The Composition and Physical Properties of Clots in COVID-19 Pathology
Abstract
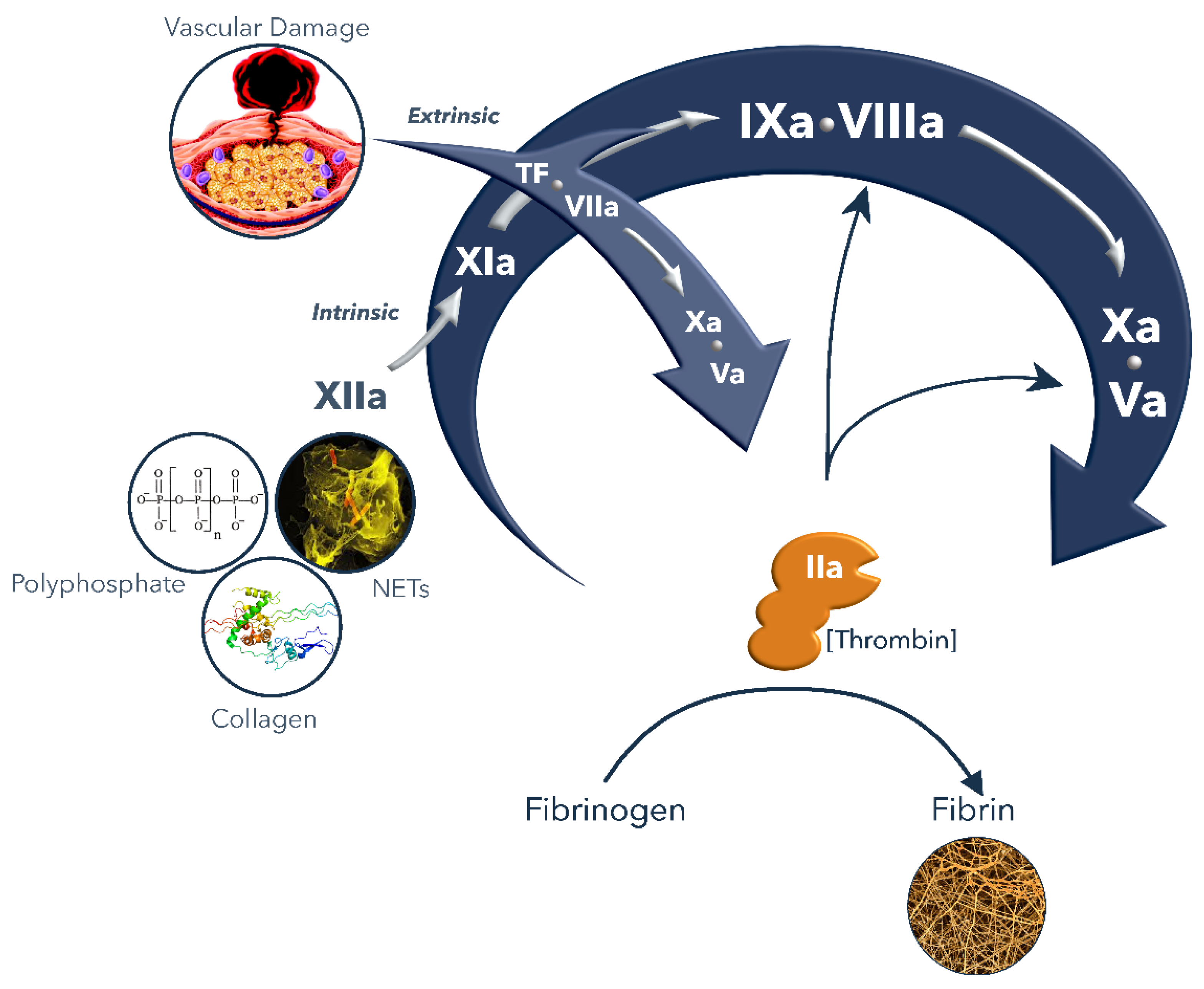
:1. Hemostasis
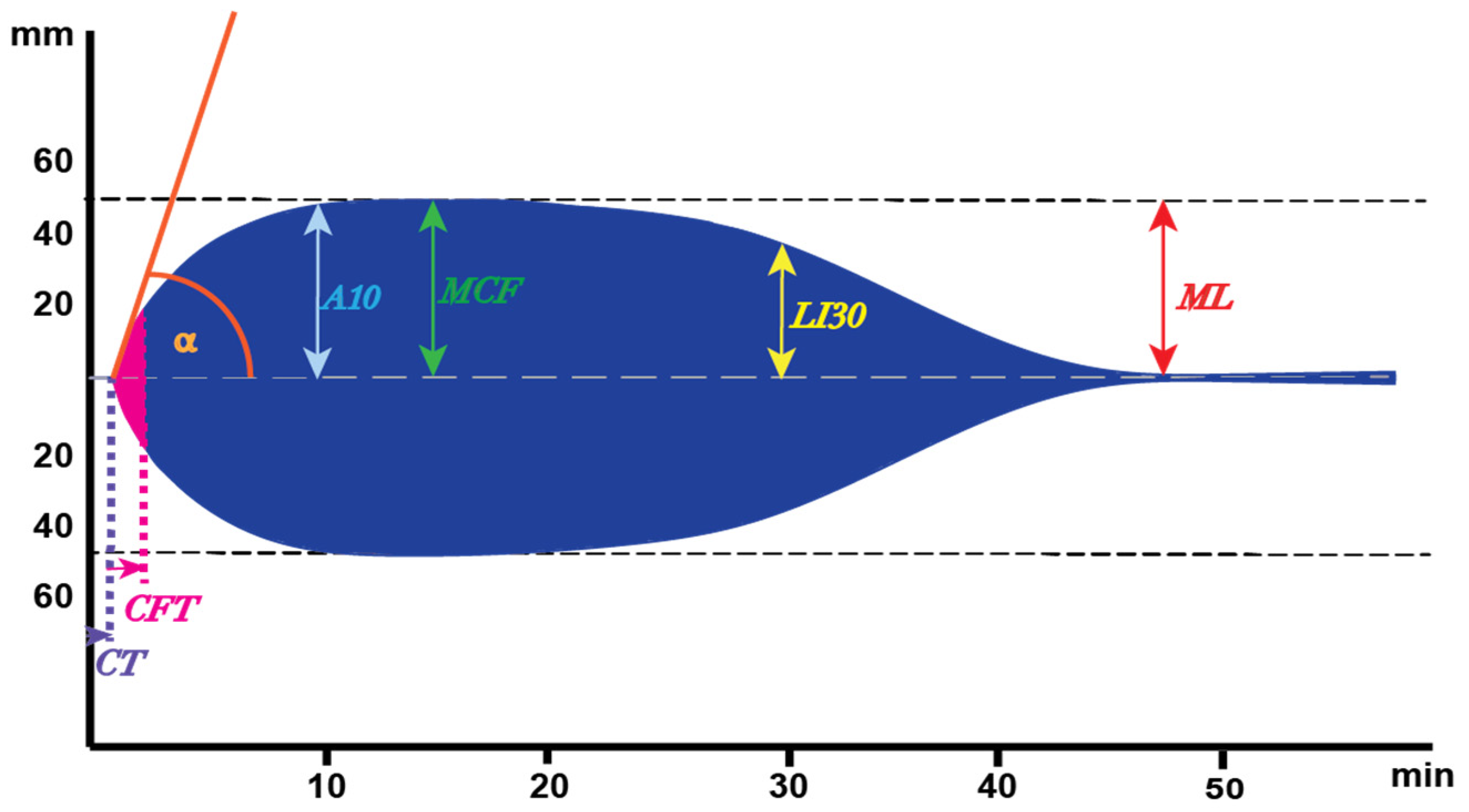
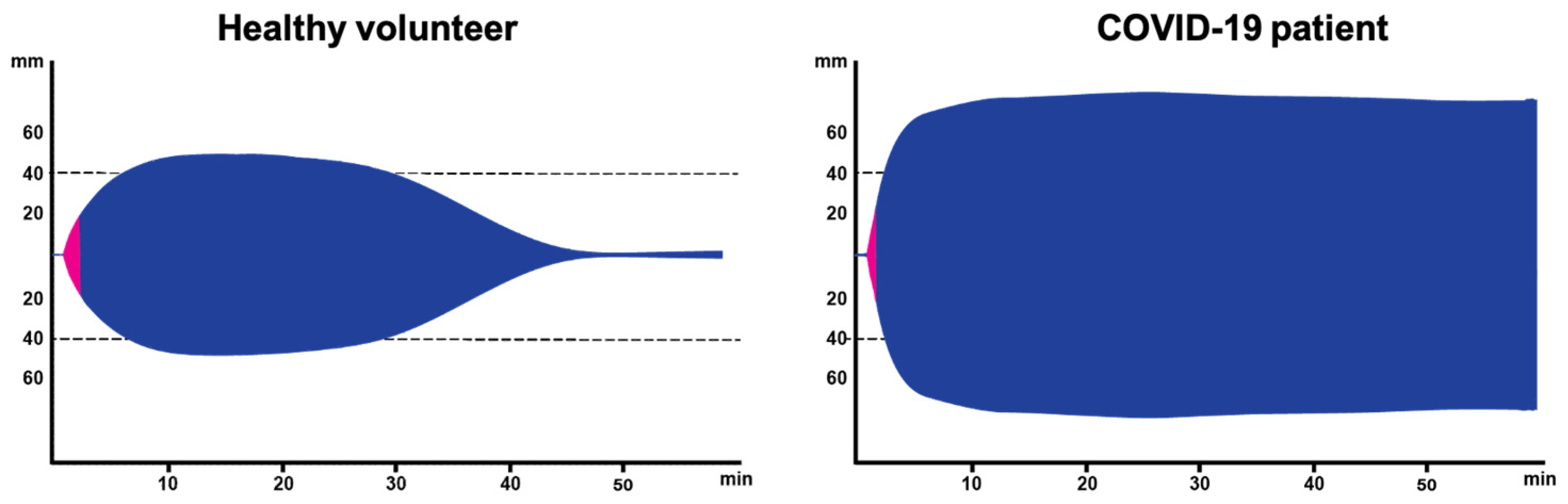
2. ROtational ThromboElastoMetry (ROTEM) and Interpretation
3. Coagulopathy in COVID-19
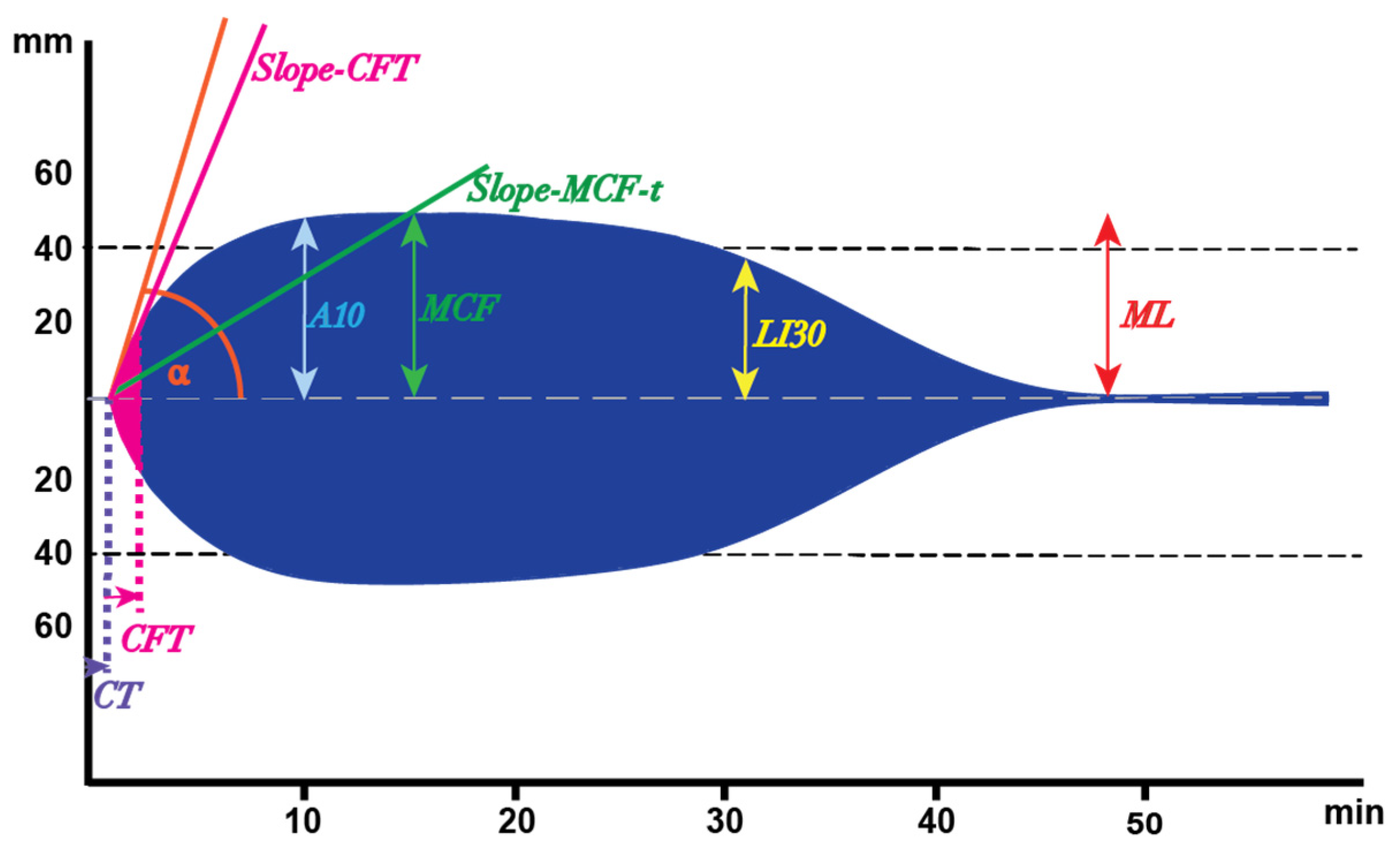
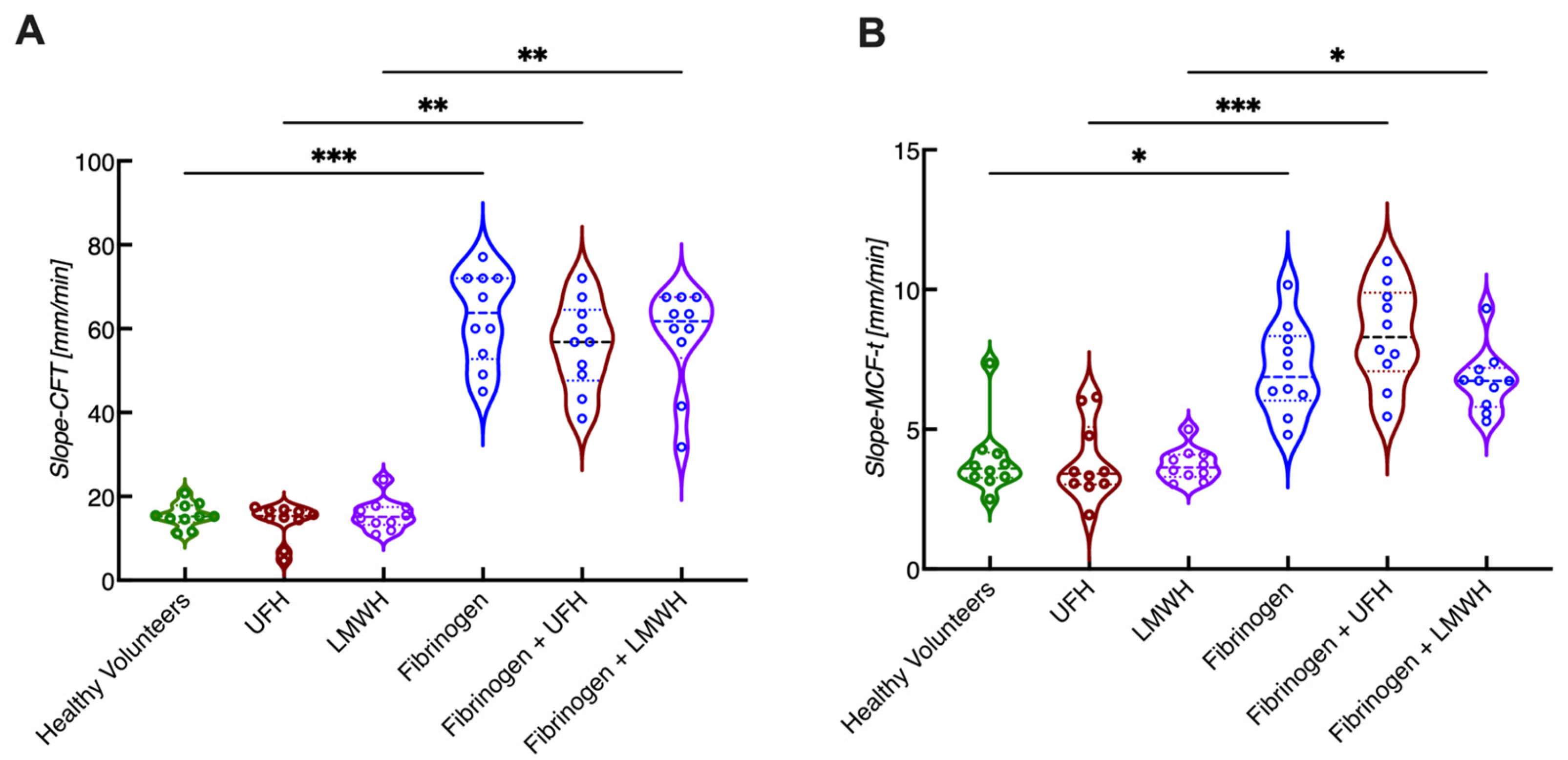
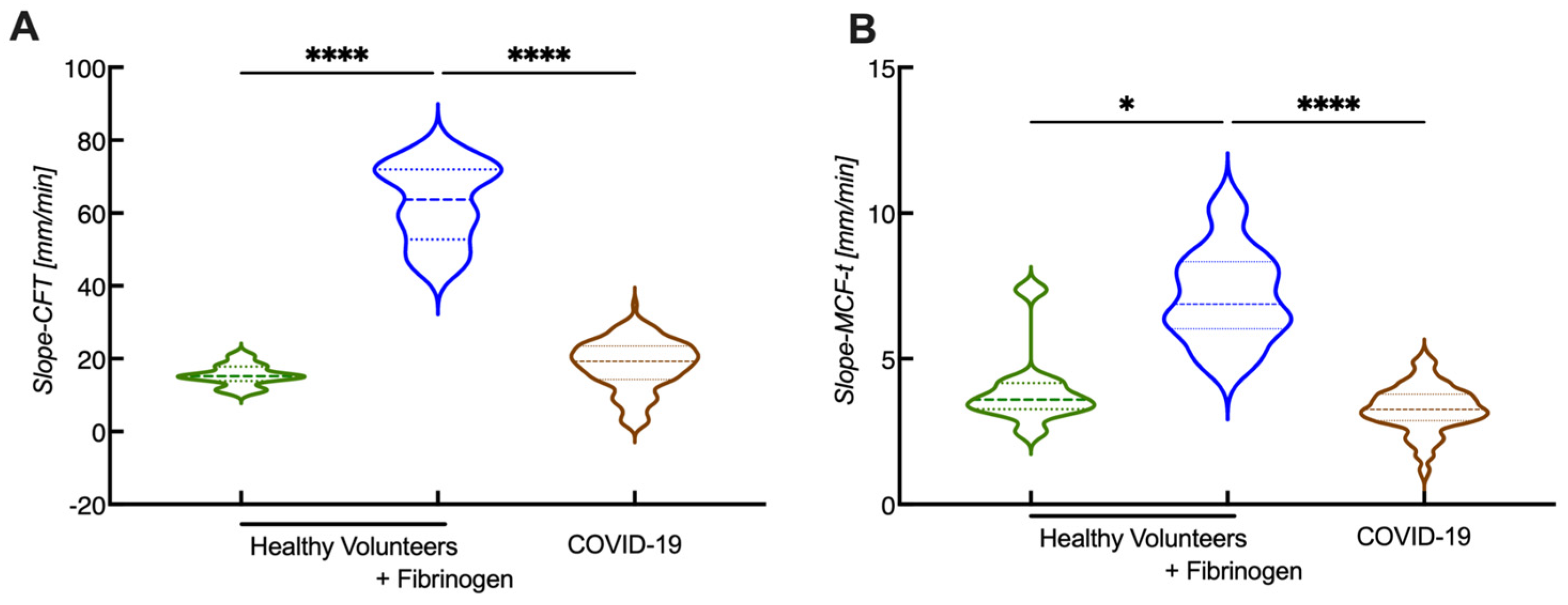
4. Novel Parameters Originated from ROTEM
5. ROTEM beyond COVID-19
6. Discussion and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Auger, J.; Munnix, I.; Cosemans, J.; Heemskerk, J. Platelet response heterogeneity in thrombus formation. Thromb. Haemost. 2009, 102, 1149–1156. [Google Scholar] [CrossRef] [Green Version]
- Farndale, R.W. Collagen-induced platelet activation. Blood Cells Mol. Dis. 2006, 36, 162–165. [Google Scholar] [CrossRef]
- Golebiewska, E.M.; Poole, A.W. Platelet secretion: From haemostasis to wound healing and beyond. Blood Rev. 2015, 29, 153–162. [Google Scholar] [CrossRef] [Green Version]
- Swieringa, F.; Spronk, H.M.; Heemskerk, J.W.; Van Der Meijden, P.E. Integrating platelet and coagulation activation in fibrin clot formation. Res. Pract. Thromb. Haemost. 2018, 2, 450–460. [Google Scholar] [CrossRef]
- D’Alessandro, E.; Posma, J.; Spronk, H.; Cate, H.T. Tissue factor (:Factor VIIa) in the heart and vasculature: More than an envelope. Thromb. Res. 2018, 168, 130–137. [Google Scholar] [CrossRef]
- Spronk, H.M.H.; Govers-Riemslag, J.W.P.; Ten Cate, H. The blood coagulation system as a molecular machine. Bioessays 2003, 25, 1220–1228. [Google Scholar] [CrossRef]
- Yun, T.H.; Baglia, F.A.; Myles, T.; Navaneetham, D.; López, J.A.; Walsh, P.N.; Leung, L.L. Thrombin activation of factor XI on activated platelets requires the interaction of factor XI and platelet glycoprotein Ib alpha with thrombin anion-binding exosites I and II, respectively. J. Biol. Chem. 2003, 278, 48112–48119. [Google Scholar] [CrossRef] [Green Version]
- Griffin, J.H.; Zlokovic, B.V.; Mosnier, L.O. Activated protein C: Biased for translation. Blood 2015, 125, 2898–2907. [Google Scholar] [CrossRef] [Green Version]
- Peraramelli, S.; Suylen, D.P.L.; Rosing, J.; Hackeng, T.M. The Kunitz 1 and Kunitz 3 domains of tissue factor pathway inhibitor are required for efficient inhibition of factor Xa. Thromb. Haemost. 2012, 108, 266–276. [Google Scholar] [CrossRef]
- Broze, G.J., Jr. Tissue factor pathway inhibitor. Thromb. Haemost. 1995, 74, 90–93. [Google Scholar] [CrossRef]
- Chapin, J.C.; Hajjar, K.A. Fibrinolysis and the control of blood coagulation. Blood Rev. 2015, 29, 17–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cesarman-Maus, G.; Hajjar, K.A. Molecular mechanisms of fibrinolysis. Br. J. Haematol. 2005, 129, 307–321. [Google Scholar] [CrossRef] [PubMed]
- Winckers, K.; ten Cate, H.; Hackeng, T.M. The role of tissue factor pathway inhibitor in atherosclerosis and arterial thrombosis. Blood Rev. 2013, 27, 119–132. [Google Scholar] [CrossRef] [PubMed]
- Levy, J.H.; Szlam, F.; Wolberg, A.S.; Winkler, A. Clinical use of the activated partial thromboplastin time and prothrombin time for screening: A review of the literature and current guidelines for testing. Clin. Lab. Med. 2014, 34, 453–477. [Google Scholar] [CrossRef] [PubMed]
- Kitchens, C.S. To bleed or not to bleed? Is that the question for the PTT? J. Thromb. Haemost. 2005, 3, 2607–2611. [Google Scholar] [CrossRef] [PubMed]
- Mezzano, D.; Quiroga, T.; Pereira, J. The level of laboratory testing required for diagnosis or exclusion of a platelet function disorder using platelet aggregation and secretion assays. Semin. Thromb. Hemost. 2009, 35, 242–254. [Google Scholar] [CrossRef]
- Hayward, C.P.M.; Pai, M.; Liu, Y.; Moffat, K.A.; Seecharan, J.; Webert, K.E.; Cook, R.J.; Heddle, N.M. Diagnostic utility of light transmission platelet aggregometry: Results from a prospective study of individuals referred for bleeding disorder assessments. J. Thromb. Haemost. 2009, 7, 676–684. [Google Scholar] [CrossRef]
- Whiting, D.; DiNardo, J.A. TEG and ROTEM: Technology and clinical applications. Am. J. Hematol. 2014, 89, 228–232. [Google Scholar] [CrossRef]
- Nielsen, V.G. A comparison of the Thrombelastograph and the ROTEM. Blood Coagul. Fibrinolysis 2007, 18, 247–252. [Google Scholar] [CrossRef]
- Hochleitner, G.; Sutor, K.; Levett, C.; Leyser, H.; Schlimp, C.J.; Solomon, C. Revisiting Hartert’s 1962 Calculation of the Physical Constants of Thrombelastography. Clin. Appl. Thromb. Hemost. 2017, 23, 201–210. [Google Scholar] [CrossRef] [Green Version]
- Lang, T.; Bauters, A.; Braun, S.L.; Pötzsch, B.; von Pape, K.-W.; Kolde, H.-J.; Lakner, M. Multi-centre investigation on reference ranges for ROTEM thromboelastometry. Blood Coagul. Fibrinolysis 2005, 16, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Theusinger, O.M.; Nürnberg, J.; Asmis, L.M.; Seifert, B.; Spahn, D.R. Rotation thromboelastometry (ROTEM) stability and reproducibility over time. Eur. J. Cardiothorac. Surg. 2010, 37, 677–683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Görlinger, K.; Dirkmann, D.; Hanke, A.A. Rotational Thromboelastometry (ROTEM®). In Trauma Induced Coagulopathy; Springer: Berlin/Heidelberg, Germany, 2016. [Google Scholar]
- Al-Samkari, H.; Karp Leaf, R.S.; Dzik, W.H.; Carlson, J.C.T.; Forgerty, A.E.; Waheed, A.; Goodarzi, K.; Bendapudi, P.K.; Bornikova, L.; Gupta, S.; et al. COVID-19 and coagulation: Bleeding and thrombotic manifestations of SARS-CoV-2 infection. Blood 2020, 136, 489–500. [Google Scholar] [CrossRef] [PubMed]
- Tang, N.; Li, D.; Wang, X.; Sun, Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J. Thromb. Haemost. 2020, 18, 844–847. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Connors, J.M.; Levy, J.H. COVID-19 and its implications for thrombosis and anticoagulation. Blood 2020, 135, 2033–2040. [Google Scholar] [CrossRef]
- Klok, F.A.; Kruip, M.J.H.A.; van der Meer, N.J.M.; Arbous, M.S.; Gommers, D.A.M.P.J.; Kant, K.M.; Kaptein, F.H.J.; van Paassen, J.; Stals, M.A.M.; Huisman, M.V.; et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb. Res. 2020, 191, 145–147. [Google Scholar] [CrossRef]
- Tan, B.K.; Mainbourg, S.; Friggeri, A.; Bertoletti, L.; Douplat, M.; Dargaud, Y.; Grange, C.; Lobbes, H.; Provencher, S.; Lega, J.-C. Arterial and venous thromboembolism in COVID-19: A study-level meta-analysis. Thorax 2021, 76, 970–979. [Google Scholar] [CrossRef]
- Fournier, M.; Faille, D.; Dossier, A.; Mageau, A.; Roland, P.N.; Ajzenberg, N.; Borie, R.; Bouadma, L.; Bunel, V.; Castier, Y.; et al. Arterial Thrombotic Events in Adult Inpatients With COVID-19. Mayo Clin. Proc. 2021, 96, 295–303. [Google Scholar] [CrossRef]
- Ruzzenenti, G.; Maloberti, A.; Giani, V.; Biolcati, M.; Leidi, F.; Monticelli, M.; Grasso, E.; Cartella, I.; Palazzini, M.; Garatti, L.; et al. COVID and Cardiovascular Diseases: Direct and Indirect Damages and Future Perspective. High Blood Press. Cardiovasc. Prev. 2021, 28, 439–445. [Google Scholar] [CrossRef]
- Levi, M.; Iba, T. COVID-19 coagulopathy: Is it disseminated intravascular coagulation? Intern. Emerg. Med. 2021, 16, 309–312. [Google Scholar] [CrossRef]
- Iba, T.; Levy, J.H.; Connors, J.M.; Warkentin, T.E.; Thachil, J.; Levi, M. Managing thrombosis and cardiovascular complications of COVID-19: Answering the questions in COVID-19-associated coagulopathy. Expert Rev. Respir. Med. 2021, 15, 1003–1011. [Google Scholar] [CrossRef] [PubMed]
- Colling, M.E.; Kanthi, Y. COVID–19-associated coagulopathy: An exploration of mechanisms. Vasc. Med. 2020, 25, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Levi, M.; Thachil, J.; Iba, T.; Levy, J.H. Coagulation abnormalities and thrombosis in patients with COVID-19. Lancet Haematol. 2020, 7, e438–e440. [Google Scholar] [CrossRef]
- Parisi, R.; Costanzo, S.; Di Castelnuovo, A.; de Gaetano, G.; Donati, M.B.; Iacoviello, L. Different Anticoagulant Regimens, Mortality, and Bleeding in Hospitalized Patients with COVID-19: A Systematic Review and an Updated Meta-Analysis. Semin. Thromb. Hemost. 2021, 47, 372–391. [Google Scholar] [CrossRef] [PubMed]
- Genovesi, S.; Rebora, P.; Occhino, G.; Rossi, E.; Maloberti, A.; Belli, M.; Bonfanti, P.; Giannattasio, C.; Rossetti, C.; Epis, O.M.; et al. Atrial Fibrillation and Clinical Outcomes in a Cohort of Hospitalized Patients with SARS-CoV-2 Infection and Chronic Kidney Disease. J. Clin. Med. 2021, 10, 4108. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Li, T.; Han, M.; Li, X.; Wu, D.; Xu, Y.; Zhu, Y.; Liu, Y.; Wang, X.; Wang, L. Diagnostic utility of clinical laboratory data determinations for patients with the severe COVID-19. J. Med. Virol. 2020, 92, 791–796. [Google Scholar] [CrossRef]
- Bi, X.; Su, Z.; Yan, H.; Du, J.; Wang, J.; Chen, L.; Peng, M.; Chen, S.; Shen, B.; Li, J. Prediction of severe illness due to COVID-19 based on an analysis of initial Fibrinogen to Albumin Ratio and Platelet count. Platelets 2020, 31, 674–679. [Google Scholar] [CrossRef]
- Hottz, E.D.; Azevedo-Quintanilha, I.G.; Palhinha, L.; Teixeira, L.; Barreto, E.A.; Pão, C.R.R.; Righy, C.; Franco, S.; Souza, T.M.L.; Kurtz, P.; et al. Platelet activation and platelet-monocyte aggregate formation trigger tissue factor expression in patients with severe COVID-19. Blood 2020, 136, 1330–1341. [Google Scholar] [CrossRef]
- Busch, M.; Timmermans, S.A.; Nagy, M.; Visser, M.; Huckriede, J.; Aendekerk, J.P.; De Vries, F.; Potjewijd, J.; Jallah, B.; Ysermans, R.; et al. Neutrophils and Contact Activation of Coagulation as Potential Drivers of COVID-19. Circulation 2020, 142, 1787–1790. [Google Scholar] [CrossRef]
- Pavoni, V.; Gianesello, L.; Pazzi, M.; Stera, C.; Meconi, T.; Frigieri, F.C. Evaluation of coagulation function by rotation thromboelastometry in critically ill patients with severe COVID-19 pneumonia. J. Thromb. Thrombolysis 2020, 50, 281–286. [Google Scholar] [CrossRef]
- Słomka, A.; Kowalewski, M.; Żekanowska, E. Hemostasis in Coronavirus Disease 2019–Lesson from Viscoelastic Methods: A Systematic Review. Thromb. Haemost. 2021, 121, 1181–1192. [Google Scholar] [CrossRef] [PubMed]
- Almskog, L.M.; Wikman, A.; Svensson, J.; Wanecek, M.; Bottai, M.; van der Liden, J.; Agren, A. Rotational Thromboelastometry predicts care level in COVID-19. J. Thromb. Thrombolysis 2020, 51, 437–445. [Google Scholar] [CrossRef] [PubMed]
- Boss, K.; Kribben, A.; Tyczynski, B. Pathological findings in rotation thromboelastometry associated with thromboembolic events in COVID-19 patients. Thromb. J. 2021, 19, 10. [Google Scholar] [CrossRef] [PubMed]
- Hulshof, A.-M.; Brüggemann, R.A.G.; Mulder, M.M.G.; van de Berg, T.W.; Sels, J.-W.E.M.; Olie, R.H.; Spaetgens, B.; Streng, A.S.; Verhezen, P.; van der Horst, I.C.C.; et al. Serial EXTEM, FIBTEM, and tPA Rotational Thromboelastometry Observations in the Maastricht Intensive Care COVID Cohort—Persistence of Hypercoagulability and Hypofibrinolysis Despite Anticoagulation. Front. Cardiovasc. Med. 2021, 8, 654174. [Google Scholar] [CrossRef]
- Mitrovic, M.; Sabljic, N.; Cvetkovic, Z.; Pantic, N.; Dakic, A.Z.; Bukumiric, Z.; Libek, V.; Savic, N.; Milenkovic, B.; Virijevic, M.; et al. Rotational thromboelastometry (ROTEM) profiling of COVID-19 patients. Platelets 2021, 32, 690–696. [Google Scholar] [CrossRef]
- van Veenendaal, N.; Scheeren, T.W.L.; Meijer, K.; van der Voort, P.H.J. Rotational thromboelastometry to assess hypercoagulability in COVID-19 patients. Thromb. Res. 2020, 196, 379–381. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Davies, N.; Harrison, N.; Sabra, A.; Lawrence, M.; Noble, S.; Davidson, S.; Evans, V.; Morris, R.; Hawkins, K.; Williams, P.; et al. Application of ROTEM to assess hypercoagulability in patients with lung cancer. Thromb. Res. 2015, 135, 1075–1080. [Google Scholar] [CrossRef]
- van de Berg, T.W.; Hulshof, A.-M.M.; Nagy, M.; van Oerle, R.; Sels, J.-W.; van Bussel, B.; ten Cate, H.; Henskens, Y.; Spronk, H.M.H. Suggestions for global coagulation assays for the assessment of COVID-19 associated hypercoagulability. Thromb. Res. 2021, 201, 84–89. [Google Scholar] [CrossRef]
- Chan, M.Y.; Andreotti, F.; Becker, R.C. Hypercoagulable states in cardiovascular disease. Circulation 2008, 118, 2286–2297. [Google Scholar] [CrossRef] [Green Version]
- Hincker, A.; Feit, J.; Sladen, R.N.; Wagener, G. Rotational thromboelastometry predicts thromboembolic complications after major non-cardiac surgery. Crit. Care 2014, 18, 549. [Google Scholar] [CrossRef] [PubMed]
- Henskens, Y.M.C.; Gulpen, A.J.W.; Van Oerle, R.; Wetzels, R.; Verhezen, P.; Spronk, H.; Schalla, S.; Crijns, H.J.; Cate, H.T.; Cate-Hoek, A.T. Detecting clinically relevant rivaroxaban or dabigatran levels by routine coagulation tests or thromboelastography in a cohort of patients with atrial fibrillation. Thromb. J. 2018, 16, 3. [Google Scholar] [CrossRef] [PubMed]
- Beiderlinden, M.; Werner, P.; Bahlmann, A.; Kemper, J.; Brezina, T.; Schäfer, M.; Görlinger, K.; Seidel, H.; Kienbaum, P.; Treschan, T.A. Monitoring of argatroban and lepirudin anticoagulation in critically ill patients by conventional laboratory parameters and rotational thromboelastometry–A prospectively controlled randomized double-blind clinical trial. BMC Anesthesiol. 2018, 18, 18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feuring, M.; Wehling, M.; Schultz, A. Dalteparin dose-dependently increases ROTEM® thrombelastography parameters only at supratherapeutic anti-factor Xa levels: An in vitro study. Clin. Exp. Pharmacol. Physiol. 2011, 38, 783–786. [Google Scholar] [CrossRef] [PubMed]
- Schaden, E.; Schober, A.; Hacker, S.; Spiss, C.; Chiari, A.; Kozek-Langenecker, S. Determination of enoxaparin with rotational thrombelastometry using the prothrombinase-induced clotting time reagent. Blood Coagul. Fibrinolysis 2010, 21, 256–261. [Google Scholar] [CrossRef] [PubMed]
- Korpallová, B.; Samoš, M.; Bolek, T.; Kühnelová, L.; Škorňová, I.; Kubisz, P.; Staško, J.; Mokáň, M. ROTEM Testing for Direct Oral Anticoagulants. Semin. Thromb. Hemost. 2021, 47, 815–823. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dauwerse, S.; ten Cate, H.; Spronk, H.M.H.; Nagy, M. The Composition and Physical Properties of Clots in COVID-19 Pathology. Diagnostics 2022, 12, 580. https://doi.org/10.3390/diagnostics12030580
Dauwerse S, ten Cate H, Spronk HMH, Nagy M. The Composition and Physical Properties of Clots in COVID-19 Pathology. Diagnostics. 2022; 12(3):580. https://doi.org/10.3390/diagnostics12030580
Chicago/Turabian StyleDauwerse, Sierk, Hugo ten Cate, Henri M. H. Spronk, and Magdolna Nagy. 2022. "The Composition and Physical Properties of Clots in COVID-19 Pathology" Diagnostics 12, no. 3: 580. https://doi.org/10.3390/diagnostics12030580
APA StyleDauwerse, S., ten Cate, H., Spronk, H. M. H., & Nagy, M. (2022). The Composition and Physical Properties of Clots in COVID-19 Pathology. Diagnostics, 12(3), 580. https://doi.org/10.3390/diagnostics12030580





