Lefebvre) (Lepidoptera: Noctuidae). Biol. Control 2005, 35, 68–77. [Google Scholar] [CrossRef]
- Bourdais, D.; Vernon, P.; Krespi, L.; van Baaren, J. Behavioural consequences of cold exposure on males and females of Aphidius rhopalosiphi De Stephani Perez (Hymenoptera: Braconidae). BioControl 2012, 57, 349–360. [Google Scholar] [CrossRef]
- Lopez, S.N.; Botto, E. Effect of cold storage on some biological parameters of Eretmocerus corni and Encarsia formosa (Hymenoptera: Aphelinidae). Biol. Control 2005, 33, 123–130. [Google Scholar] [CrossRef]
- Kivan, M.; Kilic, N. Effects of storage at low-temperature of various heteropteran host eggs on the egg parasitoid, Trissolcus semistriatus. BioControl 2005, 50, 589–600. [Google Scholar] [CrossRef]
- Chen, W.; Leopold, R.A. Progeny quality of Gonatocerus ashmeadi (Hymenoptera: Mymaridae) reared on stored eggs of Homalodisca coagulata (Homoptera: Cicadellidae). J. Econ. Entomol. 2007, 100, 685–694. [Google Scholar] [CrossRef]
- Kjarsgaard, A.; Pertoldi, C.; Loeschcke, V.; Blanckenhorn, W.U. The effect of fluctuating temperatures during development on fitness-related traits of Scatophaga stercoraria (Diptera: Scathophagidae). Environ. Entomol. 2013, 42, 1069–1078. [Google Scholar] [CrossRef]
- Consoli, F.L.; Parra, J.R.P. Effects of constant and alternating temperatures on Trichogramma galloi Zucchi (Hym., Trichogrammatidae) biology II. Parasitism capacity and longevity. J. Appl. Entomol. 1995, 119, 667–670. [Google Scholar] [CrossRef]
| Storage Treatment | Emergency Rate (%) | |||
|---|---|---|---|---|
| Encarsia sophia | Eretmocerus hayati | |||
| 10-Day-Old Pupae | 12-Day-Old Pupae | 12-Day-Old Pupae | 15-Day-Old Pupae | |
| Control (26 °C) | 86.0 ± 0.3 aA 1 | 87.0 ± 0.3 aA | 90.0 ± 0.2 aA | 85.0 ± 0.3 aA |
| 12 °C/1 week | 81.0 ± 0.3 abA | 84.0 ± 0.4 aA | 88.0 ± 0.3 aA | 88.0 ± 0.2 aA |
| 12 °C/2 weeks | 80.0 ± 0.3 ab | ⊗ 2 | ⊗ | ⊗ |
| 10 °C/1 week | 75.0 ± 0.3 abA | 85.0 ± 0.3 aA | 87.0 ± 0.3 aA | 86.0 ± 0.3 aA |
| 10 °C/2 weeks | 75.0 ± 0.3 ab | ⊗ | 77.0 ± 0.3 ab | ⊗ |
| 8 °C/1 week | 72.0 ± 0.3 abcB | 88.0 ± 0.3 aA | 62.0 ± 0.4 abcB | 86.0 ± 0.3 aA |
| 8 °C/2 weeks | 57.0 ± 0.4 cA | 53.0 ± 0.3 bA | 59.0 ± 0.3 bcA | 75.0 ± 0.3 aA |
| 6 °C/1 week | 67.0 ± 0.4 bcB | 84.0 ± 0.2 aA | 56.0 ± 0.3 bcB | 84.0 ± 0.3 aA |
| 6 °C/2 weeks | 49.0 ± 0.3 cA | 41.0 ± 0.6 bA | 45.0 ± 0.4 cdB | 77.0 ± 0.3 aA |
| 4 °C/1 week | 56.0 ± 0.5 cA | 39.0 ± 0.3 bB | 43.0 ± 0.3 cdA | 40.0 ± 0.3 bA |
| 4 °C/2 weeks | 18.0 ± 0.2 dA | 14.0 ± 0.3 cA | 22.0 ± 0.1 dA | 30.0 ± 0.3 bA |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kidane, D.; Ferrante, M.; Man, X.-M.; Liu, W.-X.; Wan, F.-H.; Yang, N.-W. Cold Storage Effects on Fitness of the Whitefly Parasitoids Encarsia sophia and Eretmocerus hayati. Insects 2020, 11, 428. https://doi.org/10.3390/insects11070428
Kidane D, Ferrante M, Man X-M, Liu W-X, Wan F-H, Yang N-W. Cold Storage Effects on Fitness of the Whitefly Parasitoids Encarsia sophia and Eretmocerus hayati. Insects. 2020; 11(7):428. https://doi.org/10.3390/insects11070428
Chicago/Turabian StyleKidane, Dawit, Marco Ferrante, Xiao-Ming Man, Wan-Xue Liu, Fang-Hao Wan, and Nian-Wan Yang. 2020. "Cold Storage Effects on Fitness of the Whitefly Parasitoids Encarsia sophia and Eretmocerus hayati" Insects 11, no. 7: 428. https://doi.org/10.3390/insects11070428
APA StyleKidane, D., Ferrante, M., Man, X.-M., Liu, W.-X., Wan, F.-H., & Yang, N.-W. (2020). Cold Storage Effects on Fitness of the Whitefly Parasitoids Encarsia sophia and Eretmocerus hayati. Insects, 11(7), 428. https://doi.org/10.3390/insects11070428







