Lymphocyte-to-Monocyte Ratio and Clinical Outcomes in Cholangiocarcinoma: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Research Question and Study Design
2.2. Register and Report Guideline
2.3. Search Strategy and Data Sources
2.4. Eligibility Criteria, Study Selection and Data Extraction
2.5. Quality Assessment
2.6. Data Synthesis and Publication Bias
3. Results
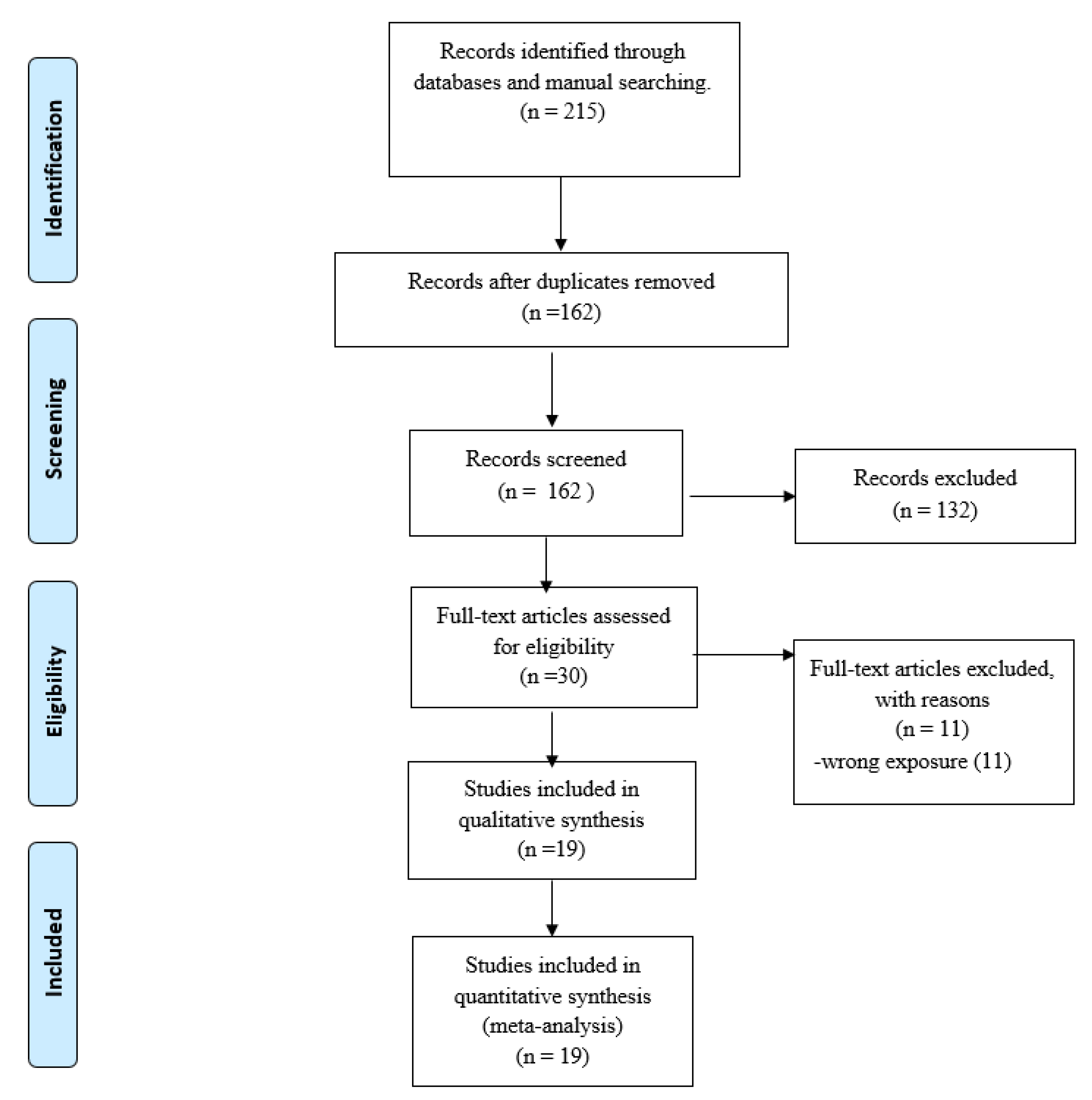
3.1. Study Selection
3.2. Study Characteristics
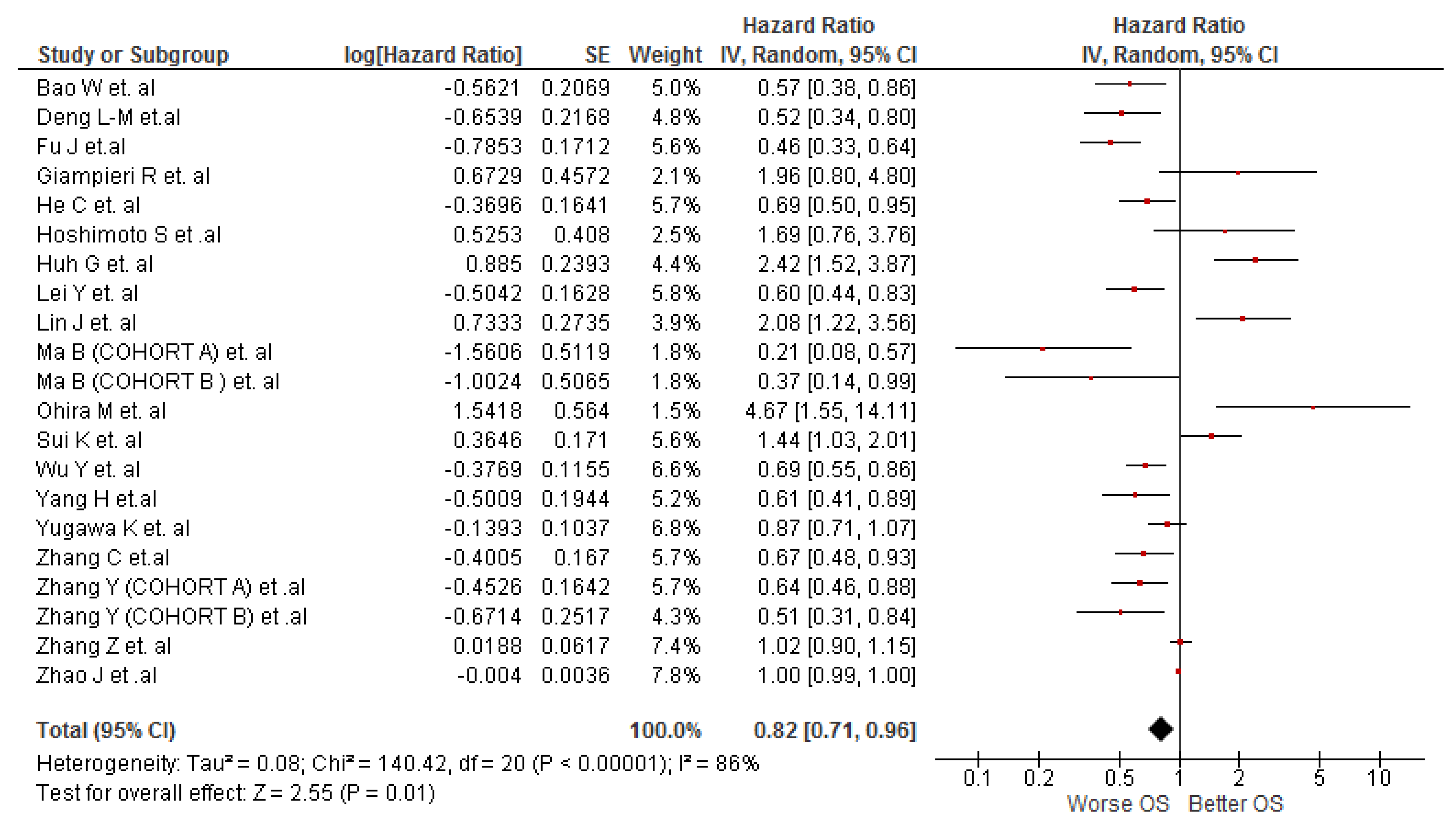
3.3. Association between LMR and OS in Cholangiocarcinoma Patients
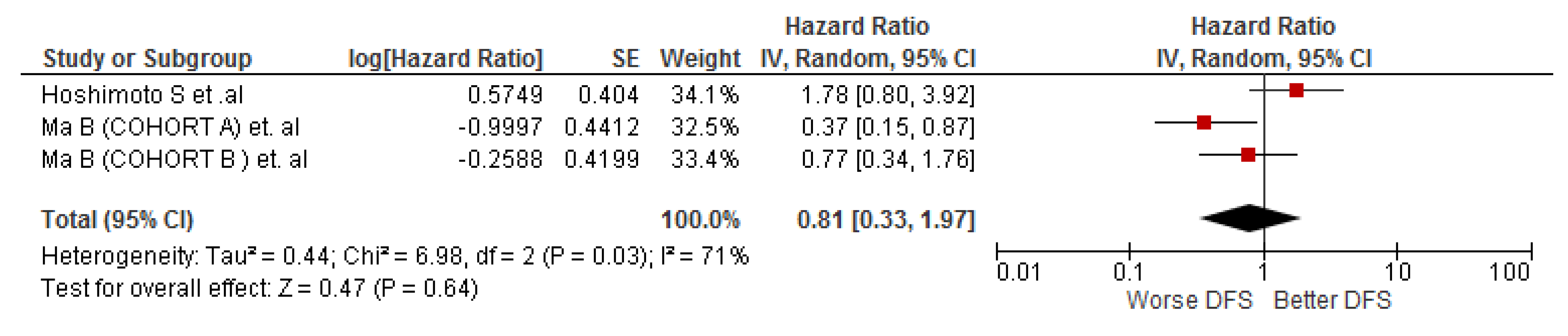
3.4. Association between LMR and DFS in Cholangiocarcinoma Patients
3.5. Association between LMR and RFS in Cholangiocarcinoma Patients
3.6. Association between LMR and TTR in Cholangiocarcinoma Patients
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cha, J.M. Early bile duct cancer. World J. Gastroenterol. 2007, 13, 3409. [Google Scholar] [CrossRef] [PubMed]
- Brindley, P.J.; Bachini, M.; Ilyas, S.I.; Khan, S.A.; Loukas, A.; Sirica, A.E.; Teh, B.T.; Wongkham, S.; Gores, G.J. Cholangiocarcinoma. Nat. Rev. Dis. Prim. 2021, 7, 65. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Zhao, C.; Zhang, Y.; Chen, C.; Lin, X. An Inflammation-Index Signature Predicts Prognosis of Patients with Intrahepatic Cholangiocarcinoma After Curative Resection. J. Inflamm. Res. 2021, 14, 1859–1872. [Google Scholar] [CrossRef] [PubMed]
- Sarcognato, S.; Sacchi, D.; Fassan, M.; Fabris, L.; Cadamuro, M.; Zanus, G.; Cataldo, I.; Capelli, P.; Baciorri, F.; Cacciatore, M.; et al. Cholangiocarcinoma. Pathologica 2021, 113, 158–169. [Google Scholar] [CrossRef]
- Sripa, B.; Pairojkul, C. Cholangiocarcinoma: Lessons from Thailand. Curr. Opin. Gastroenterol. 2008, 24, 349–356. [Google Scholar] [CrossRef]
- Florio, A.A.; Ferlay, J.; Znaor, A.; Ruggieri, D.; Alvarez, C.S.; Laversanne, M.; Bray, F.; McGlynn, K.A.; Petrick, J.L. Global trends in intrahepatic and extrahepatic cholangiocarcinoma incidence from 1993 to 2012. Cancer 2020, 126, 2666–2678. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.A.; Toledano, M.B.; Taylor-Robinson, S.D. Epidemiology, risk factors, and pathogenesis of cholangiocarcinoma. HPB 2008, 10, 77–82. [Google Scholar] [CrossRef]
- Luis. Colangiocarcinoma: Actualización, diagnóstico y terapia. Rev. Méd. Chile 2008, 136, 240–248. [Google Scholar] [CrossRef]
- Mjaess, G.; Chebel, R.; Karam, A.; Moussa, I.; Pretot, D.; Abi Tayeh, G.; Sarkis, J.; Semaan, A.; Peltier, A.; Aoun, F.; et al. Prognostic role of neutrophil-to-lymphocyte ratio (NLR) in urological tumors: An umbrella review of evidence from systematic reviews and meta-analyses. Acta Oncol. 2021, 60, 704–713. [Google Scholar] [CrossRef]
- Naszai, M.; Kurjan, A.; Maughan, T.S. The prognostic utility of pre-treatment neutrophil-to-lymphocyte-ratio (NLR) in colorectal cancer: A systematic review and meta-analysis. Cancer Med. 2021, 10, 5983–5997. [Google Scholar] [CrossRef]
- Nouri-Vaskeh, M.; Mirza-Aghazadeh-Attari, M.; Pashazadeh, F.; Azami-Aghdash, S.; Alizadeh, H.; Pouya, P.; Halimi, M.; Jadideslam, G.; Zarei, M. Prognostic Impact of Monocyte to Lymphocyte Ratio in Clinical Outcome of Patients with Hepatocellular Carcinoma: A Systematic Review and Meta-analysis. Galen Med. J. 2020, 9, e1948. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Tao, L.; Zhang, L.; Xiu, D. Prognostic role of lymphocyte to monocyte ratio for patients with pancreatic cancer: A systematic review and meta-analysis. OncoTargets Ther. 2017, 10, 3391–3397. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Yang, L.; Liu, D.; Li, W.M. Prognostic Value of Pretreatment Lymphocyte-to-Monocyte Ratio in Lung Cancer: A Systematic Review and Meta-Analysis. Technol. Cancer Res. Treat. 2021, 20, 1533033820983085. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: Explanation and elaboration. BMJ 2009, 339, b2700. [Google Scholar] [CrossRef]
- McGowan, J.; Sampson, M.; Salzwedel, D.M.; Cogo, E.; Foerster, V.; Lefebvre, C. PRESS Peer Review of Electronic Search Strategies: 2015 Guideline Statement. J. Clin. Epidemiol. 2016, 75, 40–46. [Google Scholar] [CrossRef]
- Ouzzani, M.; Hammady, H.; Fedorowicz, Z.; Elmagarmid, A. Rayyan—A web and mobile app forsystematic reviews. Syst. Rev. 2016, 5, 210. [Google Scholar] [CrossRef]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 1 July 2022).
- Wu, Y.; Ren, F.; Chai, Y.; Xue, Z.; Shen, C.; Zhang, X.; Lv, Y.; Hu, L. Prognostic value of inflammation-based indexes for intrahepatic cholangiocarcinoma following curative resection. Oncol. Lett. 2019, 17, 165–174. [Google Scholar] [CrossRef]
- Bao, W.; Deng, L.; Lin, Z.; Lin, Z.; Yang, J.; Zhao, J.; Zheng, C.; Yu, Z.; Jin, B.; Wang, Y.; et al. A nomogram based on Psoas muscle index and prognostic nutritional index predicts the prognosis of intrahepatic cholangiocarcinoma after surgery: A multi-center cohort study. BMC Cancer 2021, 1–18. [Google Scholar] [CrossRef]
- Lin, J.; Fang, T.; Zhu, M.; Xu, X.; Zhang, J.; Zheng, S.; Jing, C.; Zhang, M.; Liu, B.; Zhang, B. Comparative performance of inflammation-based prognostic scores in patients operated for intrahepatic cholangiocarcinoma. Cancer Manag. Res. 2019, 11, 9107–9119. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhou, Y.; Hu, K.; Huang, Y. Investigating effects of preoperative inflammatory biomarkers on predicting survival outcomes of intrahepatic cholangiocarcinoma after curative resection. World J. Surg. Oncol. 2020, 18, 272. [Google Scholar] [CrossRef]
- Huh, G.; Ryu, J.K.; Chun, J.W.; Kim, J.S.; Park, N.; Cho, I.R.; Paik, W.H.; Lee, S.H.; Kim, Y.T. High platelet-to-lymphocyte ratio is associated with poor prognosis in patients with unresectable intrahepatic cholangiocarcinoma receiving gemcitabine plus cisplatin. BMC Cancer 2020, 20, 907. [Google Scholar] [CrossRef] [PubMed]
- Ohira, M.; Yoshizumi, T.; Yugawa, K.; Kosai-Fujimoto, Y.; Inokuchi, S.; Motomura, T.; Mano, Y.; Toshima, T.; Itoh, S.; Harada, N.; et al. Association of inflammatory biomarkers with long-term outcomes after curative surgery for mass-forming intrahepatic cholangiocarcinoma. Surg. Today 2020, 50, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Sui, K.; Okabayashi, T.; Umeda, Y.; Oishi, M.; Kojima, T.; Sato, D.; Endo, Y.; Ota, T.; Hioki, K.; Inagaki, M.; et al. Prognostic Utility of the Glasgow Prognostic Score for the Long-Term Outcomes After Liver Resection for Intrahepatic Cholangiocarcinoma: A Multi-institutional Study. World J. Surg. 2021, 45, 279–290. [Google Scholar] [CrossRef] [PubMed]
- Ma, B.; Meng, H.; Shen, A.; Ma, Y.; Zhao, D.; Liu, G.; Zheng, S.; Tian, Y.; Zhang, W.; Li, Q.; et al. Prognostic Value of Inflammatory and Tumour Markers in Small-Duct Subtype Intrahepatic Cholangiocarcinoma after Curative-Intent Resection. Gastroenterol. Res. Pract. 2021, 2021, 6616062. [Google Scholar] [CrossRef]
- Yang, H.; Wang, J.; Li, Z.; Yang, Y.; Yang, L.; Zhang, Y.; Shi, Y.; Cao, Y.; Zhou, J.; Wang, Z.; et al. Risk Factors and Outcomes of Early Relapse After Curative Resection of Intrahepatic Cholangiocarcinoma. Front. Oncol. 2019, 9, 854. [Google Scholar] [CrossRef]
- Yugawa, K.; Itoh, S.; Yoshizumi, T.; Morinaga, A.; Iseda, N.; Toshima, T.; Harada, N.; Kohashi, K.; Oda, Y.; Mori, M. Lymphocyte-C-reactive protein ratio as a prognostic marker associated with the tumor immune microenvironment in intrahepatic cholangiocarcinoma. Int. J. Clin. Oncol. 2021, 26, 1901–1910. [Google Scholar] [CrossRef]
- Lei, Y.U.; Zhi, D.A.I.; Zheng, W.; Yong, Z.; Qing, C.; Lei, Y.U.; Zhi, D.A.I.; Zheng, W.; Yong, Z.; Qing, C. The impact of lymph node metastasis on the clinical parameters and prognosis of intrahepatic cholangiocarcinoma patients after curative resection. China Oncol. 2020, 30, 694–700. [Google Scholar] [CrossRef]
- Deng, L.M.; Wang, Y.; Yang, J.H.; Li, J.L.; Chen, Z.Y.; Bao, W.M.; Chen, K.Y.; Yao, X.F.; Zheng, C.M.; Zheng, J.Y.; et al. Diffuse reduction of spleen density is a novel prognostic marker for intrahepatic cholangiocarcinoma after curative resection. World J. Gastrointest. Oncol. 2021, 13, 930–942. [Google Scholar] [CrossRef]
- Giampieri, R.; Liguori, C.; Crocetti, S.; Pecci, F.; Bittoni, A.; Lenci, E.; Cantini, L.; Giulia, M.; Lanese, A.; Pinterpe, G.; et al. P-131 External validation of prognostic ALAN score in patients with inoperable bile duct cancer treated with second-line chemotherapy. Ann. Oncol. 2021, 32, S143. [Google Scholar] [CrossRef]
- Zhang, Y.; Shi, S.M.; Yang, H.; Yang, L.X.; Wang, Z.; Li, X.D.; Yin, D.; Shi, Y.H.; Cao, Y.; Dai, Z.; et al. Systemic inflammation score predicts survival in patients with intrahepatic cholangiocarcinoma undergoing curative resection. J. Cancer 2019, 10, 494. [Google Scholar] [CrossRef]
- Zhao, J.; Chen, Y.; Wang, J.; Wang, J.; Wang, Y.; Chai, S.; Zhang, Y.; Chen, X.; Zhang, W. Preoperative risk grade predicts the long-term prognosis of intrahepatic cholangiocarcinoma: A retrospective cohort analysis. BMC Surg. 2021, 21, 113. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Chen, Q.; Lai, Z.; Lin, K.; Fang, G.; Ding, Z.; Gao, Y.; Zeng, Y. A Novel Preoperative Inflammation Score System Established for Postoperative Prognosis Predicting of Intrahepatic Cholangiocarcinoma. BMC Cancer 2021, 1–32. [Google Scholar] [CrossRef]
- Hoshimoto, S.; Hishinuma, S.; Shirakawa, H.; Tomikawa, M.; Ozawa, I.; Ogata, Y. Association of Preoperative Platelet-to-Lymphocyte Ratio with Poor Outcome in Patients with Distal Cholangiocarcinoma. Oncology 2019, 96, 290–298. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Wang, H.; Ning, Z.; Xu, L.; Zhuang, L.; Wang, P.; Meng, Z.Q. Prognostic value of systemic inflammatory response markers in patients with intrahepatic cholangiocarcinoma. Int. J. Clin. Exp. Med. 2016, 9, 11502–11509. [Google Scholar] [CrossRef]
- He, C.; Zhao, C.; Lu, J.; Huang, X.; Chen, C.; Lin, X. Evaluation of Preoperative Inflammation-Based Prognostic Scores in Patients With Intrahepatic Cholangiocarcinoma: A Multicenter Cohort Study. Front. Oncol. 2021, 11, 672607. [Google Scholar] [CrossRef] [PubMed]
- Volcic, M.; Karl, S.; Baumann, B.; Salles, D.; Daniel, P.; Fulda, S.; Wiesmüller, L. NF-κB regulates DNA double-strand break repair in conjunction with BRCA1–CtIP complexes. Nucleic Acids Res. 2012, 40, 181. [Google Scholar] [CrossRef]
- Taniguchi, K.; Karin, M. NF-κB, inflammation, immunity and cancer: Coming of age. Nat. Rev. Immunol. 2018, 18, 309–324. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Zhu, L.; Yang, Y.; Long, Y.; Li, X.; Wang, Y. Prognostic Role of Neutrophil to Lymphocyte Ratio in Ovarian Cancer: A Meta-Analysis. Technol. Cancer Res. Treat. 2018, 17, 1533033818791500. [Google Scholar] [CrossRef]
- Zheng, J.; Cai, J.; Li, H.; Zeng, K.; He, L.; Fu, H.; Zhang, J.; Chen, L.; Yao, J.; Zhang, Y.; et al. Neutrophil to Lymphocyte Ratio and Platelet to Lymphocyte Ratio as Prognostic Predictors for Hepatocellular Carcinoma Patients with Various Treatments: A Meta-Analysis and Systematic Review. Cell. Physiol. Biochem. Int. J. Exp. Cell. Physiol. Biochem. Pharmacol. 2017, 44, 967–981. [Google Scholar] [CrossRef]
- Carr, B.I.; Pancoska, P.; Branch, R.A. Low alpha-fetoprotein hepatocellular carcinoma. J. Gastroenterol. Hepatol. 2010, 25, 1543–1549. [Google Scholar] [CrossRef]
- Peng, D.; Lu, J.; Hu, H.; Li, B.; Ye, X.; Cheng, N. Lymphocyte to Monocyte Ratio Predicts Resectability and Early Recurrence of Bismuth-Corlette Type IV Hilar Cholangiocarcinoma. J. Gastrointest. Surg. 2020, 24, 330. [Google Scholar] [CrossRef] [PubMed]
- Tateishi, R.; Enooku, K.; Shiina, S.; Koike, K. Tumor markers for hepatocellular carcinoma. Mol. Clin. Oncol. 2013, 1, 821–827. [Google Scholar] [CrossRef]
- Wu, S.J.; Lin, Y.X.; Ye, H.; Li, F.Y.; Xiong, X.Z.; Cheng, N.S. Lymphocyte to monocyte ratio and prognostic nutritional index predict survival outcomes of hepatitis B virus-associated hepatocellular carcinoma patients after curative hepatectomy. J. Surg. Oncol. 2016, 114, 202–210. [Google Scholar] [CrossRef] [PubMed]
- Li, G.J.; Ji, J.J.; Yang, F.; Xu, H.W.; Bai, Y. Preoperative lymphocyte-to-monocyte ratio predicts survival in primary hepatitis B virus-positive hepatocellular carcinoma after curative resection. OncoTargets Ther. 2017, 10, 1181–1189. [Google Scholar] [CrossRef][Green Version]
- Tham, T.; Olson, C.; Khaymovich, J.; Herman, S.W.; Costantino, P.D. The lymphocyte-to-monocyte ratio as a prognostic indicator in head and neck cancer: A systematic review and meta-analysis. Eur. Arch. Oto-Rhino-Laryngol. 2018, 275, 1663–1670. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Xu, D.; Song, H.; Qiu, B.; Tian, D.; Li, Z.; Ji, Y.; Wang, J. Inflammation and nutrition-based biomarkers in the prognosis of oesophageal cancer: A systematic review and meta-analysis. BMJ Open 2021, 11, e048324. [Google Scholar] [CrossRef]
- Wang, X.; Su, S.; Guo, Y. The clinical use of the platelet to lymphocyte ratio and lymphocyte to monocyte ratio as prognostic factors in renal cell carcinoma: A systematic review and meta-analysis. Oncotarget 2017, 8, 84506–84514. [Google Scholar] [CrossRef]
- Hu, R.j.; Liu, Q.; Ma, J.y.; Zhou, J.; Liu, G. Preoperative lymphocyte-to-monocyte ratio predicts breast cancer outcome: A meta-analysis. Clin. Chim. Acta 2018, 484, 1–6. [Google Scholar] [CrossRef]
- Labib, P.L.; Goodchild, G.; Pereira, S.P. Molecular Pathogenesis of Cholangiocarcinoma. BMC Cancer 2019, 19, 185. [Google Scholar] [CrossRef]
- Roy, S.; Glaser, S.; Chakraborty, S. Inflammation and Progression of Cholangiocarcinoma: Role of Angiogenic and Lymphangiogenic Mechanisms. Front. Med. 2019, 6, 293. [Google Scholar] [CrossRef]


| Author | Year | Study Location | Median Follow-Up Time |
Participants (Male) | Median/Mean Age (IQR/SD) | Type of Cholangiocarcinoma Evaluated | Outcome | HR(95% CI), p-Value | Cut-Off | TNM Stage (I–II/III–IV) |
|---|---|---|---|---|---|---|---|---|---|---|
| Wu Y et al. [18] | 2019 | China | 29.1 months | 123 (67) | 57 (11) | Intrahepatic | Overall Survival | 0.686 (0.547–0.819), p < 0.05 | 3.42 | 38/85 |
| He C et al. [36] | 2021 | China | NR | 292 (181) | 56 (20–77) | Intrahepatic | Overall Survival | 0.691 (0.501–0.953), p < 0.05 | 4.06 | 107/185 |
| Lin J et al. [20] | 2019 | China | NR | 123 (65) | 60 (31–85) | Intrahepatic | Overall Survival | 2.082 (1.218–3.558), p < 0.05 | 3.62 | 99/24 |
| Huh G et al. [22] | 2020 | South Korea | 35.4 months | 137 (83) | 64 (57–72) | Intrahepatic | Overall Survival | 2.423 (1.516–3.875), p < 0.05 | 3.5 | NR/NR |
| Ohira M et al. [23] | 2021 | Japan | NR | 52 (41) | 61 (39–82) | Intrahepatic | Overall Survival | 4.673 (1.547–20.165), p < 0.05 | 4.36 | 35/17 |
| Yang H et.al [26] | 2019 | China | 44 months | 299 (181) | NR | Intrahepatic | Overall Survival | 0.606 (0.414–0.885), p < 0.05 | 4.45 | 226/73 |
| Fu J et.al [33] | 2021 | China | NR | 446 (295) | 54.36 (10.71) | Intrahepatic | Overall Survival | 0.465 (0.326–0.663), p < 0.005 | 2.48 | NR/NR |
| Sui K et al. [24] | 2020 | Japan | 27.6 months | 273 (164) | 70 (9.4) | Intrahepatic | Overall Survival | 1.44 (1.03–2.43), p < 0.05 | 3.7 | NR/NR |
| Giampieri R et al. [30] | 2021 | Italy | NR | 45 (NR) | NR | Mixed | Overall Survival | 1.96 (0.80–4.8), p = 0.138 | 2.1 | NR/NR |
| Zhao J et al. [32] | 2021 | China | NR | 468 (282) | 58 (51–65) | Intrahepatic | Overall Survival | 0.996 (0.989–1.003), p = 0.302 | NR | NR/NR |
| Zhang C et al. [35] | 2016 | China | NR | 187 (117) | 58 (12) | Intrahepatic | Overall Survival | 0.67 (0.483–0.931), p < 0.05 | 3 | NR/NR |
| Bao W et al. [19] | 2021 | China | 28.7 months | 178 (85) | 64 (10) | Intrahepatic | Overall Survival | 0.57 (0.38–0.87), p < 0.05 | 3 | 126/52 |
| Recurrence-free survival | 0.57 (0.37–0.86), p < 0.05 | |||||||||
| Zhang Z et al. [21] | 2020 | China | NR | 128 (70) | 56 (10) | Intrahepatic | Overall Survival | 1.019 (0.903–1.151), p = 0.757 | NR | 53/75 |
| Recurrence-free survival | 1.039 (0.943–1.146), p = 0.435 | |||||||||
| Yugawa K et al. [27] | 2021 | Japan | NR | 78 (55) | 66 (39–87) | Intrahepatic | Overall Survival | 0.87 (0.71–1.71), p = 0.1354 | NR | NR/NR |
| Recurrence-free survival | 0.92 (0.78–1.06), p = 0.2414 | |||||||||
| Ma B (COHORT A) et al. [25] | 2021 | Tianjin, China | NR | 72 (41) | 59 (32–76) | Intrahepatic | Overall Survival | 0.21 (0.077–0.569), p < 0.05 | 2.65 | NR/NR |
| Disease Free Survival | 0.368 (0.155–0.874), p < 0.05 | |||||||||
| Ma B (COHORT B) et al. [25] | 2021 | Weifang, China | 25.1 months | 102 (57) | 49 (28–77) | Intrahepatic | Overall Survival | 0.367 (0.136–0.993), p < 0.05 | 2.7 | NR/NR |
| Disease Free Survival | 0.772 (0.339–1.758), p = 0.537 | |||||||||
| Hoshimoto S et al. [34] | 2019 | Japan | NR | 53 (31) | 70 (50–87) | Distal | Overall Survival | 1.691 (0.760–3.764), p = 0.198 | 4.633 | 50/3 |
| Disease Free Survival | 1.777 (0.805–3.925), p = 0.155 | 3.208 | ||||||||
| Deng L-M et al. [29] | 2021 | China | 29.3 months | 167 (83) | 63 (9) | Intrahepatic | Overall Survival | 0.52 (0.34–0.8), p < 0.05 | 3.13 | 116/51 |
| Recurrence-free survival | 0.51 (0.33–0.78), p < 0.05 | |||||||||
| Lei Y et al. [28] | 2020 | China | 44 months | 322 (194) | NR | Intrahepatic | Overall Survival | 0.604 (0.439–0.831), p < 0.05 | 4.45 | 248/74 |
| Time to recurrence | 0.735 (0.542–0.997), p < 0.05 | |||||||||
| Zhang Y (COHORT A) et al. [31] | 2019 | China | 44 months | 322 (194) | 58 (27–81) | Intrahepatic | Overall Survival | 0.636 (0.461– 0.878), p < 0.05 | 4.45 | 248/74 |
| Time to recurrence | 0.758 (0.557–1.032), p = 0.079 | |||||||||
| Zhang Y (COHORT B) et al. [31] | 2019 | China | 38.3 months | 104 (47) | 42 (33–56) | Intrahepatic | Overall Survival | 0.511 (0.312–0.837), p < 0.05 | 4.45 | 95/31 |
| Time to recurrence | 0.538 (0.327–0.884), p < 0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dotto-Vasquez, G.; Villacorta-Ampuero, A.K.; Ulloque-Badaracco, J.R.; Hernandez-Bustamante, E.A.; Alarcón-Braga, E.A.; Herrera-Añazco, P.; Benites-Zapata, V.A.; Hernandez, A.V. Lymphocyte-to-Monocyte Ratio and Clinical Outcomes in Cholangiocarcinoma: A Systematic Review and Meta-Analysis. Diagnostics 2022, 12, 2655. https://doi.org/10.3390/diagnostics12112655
Dotto-Vasquez G, Villacorta-Ampuero AK, Ulloque-Badaracco JR, Hernandez-Bustamante EA, Alarcón-Braga EA, Herrera-Añazco P, Benites-Zapata VA, Hernandez AV. Lymphocyte-to-Monocyte Ratio and Clinical Outcomes in Cholangiocarcinoma: A Systematic Review and Meta-Analysis. Diagnostics. 2022; 12(11):2655. https://doi.org/10.3390/diagnostics12112655
Chicago/Turabian StyleDotto-Vasquez, Giuseppe, Andrea K. Villacorta-Ampuero, Juan R. Ulloque-Badaracco, Enrique A. Hernandez-Bustamante, Esteban A. Alarcón-Braga, Percy Herrera-Añazco, Vicente A. Benites-Zapata, and Adrian V. Hernandez. 2022. "Lymphocyte-to-Monocyte Ratio and Clinical Outcomes in Cholangiocarcinoma: A Systematic Review and Meta-Analysis" Diagnostics 12, no. 11: 2655. https://doi.org/10.3390/diagnostics12112655
APA StyleDotto-Vasquez, G., Villacorta-Ampuero, A. K., Ulloque-Badaracco, J. R., Hernandez-Bustamante, E. A., Alarcón-Braga, E. A., Herrera-Añazco, P., Benites-Zapata, V. A., & Hernandez, A. V. (2022). Lymphocyte-to-Monocyte Ratio and Clinical Outcomes in Cholangiocarcinoma: A Systematic Review and Meta-Analysis. Diagnostics, 12(11), 2655. https://doi.org/10.3390/diagnostics12112655







