Association of Serum Glucose/Potassium Ratio with Injury Severity and Transfusion Requirements in Traumatic Pelvic Fractures: A Retrospective Cohort Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Setting
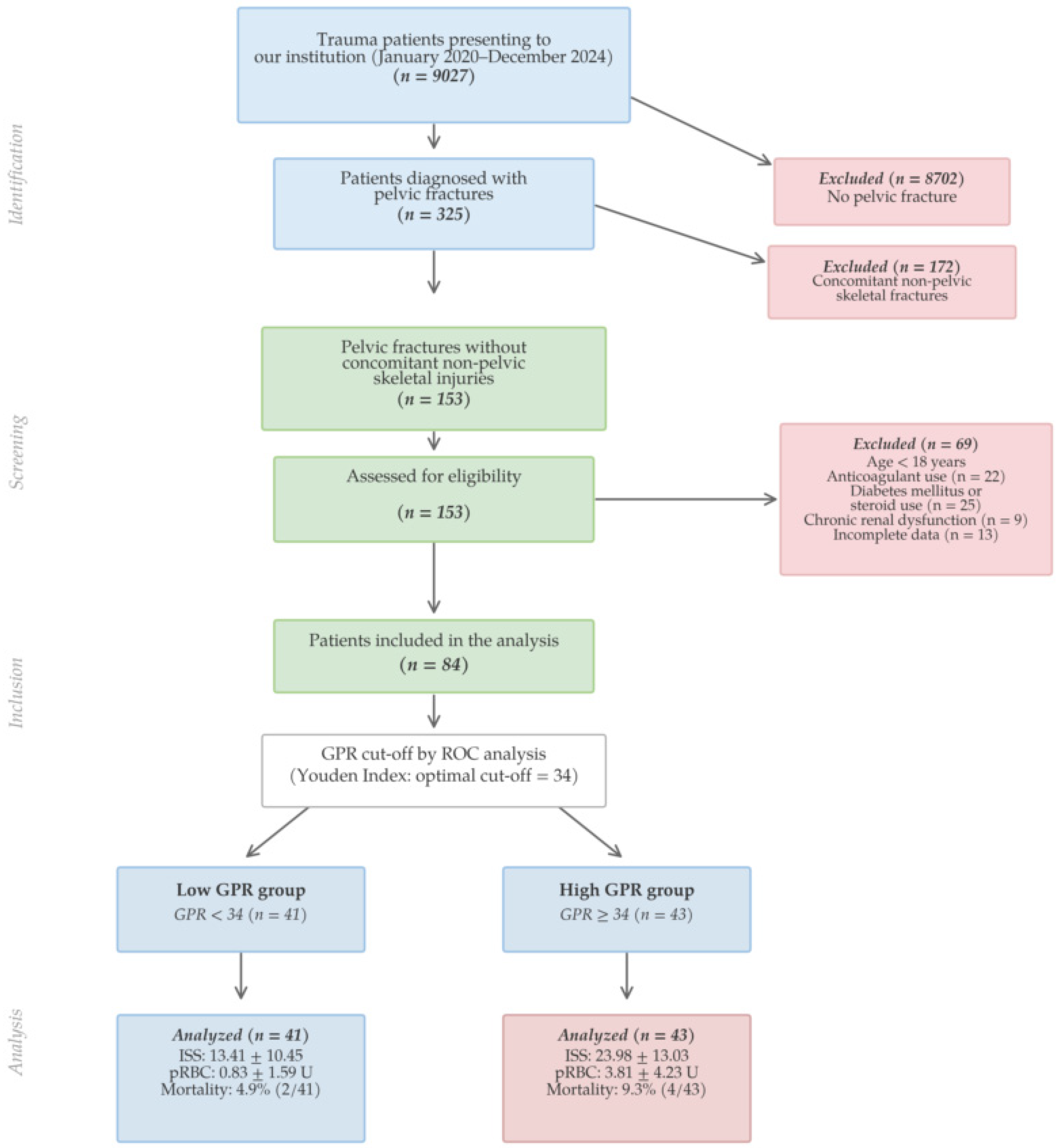
2.2. Participants
2.3. Data Collection
2.4. Statistical Analysis
2.5. Assessment of Bias
3. Results
3.1. Baseline Characteristics
3.2. ROC Analysis and GPR Cut-Off Determination
3.3. Correlation Analysis
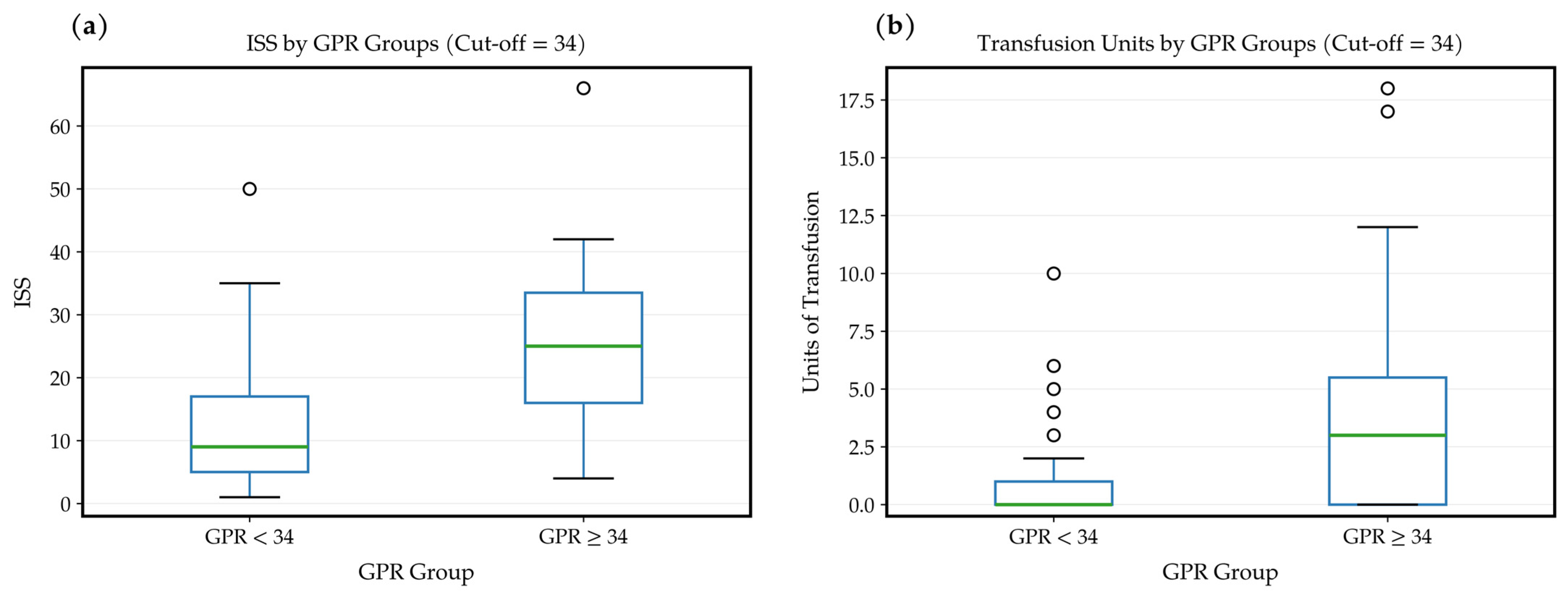
3.4. Between-Group Comparisons
3.5. Multivariable Logistic Regression
3.6. Sensitivity Analyses
4. Discussion
4.1. GPR in the Context of Traumatic Injury
4.2. GPR as a Composite Metabolic Marker
4.3. Incremental Value and Clinical Interpretation of AUC
4.4. Pathophysiological Considerations
4.5. Methodological Considerations
4.6. Limitations and Future Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AIS | Abbreviated Injury Scale |
| AUC | Area under the curve |
| CI | Confidence interval |
| df | Degrees of freedom |
| GCS | Glasgow Coma Scale |
| GPR | Glucose-to-potassium ratio |
| HGB0 | Admission hemoglobin |
| HT | Higher transfusion requirement |
| ICU | Intensive care unit |
| IDI | Integrated discrimination improvement |
| IQR | Interquartile range |
| ISS | Injury Severity Score |
| MT | Massive transfusion |
| NRI | Net reclassification improvement |
| OR | Odds ratio |
| pRBC | Packed red blood cell |
| ROC | Receiver operating characteristic |
| SAH | Subarachnoid hemorrhage |
| SI | Stress index |
| STROBE | Strengthening the Reporting of Observational Studies in Epidemiology |
References
- Adams, J.E.; Davis, G.G.; Alexander, C.B.; Alonso, J.E. Pelvic trauma in rapidly fatal motor vehicle accidents. J. Orthop. Trauma 2003, 17, 406–410. [Google Scholar] [CrossRef]
- Gabbe, B.J.; de Steiger, R.; Esser, M.; Bucknill, A.; Russ, M.K.; Cameron, P.A. Predictors of mortality following severe pelvic ring fracture: Results of a population-based study. Injury 2011, 42, 985–991. [Google Scholar] [CrossRef]
- Heetveld, M.J.; Harris, I.; Schlaphoff, G.; Balogh, Z.; D’Amours, S.K.; Sugrue, M. Hemodynamically unstable pelvic fractures: Recent care and new guidelines. World J. Surg. 2004, 28, 904–909. [Google Scholar] [CrossRef]
- Eakins, J. Blood glucose control in the trauma patient. J. Diabetes Sci. Technol. 2009, 3, 1373–1376. [Google Scholar] [CrossRef]
- Peffer, J.; McLaughlin, C. The correlation of early hyperglycemia with outcomes in adult trauma patients: A systematic review. J. Spec. Oper. Med. 2013, 13, 34–39. [Google Scholar] [CrossRef]
- Rowe, J.W.; Tobin, J.D.; Rosa, R.M.; Andres, R. Effect of experimental potassium deficiency on glucose and insulin metabolism. Metabolism 1980, 29, 498–502. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.J.; Brown, D.C.; Murphy, M.B. Hypokalemia from beta2-receptor stimulation by circulating epinephrine. N. Engl. J. Med. 1983, 309, 1414–1419. [Google Scholar] [CrossRef] [PubMed]
- Fujiki, Y.; Matano, F.; Mizunari, T.; Murai, Y.; Tateyama, K.; Koketsu, K.; Morita, A. Serum glucose/potassium ratio as a clinical risk factor for aneurysmal subarachnoid hemorrhage. J. Neurosurg. 2018, 129, 870–875. [Google Scholar] [CrossRef]
- Wu, X.Y.; Zhuang, Y.K.; Cai, Y.; Dong, X.Q.; Wang, K.Y.; Du, Q.; Yu, W.H. Serum glucose and potassium ratio as a predictive factor for prognosis of acute intracerebral hemorrhage. J. Int. Med. Res. 2021, 49, 3000605211009689. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Yang, C.S.; Shen, L.J.; Lv, Q.W.; Xu, Q.C. Usefulness of serum glucose and potassium ratio as a predictor for 30-day death among patients with severe traumatic brain injury. Clin. Chim. Acta 2020, 506, 166–171. [Google Scholar] [CrossRef]
- Demirtas, E.; Korkmaz, I.; Tekin, Y.K.; Demirtas, E.; Caltekin, I. Assessment of serum glucose/potassium ratio as a predictor for delayed neuropsychiatric syndrome of carbon monoxide poisoning. Hum. Exp. Toxicol. 2021, 40, 207–213. [Google Scholar] [CrossRef] [PubMed]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P.; STROBE Initiative. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. J. Clin. Epidemiol. 2008, 61, 344–349. [Google Scholar] [CrossRef]
- Wei, L.; Chenggao, W.; Juan, Z.; Aiping, L. Massive transfusion prediction in patients with multiple trauma by decision tree: A retrospective analysis. Indian J. Hematol. Blood Transfus. 2021, 37, 302–308. [Google Scholar] [CrossRef]
- Smith, W.; Williams, A.; Agudelo, J.; Shannon, M.; Morgan, S.; Stahel, P.; Moore, E. Early predictors of mortality in hemodynamically unstable pelvis fractures. J. Orthop. Trauma 2007, 21, 31–37. [Google Scholar] [CrossRef]
- Fluss, R.; Faraggi, D.; Reiser, B. Estimation of the Youden Index and its associated cutoff point. Biom. J. 2005, 47, 458–472. [Google Scholar] [CrossRef]
- Pencina, M.J.; D’Agostino, R.B.S.; Steyerberg, E.W. Extensions of net reclassification improvement calculations to measure usefulness of new biomarkers. Stat. Med. 2011, 30, 11–21. [Google Scholar] [CrossRef]
- Rommens, P.M.; Hessman, M.H. Staged reconstruction of pelvic ring disruption: Differences in morbidity, mortality, radiologic results, and functional outcomes between B1, B2/B3, and C-type lesions. J. Orthop. Trauma 2002, 16, 92–98. [Google Scholar] [CrossRef]
- Zhou, W.; Liu, Y.; Wang, Z.; Mao, Z.; Li, M. Serum glucose/potassium ratio as a clinical risk factor for predicting the severity and prognosis of acute traumatic spinal cord injury. BMC Musculoskelet. Disord. 2023, 24, 870. [Google Scholar] [CrossRef] [PubMed]
- Boyuk, F. The predictor potential role of the glucose to potassium ratio in the diagnostic differentiation of massive and non-massive pulmonary embolism. Clin. Appl. Thromb. Hemost. 2022, 28, 10760296221076146. [Google Scholar] [CrossRef]
- Jung, H.M.; Paik, J.H.; Kim, S.Y.; Hong, D.Y. Association of plasma glucose to potassium ratio and mortality after aneurysmal subarachnoid hemorrhage. Front. Neurol. 2021, 12, 661689. [Google Scholar] [CrossRef] [PubMed]
- Katipoğlu, B.; Demirtaş, E. Assessment of serum glucose potassium ratio as a predictor for morbidity and mortality of blunt abdominal trauma. Turkish J. Trauma Emerg. Surg. 2022, 28, 134–139. [Google Scholar] [CrossRef]
- Buz, M.; Ustaalioğlu, İ. Predicting mortality in penetrating thoracic trauma in the emergency department: The prognostic value of the glucose-to-potassium ratio. Turkish J. Trauma Emerg. Surg. 2025, 31, 40–46. [Google Scholar] [CrossRef]
- Unal, A.; Doğan, A. Comparison of glucose/potassium ratio and revised trauma score in predicting mortality in patients with isolated blunt head trauma. Sci. Rep. 2025, 15, 33463. [Google Scholar] [CrossRef] [PubMed]
- Shibata, A.; Matano, F.; Saito, N.; Fujiki, Y.; Matsumoto, H.; Mizunari, T.; Morita, A. Serum glucose-to-potassium ratio as a prognostic predictor for severe traumatic brain injury. J. Nippon Med. Sch. 2021, 88, 342–346. [Google Scholar] [CrossRef]
- Taniguchi, H.; Doi, T.; Abe, T.; Takeuchi, I. Trauma severity associated with stress index in emergency settings: An observational prediction-and-validation study. Acute Med. Surg. 2020, 7, e493. [Google Scholar] [CrossRef]
- Huang, C.Y.; Chou, S.E.; Huang, C.Y.; Tsai, C.H.; Hsu, S.Y.; Hsieh, C.H. Role of the stress index in predicting mortality among patients with traumatic femoral fractures. Diagnostics 2024, 14, 1508. [Google Scholar] [CrossRef]
- Pencina, M.J.; D’Agostino, R.B.; D’Agostino, R.B., Jr.; Vasan, R.S. Evaluating the Added Predictive Ability of a New Marker: From Area under the ROC Curve to Reclassification and Beyond. Stat. Med. 2008, 27, 157–172. [Google Scholar] [CrossRef]
- Sharma, B.; Hasan, M.; Govindarajulu, U.S.; Agriantonis, G.; Bhatia, N.D.; Dave, J.; Mestre, J.; Arora, S.; Bhatti, S.; Shafaee, Z.; et al. Kalemia Significantly Influences Clinical Outcomes in Patients with Severe Traumatic Brain Injury (TBI). Diagnostics 2025, 15, 1878. [Google Scholar] [CrossRef]
- Ramadanov, N.; Zabler, S. Ramadanov–Zabler Safe Zone for Sacroiliac Screw Placement: A CT-Based Computational Pilot Study. J. Clin. Med. 2025, 14, 3567. [Google Scholar] [CrossRef] [PubMed]
- Vadan, I.-M.; Grad, D.; Strilciuc, S.; Blesneag, A.V.; Michalak, M.; Vacaras, V.; Stan, A.; Muresanu, D.F. Evaluation of Traumatic Brain Injury Severity Using the Abbreviated Injury Scale and the Injury Severity Score: A Retrospective Study in Two Eastern European Centers. J. Clin. Med. 2025, 14, 6259. [Google Scholar] [CrossRef] [PubMed]

| Variables | Total (n = 84) |
|---|---|
| Age, years (mean ± SD) | 50.04 ± 20.52 |
| Sex, n (%) | |
| Female | 34 (40.5%) |
| Male | 50 (59.5%) |
| Mechanism of injury, n (%) | |
| Traffic accident | 33 (39.3%) |
| Fall from height (≥1 m or ≥5 steps) | 35 (41.7%) |
| Crush/compression | 2 (2.4%) |
| Other (agricultural accident, heavy object) | 14 (16.7%) |
| Mechanic energy | |
| Low | 70 (83.3%) |
| High | 14 (16.7%) |
| ISS (%) | |
| 1–15 | 35 (41.7%) |
| 16–24 | 20 (23.8%) |
| ≥25 | 29 (34.5%) |
| Unstable pelvis, n (%) | 52 (61.9%) |
| Tile classification, n (%) | |
| Type A | 32 (38.1%) |
| Type B | 50 (59.5%) |
| Type C | 2 (2.4%) |
| Treatment | |
| Conservative | 48 (57.1%) |
| Surgery | 36 (42.9%) |
| Blood transfusion (units, pRBCs) | |
| Low (<4 units/24 h) | 61 (72.6%) |
| High (≥4 units/24 h) | 23 (27.4%) |
| Hospitalization duration (days) | 7 (4–14) |
| ICU stay (days) | 1 (0–5) |
| Mortality, n (%) | 6 (7.1%) |
| Low GPR (<34), n = 41 | High GPR (≥34), n = 43 | p Value | |
|---|---|---|---|
| ISS | 9 (5–17) | 25 (16–34) | <0.001 * |
| Blood transfusion, units | 0 (0–1) | 3 (0–6) | <0.001 * |
| High transfusion (≥4 units), n (%) | 5 (12.2%) | 18 (41.9%) | 0.003 ** |
| Hospitalization duration (days) | 5 (3–10) | 9 (6–18) | 0.007 * |
| ICU stay (days) | 0 (0–1) | 3 (0–6) | <0.001 * |
| Mortality | 2 (4.9%) | 4 (9.3%) | 0.676 ** |
| Variable | Univariable | Multivariable | ||||
|---|---|---|---|---|---|---|
| OR | 95% CI | p | OR | 95% CI | p | |
| GPR (per unit) | 1.059 | 1.015–1.104 | 0.008 | 1.030 | 0.985–1.076 | 0.194 |
| ISS (per unit) | 1.135 | 1.070–1.203 | <0.001 | 1.128 | 1.062–1.198 | <0.001 |
| Metric | Value | 95% CI | p |
|---|---|---|---|
| AUC—ISS alone | 0.849 | — | — |
| AUC—ISS + GPR | 0.857 | — | — |
| ΔAUC | 0.009 | — | 0.566 |
| Optimism-corrected AUC (ISS + GPR) | 0.847 | — | — |
| Category-free NRI | 0.627 | 0.141–1.088 | 0.009 |
| IDI | 0.017 | −0.014 to 0.048 | 0.290 |
| Threshold | Events, n (%) | GPR AUC | Cut-Off | Sensitivity | Specificity | ISS + GPR AUC |
|---|---|---|---|---|---|---|
| ≥4 units (primary) | 23 (27.4%) | 0.730 | 34.0 | 78.8% | 59.0% | 0.857 |
| ≥6 units | 14 (16.7%) | 0.709 | 39.3 | 78.6% | 67.1% | 0.864 |
| ≥10 units | 4 (4.8%) | 0.684 | 46.0 | 75.0% | 75.0% | — † |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Sahin, A.A.; Özbilen, Y.E.; Akalın, Ç. Association of Serum Glucose/Potassium Ratio with Injury Severity and Transfusion Requirements in Traumatic Pelvic Fractures: A Retrospective Cohort Study. Diagnostics 2026, 16, 939. https://doi.org/10.3390/diagnostics16060939
Sahin AA, Özbilen YE, Akalın Ç. Association of Serum Glucose/Potassium Ratio with Injury Severity and Transfusion Requirements in Traumatic Pelvic Fractures: A Retrospective Cohort Study. Diagnostics. 2026; 16(6):939. https://doi.org/10.3390/diagnostics16060939
Chicago/Turabian StyleSahin, Abdullah Alper, Yunus Emre Özbilen, and Çağrı Akalın. 2026. "Association of Serum Glucose/Potassium Ratio with Injury Severity and Transfusion Requirements in Traumatic Pelvic Fractures: A Retrospective Cohort Study" Diagnostics 16, no. 6: 939. https://doi.org/10.3390/diagnostics16060939
APA StyleSahin, A. A., Özbilen, Y. E., & Akalın, Ç. (2026). Association of Serum Glucose/Potassium Ratio with Injury Severity and Transfusion Requirements in Traumatic Pelvic Fractures: A Retrospective Cohort Study. Diagnostics, 16(6), 939. https://doi.org/10.3390/diagnostics16060939






