Effects of Lethal Bronzing Disease, Palm Height, and Temperature on Abundance and Monitoring of Haplaxius crudus
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
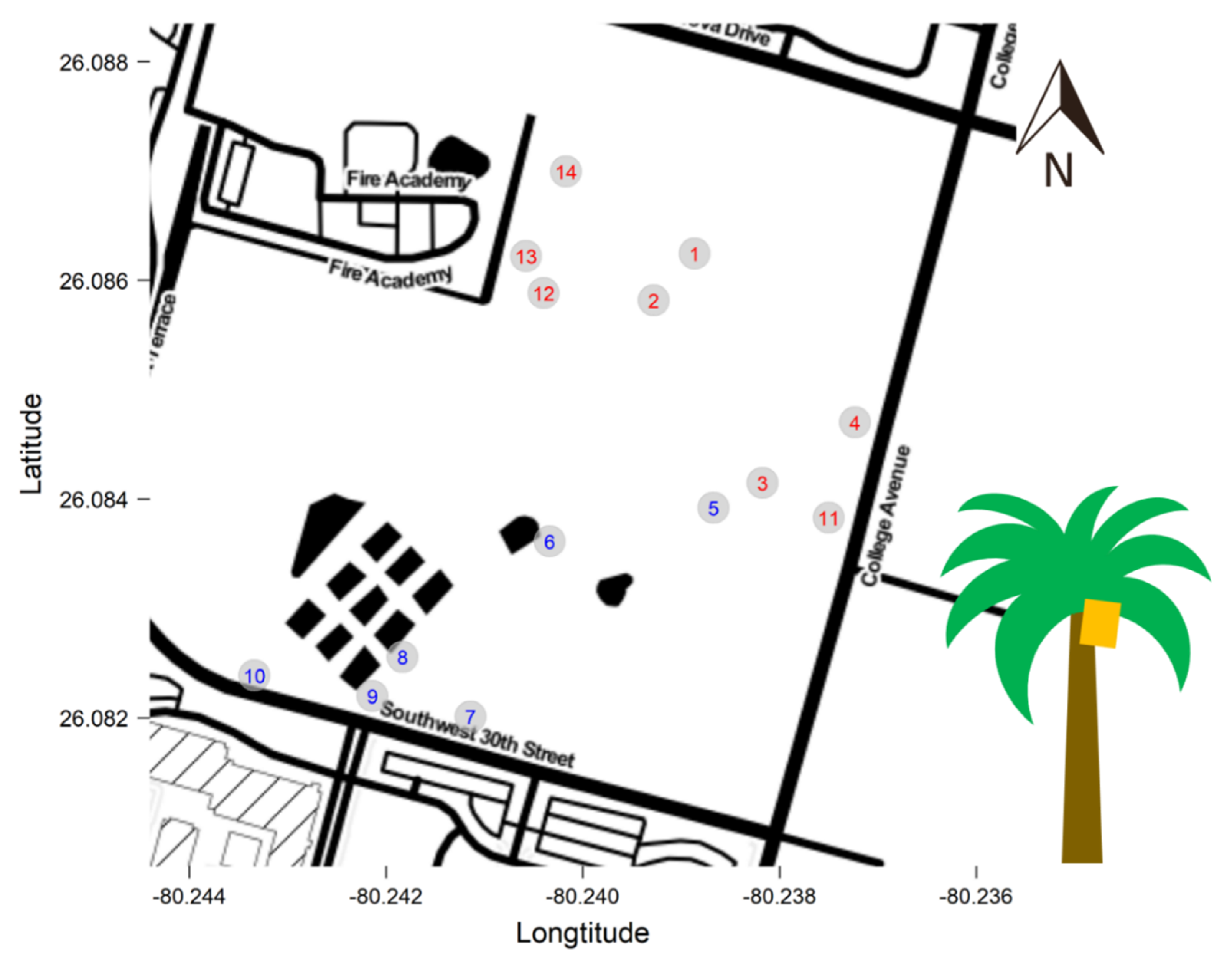
2.1. Study Site
2.2. Data Collection
2.3. Statistical Analysis
3. Results
3.1. Effects of LB on Insect Abundance
3.2. Effects of LB, Palm Height, and Temperature on H. crudus Abundance
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wilfert Eckel, R.V.; Lampert, E.P. Relative attractiveness of tobacco etch virus-infected and healthy flue-cured tobacco plants to aphids (Homoptera: Aphididae). J. Econ. Entomol. 1996, 89, 1017–1027. [Google Scholar] [CrossRef]
- Ebbert, M.A.; Nault, L.R. Survival in Dalbulus leafhopper vectors improves after exposure to maize stunting pathogens. Entomol. Exp. Appl. 2001, 100, 311–324. [Google Scholar] [CrossRef]
- Mauck, K.E.; De Moraes, C.M.; Mescher, M.C. Deceptive chemical signals induced by a plant virus attract insect vectors to inferior hosts. Proc. Natl. Acad. Sci. USA 2010, 107, 3600–3605. [Google Scholar] [CrossRef] [PubMed]
- Mauck, K.; Bosque-Pérez, N.A.; Eigenbrode, S.D.; De Moraes, C.M.; Mescher, M.C. Transmission mechanisms shape pathogen effects on host-vector interactions: Evidence from plant viruses. Funct. Ecol. 2012, 26, 1162–1175. [Google Scholar] [CrossRef]
- Mauck, K.E.; Chesnais, Q.; Shapiro, L.R. Evolutionary determinants of host and vector manipulation by plant viruses. Adv. Virus Res. 2018, 101, 189–250. [Google Scholar] [CrossRef]
- Mayer, C.J.; Vilcinskas, A.; Gross, J. Chemically mediated multitrophic interactions in a plant-insect vector-phytoplasma system compared with a partially nonvector species. Agric. For. Entomol. 2011, 13, 25–35. [Google Scholar] [CrossRef]
- Stafford, C.A.; Walker, G.P.; Ullman, D.E. Infection with a plant virus modifies vector feeding behavior. Proc. Natl. Acad. Sci. USA 2011, 108, 9350–9355. [Google Scholar] [CrossRef]
- Jiménez-Martínez, E.S.; Bosque-Pérez, N.A.; Berger, P.H.; Zemetra, R.S.; Ding, H.; Eigenbrode, S.D. Volatile cues influence the response of Rhopalosiphum padi (Homoptera: Aphididae) to barley yellow dwarf virus–infected transgenic and untransformed wheat. Environ. Entomol. 2004, 33, 1207–1216. [Google Scholar] [CrossRef]
- Mauck, K.E.; De Moraes, C.M.; Mescher, M.C. Effects of pathogens on sensory-mediated interactions between plants and insect vectors. Curr. Opin. Plant Biol. 2016, 32, 53–61. [Google Scholar] [CrossRef]
- Pradit, N.; Mescher, M.C.; Wang, Y.; Vorsa, N.; Rodriguez-Saona, C. Phytoplasma infection of cranberries benefits non-vector phytophagous insects. Front. Ecol. Evol. 2019, 7, 1–11. [Google Scholar] [CrossRef]
- Prokopy, R.J.; Owens, E.D. Visual detection of plants by herbivorous insects. Annu. Rev. Entomol. 1983, 28, 337–364. [Google Scholar] [CrossRef]
- Todd, J.L.; Harris, M.O.; Nault, L.R. Importance of color stimuli in host-finding by Dalbulus leafhoppers. Entomol. Exp. Appl. 1990, 54, 245–255. [Google Scholar] [CrossRef]
- Fereres, A.; Kampmeier, G.E.; Irwin, M.E. Aphid attraction and preference for soybean and pepper plants infected with Potyviridae. Ann. Entomol. Soc. Am. 1999, 92, 542–548. [Google Scholar] [CrossRef]
- Fereres, A.; Moreno, A. Behavioural aspects influencing plant virus transmission by homopteran insects. Virus Res. 2009, 141, 158–168. [Google Scholar] [CrossRef]
- Eigenbrode, S.D.; Ding, H.; Shiel, P.; Berger, P.H. Volatiles from potato plants infected with potato leafroll virus attract and arrest the virus vector, Myzus persicae (Homoptera: Aphididae). Proc. R. Soc. Lond. Ser. B Biol. Sci. 2002, 269, 455–460. [Google Scholar] [CrossRef]
- Ngumbi, E.; Eigenbrode, S.D.; Bosque-Pérez, N.A.; Ding, H.; Rodriguez, A. Myzus persicae is arrested more by blends than by individual compounds elevated in headspace of PLRV-infected potato. J. Chem. Ecol. 2007, 33, 1733–1747. [Google Scholar] [CrossRef]
- Brault, V.; Uzest, M.; Monsion, B.; Jacquot, E.; Blanc, S. Aphids as transport devices for plant viruses. Comptes Rendus Biol. 2010, 333, 524–538. [Google Scholar] [CrossRef]
- Mann, R.S.; Ali, J.G.; Hermann, S.L.; Tiwari, S.; Pelz-Stelinski, K.S.; Alborn, H.T.; Stelinski, L.L. Induced release of a plant-defense volatile “deceptively” attracts insect vectors to plants infected with a bacterial pathogen. PLoS Pathog. 2012, 8, e1002610. [Google Scholar] [CrossRef]
- Aksenov, A.A.; Martini, X.; Zhao, W.; Stelinski, L.L.; Davis, C.E. Synthetic blends of volatile, phytopathogen-induced odorants can be used to manipulate vector behavior. Front. Ecol. Evol. 2014, 2, 78. [Google Scholar] [CrossRef]
- Mas, F.; Vereijssen, J.; Suckling, D.M. Influence of the pathogen Candidatus Liberibacter solanacearum on tomato host plant volatiles and psyllid vector settlement. J. Chem. Ecol. 2014, 40, 1197–1202. [Google Scholar] [CrossRef]
- Doi, Y.; Teranaka, M.; Yora, K.; Asuyama, H. Mycoplasma- or PLT group-like microorganisms found in the phloem elements of plants infected with mulberry dwarf, potato witches’ broom, aster yellows, or paulownia witches’ broom. Jpn. J. Phytopathol. 1967, 33, 259–266. [Google Scholar] [CrossRef]
- Lee, I.-M.; Davis, R.E.; Gundersen-Rindal, D.E. Phytoplasma: Phytopathogenic mollicutes. Annu. Rev. Microbiol. 2000, 54, 221–255. [Google Scholar] [CrossRef]
- Hogenhout, S.A.; Oshima, K.; Ammar, E.D.; Kakizawa, S.; Kingdom, H.N.; Namba, S. Phytoplasmas: Bacteria that manipulate plants and insects. Mol. Plant Pathol. 2008, 9, 403–423. [Google Scholar] [CrossRef]
- Weintraub, P.G.; Beanland, L. Insect vectors of phytoplasmas. Annu. Rev. Entomol. 2006, 51, 91–111. [Google Scholar] [CrossRef]
- Bai, X.; Zhang, J.; Ewing, A.; Miller, S.A.; Radek, A.J.; Shevchenko, D.V.; Tsukerman, K.; Walunas, T.; Lapidus, A.; Campbell, J.W.; et al. Living with genome instability: The adaptation of phytoplasmas to diverse environments of their insect and plant hosts. J. Bacteriol. 2006, 188, 3682–3696. [Google Scholar] [CrossRef]
- Kumari, S.; Nagendran, K.; Rai, A.B.; Singh, B.; Rao, G.P.; Bertaccini, A. Global status of phytoplasma diseases in vegetable crops. Front. Microbiol. 2019, 10, 1349. [Google Scholar] [CrossRef]
- Mayer, C.J.; Vilcinskas, A.; Gross, J. Phytopathogen lures its insect vector by altering host plant odor. J. Chem. Ecol. 2008, 34, 1045–1049. [Google Scholar] [CrossRef]
- Gonzalez, J.G.; Jaramillo, M.G.; Lopes, J.R.S. Undetected infection by maize bushy stunt phytoplasma enhances host-plant preference to Dalbulus maidis (Hemiptera: Cicadellidae). Environ. Entomol. 2018, 47, 396–402. [Google Scholar] [CrossRef]
- Ramos, A.; Esteves, M.B.; Cortés, M.T.B.; Lopes, J.R.S. Maize bushy stunt phytoplasma favors its spread by changing host preference of the insect vector. Insects 2020, 11, 600. [Google Scholar] [CrossRef]
- Bahder, B.W.; Soto, N.; Helmick, E.; Dey, K.; Komondy, L.; Humphries, A.R.; Mou, D.-F.; Bailey, R.; Ascunce, M.; Goss, E. A survey of declining palms (Arecaceae) for the 16SrIV-D phytoplasma for evaluating distribution and host range in Florida. Plant Dis. 2019, 103, 2512–2519. [Google Scholar] [CrossRef]
- Bahder, B.W.; Helmick, E.E.; Harrison, N.A. Detecting and differentiating phytoplasmas belonging to subgroups 16SrIV-A and 16SrIV-D associated with lethal declines of palms in Florida using qPCR and high-resolution melt analysis (HRMA). Plant Dis. 2017, 101, 1449–1454. [Google Scholar] [CrossRef]
- Bahder, B.W.; Soto, N.; Mou, D.F.; Humphries, A.R.; Helmick, E.E. Quantification and distribution of the 16SrIV-D phytoplasma in the wild date palm, Phoenix sylvestris, at different stages of decline using quantitative PCR (qPCR) Analysis. Plant Dis. 2020, 104, 1328–1334. [Google Scholar] [CrossRef]
- Narváez, M.; Vázquez-Euán, R.; Harrison, N.A.; Nic-Matos, G.; Julia, J.F.; Dzido, J.L.; Fabre, S.; Dollet, M.; Oropeza, C. Presence of 16SrIV phytoplasmas of subgroups A, D and E in planthopper Haplaxius crudus Van Duzee insects in Yucatán, Mexico. 3 Biotech 2018, 8, 61. [Google Scholar] [CrossRef]
- Brown, S.E.; Been, B.O.; McLaughlin, W.A. Detection and variability of the lethal yellowing group (16Sr IV) phytoplasmas in the Cedusa sp. (Hemiptera: Auchenorrhyncha: Derbidae) in Jamaica. Ann. Appl. Biol. 2006, 149, 53–62. [Google Scholar] [CrossRef]
- Halbert, S.E.; Wilson, S.W.; Bextine, B.; Youngblood, S.B. Potential planthopper vectors of palm phytoplasmas in Florida with a description of a new species of the genus Omolicna (Hemiptera: Fulgoroidea). Fla. Entomol. 2014, 97, 90–97. [Google Scholar] [CrossRef]
- Mou, D.-F.; Humphries, A.R.; Soto, N.; Helmick, E.E.; Ascunce, M.S.; Goss, E.M.; Bahder, B.W. A Survey of auchenorrhynchan insects for identification of potential vectors of the 16SrIV-D phytoplasma in Florida. Fla. Entomol. 2020, 103, 344–352. [Google Scholar] [CrossRef]
- Dzido, J.-L.; Sánchez, R.; Dollet, M.; Julia, J.-F.; Narvaez, M.; Fabre, S.; Oropeza, C. Haplaxius crudus (Hemiptera: Cixiidae) transmits the lethal yellowing phytoplasmas, 16SrIV, to Pritchardia pacifica Seem. & H.Wendl (Arecaceae) in Yucatan, Mexico. Neotrop. Entomol. 2020, 1–11. [Google Scholar] [CrossRef]
- Bahder, B.W.; Helmick, E.E.; Chakrabarti, S.; Osorio, S.; Soto, N.; Chouvenc, T.; Harrison, N.A. Disease progression of a lethal decline caused by the 16SrIV-D phytoplasma in Florida palms. Plant Pathol. 2018, 67, 1821–1828. [Google Scholar] [CrossRef]
- Harrison, N.A.; Narváez, M.; Almeyda, H.; Cordova, I.; Carpio, M.L.; Oropeza, C. First report of group 16SrIV phytoplasmas infecting coconut palms with leaf yellowing symptoms on the Pacific coast of Mexico. Plant Pathol. 2002, 51, 808. [Google Scholar] [CrossRef]
- Harrison, N.A.; Helmick, E.E.; Elliott, M.L. Lethal yellowing-type diseases of palms associated with phytoplasmas newly identified in Florida, USA. Ann. Appl. Biol. 2008, 153, 85–94. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and environment for statistical computing. R foundation for statistical computing, Vienna, Austria. Available online: http://www.R-project.org/ (accessed on 4 October 2020).
- Bates, D.; Maechler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Harrison, X.A. Using observation-level random effects to model overdispersion in count data in ecology and evolution. PeerJ 2014, 2, e616. [Google Scholar] [CrossRef]
- Fox, J.; Weisberg, S. An {R} Companion to Applied Regression, 3rd ed.; Sage: Thousand Oaks, CA, USA, 2019; Available online: https://socialsciences.mcmaster.ca/jfox/Books/Companion/ (accessed on 4 October 2020).
- Length, R.; Singmann, H.; Love, J.; Buerkner, P.; Herve, M. Emmeans: Estimated marginal means, aka least-squares means. R package version 1.4.4. Available online: https://CRAN.R-project.org/package=emmeans (accessed on 4 October 2020).
- Barton, B. MuMIn: Multi-model inference. R package version 1.43.15. Available online: https://CRAN.R-project.org/package=MuMIn (accessed on 4 October 2020).
- Martini, X.; Willett, D.S.; Kuhns, E.H.; Stelinski, L.L. Disruption of vector host preference with plant volatiles may reduce spread of insect-transmitted plant pathogens. J. Chem. Ecol. 2016, 42, 357–367. [Google Scholar] [CrossRef]
- Srinivasan, R.; Alvarez, J.M.; Eigenbrode, S.D.; Bosque-pérez, N.A. Influence of hairy nightshade Solanum sarrachoides (Sendtner) and Potato leafroll virus (Luteoviridae: Polerovirus) on the host preference of Myzus persicae (Sulzer) (Homoptera: Aphididae). Environ. Entomol. 2006, 35, 546–553. [Google Scholar] [CrossRef]
- Denno, R.F.; Roderick, G.K. Population biology of planthoppers. Annu. Rev. Entomol. 1990, 35, 489–520. [Google Scholar] [CrossRef]
- Denno, R.F. The evolution of dispersal polymorphisms in insects: The influence of habitats, host plants and mates. Res. Popul. Ecol. 1994, 36, 127–135. [Google Scholar] [CrossRef]
- Bale, J.S.; Masters, G.J.; Hodkinson, I.D.; Awmack, C.; Bezemer, T.M.; Brown, V.K.; Butterfield, J.; Buse, A.; Coulson, J.C.; Farrar, J.; et al. Herbivory in global climate change research: Direct effects of rising temperature on insect herbivores. Glob. Chang. Biol. 2002, 8, 1–16. [Google Scholar] [CrossRef]
- Hall, D.G. An assessment of yellow sticky card traps as indicators of the abundance of adult Diaphorina citri (Hemiptera: Psyllidae) in citrus. J. Econ. Entomol. 2009, 102, 446–452. [Google Scholar] [CrossRef]
- Tomaseto, A.F.; Miranda, M.P.; Moral, R.A.; de Lara, I.A.R.; Fereres, A.; Lopes, J.R.S. Environmental conditions for Diaphorina citri Kuwayama (Hemiptera: Liviidae) take-off. J. Appl. Entomol. 2018, 142, 104–113. [Google Scholar] [CrossRef]
- Jonason, D.; Franzén, M.; Ranius, T. Surveying moths using light traps: Effects of weather and time of year. PLoS ONE 2014, 9, e92453. [Google Scholar] [CrossRef]
- Holopainen, J.K.; Gershenzon, J. Multiple stress factors and the emission of plant VOCs. Trends Plant Sci. 2010, 15, 176–184. [Google Scholar] [CrossRef]
- Tsai, J.H.; Woodiel, N.L.; Kirsch, O.H. Rearing techniques for Haplaxius crudus (Homoptera: Cixiidae). Fla. Entomol. 1976, 59, 41–43. [Google Scholar] [CrossRef]
- Reinert, J.A. Phenology and Density of Haplaxius crudus (Homoptera: Cixiidae) on three southern turfgrasses. Environ. Entomol. 1980, 9, 13–15. [Google Scholar] [CrossRef]
- Tsai, J.H.; Kirsch, O.H. Bionomics of Haplaxius crudus (Homoptera: Cixiidae). Environ. Entomol. 1977, 7, 305–308. [Google Scholar] [CrossRef]
- Martin, T.L.; Huey, R.B. Why “Suboptimal” ss optimal: Jensen’s inequality and ectotherm thermal preferences. Am. Nat. 2008, 171, 102–118. [Google Scholar] [CrossRef] [PubMed]
- Wilson, E.S.; Murphy, C.E.; Rinehart, J.P.; Yocum, G.; Bowsher, J.H. Microclimate temperatures impact nesting preference in Megachile rotundata (Hymenoptera: Megachilidae). Environ. Entomol. 2020, 49, 296–303. [Google Scholar] [CrossRef]
- Babin, R.; Anikwe, J.C.; Dibog, L.; Lumaret, J.P. Effects of cocoa tree phenology and canopy microclimate on the performance of the mirid bug Sahlbergella singularis. Entomol. Exp. Appl. 2011, 141, 25–34. [Google Scholar] [CrossRef]
- Penrose, L.J.; Nicol, H.I. Aspects of microclimate variation within apple tree canopies and between sites in relation to potential Venturia inaequalis infection. N. Z. J. Crop Hortic. Sci. 1996, 24, 259–266. [Google Scholar] [CrossRef]
- Ingwell, L.L.; Eigenbrode, S.D.; Bosque-Pérez, N.A. Plant viruses alter insect behavior to enhance their spread. Sci. Rep. 2012, 2, 1–6. [Google Scholar] [CrossRef]
- Roosien, B.K.; Gomulkiewicz, R.; Ingwell, L.L.; Bosque-Pérez, N.A.; Rajabaskar, D.; Eigenbrode, S.D. Conditional vector preference aids the spread of plant pathogens: Results from a model. Environ. Entomol. 2013, 42, 1299–1308. [Google Scholar] [CrossRef]
- Rajabaskar, D.; Bosque-Pérez, N.A.; Eigenbrode, S.D. Preference by a virus vector for infected plants is reversed after virus acquisition. Virus Res. 2014, 186, 32–37. [Google Scholar] [CrossRef]
- Atakan, E.; Canhilal, R. Evaluation of yellow sticky traps at various heights for monitoring cotton insect pests. J. Agric. Urban Entomol. 2004, 21, 15–24. [Google Scholar]


| Effects | χ2 Value | p-Value |
|---|---|---|
| LBD | 2.8 | 0.097 |
| Palm height | 0.5 | 0.485 |
| Temperature | 38.7 | <0.0001 |
| LBD × Palm height | 1.4 | 0.242 |
| LBD × Temperature | 9.0 | 0.003 |
| Palm height × Temperature | 35.6 | <0.0001 |
| LBD × Palm height × Temperature | 22.3 | <0.0001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mou, D.-F.; Lee, C.-C.; Hahn, P.G.; Soto, N.; Humphries, A.R.; Helmick, E.E.; Bahder, B.W. Effects of Lethal Bronzing Disease, Palm Height, and Temperature on Abundance and Monitoring of Haplaxius crudus. Insects 2020, 11, 748. https://doi.org/10.3390/insects11110748
Mou D-F, Lee C-C, Hahn PG, Soto N, Humphries AR, Helmick EE, Bahder BW. Effects of Lethal Bronzing Disease, Palm Height, and Temperature on Abundance and Monitoring of Haplaxius crudus. Insects. 2020; 11(11):748. https://doi.org/10.3390/insects11110748
Chicago/Turabian StyleMou, De-Fen, Chih-Chung Lee, Philip G. Hahn, Noemi Soto, Alessandra R. Humphries, Ericka E. Helmick, and Brian W. Bahder. 2020. "Effects of Lethal Bronzing Disease, Palm Height, and Temperature on Abundance and Monitoring of Haplaxius crudus" Insects 11, no. 11: 748. https://doi.org/10.3390/insects11110748
APA StyleMou, D.-F., Lee, C.-C., Hahn, P. G., Soto, N., Humphries, A. R., Helmick, E. E., & Bahder, B. W. (2020). Effects of Lethal Bronzing Disease, Palm Height, and Temperature on Abundance and Monitoring of Haplaxius crudus. Insects, 11(11), 748. https://doi.org/10.3390/insects11110748






