Radiological Findings Increased the Successful of COVID-19 Diagnosis in Hospitalized Patients Suspected of Respiratory Viral Infection but with a Negative First SARS-CoV-2 RT-PCR Result
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Clinical and Biochemical Data at the Hospital Admission
2.3. Clinical Criteria Used to Classify of Patients According to COVID-19 Severity
2.4. Chest CT Scan
2.5. SARS-CoV-2 RT-PCR Screening
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pascarella, G.; Strumia, A.; Piliego, C.; Bruno, F.; Del Buono, R.; Costa, F.; Scarlata, S.; Agro, F.E. COVID-19 diagnosis and management: A comprehensive review. J. Intern. Med. 2020, 288, 192–206. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Weekly Epidemiological Update on COVID-19. 28 December 2021. Available online: https://www.who.int/publications/m/item/weekly-epidemiological-update-on-covid-19---28-december-2021 (accessed on 1 December 2021).
- Rahimi, A.; Mirzazadeh, A.; Tavakolpour, S. Genetics and genomics of SARS-CoV-2: A review of the literature with the special focus on genetic diversity and SARS-CoV-2 genome detection. Genomics 2021, 113, 1221–1232. [Google Scholar] [CrossRef]
- Salud, S.D. Datos Abiertos Dirección General de Epidemiología. 2022. Available online: http://www.gob.mx/salud/documentos/datos-abiertos-152127 (accessed on 1 January 2022).
- COVID-19 Tablero México. 2020. Available online: https://datos.covid-19.conacyt.mx/ (accessed on 1 January 2022).
- Parasher, A. COVID-19: Current understanding of its Pathophysiology, Clinical presentation and Treatment. Postgrad. Med. J. 2021, 97, 312–320. [Google Scholar] [CrossRef]
- Kumar, M.; Al Khodor, S. Pathophysiology and treatment strategies for COVID-19. J. Transl. Med. 2020, 18, 353. [Google Scholar] [CrossRef] [PubMed]
- Mousavizadeh, L.; Ghasemi, S. Genotype and phenotype of COVID-19: Their roles in pathogenesis. J. Microbiol. Immunol. Infect. 2021, 54, 159–163. [Google Scholar] [CrossRef] [PubMed]
- Gobierno de México. Guía Clínica para el Tratamiento de la COVID-19 en Mexico. Consenso Interinstitucional. 2021. Available online: https://coronavirus.gob.mx/wp-content/uploads/2021/08/GuiaTx_COVID19_ConsensoInterinstitucional_2021.08.03.pdf (accessed on 1 January 2022).
- Ismail, A.A. SARS-CoV-2 (Covid-19): A short update on molecular biochemistry, pathology, diagnosis and therapeutic strategies. Ann. Clin. Biochem. 2022, 59, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Li, L.; Xu, M.; Wu, J.; Luo, D.; Zhu, Y.; Li, B.; Song, X.; Zhou, X. Prognostic value of interleukin-6, C-reactive protein, and procalcitonin in patients with COVID-19. J. Clin. Virol. Off. Publ. Pan Am. Soc. Clin. Virol. 2020, 127, 104370. [Google Scholar] [CrossRef] [PubMed]
- Lazzaroni, M.G.; Piantoni, S.; Masneri, S.; Garrafa, E.; Martini, G.; Tincani, A.; Andreoli, L.; Franceschini, F. Coagulation dysfunction in COVID-19: The interplay between inflammation, viral infection and the coagulation system. Blood Rev. 2021, 46, 100745. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Fierro, M.L.; Rios-Jasso, J.; Garza-Veloz, I.; Reyes-Veyna, L.; Cerda-Luna, R.M.; Duque-Jara, I.; Galvan-Jimenez, M.; Ramirez-Hernandez, L.A.; Morales-Esquivel, A.; Ortiz-Castro, Y.; et al. The role of close contacts of COVID-19 patients in the SARS-CoV-2 transmission: An emphasis on the percentage of nonevaluated positivity in Mexico. Am. J. Infect. Control 2021, 49, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Fierro, M.L.; Diaz-Lozano, M.; Alvarez-Zuniga, C.; Ramirez-Hernandez, L.A.; Araujo-Espino, R.; Trejo-Ortiz, P.M.; Mollinedo-Montano, F.E.; Ortiz-Castro, Y.; Vazquez-Reyes, S.; Velasco-Elizondo, P.; et al. Population-Based COVID-19 Screening in Mexico: Assessment of Symptoms and Their Weighting in Predicting SARS-CoV-2 Infection. Medicina 2021, 57, 363. [Google Scholar] [CrossRef] [PubMed]
- Parra-Ortega, I.; Vilchis-Ordonez, A.; Lopez-Martinez, B.; Angeles-Floriano, T. Analytical recommendations for SARS-CoV-2 identification by RT-PCR in pediatric patients. Bol. Med. Del Hosp. Infant. De Mex. 2021, 78, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Corman Victor, B.T.; Brünink, S.; Drosten, C.; Landt, O.; Koopmans, M.; Zambon, M. Diagnostic Detection of Wuhan Coronavirus 2019 by Real-Time RT-PCR; WHO: Geneva, Switzerland, 2020; Volume 12. [Google Scholar]
- Kanji, J.N.; Zelyas, N.; MacDonald, C.; Pabbaraju, K.; Khan, M.N.; Prasad, A.; Hu, J.; Diggle, M.; Berenger, B.M.; Tipples, G. False negative rate of COVID-19 PCR testing: A discordant testing analysis. Virol. J. 2021, 18, 13. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, W.; Simpson, S.L.; Bertsch, P.M.; Bibby, K.; Bivins, A.; Blackall, L.L.; Bofill-Mas, S.; Bosch, A.; Brandao, J.; Choi, P.M.; et al. Minimizing errors in RT-PCR detection and quantification of SARS-CoV-2 RNA for wastewater surveillance. Sci. Total Environ. 2022, 805, 149877. [Google Scholar] [CrossRef]
- Martinez, R.M. Clinical Samples for SARS-CoV-2 Detection: Review of the Early Literature. Clin. Microbiol. Newsl. 2020, 42, 121–127. [Google Scholar] [CrossRef]
- Shen, L.; Cui, S.; Zhang, D.; Lin, C.; Chen, L.; Wang, Q. Comparison of four commercial RT-PCR diagnostic kits for COVID-19 in China. J. Clin. Lab. Anal. 2021, 35, e23605. [Google Scholar] [CrossRef]
- Lippi, G.; Simundic, A.M.; Plebani, M. Potential preanalytical and analytical vulnerabilities in the laboratory diagnosis of coronavirus disease 2019 (COVID-19). Clin. Chem. Lab. Med. 2020, 58, 1070–1076. [Google Scholar] [CrossRef] [Green Version]
- Wiseman, J.; D’Amico, T.A.; Zawadzka, S.; Anyimadu, H. False negative SARS-CoV-2 PCR—A case report and literature review. Respir. Med. Case Rep. 2020, 31, 101140. [Google Scholar] [CrossRef]
- Gobierno de México. Algoritmos Interinos para la Atención del COVID-19. 2020. Available online: http://educacionensalud.imss.gob.mx/es/system/files/Algoritmos_interinos_COVID19_CTEC.pdf (accessed on 1 January 2022).
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.H.; Ou, C.Q.; He, J.X.; Liu, L.; Shan, H.; Lei, C.L.; Hui, D.S.C.; et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef] [PubMed]
- Rodilla, E.; Saura, A.; Jimenez, I.; Mendizabal, A.; Pineda-Cantero, A.; Lorenzo-Hernandez, E.; Fidalgo-Montero, M.D.P.; Lopez-Cuervo, J.F.; Gil-Sanchez, R.; Rabadan-Pejenaute, E.; et al. Association of Hypertension with All-Cause Mortality among Hospitalized Patients with COVID-19. J. Clin. Med. 2020, 9, 3136. [Google Scholar] [CrossRef] [PubMed]
- Salud, S.d. Lineamientos para la Atención de Pacientes por COVID-19. 2020. Available online: http://cvoed.imss.gob.mx/wp-content/uploads/2020/02/Linemaineto-cl%C3%Adnico-COVID-19-CCINSHAE-14feb2020.pdf.pdf.pdf.pdf (accessed on 1 January 2022).
- Mao, L.; Wang, M.; Chen, S.; He, Q.; Chang, J.; Hong, C.; Zhou, Y.; Wang, D.; Li, Y.; Jin, H.; et al. Anosmia and Ageusia: Common Findings in COVID-19 Patients. Laryngoscope 2020, 130, 1787. [Google Scholar] [CrossRef] [Green Version]
- Le Joncour, A.; Biard, L.; Vautier, M.; Bugaut, H.; Mekinian, A.; Maalouf, G.; Vieira, M.; Marcelin, A.G.; Rosenzwajg, M.; Klatzmann, D.; et al. Neutrophil-Platelet and Monocyte-Platelet Aggregates in COVID-19 Patients. Thromb. Haemost. 2020, 120, 1733–1735. [Google Scholar] [CrossRef] [PubMed]
- Ghebrehiwet, B.; Peerschke, E.I. Complement and coagulation: Key triggers of COVID-19-induced multiorgan pathology. J. Clin. Investig. 2020, 130, 5674–5676. [Google Scholar] [CrossRef] [PubMed]
- Gibson, P.G.; Qin, L.; Puah, S.H. COVID-19 acute respiratory distress syndrome (ARDS): Clinical features and differences from typical pre-COVID-19 ARDS. Med. J. Aust. 2020, 213, 54–56 e51. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Zhong, Z.; Zhao, W.; Zheng, C.; Wang, F.; Liu, J. Chest CT for Typical 2019-nCoV Pneumonia: Relationship to Negative RT-PCR Testing. Radiology 2020, 296, E41–E45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sanchez-Oro, R.; Torres Nuez, J.; Martinez-Sanz, G. Radiological findings for diagnosis of SARS-CoV-2 pneumonia (COVID-19). Med. Clin. 2020, 155, 36–40. [Google Scholar] [CrossRef]
- Ye, Z.; Zhang, Y.; Wang, Y.; Huang, Z.; Song, B. Chest CT manifestations of new coronavirus disease 2019 (COVID-19): A pictorial review. Eur. Radiol. 2020, 30, 4381–4389. [Google Scholar] [CrossRef] [Green Version]
- Wong, H.Y.F.; Lam, H.Y.S.; Fong, A.H.; Leung, S.T.; Chin, T.W.; Lo, C.S.Y.; Lui, M.M.; Lee, J.C.Y.; Chiu, K.W.; Chung, T.W.; et al. Frequency and Distribution of Chest Radiographic Findings in Patients Positive for COVID-19. Radiology 2020, 296, E72–E78. [Google Scholar] [CrossRef] [Green Version]
- Prokop, M.; van Everdingen, W.; van Rees Vellinga, T.; Quarles van Ufford, H.; Stoger, L.; Beenen, L.; Geurts, B.; Gietema, H.; Krdzalic, J.; Schaefer-Prokop, C.; et al. CO-RADS: A Categorical CT Assessment Scheme for Patients Suspected of Having COVID-19-Definition and Evaluation. Radiology 2020, 296, E97–E104. [Google Scholar] [CrossRef]
- Gupta, R. SARS-CoV-2 Omicron spike mediated immune escape and tropism shift. Res. Sq. 2022. [Google Scholar] [CrossRef]
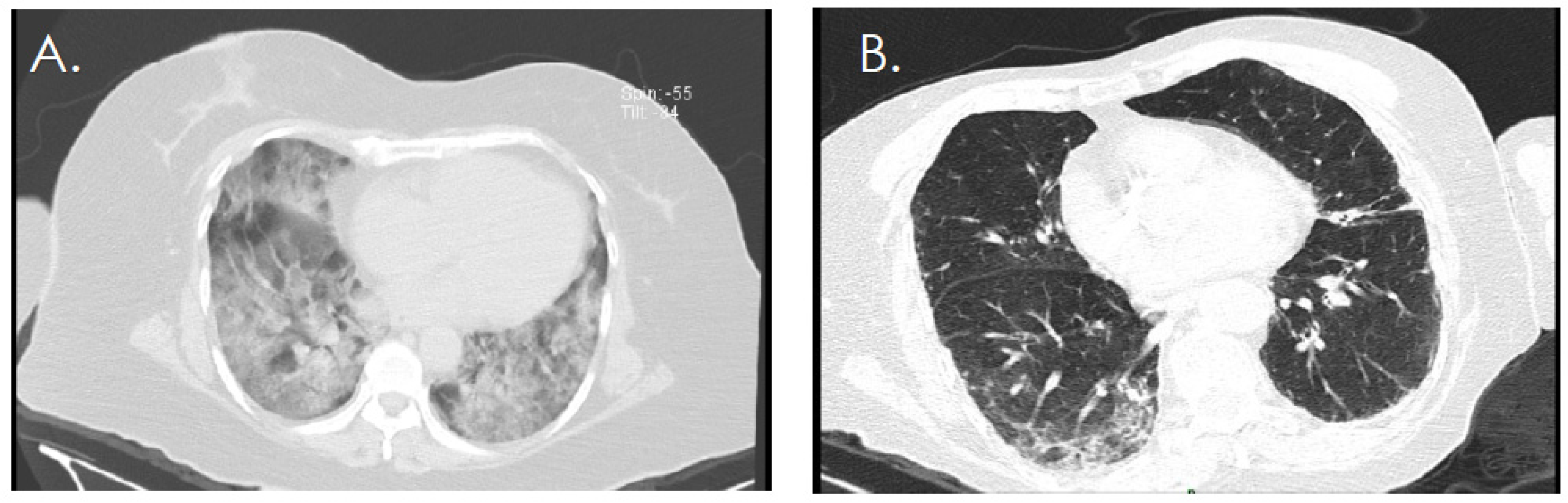
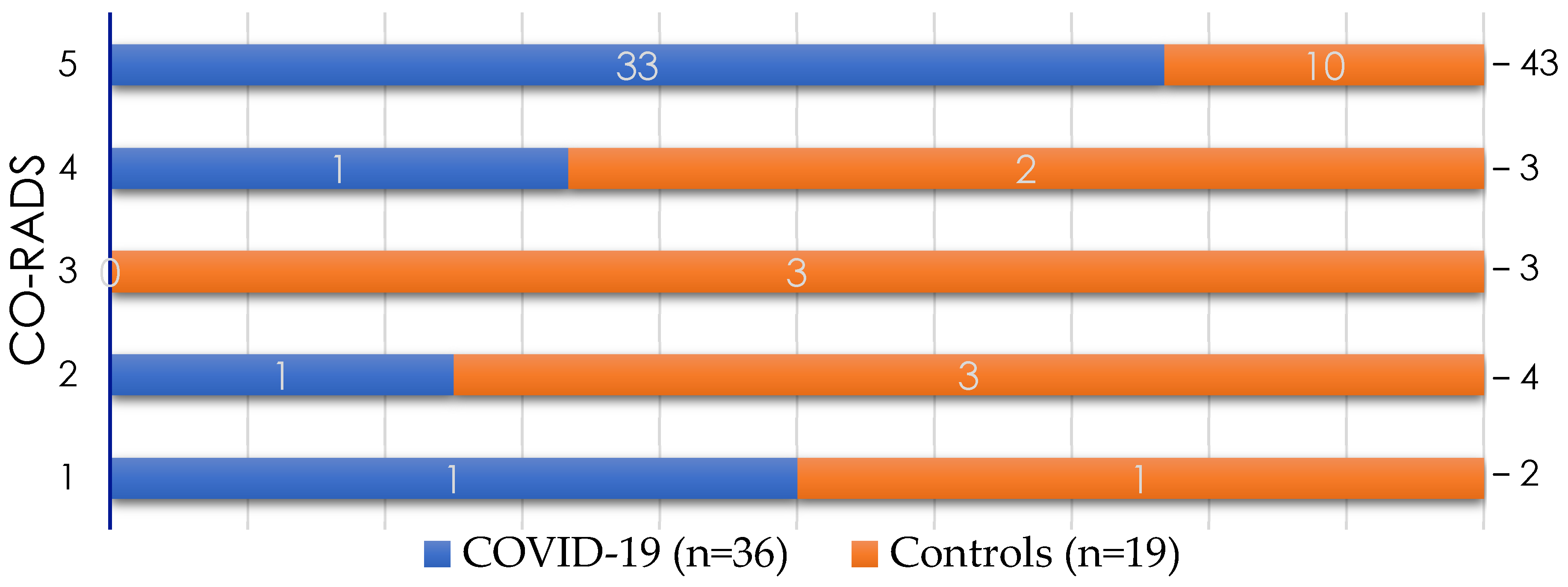
| Variable | General Population (n = 55) | Study Group | p-Value | |
|---|---|---|---|---|
| COVID-19 (n = 36) | Controls (n = 19) | |||
| Age (years) | 57.3 ± 16.77 | 57.94 ± 15.94 | 56.11 ± 18.64 | 0.69 |
| Sex female | 24 (43.6) | 14 (38.8) | 10 (52.6) | 0.39 |
| Comorbidities (n, %) | ||||
| Hypertension | 22 (40) | 12 (33.3) | 10 (52.6) | 0.17 |
| Diabetes | 18 (32.72) | 13 (36.1) | 5 (26.3) | 0.46 |
| Obesity | 19 (34.54) | 13 (36.1) | 6 (66.6) | 0.74 |
| Other comorbidity | 26 (47.27) | 15 (41.7) | 11 (57.8) | 0.25 |
| Symptoms * (n, %) | ||||
| Cough | 33 (60) | 21 (58) | 12 (63) | 0.73 |
| Dyspnoea | 53 (96.36) | 36 (100) | 17 (89) | 0.12 |
| Chest pain | 14 (25.45) | 10 (27.7) | 4 (21) | 0.75 |
| Fever | 38 (69.09) | 26 (72.2) | 12 (63.1) | 0.49 |
| Anosmia | 4 (7.27) | 4 (11.1) | 0 | 0.29 |
| Dysgeusia | 5 (9.09) | 4 (11) | 1 (5.2) | 0.65 |
| Asthenia | 34 (61.1) | 25 (69.4) | 9 (47.3) | 0.11 |
| Myalgias | 16 (29.09) | 11 (39.5) | 5 (26.3) | 0.74 |
| Arthralgias | 16 (29.09) | 13 (36.1) | 3 (15.7) | 0.12 |
| Diarrhea | 8 (14.54) | 4 (11) | 4 (21) | 0.43 |
| Headache | 14 (25.45) | 11 (30.5) | 3 (15.7) | 0.33 |
| Sickness | 12 (21.81) | 9 (25) | 3 (15.7) | 0.51 |
| Supplementary oxygen ** (n, %) | ||||
| Nasal cannula | 47 (85.45) | 30 (83.3) | 17 (89.5) | 0.7 |
| Facemask | 50 (90.90) | 33 (91.7) | 17 (89.5) | 0.78 |
| Mechanic Ventilation | 10 (18.18) | 6 (16.7) | 4 (21.1) | 0.72 |
| COVID-19 testing moment (days) | ||||
| First COVID-19 test | 10.27 ± 36.46 | 9.97 ± 5.10 | 10.84 ± 7.63 | 0.94 |
| Second COVID-19 test | 13.65 ± 6.09 | 13.72 ± 5.38 | 13.53 ± 7.43 | 0.6 |
| Cq value (mean ± SD) | ||||
| ORF1 gene | - | 29.24 ± 4.44 | - | - |
| N gene | - | 28.52 ± 5.08 | - | - |
| S gene | - | 29.17 ± 4.31 | - | - |
| Laboratory Finding | General Population (n = 55) | Study Group | p-Value | |
|---|---|---|---|---|
| COVID-19 (n = 36) | Controls (n = 19) | |||
| Hematic biometry (mean ± SD) | ||||
| Hemoglobin (g/dL) | 13.54 ± 3.11 | 13.97 ± 2.73 | 12.73 ± 3.69 | 0.20 |
| Platelets (103/dL) | 267.4 ± 117.7 | 296.5 ± 105.8 | 212.4 ± 122.1 | 0.01 * |
| Leucocytes (counts/dL) | 10,733 ± 4853 | 10,797 ± 4608 | 10,611 ± 5418 | 0.85 |
| Lymphocytes (counts/dL) | 1069 ± 620 | 1147 ± 711 | 922 ± 367 | 0.29 |
| Neutrophils (counts/dL) | 9261 ± 4462 | 9347 ± 4175 | 9098 ± 5079 | 0.85 |
| Blood chemistry (mean ± SD) | ||||
| Prothrombin time (s) | 16.08 ± 2.77 | 15.77 ± 2.4 | 16.63 ± 3.35 | 0.33 |
| Partial thromboplastin time (s) | 36.80 ± 11.10 | 36.1 ± 11.6 | 38.06 ± 10.36 | 0.26 |
| Glucose (mg/dL) | 152.58 ± 84.77 | 167.58 ± 93.09 | 124.16 ± 58.37 | 0.03 * |
| Creatinine (mg/dL) | 1.91 ± 4.67 | 1.99 ± 5.53 | 1.76 ± 2.44 | 0.42 |
| Blood urea nitrogen (mg/dL) | 28.15 ± 22.71 | 27.97 ± 25.09 | 28.511± 7.99 | 0.37 |
| Direct bilirrubin (mg/dL) | 0.10 ± 0.79 | 0.34 ± 0.23 | 0.82 ±1.29 | 0.15 |
| Albumin (mg/dL) | 3.10 ± 0.48 | 3.12 ± 0.47 | 3.07 ± 0.51 | 0.75 |
| Arterial blood gas test (mean ± SD) | ||||
| pH | 7.42 ± 0.08 | 7.43 ± 0.06 | 7.41 ± 0.10 | 0.61 |
| pO2 (mmHg) | 72.96 ± 35.13 | 68.50 ± 31.69 | 81.42 ± 40.43 | 0.04 * |
| pCO2 (mmHg) | 33.13 ± 8.95 | 32.39 ±7.47 | 34.53 ± 11.34 | 0.51 |
| HCO3 (mmol/L) | 22.22 ± 3.96 | 22.29 ± 4.30 | 22.08 ± 3.3 | 0.84 |
| Inflammatory markers (mean ± SD) | ||||
| Fibrinogen (mg/dL) | 573.97 ± 193.28 | 615.91± 186.34 | 512.47 ± 192.77 | 0.84 |
| D-dimer (pg/mL) | 4.07 ± 6.21 | 5.24 ± 7.42 | 3.23 ± 5.28 | 0.003 * |
| Lactate dehydrogenase (mg/dL) | 639.03 ± 402.47 | 620.79 ± 405.07 | 675.53 ± 407.02 | 0.89 |
| Ferritin (ng/mL) | 765.93 ± 744.26 | 692.85 ± 576.70 | 912.52 ± 1019.16 | 0.41 |
| Procalcitonin (mg/dL) | 2.41 ± 8.96 | 0.84 ± 1.17 | 4.88 ± 14.26 | 0.005 * |
| C-reactive protein | 16.55 ± 11.62 | 16.26 ± 9.32 | 17.05 ± 15.06 | 0.73 |
| Troponin (ng/mL) | 1.03 ± 4.15 | 0.31± 0.44 | 1.81 ± 5.98 | 0.42 |
| Creatine kinase (mg/dL) | 413.08 ± 1472 | 567.41 ± 1922.82 | 200.9 ± 295.2 | 0.56 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martinez-Fierro, M.L.; González-Fuentes, C.; Cid-Guerrero, D.; González Delgado, S.; Carrillo-Martínez, S.; Gutierrez-Vela, E.F.; Calzada-Luévano, J.Y.; Rocha-Pizaña, M.R.; Martínez-Rendón, J.; Castañeda-López, M.E.; et al. Radiological Findings Increased the Successful of COVID-19 Diagnosis in Hospitalized Patients Suspected of Respiratory Viral Infection but with a Negative First SARS-CoV-2 RT-PCR Result. Diagnostics 2022, 12, 687. https://doi.org/10.3390/diagnostics12030687
Martinez-Fierro ML, González-Fuentes C, Cid-Guerrero D, González Delgado S, Carrillo-Martínez S, Gutierrez-Vela EF, Calzada-Luévano JY, Rocha-Pizaña MR, Martínez-Rendón J, Castañeda-López ME, et al. Radiological Findings Increased the Successful of COVID-19 Diagnosis in Hospitalized Patients Suspected of Respiratory Viral Infection but with a Negative First SARS-CoV-2 RT-PCR Result. Diagnostics. 2022; 12(3):687. https://doi.org/10.3390/diagnostics12030687
Chicago/Turabian StyleMartinez-Fierro, Margarita L, Carolina González-Fuentes, Dagoberto Cid-Guerrero, Samantha González Delgado, Santiago Carrillo-Martínez, Edgar Fernando Gutierrez-Vela, Juan Yadid Calzada-Luévano, Maria R. Rocha-Pizaña, Jacqueline Martínez-Rendón, Maria E. Castañeda-López, and et al. 2022. "Radiological Findings Increased the Successful of COVID-19 Diagnosis in Hospitalized Patients Suspected of Respiratory Viral Infection but with a Negative First SARS-CoV-2 RT-PCR Result" Diagnostics 12, no. 3: 687. https://doi.org/10.3390/diagnostics12030687
APA StyleMartinez-Fierro, M. L., González-Fuentes, C., Cid-Guerrero, D., González Delgado, S., Carrillo-Martínez, S., Gutierrez-Vela, E. F., Calzada-Luévano, J. Y., Rocha-Pizaña, M. R., Martínez-Rendón, J., Castañeda-López, M. E., & Garza-Veloz, I. (2022). Radiological Findings Increased the Successful of COVID-19 Diagnosis in Hospitalized Patients Suspected of Respiratory Viral Infection but with a Negative First SARS-CoV-2 RT-PCR Result. Diagnostics, 12(3), 687. https://doi.org/10.3390/diagnostics12030687







