Larval Pollen Stress Increases Adult Susceptibility to Clothianidin in Honey Bees
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Colonies
2.2. Clothianidin and Laboratory Survival Assays
2.3. Stress Protein and Lipids Analysis
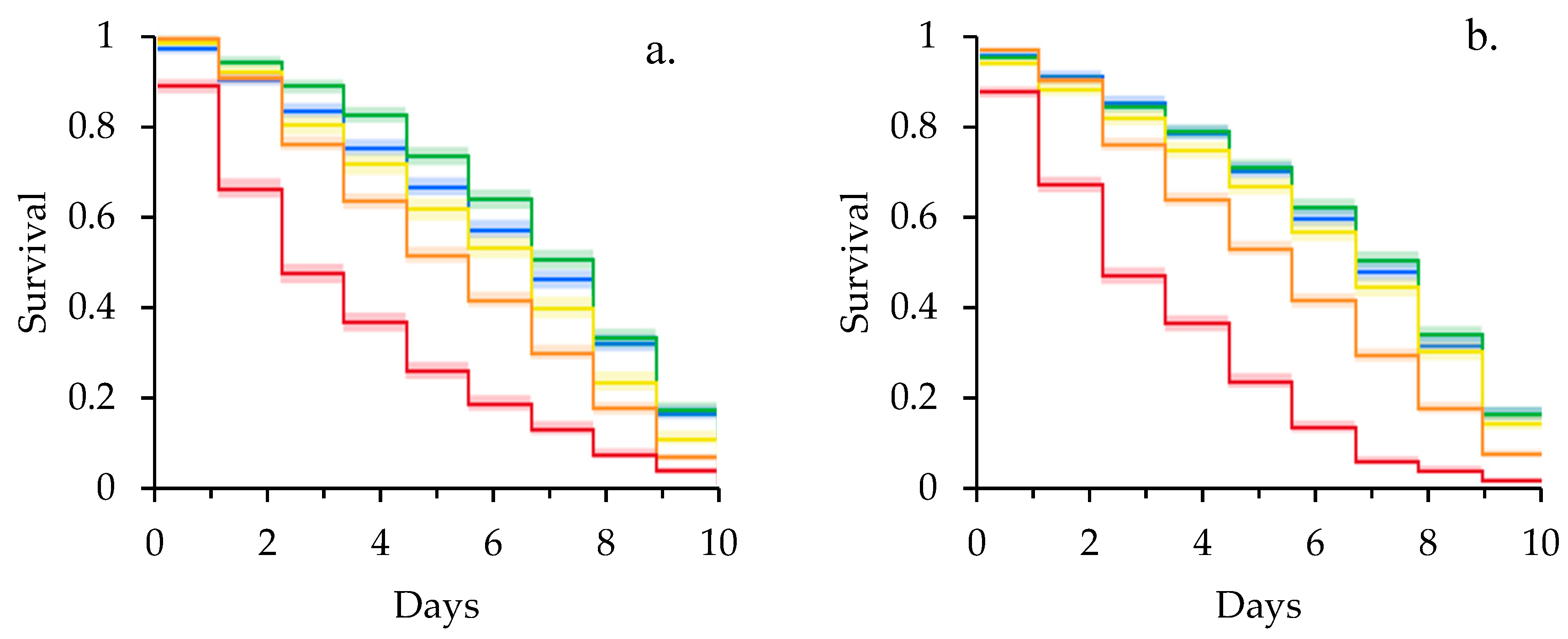
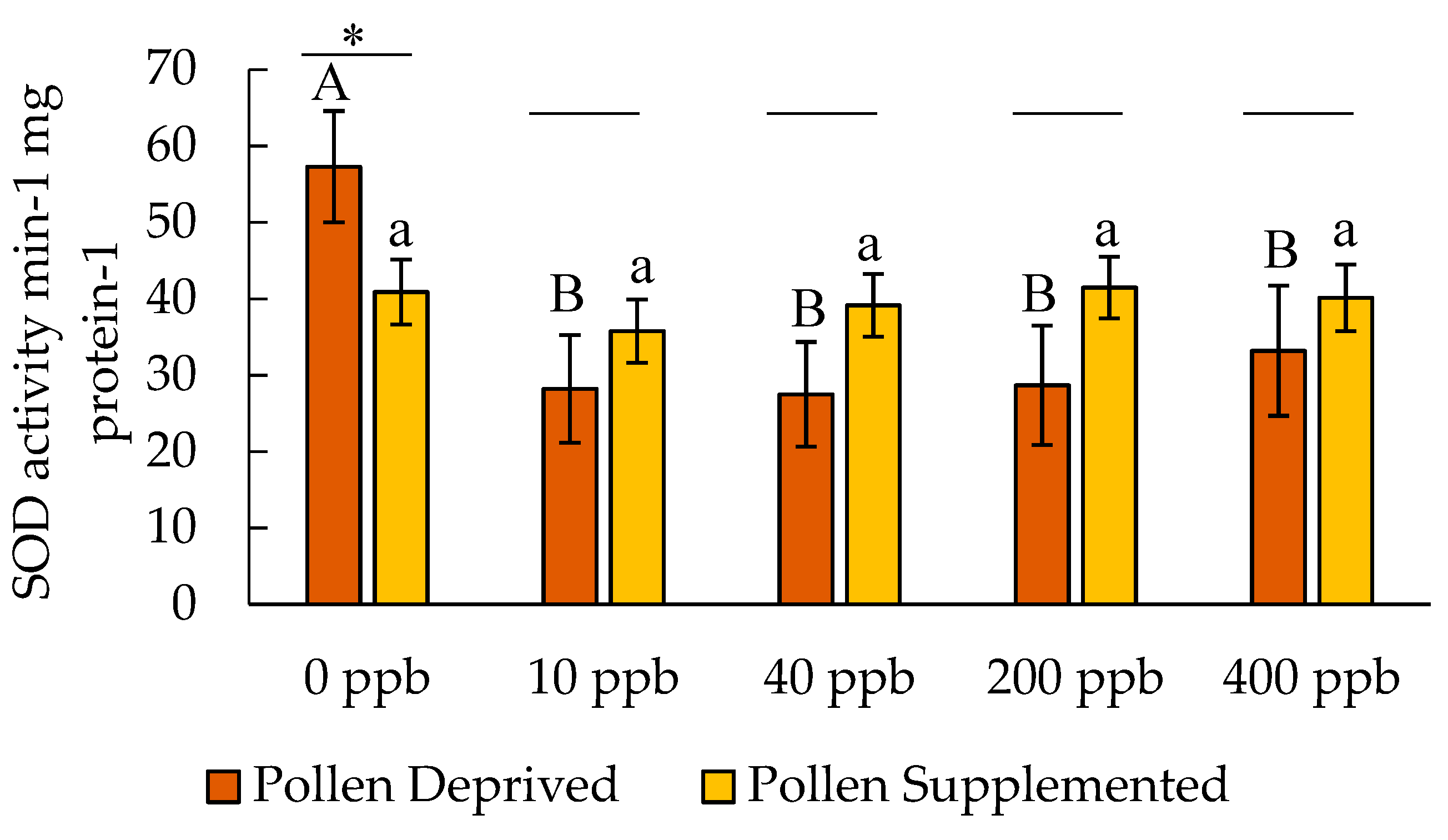
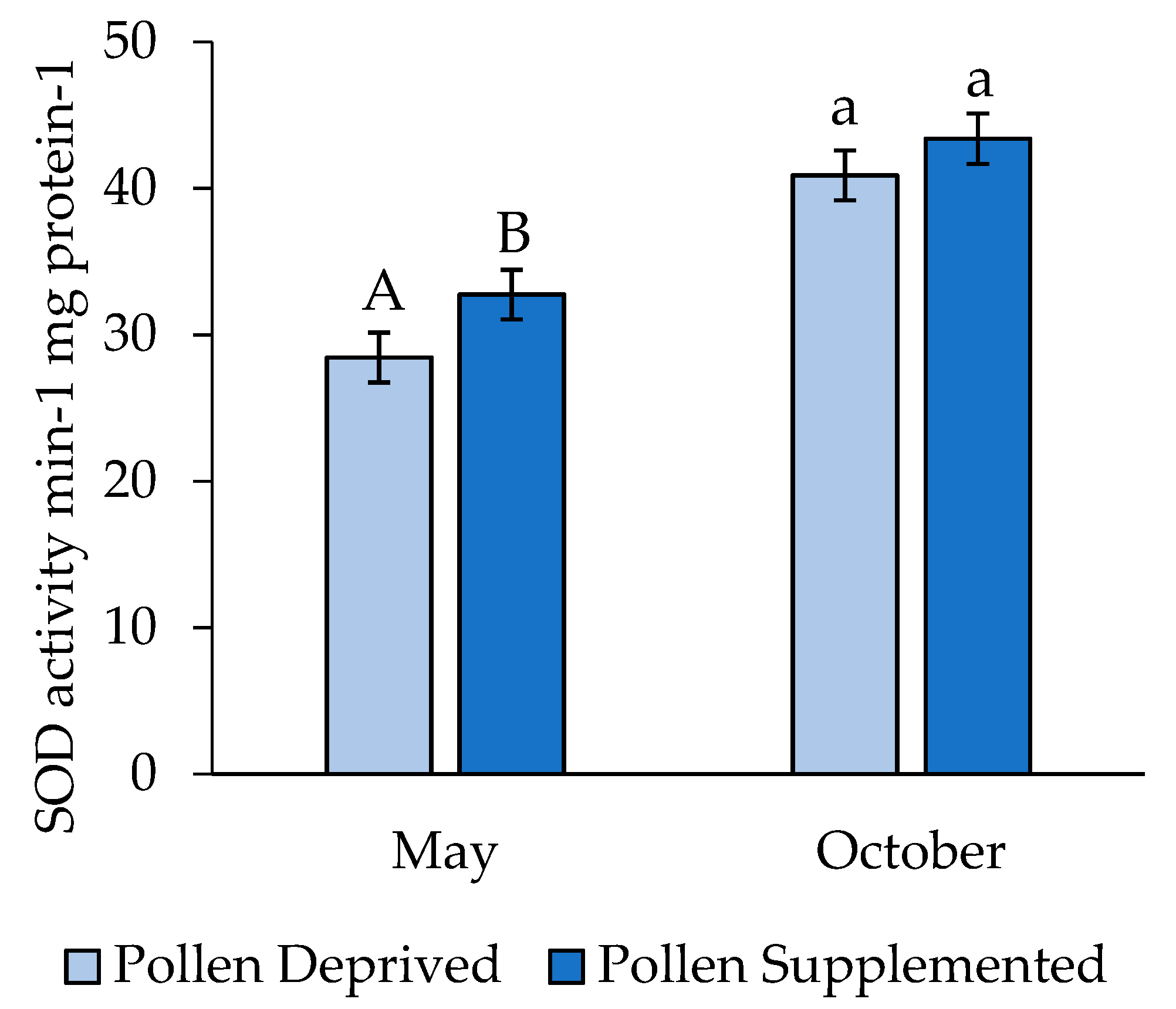
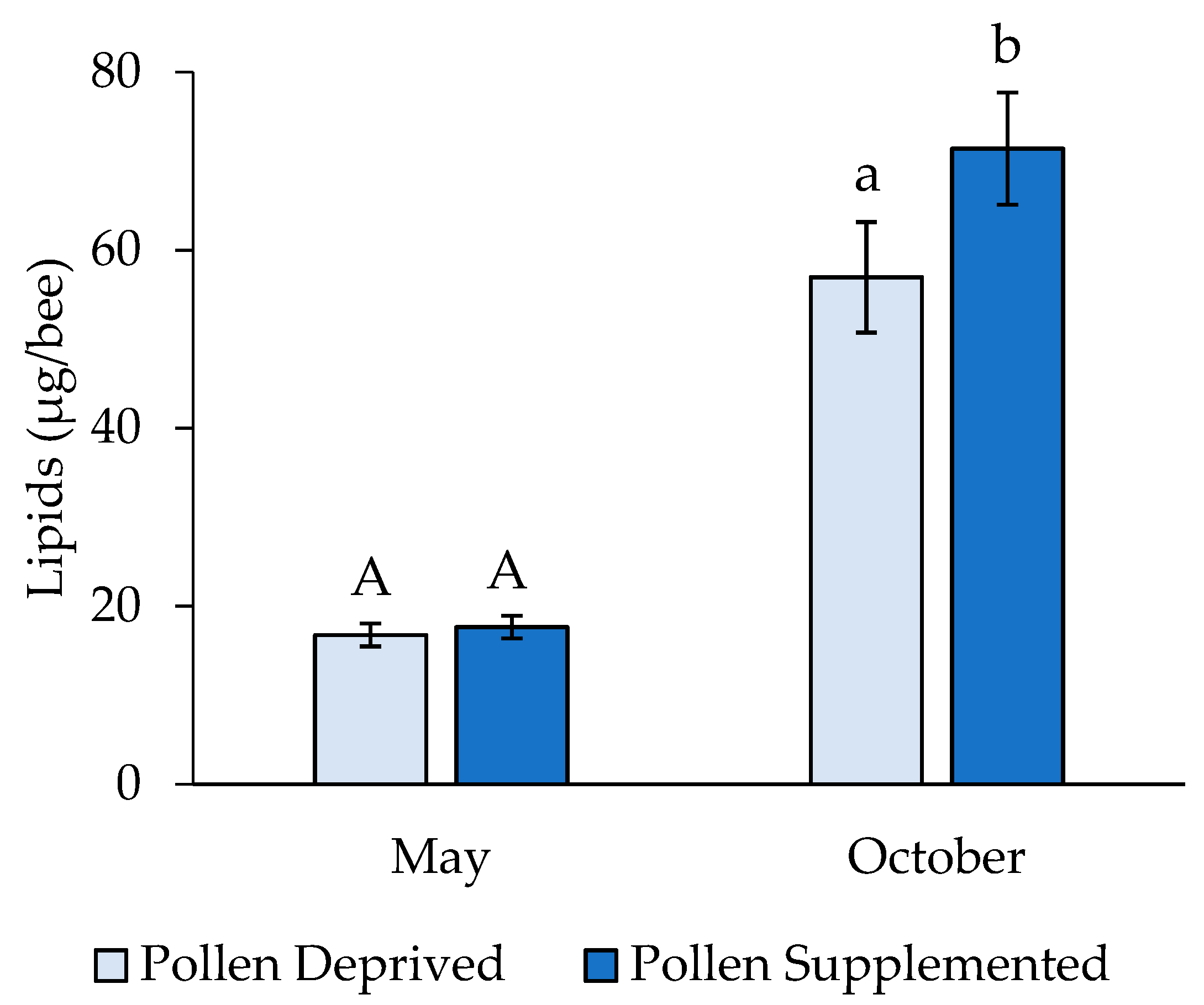
3. Results
3.1. Clothianidin and Laboratory Survival Assays
3.2. Stress Protein and Lipids Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Johnston, C.A. Agricultural expansion: Land use shell game in the US Northern Plains. Landsc. Ecol. 2014, 29, 81–95. [Google Scholar] [CrossRef]
- Mogren, C.L.; Rand, T.A.; Fausti, S.W.; Lundgren, J.G. The effects of crop intensification on the diversity of native pollinator communities. Environ. Entomol. 2016, 45, 865–872. [Google Scholar] [CrossRef] [PubMed]
- Douglas, M.R.; Tooker, J.F. Large-scale deployment of seed treatments has driven rapid increase in use of neonicotinoid insecticides and preemptive pest management in US field crops. Environ. Sci. Technol. 2015, 49, 5088–5097. [Google Scholar] [CrossRef] [PubMed]
- Hladik, M.L.; Kolpin, D.W.; Kuivila, K.M. Widespread occurrence of neonicotinoid insecticides in streams in a high corn and soybean producing region, USA. Environ. Pollut. 2014, 193, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Mogren, C.L.; Lundgren, J.G. Neonicotinoid-contaminated pollinator strips adjacent to cropland reduce honey bee health. Sci. Rep. 2016, 6, 29608. [Google Scholar] [CrossRef] [PubMed]
- Botías, C.; David, A.; Horwood, J.; Abdul-Sada, A.; Nicholls, E.; Hill, E.; Goulson, D. Neonicotinoid residues in wildflowers, a potential route of chronic exposure for bees. Environ. Sci. Technol. 2015, 49, 12731–12740. [Google Scholar] [CrossRef]
- Alaux, C.F.; Ducloz, F.; Crauser, D.; LeConte, Y. Diet effects on honeybee immunocompetence. Boil. Lett. 2010, 6, 562–565. [Google Scholar] [CrossRef]
- Fischer, J.; Müller, T.; Spatz, A.-K.; Greggers, U.; Grünewald, B.; Menzel, R. Neonicotinoids interfere with specific components of navigation in honeybees. PLoS ONE 2014, 9, e91364.Tsve. [Google Scholar] [CrossRef]
- Tsvetkov, N.; Samson-Robert, O.; Sood, K.; Patel, H.; Malena, D.; Gajiwala, P.H.; Maciukiewicz, P.; Fournier, V.; Zayed, A. Chronic exposure to neonicotinoids reduces honey bee health near corn crops. Science 2017, 356, 1395–1397. [Google Scholar] [CrossRef]
- Gallant, A.L.; Euliss, N.H.; Browning, Z. Mapping large area landscape suitability for honey bees to assess the influence of land-use change on sustainability of national pollination services. PLoS ONE 2014, 9, e99268. [Google Scholar] [CrossRef]
- Brodschneider, R.; Crailsheim, K. Nutrition and health in honey bees. Apidologie 2010, 41, 278–294. [Google Scholar] [CrossRef]
- Winston, M. The Biology of the Honey Bee; Harvard University Press: Cambridge, MA, USA, 1987; ISBN 9780674074095. [Google Scholar]
- Huang, Z. Pollen nutrition affects honey bee stress resistance. Terr. Arthropod Rev. 2012, 5, 175–189. [Google Scholar] [CrossRef]
- Wahl, O.; Ulm, K. Influence of pollen feeding and physiological condition on pesticide sensitivity of the honeybee Apis mellifera carnica. Oecologia 1983, 59, 106–128. [Google Scholar] [CrossRef] [PubMed]
- Archer, C.R.; Pirk, C.W.W.; Wright, G.A.; Nicolson, S.W. Nutrition affects survival in African honeybees exposed to interacting stressors. Funct. Ecol. 2014, 28, 913–923. [Google Scholar] [CrossRef]
- Mogren, C.L.; Margotta, J.; Danka, R.G.; Healy, K.B. Supplemental carbohydrates influence abiotic stress resistance in honey bees. J. Apic. Res. 2018, 57, 682–689. [Google Scholar] [CrossRef]
- Schmehl, D.; Teal, P.; Frazier, J.; Grozinger, C. Genomic analysis of the interaction between pesticide exposure and nutrition in honey bees (Apis mellifera). J. Insect Physiol. 2014, 71, 177–190. [Google Scholar] [CrossRef] [PubMed]
- Williamson, S.; Willis, S.; Wright, G. Exposure to neonicotinoids influences the motor function of adult worker honeybees. Ecotoxicology 2014, 23, 1409–1418. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.; Tautz, J.; Grünewald, B.; Fuchs, S. RFID tracking of sublethal effects of two neonicotinoid insecticides on the foraging behavior of Apis mellifera. PLoS ONE 2012, 7, e30023. [Google Scholar] [CrossRef]
- Tosi, S.; Burgio, G.; Nieh, J. A common neonicotinoid pesticide, thiamethoxam, impairs honey bee flight ability. Sci. Rep. 2017, 7, 1201. [Google Scholar] [CrossRef]
- Hulbert, A.; Pamplona, R.; Buffenstein, R.; Buttemer, W. Life and death: Metabolic rate, membrane composition, and life span of animals. Physiol. Rev. 2007, 87, 1175–1213. [Google Scholar] [CrossRef]
- Cui, H.; Kong, Y.; Zhang, H. Oxidative stress, mitochondrial dysfunction, and aging. J. Signal Transduct. 2012, 2012, 646354. [Google Scholar] [CrossRef]
- Margotta, J.; Roberts, S.P.; Elekonich, M.M. Effects of flight activity and age on oxidative damage in the honey bee, Apis mellifera. J. Exp. Biol. 2018, 221, 183228. [Google Scholar] [CrossRef]
- Schneider, S.S. The honey bee colony: Life history. In The Hive and the Honey Bee; Graham, J.M., Ed.; Dadant & Sons: Hamilton, IL, USA, 2015; pp. 73–109. [Google Scholar]
- United States Geological Survey. 2014 Estimated Annual Agricultural Pesticide Use. 2015. Available online: https://water.usgs.gov/nawqa/pnsp/usage/maps/compound_listing.php (accessed on 25 October 2018).
- Rinkevich, F.D.; Danka, R.G.; Healy, K.B. Influence of varroa mite (Varroa destructor) management practices on insecticide sensitivity in the honey bee (Apis mellifera). Insects 2017, 8, 9. [Google Scholar] [CrossRef]
- Williams, G.R.; Alaux, C.; Costa, C.; Csáki, T.; Doublet, V.; Eisenhardt, D.; Fries, I.; Kuhn, R.; McMahon, D.P.; Medrzycki, P.; et al. Standard methods for maintaining adult Apis mellifera in cages under in vitro laboratory conditions. J. Apic. Res. 2013, 52. [Google Scholar] [CrossRef]
- Tosi, S.; Nieh, J.; Sgolastra, F.; Cabbri, R.; Medrzycki, P. Neonicotinoid pesticides and nutritional stress synergistically reduce survival in honey bees. Proc. R. Soc. B Boil. Sci. 2017, 284, 20171711. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Chang, C.; Tao, L.; Chen, M. Distributions of neonicotinoid insecticides in the Commonwealth of Massachusetts: A temporal and spatial variation analysis for pollen and honey samples. Enviornmental Chem. 2016, 13, 4–11. [Google Scholar] [CrossRef]
- Codling, G.; Al Naggar, Y.; Giesy, J.; Robertson, A. Concentrations of neonicotinoid insecticides in honey, pollen and honey bees (Apis mellifera L.) in central Saskatchewan, Canada. Chemosphere 2016, 144, 2321–2328. [Google Scholar] [CrossRef] [PubMed]
- David, A.; Botías, C.; Abdul-Sada, A.; Nicholls, E.; Rotheray, E.L.; Hill, E.M.; Goulson, D. Widespread contamination of wildflower and bee-collected pollen with complex mixtures of neonicotinoids and fungicides commonly applied to crops. Environ. Int. 2016, 88, 169–178. [Google Scholar] [CrossRef]
- Sánchez-Hernández, L.; Hernández-Domínguez, D.; Martín, M.T.; Nozal, M.J.; Higes, M.; Bernal Yagüe, J.L. Residues of neonicotinoids and their metabolites in honey and pollen from sunflower and maize seed dressing crops. J. Chromatogr. A 2016, 1428, 220–227. [Google Scholar] [CrossRef]
- Tosi, S.; Costa, C.; Vesco, U.; Quaglia, G.; Guido, G. A 3-year survey of Italian honey bee-collected pollen reveals widespread contamination by agricultural pesticides. Sci. Total Environ. 2018, 615, 208–218. [Google Scholar] [CrossRef]
- Codling, G.; Al Naggar, Y.; Giesy, J.; Robertson, A. Neonicotinoid insecticides in pollen, honey and adult bees in colonies of the European honey bee (Apis mellifera L.) in Egypt. Ecotoxicology 2018, 27, 122–131. [Google Scholar] [CrossRef] [PubMed]
- Tavares, D.A.; Dussaubat, C.; Kretzschmar, A.; Carvalho, S.M.; Silva-Zacarin, E.C.M.; Malaspina, O.; Bérail, G.; Brunet, Je.; Belzunces, L.P. Exposure of larvae to thiamethoxam affects the survival and physiology of the honey bee at post-embryonic stages. Environ. Pollut. 2017, 229, 386–393. [Google Scholar] [CrossRef] [PubMed]
- Felton, G.W.; Summers, C.B. Antioxidant systems in insects. Arch. Insect Biochem. Physiol. 1995, 29, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Hughes, K.A.; Reynolds, R.M. Evolutionary and mechanistic theories of aging. Annu. Rev. Entomol. 2005, 50, 421–445. [Google Scholar] [CrossRef]
- Di Pasquale, G.; Alaux, C.; Le Conte, Y.; Odoux, J.-F.; Pioz, M.; Vaissière, B.E.; Belzunces, L.P.; Decourtye, A. Variations in the availability of pollen resources affect honey bee health. PLoS ONE 2016, 11, e0162818. [Google Scholar] [CrossRef]
- Herbert, E.W.; Hill, D.A. Honey bee nutrition. In The Hive and the Honey Bee; Graham, J.M., Ed.; Dadant & Sons: Hamilton, IL, USA, 2015; pp. 237–268. [Google Scholar]
- Wang, Y.; Kaftanoglu, O.; Brent, C.; Page, R.; Amdam, G. Starvation stress during larval development facilittes an adaptive response in adult worker honey bees (Apis mellifera L.). J. Exp. Boil. 2016, 219, 949–959. [Google Scholar] [CrossRef]
- Requier, F.; Odoux, J.-F.; Henry, M.; Bretagnolle, V. The carry-over effects of pollen shortage decrease the survival of honeybee colonies in farmlands. J. Appl. Ecol. 2017, 54, 1161–1170. [Google Scholar] [CrossRef]
| Replicate | Total Pollen (kg) [Range] 1 | N 2 | Distributed (g) 3 |
|---|---|---|---|
| March | 5.97 [0.77–3.07] | 9 | 664 |
| May | 2.19 [0.45–0.72] | 6 | 366 |
| October | 6.41 [1.05–2.28] | 7 | 915 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mogren, C.L.; Danka, R.G.; Healy, K.B. Larval Pollen Stress Increases Adult Susceptibility to Clothianidin in Honey Bees. Insects 2019, 10, 21. https://doi.org/10.3390/insects10010021
Mogren CL, Danka RG, Healy KB. Larval Pollen Stress Increases Adult Susceptibility to Clothianidin in Honey Bees. Insects. 2019; 10(1):21. https://doi.org/10.3390/insects10010021
Chicago/Turabian StyleMogren, Christina L., Robert G. Danka, and Kristen B. Healy. 2019. "Larval Pollen Stress Increases Adult Susceptibility to Clothianidin in Honey Bees" Insects 10, no. 1: 21. https://doi.org/10.3390/insects10010021
APA StyleMogren, C. L., Danka, R. G., & Healy, K. B. (2019). Larval Pollen Stress Increases Adult Susceptibility to Clothianidin in Honey Bees. Insects, 10(1), 21. https://doi.org/10.3390/insects10010021





