Prosthetic Knee Joint Infection Due to Candida lusitaniae: A Diagnostic and Therapeutic Odyssey: A Case Study
Abstract
1. Introduction
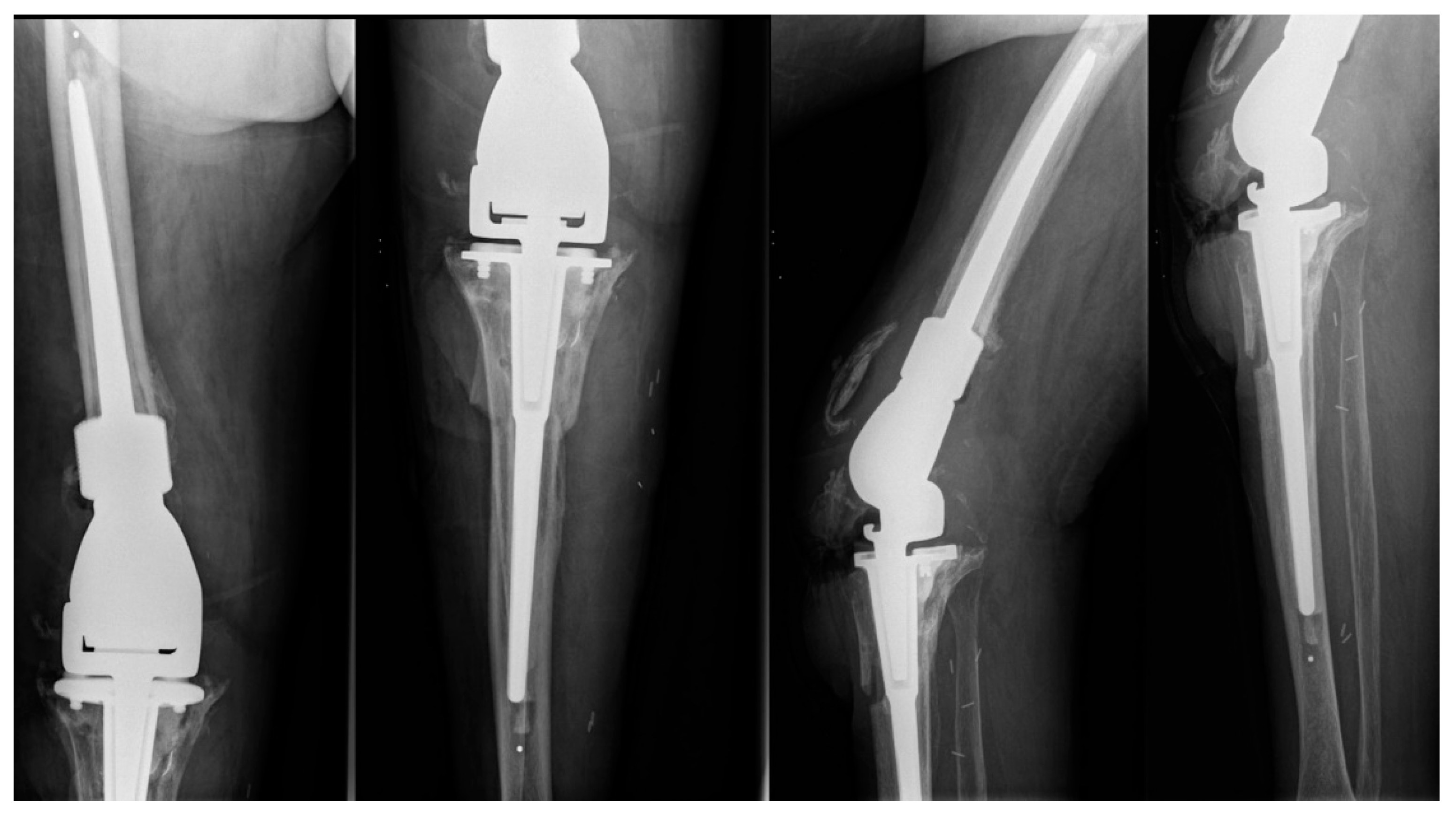
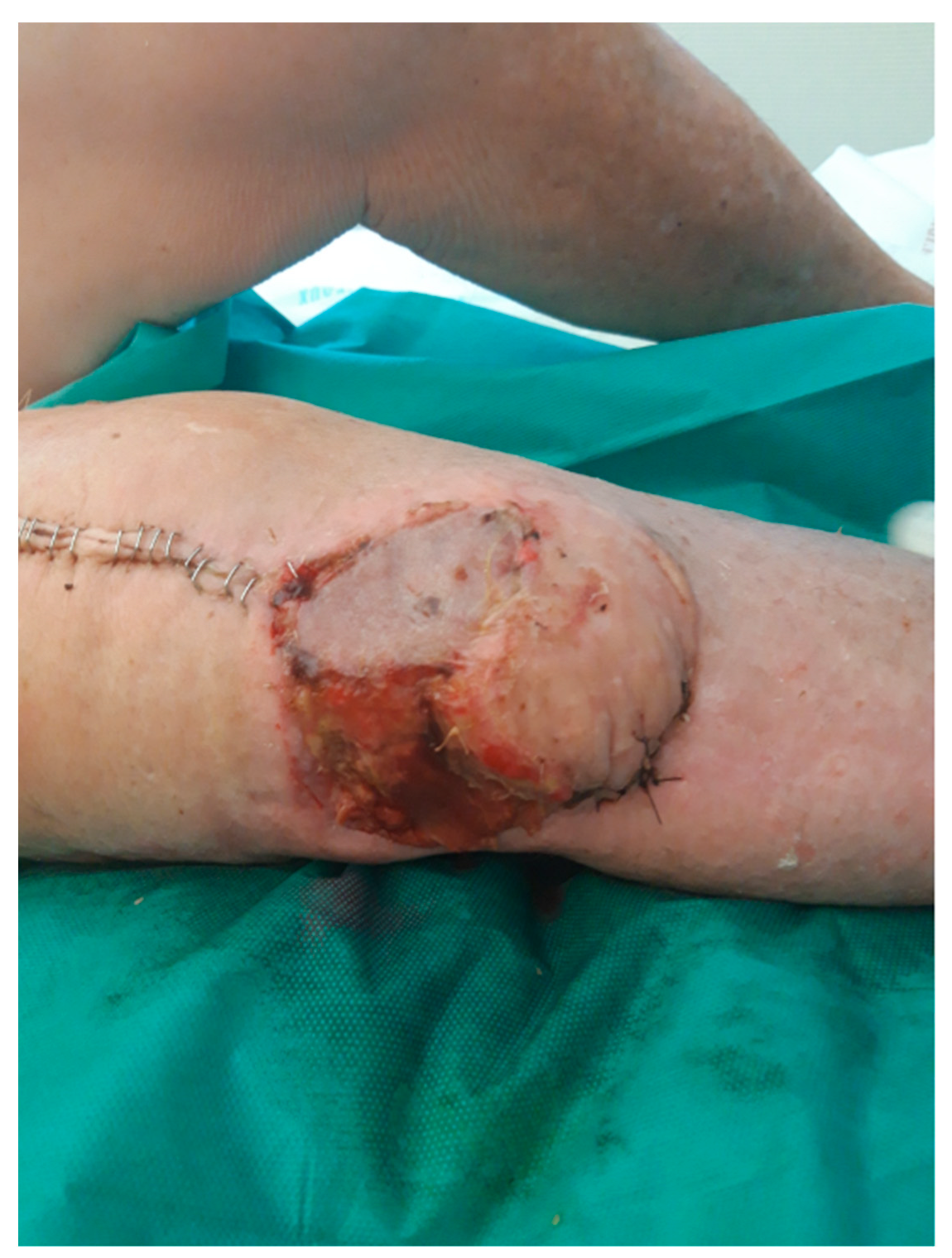
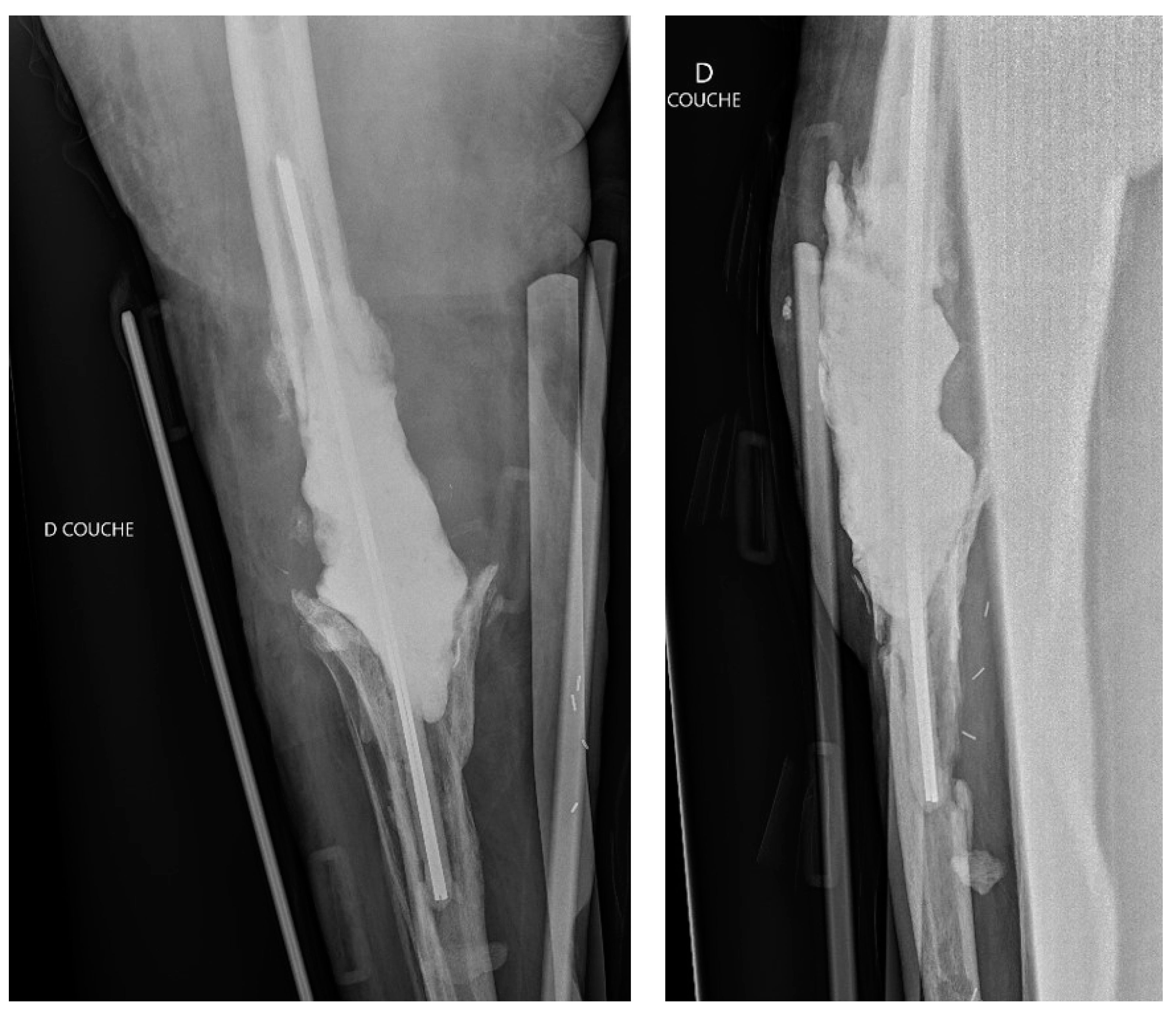
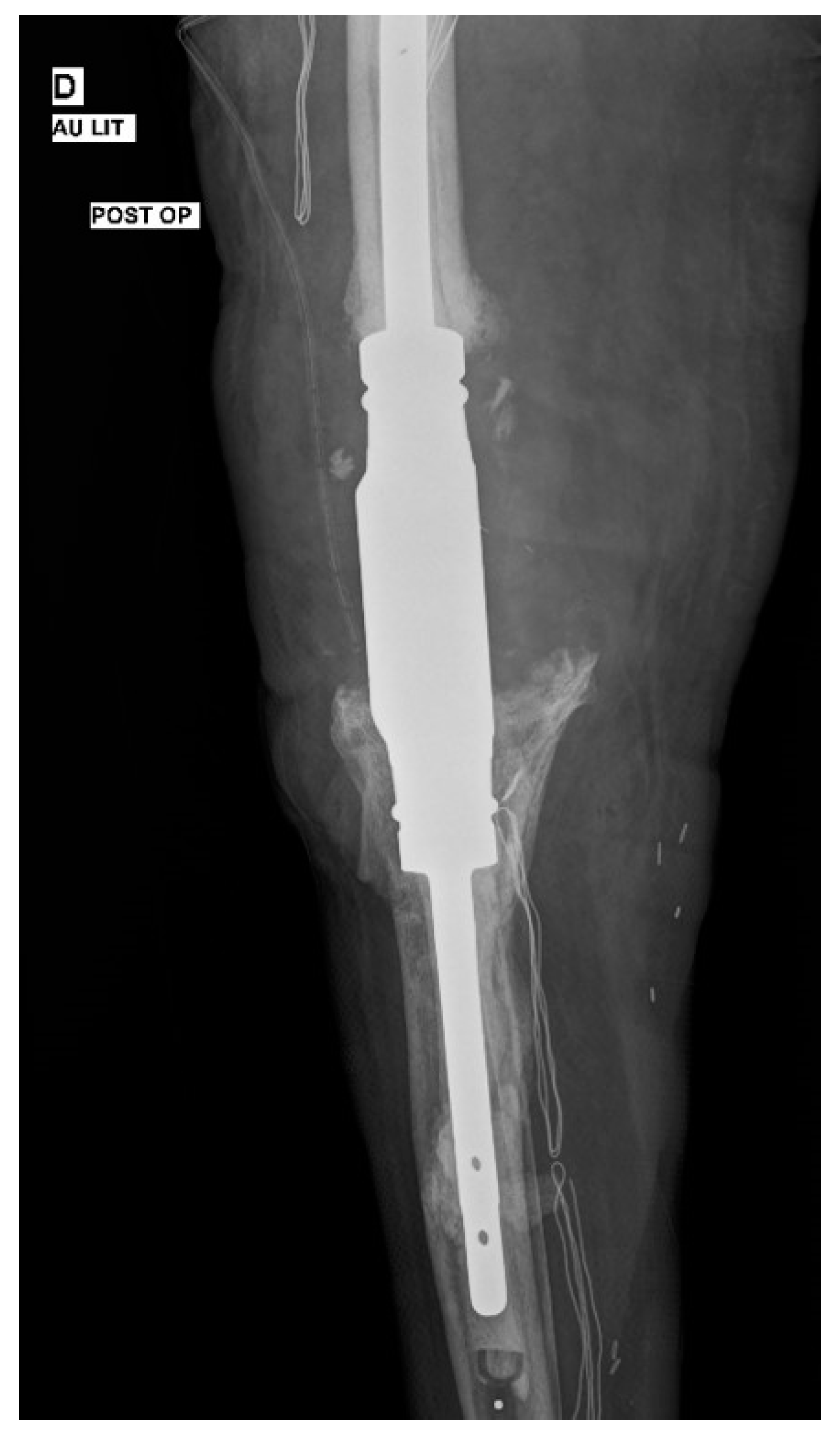
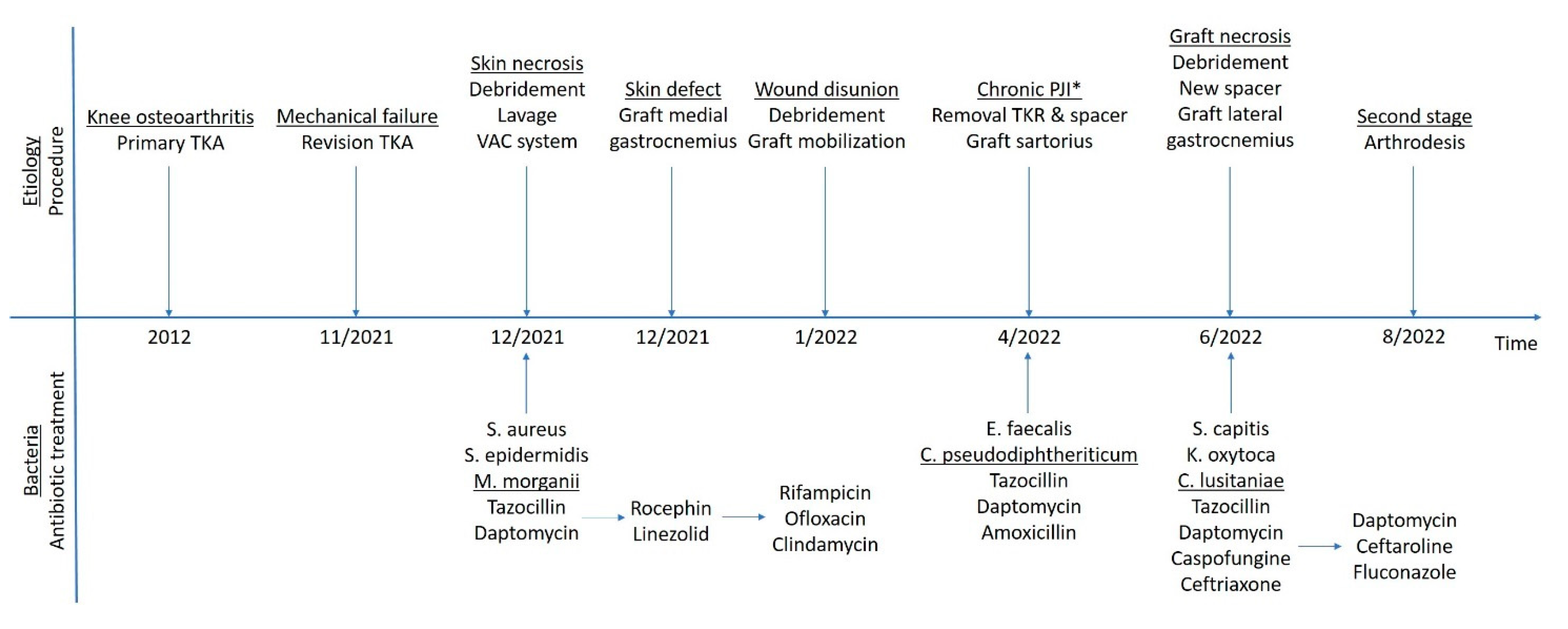
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Inacio, M.C.S.; Paxton, E.W.; Graves, S.E.; Namba, R.S.; Nemes, S. Projected increase in total knee arthroplasty in the United States—An alternative projection model. Osteoarthr. Cartil. 2017, 25, 1797–1803. [Google Scholar] [CrossRef] [PubMed]
- Dall’Oca, C.; Ricci, M.; Vecchini, E.; Giannini, N.; Lamberti, D.; Tromponi, C.; Magnan, B. Evolution of TKA design. Acta Biomed. 2017, 88, 17–31. [Google Scholar] [PubMed]
- Kalavrytinos, D.; Koutserimpas, C.; Kalavrytinos, I.; Dretakis, K. Expanding Robotic Arm-Assisted Knee Surgery: The First Attempt to Use the System for Knee Revision Arthroplasty. Case Rep. Orthop. 2020, 2020, 4806987. [Google Scholar] [CrossRef] [PubMed]
- Besiris, G.T.; Koutserimpas, C.; Karamitros, A.; Karaiskos, I.; Tsakalou, D.; Raptis, K.; Kourelis, K.; Paxinos, O.; Kotsirakis, A.; Vlasis, K. Topical use of tranexamic acid in primary total knee arthroplasty: A comparative study. G. Chir. -J. Ital. Surg. Assoc. 2020, 41, 126–130. [Google Scholar]
- Leung, T.P.; Lee, C.H.; Chang, E.W.Y.; Lee, Q.J.; Wong, Y.C. Clinical outcomes of fast-track total knee arthroplasty for patients aged >80 years. Hong Kong Med. J. 2022, 28, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Vasiliadis, A.V.; Poutoglidou, F.; Chatziravdeli, V.; Metaxiotis, D.; Beletsiotis, A. Acute Periprosthetic Hip Joint Infection Caused by Multidrug-Resistant Acinetobacter Baumannii: Is Debridement, Antibiotics, Irrigation, and Implant Retention a Viable Treatment Option? Cureus 2021, 13, e13090. [Google Scholar] [CrossRef]
- Koutserimpas, C.; Zervakis, S.G.; Maraki, S.; Alpantaki, K.; Ioannidis, A.; Kofteridis, D.P.; Samonis, G. Non-albicans Candida prosthetic joint infections: A systematic review of treatment. World J. Clin. Cases 2019, 7, 1430–1443. [Google Scholar] [CrossRef]
- Koutserimpas, C.; Samonis, G.; Velivassakis, E.; Iliopoulou-Kosmadaki, S.; Kontakis, G.; Kofteridis, D.P. Candida glabrata prosthetic joint infection, successfully treated with anidulafungin: A case report and review of the literature. Mycoses 2018, 61, 266–269. [Google Scholar] [CrossRef]
- Beam, E.; Osmon, D. Prosthetic Joint Infection Update. Infect. Dis. Clin. N. Am. 2018, 32, 843–859. [Google Scholar] [CrossRef]
- Abblitt, W.P.; Chan, E.W.; Shinar, A.A. Risk of Periprosthetic Joint Infection in Patients with Multiple Arthroplasties. J. Arthroplast. 2018, 33, 840–843. [Google Scholar] [CrossRef]
- Koutserimpas, C.; Naoum, S.; Alpantaki, K.; Raptis, K.; Dretakis, K.; Vrioni, G.; Samonis, G. Fungal Prosthetic Joint Infection in Revised Knee Arthroplasty: An Orthopaedic Surgeon’s Nightmare. Diagnostics 2022, 12, 1606. [Google Scholar] [CrossRef] [PubMed]
- Delgado-González, A.; Morales-Viaji, J.J.; Criado-Albillos, G.; Martín-Rodríguez, A.D.P.; González-Santos, J.; López-Liria, R.; Collazo-Riobo, C.; Soto-Cámara, R.; González-Bernal, J.J. Explanatory Factors for Periprosthetic Infection in Total Knee Arthroplasty. J. Clin. Med. 2021, 10, 2315. [Google Scholar] [CrossRef] [PubMed]
- Koutserimpas, C.; Chamakioti, I.; Zervakis, S.; Raptis, K.; Alpantaki, K.; Kofteridis, D.P.; Vrioni, G.; Samonis, G. Non-Candida Fungal Prosthetic Joint Infections. Diagnostics 2021, 11, 1410. [Google Scholar] [CrossRef]
- Koutserimpas, C.; Chamakioti, I.; Raptis, K.; Alpantaki, K.; Vrioni, G.; Samonis, G. Osseous Infections Caused by Aspergillus Species. Diagnostics 2022, 12, 201. [Google Scholar] [CrossRef] [PubMed]
- Blanco, J.F.; Díaz, A.; Melchor, F.R.; da Casa, C.; Pescador, D. Risk factors for periprosthetic joint infection after total knee arthroplasty. Arch. Orthop. Trauma Surg. 2020, 140, 239–245. [Google Scholar] [CrossRef]
- Pappas, P.G.; Kauffman, C.A.; Andes, D.R.; Clancy, C.J.; Marr, K.A.; Ostrosky-Zeichner, L.; Reboli, A.C.; Schuster, M.G.; Vazquez, J.A.; Walsh, T.J.; et al. Executive Summary: Clinical Practice Guideline for the Management of Candidiasis: 2016 Update by the Infectious Diseases Society of America. Clin. Infect. Dis. 2016, 62, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Postler, A.; Lützner, C.; Beyer, F.; Tille, E.; Lützner, J. Analysis of Total Knee Arthroplasty revision causes. BMC Musculoskelet. Disord. 2018, 19, 55. [Google Scholar] [CrossRef] [PubMed]
- Jeschke, E.; Citak, M.; Günster, C.; Matthias Halder, A.; Heller, K.D.; Malzahn, J.; Niethard, F.U.; Schräder, P.; Zacher, J.; Gehrke, T. Are TKAs Performed in High-volume Hospitals Less Likely to Undergo Revision Than TKAs Performed in Low-volume Hospitals? Clin. Orthop. Relat. Res. 2017, 475, 2669–2674. [Google Scholar] [CrossRef]
- Batty, L.M.; Lanting, B. Contemporary Strategies to Prevent Infection in Hip and Knee Arthroplasty. Curr. Rev. Musculoskelet. Med. 2020, 13, 400–408. [Google Scholar] [CrossRef]
- Kritikou, G.; Avgerinos, K.I.; Koutserimpas, C.; Sourri, F.; Hatzigeorgiou, D.; Kottaridi, C.; Bountouris, I. Effectiveness of a preventive bundle of measures for reducing surgical site infections in patients undergoing elective orthopedic procedures in a Hellenic Air Force Hospital. G. Chir. -J. Ital. Surg. Assoc. 2019, 40, 120–126. [Google Scholar]
- Lee, Y.R.; Kim, H.J.; Lee, E.J.; Sohn, J.W.; Kim, M.J.; Yoon, Y.K. Prosthetic Joint Infections Caused by Candida Species: A Systematic Review and a Case Series. Mycopathologia 2019, 184, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Saconi, E.S.; de Carvalho, V.C.; de Oliveira, P.R.D.; Lima, A.L.L.M. Prosthetic joint infection due to Candida species: Case series and review of literature. Medicine 2020, 99, e19735. [Google Scholar] [CrossRef] [PubMed]
- Bini Viotti, J.; Corzo-Pedroza, M.; Gonzales Zamora, J.A. Prosthetic joint infection caused by Candida lusitaniae: Report of a unique case. Acta Clin. Belg. 2019, 74, 286–291. [Google Scholar] [CrossRef] [PubMed]
- Klatte, T.O.; Kendoff, D.; Kamath, A.F.; Jonen, V.; Rueger, J.M.; Frommelt, L.; Gebauer, M.; Gehrke, T. Single-stage revision for fungal peri-prosthetic joint infection: A single-centre experience. Bone Jt. J. 2014, 96-B, 492–496. [Google Scholar] [CrossRef]
- Hwang, B.H.; Yoon, J.Y.; Nam, C.H.; Jung, K.A.; Lee, S.C.; Han, C.D.; Moon, S.H. Fungal peri-prosthetic joint infection after primary total knee replacement. J. Bone Jt. Surg. Br. 2012, 94, 656–659. [Google Scholar] [CrossRef]
- Sidhu, M.S.; Cooper, G.; Jenkins, N.; Jeys, L.; Parry, M.; Stevenson, J.D. Prosthetic fungal infections: Poor prognosis with bacterial co-infection. Bone Jt. J. 2019, 101-B, 582–588. [Google Scholar] [CrossRef]
- Berbudi, A.; Rahmadika, N.; Tjahjadi, A.I.; Ruslami, R. Type 2 Diabetes and its Impact on the Immune System. Curr. Diabetes Rev. 2020, 16, 442–449. [Google Scholar]
- Lao, M.; Li, C.; Li, J.; Chen, D.; Ding, M.; Gong, Y. Opportunistic invasive fungal disease in patients with type 2 diabetes mellitus from Southern China: Clinical features and associated factors. J. Diabetes Investig. 2020, 11, 731–744. [Google Scholar] [CrossRef]
- Anagnos, K.; Becker, S.L.; Sahan, I. Antifungal-Loaded Acrylic Bone Cement in the Treatment of Periprosthetic Hip and Knee Joint Infections: A Review. Antibiotics 2022, 11, 879. [Google Scholar] [CrossRef]
- Mendoza-Reyes, D.F.; Gómez-Gaviria, M.; Mora-Montes, H.M. Candida lusitaniae: Biology, Pathogenicity, Virulence Factors, Diagnosis, and Treatment. Infect. Drug Resist. 2022, 15, 5121–5135. [Google Scholar] [CrossRef]
- Osmon, D.R.; Berbari, E.F.; Berendt, A.R.; Lew, D.; Zimmerli, W.; Steckelberg, J.M.; Rao, N.; Hanssen, A.; Wilson, W.R. Infectious Diseases Society of America. Diagnosis and management of prosthetic joint infection: Clinical practice guidelines by the Infectious Diseases Society of America. Clin. Infect. Dis. 2013, 56, e1–e25. [Google Scholar] [CrossRef] [PubMed]
- Mamali, V.; Koutserimpas, C.; Manoloudaki, K.; Zarkotou, O.; Samonis, G.; Vrioni, G. Necrotizing Skin and Soft Tissue Infection Due to Syncephalastrum Species and Fusarium solani Species Complex Following Open Tibia Fracture. Diagnostics 2022, 12, 1163. [Google Scholar] [CrossRef] [PubMed]
- Samonis, G.; Koutserimpas, C.; Vrioni, G.; Kampos Martinez, E.; Kouloumentas, P.; Alpantaki, K.; Saroglou, G. Fungal Septic Knee Arthritis Caused by Aspergillus fumigatus following Anterior Cruciate Ligament Reconstruction. Diagnostics 2021, 11, 1975. [Google Scholar] [CrossRef] [PubMed]
- Oviaño, M.; Rodríguez-Sánchez, B. MALDI-TOF mass spectrometry in the 21st century clinical microbiology laboratory. Enferm. Infecc. Y Microbiol. Clin. (Engl. Ed.) 2021, 39, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Gehrke, T.; Alijanipour, P.; Parvizi, J. The management of an infected total knee arthroplasty. Bone Jt. J. 2015, 97-B, 20–29. [Google Scholar] [CrossRef]
- Zajonz, D.; Zimmerlich, B.; Möbius, R.; Edel, M.; Przybyl, J.; Höch, A.; Fakler, J.K.M.; Roth, A.; Ghanem, M. Knee arthrodesis as last resort for persistent knee joint infections: Comparison of extramedullary and intramedullary treatment. Orthopade 2021, 50, 207–213. [Google Scholar] [CrossRef]
- Trouillez, T.; Faure, P.A.; Martinot, P.; Migaud, H.; Senneville, E.; Pasquier, G.; Dartus, J.; Putman, S. Above-the-knee amputation versus knee arthrodesis for revision of infected total knee arthroplasty: Recurrent infection rates and functional outcomes of 43 patients at a mean follow-up of 6.7 years. Orthop. Traumatol. Surg. Res. 2021, 107, 102914. [Google Scholar] [CrossRef]
- Mahmoud, S.S.; Sukeik, M.; Alazzawi, S.; Shaath, M.; Sabri, O. Salvage Procedures for Management of Prosthetic Joint Infection After Hip and Knee Replacements. Open Orthop. J. 2016, 10, 600–614. [Google Scholar] [CrossRef]
- Koutserimpas, C.; Chamakioti, I.; Naoum, S.; Raptis, K.; Alpantaki, K.; Samonis, G. Native Joint Infections by Aspergillus Species. Diagnostics 2021, 11, 2335. [Google Scholar] [CrossRef]
- Pasternak, B.; Wintzell, V.; Furu, K.; Engeland, A.; Neovius, M.; Stephansson, O. Oral Fluconazole in Pregnancy and Risk of Stillbirth and Neonatal Death. JAMA 2018, 319, 2333–2335. [Google Scholar] [CrossRef]
- Hamill, R.J. Amphotericin B formulations: A comparative review of efficacy and toxicity. Drugs 2013, 73, 919–934. [Google Scholar] [CrossRef] [PubMed]
- Nett, J.E.; Andes, D.R. Antifungal Agents: Spectrum of Activity, Pharmacology, and Clinical Indications. Infect. Dis. Clin. N. Am. 2016, 30, 51–83. [Google Scholar] [CrossRef] [PubMed]
| Study | Gender/ Age | Joint | Bacterial Co-Infection | CRP (mg/L) | ESR (mm/h) | Comorbidities | Number of Revisions | Time from Implantation to Symptoms’ Onset (Months) | Time from Symptoms Onset to Diagnosis (Months) |
|---|---|---|---|---|---|---|---|---|---|
| Present study, 2022 | F/74 | Knee | K. oxytoca S. capitis C. lusitaniae | 29 | - | DM, HTN | 3 | 1 | 1 |
| Saconi et al., 2020 [21] | M/63 | Hip | No | - | - | Kidney transplantation | - | - | - |
| Viotti et al., 2019 [22] | F/79 | Knee | - | 8.4 | 118 | RA | - | 120 | - |
| Klatte et al., 2014 [23] | M/74 | Knee | S. aureus S. mitis | >22 | - | - | 2 | 17 | 6 |
| Hwang et al., 2012 [24] | F/66 | Knee | NR | 4.3 | 29 | - | - | - | 48 |
| Study | ST | Time between Stages in TSRA (Months) | AFT | AFT (Duration) | Follow-Up (Months) | Outcome |
|---|---|---|---|---|---|---|
| Present study, 2022 | Arthrodesis | - | Fluconazole | 4 | 4 | Success |
| Saconi et al., 2020 [1] | OSRA | - | Micafungin, fluconazole | 24 | Lost | - |
| Viotti et al., 2019 [13] | TSRA | 11 | Micafungin | 6 | 18 | Success |
| Klatte et al., 2014 [19] | NS | - | Voriconazole | >2 | 30 | Success |
| Hwang et al., 2012 [20] | TSRA | 3 | Amphotericin B, then oral fluconazole | - | 43 | Success |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giovanoulis, V.; Vasiliadis, A.V.; Koutserimpas, C.; Samonis, G.; Batailler, C.; Ferry, T.; Lustig, S. Prosthetic Knee Joint Infection Due to Candida lusitaniae: A Diagnostic and Therapeutic Odyssey: A Case Study. Diagnostics 2022, 12, 2640. https://doi.org/10.3390/diagnostics12112640
Giovanoulis V, Vasiliadis AV, Koutserimpas C, Samonis G, Batailler C, Ferry T, Lustig S. Prosthetic Knee Joint Infection Due to Candida lusitaniae: A Diagnostic and Therapeutic Odyssey: A Case Study. Diagnostics. 2022; 12(11):2640. https://doi.org/10.3390/diagnostics12112640
Chicago/Turabian StyleGiovanoulis, Vasileios, Angelo V. Vasiliadis, Christos Koutserimpas, George Samonis, Cécile Batailler, Tristan Ferry, and Sébastien Lustig. 2022. "Prosthetic Knee Joint Infection Due to Candida lusitaniae: A Diagnostic and Therapeutic Odyssey: A Case Study" Diagnostics 12, no. 11: 2640. https://doi.org/10.3390/diagnostics12112640
APA StyleGiovanoulis, V., Vasiliadis, A. V., Koutserimpas, C., Samonis, G., Batailler, C., Ferry, T., & Lustig, S. (2022). Prosthetic Knee Joint Infection Due to Candida lusitaniae: A Diagnostic and Therapeutic Odyssey: A Case Study. Diagnostics, 12(11), 2640. https://doi.org/10.3390/diagnostics12112640









